First Detection of mcr-9 in a Multidrug-Resistant Escherichia coli of Animal Origin in Italy Is Not Related to Colistin Usage on a Pig Farm
Abstract
:1. Introduction
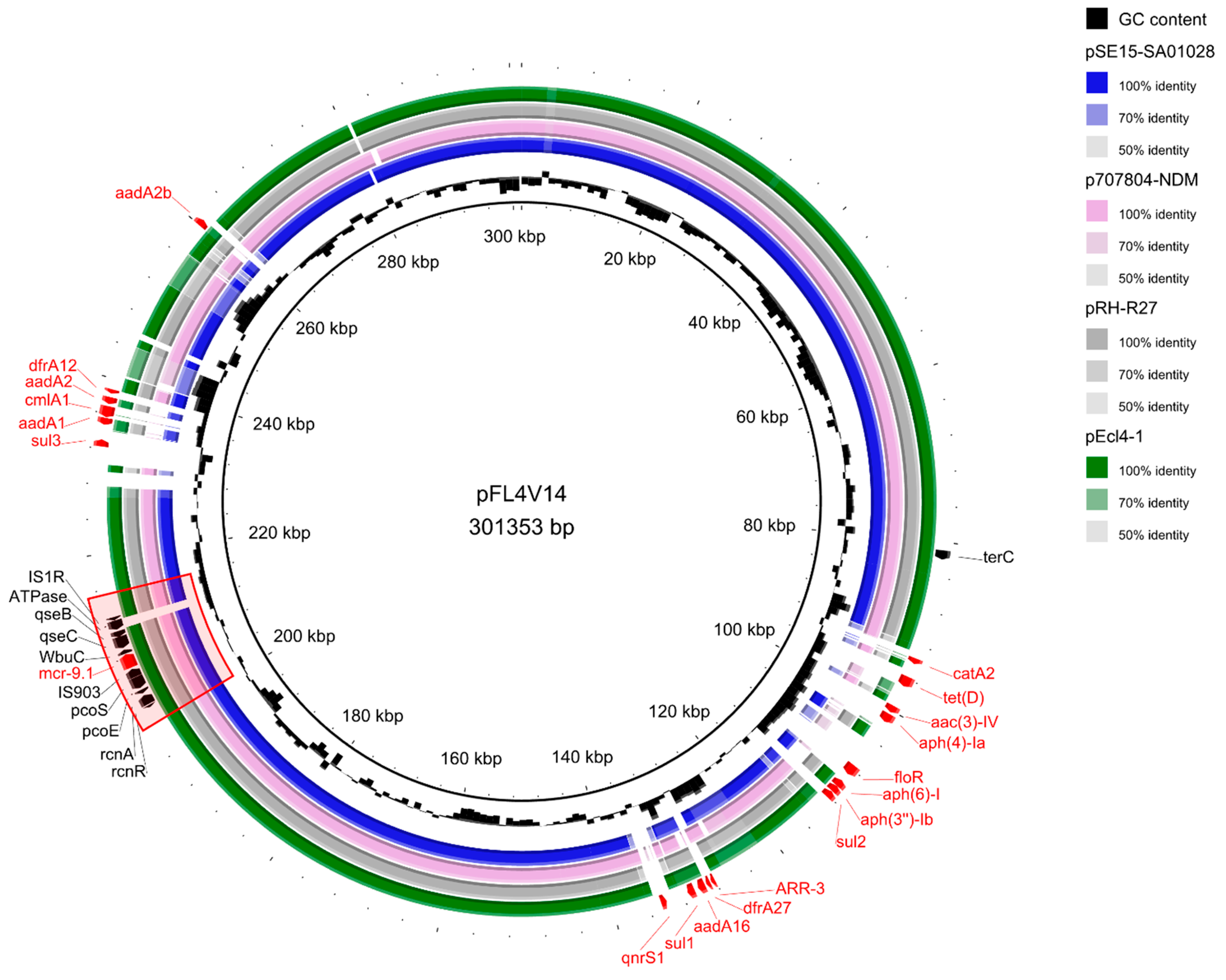
2. Results
3. Discussion
4. Materials and Methods
4.1. Farm Characteristics, Sampling and Isolation of the Bacterial Strain
4.2. PCR Detection
4.3. Whole-Genome Sequencing and Bioinformatic Analyses
4.4. Antimicrobial Susceptibility Testing
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Roca, I.; Akova, M.; Baquero, F.; Carlet, J.; Cavaleri, M.; Coenen, S.; Cohen, J.; Findlay, D.; Gyssens, I.; Heure, O.E.; et al. The Global Threat of Antimicrobial Resistance: Science for Intervention. New Microbes New Infect. 2015, 6, 22–29. [Google Scholar] [CrossRef] [Green Version]
- Terreni, M.; Taccani, M.; Pregnolato, M. New Antibiotics for Multidrug-Resistant Bacterial Strains: Latest Research Developments and Future Perspectives. Molecules 2021, 26, 2671. [Google Scholar] [CrossRef]
- Van Duin, D.; Paterson, D.L. Multidrug-Resistant Bacteria in the Community: Trends and Lessons Learned. Infect. Dis. Clin. 2016, 30, 377–390. [Google Scholar] [CrossRef] [Green Version]
- World Health Organization. Critically Important Antimicrobials for Human Medicine, 6th ed.; World Health Organization: Geneva, Switzerland, 2019; ISBN 978-92-4-151552-8. [Google Scholar]
- Ara, B.; Urmi, U.L.; Haque, T.A.; Nahar, S.; Rumnaz, A.; Ali, T.; Alam, M.S.; Mosaddek, A.S.M.; Rahman, n.A.a.; Haque, M.; et al. Detection of Mobile Colistin-Resistance Gene Variants (Mcr-1 and Mcr-2) in Urinary Tract Pathogens in Bangladesh: The Last Resort of Infectious Disease Management Colistin Efficacy Is under Threat. Expert Rev. Clin. Pharmacol. 2021, 14, 513–522. [Google Scholar] [CrossRef]
- Mendelson, M.; Brink, A.; Gouws, J.; Mbelle, N.; Naidoo, V.; Pople, T.; Schellack, N.; van Vuuren, M.; Rees, H.; Banoo, S.; et al. The One Health Stewardship of Colistin as an Antibiotic of Last Resort for Human Health in South Africa. Lancet Infect. Dis. 2018, 18, e288–e294. [Google Scholar] [CrossRef]
- Lima, T.; Domingues, S.; Da Silva, G.J. Plasmid-Mediated Colistin Resistance in Salmonella Enterica: A Review. Microorganisms 2019, 7, 55. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.; Abbas, M.; Rehman, M.U.; Wang, M.; Jia, R.; Chen, S.; Liu, M.; Zhu, D.; Zhao, X.; Gao, Q.; et al. Updates on the Global Dissemination of Colistin-Resistant Escherichia Coli: An Emerging Threat to Public Health. Sci. Total Environ. 2021, 799, 149280. [Google Scholar] [CrossRef]
- Binsker, U.; Käsbohrer, A.; Hammerl, J.A. Global Colistin Use: A Review of the Emergence of Resistant Enterobacterales and the Impact on Their Genetic Basis. FEMS Microbiol. Rev. 2022, 46, fuab049. [Google Scholar] [CrossRef]
- Jeannot, K.; Bolard, A.; Plésiat, P. Resistance to Polymyxins in Gram-Negative Organisms. Int. J. Antimicrob. Agents 2017, 49, 526–535. [Google Scholar] [CrossRef]
- Poirel, L.; Jayol, A.; Nordmann, P. Polymyxins: Antibacterial Activity, Susceptibility Testing, and Resistance Mechanisms Encoded by Plasmids or Chromosomes. Clin. Microbiol. Rev. 2017, 30, 557–596. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.-Y.; Wang, Y.; Walsh, T.R.; Yi, L.-X.; Zhang, R.; Spencer, J.; Doi, Y.; Tian, G.; Dong, B.; Huang, X.; et al. Emergence of Plasmid-Mediated Colistin Resistance Mechanism MCR-1 in Animals and Human Beings in China: A Microbiological and Molecular Biological Study. Lancet Infect. Dis. 2016, 16, 161–168. [Google Scholar] [CrossRef]
- Hussein, N.H.; AL-Kadmy, I.M.S.; Taha, B.M.; Hussein, J.D. Mobilized Colistin Resistance (Mcr) Genes from 1 to 10: A Comprehensive Review. Mol. Biol. Rep. 2021, 48, 2897–2907. [Google Scholar] [CrossRef]
- Tyson, G.H.; Li, C.; Hsu, C.-H.; Ayers, S.; Borenstein, S.; Mukherjee, S.; Tran, T.-T.; McDermott, P.F.; Zhao, S. The Mcr-9 Gene of Salmonella and Escherichia Coli Is Not Associated with Colistin Resistance in the United States. Antimicrob. Agents Chemother. 2020, 64, e00573-20. [Google Scholar] [CrossRef]
- Carattoli, A.; Villa, L.; Feudi, C.; Curcio, L.; Orsini, S.; Luppi, A.; Pezzotti, G.; Magistrali, C.F. Novel Plasmid-Mediated Colistin Resistance Mcr-4 Gene in Salmonella and Escherichia Coli, Italy 2013, Spain and Belgium, 2015 to 2016. Eurosurveillance 2017, 22, 30589. [Google Scholar] [CrossRef] [Green Version]
- Formenti, N.; Guarneri, F.; Bertasio, C.; Parisio, G.; Romeo, C.; Scali, F.; Birbes, L.; Boniotti, M.B.; Diegoli, G.; Candela, L.; et al. Wastewater-Based Surveillance in Italy Leading to the First Detection of Mcr-10-Positive Klebsiella Quasipneumoniae. Antimicrob. Resist. Infect. Control 2022, 11, 155. [Google Scholar] [CrossRef]
- Liao, W.; Cui, Y.; Quan, J.; Zhao, D.; Han, X.; Shi, Q.; Wang, Q.; Jiang, Y.; Du, X.; Li, X.; et al. High Prevalence of Colistin Resistance and Mcr-9/10 Genes in Enterobacter Spp. in a Tertiary Hospital over a Decade. Int. J. Antimicrob. Agents 2022, 59, 106573. [Google Scholar] [CrossRef]
- Wang, Y.; Liu, F.; Hu, Y.; Zhang, G.; Zhu, B.; Gao, G.F. Detection of Mobile Colistin Resistance Gene Mcr-9 in Carbapenem-Resistant Klebsiella Pneumoniae Strains of Human Origin in Europe. J. Infect. 2020, 80, 578–606. [Google Scholar] [CrossRef]
- Wang, Y.; Xu, C.; Zhang, R.; Chen, Y.; Shen, Y.; Hu, F.; Liu, D.; Lu, J.; Guo, Y.; Xia, X.; et al. Changes in Colistin Resistance and Mcr-1 Abundance in Escherichia Coli of Animal and Human Origins Following the Ban of Colistin-Positive Additives in China: An Epidemiological Comparative Study. Lancet Infect. Dis. 2020, 20, 1161–1171. [Google Scholar] [CrossRef]
- European Commission. Commission Implementing Decision of 14.7.2016 Concerning in the Framework of Article 35 of Directive 2001/82/EC of the European Parliament and of the Council, the Marketing Authorisations for All Veterinary Medicinal Products Containing “Colistin” in Combination with Other Antimicrobial Substances to Be Administered Orally; European Commission: Brussels, Belgium, 2016. [Google Scholar]
- European Medicines Agency Sales of Veterinary Antimicrobial Agents in 31 European Countries in 2021—Trends from 2010 to 2021 (Twelfth ESVAC Report); ESVAC Report: Luxembourg, 2022.
- Gagliotti, C.; Bolzoni, L.; Carretto, E.; Sarti, M.; Ricchizzi, E.; Ambretti, S.; Barozzi, A.; Bracchi, C.; Confalonieri, M.; Menozzi, I.; et al. Reduction Trend of Mcr-1 Circulation in Emilia-Romagna Region, Italy. Eur. J. Clin. Microbiol. Infect Dis. 2021, 40, 2585–2592. [Google Scholar] [CrossRef]
- Kohler, P.; Tijet, N.; Kim, H.C.; Johnstone, J.; Edge, T.; Patel, S.N.; Seah, C.; Willey, B.; Coleman, B.; Green, K.; et al. Dissemination of Verona Integron-Encoded Metallo-β-Lactamase among Clinical and Environmental Enterobacteriaceae Isolates in Ontario, Canada. Sci. Rep. 2020, 10, 18580. [Google Scholar] [CrossRef]
- Diaconu, E.L.; Alba, P.; Feltrin, F.; Di Matteo, P.; Iurescia, M.; Chelli, E.; Donati, V.; Marani, I.; Giacomi, A.; Franco, A.; et al. Emergence of IncHI2 Plasmids With Mobilized Colistin Resistance (Mcr)-9 Gene in ESBL-Producing, Multidrug-Resistant Salmonella Typhimurium and Its Monophasic Variant ST34 From Food-Producing Animals in Italy. Front. Microbiol. 2021, 12, 705230. [Google Scholar] [CrossRef]
- Marchetti, V.M.; Bitar, I.; Sarti, M.; Fogato, E.; Scaltriti, E.; Bracchi, C.; Hrabak, J.; Pongolini, S.; Migliavacca, R. Genomic Characterization of VIM and MCR Co-Producers: The First Two Clinical Cases, in Italy. Diagnostics 2021, 11, 79. [Google Scholar] [CrossRef]
- Gaiarsa, S.; Merla, C.; Corbella, M.; Mariani, B.; Zatelli, M.; Sciabica, I.; Castelli, M.; Piazza, A.; Zecca, M.; Sassera, D.; et al. Isolation of a Colistin-Susceptible MDR Pantoea Calida Harboring the Mcr-9 Gene Suggests the Silent Spread of the Resistance Factor. Microb. Drug Resist. 2022, 28, 408–412. [Google Scholar] [CrossRef]
- Simoni, S.; Mingoia, M.; Brenciani, A.; Carelli, M.; Lleò, M.M.; Malerba, G.; Vignaroli, C. First IncHI2 Plasmid Carrying Mcr-9.1, BlaVIM-1, and Double Copies of BlaKPC-3 in a Multidrug-Resistant Escherichia Coli Human Isolate. mSphere 2021, 6, e00302-21. [Google Scholar] [CrossRef]
- Zhang, Y.; Sun, Y.-H.; Wang, J.-Y.; Chang, M.-X.; Zhao, Q.-Y.; Jiang, H.-X. A Novel Structure Harboring BlaCTX-M-27 on IncF Plasmids in Escherichia Coli Isolated from Swine in China. Antibiotics 2021, 10, 387. [Google Scholar] [CrossRef]
- Yuan, J.; Wang, X.; Shi, D.; Ge, Q.; Song, X.; Hu, W.; Wei, D.; Ge, C.; Li, X.; Hu, C. Extensive Antimicrobial Resistance and Plasmid-Carrying Resistance Genes in Mcr-1-Positive E. Coli Sampled in Swine, in Guangxi, South China. BMC Vet. Res. 2021, 17, 86. [Google Scholar] [CrossRef]
- He, D.-D.; Cui, M.-M.; Zhang, T.-L.; Hu, G.-Z.; Liu, J.-H.; Pan, Y.-S. Characterization of BlaCMY-2-Carrying IncC and RmtB-Carrying IncI1/ST136 Plasmids in an Avian Escherichia Coli ST224 Strain. Plasmid 2021, 114, 102555. [Google Scholar] [CrossRef]
- Liu, Z.; Li, W.; Wang, J.; Pan, J.; Sun, S.; Yu, Y.; Zhao, B.; Ma, Y.; Zhang, T.; Qi, J.; et al. Identification and Characterization of the First Escherichia Coli Strain Carrying NDM-1 Gene in China. PLoS ONE 2013, 8, e66666. [Google Scholar] [CrossRef] [Green Version]
- Cao, X.; Zhang, Z.; Shen, H.; Ning, M.; Chen, J.; Wei, H.; Zhang, K. Genotypic Characteristics of Multidrug-Resistant Escherichia Coli Isolates Associated with Urinary Tract Infections. APMIS 2014, 122, 1088–1095. [Google Scholar] [CrossRef]
- Silva, M.M.; Sellera, F.P.; Fernandes, M.R.; Moura, Q.; Garino, F.; Azevedo, S.S.; Lincopan, N. Genomic Features of a Highly Virulent, Ceftiofur-Resistant, CTX-M-8-Producing Escherichia Coli ST224 Causing Fatal Infection in a Domestic Cat. J. Glob. Antimicrob. Resist. 2018, 15, 252–253. [Google Scholar] [CrossRef]
- Alonso, C.A.; González-Barrio, D.; Tenorio, C.; Ruiz-Fons, F.; Torres, C. Antimicrobial Resistance in Faecal Escherichia Coli Isolates from Farmed Red Deer and Wild Small Mammals. Detection of a Multiresistant E. Coli Producing Extended-Spectrum Beta-Lactamase. Comp. Immunol. Microbiol. Infect. Dis. 2016, 45, 34–39. [Google Scholar] [CrossRef]
- Delgado-Blas, J.F.; Ovejero, C.M.; David, S.; Montero, N.; Calero-Caceres, W.; Garcillan-Barcia, M.P.; de la Cruz, F.; Muniesa, M.; Aanensen, D.M.; Gonzalez-Zorn, B. Population Genomics and Antimicrobial Resistance Dynamics of Escherichia Coli in Wastewater and River Environments. Commun. Biol. 2021, 4, 457. [Google Scholar] [CrossRef]
- Ling, Z.; Yin, W.; Shen, Z.; Wang, Y.; Shen, J.; Walsh, T.R. Epidemiology of Mobile Colistin Resistance Genes Mcr-1 to Mcr-9. J. Antimicrob. Chemother. 2020, 75, 3087–3095. [Google Scholar] [CrossRef]
- Khedher, M.B.; Baron, S.A.; Riziki, T.; Ruimy, R.; Raoult, D.; Diene, S.M.; Rolain, J.-M. Massive Analysis of 64,628 Bacterial Genomes to Decipher Water Reservoir and Origin of Mobile Colistin Resistance Genes: Is There Another Role for These Enzymes? Sci. Rep. 2020, 10, 5970. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.; Dai, X.; Zeng, J.; Gao, Y.; Zhang, Z.; Zhang, L. Characterization of the Global Distribution and Diversified Plasmid Reservoirs of the Colistin Resistance Gene Mcr-9. Sci. Rep. 2020, 10, 8113. [Google Scholar] [CrossRef]
- Carroll, L.M.; Gaballa, A.; Guldimann, C.; Sullivan, G.; Henderson, L.O.; Wiedmann, M. Identification of Novel Mobilized Colistin Resistance Gene Mcr-9 in a Multidrug-Resistant, Colistin-Susceptible Salmonella Enterica Serotype Typhimurium Isolate. mBio 2019, 10, e00853-19. [Google Scholar] [CrossRef] [Green Version]
- Kieffer, N.; Royer, G.; Decousser, J.-W.; Bourrel, A.-S.; Palmieri, M.; Ortiz De La Rosa, J.-M.; Jacquier, H.; Denamur, E.; Nordmann, P.; Poirel, L. Mcr-9, an Inducible Gene Encoding an Acquired Phosphoethanolamine Transferase in Escherichia Coli, and Its Origin. Antimicrob. Agents Chemother. 2019, 63, e00965-19. [Google Scholar] [CrossRef] [Green Version]
- Breland, E.J.; Zhang, E.W.; Bermudez, T.; Martinez, C.R.; Hadjifrangiskou, M. The Histidine Residue of QseC Is Required for Canonical Signaling between QseB and PmrB in Uropathogenic Escherichia Coli. J. Bacteriol. 2017, 199, e00060-17. [Google Scholar] [CrossRef] [Green Version]
- Phan, M.-D.; Peters, K.M.; Sarkar, S.; Forde, B.M.; Lo, A.W.; Stanton-Cook, M.; Roberts, L.W.; Upton, M.; Beatson, S.A.; Schembri, M.A. Third-Generation Cephalosporin Resistance Conferred by a Chromosomally Encoded BlaCMY-23 Gene in the Escherichia Coli ST131 Reference Strain EC958. J. Antimicrob. Chemother. 2015, 70, 1969–1972. [Google Scholar] [CrossRef] [Green Version]
- Manageiro, V.; Ferreira, E.; Pinto, M.; Fonseca, F.; Ferreira, M.; Bonnet, R.; Caniça, M. Two Novel CMY-2-Type β-Lactamases Encountered in Clinical Escherichia Coli Isolates. Ann. Clin. Microbiol. Antimicrob. 2015, 14, 12. [Google Scholar] [CrossRef] [Green Version]
- Formenti, N.; Grassi, A.; Parisio, G.; Romeo, C.; Guarneri, F.; Birbes, L.; Pitozzi, A.; Scali, F.; Maisano, A.M.; Boniotti, M.B.; et al. Extended-Spectrum-β-Lactamase- and AmpC-Producing Escherichia Coli in Domestic Dogs: Spread, Characterisation and Associated Risk Factors. Antibiotics 2021, 10, 1251. [Google Scholar] [CrossRef] [PubMed]
- Macesic, N.; Blakeway, L.V.; Stewart, J.D.; Hawkey, J.; Wyres, K.L.; Judd, L.M.; Wick, R.R.; Jenney, A.W.; Holt, K.E.; Peleg, A.Y. Silent Spread of Mobile Colistin Resistance Gene Mcr-9.1 on IncHI2 ‘Superplasmids’ in Clinical Carbapenem-Resistant Enterobacterales. Clin. Microbiol. Infect. 2021, 27, 1856.e7–1856.e13. [Google Scholar] [CrossRef]
- Deekshit, V.K.; Srikumar, S. ‘To Be, or Not to Be’—The Dilemma of ‘Silent’ Antimicrobial Resistance Genes in Bacteria. J. Appl. Microbiol. 2022, 133, 2902–2914. [Google Scholar] [CrossRef]
- Clermont, O.; Christenson, J.K.; Denamur, E.; Gordon, D.M. The Clermont Escherichia Coli Phylo-Typing Method Revisited: Improvement of Specificity and Detection of New Phylo-Groups. Environ. Microbiol. Rep. 2013, 5, 58–65. [Google Scholar] [CrossRef]
- Fang, H.; Ataker, F.; Hedin, G.; Dornbusch, K. Molecular Epidemiology of Extended-Spectrum β-Lactamases among Escherichia Coli Isolates Collected in a Swedish Hospital and Its Associated Health Care Facilities from 2001 to 2006. J. Clin. Microbiol. 2008, 46, 707–712. [Google Scholar] [CrossRef] [Green Version]
- Rehman, M.A.; Hasted, T.-L.; Persaud-Lachhman, M.G.; Yin, X.; Carrillo, C.; Diarra, M.S. Genome Analysis and Multiplex PCR Method for the Molecular Detection of Coresistance to Cephalosporins and Fosfomycin in Salmonella Enterica Serovar Heidelberg. J. Food Prot. 2019, 82, 1938–1949. [Google Scholar] [CrossRef]
- Borowiak, M.; Baumann, B.; Fischer, J.; Thomas, K.; Deneke, C.; Hammerl, J.A.; Szabo, I.; Malorny, B. Development of a Novel Mcr-6 to Mcr-9 Multiplex PCR and Assessment of Mcr-1 to Mcr-9 Occurrence in Colistin-Resistant Salmonella Enterica Isolates From Environment, Feed, Animals and Food (2011–2018) in Germany. Front. Microbiol. 2020, 11, 80. [Google Scholar] [CrossRef] [Green Version]
- Rebelo, A.R.; Bortolaia, V.; Kjeldgaard, J.S.; Pedersen, S.K.; Leekitcharoenphon, P.; Hansen, I.M.; Guerra, B.; Malorny, B.; Borowiak, M.; Hammerl, J.A.; et al. Multiplex PCR for Detection of Plasmid-Mediated Colistin Resistance Determinants, Mcr-1, Mcr-2, Mcr-3, Mcr-4 and Mcr-5 for Surveillance Purposes. Eurosurveillance 2018, 23, 17. [Google Scholar] [CrossRef] [Green Version]
- Wang, C.; Feng, Y.; Liu, L.; Wei, L.; Kang, M.; Zong, Z. Identification of Novel Mobile Colistin Resistance Gene Mcr-10. Emerg. Microbes Infect. 2020, 9, 508–516. [Google Scholar] [CrossRef] [Green Version]
- Bolger, A.M.; Lohse, M.; Usadel, B. Trimmomatic: A Flexible Trimmer for Illumina Sequence Data. Bioinformatics 2014, 30, 2114–2120. [Google Scholar] [CrossRef] [Green Version]
- Wick, R.R.; Judd, L.M.; Gorrie, C.L.; Holt, K.E. Unicycler: Resolving Bacterial Genome Assemblies from Short and Long Sequencing Reads. PLoS Comput. Biol. 2017, 13, e1005595. [Google Scholar] [CrossRef] [Green Version]
- Li, X.; Xie, Y.; Liu, M.; Tai, C.; Sun, J.; Deng, Z.; Ou, H.-Y. OriTfinder: A Web-Based Tool for the Identification of Origin of Transfers in DNA Sequences of Bacterial Mobile Genetic Elements. Nucleic Acids Res. 2018, 46, W229–W234. [Google Scholar] [CrossRef] [Green Version]

| Antimicrobial Class | Resistance Genes | MIC | ||
|---|---|---|---|---|
| Antimicrobial | MIC Values (mg/L) | Cut-Off | ||
| Amphenicols | catA2, cmIa1, floR | Florfenicol | >64 | >16 a |
| Aminoglycosides | aadA1, aph(3‴)-Ib, aadA2b, aac(3)-IV, aph(4)-Ia, aadA2, aph(6)-Id, aadA16, rmtB | Aminosidine | >32 | NA |
| Gentamicin | >32 | >2 a | ||
| Kanamycin | >32 | >8 a | ||
| Beta-lactams (Carbapenems) | Ertapenem | 0.03 | >0.03 a | |
| Imipenem | ≤0.12 | >0.5 a | ||
| Meropenem | ≤0.03 | >0.06 a | ||
| Beta-lactams (Cephalosporines and Penicillins) | blaTEM-1B, blaTEM-214, blaTEM-206, blaTEM 141, blaCTX-M-55 | Cefazolin | >8 | >4 a |
| Cefepime | 4 | >0.125 a | ||
| Cefotaxime | 64 | >0.25 a | ||
| Cefotaxime/clavulanic acid | ≤0.06 | >0.25 a | ||
| Cefoxitin | 4 | >16 a | ||
| Ceftazidime | 8 | >1 a | ||
| Ceftazidime/clavulanic acid | 0.25 | >1 a | ||
| Amoxicillin/clavulanic acid | 8 | >8 b | ||
| Ampicillin | >32 | >8 a | ||
| Temocillin | 8 | >16 a | ||
| Quinolones and Fluoroquinolones | qnrS1 | Enrofloxacin | >32 | >0.125 a |
| Flumequine | >16 | >2 a | ||
| Polymixins | mcr-9 | Colistin | 0.25 | >2 a |
| Sulfonamides and Diaminopyrimidines | sul1, sul2, sul3 (sulfonamides); dfrA12, dfrA27 (diaminopyrimidines) | Sulfisoxazole | >512 | ≥ 512 c |
| Trimethoprim/Sulfamethoxazole | >16 | >0.5 a | ||
| Tetracyclines | tet(D) | Tetracycline | >16 | >8 a |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guarneri, F.; Bertasio, C.; Romeo, C.; Formenti, N.; Scali, F.; Parisio, G.; Canziani, S.; Boifava, C.; Guadagno, F.; Boniotti, M.B.; et al. First Detection of mcr-9 in a Multidrug-Resistant Escherichia coli of Animal Origin in Italy Is Not Related to Colistin Usage on a Pig Farm. Antibiotics 2023, 12, 689. https://doi.org/10.3390/antibiotics12040689
Guarneri F, Bertasio C, Romeo C, Formenti N, Scali F, Parisio G, Canziani S, Boifava C, Guadagno F, Boniotti MB, et al. First Detection of mcr-9 in a Multidrug-Resistant Escherichia coli of Animal Origin in Italy Is Not Related to Colistin Usage on a Pig Farm. Antibiotics. 2023; 12(4):689. https://doi.org/10.3390/antibiotics12040689
Chicago/Turabian StyleGuarneri, Flavia, Cristina Bertasio, Claudia Romeo, Nicoletta Formenti, Federico Scali, Giovanni Parisio, Sabrina Canziani, Chiara Boifava, Federica Guadagno, Maria Beatrice Boniotti, and et al. 2023. "First Detection of mcr-9 in a Multidrug-Resistant Escherichia coli of Animal Origin in Italy Is Not Related to Colistin Usage on a Pig Farm" Antibiotics 12, no. 4: 689. https://doi.org/10.3390/antibiotics12040689





