Diamine Fungal Inducers of Secondary Metabolism: 1,3-Diaminopropane and Spermidine Trigger Enzymes Involved in β-Alanine and Pantothenic Acid Biosynthesis, Precursors of Phosphopantetheine in the Activation of Multidomain Enzymes
Abstract
1. Introduction: The Search for Inducers of the Biosynthesis of Secondary Metabolites in Fungi
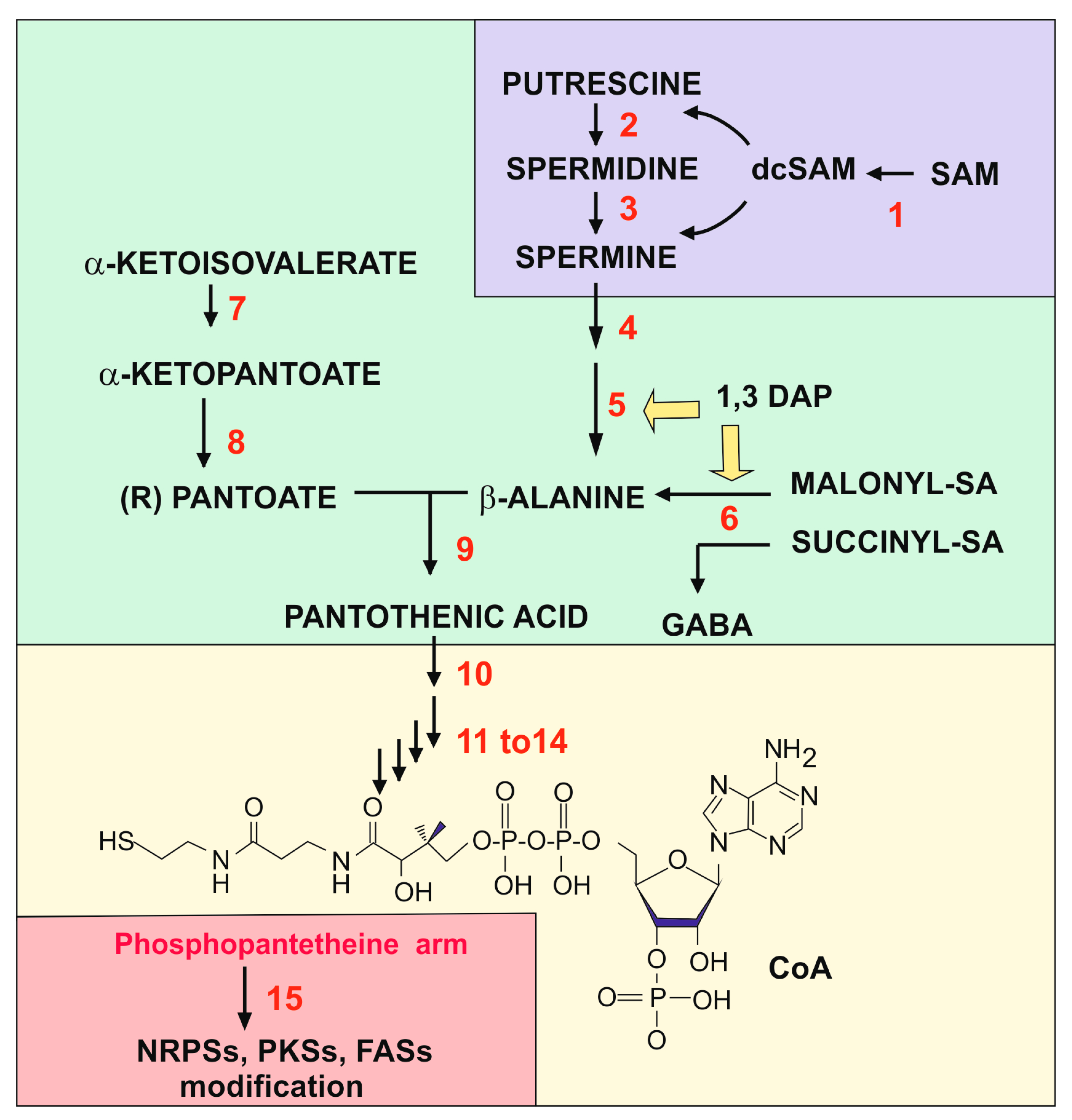
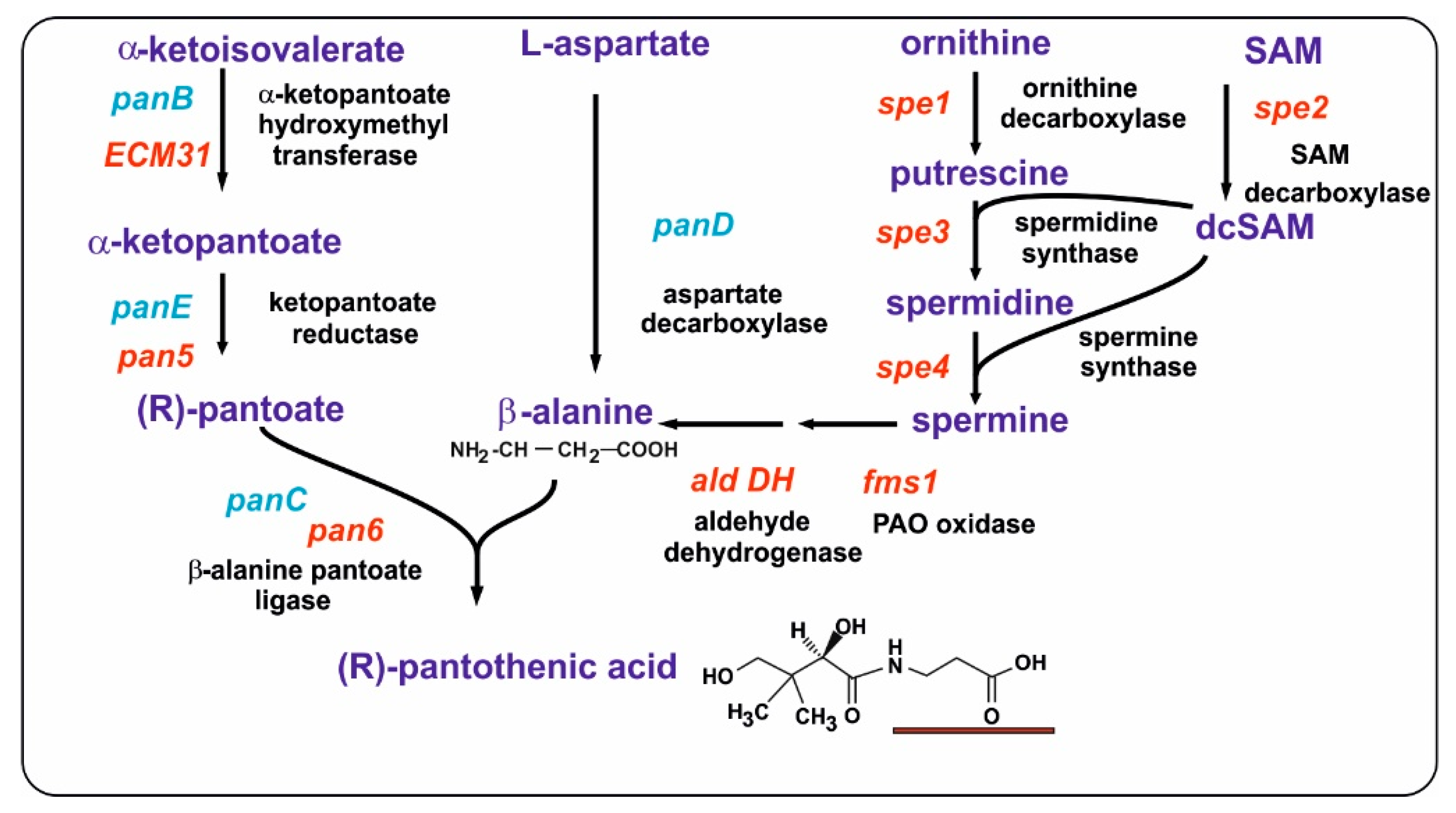
2. Polyamines in Fungi and the Origin of the Propylamine Unit for the Biosynthesis of β-Alanine
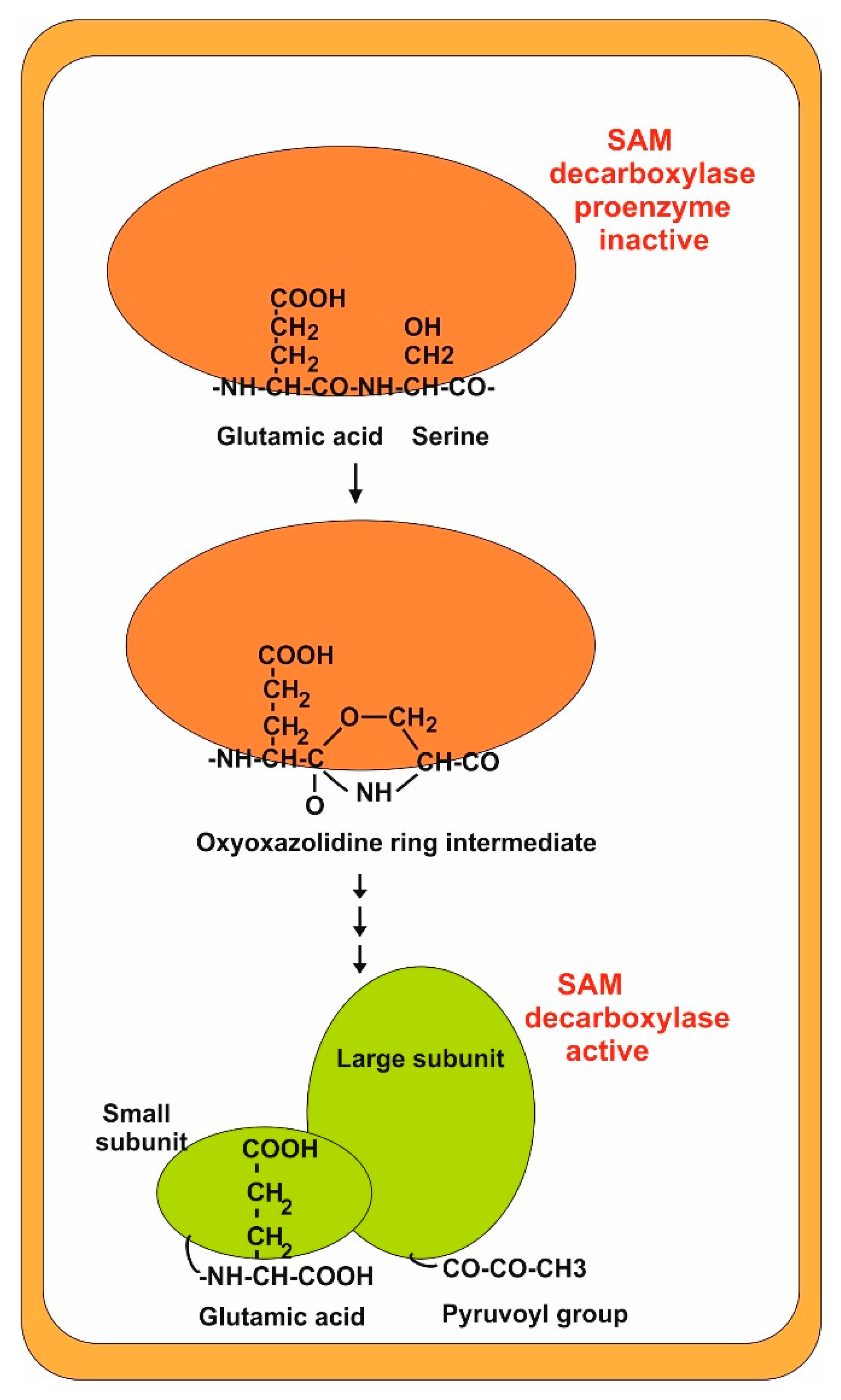
2.1. Aminopropyl Transferases: Formation of Spermidine and Spermine
2.2. Conversion of Spermidine in Spermine
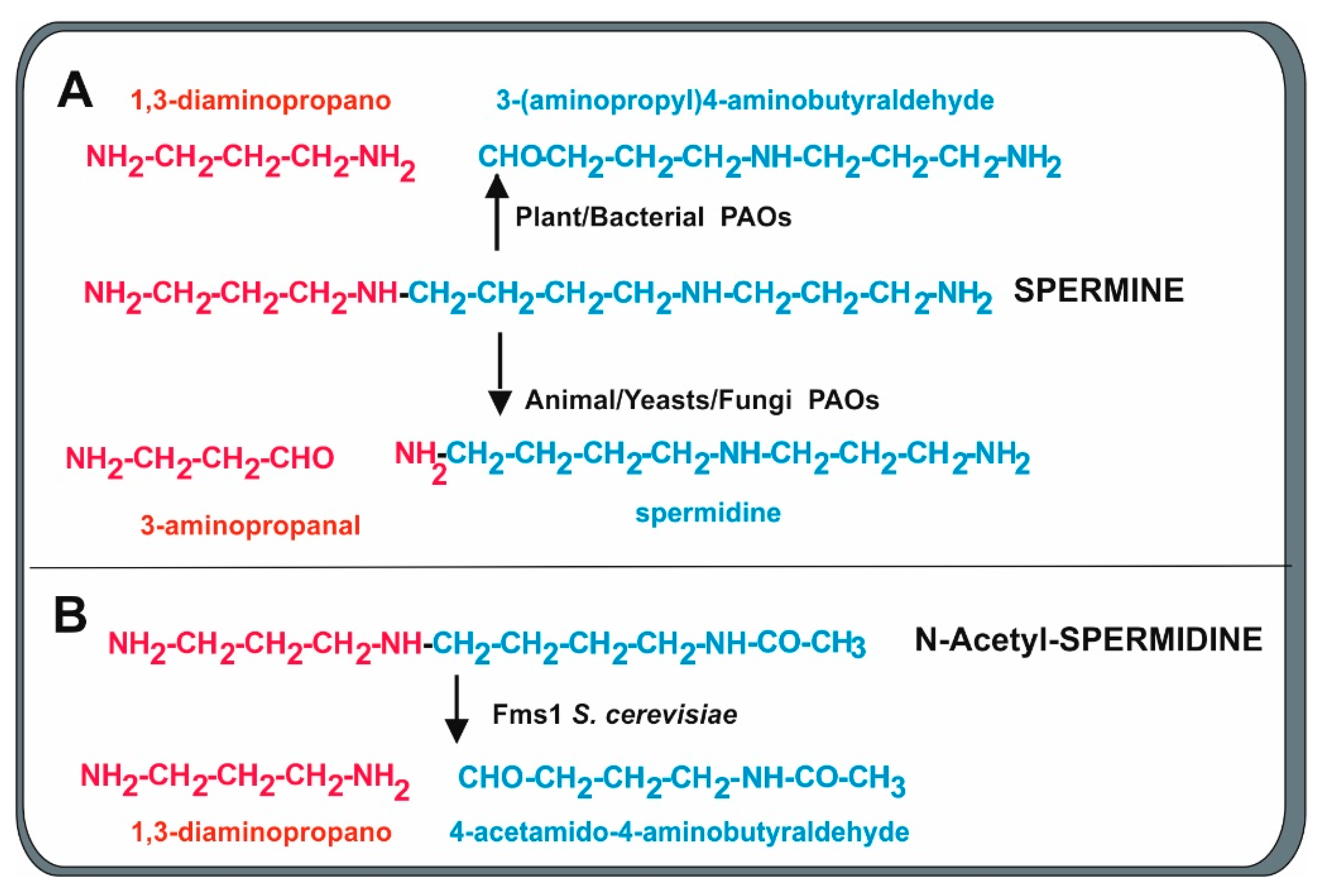
2.3. Polyamine Oxidases
Polyamine Oxidase in Penicillium chrysogenum
2.4. The 1,3-DAP Inducer Triggers Formation of Aldehyde Dehydrogenases That Convert 3-Aminopropanal to β-Alanine
2.5. Other Proteins Related to β-Alanine Biosynthesis in P. chrysogenum
2.5.1. Duplicated GABA Aminotransferases in Some Yeasts: Specificity for GABA or for β-Alanine as Substrates
2.5.2. GABA Aminotransferases in Filamentous Fungi
3. From β-Alanine to Phosphopantetheinyl Activation of Multimodular Enzymes
3.1. Pantothenic Acid in Yeast and Filamentous Fungi
3.1.1. The α-Ketopantoate Reductase: An Enigmatic Enzyme
3.1.2. The Pantoate and β-Alanine Condensing Enzyme
3.1.3. Intercellular Cross Feeding of Pantothenic Acid
3.2. Formation of Coenzyme A from Pantothenic Acid
3.2.1. The Pantothenate Kinase
3.2.2. Characterization of the Saccharomyces Cerevisiae PanK Gene
4. Activation of Antibiotic Biosynthetic Enzymes by Phosphopantetheinylation
How Many PPTases Are Involved in the Activation of Multidomain Synthetases?
5. Conclusions and Future Outlook
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| ATP | adenosine triphosphate |
| CoA | coenzyme A |
| 1,3-DAP | 1,3-diaminopropane |
| DPCK | dephospho-CoA kinase |
| FAD | flavin adenine dinucleotide |
| FAS | fatty acid synthase |
| GABA | γ-aminobutyric acid |
| malonyl-SA | malonyl semialdehyde |
| NADH | nicotinamide adenine dinucleotide reduced form |
| NRPS | non-ribosomal peptide synthetase |
| PAO | polyamine oxidase |
| PAS | pantothenic acid synthetase |
| PKS | polyketide synthetase |
| PLP | pyridoxal phosphate |
| PPTase | phosphopantetheine transferase |
| PPCS | phosphopanthotenoylcysteine synthetase |
| PPCDC | 4′-phosphopantothenoylcysteine decarboxylase |
| succinyl-SA | succinyl semialdehyde |
| SAM | S-adenosylmethionine |
| dcSAM | decarboxylated S-adenosylmethionine |
| SMO | spermidine oxidase |
References
- Velasco, J.; Gutiérrez, S.; Fernández, F.J.; Marcos, A.T.; Arenós, C.; Martín, J.F. Exogenous methionine increases levels of mRNAs transcribed from pcbAB, pcbC, and cefEF genes, encoding enzymes of the cephalosporin biosynthetic pathway, in Acremonium chrysogenum. J. Bacteriol. 1994, 176, 985–991. [Google Scholar] [CrossRef] [PubMed]
- Liu, G.; Casqueiro, J.; Bañuelos, O.; Cardoza, R.E.; Gutiérrez, S.; Martín, J.F. Targeted inactivation of the mecB gene encoding cystathionine-γ-lyase shows that the transsulfuration pathway is required for high level cephalosporin biosynthesis in Acremonium chrysogenum C10 but not for methionine-induction of the cephalosporin genes. J. Bacteriol. 2001, 183, 1765–1772. [Google Scholar] [CrossRef]
- Marcos, A.T.; Kosalková, K.; Cardoza, R.E.; Fierro, F.; Gutiérrez, S.; Martín, J.F. Characterization of the reverse transsulfuration gene mecB of Acremonium chrysogenum, which encodes a functional cystathionine-gamma-lyase. Mol. Gen. Genet. 2001, 264, 746–754. [Google Scholar] [CrossRef] [PubMed]
- Martín, J.F.; Demain, A.L. Unraveling the methionine-cephalosporin puzzle in Acremonium chrysogenum. Trends Biotechnol. 2002, 20, 12502–12507. [Google Scholar] [CrossRef] [PubMed]
- Long, L.-K.; Wang, Y.; Yang, J.; Xu, X.; Liu, G. A septation related gene AcsepH in Acremonium chrysogenum is involved in the cellular differentiation and cephalosporin production. Fungal Genet. Biol. 2013, 50, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Kluge, J.; Kück, U. AcAxl2 and AcMst1 regulate arthrospore development and stress resistance in the cephalosporin C producer Acremonium chrysogenum. Curr. Genet. 2018, 64, 713–727. [Google Scholar] [CrossRef]
- Xu, Y.; Liu, L.; Chen, Z.; Tian, X.; Chu, J.J. The arthrospore-related gene Acaxl2 is involved in cephalosporin C production in industrial Acremonium chrysogenum by the regulatory factors AcFKH1 and CPCR1. J. Biotechnol. 2022, 347, 26–39. [Google Scholar] [CrossRef]
- Cepeda-García, C.; Domínguez-Santos, R.; García-Rico, R.O.; García-Estrada, C.; Cajiao, A.; Fierro, F.; Martín, J.F. Direct involvement of the CreA transcription factor in penicillin biosynthesis and expression of the pcbAB gene in Penicillium chrysogenum. Appl. Microbiol. Biotechnol. 2014, 98, 7113–7124. [Google Scholar] [CrossRef]
- Barredo, J.L.; Alvarez, E.; Cantoral, J.M.; Díez, B.; Martín, J.F. Glucokinase-deficient mutant of Penicillium chrysogenum is derepressed in glucose catabolite regulation of both β-galactosidase and penicillin biosynthesis. Antimicrob. Agents Chemother. 1988, 32, 1061–1067. [Google Scholar] [CrossRef]
- Pérez, E.A.; Fernández, F.J.; Fierro, F.; Mejía, A.; Marcos, A.T.; Martín, J.F.; Barrios-González, J. Yeast HXK2 gene reverts glucose regulation mutation of penicillin biosynthesis in P. chrysogenum. Braz. J. Microbiol. 2014, 45, 873–883. [Google Scholar] [CrossRef]
- Domínguez-Santos, R.; Martín, J.F.; Kosalková, K.; Prieto, C.; Ullán, R.V.; García-Estrada, C. The regulatory factor PcRFX1 controls the expression of the three genes of β-lactam biosynthesis in Penicillium chrysogenum. Fungal Genet. Biol. 2012, 49, 866–881. [Google Scholar] [CrossRef] [PubMed]
- Domínguez-Santos, R.; García Estrada, C.; Kosalkova, K.; Prieto, C.; Santamarta, I.; Martín, J.F. PcFKH1, a novel regulatory factor from the forkhead family, controls the biosynthesis of penicillin in Penicillium chrysogenum. Biochimie 2015, 115, 162–176. [Google Scholar] [CrossRef]
- García-Estrada, C.; Domínguez-Santos, R.; Kosalková, K.; Martín, J.F. Transcription Factors controlling primary and secondary metabolism in filamentous fungi: The b-Lactam Paradigm. Fermentation 2018, 4, 47. [Google Scholar] [CrossRef]
- Huang, X.; Li, H. Cloning and bioinformatic analysis of lovastatin biosynthesis regulatory gene lovE. Chin. Med. J. 2009, 122, 1800–1805. [Google Scholar] [PubMed]
- Barrios-González, J.; Pérez-Sánchez, A.; Bibián, M.E. New knowledge about the biosynthesis of lovastatin and its production by fermentation of Aspergillus terreus. Appl. Microbiol. Biotechnol. 2020, 104, 8979–8998. [Google Scholar] [CrossRef]
- Zhgun, A.A.; Nuraeva, G.K.; Eldarov, M.A. The Role of LaeA and LovE Regulators in Lovastatin Biosynthesis with Exogenous Polyamines in Aspergillus terreus. Appl. Biochem. Microbiol. 2019, 55, 639–648. [Google Scholar] [CrossRef]
- Shimizu, K.; Hicks, J.K.; Huang, T.P.; Keller, N.P. Pka, Ras and RGS protein interactions regulate activity of AflR, a Zn(II)2Cys6 transcription factor in Aspergillus nidulans. Genetics 2003, 165, 1095–1104. [Google Scholar] [CrossRef]
- Majumdar, R.; Lebar, M.; Mack, B.; Minocha, R.; Minocha, S.; Carter-Wientjes, C.; Sickler, C.; Rajasekaran, K.; Cary, J.C. The Aspergillus flavus Spermidine Synthase (spds) Gene, Is Required for Normal Development, Aflatoxin Production, and Pathogenesis during Infection of Maize Kernels. Front. Plant Sci. 2018, 9, 317. [Google Scholar] [CrossRef]
- Martín, J.; García-Estrada, C.; Rumbero, A.; Recio, E.; Albillos, S.M.; Ullán, R.V.; Martín, J.F. Characterization of an Autoinducer of Penicillin Biosynthesis in Penicillium chrysogenum. Appl. Environm. Microbiol. 2011, 77, 5688–5696. [Google Scholar] [CrossRef]
- Martín, J.; García-Estrada, C.; Kosalková, K.; Ullán, R.V.; Albillos, S.M.; Martín, J.F. The inducers 1,3-diaminopropane and spermidine produce a drastic increase in the expression of the penicillin biosynthetic genes for prolonged time, mediated by the LaeA regulator. Fungal Gen. Biol. 2012, 49, 1004–1013. [Google Scholar] [CrossRef]
- Zhgun, A.A.; Nuraeva, G.K.; Dumina, M.V.; Voinova, T.M.; Dzhavakhiya, V.V.; Eldarov, M.A. 1,3-Diaminopropane and Spermidine Upregulate Lovastatin Production and Expression of Lovastatin Biosynthetic Genes in Aspergillus terreus via LaeA Regulation. Appl. Biochem. Microbiol. 2019, 55, 243–254. [Google Scholar] [CrossRef]
- Zhgun, A.A.; Eldarov, M.A. Spermidine and 1,3-Diaminopropane Have Opposite Effects on the Final Stage of Cephalosporin C Biosynthesis in High-Yielding Acremonium chrysogenum Strain. Int. J. Mol. Sci. 2022, 23, 14625. [Google Scholar] [CrossRef] [PubMed]
- Zhgun, A.A.; Eldarov, M.A. Polyamines Upregulate Cephalosporin C Production and Expression of b-Lactam Biosynthetic Genes in High-Yielding Acremonium chrysogenum Strain. Molecules 2021, 26, 6636. [Google Scholar] [CrossRef] [PubMed]
- García-Estrada, C.; Barreiro, C.; Jami, M.S.; Martín-González, J.; Martín, J.F. The inducers 1,3-diaminopropane and spermidine cause the reprogramming of metabolism in Penicillium chrysogenum, leading to multiple vesicles and penicillin overproduction. J. Proteom. 2013, 85, 129–159. [Google Scholar] [CrossRef] [PubMed]
- Valdés-Santiago, L.; Cervantes-Chávez, J.A.; Winkler, R.; León-Ramírez, C.G.; Ruiz-Herrera, J. Phenotypic comparison of samdc and spe mutants reveals complex relationships of polyamine metabolism in Ustilago maydis. J. Microbiol. 2012, 158, 674–684. [Google Scholar] [CrossRef]
- Guo, K.; Chang, W.-T.; Newell, P.C. Isolation of spermidine synthase gene (spsA) of Dictyostelium discoideum. Biochim. Biophys. Acta 1999, 1449, 211–216. [Google Scholar] [CrossRef]
- Landry, J.; Sternglanzb, R. Yeast Fms1 is a FAD-utilizing polyamine oxidase Biochim. Biophys. Res. Comm. 2003, 303, 771–776. [Google Scholar] [CrossRef]
- Miller-Fleming, L.; Olin-Sandoval, V.; Campbell, K.; Ralser, M. Remaining Mysteries of Molecular Biology: The Role of Polyamines in the Cell. J. Mol Biol. 2015, 427, 3389–3406. [Google Scholar] [CrossRef] [PubMed]
- Rocha, R.O.; Wilson, R.A. Essential, deadly, enigmatic: Polyamine metabolism and roles in fungal cells. Fungal Biol. Rev. 2019, 33, 47–57. [Google Scholar] [CrossRef]
- Hoyt, M.A.; Williams-Abbott, J.W.; Pitkin, J.W.; Davis, R.H. Cloning and expression of the S-adenosylmethionine decarboxylase gene of Neurospora crassa and processing of its product. Mol. Gen. Genet. 2000, 263, 664–673. [Google Scholar] [CrossRef][Green Version]
- Kummasook, A.; Cooper, C.R., Jr.; Sakamoto, A.; Terui, Y.; Kashiwagi, K.; Vanittanakom, N. Spermidine is required for morphogenesis in the human pathogenic fungus, Penicillium marneffei. Fungal Genet. Biol. 2013, 58–59, 25–32. [Google Scholar] [CrossRef] [PubMed]
- Van Poelje, P.D.; Snell, E.E. Pyruvoyl-dependent enzymes. Annu. Rev. Biochem. 1990, 59, 29–59. [Google Scholar] [CrossRef]
- Ekstrom, J.L.; Tolbert, W.D.; Xiong, H.; Pegg, A.E.; Ealick, S.E. Structure of a human S-adenosylmethionine decarboxylase self-processing ester intermediate and mechanism of putrescine stimulation of processing as revealed by the H243A mutant. Biochemistry 2001, 40, 9495–9504. [Google Scholar] [CrossRef] [PubMed]
- Tolbert, W.D.; Zhang, Y.; Cottet, S.E.; Bennett, E.M.; Ekstrom, J.L.; Pegg, A.E.; Ealick, S.E. Mechanism of human S-adenosylmethionine decarboxylase proenzyme processing as revealed by the structure of the S68A mutant. Biochemistry 2003, 42, 2386–2395. [Google Scholar] [CrossRef] [PubMed]
- Stanley, B.A.; Pegg, A.E.; Holm, I. Site of pyruvate formation and processing of mammalian S-adenosylmethionine decarboxylase proenzyme. J. Biol. Chem. 1989, 264, 21073–21079. [Google Scholar] [CrossRef]
- Bale, S.; Ealick, S.E. Structural Biology of S-Adenosylmethionine Decarboxylase. Amino Acids. 2010, 38, 451–460. [Google Scholar] [CrossRef]
- Bale, S.; Lopez, M.M.; Makhatadze, G.I.; Fang, Q.; Pegg, A.E.; Ealick, S.E. Structural Basis for Putrescine Activation of Human S-Adenosylmethionine Decarboxylase. Biochemistry 2008, 47, 13404–13417. [Google Scholar] [CrossRef]
- Strope, P.K.; Skelly, D.A.; Kozmin, S.G.; Mahadevan, G.; Stone, E.A.; Magwene, P.M.; Dietrich, F.S.; McCusker, J.H. The 100-genomes strains, an S. cerevisiae resource that illuminates its natural phenotypic and genotypic variation and emergence as an opportunistic pathogen. Genome Res. 2015, 25, 762–774. [Google Scholar] [CrossRef]
- Hamasaki-Katagiri, N.; Tabor, C.W.; Tabor, H. Spermidine biosynthesis in Saccharomyces cerevisiae: Polyamine requirement of a null mutant of the SPE3 gene (spermidine synthase). Gene 1997, 187, 35–43. [Google Scholar] [CrossRef]
- Ikeguchi, Y.; Bewley, M.C.; Pegg, A.E. Aminopropyltransferases: Function, Structure and Genetics. J. Biochem. 2006, 139, 1–9. [Google Scholar] [CrossRef]
- Jin, Y.; Bok, J.W.; Guzman-De-Peña, D.; Keller, N.P. Requirement of spermidine for developmental transitions in Aspergillus nidulans. Mol. Microbiol. 2002, 46, 801–812. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, R.; Minocha, R.; Lebar, M.D.; Rajasekaran, K.; Long, S.; Carter-Wientjes, C.; Minocha, S.; Caryl, J.W. Contribution of Maize Polyamine and Amino Acid Metabolism toward Resistance against Aspergillus flavus Infection and Aflatoxin Production. Front. Plant Sci. 2019, 10, 692. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, M.K.; Park, M.H.; Tabor, H. Hypusine modification for growth is the major function of spermidine in Saccharomyces cerevisiae polyamine auxotrophs grown in limiting spermidine. Proc. Natl. Acad. Sci. USA 2008, 105, 6554–6559. [Google Scholar] [CrossRef] [PubMed]
- Cano, V.S.; Jeon, G.A.; Johansson, H.E.; Henderson, C.A.; Park, J.H.; Valentini, S.R.; Hershey, J.W.; Park, M.H. Mutational analyses of human eIF5A-1--identification of amino acid residues critical for eIF5A activity and hypusine modification. FEBS J. 2008, 275, 44–58. [Google Scholar] [CrossRef]
- Martínez-Rocha, A.L.; Woriedh, M.; Chemnitz, J.; Willingmann, P.; Kröger, C.; Hadeler, B.; Hauber, J.; Schäfer, W. Posttranslational hypusination of the eukaryotic translation initiation factor-5A regulates Fusarium graminearum virulence. Sci. Rep. 2016, 6, 24698. [Google Scholar] [CrossRef]
- Patel, M.M.; Anchordoquy, T.J. Ability of spermine to differentiate between DNA sequences-preferential stabilization of A-tracts. Biophys. Chem. 2006, 122, 5–15. [Google Scholar] [CrossRef]
- Choi, W.B.; Kang, S.H.; Lee, Y.W.; Lee, Y.H. Cyclic AMP restores appressorium formation inhibited by polyamines in Magnaporthe grisea. Phytopathology 1998, 88, 58–62. [Google Scholar] [CrossRef][Green Version]
- Gárriz, A.; Gonzalez, M.E.; Marina, M.; Ruiz, O.A.; Pieckenstain, F.L. Polyamine metabolism during sclerotial development of Sclerotinia sclerotiorum. Mycol. Res. 2008, 112, 414–422. [Google Scholar] [CrossRef]
- Keinänen, T.A.; Grigorenko, N.; Khomutov, A.R.; Huang, Q.; Uimari, A.; Alhonen, L.; Hyvönen, M.T.; Vepsäläinen, J. Controlling the regioselectivity and stereospecificity of FAD-dependent polyamine oxidases with the use of amine-attached guide molecules as conformational modulators. Biosci. Rep. 2018, 38, BSR20180527. [Google Scholar] [CrossRef]
- Joets, J.; Pousset, D.; Marcireau, C.; Karst, F. Characterization of the Saccharomyces cerevisiae FMS1 gene related to Candida albicans corticosteroid-binding protein 1. Curr. Genet. 1996, 30, 115–120. [Google Scholar] [CrossRef]
- White, W.H.; Gunyuzlu, P.L.; Toyn, J.H. Saccharomyces cerevisiae is capable of De Novo pantothenic acid biosynthesis involving a novel pathway of beta-alanine production from spermine. J. Biol. Chem. 2001, 276, 10794–10800. [Google Scholar] [CrossRef] [PubMed]
- Huang, Q.; Liu, Q.; Hao, Q. Crystal Structures of Fms1 and its Complex with Spermine Reveal Substrate Specificity. J. Mol. Biol. 2005, 348, 951–959. [Google Scholar] [CrossRef]
- Binda, C.; Coda, A.; Angelini, R.; Federico, R.; Ascenzi, P.; Mattevi, A. A 30-angstrom-long U-shaped catalytic tunnel in the crystal structure of polyamine oxidase. Struct. Fold. Des. 1999, 7, 265–276. [Google Scholar] [CrossRef]
- Wang, Y.; Murray-Stewart, T.; Devereux, W.; Hacker, A.; Frydman, B.; Woster, P.M.; Casero, R.A., Jr. Properties of purified recombinant human polyamine oxidase, PAOh1/SMO. Biochem. Biophys. Res. Commun. 2003, 304, 605–611. [Google Scholar] [CrossRef]
- Wang, Y.; Devereux, W.; Woster, P.M.; Stewart, T.M.; Hacker, A.; Casero, R.A., Jr. Cloning and characterization of a human polyamine oxidase that is inducible by polyamine analogue exposure. Cancer Res. 2001, 61, 5370–5373. [Google Scholar] [PubMed]
- Vujcic, S.; Diegelman, P.; Bacchi, C.J.; Kramer, D.L.; Porter, C.W. Identification and characterization of a novel flavin-containing spermine oxidase of mammalian cell origin. Biochem. J. 2002, 367, 665–667. [Google Scholar] [CrossRef]
- Adachi, M.S.; Torres, J.M.; Fitzpatrick, P.F. Mechanistic studies of the yeast polyamine oxidase Fms1: Kinetic mechanism, substrate specificity, and pH dependence. Biochemistry 2010, 49, 10440–10448. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Murray-Stewart, T.; Wang, Y.; Devereux, W.; Casero, R.A. Cloning and characterization of multiple human polyamine oxidase splice variants that code for isoenzymes with different biochemical characteristics. Biochem. J. 2002, 368, 673–677. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Higashi, T.; Machida, H.; Iwasaki, S.; Horikoshi, K. Oxidation of acetylpolyamines by extracellular polyamine oxidase produced by Penicillium sp. nov. PO-I. Biochim. Biophys. Acta 1983, 743, 431–436. [Google Scholar] [CrossRef]
- Hammerstad, M.; Hersleth, H.-P. Overview of structurally homologous flavoprotein oxidoreductases containing the low Mr thioredoxin reductase-like fold—A functionally diverse group. Arch. Biochem. Biophys. 2021, 702, 108826. [Google Scholar] [CrossRef]
- van den Berg, M.A.; Albang, R.; Albermann, K.; Badger, J.H.; Daran, J.-M.; Driessen, A.J.; García-Estrada, C.; Fedorova, N.D.; Harris, D.M.; Heijne, W.H.; et al. Genome sequencing and analysis of the filamentous fungus Penicillium chrysogenum. Nat. Biotechnol. 2008, 26, 1161–1168. [Google Scholar] [CrossRef]
- White, W.H.; Skatrud, P.L.; Xue, Z.; Toyn, J.H. Specialization of function among aldehyde dehydrogenases: The ALD2 and ALD3 genes are required for beta-alanine biosynthesis in Saccharomyces cerevisiae. Genetics 2003, 163, 69–77. [Google Scholar] [CrossRef]
- Richardson, I.B.; Hurley, S.K.; Hynes, M.J. Cloning and molecular characterisation of the amdR controlled gatA gene of Aspergillus nidulans. Mol. Gen. Genet. 1989, 217, 118–125. [Google Scholar] [CrossRef] [PubMed]
- Ramos, F.; el Guezzar, M.; Grenson, M.; Wiame, J.M. Mutations affecting the enzymes involved in the utilization of 4-aminobutyric acid as nitrogen source by the yeast Saccharomyces cerevisiae. Eur. J. Biochem. 1985, 149, 401–404. [Google Scholar] [CrossRef]
- der Garabedian, P.A.; Lotti, A.-M.; Vermeersch, J.J. 4-Aminobutyrate: 2-oxoglutarate amino transferase from Candida Purification and properties. Eur. J. Biochem. 1986, 156, 589–596. [Google Scholar] [CrossRef] [PubMed]
- Schor, D.S.; Struys, E.A.; Hogema, B.M.; Gibson, K.M.; Jakobs, C. Development of a stable-isotope dilution assay for gamma-aminobutyric acid (GABA) transaminase in isolated leukocytes and evidence that GABA and beta-alanine transaminases are identical. Clin. Chem. 2001, 47, 525–531. [Google Scholar] [CrossRef]
- Andersen, G.; Andersen, B.; Dobritzsch, D.; Klaus, D.; Schnackerz, K.D.; Piskur, J.P. A gene duplication led to specialized g-aminobutyrate and b-alanine aminotransferase in yeast. FEBS J. 2007, 274, 1804–1817. [Google Scholar] [CrossRef] [PubMed]
- Liu, W.; Peterson, P.E.; Carter, R.J.; Zhou, X.; Langston, J.A.; Fisher, A.J.; Toney, M.D. Crystal structures of unbound and aminooxyacetate-bound Escherichia coli gamma-aminobutyrate aminotransferase. Biochemistry 2004, 43, 10896–10905. [Google Scholar] [CrossRef]
- Liu, W.; Peterson, P.E.; Langston, J.A.; Jin, X.; Zhou, X.; Fisher, A.J.; Toney, M.D. Kinetic and crystallographic analysis of active site mutants of Escherichia coli gamma-aminobutyrate aminotransferase. Biochemistry 2005, 44, 2982–2992. [Google Scholar] [CrossRef]
- Bönnighausen, J.; Gebhard, D.; Kröger, C.; Hadeler, B.; Tumforde, T.; Lieberei, R.; Bergemann, J.; Schäfer, W.; Bormann, J. Disruption of the GABA shunt affects mitochondrial respiration and virulence in the cereal pathogen Fusarium graminearum. Mol. Microbiol. 2015, 98, 1115–1132. [Google Scholar] [CrossRef]
- Ďurišová, K.; Šimkovič, M.; Varečka, L.; Kaliňák, M.; Šoltys, K.; Mosná, B.; Kryštofová, S. Functional dissection of gamma-aminobutyric acid metabolism in Neurospora crassa. Gen. Physiol. Biophys. 2020, 39, 205–218. [Google Scholar] [CrossRef] [PubMed]
- Straffon, M.J.; Hynes, M.J.; Davis, M.A. Characterization of the ugatA gene of Ustilago maydis, isolated by homology to the gatA gene of Aspergillus nidulans. Curr. Genet. 1996, 29, 360–369. [Google Scholar] [CrossRef] [PubMed]
- Shimoi, H.; Okuda, M.; Ito, K.J. Molecular cloning and application of a gene complementing pantothenic acid auxotrophy of sake yeast Kyokai no. 7. Biosci. Bioeng. 2000, 90, 643–647. [Google Scholar] [CrossRef]
- Rueda-Mejia, M.P.; Bühlmann, A.; Ortiz-Merino, R.A.; Lutz, S.; Ahrens, C.H.; Künzler, M.; Freimoser, F.M. Pantothenate Auxotrophy in a Naturally Occurring Biocontrol Yeast. Appl. Environ. Microbiol. 2023, 89, e0088423. [Google Scholar] [CrossRef]
- Martín, J.F.; Liras, P.; Sánchez, S. Modulation of Gene Expression in Actinobacteria by Translational Modification of Transcriptional Factors and Secondary Metabolite Biosynthetic Enzymes. Front. Microbiol. 2021, 12, 630694. [Google Scholar] [CrossRef]
- Genschel, U. Coenzyme A Biosynthesis: Reconstruction of the Pathway in Archaea and an Evolutionary Scenario Based on Comparative Genomics. Mol. Biol. Evol. 2004, 21, 1242–1251. [Google Scholar] [CrossRef]
- Dansie, L.E.; Reeves, S.; Miller, K.; Zano, S.P.; Frank, M.; Pate, C.; Wang, J.; Jackowski, S. Physiological roles of the pantothenate kinases. Biochem. Soc. Trans. 2014, 42, 1033–1036. [Google Scholar] [CrossRef]
- Pietrocola, F.; Lorenzo Galluzzi, L.; Bravo-San Pedro, J.M.; Madeo, F.; Kroemer, G. Acetyl Coenzyme A: A Central Metabolite and Second Messenger. Cell Metab. 2015, 21, 805–821. [Google Scholar] [CrossRef]
- Keszenman-Pereyra, D.; Lawrence, S.; Twfieg, M.E.; Price, J.; Turner, G. The npgA/cfwA gene encodes a putative 4_-phosphopantetheinyl transferase which is essential for penicillin biosynthesis in Aspergillus nidulans. Curr. Genet. 2003, 43, 186–190. [Google Scholar] [CrossRef]
- Márquez-Fernández, O.; Trigos, A.; Ramos-Balderas, J.L.; Viniegra-Gonzalez, G.; Deising, H.B.; Aguirre, J. Phosphopantetheinyl transferase CfwA/NpgA is required for Aspergillus nidulans secondary metabolism and asexual development. Eukaryot. Cell 2007, 6, 710–720. [Google Scholar] [CrossRef]
- García-Estrada, C.; Ullán, R.V.; Velasco-Conde, T.; Godio, R.P.; Teijeira, F.; Vaca, I.; Feltrer, R.; Kosalková, K.; Mauriz, E.; Martín, J.F. Post-translational enzyme modification by the phosphopantetheinyl transferase is required for lysine and penicillin biosynthesis but not for roquefortine or fatty acid formation in Penicillium chrysogenum. Biochem. J. 2008, 415, 317–324. [Google Scholar] [CrossRef]
- Hodges, P.E.; McKee, A.H.; Davis, B.P.; Payne, W.E.; Garrels, J.I. The Yeast Proteome Database (YPD): A model for the organization and presentation of genome-wide functional data. Nucleic Acids Res. 1999, 27, 69–73. [Google Scholar] [CrossRef] [PubMed]
- Kurtov, D.; Kinghorn, J.R.; Unkles, S.E. The Aspergillus nidulans panB gene encodes ketopantoate hydroxymethyltransferase, required for biosynthesis of pantothenate and Coenzyme, A. Mol. Gen. Genet. 1999, 262, 115–120. [Google Scholar] [CrossRef] [PubMed]
- Genschel, U.; Powell, C.A.; Abell, C.; Smith, A.G. The final step of pantothenate biosynthesis in higher plants: Cloning and characterization of pantothenate synthetase from Lotus japonicus and Oryza sativum (rice). Biochem. J. 1999, 341, 669–678. [Google Scholar] [CrossRef] [PubMed]
- Olzhausen, J.; Schübbe, S.; Schüller, H.-J. Genetic analysis of coenzyme A biosynthesis in the yeast Saccharomyces cerevisiae: Identification of a conditional mutation in the pantothenate kinase gene CAB1. Curr. Genet. 2009, 55, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Stolz, J.; Sauer, N. The fenpropimorph resistance gene FEN2 from Saccharomyces cerevisiae encodes a plasma membrane H1-pantothenate symporter. J. Biol. Chem. 1999, 274, 18747–18752. [Google Scholar] [CrossRef]
- Daugherty, M.; Polanuyer, B.; Farrell, M.; Scholle, M.; Lykidis, A.; de Crécy-Lagard, V.; Osterman, A.J. Complete reconstitution of the human coenzyme A biosynthetic pathway via comparative genomics. Biol. Chem. 2002, 277, 21431–21439. [Google Scholar] [CrossRef]
- Calder, R.B.; Williams, R.S.; Ramaswamy, G.; Rock, C.O.; Campbell, E.; Unkles, S.E.; Kinghorn, J.R.; Jackowski, S. Cloning and Characterization of a Eukaryotic Pantothenate Kinase Gene (panK) from Aspergillus nidulans. J. Biol. Chem. 1999, 274, 2014–2020. [Google Scholar] [CrossRef]
- de la Sierra-Gallay, I.L.; Collinet, B.; Graille, M.; Quevillon-Cheruel, S.; Liger, D.; Minard, P.; Blondeau, K.; Henckes, G.; Aufrere, R.; Leulliot, N.; et al. Crystal structure of the YGR205w protein from Saccharomyces cerevisiae: Close structural resemblance to E. coli pantothenate kinase. Proteins 2004, 54, 776–783. [Google Scholar] [CrossRef]
- Lambalot, R.H.; Gehring, A.M.; Flugel, R.S.; Zuber, P.; LaCelle, M.; Marahiel, M.A.; Reid, R.; Khosla, C.; Walsh, C.T. A new enzyme superfamily—The phosphopantetheinyl transferases. Chem. Biol. 1996, 3, 923–936. [Google Scholar] [CrossRef]
- Mootz, H.D.; Finking, R.; Marahiel, M.A. 4’-phosphopantetheine transfer in primary and secondary metabolism of Bacillus subtilis. J. Biol. Chem. 2001, 276, 37289. [Google Scholar] [CrossRef]
- Casqueiro, J.; Gutiérrez, S.; Bañuelos, O.; Fierro, F.; Velasco, J.; Martín, J.F. Characterization of the lys2 gene of Penicillium chrysogenum encoding α-aminoadipic acid reductase. Mol. Gen. Genet. 1998, 259, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Hijarrubia, M.J.; Aparicio, J.F.; Casqueiro, J.; Martín, J.F. Characterization of the lys2 gene of Acremonium chrysogenum encoding a functional α-aminoadipate activating and reducing enzyme. Mol. Gen. Genet. 2001, 264, 755–762. [Google Scholar] [CrossRef]
- Hijarrubia, M.J.; Aparicio, J.F.; Martín, J.F. Domain structure characterization of the mulfunctional α-aminoadipate reductase from Penicillium chrysogenum by limited proteolysis. J. Biol. Chem. 2003, 278, 8250–8256. [Google Scholar] [CrossRef] [PubMed]
- Walsh, C.T.; Gehring, A.M.; Weinreb, P.H.; Quadri, L.; Flugel, R.S. Post-translational modification of polyketide and nonribosomal peptide synthases. Curr. Opin. Chem. Biol. 1997, 1, 309–315. [Google Scholar] [CrossRef]
- Wang, Y.Y.; Zhang, X.S.; Luo, H.D.; Ren, N.N.; Jiang, X.H.; Jiang, H.; Jiang, H.; Li, Y.-Q. Characterization of discrete phosphopantetheinyl transferases in Streptomyces tsukubaensis L19 unveils a complicate phosphopantetheinylation network. Sci. Rep. 2016, 6, 24255. [Google Scholar] [CrossRef] [PubMed]
- Caboche, S.; Pupin, M.; Leclère, V.; Fontaine, A.; Jacques, P.; Kucherov, G. NORINE: A database of nonribosomal peptides. Nucleic Acids Res. 2008, 36, D326–D331. [Google Scholar] [CrossRef]
- Galagan, J.E.; Calvo, S.E.; Cuomo, C.; Ma, L.-J.; Wortman, J.R.; Batzoglou, S.; Lee, S.-I.; Türkmen, M.B.; Spevak, C.; Clutterbuck, J.; et al. Sequencing of Aspergillus nidulans and comparative analysis with A. fumigatus and A. oryzae. Nature 2005, 438, 22–29. [Google Scholar] [CrossRef]
- Suo, Z.; Tseng, C.C.; Walsh, C.T. Purification, priming and catalytic acylation of carrier protein domains in the polyketide synthase and nonribosomal peptidyl synthetase modules of the HMWP1 subunit of yersiniabactin synthetase. Proc. Natl. Acad. Sci. USA 2001, 98, 99–104. [Google Scholar] [CrossRef]
- Fichtlscherer, F.; Wellein, C.; Mittag, M.; Schweizer, E. A novel function of yeast fatty acid synthetase. Subunit alpha is capable of self-pantetheinylation. Eur. J. Biochem. 2000, 267, 2666–2671. [Google Scholar] [CrossRef]
- Mofid, M.R.; Finking, R.; Marahiel, M.A. Recognition of hybrid peptidyl carrier proteins/acyl carrier proteins in nonribosomal peptide synthetase modules by the 4-phosphopantetheinyl transferases AcpS and Sfp. J. Biol. Chem. 2002, 277, 17023–17031. [Google Scholar] [CrossRef]
- Beld, J.; Sonnenschein, E.C.; Vickery, C.R.; Noel, J.P.; Burkart, M.D. The phosphopantetheinyl transferases: Catalysis of a post-translational modification crucial for life. Nat. Prod. Rep. 2014, 31, 61–108. [Google Scholar] [CrossRef]
- Ehmann, D.E.; Gehring, A.M.; Walsh, C.T. Lysine biosynthesis in Saccharomyces cerevisiae: Mechanism of alpha-aminoadipate reductase (Lys2) involves posttranslational phosphopantetheinylation by Lys5. Biochemistry 1999, 38, 6171–6177. [Google Scholar] [CrossRef] [PubMed]
- Oberegger, H.; Eisendle, M.; Schrettl, M.; Graessle, S.; Haas, H. 4-Phosphopantetheinyl transferase-encoding npgA is essential for siderophore biosynthesis in Aspergillus nidulans. Curr. Genet. 2003, 44, 211–215. [Google Scholar] [CrossRef] [PubMed]
- Liras, P.; Martín, J.F. Interconnected Set of Enzymes provide Lysine Biosynthetic Intermediates and Ornithine Derivatives as Key Precursors for the Biosynthesis of Bioactive Secondary. Antibiotics 2023, 12, 159. [Google Scholar] [CrossRef]
- Fierro, F.; Barredo, J.L.; Díez, B.; Gutiérrez, S.; Fernández, F.J.; Martín, J.F. The penicillin gene cluster is amplified in tandem repeats linked by conserved hexanucleotide sequences. Proc. Natl. Acad. Sci. USA 1995, 92, 6200–6204. [Google Scholar] [CrossRef]
- Fierro, F.; García-Estrada, C.; Castillo, I.; Rodríguez, R.; Velasco-Conde, T.; Martín, J.F. Transcriptional and bioinformatic analysis of the 56.8 kb DNA region amplified in tandem repeats containing the penicillin gene cluster in Penicillium chrysogenum. Fungal Genet. Biol. 2006, 43, 618–629. [Google Scholar] [CrossRef]
- García-Estrada, C.; Ullán, R.V.; Albillos, S.M.; Fernández-Bodega, M.Á.; Durek, P.; von Döhren, H.; Martín, J.F. A single cluster of coregulated genes encodes the biosynthesis of the mycotoxins roquefortine C and meleagrin in Penicillium chrysogenum. Chem. Biol. 2011, 18, 1499–1512. [Google Scholar] [CrossRef] [PubMed]
- Kosalková, K.; Domínguez-Santos, R.; Coton, M.; Coton, E.; García-Estrada, C.; Liras, P.; Martín, J.F. A natural short pathway synthesizes roquefortine C but not meleagrin in three different Penicillium roqueforti strains. Appl. Microbiol. Biotechnol. 2015, 99, 7601–7612. [Google Scholar] [CrossRef]
- Casqueiro, J.; Gutiérrez, S.; Bañuelos, O.; Hijarrubia, M.J.; Martín, J.F. Gene targeting in Penicillium chrysogenum: Disruption of the lys2 gene leads to penicillin overproduction. J. Bacteriol. 1999, 181, 1181–1188. [Google Scholar] [CrossRef]
- Eisenberg, T.; Knauer, H.; Schauer, A.; Büttner, S.; Ruckenstuhl, C.; Carmona-Gutierrez, D.; Ring, J.; Schroeder, S.; Magnes, C.; Antonacci, L.; et al. Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 2009, 11, 1305–1314. [Google Scholar] [CrossRef]
- Madeo, F.; Eisenberg, T.; Büttner, S.; Ruckenstuhl, C.; Kroemer, G. Spermidine: A novel autophagy inducer and longevity elixir. Autophagy 2010, 6, 160–162. [Google Scholar] [CrossRef] [PubMed]
- Leitao, A.L.; Enguita, F.J.; de la Fuente, J.L.; Liras, P.; Martín, J.F. Inducing effect of diamines on transcription of the cephamycin C genes from the lat and pcbAB promoters in Nocardia lactamdurans. J. Bacteriol. 1999, 181, 2379–2384. [Google Scholar] [CrossRef]
- Ordóñez-Robles, M.; Rodríguez-García, A.; Martín, J.F. Target genes of the Streptomyces tsukubaensis FkbN regulator include most of the tacrolimus biosynthesis genes, a phosphopantetheinyl transferase and other PKS genes. Appl. Microbiol. Biotechnol. 2016, 100, 8091–8103. [Google Scholar] [CrossRef] [PubMed]
- Large, P.J. Enzymes and pathways of polyamine breakdown in microorganisms. FEMS Microbiol. 1992, 8, 249–262. [Google Scholar] [CrossRef] [PubMed]
- Blackwell, M. The fungi: 1,2,3…5.1 million species? Am. J. Bot. 2011, 98, 426–438. [Google Scholar] [CrossRef]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Martín, J.F.; Liras, P. Diamine Fungal Inducers of Secondary Metabolism: 1,3-Diaminopropane and Spermidine Trigger Enzymes Involved in β-Alanine and Pantothenic Acid Biosynthesis, Precursors of Phosphopantetheine in the Activation of Multidomain Enzymes. Antibiotics 2024, 13, 826. https://doi.org/10.3390/antibiotics13090826
Martín JF, Liras P. Diamine Fungal Inducers of Secondary Metabolism: 1,3-Diaminopropane and Spermidine Trigger Enzymes Involved in β-Alanine and Pantothenic Acid Biosynthesis, Precursors of Phosphopantetheine in the Activation of Multidomain Enzymes. Antibiotics. 2024; 13(9):826. https://doi.org/10.3390/antibiotics13090826
Chicago/Turabian StyleMartín, Juan Francisco, and Paloma Liras. 2024. "Diamine Fungal Inducers of Secondary Metabolism: 1,3-Diaminopropane and Spermidine Trigger Enzymes Involved in β-Alanine and Pantothenic Acid Biosynthesis, Precursors of Phosphopantetheine in the Activation of Multidomain Enzymes" Antibiotics 13, no. 9: 826. https://doi.org/10.3390/antibiotics13090826
APA StyleMartín, J. F., & Liras, P. (2024). Diamine Fungal Inducers of Secondary Metabolism: 1,3-Diaminopropane and Spermidine Trigger Enzymes Involved in β-Alanine and Pantothenic Acid Biosynthesis, Precursors of Phosphopantetheine in the Activation of Multidomain Enzymes. Antibiotics, 13(9), 826. https://doi.org/10.3390/antibiotics13090826





