1. Introduction
Bioplastics issued from renewable resources represent an interesting alternative to reduce carbon footprints instead of using polymers made of fossil carbon [
1]. Therefore, significant progress in the development of bio-based polymers with renewable feedstock has generated great interest in the polymer industry [
2]. Starch is one of the abundant biopolymers with diverse applications in many fields such as food processing, sizing material in textiles as binding agent to reduce friction and in vat dyes for the printing of 100% cotton fabrics by virtue of its cost effectiveness, biodegradability and renewability [
3]. It consists of amylose and amylopectin. Amylose is a linear polymer consisting of α-(1–4) glucose units, while amylopectin is a branched polymer consisting of α-(1–4) glucose units with intermittent divisions of α-(1–6) connections [
4]. Therefore, thermoplastic processing of starch is difficult due to the presence of intermolecular and intramolecular hydrogen bond linkages in its macromolecules that restrict the molecular chain movement due to the presence of resilient interactive forces [
5]. Such interactive forces are repelled by the use of plasticizers such as glycol, glycerol, sorbitol or urea that weaken the hydrogen bonds present within the macromolecules and thermoplastic processing of starch is improved [
6]. These plasticizers disrupt the crystalline structure of starch macromolecules and allows free molecular chain movement by nullifying the resilient interactive forces by a process called gelatinization [
7]. However, the foremost disadvantage of using plasticizers in starch is the emergence of a hydrophilic characteristic that induces poor water resistance in the polymer hence negatively impacts the mechanical properties of polymer [
8]. The other drawbacks associated with native starch are lack of fluidity and higher viscosity that limits its uses in many technical applications [
9].
Such shortcomings can be remedied by the oxidation of starch by using oxidizing agents [
10]. Starch oxidation results in de-polymerization by the hydrolysis of glucose units hence oxidized starches present low viscosities at higher concentrations: a property desired for thermoplastic processing of starch [
11]. Many oxidizing agents for starch oxidation have been used in the past such as sodium hypochlorite, hydrogen peroxide and ammonium persulfate. However, sodium hypochlorite has been found to be the most efficient one and widely used in industrial production, but in the recent past, this oxidizing agent has been abandoned due to the formation of hazardous chlorinated byproducts that are harmful to the environment [
12]. Therefore, we used maize starch that was oxidized by sodium perborate (SPB) which is an efficient and environment-friendly oxidizing agent for the oxidation of native starch. In practice, SPB is not only cost effective but also presents distinctive benefits such as being available as a solid form of hydrogen peroxide [
13]. However, SPB has proven to be a better alternative to hydrogen peroxide since the former has not only provided superior stability at higher temperatures but also proven to be an efficient source of per hydroxyl anions, superoxide and active oxygen [
14].
Blending two different polymers not only can offer valuable properties but also a new composite material can be developed that can present desired functionality for a certain application [
15]. Polylactic acid (PLA) is a linear aliphatic polyester [
16] that is derived from renewable resources and one of the most important bio-based polymers with attractive physical and performance properties can serve as an alternative to petroleum based polymers in many applications [
17]. The blending of starch and PLA will not only make the composite material cost effective but also certain functional properties can be enhanced such as flame retardancy [
18]. Although PLA is less flammable than other synthetic thermoplastics [
19] in its pure form by virtue of having higher limiting oxygen index (24–26) than other polyesters (20–22) and lower peak heat release rate, PLA is still combustible, which limits its uses in many flame-retardant applications [
20]. To improve the flame retardancy of PLA, intumescent flame retardants (IFRs) present an extremely efficient approach through which a char layer is formed on the polymer surface that not only can protect it from further burning but also restricts the passage of heat, oxygen and volatile compounds to the sight of burning [
21]. Although, IFRs generally contain a petroleum based carbonic source together with a halogen-free acidic source, but in the recent past, a more sustainable approach towards flame retardancy has been promoted by using biopolymers that contain a bio-based carbonic source [
22]. Starch could be a suitable contender as a bio-based carbonic source in intumescent formulations by virtue of its natural charring capability and presence of excessive polyhydric compounds. Wang et al. used starch as natural charring agent in PLA foams [
23] however, the spinning of PLA/starch compounds containing intumescent formulations is still unknown.
Therefore, in this study we have investigated the melt spinnability of PLA/oxidized starch (OS) composites containing halogen free intumescent flame-retardants with OS being a bio-based carbonic source presenting sustainable approach towards flame retardancy. We used maize starch that was oxidized by sodium perborate (SPB) which is an efficient and environment friendly oxidizing agent for the oxidation of native starch to improve its thermoplastic processing and spinnability. Composites were melt-spun to multifilament fibers on pilot-scale melt spinning machine and fibers were then needle-punched to form non-woven structures. The changes in fiber crystallinity and mechanical properties were measured in response to the variations in spinning process parameters. We also tested the thermal stability of fibers by thermogravimetric analysis, while surface morphology and dispersion of the additives was recorded by scanning electron microscopy. The fire characteristics such as time to ignition, heat release rate, and total heat release were tested by cone calorimetry as standard testing methods.
2. Materials and Methods
2.1. Materials
Oxidized starch (OS) 400L-NF, a white fine powder, containing 20 mg kg−1 of SPB as an oxidizing agent was kindly provided by Roquette Freres SA (Lestrem, France). PLA Luminy L130 which is a highly-crystalline polymer (L-isomer ≥ 99) with a density of 1.24 g cm−3 and melting temperature of 175–180 °C was purchased from Total-Corbion NV (Gorinchem, Netherlands). Halogen-free flame retardant (FR, Exolit AP 422) having decomposition temperature higher than 275 °C was attained from Clariant AG (Muttenz, Switzerland). Exolit AP 422 is a fine-particle ammonium polyphosphate (APP) containing 31% (w/w) phosphorous and 14% (w/w) nitrogen, having density of 1.9 g cm−3 and average particle size of 17 µm, used as acid donor in intumescent formulations. PLA, APP and OS were vacuum dried at 100 °C for 4 h before compounding.
2.2. Preparation of Composites
PLA/OS/APP composites were prepared on twin-screw extruder (ZSK Mc
18) from Coperion GmbH (Stuttgart, Germany). OS was mixed together with PLA pellets in four different concentrations, i.e., 1, 3, 5 and 7 wt% in the first feeding zone which were then compounded together with APP present in the second feeding zone. PLA/OS1/APP10, PLA/OS3/APP10, PLA/OS5/APP10 and PLA/OS7/APP10 composites were prepared at screw rotation speed of 500 rpm. The formulations with content of each component (w/w) of as prepared composites are presented in
Table 1. The temperatures of the three heating zones were kept at 160 °C, 170 °C and 180 °C, respectively.
2.3. Melt Spinning of Composites
PLA/OS/APP composite pellets were vacuum dried at 100 °C for 4 h prior to melt spinning. Drying the pellets before melt spinning is necessary to control moisture (%) in the polymer to avoid hydrolysis that can lower the viscosity of the melt and ultimately fiber breakage during spinning can occur. Karl Fischer titrations analyzed by ASTM D6869-17 method confirmed that the moisture content in the pellets were lower than 80 ppm. Pilot scale melt spinning machine (
Figure 1) with a throughput of few kilograms an hour was used to spin PLA/OS/APP composite pellets to multifilament fibers. The composite pellets were dosed into the hopper of the melt-spinning machine and were then transferred to the extruder where they were melted at a temperature range of 190 °C to 215 °C. Constant rpm of spin pump ensured a homogeneous supply of the melt to the spinneret die containing 24 monofilament holes. After ejecting from spinneret die, monofilaments were delivered through the quenching section where chilled air running at a velocity of 0.5 m s
−1 helped in lowering the temperature of the monofilaments. They were joined together into multifilaments by passing through the spin finish before being collected by the take-up roller. The bobbins carrying multifilaments were placed in standard atmospheric conditions before further analysis. The spinning parameters used to produce multifilament fibers are presented in
Table 2.
2.4. Scanning Electron Microscopy
Hitachi S-3200 scanning electron microscope (SEM, Chiyoda, Tokyo, Japan) was used to determine surface morphology and dispersion of the additives in melt spun fibers. Multifilament fibers were first frozen in liquid nitrogen and then were delicately cut with a razor blade to obtain clear SEM images. The cut samples were attached to the silver coated standard specimen stubs which were then sputter coated with gold. The system used for sputter coating was PECS 682, USA. The thickness of the gold sputter coating was 150 Å. Fiber samples were examined at a magnification of 1000 × and at an accelerating voltage of 20 kV.
2.5. Apparent Viscosity Measurement
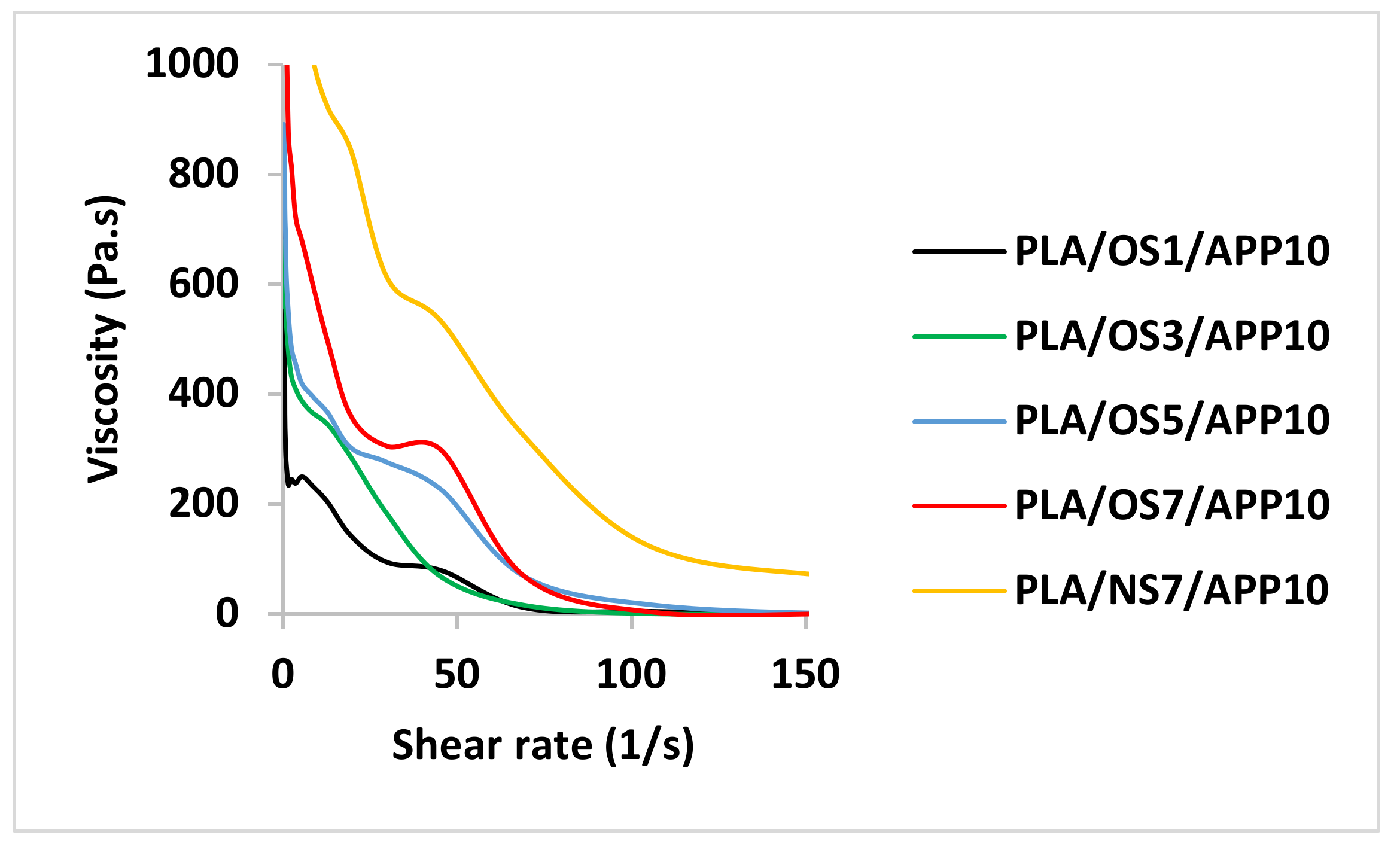
A Brookfield digital rheometer (Model, DV-III, Middleboro, USA) was used to measure the apparent viscosity of the composite samples. The samples were first dried and then around 5–10 grams from each sample were dispersed in 100 mL of distilled water to prepare the solutions. The as prepared solutions were stirred continuously for an hour and kept for 15 min at a temperature of 80 °C. The volume to be used from each sample was adjusted to 100 mL and the pH used was 9.0. The solutions were then left for cooling at 25 °C and then apparent viscosity of each sample was measured at a shear rate of up to 150 s−1.
2.6. Mechanical Testing
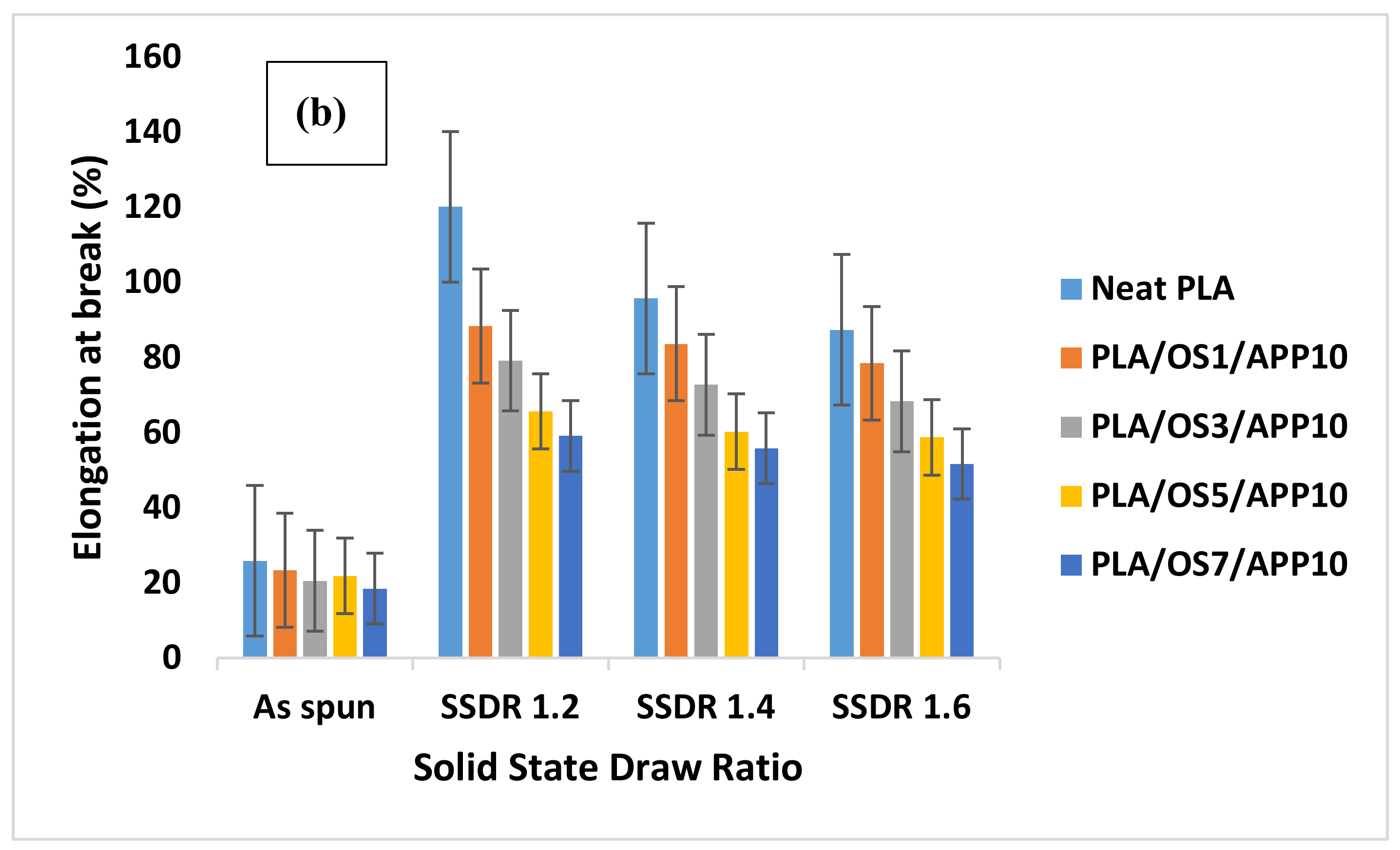
Mechanical properties of multifilament fibers such as tenacity (cN/tex) and elongation at break (%) were tested on Zwick Roell testing machine by using EN ISO 5079 standard method. The specimen lengths (50 mm) and rate of deformation (50 mm min−1) were kept constant for all samples. Ten specimens were prepared from each sample and their average results with standard deviations were recorded.
2.7. Thermogravimetric Analysis
Thermal stabilities and residual mass (%) of the fiber samples up to 500 °C were analyzed by thermogravimetric analysis using TGA Q5000 equipment (TA Instruments, New Castle, Delaware, USA). Fiber samples of 10–15 mg were heated at a constant rate of 10 °C min−1 up to 500 °C under nitrogen atmosphere at a flow rate of 50 mL min−1. The initial temperature of measurement for TGA was 20 °C. The temperatures at which maximum decomposition of fibers occurred were noted and the residual mass (%) of the samples were compared with the initial mass of the samples. Thermogravimetric (TG) curves of the samples were plotted and were analyzed in detail.
2.8. Differential Scanning Calorimetry
Thermal properties such as glass transition temperature (Tg), cold crystallization temperature (Tcc), melting temperature (Tm) and crystallinity (%) of multifilament fibers were investigated by Differential scanning calorimetry (DSC) under nitrogen atmosphere at a stream rate of 50 mL min
−1. The samples were heated at a constant rate of 10 °C min
−1 starting from 0 °C to 230 °C and were then cooled at the same rate followed by heating again as above. The degree of crystallinity (Xc) of multifilament fibers was calculated by Equation (1),
where Xc corresponds to degree of crystallinity of the sample; ΔHm implies to heat of fusion of the sample; ΔHcc corresponds to cold crystallization enthalpy; ΔHf relates to the heat of fusion of 100% crystalline material, and Wfr is the net weight fraction of the polymer. The heat of fusion of 100% crystalline PLA (ΔHf) is approximately 93.6 J g
−1.
2.9. Needlepunched Non-Wovens
Needlepunching is a process by which fibers are mechanically bonded by entangling them together with the barbed needles penetrating through the fibrous web to form non-woven fabrics. Multifilament fibers produced by melt spinning were cut to short fibers and passed through a carding machine: a process by which fibers were opened and homogeneously blended to form fibrous web of uniform areal density. The unbonded and voluminous fibrous web was fed to the needlepunching machine by a pair of feed rollers, and then passed between a pair of perforated plates as shown in
Figure 2. The barbed needles responsible for entangling the fibrous web were arranged on the needle board that was placed on a reciprocating beam operated through an eccentric crank mechanism. In the downward motion, barbed needles ran down through the perforations of the top and bottom bed plates and during the upward motion were withdrawn upwards; therefore, by doing so fibrous webs were mechanically bonded and mechanical strength of the non-woven fabric was improved. The delivery rollers delivered the mechanically bonded non-woven fabric, which was then thermally molded to impart uniform thickness and areal density.
2.10. Fire Testing
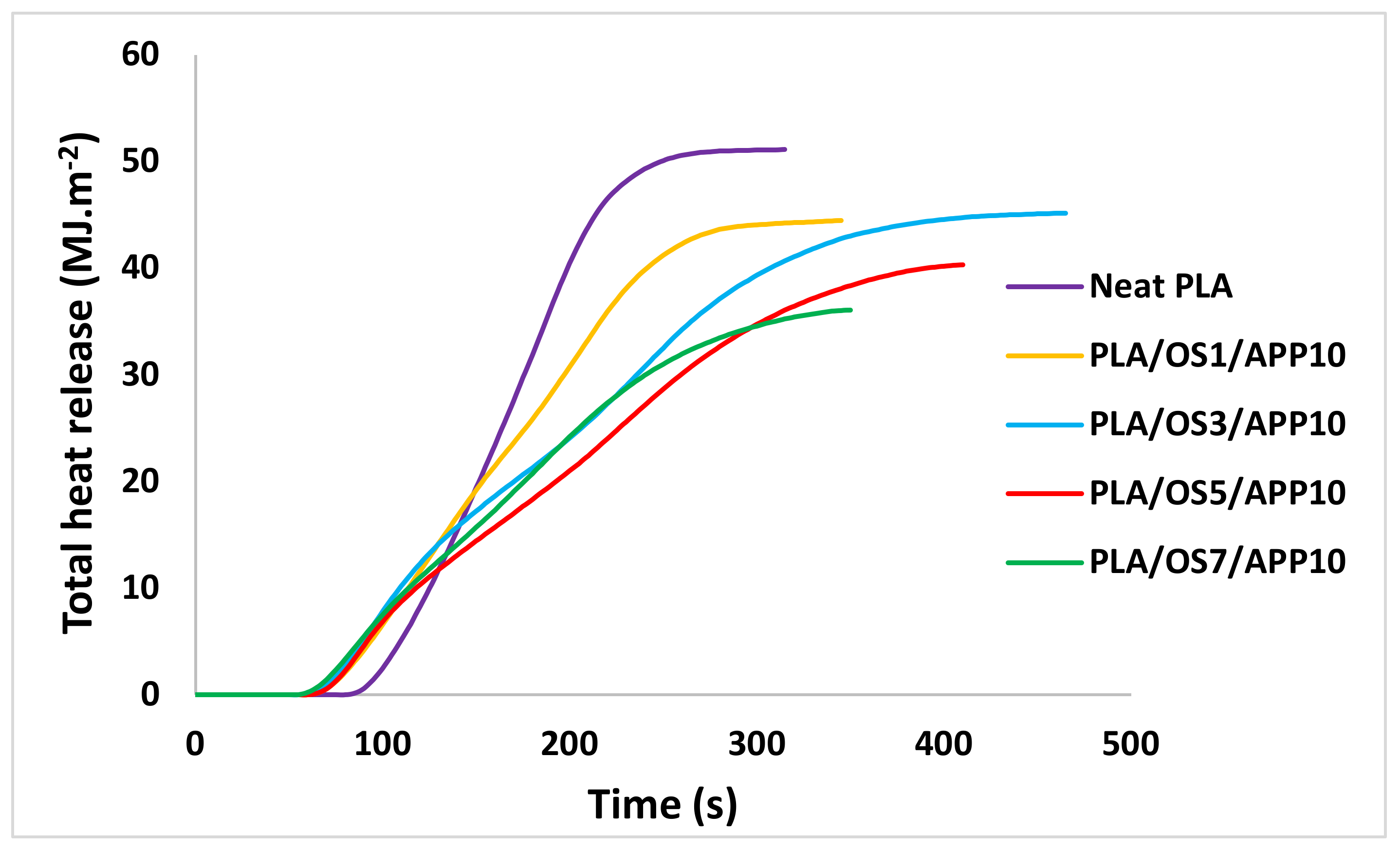
The cone calorimeter is one of the most important fire testing equipment that is used to measure the heat release rate (HRR) of the sample. The basic principle of this equipment is based on the consumption of oxygen which is considered equivalent to the amount of heat released during combustion. This equipment can also measure the amount of smoke produced during combustion. The non-woven fabric samples with dimensions of 100 × 100 × 3 mm3 were prepared to perform a cone calorimetry test, according to ISO 5660 standard testing method, with heat flux of 35 kW m−2 by using Stanton Redcroft instrument (Illinois Toolworks, Glenview, IL, USA). The other important fire testing results that can be obtained through this equipment are time to ignition (TTI), total heat release (THR), total smoke production (TSP) and residual mass % of the sample.
2.11. Fourier Transform Infrared Spectra (FTIR)
Fourier transform infrared (FTIR) spectra of non-woven fabrics before and after burning were recorded on a Perkin Elmer Spectrum 1000 FTIR spectrometer at room temperature and the sample powders of around 10 mg were mixed with 200 mg KBr powder and subsequently a mixture of about 50 mg was pressed to form a tablet for IR measurements. FTIR spectra were recorded in the range of 500 to 4000 cm−1.
4. Conclusions
In this study, modified starch oxidized by SPB was used as carbonization agent in intumescent flame-retardant blends. PLA/OS/APP blends containing different wt% of OS were melt-blended using a twin screw extruder and then melt spun on a pilot-scale melt-spinning machine to develop flame-retardant multifilament fibers. The impact of oxidized starch together with ammonium polyphosphate was thoroughly investigated by optimizing melt-spinnability, mechanical and thermal behavior of PLA multifilament fibers. These multifilament fibers were cut into short fibers, carded to form fibrous web and later needle-punched together to form non-woven fabrics. The fire-related properties of these non-woven fabrics were tested by a cone calorimetry test. SEM analysis of multifilament fibers revealed reasonably uniform dispersion of the additives incorporated in the blends; however, some small agglomerates were also observed. The multifilament fibers containing 7 wt% of OS showed the highest thermal stability as confirmed by TG analysis alongside the residual mass% up to 14.1%. Cone calorimetry revealed that the interaction between OS and APP stimulated a remarkable decrease in PHRR and THR of the non-woven fabric samples. For instance, the lowest PHRR (216.1 kW m−2) was observed in the case of a fabric sample containing 7 wt% of OS which, is 51.8% lower than the PHRR of the pure PLA sample. Similarly, THR of the same sample (7 wt% OS) was 36.0 MJ m−2 which, is 29.5% lower than the THR of pure PLA sample. On the other hand, effective heat of combustion and total smoke production were significantly reduced in the case of samples containing higher wt% of OS. The morphology of char leftovers confirmed that compact char structure is mainly responsible for enhanced flame-retardant properties of the samples. To conclude this concisely, oxidized starch (OS) not only improved the spinnability of PLA/OS/APP blends compared to native starch (NS) but also the flame-retardant properties of non-wovens were very promising for use in industrial applications.