3.2. Optimization of Base Formulation and Effect of H-MWT as a Complement to the Carbon Source
To optimize the percentages of the main components in the formulation, it is necessary to understand the intumescence process, which depends on the thermal decomposition temperature of the components involved. Thus, the reaction process begins with the decomposition of the catalytic acid that subsequently reacts with the polyol, dehydrating it and forming a phosphate ester and H
2O. Subsequently, the decomposition of MEL occurs, which generates the evolution of NH
3 and CO
2 gases and the consequent foaming of the previously formed complex [
9]. In the case of using sodium polyphosphate (SPP) as a catalyst agent, it presents a decomposition temperature of approximately 593 °C [
22]. This is a higher value when compared to the MEL present in the pigment mixture of the tested base formulation (300 °C) [
23], which leads to the generation of CO
2 and ammonium vapors from the decomposition of MEL. The above occurs prior to the decomposition of the acid and to the reaction with the polyol, for which no carbonaceous layer or intumescence is generated.
When using MAP, decomposition occurs at temperatures below the decomposition temperature of SPP (210 ° C) [
24,
25], generating the phosphoric acid necessary for reaction with the carbon source (PER), which decomposes into short chains at 250 °C [
26,
27]. In this way, the type of polyphosphate used in the formulation indisputably marks the intumescent reaction, because of its ability to generate the main compound that acts as a catalyst for the dehydration reaction of the polyol and formation of the ester. Wang et al. studied the thermal decomposition of coatings developed with ammonium polyphosphate, which decomposes at temperatures of 290 °C [
28]. This allows it to react with PER at these temperature ranges, generating water and ammonium gas [
18]. Therefore, from the results obtained, it is observed that using SPP in the base formulation as an acid source (BF-SPP) does not generate a voluminous carbonaceous layer, unlike when using MAP (BF-MAP) (BF: base formulation), which generates superior intumescence properties in the coating when exposed to fire (see
Figure S1). For this reason, MAP was chosen as the acid catalyst in this work.
Table S1 presents the formulation results from varying the main component (MAP, PER, and MEL) percentages. Three molar ratios were used of phosphate:polyol (MAP:PER), 1:1, 2:1, and 4:1, to alter the reactivity of the polyol. In the first case, there is a reaction between a MAP molecule with only one OH group from PER. In the second case, there is a reaction with two MAP molecules, and in the last one, all OH from the PER are involved, forming a polymer complex [
29]. In the latter case, the 4:1 ratio contemplates the reaction between each OH group of the PER molecule with a MAP molecule. This relationship is unlikely due to the possible steric hindrance, leading to excess phosphate. Therefore, a greater probability of a complete reaction is expected when using molar ratios where there are excess phosphate groups in the medium. Thus, the MAP/PER percentage ratios with indices of 2.7, 1.9, and 1.5 presented better properties of fire resistance and intumescence. When altering the ratio of foaming agent (MEL) used, greater gas evolution was observed when the percentage was increased. The expansion area of the flame on the sample surface was smallest for BF2, and the most voluminous carbonaceous layer was seen with BF3 (see
Figure S2). Therefore, a greater amount of foaming agent and carbon source presented a better response to the flame, forming a carbon layer of greater thickness and less area affected by the flame. However, in BF4, the amount of acid present in the formulation was controlled by a MAP/PER ratio < 1.0, which led to a loss of carbonaceous foaming capacity due to excess polyol and a deficiency of phosphate groups in the medium that form the phosphate ester complex with OH groups (
Figure S2). In this way, MAP/PER ratios > 1, as presented in formulations BF1, BF2, and BF3, allow for a complete reaction and better intumescence properties.
Other factors had to be considered prior to creating the formulation with H-MWT, as previous wetting of the tannins was required to improve the rheological properties of the formulations due to their hydrophilicity. The water absorption results of the H-MWT and L-MWT extracts (
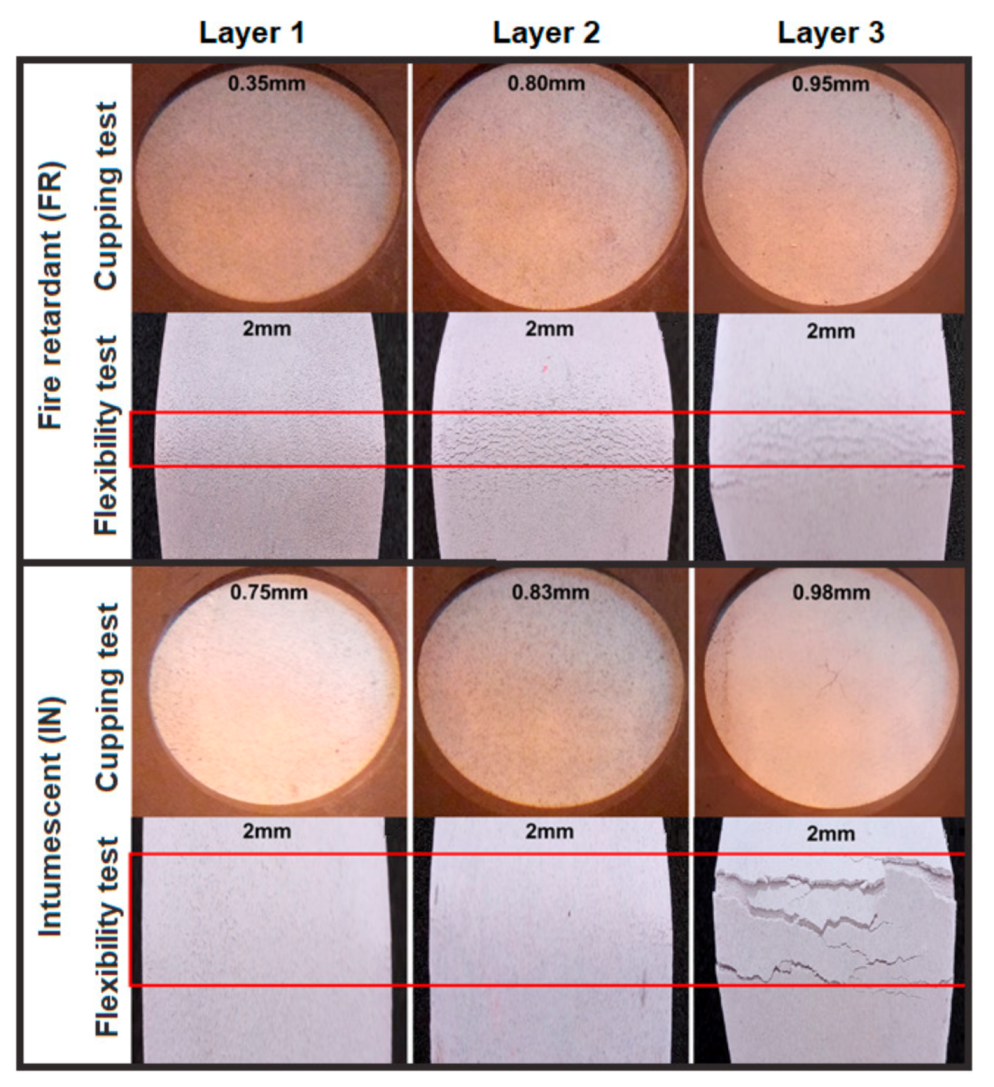
Figure S3) indicated that, by increasing the contact time of the particles with the aqueous medium, their swelling increased until reaching an absorption maximum of 9 and 31 times their weight in a period of 77 and 207 h for H-MWT and L-MWT, respectively. After this saturation time, the amount of water absorbed plateaued due to the complete swelling of the tannin particles. Since adsorption is a physical phenomenon influenced by the surface area of the particles, it was expected that the L-MWT would adsorb greater amounts water because they have shorter molecular chains compared to the H-MWT. Regarding these results, in the formulations with H-MWT, 9 L of water were required for each kg of tannins to moisten all the particles in the established period of time, while adjusting the viscosity of the formulation with rheological agents until obtaining values of 127 KU (10–15% dilution at 15 °C). After formulation, the coating was applied on opacity drawdown charts to observe the covering power and wet film thicknesses of 400 µm, as shown in
Figure 1. Those layers with fire-retardant properties presented greater opacity and covering power, with brown tones due to the high amount of tannins in the formulation, whereas the surfaces coated with the intumescent formulation presented lighter shades.
The intumescent and fire-retardant properties of the coatings developed with H-MWT were determined by the response of both wood and steel to flame. It was found that the FT1 and FT5 (FT: tannin formulation) coatings formulated according to
Table 2 showed intermediate properties between the fire retardant and intumescent coatings (
Figure 1). For the FT1 formulation, exposure to fire triggered the formation of a carbonaceous layer on the surface that acted as an active thermal barrier. For FT2, a spongy carbonaceous layer with an intumescent response was formed on the coated surface. The quantification of this response was carried out by measuring the mass loss of the wood substrates and comparing to the uncoated wood substrate mass loss of 14.7 ± 0.1%. The mass loss measurements showed better results for the FT1 and FT5 coating formulations in
Table 3, with values of 7.3 ± 0.5% and 9.3 ± 0.8%, respectively. It should be noted that FT1 did not show carbonaceous foam formation, but there was less mass loss. This is due to the fact that it contained a large amount of tannins, exceeding the optimal value of the phosphate/polyol ratio, providing a carbon source that acts as a thermal insulator and preventing the advance of the flame. Furthermore, when high amounts of H-MWT are added to the formulation, the phosphate/polyol ratio is not the only factor to take into account for the generation of intumescence, since it is necessary to use a greater amount of MEL. This is because the tannin molecule has available OH groups that react with the phosphoric acid produced by the decomposition of MAP, forming esters in the same way as PER. Additionally, these molecules in the formulation form complex structures after the reaction is activated by temperature, which turns into carbonaceous foam with a minimal amount of added MEL. According to studies by Braghiroli et al. [
30], three-dimensional structures can be formed after the reaction between tannins and ammonia derivatives, so the H-MWT used here also forms complex structures not only due to the presence of phosphates from the catalyst, but also due to the presence of ammonium ions.
On the other hand, the FT5 formulation also presented a low percentage of mass loss and generated foaming, an effect attributable to the lower amount of tannins, not unbalancing the MAP/PER ratio and favoring the growth of the carbonaceous layer. The results show that the best formulations were FT1 and FT5, which presented MAP/H-MWT ratios of 1.6 and 8, respectively. The coating obtained with FT1 had a high tannin content and exhibited fire-retardant behavior, whereas FT5 had intumescent properties due to its lower tannin amount, allowing the generation of a carbonaceous foam with greater volume when exposed to fire.