1. Introduction
Increasing the volume and efficiency of production during metal treatment is accompanied by the development of new and expanding the scope of existing types of tool complications during operation. This causes an increase in the tool’s temperature and the load in the contact area resulting in a reduction in product’s service life. Improving the stability of tools is now possible in the following way: improvement of design, development of new grades of alloys, and application of wear-resistant coatings.
It should be noted that from the economic standpoint of the technological capabilities of modern methods, it can be considered that the application of protective coatings is more promising than the development of new tool alloys.
Currently, there are some ways to obtain coatings on the working surfaces of steel and carbide tools [
1,
2,
3,
4,
5,
6,
7,
8]. After considering the peculiarities of the saturation processes, the following three methods can be distinguished: the method of physical deposition from the gas phase, the method of chemical deposition from the gas phase, and the method of chemical–thermal treatment, in which the formation of coatings is due to the diffusion processes of solid-phase, liquid, and gas-phase saturation.
Depending on the operational requirements, the gas-phase chemical deposition method is applied to the carbide plates with mechanical fastening multilayer coatings with the participation of TiN, TiC, and Al
2O
3 compounds [
4,
9,
10]. The arrangement of the layers of compounds in the coating determines the advantage of a particular property. It is necessary to note that this technology’s formation of nitride and carbide layers is due to the nitrogen–oxygen of the saturation medium. The obtained multilayer composition provides a complex of favorable properties: heat resistance, abrasive wear resistance, and resistance to indentation formation.
Using technologies of chemical–thermal treatment with a source of nitrogen–carbon to form, respectively, TiN and TiC layers will be the basis of tool steels. Nitrogen in steels or carbide may appear after nitriding. The results of studies on the formation of diffuser carbide and carbon–nitride coatings on tool steels and hard alloys are reported in [
9,
10]. Literature sources on the formation of TiN, TiC, and Al
2O
3 diffusive multilayer coatings on tool steels on hard alloys are unknown.
The coatings are also widely used in various technological fields. Particularly, the impact of diamond-like carbon coatings on the wear of the press joint components was studied in [
11]. In addition, control of structural and phase transformations in Ti materials, WC-Co substrate, and Ti-Al coatings was studied in [
12]. As a result, physicomechanical and wear properties were improved. Moreover, in [
13], the influence of coatings on fretting wear in a clamped joint for ensuring the maintenance and reliability of structures has been investigated.
The quality of TiN coating on steels can be estimated from [
14]. WC tungsten carbide and Co cobalt cutting tools have been the main components of hard alloy for decades. Concerns about cobalt toxicity have increased recently. This led to the search for new material for the binding phase [
15]. At the same time, requirements for improving the quality of tools and working productivity in metalworking are increasing. In [
14], it was shown that the deposition of TiN on iron causes strong corrosion of the substrate because of the formation of FeCl
x compounds in the gas phase. This is due to the exchange reactions between iron and TiCl
4 chloride. It is possible to assume the possibility of forming diffusion coatings without corrosion destruction of the steel surface during the reactions of titanium chloride disproportionation and elimination of reactions with the formation of iron chlorides [
10].
Multilayer multicomponent coatings with aluminum are particularly interesting [
16,
17,
18]. It can be assumed that in titanium aluminizing and chrome aluminizing, the coatings will combine the heat resistance of aluminum compounds with a hardness of carbides, titanium nitride, and chromium (TiN, TiC, Cr
7C
3, and Cr
23C
6) on the surface of carbon steel. The titanium alloys obtained by the powder method on the surface of carbon steels consist of a layer of TiC carbide outside the diffusion zone and a layer based on a solid Fe
α(Al) solution adjacent to the base. The latter’s formation is due to the diffusion of aluminum through the layer of titanium carbide, dissolution of aluminum in austenite, and time of the maximum concentration and austenite flow Fe
γ → Fe
α transformation. A similar structure is formed on medium-carbon steel during chrome aluminizing. Layers of Cr
7C
3 and Cr
23C
6 carbides are formed on the surface; layers of FeAl Fe
3Al compounds are located closer to the base. The disadvantage of titanium alloy and chrome-aluminized coatings on the investigated steels is the poor location of the layers in the coating. Under conditions of contact interaction, the composition is destroyed and does not realize the potential properties of hardness and heat resistance.
The functionality of the TiN, TiC, and Al
2O
3 compositions depends on the properties of the individual layers. It was shown in [
19] that the TiN layer is consistently deposited in elections with good quality at a wide temperature range, even at low temperatures. There are difficulties in the precipitation of TiC compounds in forming carbon hydrides. During chemical deposition from the gas phase, the latter can consistently exist in a wide temperature range and thus inhibit the formation of TiC carbide. In addition, in the TiC layers, there are significant compressive stresses, which lead to the delamination of the coatings. The detachment of the coatings from the base is quite clear.
The purpose of the work is to determine the effects of the composition of the reaction medium and the composition of the surface of AISI W1-7 steel after prenitriding, predeposited by the method of physical deposition from the TiN gas-phase layer and a preapplied layer of chromium carbides Cr7C3 and Cr23C6 on phase, chemical composition, structure, thickness, and properties of the obtained coatings on AISI W1-7 steel.
3. Results and Discussion
The results of studies of the phase and chemical compositions of the structure of the properties investigated in the work of coatings on AISI W1-7 steel are shown in
Table 3,
Figure 1.
The titanium coatings on AISI W1-7 steel consist, as already noted, of a layer of titanium carbide with a crystalline lattice period close to stoichiometric layers of intermetallic Fe
2Ti and FeTi, as well as a transition zone with high titanium content (
Table 3). The TiC layer is formed along with the entire thickness of equilibrium grains of 0.5–0.7 μm. TiC fracture is transcrystalline. Pores are not detected in the structure (
Figure 1).
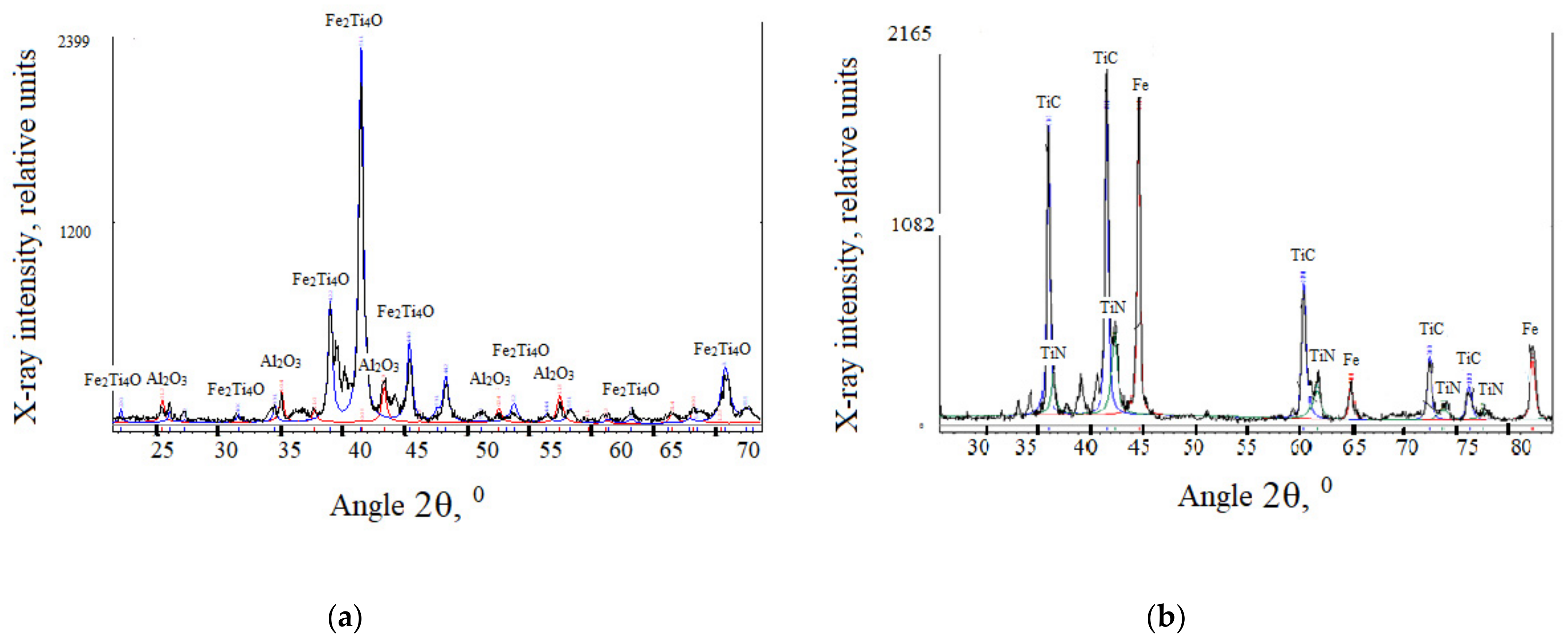
As a result of X-ray diffraction analysis of AISI W1-7 steel after titanium aluminizing with TiN-(PVD) layer, it was found that a small amount of alumina Al
2O
3 is formed on the surface (
Figure 2,
Table 3). The main phase of the surface layer of the protective coating is FeTi
4O. X-ray diffraction analysis of AISI W1-7 steel at a distance of 10 μm from the surface reveals the presence of two main phases: titanium carbide TiC and titanium nitride TiN.
The outer layer of intermetallic (σ-phase) and the transition zone is characteristic of chromium coatings. In this case, the layers of Cr
7C
3 and Cr
23C
6 carbides in the light microscope and raster microscope are revealed with a columnar structure (
Figure 1c,d).
Part of the samples, before titanium alloying, was applied by physically precipitating, from the gas phase, the titanium nitride TiN thickness of 4.5–5.0 μm. The TiN compound layer is yellow in a light microscope, corresponding to a stoichiometric nitride composition. The latter is confirmed by X-ray analysis, according to which the period of the FCC lattice of the compound TiN is 0.4245 nm (
Table 3).
Nitriding occurred in ammonia at 25–35% dissociation at 540 °C for 20 h. A diffusion layer is formed during the process of treatment on the surface of AISI W1-7 steel, which consists of a zone of compounds: these are the layers of Fe
4N, Fe
2-3N, phases, as well as the internal nitriding zone (
Table 3). The last in the pearlitic structure of AISI W1-7 steel is practically undetectable.
The titanium alloys are formed by the simultaneous saturation of titanium and aluminum in the powder mixture consisting of a solid solution of Fe
α(Al) at the boundary with the base. The layers of TiC and Fe
2(Al, Ti)
4O are formed outside. The Fe
α(Al) layer was formed during the diffusion of aluminum into austenite due to the polymorphic Feγ
→ Feα transformation. At a temperature of 1050 °C, the conversion of austenite into ferrite occurs at an aluminum content of 0.5–0.8% mass. Because of the low solubility of carbon in ferrite, carbon is displaced from the surface (
Table 3). TiC layers are formed by diffusion of the substrate carbon to the surface by adsorbed titanium. Over time, the growth rate of the TiC layer decreases. This effect allows forming compounds with iron on the outside. In the same way, Fe
2Ti
4O layers are formed in steels by diffusion from the Fe base.
After etching with a 3% alcohol solution of nitric acid, the Fe
α(Al) layer appears as a light zone with characteristic column crystals (
Figure 3). It should be noted that titanium is practically absent in the layer of Fe
α(Al) both at some distance from the TiC layer and directly beyond the boundary of TiC–Fe
α(Al) layers.
The absence of titanium is caused by its interaction on the surface with the carbon base and a layer based on TiC carbide. It is essential to mention that aluminum does not form carbides in the diffusion zone under acceptable conditions but diffuses through the TiC layer into the base where the Feα(Al) layer is formed. The aluminum concentration on the surface of AISI W1-7 titanium alloy steel and in Feα(Al) solid solution is 19.8 and 7.5–8.5% wt., respectively.
A common feature of multilayer coatings obtained by nitriding methods, physical precipitation from the gas phase, and diffusion chromium plating with titanium alloying is the obtained coating layer of TiC compounds (coating 6–8,
Table 3). TiC carbide in these coatings is formed from the carbon diffusion from the base and the interaction on the surface with titanium. The thickness of the TiC layers in coatings 6–8 (
Table 3) is less than the thickness of the TiC layer in coating 1 (
Table 3) because of the presence in the coatings 6–8 (
Table 3) of the intermediate zone with barrier functions; these are layers TiC in coatings 6 and 7 (
Table 3), and layers Cr7C3 andCr23C6 in coating 8 (
Table 3). TiN layer in coating 6 (
Table 3) begins to form in the initial stages of titanium alloying, and its barrier properties increase with thickness, which causes a slightly larger thickness of the TiC layer compared with the thickness of the TiC layer in coating 7 (
Table 3).
The difference between the phase composition of coatings No 6–8 (
Table 3) from titanium alloy No 5 (
Table 3) is the absence of a layer of Fe (Al). This fact is due to the presence of barrier TiN layers in coatings No 6 and 7 (
Table 3) and layers Cr
7C
3 and Cr
23C
6 in coating No 8 (
Table 3), which completely stop the penetration of aluminum into the steel substrate. The Feα(Al) layer was not detected in the coating data after titanium alloying. In the coatings No 6 and 7 (
Table 3), carbon and iron diffuse through the TiN and TiC layers from the base to the surface.
The analysis of the obtained data showed the effect of titanium aluminizing on the phase composition of the nitrided and chromium layers, which becomes almost unchanged after titanium alloying of the TiN layer. The layers of Fe4N, Fe3N, and Fe3O4 compounds disappear on nitrous steel, and the composition of the internal nitriding zone changes. In the chromium coating of Cr7C3, Cr23C6, and σ-phase carbides, the following changes occur: the total thickness of the carbide layer decreases from 15.0 to 10.0 μm mainly because of the thickness of the Cr7C3 layer; the σ-phase layer disappears.
These research results suggest that the carbon source for TiC formation is chromium carbide carbon, not the base carbon. Thus, the formation of the TiC layer occurs during the dissociation of chromium carbides. Thus, released carbon diffuses to the surface and participates in the formation of the TiC layer. Chromium and iron, together with the saturating elements of titanium and aluminum, form the compounds of the outer layers AlCrTi, Cr2Ti, and Ti3Al.
The crystalline lattice periods of TiC titanium carbide and TiN titanium nitride correspond to compounds close in composition to stoichiometric [
20,
21]. It is known [
21] that the period of TiN crystalline lattice increases with increasing nitrogen content in the homogeneity region, which changes at 1400 °C from TiN 0.6 to TiN 1.0. It was found that the lattice period of the TiN phase of the penetration obtained by physical deposition on AISI W1-7 steel practically did not change after the subsequent titanium alloying and was higher than the lattice period of TiN formed after the titanium alloying of nitrous steel. The latter may indicate a lack of nitrogen source power (nitrous steel). In this case, the lattice period of the TiC layer on nitrous steel (coating No 6,
Table 3) was a more considerable period of the lattice TiC on steel with a layer of TiN (coating No 7,
Table 3). The established dependence can be explained by the inhibition of carbon flux from the base to the surface by a layer of TiN and the absence of such a layer on the samples with coating No 6 (
Table 3).
Analysis of the results of studies of the phase composition showed that the layers of TiC and TiN in coatings 1, 5–8 (
Table 3) are formed by the extraction of titanium carbon and nitrogen to the surface with the formation of phases of penetration. TiN coatings acting as an aluminum barrier prevent the formation of a Feα(Al) layer. In addition, titanium aluminizing may lead to TiC, TiN by iron, and chromium layers, which diffuse to the surface through these layers.
So, the concentration of aluminum and iron in the layer of TiC in conventional titanium alloy (coating No 5,
Table 3) is aluminum at the level of 0.2% and has 2.4–4.6% wt. Simultaneously, the aluminum content on the outside of the coatings No 5–8 (
Table 3) is 18.5–19.8% of the mass, iron 20.0–38.0% wt. Titanium alloying nitrous steel with a layer of TiN increases the TiC layer of aluminum content to 0.5–0.6% wt. and reduces the iron content to 1.1% wt. The aluminum content in the TiN layer reaches 1.3–1.4% wt., and iron—2.3–2.4% wt.
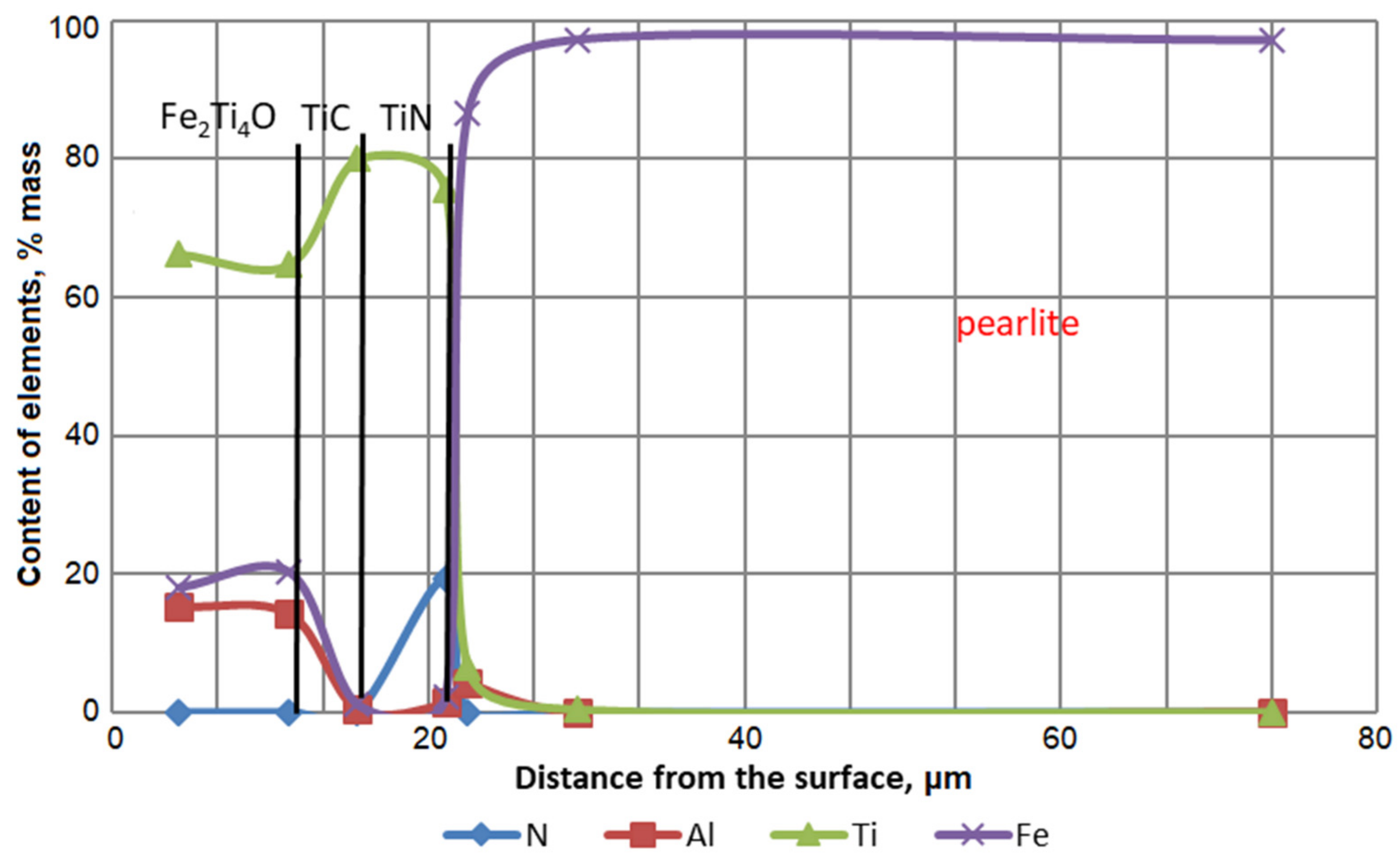
Figure 4 and
Figure 5 show the results of studies of the distribution of saturating elements and substrate elements by the thickness of titanium alloy coatings on AISI W1-7 nitrous steel and a distribution map. As already noted, the main difference between coatings No 5 and coatings 6–8 (
Table 3) is the lack of coatings 6–8 (
Table 3) Feα(Al) layer. The established feature of the structure of coatings 6–8 is the presence of barrier layers, which inhibit the penetration of aluminum into the substrate and prevent the formation of the Feα(Al) layer.
In the Fe
2(Ti, Al)4O layer, a significant amount of nitrogen was revealed. The nitrogen source can be in samples No 6 (
Table 3), nitrogen residues in steel after titanium, titanium nitride, and in samples No 7 (
Table 3)—titanium nitride. Titanium nitride nitrogen diffuses to the surface and dissolves in a layer of Fe
2(Ti, Al)
4O. The solubility of titanium nitride in Fe
2Ti
4O was shown in [
22], in which the (Ti, Zr)N compound was used as a barrier layer. Probably in equilibrium with the TiC layer, there must be a compound with certain nitrogen content. Thus, the TiN layer is dissociated during the chemical–thermal treatment, followed by the diffusion of nitrogen and titanium into the Fe
2Ti
4O layer.
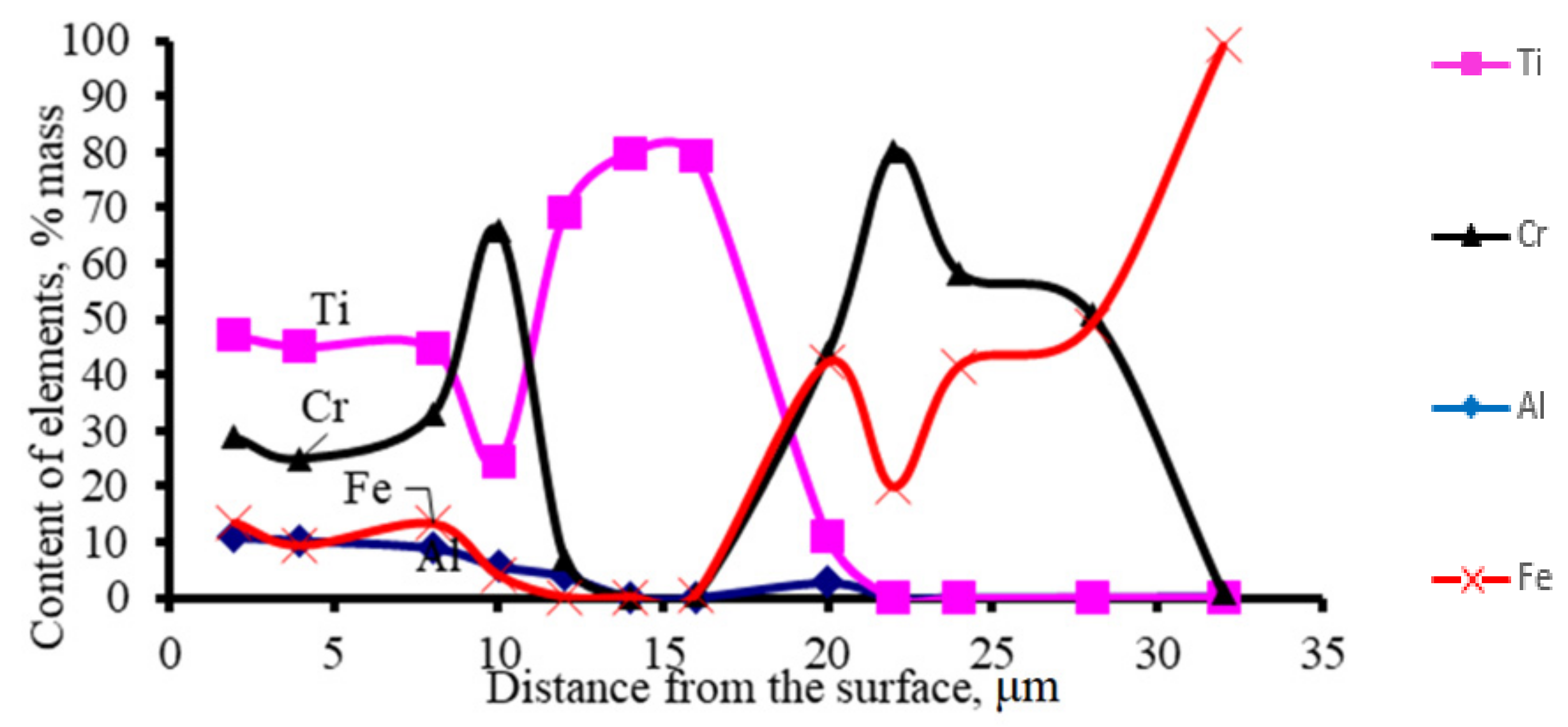
Figure 6 shows the results of the studies of the distribution of elements by the thickness of coating No. 5 (
Table 3) and coatings obtained by diffusion titanium aluminizing of chromium steel. According to the results obtained in coating No 8 (
Table 3), titanium alloying is the dissociation of chromium carbides, which is confirmed by reducing the thickness of their layers. The carbon released from chromium carbides interacts with titanium to form TC carbide, and chromium interacts with the metal. The thickness of the TiC layer in coating No 8 (
Table 3) is 7.0 μm.
Metallographic analysis of the obtained coatings showed a significant difference between complex coatings with TiN nitride layers and chromium carbides (coatings 6–8,
Table 3). First of all, this concerns the absence and complex coatings of the Feα(Al) layer. Directly to the base in coatings 0 and 7 (
Table 3) is the adjacent layer of TiN or a layer of chromium carbides in coating No 8 (
Table 3). Outside there is a layer of titanium carbide TiC and a layer of Fe
2(Ti, Al)
4O in coatings No 6 and 7 (
Table 3), or a layer of TiC and layers of intermetallics in coating No 8 (
Table 3).
It should be noted that the TiN nitride layer is well identified in the structure during analysis and light microscope because of its yellow-gold color.
The microhardness of individual component coatings varies over a wide range: from the microhardness of TiC carbide layers—30.3–37.8 GPa, TiN nitride—19.8–22.0 GPa, of Cr
7C
3 carbides and Cr
23C
6—16.5–17.2 GPa, to microhardness of intermetallics, oxides—6.1–14.0 GPa. The microhardness of the outer layers in the TiN coatings (No 6, 7,
Table 3) was higher than the microhardness of the similar layers in coatings No 5. This fact is due, as already mentioned, to the alloying of these zones in coatings No 6 and 7 (
Table 3) with nitrogen.
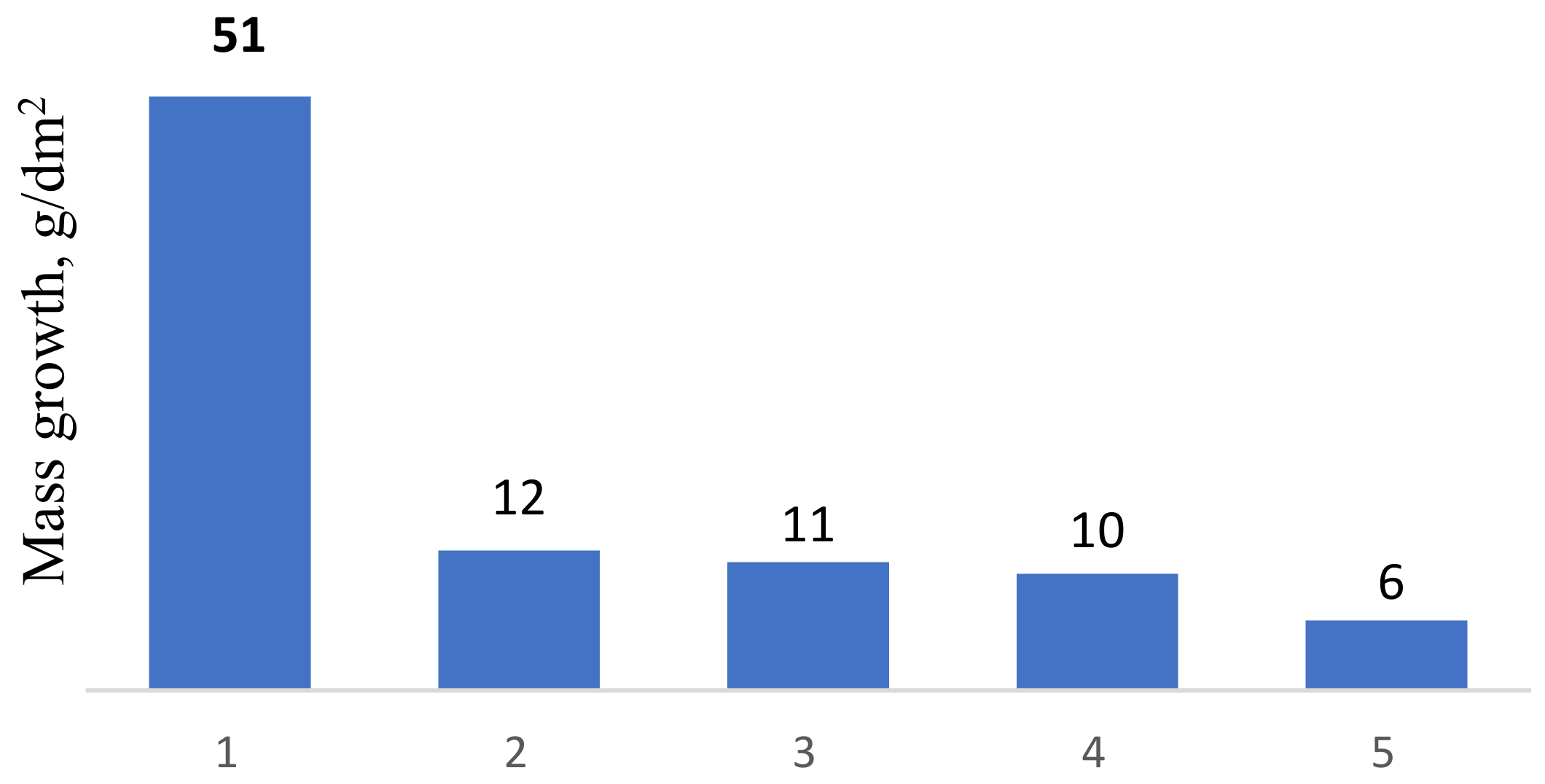
Analysis of test results of coatings investigated in the work showed that the heat resistance of AISI W1-7 steel with coatings increased 4.2–8.5 times (
Figure 7). The maximum heat resistance was shown by coating No 8 (
Table 3) obtained by titanium alloying of chromium steel. Analysis of the literature sources [
23,
24] shows that at elevated temperatures under continuous oxidation of Ti–Al–Cr alloys on the surface, an oxide film (Al, Cr)
2O
3 is formed at high oxidation resistance. The properties of coatings with the participation of titanium on aluminum at high temperatures, under conditions of friction of the sliding, depend on the presence on the outside of the layer of compound Al
2O
3 [
23,
24]. Increasing the heat resistance of coatings with titanium and aluminum is important. The heat resistance of titanium and aluminum alloys depends on the composition and increases with increasing aluminum content up to 60–70% wt. In this case, the binary Ti–Al alloy with significant aluminum content becomes brittle, limiting its practical use. This solution is made possible by using chromium as an alloying element. Ti–Al–Cr alloys consisting of the Laves Ti(Al, Cr)
2 and θ-phase (Ti
0,25A
l0,
67Cr
0,08) are characterized by high heat resistance and mechanical properties [
23,
24]. First of all, it is an alloy containing 25–35% of the Laves phase and 70–75% having the θ-phase.
The oxidation of compounds involving titanium and aluminum occurs by forming an oxidation zone, the phase composition of which depends on the content of aluminum in the coating and temperature [
25,
26,
27]. In Tix Alx N films [
26], the aluminum content was changed from 25.0 to 67.0% wt. The heat resistance, in this case, increased. The oxidation of the TiAlN film aggregates at 700 °C and below is accompanied by the heterogeneous zone of the oxides Al
2O
3 and TiO
2. At temperatures above 800 °C, the heat resistance increases, and a two-layer oxide structure is formed from the Al
2O
3 compounds—the outer layer and the inner layer TiO
2. Al
2O
3 layer performs the main protective functions.
The aluminum stock determines the heat resistance of the coating material. The higher the aluminum content, the longer the coating service life. When tested, the aluminum part of the coating forms an Al
2O
3 oxide film. The part diffuses into the substrate. In addition, it is possible to diffuse the base elements in the coating, which also reduces the concentration of aluminum. The results show that the TiN barrier layer almost completely blocks aluminum diffusion to the base. It was shown in [
27] that the amount of aluminum transferred from the coating to the substrate is the same as the coating in Al
2O
3 oxide.
Analysis of the results of studies of AISI W1-7 steel wear resistance with multicomponent coatings in the conditions of sliding friction without lubrication for the accepted sliding speed (10 m/s) and loadings (5.0, 10.0, and 15.0 N) showed the feasibility of using developed coatings for tool protection (
Figure 8). Titanium alloy coatings showed the highest wear resistance with preapplied layers of chromium carbides and titanium nitride.
Sliding friction on the contact surfaces of solids at full pressure occurs in small areas of the so-called contact spots [
28,
29]. The total area of contact spots corresponds to the actual contact area, which is a small fraction of the nominal one. In the friction process, the shape and position of the contact spots are constantly changing. Studies have established [
29] that the average diameter of contact spots is 6.0–30.0 μm at a distance between them of 80.0–120.0 μm.
Notably, individual properties and characteristics of coatings have been positively affected. The actual working conditions of the cutting tool are complex, while some physical and chemical processes occur, which lead to the destruction of the product. The paper discusses some processes related to the impact on the wear resistance of barrier coatings based on TiN titanium nitride.
The temperature in the area of contact interaction during cutting can reach significant values, accompanied by the interaction of the coating elements and the substrate with the environment. Formed oxide films, which are well bonded to the substrate and have anti-friction properties, increase the wear resistance of the tools. The formation of oxides of the desired composition is possible only on an alloy of a particular composition. It is known that the wear resistance of multicomponent coatings is positively influenced by the layers of Al2O3 aluminum oxide and TiO2 titanium oxide. A layer of Al2O3 alumina was fixed on the surface of the coating after chemical–thermal treatment partially formed during the friction process. A layer of TiO2 oxide is formed in testing at increased temperatures. It can be assumed that the presence of a barrier layer based on TiN titanium nitride stabilizes the chemical composition by inhibiting the diffusion drainage at the base of the coating elements, primarily aluminum. In addition, the barrier layers inhibit the diffusion of the coating elements into the treated alloy and the processed alloy (counter) elements into the coating.
The properties of the coatings and their structure are formed by the friction layers of aluminum and titanium oxides, causing a decrease in the friction coefficients of AISI W1-7 steel with coatings in contact with the rider and thus contributing to the increase in wear resistance. The coefficient of friction was determined in the conditions of the formed indentation wear, which corresponds to the actual operating conditions of the cutting tools. An analysis of the literature [
30,
31] showed the effect of coatings on the coefficient of friction during tests under sliding friction conditions without lubrication according to the scheme of the pin-disk. It is established that the coating of TiN titanium nitride in friction with steel contributes to reducing the coefficient of friction compared with the coefficient of friction of steel pair by 2.1 times. In the same work [
31], TiN coatings reduce the friction coefficient to a small extent, and even the chromium carbide coatings increase.
Under the considered friction conditions, an indentation with a depth more significant than the thickness of the coating is formed in the contact zone. Thus, the indentation edges will consist of layers of TiN, TiC, intermetallics, and oxides Al2O3 and TiO2. The central zone of the indentation corresponds to the base material with possible inclusions of the destroyed coating—TiN and TiC particles, which corresponds to the Sharpey structure. The proposed complex coatings, by their structure and properties, can enhance the functional properties of AISI W1-7 tool steel.