Corrosion Protection Efficacy of the Electrodeposit of Poly (N-Methyl Pyrrole-Tween20/3-Methylthiophene) Coatings on Carbon Steel in Acid Medium
Abstract
:1. Introduction
2. Experimental
Materials and Methods
3. Results and Discussion
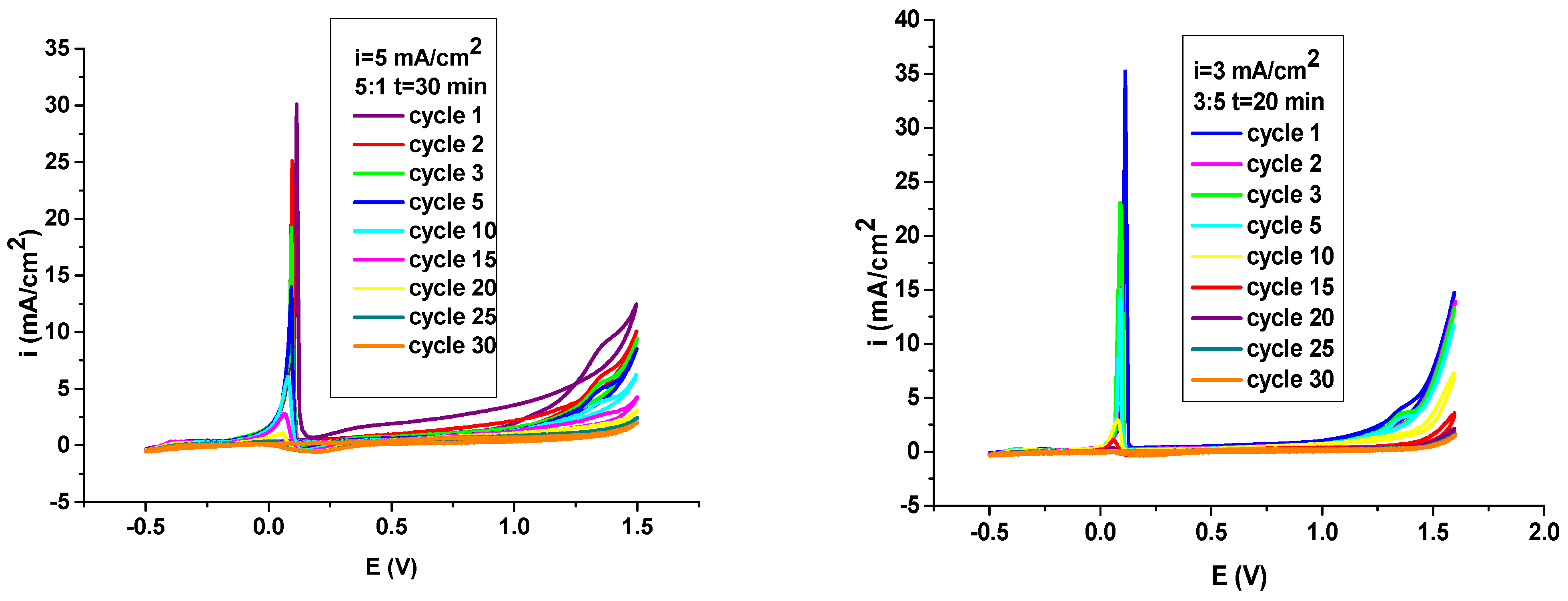
3.1. Electrodeposition PNMPY-TW20/P3MT Coating
3.2. Electrochemical Characterization of PNMPY-Tw20/P3MT Composite Coating
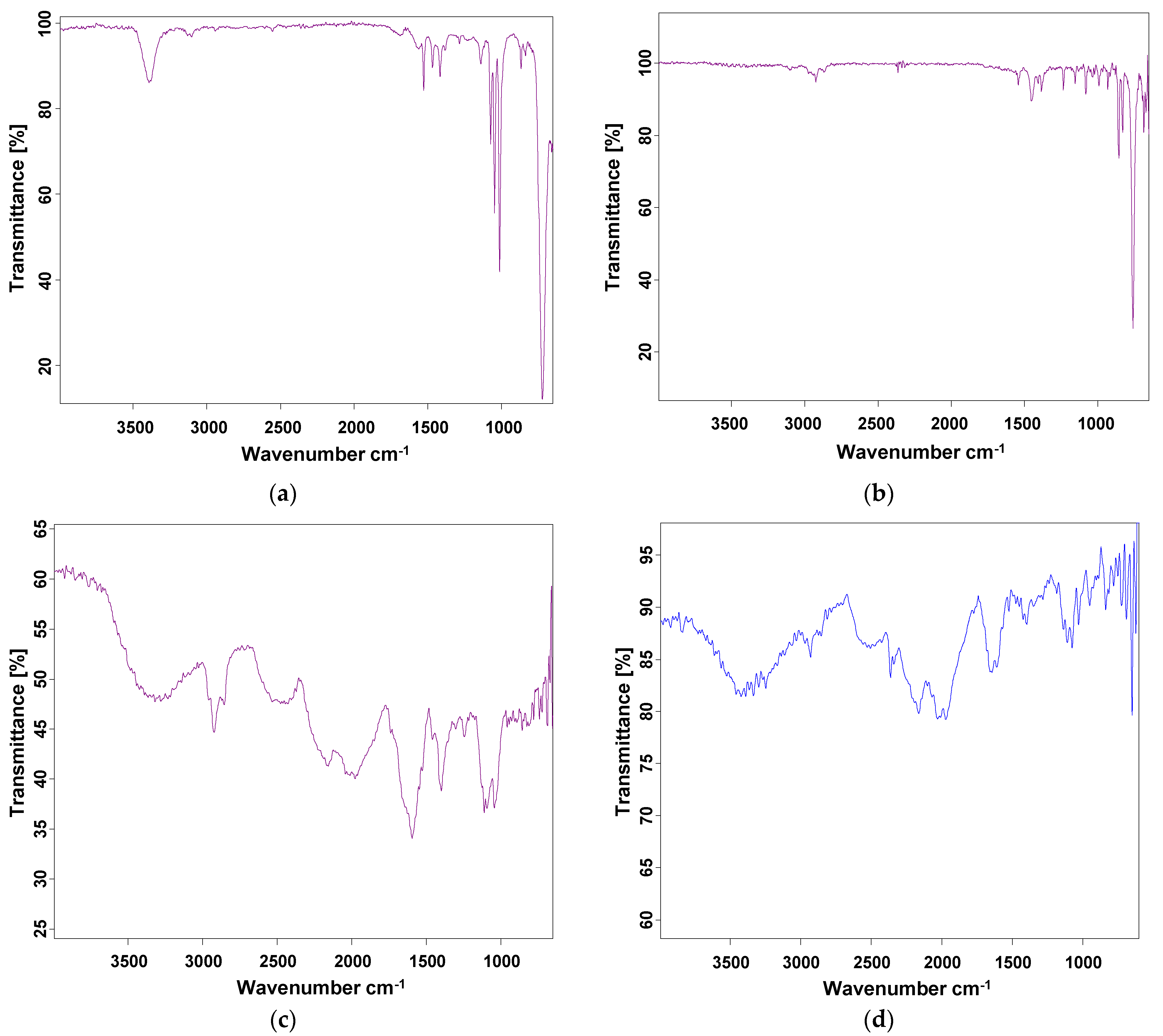
3.3. FT-IR Studies
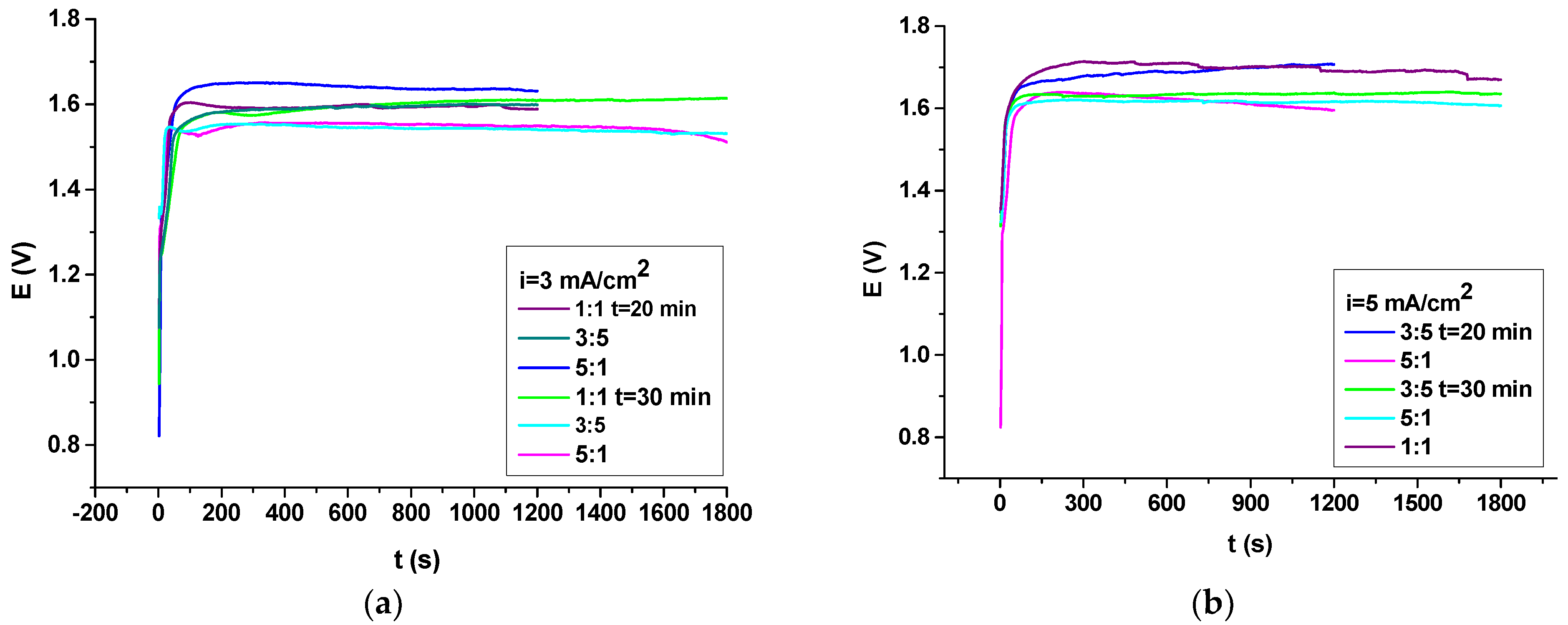
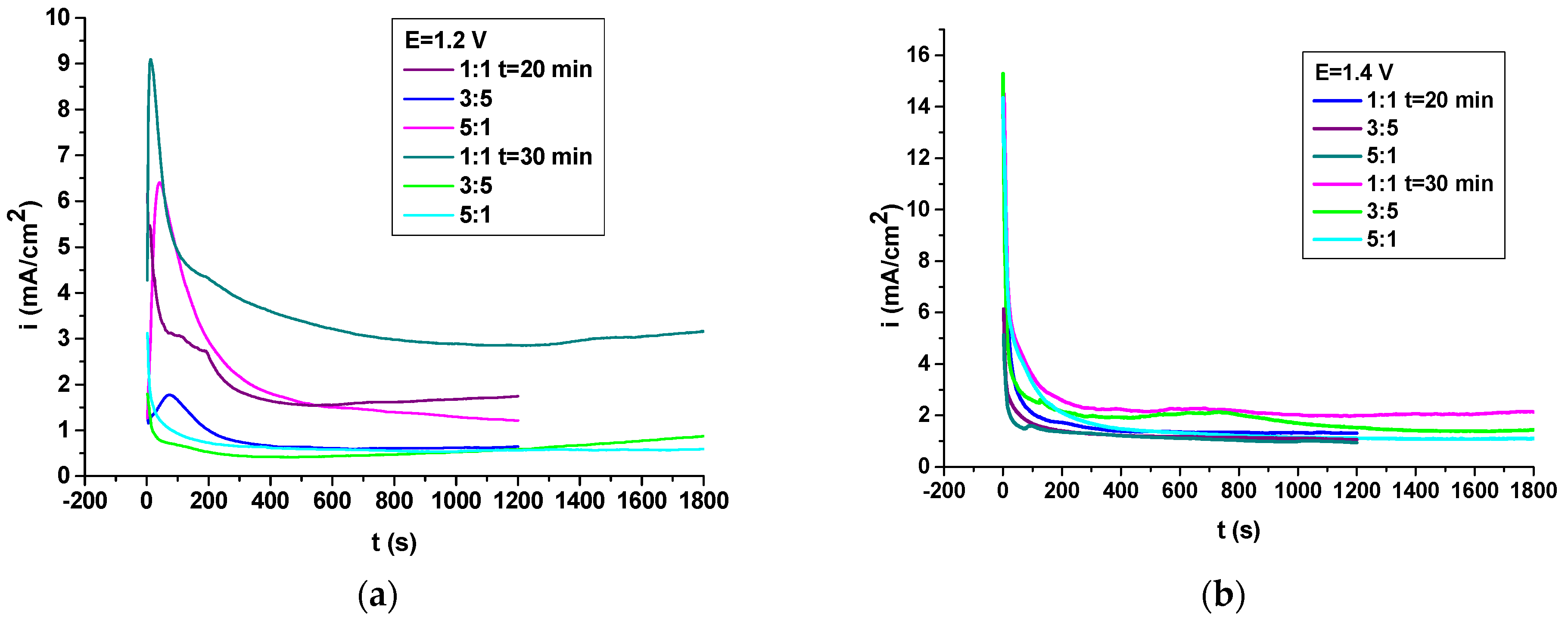
3.4. Electrochemical Evaluations
3.4.1. Potentiodynamic Polarization Procedure
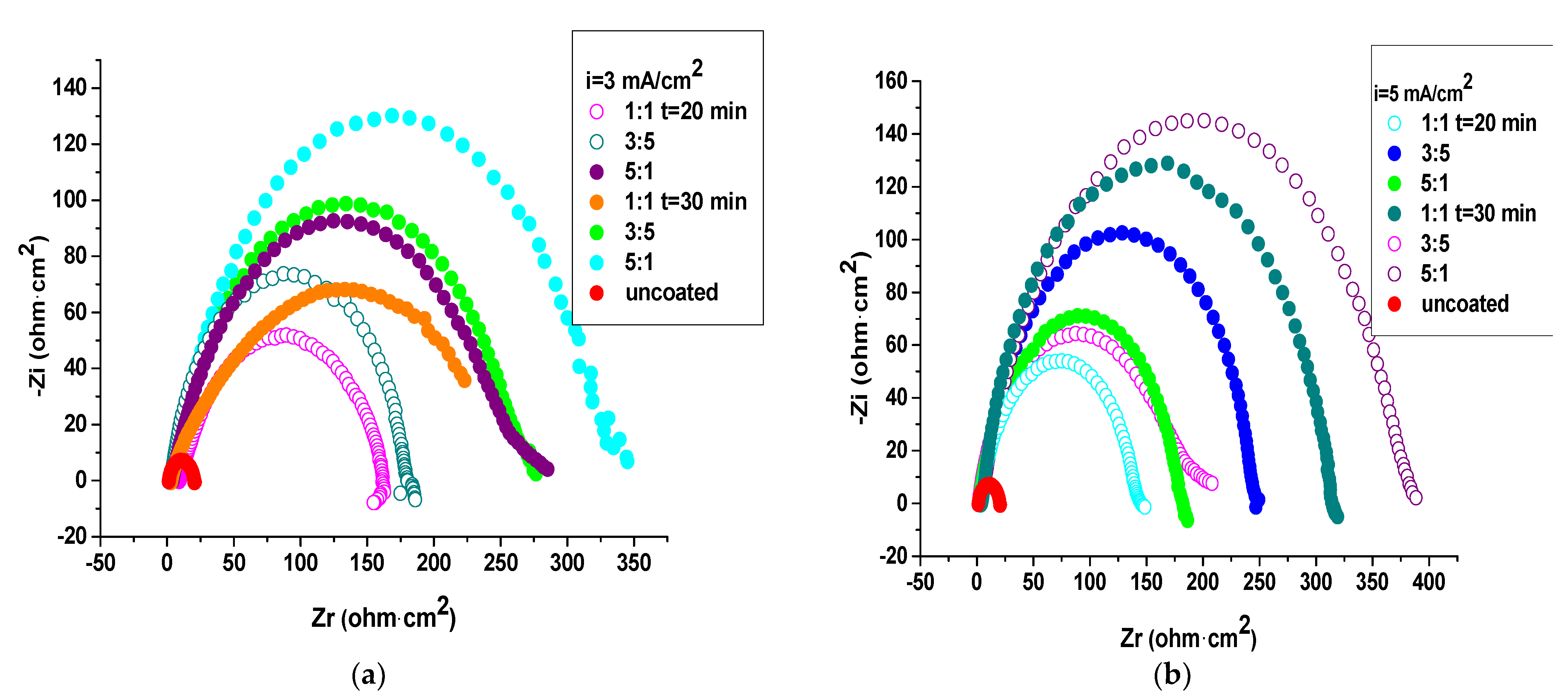
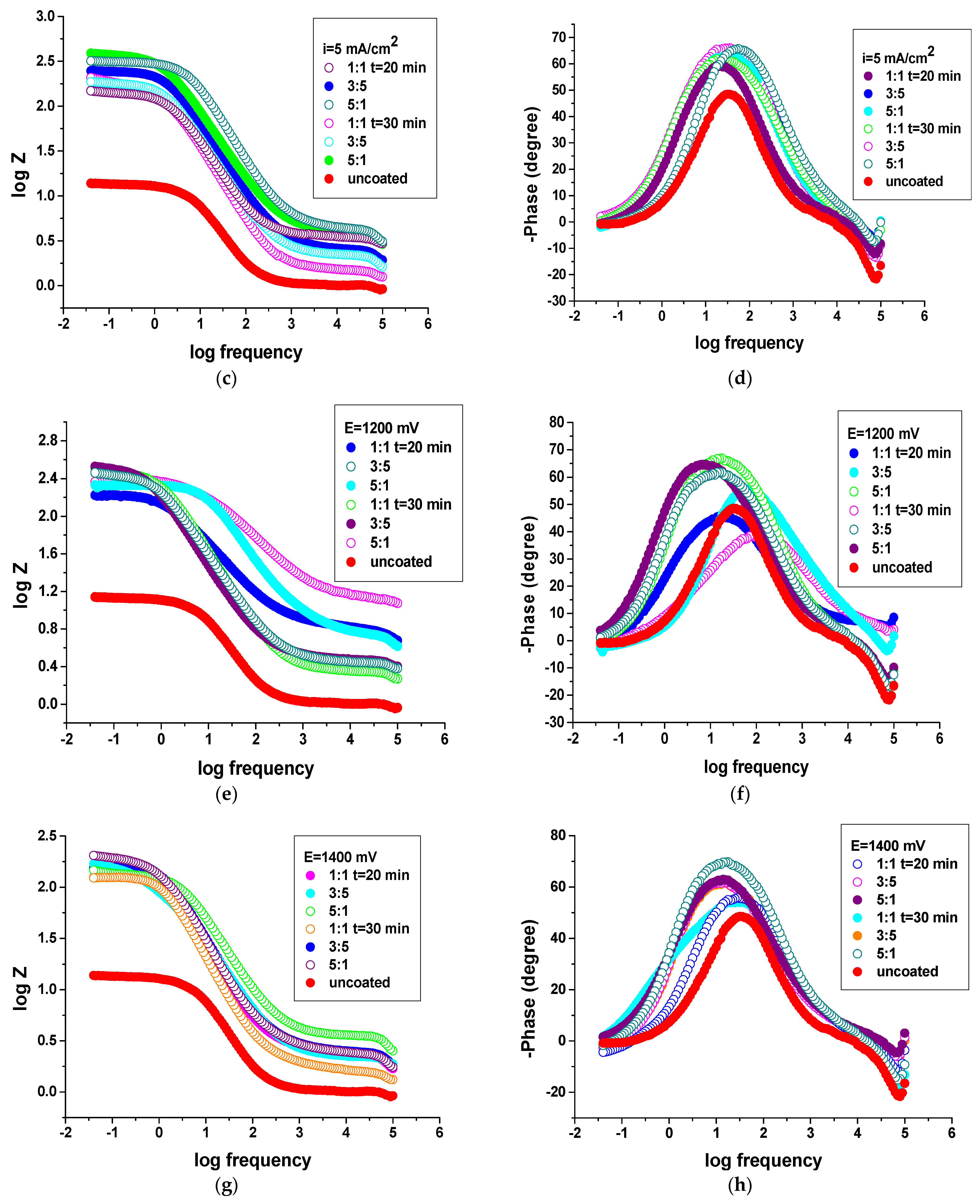
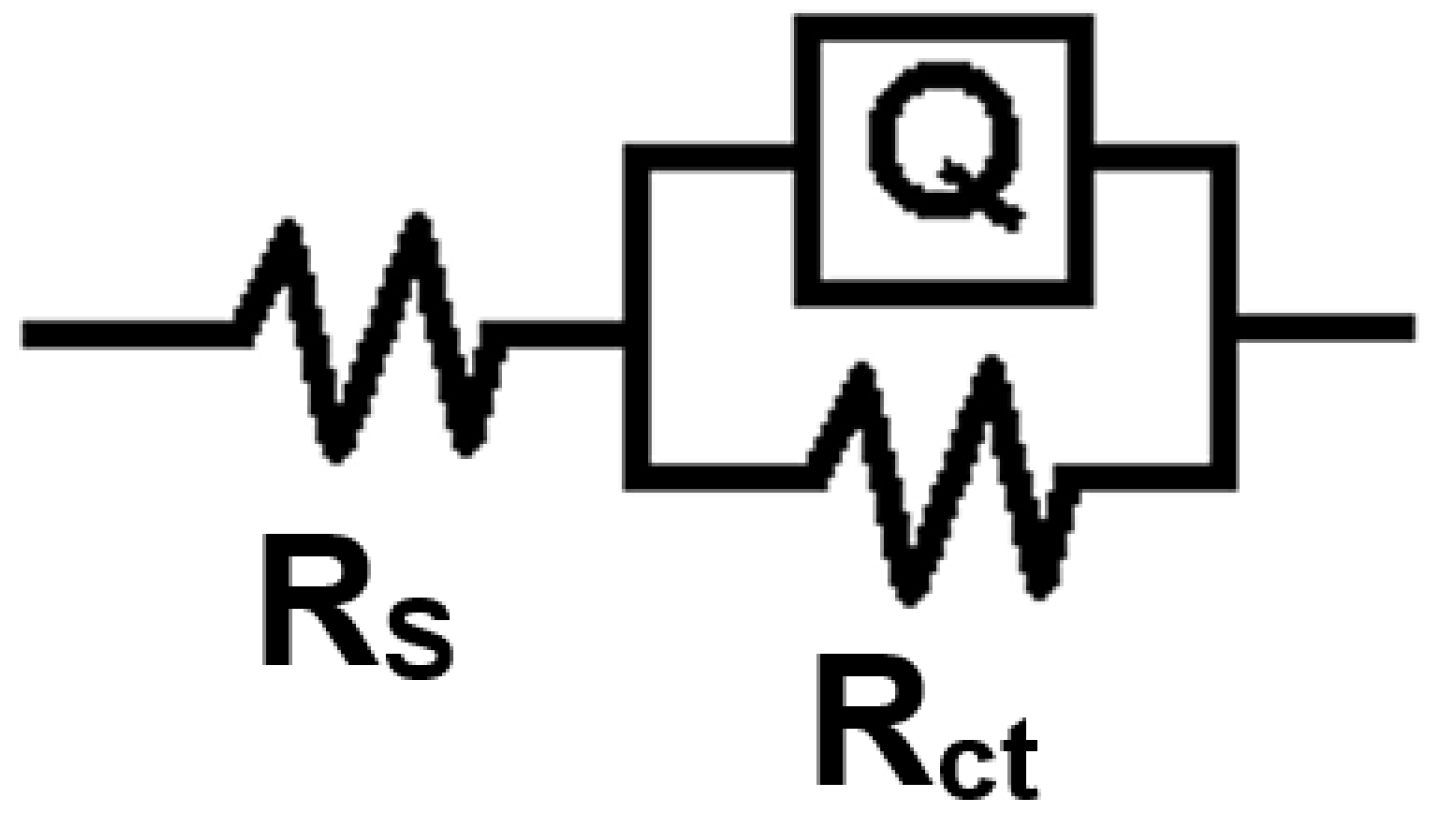
3.4.2. Electrochemical Impedance Spectroscopy (EIS) Studies
3.5. SEM Studies
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Abdel Hamid, Z.; Hasan Gomaa, M.; Abdel Rehim, S.S.; Abdel Hamid, M.; Ibrahim, A. Synthesis and Characterization of 400 Nanostructured Polyaniline Thin Films with Super hydrophobic Properties. Coatings 2019, 9, 748. [Google Scholar] [CrossRef] [Green Version]
- Zhai, Y.; Pan, K.; Zhang, E. Anti-Corrosive Coating of Carbon-Steel Assisted by Polymer-Camphor sulfonic Acid Embedded within Graphene. Coatings 2020, 10, 879. [Google Scholar] [CrossRef]
- Guptad, D.K.; Neupane, S.; Singh, S.; Karki, N.; Yadav, A.P. The effect of electrolytes on the coating of polyaniline on mild steel by electrochemical methods and its corrosion behavior. Prog. Org. Coat. 2021, 152, 106127. [Google Scholar] [CrossRef]
- Branzoi, F.; Branzoi, V. Enzymatic electrode obtained by immobilizing of urease into a nanocomposite film based on conducting polymers and different additives. Int. J. Polym. Mater. Polym. Biomater. 2014, 63, 549–556. [Google Scholar] [CrossRef]
- Silva, R.S.; Meneguzzi, Á. Passivation of Carbon Steel Using Intelligent Epoxy Paint. Coatings 2020, 10, 452. [Google Scholar] [CrossRef]
- Pruna, A.; Brânzoi, F. Electrochemical activity and microscopy of electrosynthesised poly (o-phenylenediamine) nanotubes. J. Polym. Res. 2012, 19, 9879. [Google Scholar] [CrossRef]
- Gallegos-Melgar, A.; Serna, S.A.; Lázaro, I.; Gutiérrez-Castañeda, E.-J.; Mercado-Lemus, V.H.; Arcos-Gutierrez, H.; Hernández-Hernández, M.; Porcayo-Calderón, J.; Mayen, J.; Monroy, M.D.A. Potentiodynamic Polarization Performance of a Novel Composite Coating System of Al2O3/Chitosan-Sodium Alginate, Applied on an Aluminum AA6063 Alloy for Protection in a Chloride Ions Environment. Coatings 2020, 10, 45. [Google Scholar] [CrossRef] [Green Version]
- Tavandashti, N.P.; Almas, S.M.; Esmaeilzadeh, E. Corrosion protection performance of epoxy coating containing alumina/PANI nanoparticles doped with cerium nitrate inhibitor on Al-2024 substrates. Prog. Org. Coat. 2021, 152, 106133. [Google Scholar] [CrossRef]
- Liu, A.; Tian, H.; Li, S.; Ju, X.; Yang, H.; Sun, Y.; Wang, L.; Li, W. Bioinspired layered hybrid coatings with greatly enhanced barrier effect and active corrosion protection performance. Prog. Org. Coat. 2021, 152, 106131. [Google Scholar] [CrossRef]
- Branzoi, F.; Băran, A.; Petrescu, S. Evaluation of Corrosion Protection Performance of New Polymer Composite Coatings on Carbon Steel in Acid Medium by Electrodeposition Methods. Coatings 2021, 11, 903. [Google Scholar] [CrossRef]
- Ji, S.; Gui, H.; Guan, G.; Zhou, M.; Guo, Q.; Tan, M.Y.J. Molecular design and copolymerization to enhance the anti-corrosion performance of waterborne acrylic coatings. Prog. Org. Coat. 2021, 153, 106140. [Google Scholar] [CrossRef]
- Branzoi, F.; Băran, A. The inhibition effect of some organic compounds on corrosion of brass and carbon steel in aggressive medium. Int. J. Electrochem. Sci. 2019, 14, 2780–2803. [Google Scholar] [CrossRef]
- Pekmez, N.O.; Abaci, E.; Cinkilli, K.; Yagan, A. Polybithiophene and its bilayers with polyaniline coatings on stainless steel by electropolymerization in aqueous medium. Prog. Org. Coat. 2009, 65, 462–468. [Google Scholar] [CrossRef]
- Branzoi, F.; Branzoi, V. Investigation of Protective Effect of Polymeric Film Coatings on Carbon Steel in Aggressive Solutions. Int. J. Electrochem. Sci. 2016, 11, 6564–6579. [Google Scholar] [CrossRef]
- Yagan, A.; Pekmez, N.Ö.; Yıldız, A. Poly (N-methylaniline) coatings on stainless steel by electropolymerization. Corros. Sci. 2007, 49, 2905–2919. [Google Scholar] [CrossRef]
- Zeybek, B.; Aksun, E. Electrodeposition of poly(N-methylpyrrole) on stainless steel in the presence of sodium dodecylsulfate and its corrosion performance. Prog. Org. Coat. 2015, 81, 1–10. [Google Scholar] [CrossRef]
- Pekmeza, N.Ö.; Cinkillia, K.; Zeybekba, B. The electrochemical copolymerization of pyrrole and bithiophene on stainless steel in the presence of SDS in aqueous medium and its anticorrosive performance. Prog. Org. Coat. 2014, 77, 1277–1287. [Google Scholar] [CrossRef]
- Redondo, M.I.; de la Blanca, E.S.; García, M.V.; González-Tejera, M.J. Poly(N-methylpyrrole) Electrodeposited on copper: Corrosion protection properties. Prog. Org. Coat. 2009, 65, 386–391. [Google Scholar] [CrossRef]
- Su, W.; Iroh, J.O. Electrodeposition mechanism, adhesion and corrosion performance of polypyrrole and poly (N-methylpyrrole) coatings on steel substrates. Synth. Met. 2000, 114, 225–234. [Google Scholar] [CrossRef]
- Çakmakci, I.; Duran, B.; Duran, M.; Bereket, G. Experimental and theoretical studies on protective properties of poly (pyrrole-co-n-methyl pyrrole) coatings on copper in chloride media. Corros. Sci. 2013, 69, 252–261. [Google Scholar] [CrossRef]
- Ren, S.; Barkey, D. Electrochemically Prepared Poly (3-methylthiophene) Films for Passivation of 430 Stainless Steel. J. Electrochem. Soc. 1992, 139, 1021. [Google Scholar] [CrossRef]
- Duran, B.; Bereket, G. Cyclic Voltammetric Synthesis of Poly(N-methyl pyrrole) on Copper and Effects of Polymerization Parameters on Corrosion Performance. Ind. Eng. Chem. Res. 2012, 51, 5246–5255. [Google Scholar] [CrossRef]
- Rui, M.; Zhu, A. The synthesis and corrosion protection mechanisms of PANI/CNT nanocomposite doped with organic phosphoric acid. Prog. Org. Coat. 2021, 153, 106134. [Google Scholar] [CrossRef]
- Martí, M.; Armelin, E.; Iribarren, J.I.; Alemán, C. Soluble polythiophenes as anticorrosive additives for marine epoxy paints. Mater. Corros. 2015, 66, 23–30. [Google Scholar]
- Gopi, D.; Saraswathy, R.; Kavitha, L.; Kim, D.-K. Electrochemical synthesis of poly(indole-co-thiophene) on low-nickel stainless steel and its anticorrosive performance in 0.5 mol L−1H2SO4. Polym. Int. 2013, 63, 280–289. [Google Scholar] [CrossRef]
- Sazou, D.; Kourouzidou, M.; Pavlidou, E. Potentiodynamic and potentiostatic deposition of polyaniline on stainless steel: Electrochemical and structural studies for a potential application to corrosion control. Electrochim. Acta 2007, 52, 4385–4397. [Google Scholar] [CrossRef]
- Branzoi, F.; Brânzoi, V.; Musina, A. Fabrication and characterisation of conducting composite films based on conducting polymers and functionalised carbon nanotubes. Surf. Interface Anal. 2012, 44, 1076–1081. [Google Scholar] [CrossRef]
- Zhou, W.; Wu, K.; Zhang, K.; Wang, Z.; Liu, Z.; Hu, S.; Fang, Y.; He, C. Studies on Corrosion Behaviors of Q235 Steel Coated by the Polypyrrole Films Doped with different dopants. Int. J. Electrochem. Sci. 2020, 15, 2594–2603. [Google Scholar] [CrossRef]
- Branzoi, F.; Branzoi, V.; Musina, A. Coatings based on conducting polymers and functionalized carbon nanotubes obtained by electropolymerization. Prog. Org. Coat. 2013, 76, 632–638. [Google Scholar] [CrossRef]
- Fuseini, M.; Zaghloul, M.M.Y. Statistical and qualitative analyses of the kinetic models using electrophoretic deposition of polyaniline. J. Ind. Eng. Chem. 2022. [Google Scholar] [CrossRef]
- Zaghloul, M.Y.; Zaghloul, M.M.Y.; Zaghloul, M.M.Y. Influence of Stress Level and Fibre Volume Fraction on Fatigue Performance of Glass Fibre-Reinforced Polyester Composites. Polymers 2022, 14, 2662. [Google Scholar] [CrossRef]
- Fuseini, M.; Zaghloul, M.M.Y.; Elkady, M.F.; El-Shazly, A.H. Evaluation of synthesized polyaniline nanofibres as corrosion protection film coating on copper substrate by electrophoretic deposition. J. Mater. Sci. 2022, 57, 6085–6101. [Google Scholar] [CrossRef]
- Zhang, J.; Zhu, A. Study on the synthesis of PANI/CNT nanocomposite and its anticorrosion mechanism in waterborne Coatings. Prog. Org. Coat. 2021, 159, 106447. [Google Scholar] [CrossRef]
- Pan, Y.; Pu, D.L.; Li, Y.Q.; Zheng, Q.H. Origin of the antioxidation mechanism of RuAl(1 1 0) surface from first-principles calculations. Mater. Sci. Eng. B 2020, 259, 114580. [Google Scholar] [CrossRef]
- Pan, Y.; Wen, M. Insight into the oxidation mechanism of Nb3Si(111) surface: First-principles calculations. Mater. Res. Bull. 2018, 107, 484–491. [Google Scholar] [CrossRef]
- Pan, Y.; Pu, D.; Jia, Y. Adjusting the correlation between the oxidation resistance and mechanical properties of Pt-based thermal barrier coating. Vacuum 2020, 172, 109067. [Google Scholar] [CrossRef]
- Branzoi, F.; Băran, A.; Ludmila, A.; Alexandrescu, E. The inhibition action of some organic polymers on the corrosion carbon steel in acidic media. Chem. Pap. 2020, 74, 4315–4335. [Google Scholar] [CrossRef]
- Menkuer, M.; Ozkazanc, H. Anticorrosive polypyrrole/zirconium-oxide composite film prepared in oxalic acid and dodecylbenzene sulfonic acid mix electrolyte. Prog. Org. Coat. 2020, 147, 105815. [Google Scholar] [CrossRef]
- Branzoi, F.; Branzoi, V. Investigation of some nonionic surfactants as corrosion inhibitors for carbon steel in sulfuric acid medium. Int. J. Electrochem. Sci. 2017, 12, 7638–7658. [Google Scholar] [CrossRef]
- Zeybek, B.; Pekmez, N.O.; Kilic, E. Electrochemical synthesis of bilayer coatings of poly(N-methylaniline) and polypyrrole on mild steel and their corrosion protection performances. Electrochim. Acta 2011, 56, 9277–9286. [Google Scholar] [CrossRef]
- González, M.B.; Saidman, S.B. Electrodeposition of polypyrrole on 316L stainless steel for corrosion prevention. Corros. Sci. 2011, 53, 276–282. [Google Scholar] [CrossRef]
- Herrasti, P.; Patil, P. Corrosion protective poly (o-ethoxyaniline) coatings on copper. Electrochim. Acta 2007, 53, 927–933. [Google Scholar]
- Duran, B.; Turhan, M.C.; Bereket, G.; Sarac, A.S. Electropolymerization, characterization and corrosion performance of poly(N-ethylaniline) on copper. Electrochim. Acta 2009, 55, 104–112. [Google Scholar] [CrossRef]
- Yagan, A.; Pekmez, O.N.; Yildiz, A. Poly(N-ethylaniline) coatings on 304 stainless steel for corrosion protection in aqueous HCl and NaCl solutions. Electrochim. Acta 2008, 53, 2474–2482. [Google Scholar] [CrossRef]
- Shi, S.; Zhao, Y.; Zhang, Z.; Yu, L. Corrosion protection of a novel SiO2/PANI coating for Q235 carbon steel. Prog. Org. Coat. 2019, 132, 227–234. [Google Scholar] [CrossRef]
- Yagan, A.; Pekmez, O.N.; Yildiz, A. Electrodeposition of poly (N-methyl aniline) on mild steel: Synthesis, Characterization and corrosion protection. J. Electroanal. Chem. 2005, 578, 231–238. [Google Scholar] [CrossRef]

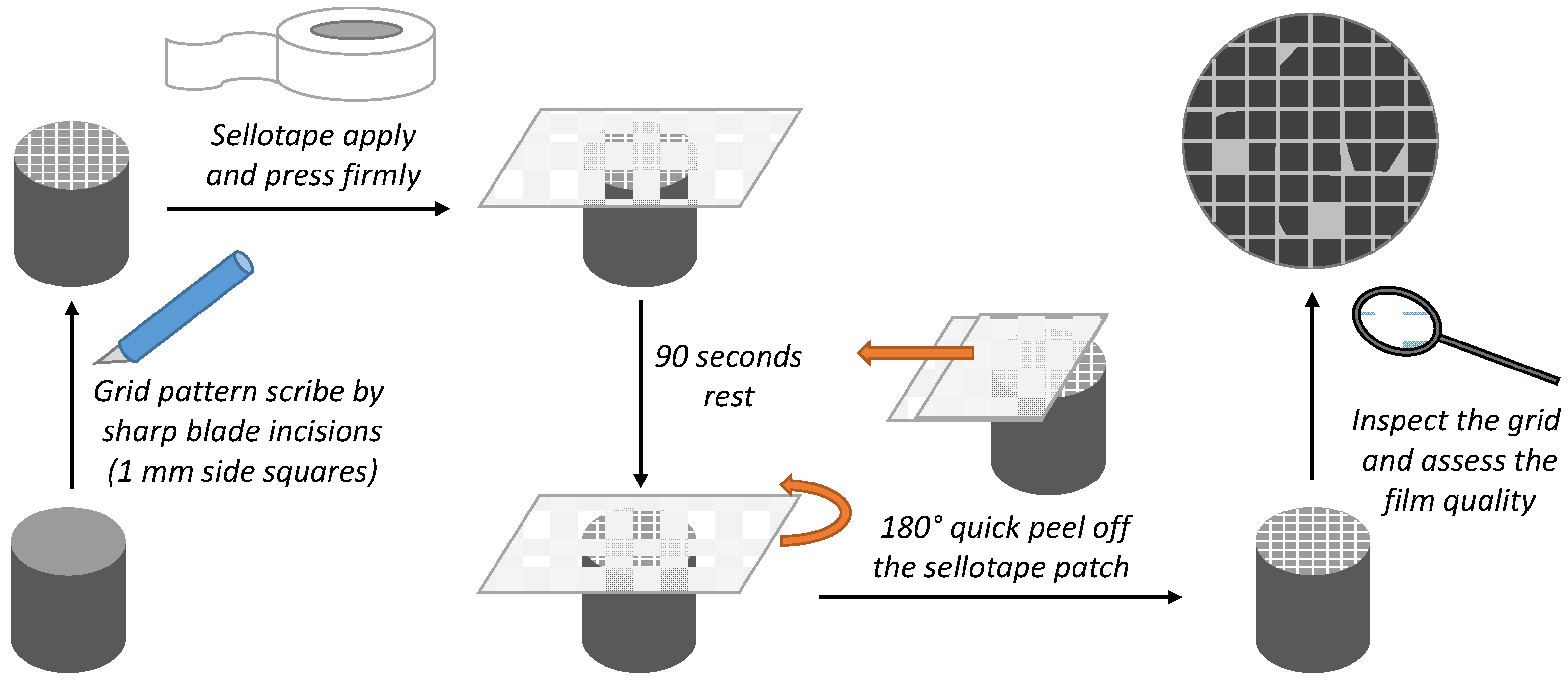
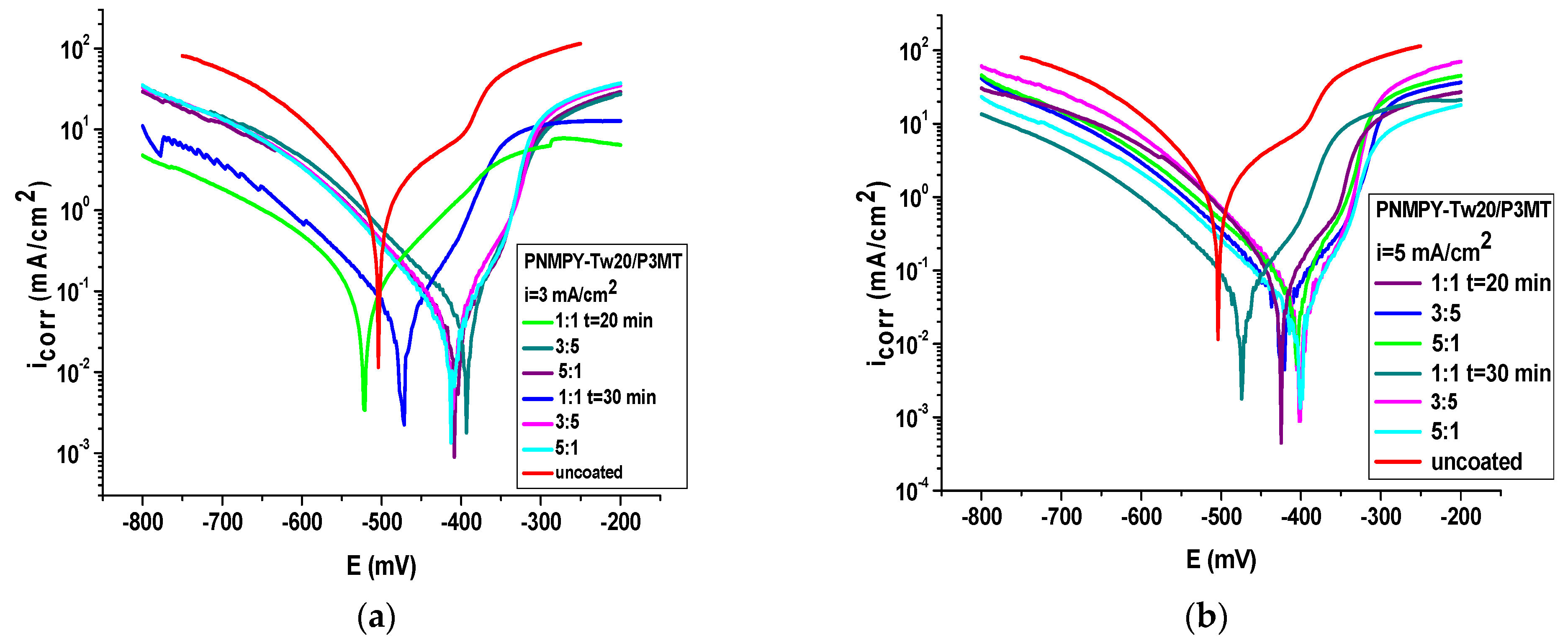





| PNMPY-TW20/P3MT/OLC 45 + H2SO4 | E (mV) | icorr (mA/cm2) | Rp (Ωcm2) | ba (mV/Decade) | bc (mV/Decade) | E% | Rmpy | Pm/year | %P |
|---|---|---|---|---|---|---|---|---|---|
| OLC 45 uncoated | −496 | 0.887 | 17 | 94 | −93 | - | 413 | 11 | - |
| PNMPY-TW20/P3MT i = 3 mA 1:1 t = 20 min | −403 | 0.044 | 286 | 40 | −103 | 94 | 20.53 | 0.52 | 0.0055 |
| PNMPY-TW20/P3MT i = 3 mA 3:5 | −410 | 0.033 | 355 | 60 | −84 | 96 | 15.4 | 0.39 | 0.0054 |
| PNMPY-TW20/P3MT i = 3 mA 5:1 | −416 | 0.0415 | 343 | 68 | −88 | 95 | 19.46 | 0.49 | 0.0052 |
| PNMPY-TW20/P3MT i =3 mA 1:1 t = 30 min | −413 | 0.045 | 306 | 60 | −92 | 94 | 21 | 0.53 | 0.0066 |
| PNMPY-TW20/P3MT i = 3 mA 3:5 | −403 | 0.051 | 245 | 48 | −102 | 95 | 20 | 0.5 | 0.0066 |
| PNMPY-TW20/P3MT i = 3 mA 5:1 | −414 | 0.031 | 392 | 60 | −78 | 96 | 14 | 0.36 | 0.0046 |
| PNMPY-TW20/P3MT/OLC 45 + H2SO4 | E (mV) | icorr (mA/cm2) | Rp (Ωcm2) | ba (mV/Decade) | bc (mV/Decade) | E% | Rmpy | Pmm/year | %P |
|---|---|---|---|---|---|---|---|---|---|
| OLC 45 uncoated | −496 | 0.887 | 17 | 94 | −93 | - | 413 | 11 | - |
| PNMPY-TW20/P3MT i = 5 mA 1:1 t = 20 min | −428 | 0.056 | 190 | 69 | −68 | 94 | 26.13 | 0.66 | 0.016 |
| PNMPY-TW20/P3MT i = 5 mA 3:5 | −426 | 0.038 | 321 | 78 | −79 | 95.6 | 17.73 | 0.45 | 0.0076 |
| PNMPY-TW20/P3MT i = 5 mA 5:1 | −410 | 0.041 | 233 | 56 | −92 | 95 | 21.46 | 0.54 | 0.0078 |
| PNMPY-TW20/P3MT i = 5 mA 1:1 t = 30 min | −467 | 0.045 | 295 | 69 | −80 | 95 | 20.76 | 0.527 | 0.027 |
| PNMPY-TW20/P3MT i = 5 mA 3:5 | −402 | 0.039 | 349 | 61 | −67 | 95.6 | 18.66 | 0.473 | 0.0046 |
| PNMPY-TW20/P3MT i = 5 mA 5:1 | −405 | 0.027 | 517 | 56 | −99 | 97 | 12.6 | 0.319 | 0.0034 |
| PNMPY-TW20/P3MT/OLC 45 + H2SO4 | E (mV) | icorr (mA/cm2) | Rp (Ωcm2) | ba (mV/Decade) | bc (mV/Decade) | E% | Rmpy | Pmm/year | %P |
|---|---|---|---|---|---|---|---|---|---|
| OLC 45 uncoated | −496 | 0.887 | 17 | 94 | −93 | - | 413 | 11 | - |
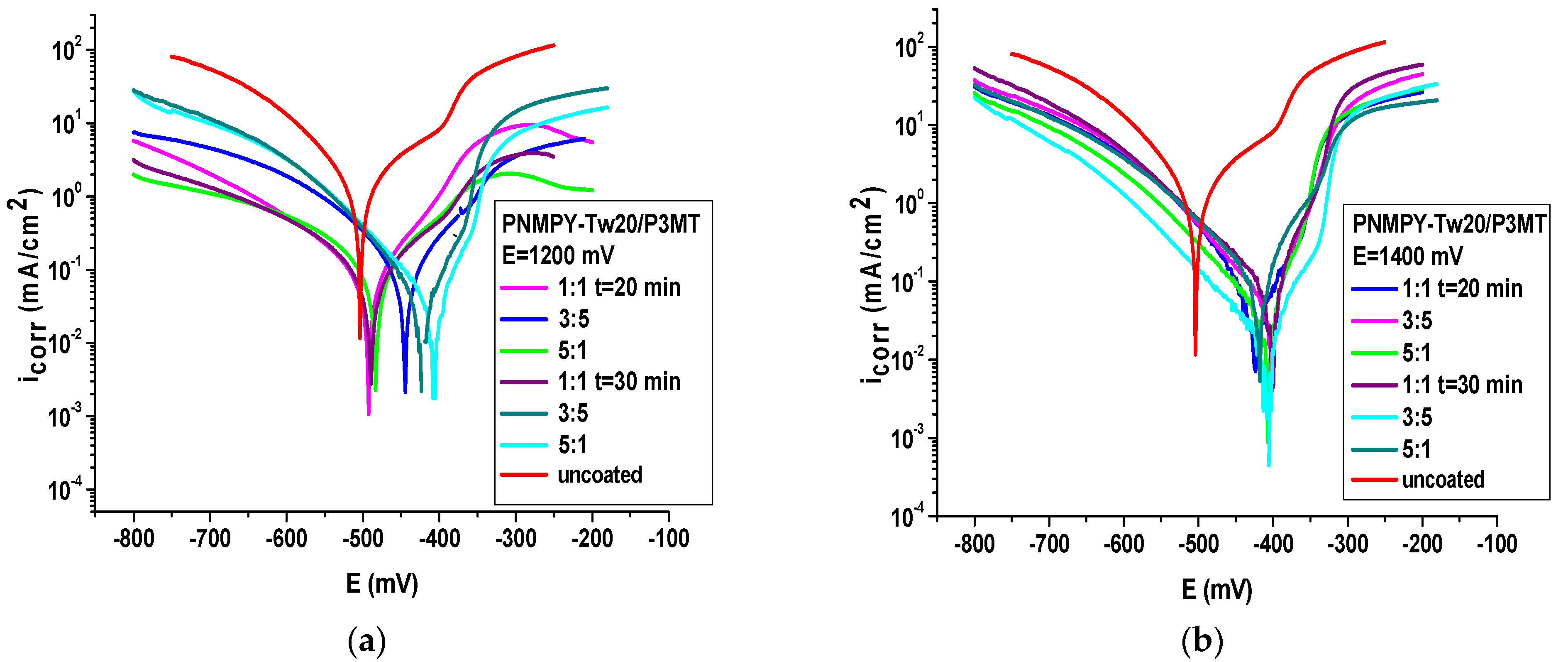
| PNMPY-TW20/P3MT E = 1.2 V 1:1 t = 20 min | −437 | 0.074 | 191 | 77 | −86 | 91 | 34.53 | 0.867 | 0.017 |
| PNMPY-TW20/P3MT E = 1.2 V 3:5 | −440 | 0.057 | 240 | 73 | −97 | 94 | 26.6 | 0.67 | 0.015 |
| PNMPY-TW20/P3MT E = 1.2 V 5:1 | −428 | 0.061 | 217 | 87 | 104 | 93 | 28.46 | 0.72 | 0.012 |
| PNMPY-TW20/P3MT E = 1.2 V 1:1 t = 30 min | −453 | 0.055 | 262 | 87 | −95 | 94 | 25.12 | 0.63 | 0.018 |
| PNMPY-TW20/P3MT E = 1.2 V 3:5 | −426 | 0.041 | 338 | 60 | −77 | 95 | 19.13 | 0.48 | 0.007 |
| PNMPY-TW20/P3MT E = 1.2 V 5:1 | −410 | 0.036 | 358 | 59 | 90 | 96 | 10.73 | 0.272 | 0.0052 |
| PNMPY-TW20/P3MT/OLC 45 + H2SO4 | E (mV) | icorr (mA/cm2) | Rp (Ωcm2) | ba (mV/Decade) | bc (mV/Decade) | E% | Rmpy | Pmm/year | %P |
|---|---|---|---|---|---|---|---|---|---|
| OLC 45 uncoated | −496 | 0.887 | 17 | 94 | −93 | - | 413 | 11 | - |
| PNMPY-TW20/P3MT E = 1.4 V 1:1 t = 20 min | −426 | 0.051 | 222 | 53 | −91 | 94 | 24 | 0.51 | 0.0114 |
| PNMPY-TW20/P3MT E = 1.4 V 3:5 | −405 | 0.046 | 285 | 67 | −63 | 94 | 21.4 | 0.544 | 0.006 |
| PNMPY-TW20/P3MT E = 1.4 V 5:1 | −410 | 0.032 | 371 | 50 | −93 | 96 | 15 | 0.38 | 0.0052 |
| PNMPY-TW20/P3MT E = 1.4 V 1:1 t = 30 min | −405 | 0.067 | 169 | 56 | −94 | 92 | 31.26 | 0.79 | 0.0096 |
| PNMPY-TW20/P3MT E = 1.4 V 3:5 | −410 | 0.056 | 196 | 60 | −84 | 93 | 26 | 0.66 | 0.0091 |
| PNMPY-TW20/P3MT E = 1.4 V 5:1 | −412 | 0.044 | 228 | 50 | −74 | 95 | 20.5 | 0.521 | 0.0084 |
| PNMPY-TW20/P3MT/OLC 45 + H2SO4 | E (mV) | icorr (mA/cm2) | Rp (Ωcm2) | ba (mV/Decade) | bc (mV/Decade) | Rmpy | Pmm/year | E% |
|---|---|---|---|---|---|---|---|---|
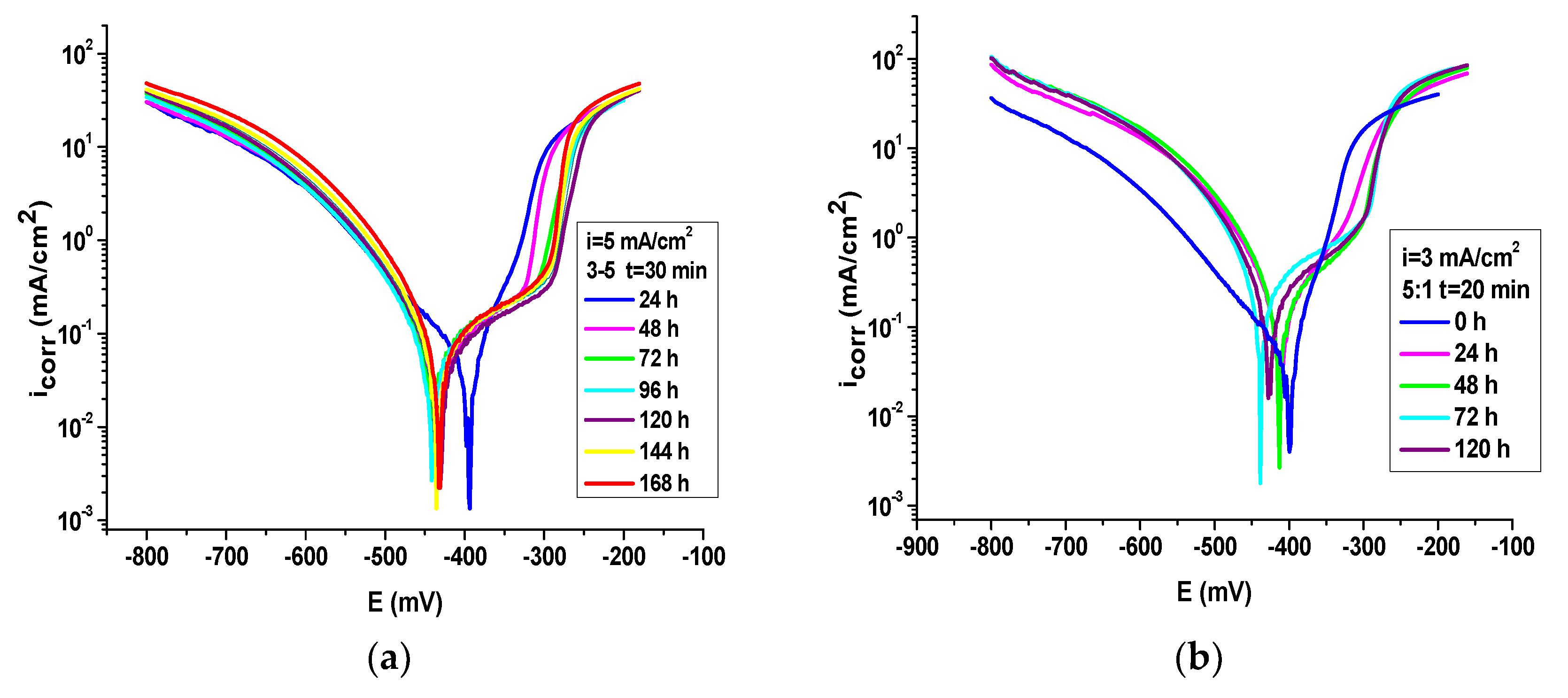
| i = 5 mA/cm2 3:5 t = 30 min 0 h | −402 | 0.039 | 334 | 61 | −67 | 18.6 | 0.47 | 95 |
| i = 5 mA/cm2 3:5 t = 30 min 24 h | −435 | 0.051 | 310 | 120 | −69 | 24 | 0. 61 | 94 |
| i = 5 mA/cm2 3:5 t = 30 min 48 h | −443 | 0.058 | 296 | 134 | −70 | 27 | 0. 68 | 93 |
| i = 5 mA/cm2 3:5 t = 30 min 72 h | −441 | 0.057 | 302 | 129 | −71 | 26 | 0. 67 | 94 |
| i = 5 mA/cm2 3:5 t = 30 min 96 h | −432 | 0.049 | 398 | 125 | −68 | 22 | 0. 58 | 94 |
| i = 5 mA/cm2 3:5 t = 30 min 120 h | −437 | 0.059 | 300 | 137 | −69 | 27 | 0. 69 | 93 |
| i = 5 mA/cm2 3:5 t = 30 min 144 h | −436 | 0.063 | 278 | 132 | −67 | 29 | 0. 74 | 93 |
| i = 5 mA/cm2 3:5 t = 30 min 168 h | −430 | 0.073 | 270 | 133 | −68 | 34 | 0. 86 | 92 |
| i = 3 mA/cm2 5:1 t = 20 min 0 h | −403 | 0.044 | 286 | 46 | −92 | 20.5 | 0.52 | 95 |
| i = 3 mA/cm2 5:1 t = 20 min 24 h | −413 | 0.072 | 106 | 77 | −57 | 33 | 0.85 | 92 |
| i = 3 mA/cm2 5:1 t = 20 min 48 h | −414 | 0.117 | 99 | 130 | −68 | 54 | 1.35 | 87 |
| i = 3 mA/cm2 5:1 t = 20 min 96 h | −427 | 0.18 | 87 | 131 | −67 | 81 | 2.11 | 80 |
| i = 3 mA/cm2 5:1 t = 20 min 120 h | −431 | 0.19 | 81 | 133 | −69 | 86 | 2.18 | 80 |
| E = 1.2 V 1:1 t = 30 min 0 h | −453 | 0.055 | 262 | 87 | −95 | 25.12 | 0.63 | 94 |
| E = 1.2 V 1:1 t = 30 min 24 h | −525 | 0.11 | 150 | 112 | −100 | 51.3 | 1.30 | 88 |
| E = 1.2 V 1:1 t = 30 min 48 h | −540 | 0.16 | 144 | 105 | −93 | 74.6 | 1.89 | 82 |
| E = 1.2 V 1:1 t = 30 min 96 h | −501 | 0.32 | 71 | 103 | −98 | 148 | 3.75 | 65 |
| E = 1.2 V 1:1 t = 30 min 120 h | −482 | 0.46 | 69 | 110 | −78 | 149 | 3.79 | 50 |
| The System PNMPY-TW20/P3MT/OLC 45 + H2SO4 | Rs (ohm × cm2) | Q−Yo S⋅s-n⋅cm−2 | Q−n | Rct (ohm × cm2) | χ2 | E% |
|---|---|---|---|---|---|---|
| OLC 45 uncoated | 0.881 | 0.0067 | 0.773 | 13 | 4.764 × 10−3 | |
| PNMPY-TW20/P3MT i = 3 mA/cm2 1:1 t = 20 min | 1.03 | 0.00202 | 0.733 | 85 | 4.514 × 10−3 | 85 |
| PNMPY-TW20/P3MT i = 3 mA/cm2 3:5 | 1.535 | 0.00037 | 0.852 | 119 | 2.500 × 10−3 | 90 |
| PNMPY-TW20/P3MT i = 3 mA/cm2 5:1 | 2.461 | 0.001093 | 0.791 | 159 | 2.543 × 10−3 | 92 |
| PNMPY-TW20/P3MT i = 3 mA/cm2 1:1 t = 30 min | 1.842 | 0.003359 | 0.619 | 146 | 4.321 × 10−3 | 91 |
| PNMPY-TW20/P3MT i = 3 mA/cm2 3:5 | 1.576 | 0.001033 | 0.810 | 162 | 4.986 × 10−3 | 93 |
| PNMPY-TW20/P3MT i = 3 mA/cm2 5:1 | 1.512 | 0.001105 | 0.815 | 205 | 5.590 × 10−3 | 94 |
| The System PNMPY-TW20/P3MT/OLC 45 + H2SO4 | Rs (ohm × cm2) | Q−Yo S⋅s-n⋅cm−2 | Q−n | Rct (ohm × cm2) | χ2 | E% |
|---|---|---|---|---|---|---|
| OLC 45 uncoated | 0.881 | 0.0067 | 0.773 | 13 | 4.764 × 10−3 | |
| PNMPY-TW20/P3MT i = 5 mA/cm2 1:1 t = 20 min | 2.126 | 0.00114 | 0.8454 | 87 | 2.279 × 10−3 | 86 |
| PNMPY-TW20/P3MT i = 5 mA/cm2 3:5 | 1.543 | 0.00078 | 0.828 | 152 | 2.372 × 10−3 | 92 |
| PNMPY-TW20/P3MT i = 5 mA/cm2 5:1 | 1.322 | 0.000910 | 0.831 | 123 | 2.752 × 10−3 | 90 |
| PNMPY-TW20/P3MT i = 5 mA/cm2 1:1 t = 30 min | 0.744 | 0.00262 | 0.8512 | 94 | 5.937 × 10−3 | 87 |
| PNMPY-TW20/P3MT i = 5 mA/cm2 3:5 | 2.655 | 0.0002435 | 0.8662 | 188 | 2.559 × 10−3 | 94 |
| PNMPY-TW20/P3MT i = 5 mA/cm2 5:1 | 2.37 | 0.0006286 | 0.8336 | 233 | 4.505 × 10−3 | 95 |
| PNMPY-TW20/P3MT/OLC 45 + H2SO4 | Rs (ohm × cm2) | Q−Yo S⋅s-n⋅cm−2 | Q−n | Rct (ohm × cm2) | χ2 | E% |
|---|---|---|---|---|---|---|
| OLC 45 uncoated | 0.881 | 0.0067 | 0.773 | 13 | 4.764 × 10−3 | |
| PNMPY-TW20/P3MT E = 1.2 V 1:1 t = 20 min | 3.792 | 0.001749 | 0.7803 | 109 | 3.247 × 10−3 | 88 |
| PNMPY-TW20/P3MT E = 1.2 V 3:5 | 3.504 | 0.000287 | 0.793 | 132 | 4.632 × 10−3 | 91 |
| PNMPY-TW20/P3MT E = 1.2 V 5:1 | 1.374 | 0.001088 | 0.8548 | 182 | 3.952 × 10−3 | 93 |
| PNMPY-TW20/P3MTE = 1.2 V 1:1 t = 30 min | 1.708 | 0.001408 | 0.8117 | 172 | 4.271 × 10−3 | 93 |
| PNMPY-TW20/P3MT E = 1.2 V 3:5 | 1.831 | 0.001781 | 0.8228 | 206 | 4.271 × 10−3 | 94 |
| PNMPY-TW20/P3MT E = 1.2 V 5:1 | 7.612 | 0.000453 | 0.7426 | 246 | 6.451 × 10−3 | 95 |
| PNMPY-TW20/P3MT/OLC 45 + H2SO4 | Rs (ohm × cm2) | Q−Yo S⋅s-n⋅cm−2 | Q−n | Rct (ohm × cm2) | χ2 | E% |
|---|---|---|---|---|---|---|
| OLC 45 uncoated | 0.881 | 0.0067 | 0.773 | 13 | 4.764 × 10−3 | |
| PNMPY-TW20/P3MT E = 1.4 V 1:1 t = 20 min | 1.084 | 0.003338 | 0.7911 | 85 | 6.073 × 10−3 | 85 |
| PNMPY-TW20/P3MT E = 1.4 V 3:5 | 2.147 | 0.000966 | 0.8165 | 90 | 3.865 × 10−3 | 86 |
| PNMPY-TW20/P3MT E = 1.4 V 5:1 | 1.497 | 0.001813 | 0.8128 | 123 | 3.370 × 10−3 | 90 |
| PNMPY-TW20/P3MT E = 1.4 V 1:1 t = 30 min | 1.057 | 0.002098 | 0.8644 | 81 | 4.558 × 10−3 | 84 |
| PNMPY-TW20/P3MT E = 1.4 V 3:5 | 1.558 | 0.001551 | 0.829 | 99 | 3.773 × 10−3 | 87 |
| PNMPY-TW20/P3MT E = 1.4 V 5:1 | 3.563 | 0.000888 | 0.8339 | 159 | 5.875 × 10−3 | 92 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Branzoi, F.; Mihai, M.A.; Petrescu, S. Corrosion Protection Efficacy of the Electrodeposit of Poly (N-Methyl Pyrrole-Tween20/3-Methylthiophene) Coatings on Carbon Steel in Acid Medium. Coatings 2022, 12, 1062. https://doi.org/10.3390/coatings12081062
Branzoi F, Mihai MA, Petrescu S. Corrosion Protection Efficacy of the Electrodeposit of Poly (N-Methyl Pyrrole-Tween20/3-Methylthiophene) Coatings on Carbon Steel in Acid Medium. Coatings. 2022; 12(8):1062. https://doi.org/10.3390/coatings12081062
Chicago/Turabian StyleBranzoi, Florina, Marius Alexandru Mihai, and Simona Petrescu. 2022. "Corrosion Protection Efficacy of the Electrodeposit of Poly (N-Methyl Pyrrole-Tween20/3-Methylthiophene) Coatings on Carbon Steel in Acid Medium" Coatings 12, no. 8: 1062. https://doi.org/10.3390/coatings12081062
APA StyleBranzoi, F., Mihai, M. A., & Petrescu, S. (2022). Corrosion Protection Efficacy of the Electrodeposit of Poly (N-Methyl Pyrrole-Tween20/3-Methylthiophene) Coatings on Carbon Steel in Acid Medium. Coatings, 12(8), 1062. https://doi.org/10.3390/coatings12081062






