In Situ Synthesis of CuO/Cu2O Nanoparticle-Coating Nanoporous Alumina Membranes with Photocatalytic Activity under Visible Light Radiation
Abstract
:1. Introduction
2. Experimental
2.1. Chemicals
2.2. Membrane Fabrication
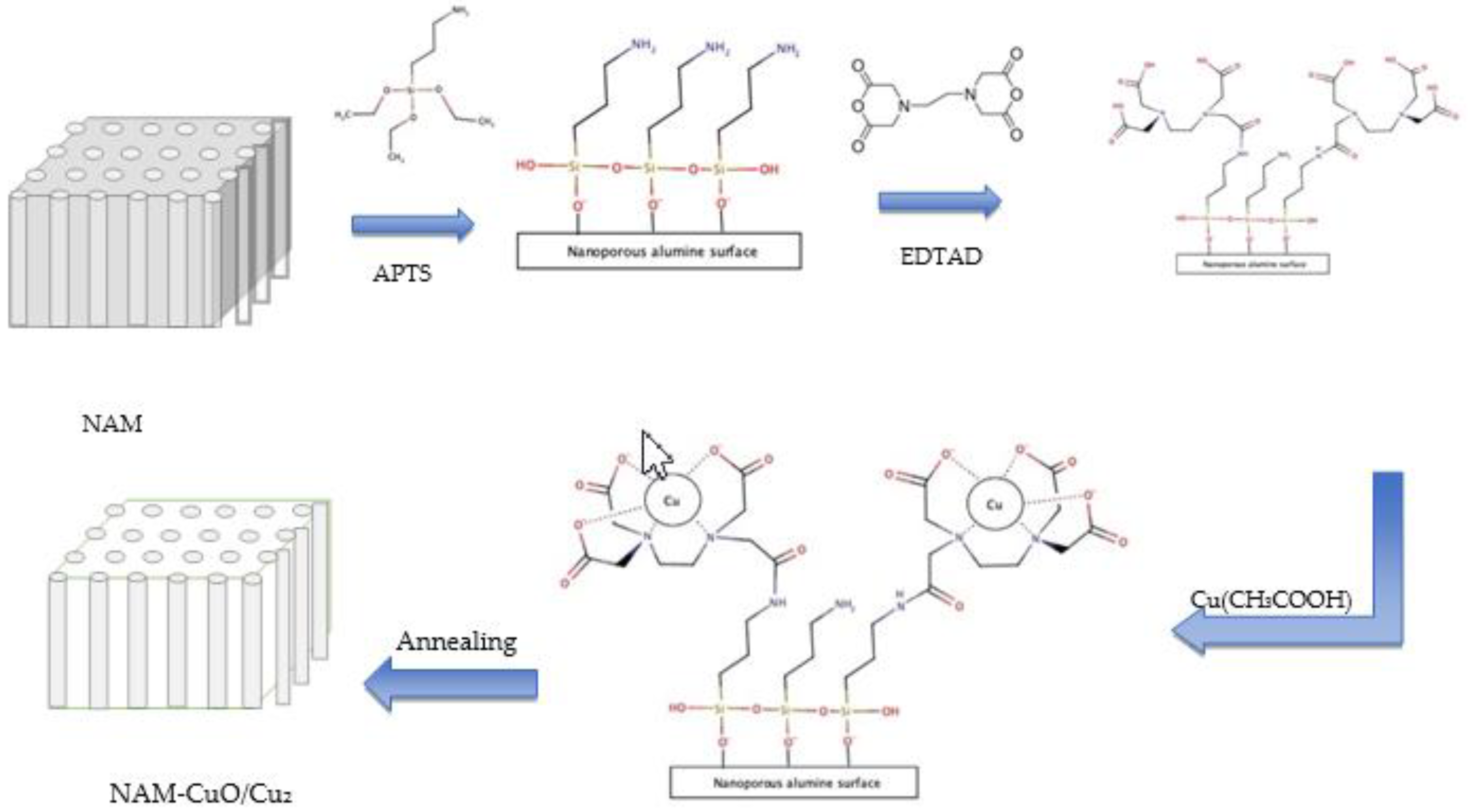
2.3. Membrane Modification
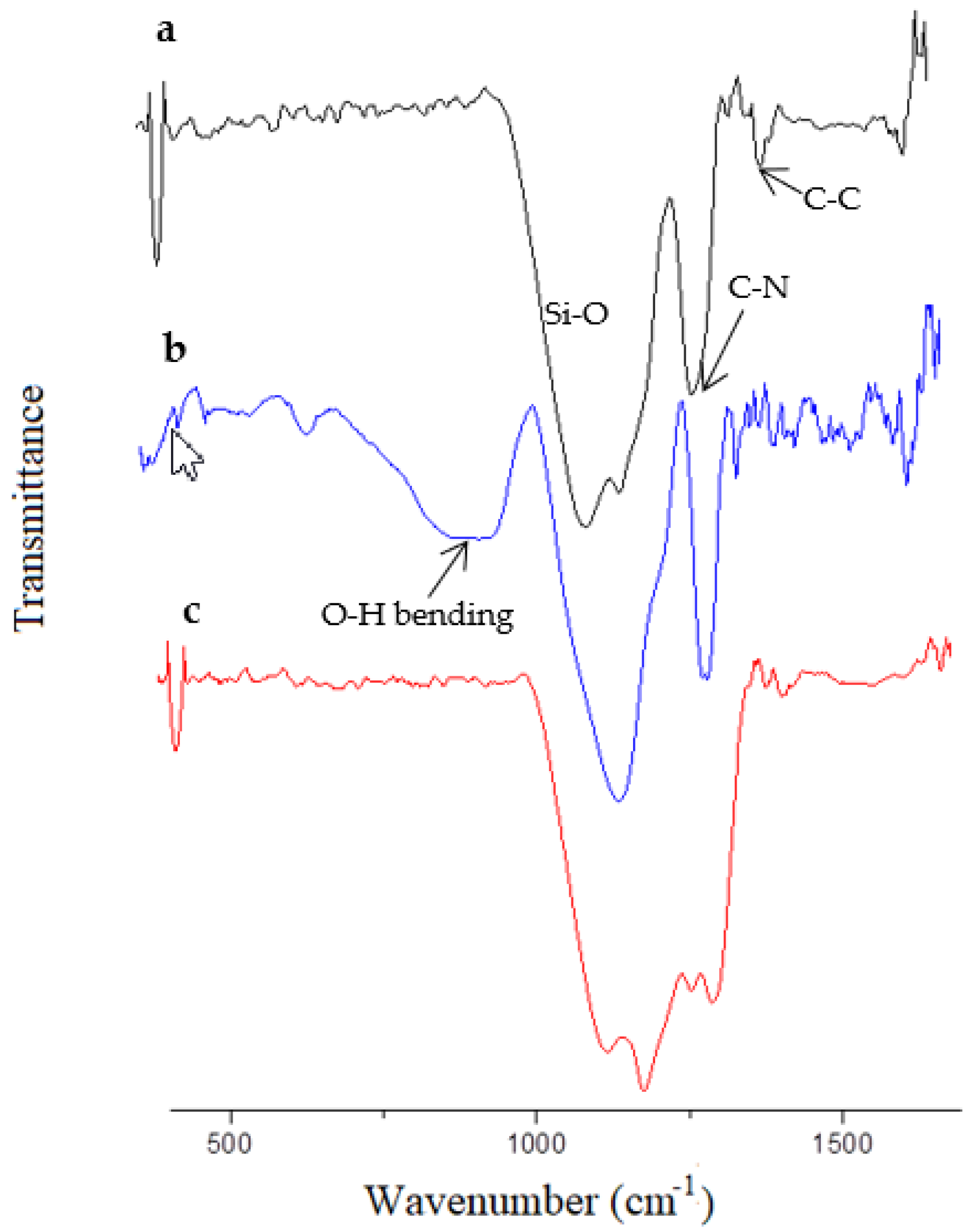
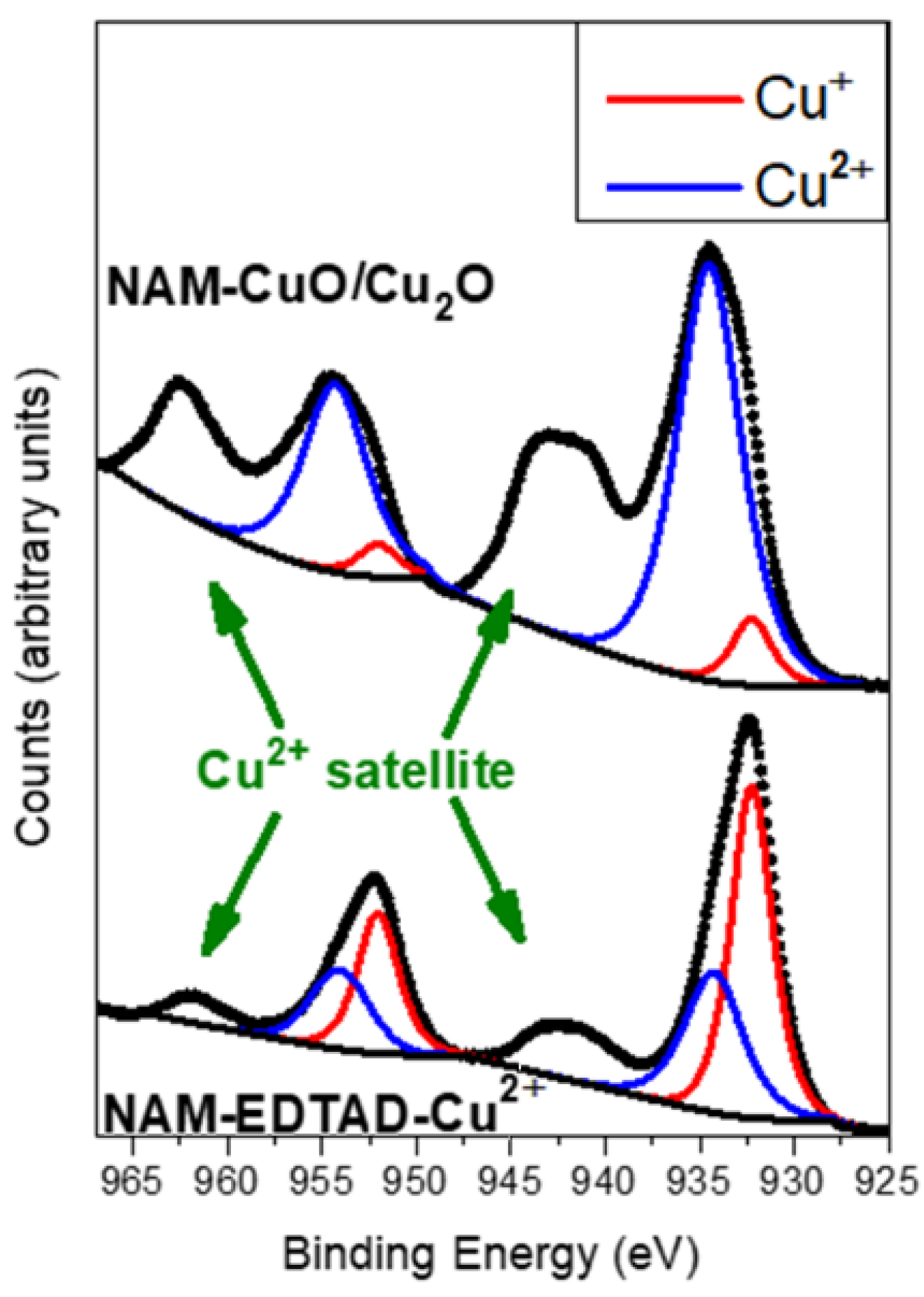
2.4. Membrane Characterization
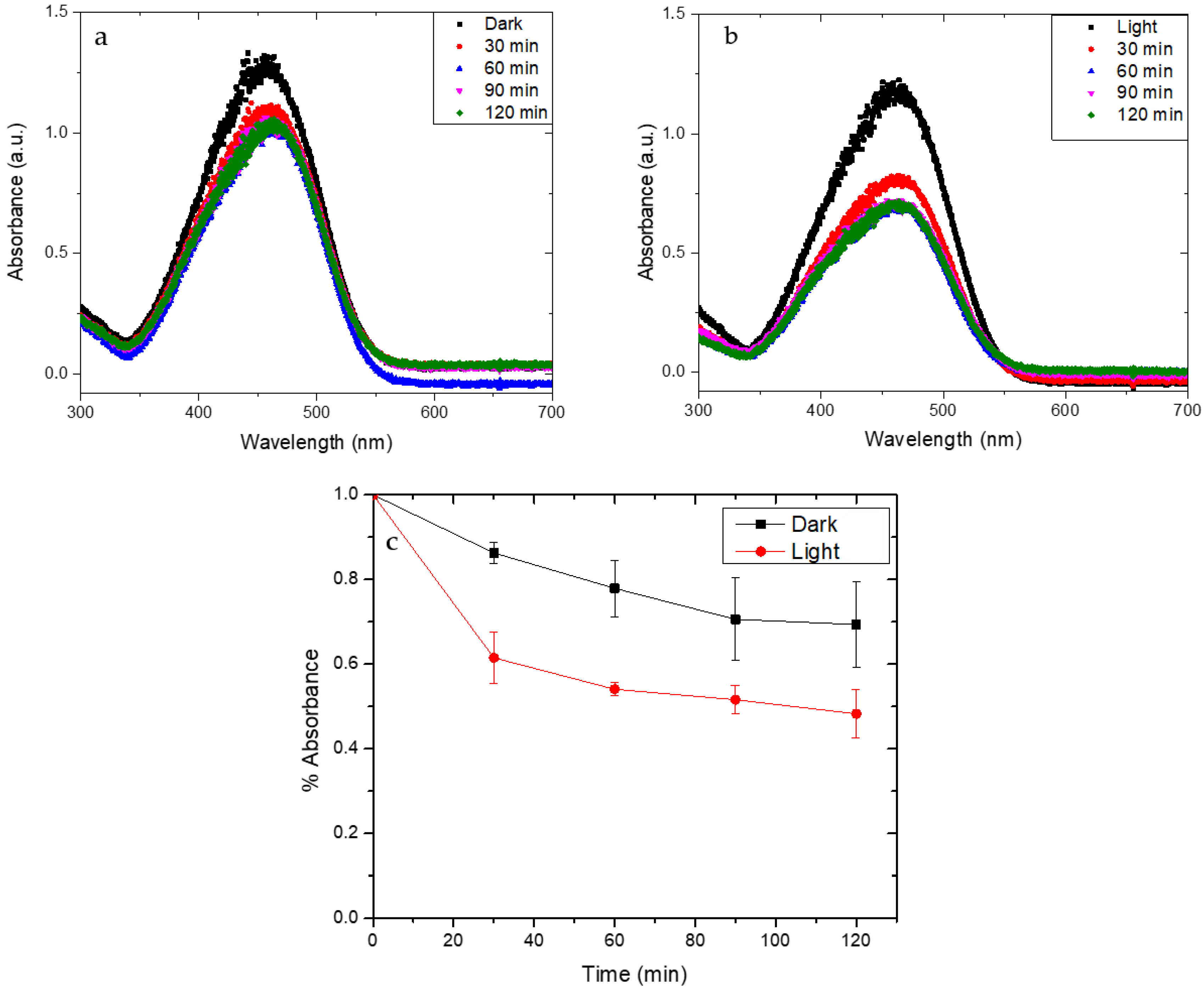
2.5. Photocatalytic Activity Tests
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tishkevich, D.I.; Vorobjova, A.I.; Bondaruk, A.A.; Dashkevich, E.S.; Shimanovich, D.L.; Razanau, I.U.; Zubar, T.I.; Yakimchuk, D.V.; Dong, M.G.; Sayyed, M.I.; et al. The Interrelation of Synthesis Conditions and Wettability Properties of the Porous Anodic Alumina Membranes. Nanomaterials 2022, 12, 2382. [Google Scholar] [CrossRef]
- Nagaoka, S.; Yoshida, K.; Hirota, Y.; Komachi, Y.; Takafuji, M.; Ihara, H. Functionalized Aluminum Oxide by Immobilization of Totally Organic Aromatic Polymer Spherical Nanoparticles. Colloids Surf. A Physicochem. Eng. Asp. 2022, 640, 128438. [Google Scholar] [CrossRef]
- Li, Z.; Zhu, G.; Mo, R.; Wu, M.; Ding, X.; Huang, L.; Wu, Z.; Xia, X. Light-Enhanced Osmotic Energy Harvester Using Photoactive Porphyrin Metal–Organic Framework Membranes. Angew. Chem. 2022, 134. [Google Scholar] [CrossRef]
- Makela, M.; Lin, Z.; Lin, P.T. Surface Functionalized Anodic Aluminum Oxide Membrane for Opto-Nanofluidic SARS-CoV-2 Genomic Target Detection. IEEE Sens. J. 2021, 21, 22645–22650. [Google Scholar] [CrossRef]
- Bruera, F.A.; Kramer, G.R.; Velázquez, J.E.; Sadañoski, M.A.; Fonseca, M.I.; Ares, A.E.; Zapata, P.D. Laccase Immobilization on Nanoporous Aluminum Oxide for Black Liquor Treatment. Surf. Interfaces 2022, 30, 101879. [Google Scholar] [CrossRef]
- Previdi, R.; Levchenko, I.; Arnold, M.; Gali, M.; Bazaka, K.; Xu, S.; Ostrikov, K.K.; Bray, K.; Jin, D.; Fang, J. Plasmonic Platform Based on Nanoporous Alumina Membranes: Order Control via Self-Assembly. J. Mater. Chem. A Mater. 2019, 7, 9565–9577. [Google Scholar] [CrossRef]
- Shang, G.; Bi, D.; Gorelik, V.S.; Fei, G.; Zhang, L. Anodic Alumina Photonic Crystals: Structure Engineering, Optical Properties and Prospective Applications. Mater. Today Commun. 2023, 34, 105052. [Google Scholar] [CrossRef]
- Vorobjova, A.; Tishkevich, D.; Shimanovich, D.; Zubar, T.; Astapovich, K.; Kozlovskiy, A.; Zdorovets, M.; Zhaludkevich, A.; Lyakhov, D.; Michels, D.; et al. The Influence of the Synthesis Conditions on the Magnetic Behaviour of the Densely Packed Arrays of Ni Nanowires in Porous Anodic Alumina Membranes. RSC Adv. 2021, 11, 3952–3962. [Google Scholar] [CrossRef]
- Jiang, N.; Shrotriya, P.; Dassanayake, R.P. NK-Lysin Antimicrobial Peptide-Functionalized Nanoporous Alumina Membranes as Biosensors for Label-Free Bacterial Endotoxin Detection. Biochem. Biophys. Res. Commun. 2022, 636, 18–23. [Google Scholar] [CrossRef]
- Niazi, F.K.; Umer, M.A.; Ahmed, A.; Hafeez, M.A.; Khan, Z.; Butt, M.S.; Razzaq, A.; Luo, X.; Park, Y.-K. Nanoporous Alumina Membranes for Sugar Industry: An Investigation of Sintering Parameters Influence OnUltrafiltration Performance. Sustainability 2021, 13, 7593. [Google Scholar] [CrossRef]
- Repo, E.; Warchoł, J.K.; Bhatnagar, A.; Sillanpää, M. Heavy Metals Adsorption by Novel EDTA-Modified Chitosan–Silica Hybrid Materials. J. Colloid Interface Sci. 2011, 358, 261–267. [Google Scholar] [CrossRef] [PubMed]
- Li, F.; Li, Y. Preparation of Efficient and Environment-Friendly Silica-Supported EDTA Platinum Catalyst and Its Applications in Hydrosilylation of Olefins and Methyldichlorosilane. J. Mol. Catal. A Chem. 2016, 420, 254–263. [Google Scholar] [CrossRef]
- Repo, E.; Warchol, J.K.; Kurniawan, T.A.; Sillanpää, M.E.T. Adsorption of Co(II) and Ni(II) by EDTA- and/or DTPA-Modified Chitosan: Kinetic and Equilibrium Modeling. Chem. Eng. J. 2010, 161, 73–82. [Google Scholar] [CrossRef]
- Repo, E.; Malinen, L.; Koivula, R.; Harjula, R.; Sillanpää, M. Capture of Co(II) from Its Aqueous EDTA-Chelate by DTPA-Modified Silica Gel and Chitosan. J. Hazard. Mater. 2011, 187, 122–132. [Google Scholar] [CrossRef]
- Pereira, F.V.; Gurgel, L.V.A.; Gil, L.F. Removal of Zn2+ from Aqueous Single Metal Solutions and Electroplating Wastewater with Wood Sawdust and Sugarcane Bagasse Modified with EDTA Dianhydride (EDTAD). J. Hazard. Mater. 2010, 176, 856–863. [Google Scholar] [CrossRef] [Green Version]
- Córdoba, A.; Durán, B.; Bonardd, S.; Diaz Diaz, D.; Leiva, A.; Saldías, C. In Situ Synthesis and Immobilization of CuO Nanoparticles in Alginate-Poly(Amido Amine) Nanogels for Photocatalytic Applications. Mater. Lett. X 2022, 14, 100148. [Google Scholar] [CrossRef]
- Liu, S.-H.; Wei, Y.-S.; Lu, J.-S. Visible-Light-Driven Photodegradation of Sulfamethoxazole and Methylene Blue by Cu2O/RGO Photocatalysts. Chemosphere 2016, 154, 118–123. [Google Scholar] [CrossRef]
- Shoeib, M.A.; Abdelsalam, O.E.; Khafagi, M.G.; Hammam, R.E. Synthesis of Cu2O Nanocrystallites and Their Adsorption and Photocatalysis Behavior. Adv. Powder Technol. 2012, 23, 298–304. [Google Scholar] [CrossRef]
- Kobayashi, Y.; Yasuda, Y.; Morita, T. Recent Advances in the Synthesis of Copper-Based Nanoparticles for Metal–Metal Bonding Processes. J. Sci. Adv. Mater. Devices 2016, 1, 413–430. [Google Scholar] [CrossRef] [Green Version]
- Hevia, S.A.; Homm, P.; Guzmán, F.; Ruiz, H.M.; Muñoz, G.; Caballero, L.S.; Favre, M.; Flores, M. Pulsed Laser Deposition of Carbon Nanodot Arrays Using Porous Alumina Membranes as a Mask. Surf. Coat. Technol. 2014, 253, 161–165. [Google Scholar] [CrossRef]
- Chan, K.Y.; Ye, W.W.; Zhang, Y.; Xiao, L.D.; Leung, P.H.M.; Li, Y.; Yang, M. Ultrasensitive Detection of E. Coli O157:H7 with Biofunctional Magnetic Bead Concentration via Nanoporous Membrane Based Electrochemical Immunosensor. Biosens. Bioelectron. 2013, 41, 532–537. [Google Scholar] [CrossRef] [PubMed]
- Baranowska, M.; Slota, A.J.; Eravuchira, P.J.; Macias, G.; Xifré-Pérez, E.; Pallares, J.; Ferré-Borrull, J.; Marsal, L.F. Protein Attachment to Nanoporous Anodic Alumina for Biotechnological Applications: Influence of Pore Size, Protein Size and Functionalization Path. Colloids Surf. B Biointerfaces 2014, 122, 375–383. [Google Scholar] [CrossRef]
- Xie, Y.; Hill, C.A.S.; Xiao, Z.; Militz, H.; Mai, C. Silane Coupling Agents Used for Natural Fiber/Polymer Composites: A Review. Compos. Part A Appl. Sci. Manuf. 2010, 41, 806–819. [Google Scholar] [CrossRef]
- Du, F.; Chen, Q.-Y.; Wang, Y.-H. Effect of Annealing Process on the Heterostructure CuO/Cu2O as a Highly Efficient Photocathode for Photoelectrochemical Water Reduction. J. Phys. Chem. Solids 2017, 104, 139–144. [Google Scholar] [CrossRef]
- Kim, T.G.; Oh, H.; Ryu, H.; Lee, W.-J. The Study of Post Annealing Effect on Cu2O Thin-Films by Electrochemical Deposition for Photoelectrochemical Applications. J. Alloy. Compd. 2014, 612, 74–79. [Google Scholar] [CrossRef]
- Zhang, M.; Salje, E.K.H. Infrared Spectroscopic Analysis of Zircon: Radiation Damage and the Metamict State. J. Phys. Condens. Matter 2001, 13, 3057–3071. [Google Scholar] [CrossRef]
- Wong, K.C. Review of Spectrometric Identification of Organic Compounds, 8th Edition. J. Chem. Educ. 2015, 92, 1602–1603. [Google Scholar] [CrossRef]
- Ma, D.-L.; He, H.-Z.; Chan, D.S.-H.; Wong, C.-Y.; Leung, C.-H. A Colorimetric and Luminescent Dual-Modal Assay for Cu(II) Ion Detection Using an Iridium(III) Complex. PLoS ONE 2014, 9, e99930. [Google Scholar] [CrossRef] [PubMed]
- An, X.; Li, K.; Tang, J. Cu2O/Reduced Graphene Oxide Composites for the Photocatalytic Conversion of CO2. ChemSusChem 2014, 7, 1086–1093. [Google Scholar] [CrossRef] [Green Version]
- Januário, E.; Nogueira, A.; Pastore, H. ETS-10 Modified with CuxO Nanoparticles and Their Application for the Conversion of CO2 and Water into Oxygenates. J. Braz. Chem. Soc. 2018. [Google Scholar] [CrossRef]
- Bhargava, R.; Khan, S. Enhanced Optical Properties of Cu2O Anchored on Reduced Graphene Oxide (RGO) Sheets. J. Phys. Condens. Matter 2018, 30, 335703. [Google Scholar] [CrossRef]
- Prashanth, P.A.; Raveendra, R.S.; Hari Krishna, R.; Ananda, S.; Bhagya, N.P.; Nagabhushana, B.M.; Lingaraju, K.; Raja Naika, H. Synthesis, Characterizations, Antibacterial and Photoluminescence Studies of Solution Combustion-Derived α-Al2O3 Nanoparticles. J. Asian Ceram. Soc. 2015, 3, 345–351. [Google Scholar] [CrossRef] [Green Version]
- He, H.; Liu, Y.; Wu, D.; Guan, X.; Zhang, Y. Ozonation of Dimethyl Phthalate Catalyzed by Highly Active CuO-Fe3O4 Nanoparticles Prepared with Zero-Valent Iron as the Innovative Precursor. Environ. Pollut. 2017, 227, 73–82. [Google Scholar] [CrossRef]
- Ding, Y.; Zhu, L.; Wang, N.; Tang, H. Sulfate Radicals Induced Degradation of Tetrabromobisphenol A with Nanoscaled Magnetic CuFe2O4 as a Heterogeneous Catalyst of Peroxymonosulfate. Appl. Catal. B 2013, 129, 153–162. [Google Scholar] [CrossRef]
- Li, H.; Su, Z.; Hu, S.; Yan, Y. Free-Standing and Flexible Cu/Cu2O/CuO Heterojunction Net: A Novel Material as Cost-Effective and Easily Recycled Visible-Light Photocatalyst. Appl. Catal. B 2017, 207, 134–142. [Google Scholar] [CrossRef]
- Akgul, F.A.; Akgul, G.; Yildirim, N.; Unalan, H.E.; Turan, R. Influence of Thermal Annealing on Microstructural, Morphological, Optical Properties and Surface Electronic Structure of Copper Oxide Thin Films. Mater. Chem. Phys. 2014, 147, 987–995. [Google Scholar] [CrossRef]
- John, S.; Roy, S.C. CuO/Cu2O Nanoflake/Nanowire Heterostructure Photocathode with Enhanced Surface Area for Photoelectrochemical Solar Energy Conversion. Appl. Surf. Sci. 2020, 509, 144703. [Google Scholar] [CrossRef]
- Kumar, M.; Das, R.R.; Samal, M.; Yun, K. Highly Stable Functionalized Cuprous Oxide Nanoparticles for Photocatalytic Degradation of Methylene Blue. Mater. Chem. Phys. 2018, 218, 272–278. [Google Scholar] [CrossRef]
- Zhang, F.; Dong, G.; Wang, M.; Zeng, Y.; Wang, C. Efficient Removal of Methyl Orange Using Cu2O as a Dual Function Catalyst. Appl. Surf. Sci. 2018, 444, 559–568. [Google Scholar] [CrossRef]



Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Durán, B.; Saldías, C.; Villarroel, R.; Hevia, S.A. In Situ Synthesis of CuO/Cu2O Nanoparticle-Coating Nanoporous Alumina Membranes with Photocatalytic Activity under Visible Light Radiation. Coatings 2023, 13, 179. https://doi.org/10.3390/coatings13010179
Durán B, Saldías C, Villarroel R, Hevia SA. In Situ Synthesis of CuO/Cu2O Nanoparticle-Coating Nanoporous Alumina Membranes with Photocatalytic Activity under Visible Light Radiation. Coatings. 2023; 13(1):179. https://doi.org/10.3390/coatings13010179
Chicago/Turabian StyleDurán, Boris, César Saldías, Roberto Villarroel, and Samuel A. Hevia. 2023. "In Situ Synthesis of CuO/Cu2O Nanoparticle-Coating Nanoporous Alumina Membranes with Photocatalytic Activity under Visible Light Radiation" Coatings 13, no. 1: 179. https://doi.org/10.3390/coatings13010179
APA StyleDurán, B., Saldías, C., Villarroel, R., & Hevia, S. A. (2023). In Situ Synthesis of CuO/Cu2O Nanoparticle-Coating Nanoporous Alumina Membranes with Photocatalytic Activity under Visible Light Radiation. Coatings, 13(1), 179. https://doi.org/10.3390/coatings13010179





