Abstract
Microbiologically influenced corrosion (MIC) is the process of material degradation in the presence of microorganisms and their biofilms. This is an environmentally assisted type of corrosion, which is highly complex and challenging to fully understand. Different metallic materials, such as steel alloys, magnesium alloys, aluminium alloys, and titanium alloys, have been reported to have adverse effects of MIC on their applications. Though many researchers have reported bacteria as the primary culprit of microbial corrosion, several other microorganisms, including fungi, algae, archaea, and lichen, have been found to cause MIC on metal and non-metal surfaces. However, less attention is given to the MIC caused by fungi, algae, archaea, and lichens. In this review paper, the effects of different microorganisms, including bacteria, fungi, algae, archaea, and lichens, on the corrosion properties of engineering materials have been discussed in detail. This review aims to summarize all of the corrosive microorganisms that directly or indirectly cause the degradation of structural materials. Accusing bacteria of every MIC case without a proper investigation of the corrosion site and an in-depth study of the biofilm and secreted metabolites can create problems in understanding the real cause of the materials’ failure. To identify the real corrosion agent in any environment, it is highly important to study all kinds of microorganisms that exist in that specific environment.
1. Introduction
The process of material deterioration by the direct or indirect involvement of various microscopic microorganisms is called microbiologically influenced corrosion (MIC). MIC is also termed as microbial corrosion, bio-corrosion, and microbially induced corrosion. In 1891, Garret, for the first time, discussed MIC when he found degradation of lead-covered cables by microorganisms [1]. Later on, in 1910, Gaines reported the corrosive activities of microorganisms by relating the sulphur present in the corrosion product with the activities of microbes [2]. However, more attention was paid to this problem in the middle of the last century, and intensive research was conducted, reporting the involvement of microorganisms in the deterioration of various materials [3].
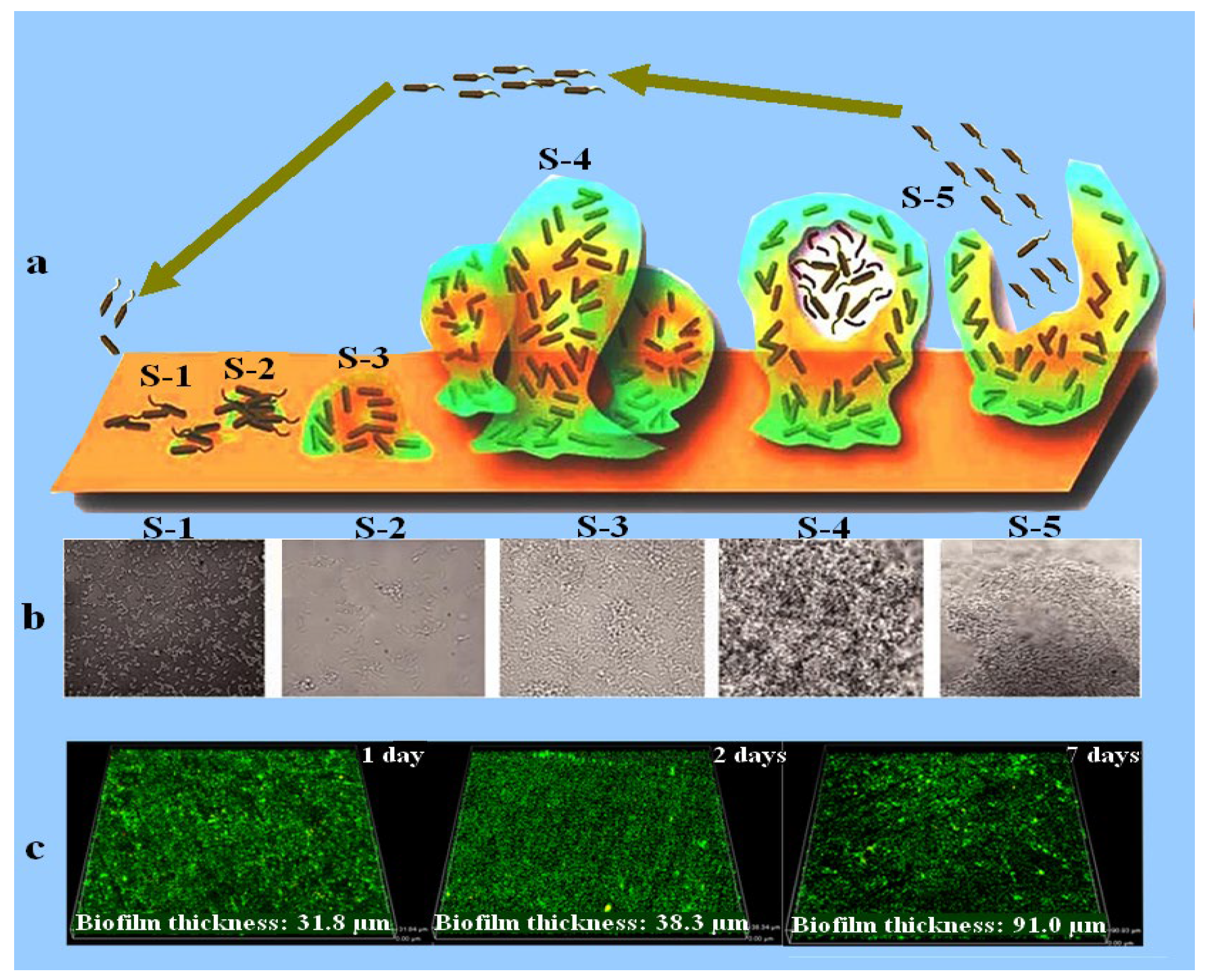
MIC is the main cause of localized corrosion, including pitting corrosion, crevice corrosion, and stress corrosion cracking. Microorganisms become attached to the material surface, secrete extracellular polymeric substances (EPS), and form a biofilm, usually growing in the shape of a continuous sheet, sludge, or tubercle [4]. The name biofilm was coined in 1978 and defined as a matrix enclosing bacterial cells adherent to each other or to surfaces and interfaces [5,6]. Bacterial attachment and biofilm development on surfaces have been shown in Figure 1 [7]. Biofilms formed by corrosive microbes have been considered a topic of interest since 1943 [8]. Different factors, such as the type of microorganisms, surface morphology, chemical composition of the medium, and hydrodynamics, affect biofilm formation on material surfaces [9,10].
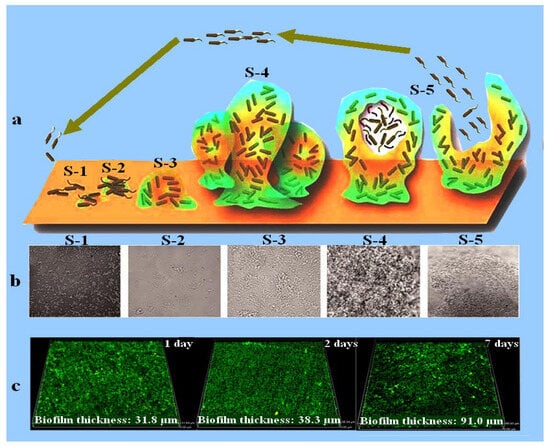
Figure 1.
Schematic diagram displaying biofilm development (a) [11]. (b) SEM micrographs of biofilm development with increasing immersion time, where S-1 represents the bacterial cells attachment, S-2 represents the establishment of bacterial cells in the form of a colony and EPS secretion, S-3 denotes the early stage of biofilm formation, S-4 characterizes the fully grown biofilm, and S-5 is the final stage of the biofilm where bacterial cells leave the biofilm and are mature enough to attach to a new surface. Reprinted with permission from ref. [11]. Copyright 2023 Frontiers in Chemistry. (c) CLSM images showing the biofilm thickness measured after 1, 2, and 7 days of immersion in a medium inoculated with marine bacteria Marinobacter aquaeolei. Reprinted with permission from ref. [12]. Copyright 2023 Springer Nature.
According to the published research, the microorganisms that develop biofilms and secrete aggressive metabolites (such as sulphide ions and acids) along with the secretion of EPS are the most corrosive types of bacteria [3]. EPS is a highly important component of a biofilm, performing the function of glue, keeping microorganisms together in a safe sheath where they can securely grow and proliferate in different shapes. MIC cooperates in the process of wear, cavitation, and erosion, triggering the peeling of the material surface. Since microorganisms usually colonize the material surface, they are more aggressive in producing pits on the surface, which is a worse form of corrosion compared to uniform corrosion [13,14]. Pits are usually initiated beneath the biofilm, attributed to the secretion of corrosive metabolites and the formation of concentrated cells.
Both metallic and non-metallic materials are affected by corrosion caused by microorganisms. Numerous cases of the deterioration of stonework, polymers, architectural building materials, and fibre-reinforced composites caused by microorganisms have been reported [15,16]. MIC is a globally identified problem, having high damaging effects on various engineering alloys in different locations, such as oil and gas fields, water pipelines, fresh water, demineralized water, seawater, chemical processing facilities, food stuffs, aircraft fuel, soil, human plasma, sewage, pulp and paper processing facilities, exploration equipment, transportation vehicles, production facilities, nuclear power generation plants, marine environments, storage facilities, and fire protection systems [17,18,19,20]. Huge economic losses have been reported worldwide that were caused by MIC [21,22]. Material failure owing to MIC greatly increases the potential safety concerns [23]. A crude oil pipeline leakage that occurred in 2006 at Alaska Prudhoe Bay created many problems in the global oil market, and according to the investigation, MIC was the main culprit responsible for the pipeline failure [24]. In another case, the failure due to MIC of an oil transporting pipeline with an 8-inch diameter was reported just 8 months after its installation [25]. Billions of dollars are spent every year on material renovation due to corrosion in the USA alone [26]. It has been observed that 50% of the material failures in the oil and gas industries are due to the activities of microorganisms, and 20%–40% of overall corrosion is due to MIC [27].
The majority of corrosive microorganisms live in aquatic environments. This is because water makes available all of the necessary chemicals for their activities and provides them with nutrients in the best possible form that they can easily digest. MIC is not a separate type of corrosion; indeed, it is electrochemical corrosion in origin and can be explained within the known framework of corrosion control measures already used for controlling electrochemical corrosion [28]. This definition of MIC makes it possible for researchers to find ways for its treatment and control. The parameters needed for MIC are corrosive microorganisms, nutrients, and water-containing medium.
The most common microorganisms frequently found to be involved in initiating and accelerating the corrosion processes are bacteria, fungi, algae, archaea, and lichens. This review focuses on the study of different types of microorganisms involved in MIC. In addition, the habitats of the corrosive microorganisms and the course by which they take part in corrosion processes will be discussed in detail.
2. Bacteria
Most bacteria are unicellular microorganisms and are frequently present in various environments ranging from soil to water, acidic hot springs, and radioactive waste products [29]. This group of microorganisms can survive at temperatures ranging from −10 °C to 100 °C and pHs from 0 to 10.5. The oxygen concentration needed for their growth is 0 to saturated medium, and the pressure required is from vacuum to 31 MPa. Some bacteria are extremely halophilic, living in a salt concentration of 30% (parts per billion). Bacteria that increase the corrosion process are observed to have good growth at a temperature range of 15 °C to 45 °C and pHs of 6–8 [30].
Bacteria have the ability to accumulate on a metal surface and establish a biofilm, which contains live and dead bacterial cells, extracellular polymeric substances (EPS), corrosion products, as well as organic and inorganic debris. The percentage of the corrosion products and EPS in a biofilm is about 75%–95%, while 5%–25% of a biofilm is made up of bacterial cells [31,32]. Biofilm formation leads to gradients of pH and oxygen concentration at the metal/biofilm interface, resulting in severe localized corrosion attacks, such as pitting, crevices, and stress corrosion cracking (Figure 2) [33,34].

Figure 2.
SEM micrographs of carbon steel inoculated with Desulfovibrio alaskensis (a) and Desulfovibrio desulfuricans (b). Reprinted with permission from ref. [34]. Copyright 2023 Elsevier.
Based on oxygen use, bacteria are further divided into aerobic bacteria, anaerobic bacteria, facultative anaerobe, and micro-aerophilic bacteria. According to the published research, bacteria live both in aerobic and anaerobic environments and have the ability to enhance the bio-deterioration of metallic and non-metallic materials; however, it has been reported that anaerobic corrosion is more destructive compared to aerobic corrosion [35]. Anaerobic bacteria, including iron-reducing bacteria (IRB), acid-producing bacteria (APB), iron- and manganese-oxidizing bacteria (IOB, MOB), and sulphate-reducing bacteria (SRB), are reported to have detrimental effects on the properties of metals. Among them, SRB have been considered to play a key role in the process of corrosion [36,37]. SRB usually use SO42− as a terminal electron acceptor for energy generation in their metabolism, which indirectly contributes to the accumulation of corrosive sulphide and organic acid end-products, causing localized pitting corrosion of metals. Different steel alloys have been reported with severe corrosion rates in the presence of SRB [31,38]. The black-coloured sulphide corrosion products usually found at the localized corrosion sites of ferrous alloys indicate the presence of SRB [39]. According to the cathodic depolarization theory, SRB utilize hydrogen produced by the reduction of protons to reduce sulphate. Hydrogen consumption by SRB leads to an imbalance and encourages proton reduction, resulting in an increased oxidation of the steel surface by an oxidation reaction.
The mechanism involved is given as follows:
The iron sulphide (FeS) formed during this process works as a catalyst, promoting the reduction of protons on the material surface, which increases electron transfer, resulting in fast dissolution of the metal. Gu et al. proposed another theory called the bio-catalytic cathodic sulphate reduction (BCSR) theory [40]. The BCSR theory postulates that in MIC caused by SRB, sulphate works as a cathode while iron works as an anode. With the help of a biocatalyst, sulphate consumes the electrons released during iron oxidation and becomes reduced [40,41]. According to this theory, the following anodic and cathodic reactions are involved in MIC caused by an SRB biofilm:
Damaging effects of SRB on different metallic alloys ranging from iron alloys to titanium alloys have been reported [42,43,44,45]. Different researchers have proposed different theories to describe the role of SRB in the process of microbial corrosion. It has been stated that SRB consume hydrogen during their metabolism and accelerate cathodic reactions [46]. Silva et al. reported that the cathodic reactions on the surfaces of carbon steel and stainless steel were highly catalysed by hydrogenase enzymes [47,48]. In another study, Dihn et al. stated that SRB obtain electrons from the iron surface when zero-valent iron is available as a single electron donor [49].
IOB, also called metal oxidizing bacteria, are referred to as causing MIC [50,51]. The synergistic effect of aerobic and anaerobic bacteria is more aggressive than a single bacterial species. A community of aerobic IOB and anaerobic SRB severely increased the corrosion rate of X65 steel in a simulated marine environment [52].
3. Fungi
Fungi are eukaryotic organisms, such as yeast, moulds, and some well-known mushrooms. Contrary to bacteria, fungi are heterotrophs that secrete digestive enzymes into the surrounding environment and absorb dissolved molecules for their nutrition. Fungi can survive and grow in an environment highly deficient in water. Two species of this group, Aspergillus and Penicillium, have been found to tolerate very high and low pHs (above 12 and below 2) in their environment [53].
The involvement of fungi in MIC has been reported. Alekhova et al. found degradation of aluminium alloys by fungi-induced MIC on the Mir Space Station [54]. Carbon steel and aluminium alloys exposed to hydrocarbon fuel have been found to have an increased corrosion rate caused by fungi [55]. Videla et al. clarified the MIC mechanism in a fuel/water system as follows: (1) organic acidic metabolites locally increase the proton concentration, (2) metabolites of microorganisms decrease the surface energy of the interface passive oxides film to electrolytes, (3) the presence of microorganisms increases the oxidizing properties of the medium, thus increasing the chances of pitting corrosion, (4) microorganisms utilize corrosion inhibitors from the medium, and (5) adhesion of microbes speeds up the dissolution of metals [55]. Differential aeration cells created by fungi on the metal surface have been reported to enhance the corrosion process [55].
Metabolically, fungi are highly diverse microorganisms that are able to obtain nutrition from the degradation of various organic materials, such as polymeric organic compounds and hydrocarbons. Some fungi produce organic compounds, including organic acids and complexants, which affect the properties of metals [56,57]. Fungus-induced degradation of the coatings and underlying metals has been reported in several studies [58,59]. Stranger-Johannessen reported fungal degradation of ship cargo coated with chlorinated rubber. In another study, the deterioration of ship holds coated with epoxy resin and filled with molasses and fatty oils, as well as other fluid cargoes, was reported by Stranger-Johannessen [60]. In addition, Stranger-Johannessen investigated and confirmed the fungal-induced corrosion of polyurethane cable sheathing in a marine environment [60].
It has been stated that fungal-induced corrosion of metallic materials and coatings is associated with the production of organic acids [61]. Fungal degradation of aluminium has attracted more attention from researchers due to its impact on the integrity of aircraft [62]. Damage to aircraft integrity caused by fungal biofilms has been reported on several occasions [62]. Fungi degrade organic materials, such as lubricants, cladding, and jet fuel, and generate organic acids [62,63,64,65]. Fungi usually produce a wide range of organic acids in an aerobic environment, while most bacteria generate organic acids in an anaerobic environment [66]. Most of these acids are corrosive, causing the deterioration of different materials. For example, it has been reported that formic acid and acetic acid highly increased the corrosion of radioactive waste containers made of carbon steel [67].
4. Algae
Algae are unicellular aquatic microorganisms and are able to produce their own food by the process of photosynthesis. Algae are not closely related to each other in an evolutionary sense. For instance, they can live as single-cell microscopic algae or can be found in macroscopic and multicellular forms, which exist in the form of a colony.
Besides their adverse effects on water quality, these microorganisms considerably influence the corrosion process of engineering materials in marine environments [68]. Figure 3 represents different diatoms attached to the surface of stainless steel. The attachment of single-cell algae (diatoms) to stainless steel surfaces has been observed by different researchers [69,70,71,72,73]. It was found that the colonization of diatoms on a stainless steel surface was more active and fast in the light compared to the dark [74]. The process of photosynthesis plays a key role in enhancing corrosion by changing the surface state (such as dissolved oxygen and pH) of metallic materials [75,76]. Degradation due to algal biofilms has been reported for metallic materials. For instance, the corrosion process of Q235 steel was highly increased in the presence of Chlorella vulgaris, which is an algae species [68].

Figure 3.
Diatoms attached to a SS surface exposed to a natural river (Oise, France), where (a) represents the presence of Cocconeis, (b) represents the presence of Cymbella sp., while (c) represents Amphora sp. Reprinted with permission from ref. [75]. Copyright 2023 Taylor & Francis.
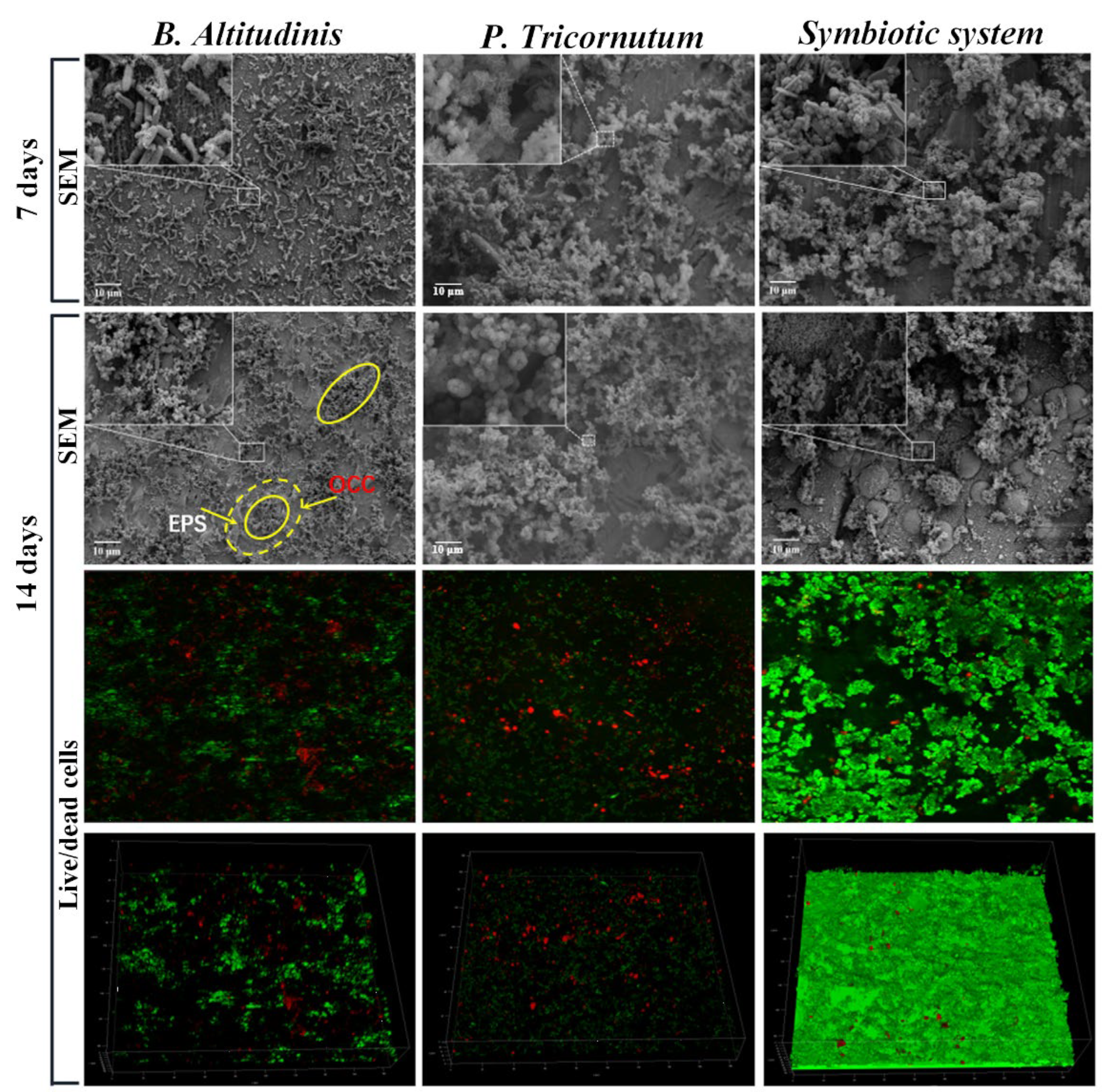
Furthermore, through ecological studies, it has been specified that algae and bacteria living together in a fouled part of the material maintain an extremely close association with each other [77,78]. A study on symbiosis-induced biofouling in a marine micro-fouling system, where bacterial biofilms form the underlying layer and microalgae work as the elementary biofouling layer, has been reported [79]. It was found that the corrosion rate of carbon steel (Q235) immersed in a culture medium inoculated with the bacterium Bacillus altitudinis was 2.2 times higher than that in a sterile medium. Meanwhile, the corrosion rate of Q235 steel in the presence of both Phaeodactylum tricornutum and its symbiotic bacterium B. altitudinis was about 7 times higher compared to the effects of the individual bacterial strain.
Microalgae have been found to secrete EPS, which further triggers the corrosion process by complexation with metals [68]. Multispecies biofilms, as shown in Figure 4, are able to form stable micro-consortia that strengthen the three-dimensional structure of the adhesive layer and accelerate biofouling. Owing to the combined effect of the symbiotic system, the anodic part underneath the biofilms, which lacks oxygen and is abundant in EPS, would continue dissolving. This further enhances the oxygen concentration cell (OCC) corrosion of the metal, leading to an increased accumulation of corrosion products on the material surface [80].
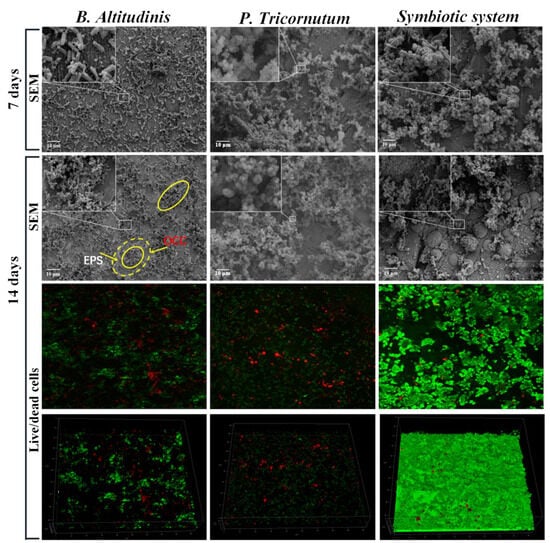
Figure 4.
Biofilm morphologies on the surface of Q235 steel immersed in B. altitudinis, P. tricornutum, and a symbiotic system for 7 and 14 days. Here, the small square areas linked with the insets represent that the insets are the magnified pictures of these areas. The solid yellow circle represents the biofilm, while the dotted yellow circle with a small solid circle in its centre represents the biofilm, the EPS and the oxygen concentration cell. Reprinted with permission from ref. [79]. Copyright 2023 Elsevier.
The reactions involved in this process are given as follows [79]:
5. Archaea
Archaea are a group of microorganisms originally believed to be bacteria and called archaebacteria owing to their physical similarities. But later, through genetic analysis, it was found that archaea are different organisms from bacteria and eukaryotes. This analysis earned them their own domain in the three domain classification originally proposed by Woese in 1977, alongside the eukaryotes and the bacteria [81]. In addition to bacteria, archaea are also an important part of the microbial system [82].
Archaea are broadly distributed in the world. The majority of archaea have the ability to inhabit and thrive in some extreme environments, such as those with enormously low oxygen levels, high acidity, high salinity, and very high temperatures, which provide archaea with distinctive cell structures and metabolic characteristics [83].
Archaea have been found to cause MIC of metallic materials [84,85]. For instance, the presence of methanogenic and thermophilic archaea has been reported in high-temperature, anaerobic oil production fluids collected from the North Sea and North Slope of Alaskan oil fields [86]. Both the methanogenic and thermophilic archaea found in the above-mentioned locations were reported to have corrosion triggering effects [87,88]. It was stated that the methanogenic archaea (Methanothermobacter sp.) used carbon steel as an energy source and accelerated its corrosion process, while the thermophilic archaea (Thermococcales sp.) enhanced carbon steel degradation through its iron reduction ability as well as the secretion of fatty acid metabolites. Furthermore, Usher et al. observed the colonization and corrosive effects of methanogenic archaeal communities on a carbon steel surface [89]. Moreover, it has been noted that halophilic archaea such as Natronorubrum tibetense, which inhabits alkali soil with a high level of salinity, is capable of using iron in carbon steel and stainless steel as a source of energy to promote their corrosion processes [90,91]. Carbon steel Q235 and stainless steel 304 were investigated during these studies, and it was noted that samples immersed in archaea-inoculated media possessed weak linear polarization resistance, increased pitting corrosion, and high corrosion current density, indicating that archaea significantly deteriorated the materials.
Methane-producing microbes trigger the corrosion process of iron-containing metals. H2 has been considered as an electron shuttle between Fe(0) and methanogens. Some of the methanogens, such as the Methanosarcina acetivorans, catalyse direct electron transfer from metal-to-microbe to support methane production [92]. In M. acetivorans, deletion of the gene for multiheme eliminated methane production from Fe(0) by the outer-surface c-type cytochrome MmcA, which is consistent with the basic role of MmcA in other forms of extracellular electron transfer. Dawn et al. proposed that these findings, together with the earlier statement that outer surface c-type cytochromes also work as electron carriers between Fe(0) and microbes, such as Geobacter and Shewanella species, suggest that the presence of multiheme c-type cytochromes on corrosion surfaces might be indicative of direct metal-to-microbe electron transfer and that deactivating the cytochrome function could be an important strategy to inhibit corrosion.
As mentioned before, thermophilic archaea can survive at high temperatures (60–80 °C) [93]. A thermophilic sulphate-reducing archaeon called Archaeoglobus fulgidus was cultivated on the surface of C1018 carbon steel at 80 °C under organic carbon starvation. C1018 carbon steel immersed in archaea-inoculated medium for 7 days was observed to have more sessile cell accumulation on its surface, which aggravated the material, leading to a high corrosion rate and increased pitting corrosion (Figure 5) [84]. It was stated that the EET-MIC mechanism could apply to the corrosion of C1018 carbon steel caused by A. fulgidus, where carbon source starvation forced A. fulgidus sessile cells to start utilizing extracellular elemental iron for energy production. Based on this study, it was concluded that SRB and NRB sulphate-reducing archaea have the capability to use elemental iron as an electron donor to obtain energy for their metabolic activities.

Figure 5.
Pitting corrosion of abiotic control material after 10-day incubation in sterile enriched artificial seawater (EASW) medium with a 100% carbon source (A,A’) and biotic coupons in archaea-inoculated EASW with 100% availability of a carbon source after a 3-day pre-growth (B,B’), and biotic coupons after 7 days of incubation in EASW with 0% (C,C′), 90% (D,D’), and 100% (E,E’) carbon source reductions. Reprinted with permission from ref. [84]. Copyright 2023 Elsevier.
6. Lichens
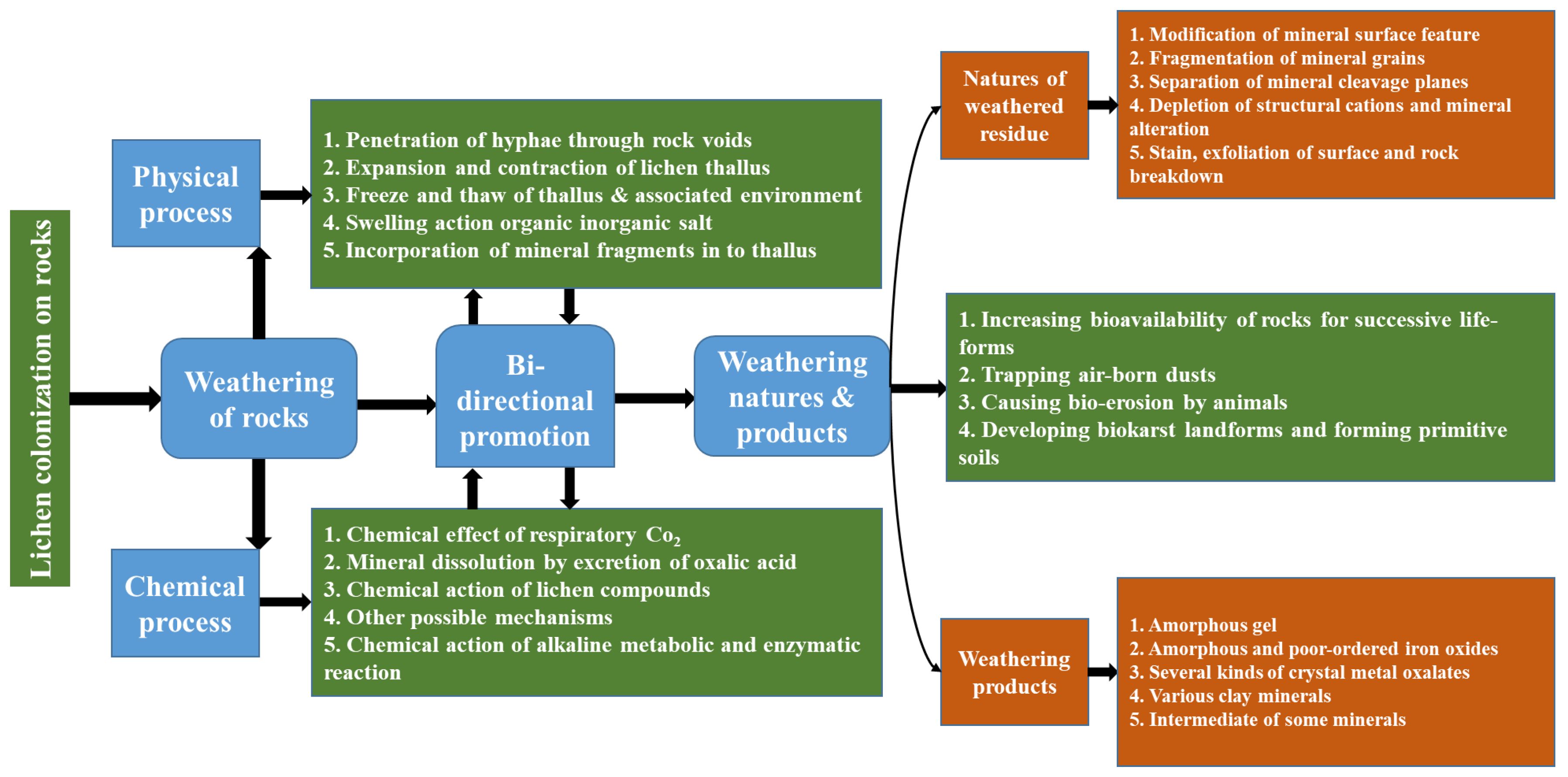
A lichen is actually two organisms working as a single stable unit. Lichens are plant-like organisms that consist of a symbiotic association of algae or cyanobacteria and fungi. Lichens have about 20,000 known species worldwide that have been found surviving in different environmental conditions. This is a diverse group of organisms, having the ability to colonize a wide range of surfaces, including tree bark, exposed rock, biological soil crust, and other metallic and non-metallic materials in various environments. Through metabolism, lichens discharge different kinds of organic molecules, such as oxalic acid and polyphenolic acids, indicated as “lichen acids”, that have been confirmed to play a vital role in weathering and neogenesis [94].
The deterioration caused by lichens occurs at the interface between the lichen and metal substrate. This interface has been considered a place of significant physical and chemical activities, presenting a very complex heterogeneity in which both primary and secondary minerals, organic acids, and compounds, as well as all kinds of organisms, including lichens, free-living fungi, free-living algae, and bacteria, are involved [95]. This phenomenon is happening on all kinds of substrates, whether natural or man-made. In the case of metal, lichen can directly affect the surface or affect the antirust paint applied to the surface of the metal, thus contributing to the oxidation process.
The deterioration of ceramics due to lichen development on their surfaces has been reported [96]. It has been stated that the oxalic acid released by lichens was the main reason for ceramic deterioration and aging [96]. The deteriorating effects of lichens on natural rocks and building stones have been recognized long before [97,98]. The mycobiont of lichens, which is always in close contact with the substrate, makes them able to cause deterioration. Bio-deterioration by lichens is, in general, attributed to a combination of physical mechanisms (such as the pressure exerted by the expansion and contraction of thalli, rhizine adhesion, and hyphal penetration) and chemical factors, which include the interaction of carbon dioxide, organic acids, and lichen substances with complex properties [99]. The effects of lichens on the host substrate are shown in Figure 6 [95].
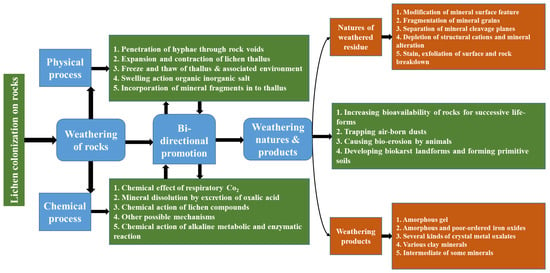
Figure 6.
The effects of lichens on the substrate. Reprinted with permission from ref. [95]. Copyright 2023 Elsevier.
7. Conclusions
MIC has been considered a major threat to material applications in various fields. However, until now, most researchers only considered bacteria as a key player involved in MIC. According to this short review, there are a number of microorganisms, such as bacteria, fungi, algae, archaea, and lichens, that are directly or indirectly participating in the process of MIC. Therefore, it is highly important to identify the microorganisms actually involved in the process of corrosion in the real environment. In addition, genetic manipulation of corrosion-causing microorganisms is highly important to better understand the process and mechanisms of MIC.
Author Contributions
Conceptualization, M.S.K.; methodology, Z.L.; validation, L.Z.; data curation, W.L.; formal analysis, S.L.; writing original draft preparation, X.W.; writing review and editing, K.Y.; supervision, C.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the Natural Science Foundation of China (No. U1906226) and Application Technology of Antibacterial Stainless Steel (RCZX2023001).
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- González, J.E.G.; Santana, F.J.H.; Mirza-Rosca, J.C. Effect of bacterial biofilm on 316 SS corrosion in natural seawater by EIS. Corros. Sci. 1998, 40, 2141–2154. [Google Scholar] [CrossRef]
- Gaines, H.A. Bacterial activity as a corrosion induced in the soil. J. Eng. Ind. Chem. 1910, 2, 128–130. [Google Scholar] [CrossRef]
- Telegdi, J.; Trif, L.; Roma, L. Smart anti-biofouling composite coatings for naval applications. In Smart Composite Coatings and Membranes; Elsevier: Amsterdam, The Netherlands, 2016; pp. 123–155. [Google Scholar] [CrossRef]
- Bessone, J.B.; Baldo, R.A.S.; de de Micheli, S.M. Sea water testing OF Al-Zn, Al-Zn-Sn, and Al-Zn-ln sacrificial anodes. Corrosion 1981, 37, 533–540. [Google Scholar] [CrossRef]
- Vu, B.; Chen, M.; Crawford, R.J.; Ivanova, E.P. Bacterial extracellular polysaccharides involved in biofilm formation. Molecules 2009, 14, 2535–2554. [Google Scholar] [CrossRef]
- Costerton, J.; Lewandownski, Z.; Caldwell, D.; Korber, D. Lappin-Scott H Microbial Biofilms. Annu. Rev. Microbiol. 1995, 49, 711–745. [Google Scholar] [CrossRef]
- Khan, M.S.; Liang, T.; Liu, Y.; Shi, Y.; Zhang, H.; Li, H.; Guo, S.; Pan, H.; Yang, K.; Zhao, Y. Microbiologically Influenced Corrosion Mechanism of Ferrous Alloys in Marine Environment. Metals 2022, 12, 1458. [Google Scholar] [CrossRef]
- Zobell, C.E. The Effect of Solid Surfaces upon Bacterial Activity. J. Bacteriol. 1943, 46, 39–56. [Google Scholar] [CrossRef]
- Beyenal, H.; Lewandowski, Z. Internal and external mass transfer in biofilms grown at various flow velocities. Biotechnol. Prog. 2002, 18, 55–61. [Google Scholar] [CrossRef]
- Allan, V.J.M.; Callow, M.E.; Macaskie, L.E.; Paterson-beedle, M.; Citrobacter, A. Effect of nutrient limitation on biofilm formation and phosphatase activity of a Citrobacter sp. Microbiology 2002, 148, 277–288. [Google Scholar] [CrossRef] [PubMed]
- Verderosa, A.D.; Totsika, M.; Fairfull-Smith, K.E. Bacterial Biofilm Eradication Agents: A Current Review. Front. Chem. 2019, 7, 824. [Google Scholar] [CrossRef]
- Saleem Khan, M.; Xu, D.; Liu, D.; Lekbach, Y.; Yang, K.; Yang, C. Corrosion Inhibition of X80 Steel in Simulated Marine Environment with Marinobacter aquaeolei. Acta Metall. Sin. 2019, 32, 1373–1384. [Google Scholar] [CrossRef]
- Yuan, S.J.; Pehkonen, S.O. AFM study of microbial colonization and its deleterious effect on 304 stainless steel by Pseudomonas NCIMB 2021 and Desulfovibrio desulfuricans in simulated seawater. Corros. Sci. 2009, 51, 1372–1385. [Google Scholar] [CrossRef]
- Miranda, E.; Bethencourt, M.; Botana, F.J.; Cano, M.J.; Sánchez-Amaya, J.M.; Corzo, A.; de Lomas, J.G.; Fardeau, M.L.; Ollivier, B. Biocorrosion of carbon steel alloys by an hydrogenotrophic sulfate-reducing bacterium Desulfovibrio capillatus isolated from a Mexican oil field separator. Corros. Sci. 2006, 48, 2417–2431. [Google Scholar] [CrossRef]
- McNamara, C.J.; Perry, T.D., IV; Bearce, K.A.; Hernandez-Duque, G.; Mitchell, R. Epilithic and endolithic bacterial communities in limestone from a Maya archaeological site. Microb. Ecol. 2006, 51, 51–64. [Google Scholar] [CrossRef] [PubMed]
- McNamara, C.J.; Perry, T.D., IV; Bearce, K.; Hernandez-Duque, G.; Mitchell, R. Measurement of limestone biodeterioration using the Ca2+ binding fluorochrome Rhod-5N. J. Microbiol. Methods 2005, 61, 245–250. [Google Scholar] [CrossRef]
- Rao, T.S. Microbial Fouling and Corrosion: Fundamentals and Mechanisms. In Operational and Environmental Consequences of Large Industrial Cooling Water Systems; Springer: Boston, MA, USA, 2012; ISBN 9781461416982. [Google Scholar]
- Rouvre, I.; Gauquelin, C.; Meynial-salles, I.; Basseguy, R. Impact of the chemicals, essential for the puri fi cation process of strict Fe-hydrogenase, on the corrosion of mild steel. Bioelectrochemistry 2016, 109, 9–23. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Xu, D.; Jia, R.; Li, Y.; Gu, T. Advances in the treatment of problematic industrial biofilms. World J. Microbiol. Biotechnol. 2017, 33, 97. [Google Scholar] [CrossRef]
- Kilbane, J.J. Determining the source of H2S on an offshore oil production platform. In Microbiologically Influenced Corrosion in the Upstream Oil and Gas Industry; CRC Press: Boca Raton, FL, USA, 2017; pp. 351–360. ISBN 9781498726603. [Google Scholar]
- Zhao, Y.; Zhou, E.; Liu, Y.; Liao, S.; Li, Z.; Xu, D.; Zhang, T.; Gu, T. Comparison of different electrochemical techniques for continuous monitoring of the microbiologically influenced corrosion of 2205 duplex stainless steel by marine Pseudomonas aeruginosa biofilm. Corros. Sci. 2017, 126, 142–151. [Google Scholar] [CrossRef]
- Sabel, C.F.; Victor, D.G. Governing global problems under uncertainty: Making bottom-up climate policy work. Clim. Chang. 2017, 144, 15–27. [Google Scholar] [CrossRef]
- Videla, H.A. Prevention and control of biocorrosion. Int. Biodeterior. Biodegrad. 2002, 49, 259–270. [Google Scholar] [CrossRef]
- Jacobson, G. Corrosion at Prudhoe Bay—A lesson on the line. Mater. Perform. 2007, 46, 26–34. [Google Scholar]
- Bhat, S.; Kumar, B.; Prasad, S.R.; Katarki, M.V. Failure of a new 8-in pipeline from group gathering station to central tank farm. Mater. Perform. 2011, 50, 50–54. [Google Scholar]
- Walsh, D.; Pope, D.; Danford, M.; Huff, T. The effect of microstructure on microbiologically influenced corrosion. J. Miner. Met. Mater. Soc. 1993, 45, 22–30. [Google Scholar] [CrossRef]
- Little, B.J.; Lee, J.S. Microbiologically Influenced Corrosion. In Oil and Gas Pipelines; John Wiley & Sons: Hoboken, NJ, USA, 2015; Volume 2, ISBN 9781119019213. [Google Scholar]
- Javaherdashti, R.; Alasvand, K. An Introduction to Microbial Corrosion. In Biological Treatment of Microbial Corrosion; Elsevier: Amsterdam, The Netherlands, 2019; pp. 25–70. ISBN 9780128161081. [Google Scholar]
- Fredrickson, J.K.; Zachara, J.M.; Balkwill, D.L.; Kennedy, D.; Li, S.M.W.; Kostandarithes, H.M.; Daly, M.J.; Romine, M.F.; Brockman, F.J. Geomicrobiology of high-level nuclear waste-contaminated vadose sediments at the Hanford Site, Washington State. Appl. Environ. Microbiol. 2004, 70, 4230–4241. [Google Scholar] [CrossRef]
- Metals Handbook, 9th ed.; ASM International: Metals park, OH, USA, 1987; Volume 13.
- Alabbas, F.M.; Williamson, C.; Bhola, S.M.; Spear, J.R.; Olson, D.L.; Mishra, B.; Kakpovbia, A.E. Microbial corrosion in linepipe steel under the influence of a sulfate-reducing consortium isolated from an oil field. J. Mater. Eng. Perform. 2013, 22, 3517–3529. [Google Scholar] [CrossRef]
- Videla, H.A.; Herrera, L.K. Microbiologically influenced corrosion: Looking to the future. Int. Microbiol. 2005, 8, 169–180. [Google Scholar]
- Shahryari, Z.; Gheisari, K.; Motamedi, H. Corrosion behavior of API X70 microalloyed pipeline steel in a simulated soil solution in the absence and presence of aerobic Pseudomonas species. Mater. Res. Express 2019, 6, 065409. [Google Scholar] [CrossRef]
- Stipaničev, M.; Turcu, F.; Esnault, L.; Schweitzer, E.W.; Kilian, R.; Basseguy, R. Corrosion behavior of carbon steel in presence of sulfate-reducing bacteria in seawater environment. Electrochim. Acta 2013, 113, 390–406. [Google Scholar] [CrossRef]
- El Hajj, H.; Abdelouas, A.; El Mendili, Y.; Karakurt, G.; Grambow, B.; Martin, C. Corrosion of carbon steel under sequential aerobic-anaerobic environmental conditions. Corros. Sci. 2013, 76, 432–440. [Google Scholar] [CrossRef]
- Lee, W.; Characklis, W.G. Corrosion of mild steel under anaerobic biofilm. Corrosion 1993, 49, 186–199. [Google Scholar] [CrossRef]
- Lee, W.; Lewandowski, Z.; Nielsen, P.H.; Allan Hamilton, W. Role of sulfate-reducing bacteria in corrosion of mild steel: A review. Biofouling 1995, 8, 165–194. [Google Scholar] [CrossRef]
- Chen, S.; Wang, P.; Zhang, D. Corrosion behavior of copper under biofilm of sulfate-reducing bacteria. Corros. Sci. 2014, 87, 407–415. [Google Scholar] [CrossRef]
- Marchal, R. Rôle des bactéries sulfurogènes dans la corrosion du fer. Oil Gas Sci. Technol. 1999, 54, 649–659. [Google Scholar] [CrossRef]
- Gu, T.; Zhao, K.; Nesic, S. A New Mechanistic Model for Mic Based on A Biocatalytic Cathodic Sulfate Reduction Theory. In Proceedings of the Corrosion 2009, Atlanta, GA, USA, 22–26 March 2009; pp. 1–12. [Google Scholar]
- Zhao, K. Investigation of Microbiologically Influenced Corrosion (Mic) and Biocide Treatment in Anaerobic Salt Water and Development of a Mechanistic Mic Model. Ph.D. Thesis, Ohio University, Athens, OH, USA, 2008. [Google Scholar]
- Chen, J.; Wu, J.; Wang, P.; Zhang, D.; Chen, S.; Tan, F. Correction to: Corrosion of 907 Steel Influenced by Sulfate-Reducing Bacteria. J. Mater. Eng. Perform. 2019, 28, 5913. [Google Scholar] [CrossRef]
- Fang, S.J.; Liu, Y.H.; Wang, Q.; Yu, S.R.; Song, Y.L. Influence of SRB on corrosion of AZ91 magnesium alloy in solution containing chlorine ions. Huanan Ligong Daxue Xuebao/J. South China Univ. Technol. (Nat. Sci.) 2008, 36, 92–96. [Google Scholar]
- Guan, F.; Zhai, X.; Duan, J.; Zhang, J.; Li, K.; Hou, B. Influence of sulfate-reducing bacteria on the corrosion behavior of 5052 aluminum alloy. Surf. Coatings Technol. 2017, 316, 171–179. [Google Scholar] [CrossRef]
- Zheng, X.; Zhuang, X.; Lei, Y.; Chu, Z.; Xu, J.; Gao, L.; Sun, X. Corrosion behavior of the Ti-6Al-4V alloy in sulfate-reducing bacteria solution. Coatings 2020, 10, 24. [Google Scholar] [CrossRef]
- Costello, J.A. Cathodic depolarization by sulphate reducing bacteria. S. Afr. J. Sci. 1974, 70, 202–204. [Google Scholar]
- Silva, D.; Basséguy, R.; Bergel, A. The role of hydrogenases in the anaerobic microbiologically influenced corrosion of steels. Bioelectrochemistry 2002, 56, 77–79. [Google Scholar] [CrossRef]
- Da Silva, S.; Basséguy, R.; Bergel, A. Electrochemical deprotonation of phosphate on stainless steel. Electrochim. Acta 2004, 49, 4553–4561. [Google Scholar] [CrossRef]
- Dinh, H.T.; Kuever, J.; Mußmann, M.; Hassel, A.W.; Stratmann, M.; Widdel, F. Iron corrosion by novel anaerobic microorganisms. Nature 2004, 427, 829–832. [Google Scholar] [CrossRef] [PubMed]
- Yuan, S.J.; Pehkonen, S.O. Microbiologically influenced corrosion of 304 stainless steel by aerobic Pseudomonas NCIMB 2021 bacteria: AFM and XPS study. Colloids Surf. B Biointerfaces 2007, 59, 87–99. [Google Scholar] [CrossRef] [PubMed]
- Lekbach, Y.; Liu, T.; Li, Y.; Moradi, M.; Dou, W.; Xu, D.; Smith, J.A.; Lovley, D.R. Microbial corrosion of metals: The corrosion microbiome. Adv. Microb. Physiol. 2021, 78, 317–390. [Google Scholar]
- Lv, M.; Du, M.; Li, X.; Yue, Y.; Chen, X. Mechanism of microbiologically influenced corrosion of X65 steel in seawater containing sulfate-reducing bacteria and iron-oxidizing bacteria. J. Mater. Res. Technol. 2019, 8, 4066–4078. [Google Scholar] [CrossRef]
- Ghali, E. Corrosion Resistance of Al and Mg Alloys; Wiley: Hoboken, NJ, USA, 2010; ISBN 9780471715764. [Google Scholar]
- Alekhova, T.A.; Aleksandrova, A.A.; Novozhilova, T.Y.; Lysak, L.V.; Zagustina, N.A.; Bezborodov, A.M. Monitoring of microbial degraders in manned space stations. Appl. Biochem. Microbiol. 2005, 41, 382–389. [Google Scholar] [CrossRef]
- Little, B.J.; Ray, R.I. The Role of Fungi in Microbiologically Influenced Corrosion. In Proceedings of the 15th International Corrosion Congress, Granada, Spain, 22–27 September 2002. [Google Scholar]
- Gadd, G.M. Metals, minerals and microbes: Geomicrobiology and bioremediation. Microbiology 2010, 156, 609–643. [Google Scholar] [CrossRef]
- Gadd, G.M. Geomycology: Biogeochemical transformations of rocks, minerals, metals and radionuclides by fungi, bioweathering and bioremediation. Mycol. Res. 2007, 111, 3–49. [Google Scholar] [CrossRef]
- Webb, J.S.; Nixon, M.; Eastwood, I.M.; Greenhalgh, M.; Robson, G.D.; Handley, P.S. Fungal colonization and biodeterioration of plasticized polyvinyl chloride. Appl. Environ. Microbiol. 2000, 66, 3194–3200. [Google Scholar] [CrossRef]
- There, I.; Polymers, E.; Pcl, F.; Corporation, U.C.; Brook, B. Scanning electron microscopic visualization of biodegradation of polycaprolactones by fungi. J. Polym. Sci. Polym. Lett. Ed. 1981, 19, 159–165. [Google Scholar]
- Date, R.; Use, A.; Leave, O.; No, J.O.; Little, B.; Ray, R. Fungal Influenced Corrosion of Metals in Humid Environments. NACE Int. Corros. Soc. 1999, 298, 1–6. [Google Scholar]
- Lugauskas, A.; Demčenko, I.; Selskiene, A.; Pakštas, V.; Jaskelevičius, B.; Narkevičius, A.; Bučinskiene, D. Resistance of chromated zinc coatings to the impact of microscopic fungi. Medziagotyra 2011, 17, 20–26. [Google Scholar] [CrossRef]
- Lee, J.S.; Ray, R.I.; Little, B.J. Microbiologically Influenced Corrosion in Military Environments. In Corrosion: Environments and Industries; ASM International: Materials Park, OH, USA, 2018. [Google Scholar]
- Little, B.; Staehle, R.; Davis, R. Fungal influenced corrosion of post-tensioned cables. Int. Biodeterior. Biodegrad. 2001, 47, 71–77. [Google Scholar] [CrossRef]
- McNamara, C.; Perry, T.; Leard, R.; Bearce, K.; Dante, J.; Mitchell, R. Corrosion of aluminum alloy 2024 by microorganisms isolated from aircraft fuel tanks. Biofouling 2005, 21, 257–265. [Google Scholar] [CrossRef] [PubMed]
- Rauch, M.E.; Graef, H.W.; Rozenzhak, S.M.; Jones, S.E.; Bleckmann, C.A.; Kruger, R.L.; Naik, R.R.; Stone, M.O. Characterization of microbial contamination in United States Air Force aviation fuel tanks. J. Ind. Microbiol. Biotechnol. 2006, 33, 29–36. [Google Scholar] [CrossRef]
- Plassard, C.; Fransson, P. Regulation of low-molecular weight organic acid production in fungi. Fungal Biol. Rev. 2009, 23, 30–39. [Google Scholar] [CrossRef]
- Humphreys, P. The Potential Role of Fungi and Algae in the Atmospheric Corrosion of Waste Packages in Interim Storage and During the Operational Period of a GDF. Nuclear Decommissioning Authority: Oxfordshire, UK, 2012; pp. 1–40. [Google Scholar]
- Liu, H.; Xu, D.; Dao, A.Q.; Zhang, G.; Lv, Y.; Liu, H. Study of corrosion behavior and mechanism of carbon steel in the presence of Chlorella vulgaris. Corros. Sci. 2015, 101, 84–93. [Google Scholar] [CrossRef]
- Scotto, V.; Alabiso, G.; Marcenaro, G. An example of microbiologically influenced corrosion: The Behaviour of Stainless Steels in Natural Seawater. Bioelectrochemistry 1986, 212, 325–331. [Google Scholar]
- Mattila, K.; Carpen, L.; Hakkarainen, T.; Salkinoja-Salonen, M.S. Biofilm development during ennoblement of stainless steel in Baltic Sea water: A microscopic study. Int. Biodeterior. Biodegrad. 1997, 40, 1–10. [Google Scholar] [CrossRef]
- Videla, H.A. Biofilms and corrosion interactions on stainless steel in seawater. Int. Biodeterior. Biodegrad. 1994, 34, 245–257. [Google Scholar] [CrossRef]
- Mansfeld, F.; Liu, G.; Xiao, H.; Tsai, C.H.; Little, B.J. The corrosion behavior of copper alloys, stainless steels and titanium in seawater. Corros. Sci. 1994, 36, 2063–2095. [Google Scholar] [CrossRef]
- Motoda, S.; Suzuki, Y.; Shinohara, T.; Tsujikawa, S. The effect of marine fouling on the ennoblement of electrode potential for stainless steels. Corros. Sci. 1990, 31, 515–520. [Google Scholar] [CrossRef]
- Cooksey, B.; Cooksey, K.E. Calcium is Necessary for Motility in the Diatom Amphora coffeaeformis. Plant Physiol. 1980, 65, 129–131. [Google Scholar] [CrossRef] [PubMed]
- Landoulsi, J.; Cooksey, K.E.; Dupres, V. Review—Interactions between diatoms and stainless steel: Focus on biofouling and biocorrosion. Biofouling 2011, 27, 1105–1124. [Google Scholar] [CrossRef]
- Edyvean, R.; Evans, L.V.; Hoagland, K.D. Algal Biofouling. J. Ecol. 1987, 28, 1206–1207. [Google Scholar] [CrossRef]
- Christie-Oleza, J.A.; Sousoni, D.; Lloyd, M.; Armengaud, J.; Scanlan, D.J. Nutrient recycling facilitates long-term stability of marine microbial phototroph-heterotroph interactions. Nat. Microbiol. 2017, 2, 17100. [Google Scholar] [CrossRef] [PubMed]
- Ji, X.; Jiang, M.; Zhang, J.; Jiang, X.; Zheng, Z. The interactions of algae-bacteria symbiotic system and its effects on nutrients removal from synthetic wastewater. Bioresour. Technol. 2018, 247, 44–50. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Song, G.L.; Zhang, J.; Gao, Y.; Wang, Z.M.; Zheng, D. Biocorrosion induced by red-tide alga-bacterium symbiosis and the biofouling induced by dissolved iron for carbon steel in marine environment. J. Mater. Sci. Technol. 2022, 128, 107–117. [Google Scholar] [CrossRef]
- Guzman, M.S.; Rengasamy, K.; Binkley, M.M.; Jones, C.; Ranaivoarisoa, T.O.; Singh, R.; Fike, D.A.; Meacham, J.M.; Bose, A. Phototrophic extracellular electron uptake is linked to carbon dioxide fixation in the bacterium Rhodopseudomonas palustris. Nat. Commun. 2019, 10, 1355. [Google Scholar] [CrossRef]
- Woese, C.R.; Fox, G.E. Phylogenetic structure of the prokaryotic domain: The primary kingdoms (archaebacteria/eubacteria/urkaryote/16S ribosomal RNA/molecular phylogeny). Proc. Natl. Acad. Sci. USA 1977, 74, 5088–5090. [Google Scholar] [CrossRef]
- Woese, C.R.; Kandler, O.; Wheelis, M.L. Towards a natural system of organisms: Proposal for the domains Archaea, Bacteria, and Eucarya. Proc. Natl. Acad. Sci. USA 1990, 87, 4576–4579. [Google Scholar] [CrossRef]
- Cavicchioli, R. Archaea—Timeline of the third domain. Nat. Rev. Microbiol. 2011, 9, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Jia, R.; Yang, D.; Xu, D.; Gu, T. Carbon steel biocorrosion at 80 °C by a thermophilic sulfate reducing archaeon biofilm provides evidence for its utilization of elemental iron as electron donor through extracellular electron transfer. Corros. Sci. 2018, 145, 47–54. [Google Scholar] [CrossRef]
- Larsen, J.; Rasmussen, K.; Pedersen, H.; Sørensen, K.; Lundgaard, T. Consortia of MIC bacteria and archaea causing pitting corrosion in top side oil production facilities. In Proceedings of the Corrosion 2010, San Antonio, TX, USA, 14–18 March 2010. [Google Scholar]
- Stetter, K.O.; Huber, R.; Blöchl, E.; Kurr, M.; Eden, R.D.; Fielder, M.; Cash, H.; Vance, I. Hyperthermophilic archaea are thriving in deep North Sea and Alaskan oil reservoirs. Nature 1993, 365, 743–745. [Google Scholar] [CrossRef]
- Davidova, I.A.; Duncan, K.E.; Perez-Ibarra, B.M.; Suflita, J.M. Involvement of thermophilic archaea in the biocorrosion of oil pipelines. Environ. Microbiol. 2012, 14, 1762–1771. [Google Scholar] [CrossRef]
- Liang, R.; Grizzle, R.S.; Duncan, K.E.; McInerney, M.J.; Suflita, J.M. Roles of thermophilic thiosulfate-reducing bacteria and methanogenic archaea in the biocorrosion of oil pipelines. Front. Microbiol. 2014, 5, 89. [Google Scholar] [CrossRef] [PubMed]
- Usher, K.M.; Kaksonen, A.H.; MacLeod, I.D. Marine rust tubercles harbour iron corroding archaea and sulphate reducing bacteria. Corros. Sci. 2014, 83, 189–197. [Google Scholar] [CrossRef]
- Qian, H.; Ma, L.; Zhang, D.; Li, Z.; Huang, L.; Lou, Y.; Du, C. Microbiologically influenced corrosion of 304 stainless steel by halophilic archaea Natronorubrum tibetense. J. Mater. Sci. Technol. 2020, 46, 12–20. [Google Scholar] [CrossRef]
- Qian, H.; Zhang, D.; Lou, Y.; Li, Z.; Xu, D.; Du, C.; Li, X. Laboratory investigation of microbiologically influenced corrosion of Q235 carbon steel by halophilic archaea Natronorubrum tibetense. Corros. Sci. 2018, 145, 151–161. [Google Scholar] [CrossRef]
- Holmes, D.E.; Tang, H.; Woodard, T.; Liang, D.; Zhou, J.; Liu, X.; Lovley, D.R. Cytochrome-mediated direct electron uptake frommetallic iron by Methanosarcina acetivorans. mLife 2022, 1, 443–447. [Google Scholar] [CrossRef]
- Wang, Q.; Cen, Z.; Zhao, J. The survival mechanisms of thermophiles at high temperatures: An angle of omics. Physiology 2015, 30, 97–106. [Google Scholar] [CrossRef]
- Adamo, P.; Vingiani, S.; Violante, P. Lichen-rock interactions and bioformation of minerals. Dev. Soil Sci. 2002, 28, 377–391. [Google Scholar]
- Chen, J.; Blume, H.-P.; Beyer, L. Weathering of rocks induced by lichen colonization—A review. Catena 2000, 39, 121–146. [Google Scholar] [CrossRef]
- Radeka, M.; Ranogajec, J.; Kiurski, J.; Markov, S.; Marinković-Nedučin, R. Influence of lichen biocorrosion on the quality of ceramic roofing tiles. J. Eur. Ceram. Soc. 2007, 27, 1763–1766. [Google Scholar] [CrossRef]
- Seaward, M.R.D. Major impacts made by lichens in biodeterioration processes. Int. Biodeterior. Biodegrad. 1997, 40, 269–273. [Google Scholar] [CrossRef]
- Silva, B.; Prieto, B.; Rivas, T.; Sanchez-Biezma, M.J.; Paz, G.; Carballal, R. Rapid biological colonization of a granitic building by lichens. Int. Biodeterior. Biodegrad. 1997, 40, 263–267. [Google Scholar] [CrossRef]
- Salvadori, O.; Municchia, A.C. The Role of Fungi and Lichens in the Biodeterioration of Stone Monuments. Open Conf. Proc. J. 2016, 7, 39–54. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).