Alginate-Based Zinc Oxide Nanoparticles Coating Extends Storage Life and Maintains Quality Parameters of Mango Fruits “cv. Kiett”
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation of Zinc Oxide Nanoparticles
2.2. Characterization of ZnO NPs
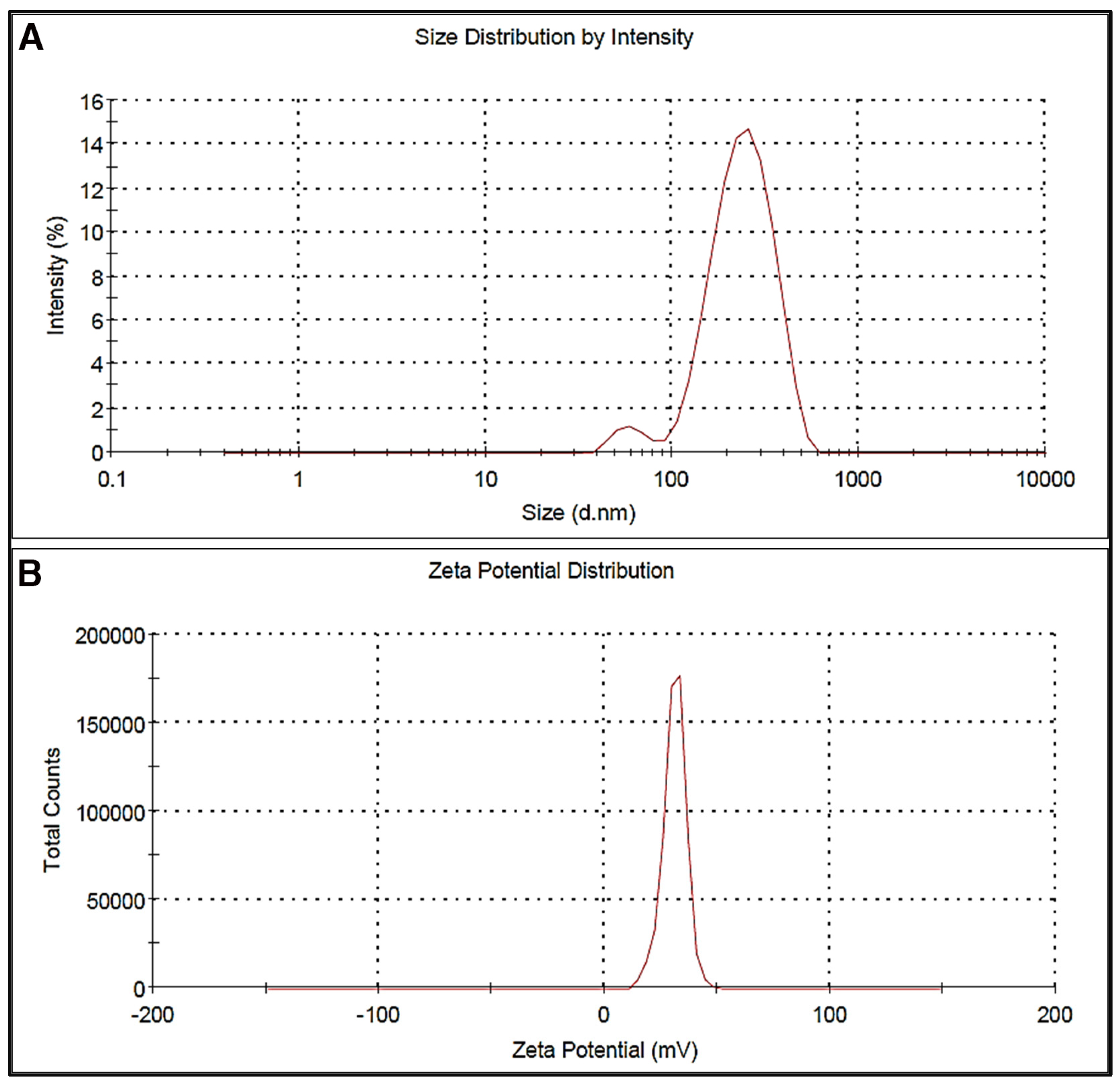
2.2.1. Particle Size and Zeta Potential
2.2.2. Transmission Electron Microscopy (TEM)
2.2.3. Scanning Electron Microscopy (SEM) and Energy-Dispersive X-ray Spectroscopy (EDX)
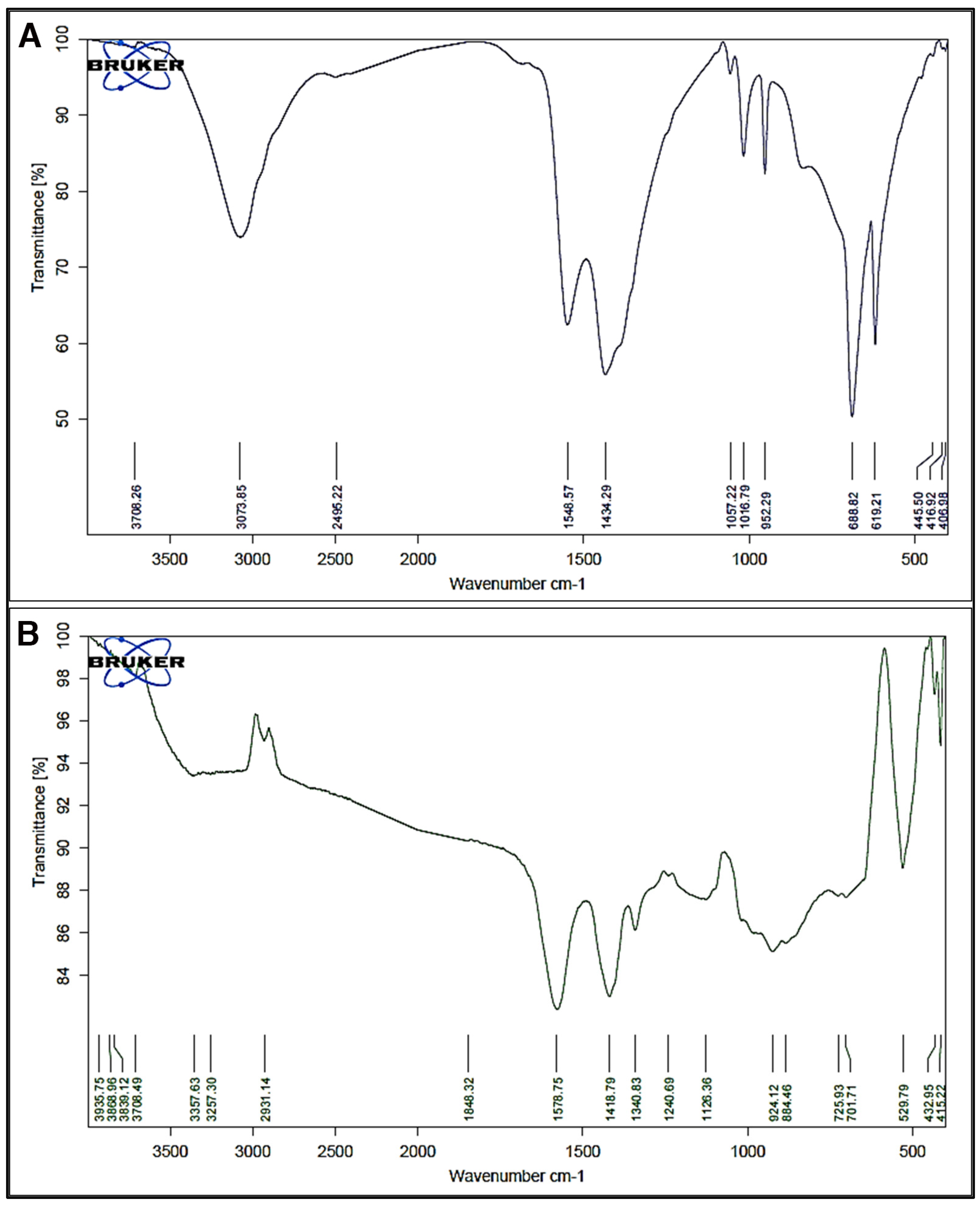
2.2.4. Fourier Transform Infrared Spectroscopy
2.3. Antimicrobial Properties of Alg–ZnO NPs
2.4. Preparation of Alginate Coatings
2.5. Fruits Material
2.6. Coating Application and Storage Conditions
2.7. Fruit Quality Assessment
2.7.1. Respiration Rate
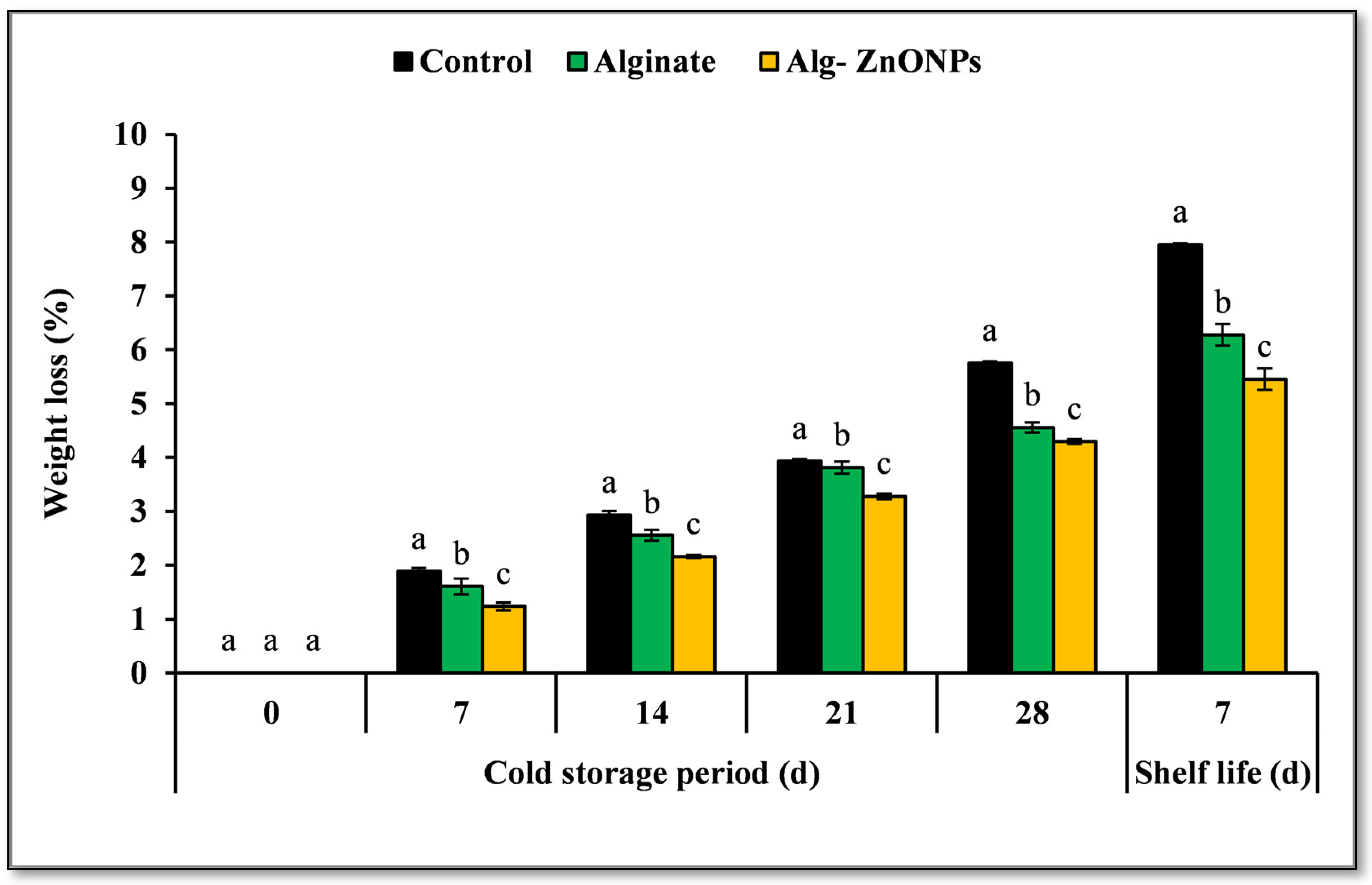
2.7.2. Weight Loss
2.7.3. Fruit Pulp Firmness
2.7.4. Total Soluble Solids (TSS), Titratable Acidity (TA) and TSS/TA Ratio
2.7.5. Total Sugars Content
2.7.6. Reducing Sugar Content
2.7.7. Total Carotenoid Content
2.7.8. Total Phenol Content
2.7.9. Decay Percentage
2.8. Statistical Analysis
3. Results and Discussion
3.1. Particle Size and Zeta Potential
3.2. Transmission Electron Microscopy (TEM) of ZnO NPs
3.3. Scanning Electron Microscopy (SEM) and Energy-Dispersive X-ray Spectroscopy (EDX) of ZnO NPs
3.4. Fourier-Transform Infrared Spectroscopy
3.5. Antimicrobial Properties of Alg–ZnO NPs
3.6. Fruit Quality Assessment
3.6.1. Respiration Rate
3.6.2. Weight Loss
3.6.3. Fruit Pulp Firmness
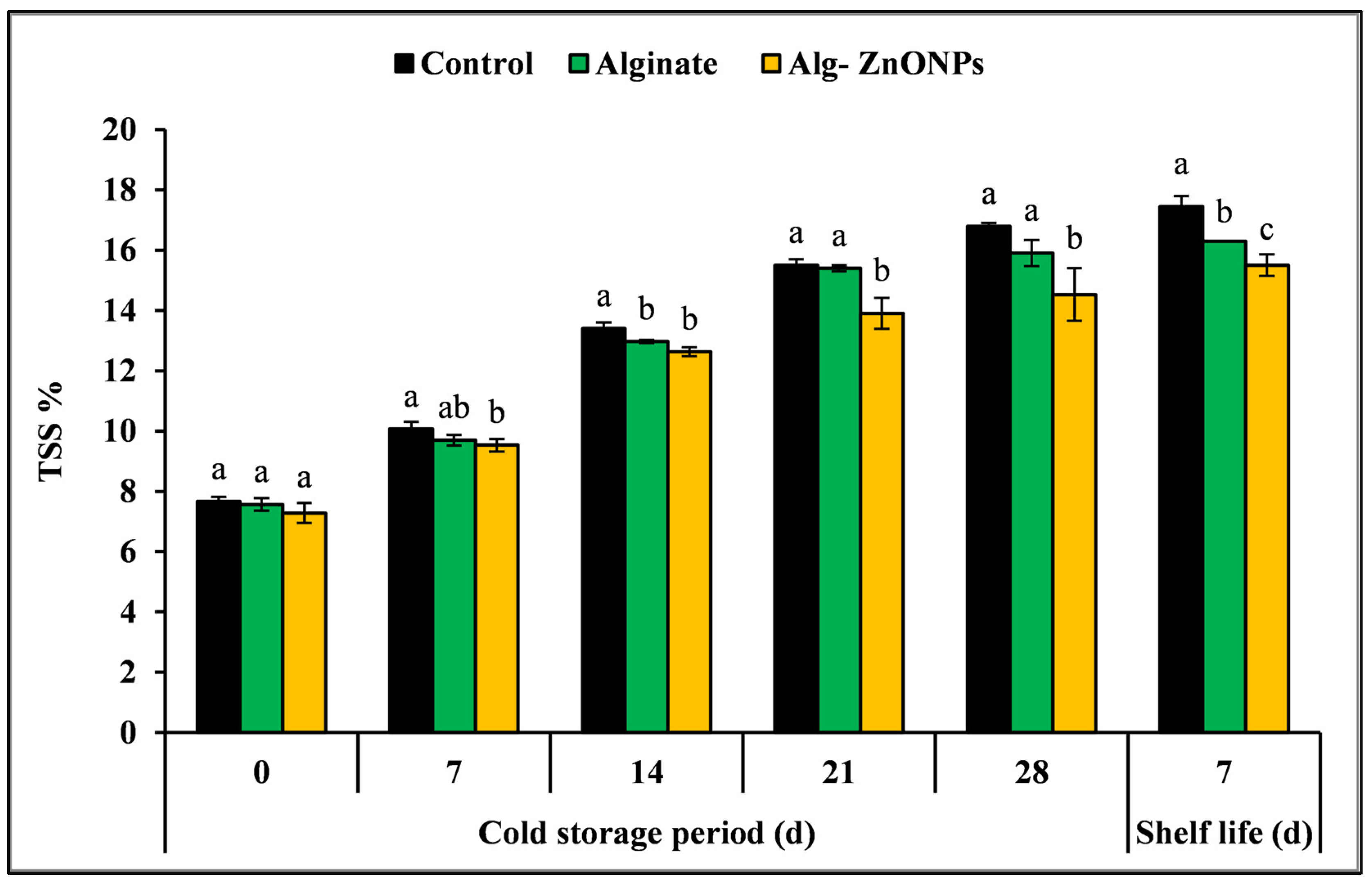
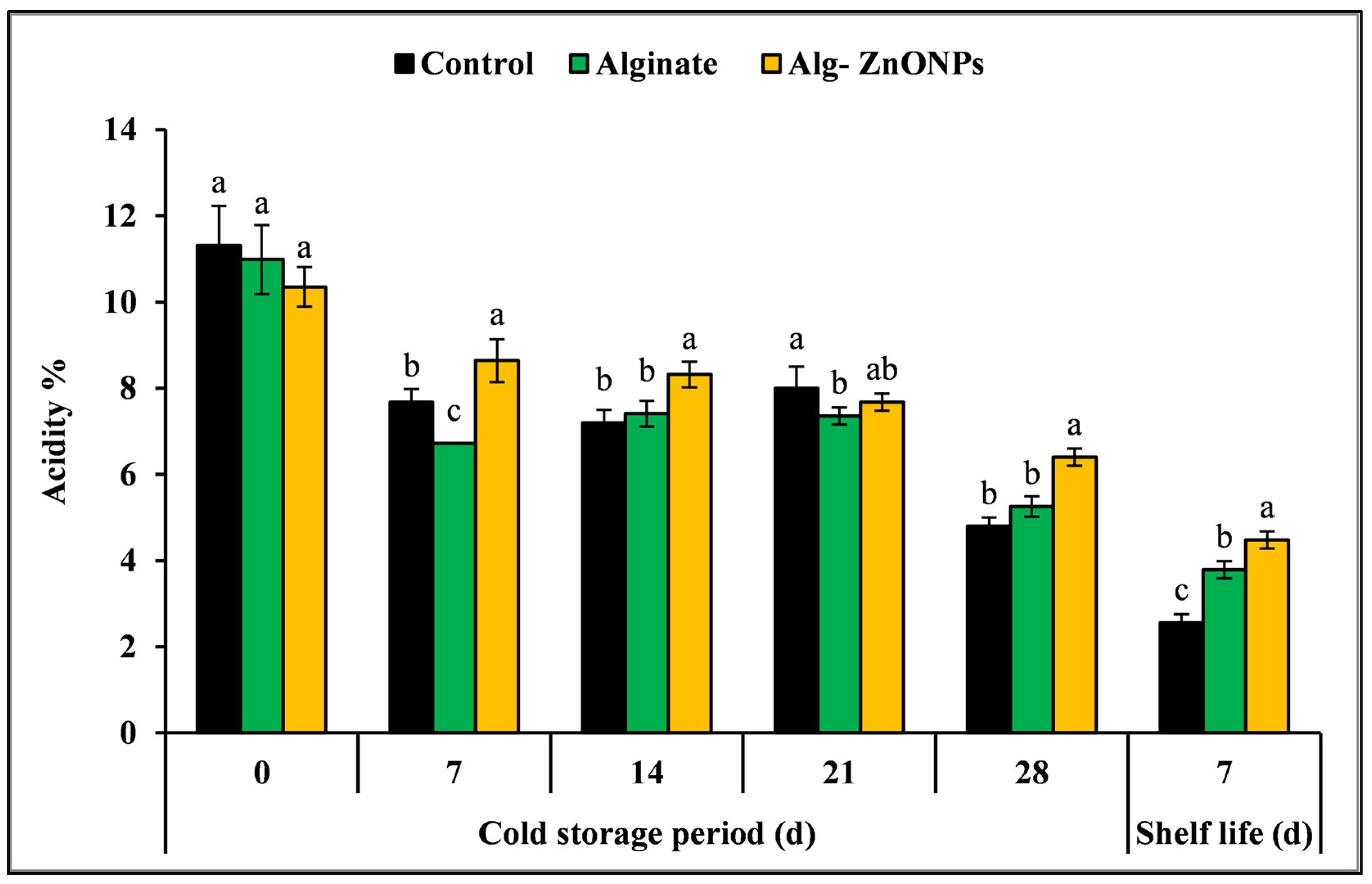
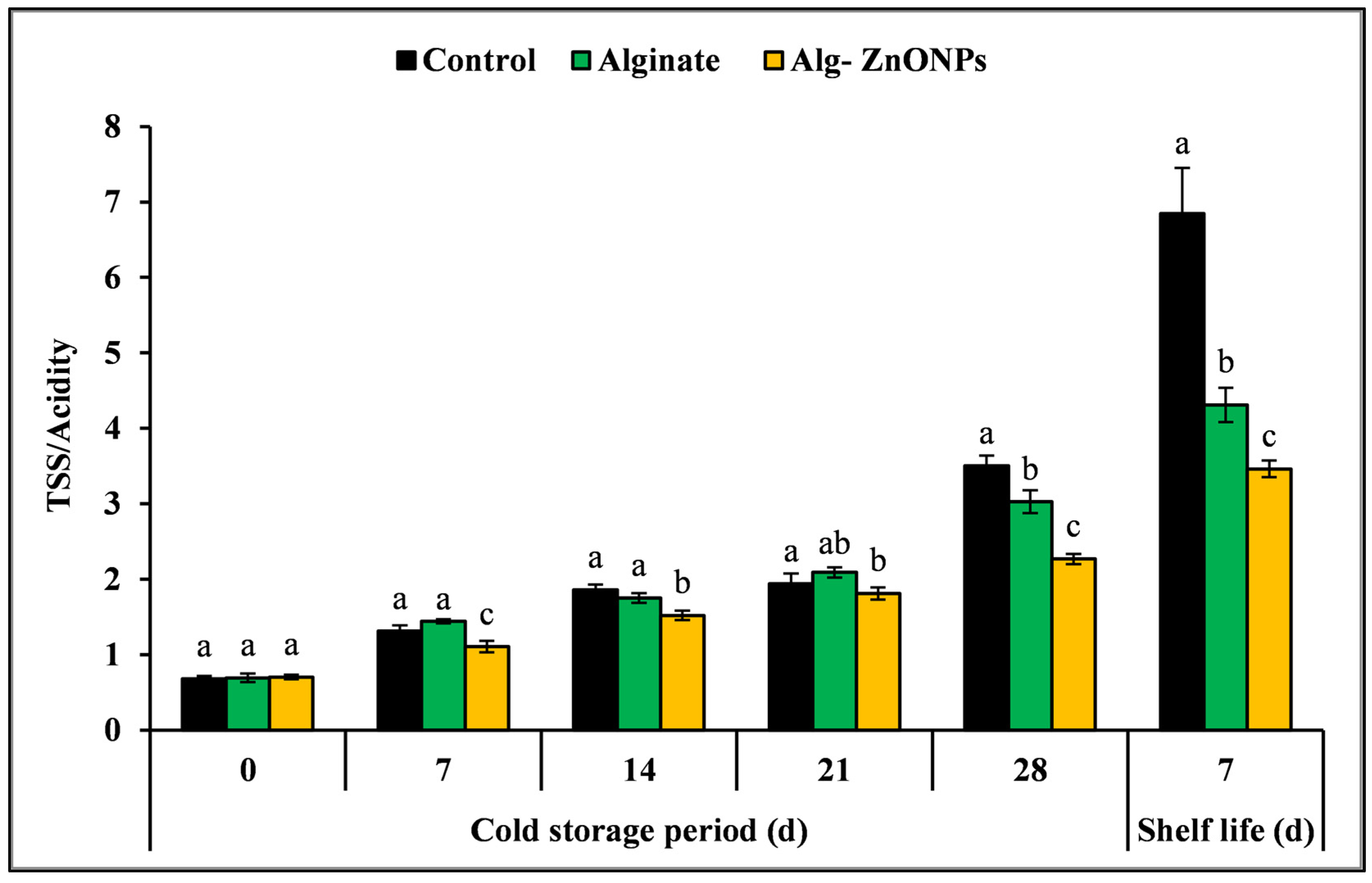
3.6.4. Total Soluble Solids (TSS), Titratable Acidity (TA), and TSS/TA Ratio
3.6.5. Total and Reducing Sugar Content
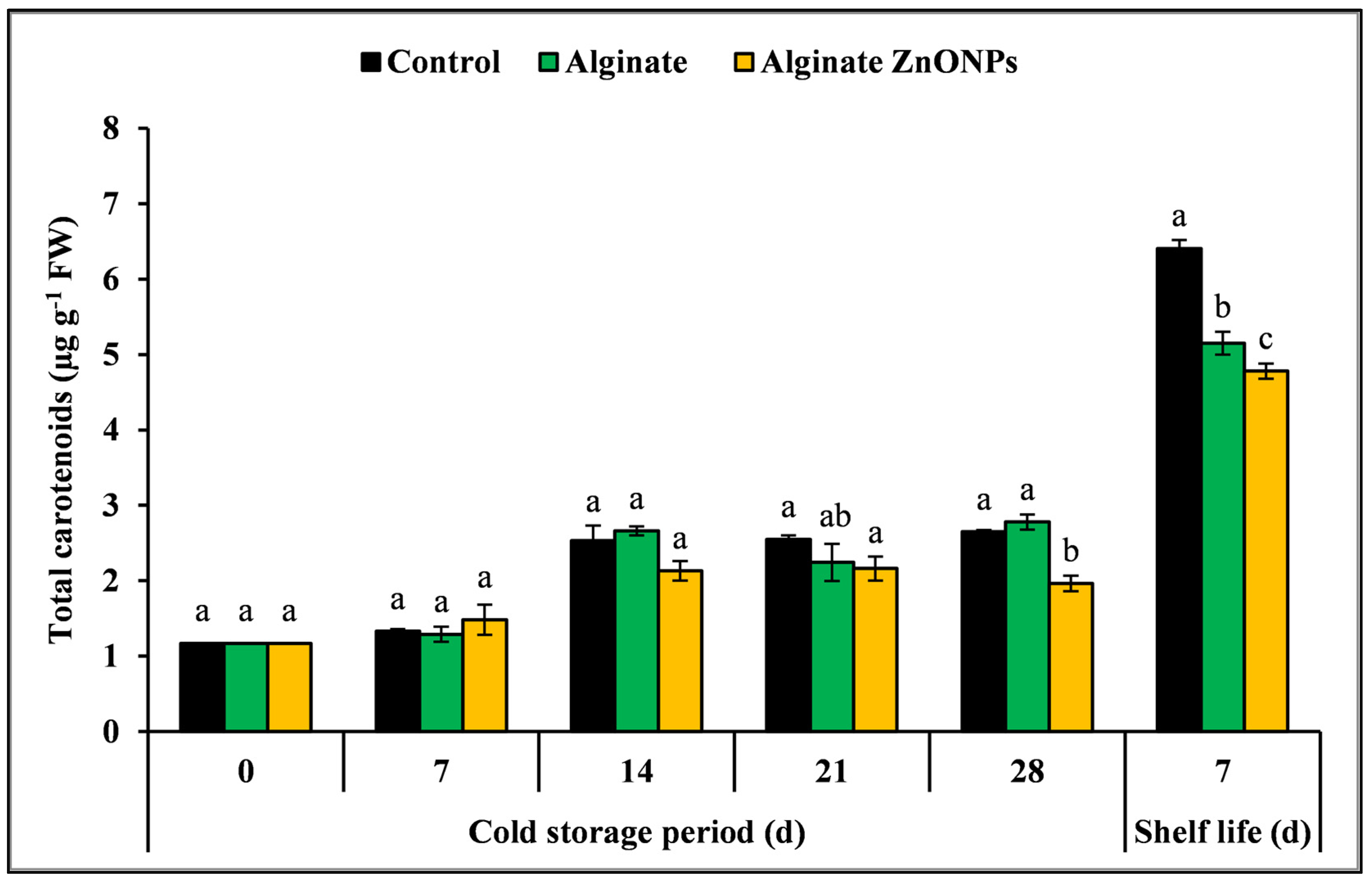
3.6.6. Total Carotenoids Content
3.6.7. Total Phenol Content
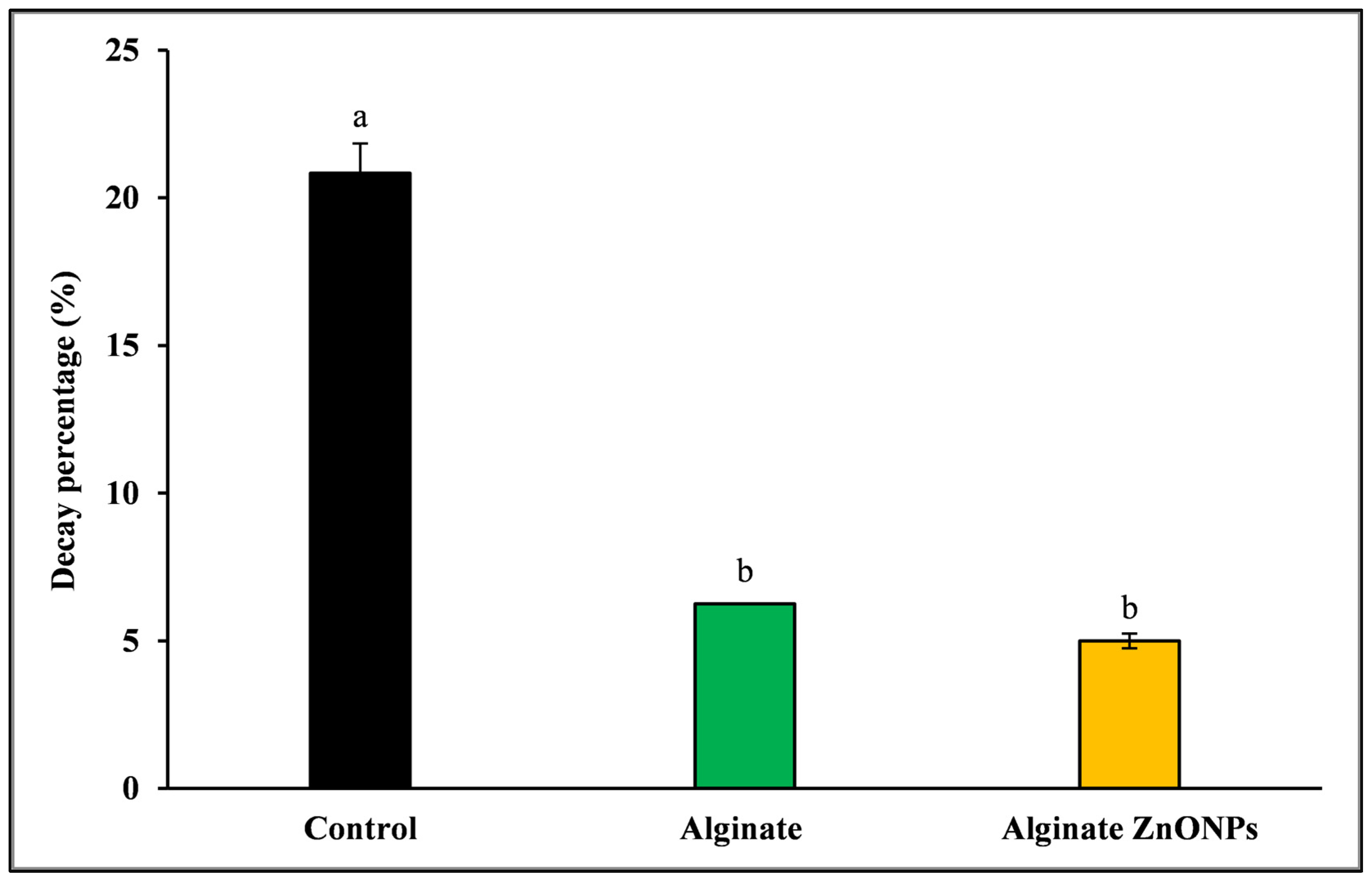
3.6.8. Decay Percentage
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Singh, Z.; Singh, R.K.; Sane, V.A.; Nath, P. Mango-postharvest biology and biotechnology. Crit. Rev. Plant Sci. 2013, 32, 217–236. [Google Scholar] [CrossRef]
- Jahurul, M.H.A.; Zaidul, I.S.M.; Ghafoor, K.; Al-Juhaimi, F.Y.; Nyam, K.L.; Norulaini, N.A.N.; Sahena, F.; Omar, A.M. Mango (Mangifera indica L.) by-products and their valuable components: A review. Food Chem. 2015, 183, 173–180. [Google Scholar] [CrossRef]
- Sivakumar, D.; Jiang, Y.; Yahia, E.M. Maintaining mango (Mangifera indica L.) fruit quality during the export chain. Food Res. Int. 2011, 44, 1254–1263. [Google Scholar] [CrossRef]
- Brecht, J.K.; Yahia, E.M. Postharvest Physiology. In The Mango: Botany, Production and Uses, 2nd ed.; Litz, R.E., Ed.; CAB International: Wallingford, UK, 2009; pp. 484–516. [Google Scholar]
- Knight, R.J., Jr.; Campbell, R.J.; Maguire, I. Important Mango Cultivars and Their Descriptors. In The Mango: Botany, Production and Uses, 2nd ed.; Litz, R.E., Ed.; CAB International: Wallingford, UK, 2009; pp. 42–66. [Google Scholar]
- Riad, M. Mango production in Egypt. Acta Hortic. 1997, 1, 1–6. [Google Scholar] [CrossRef]
- Ntsoane, M.L.; Zude-Sasse, M.; Mahajan, P.; Sivakumar, D. Quality assesment and postharvest technology of mango: A review of its current status and future perspectives. Sci. Hortic. 2019, 249, 77–85. [Google Scholar] [CrossRef]
- Sane, V.A.; Chourasia, A.; Nath, P. Softening in mango (Mangifera indica cv. Dashehari) is correlated with the expression of an early ethylene responsive, ripening related expansin gene, MiExpA1. Postharvest Biol. Technol. 2005, 38, 223–230. [Google Scholar] [CrossRef]
- Cárdenas-Coronel, W.G.; Velez-de la Rocha, R.; Siller-Cepeda, J.H.; Osuna-Enciso, T.; Muy-Rangel, M.D.; Sañudo-Barajas, J.A. Changes in the composition of starch, pectin and hemicellulose during ripening of mango (Mangifera indica cv. Kent). Rev. Chapingo Ser. Hortic. 2012, 18, 5–19. [Google Scholar] [CrossRef]
- Singh, Z.; Singh, S.P. Mango. In Crop Post-Harvest: Science and Technology; Rees, D., Orchard, J., Eds.; Blackwell Pub.: Perishables, UK, 2012; Volume 3, pp. 108–142. [Google Scholar]
- Ridoutt, B.G.; Juliano, P.; Sanguansri, P.; Sellahewa, J. The water footprint of food waste: Case study of fresh mango in Australia. J. Clean Prod. 2010, 18, 1714–1721. [Google Scholar] [CrossRef]
- Sheahan, M.; Barrett, C.B. Food loss and waste in Sub-Saharan Africa. Food Policy 2017, 70, 1–12. [Google Scholar] [CrossRef]
- Carrillo-Lopez, A.; Ramirez-Bustamante, F.; Valdez-Torres, J.B.; Rojas-Villegas, R.; Yahia, E.M. Ripening and quality changes in mango fruit as affected by coating with an edible film. J. Food Qual. 2000, 23, 479–486. [Google Scholar] [CrossRef]
- Parfitt, J.; Barthel, M.; Macnaughton, S. Food waste within food supply chains: Quantification and po-tential for change to 2050. Philos. Transac-Tions R. Soc. Biol. Sci. 2010, 365, 3065–3081. [Google Scholar] [CrossRef] [PubMed]
- Nair, M.S.; Saxena, A.; Kaur, C. Effect of chitosan and alginate based coatings enriched with pomegranate peel extract to extend the postharvest quality of guava (Psidiumguajava L.). Food Chem. 2018, 240, 245–252. [Google Scholar] [CrossRef]
- Hassan, B.; Chatha, S.A.S.; Hussain, A.I.; Zia, K.M.; Akhtar, N. Recent advances on polysaccharides, lipids and protein based edible films and coatings: A review. Int. J. Biol. Macromol. 2018, 109, 1095–1107. [Google Scholar] [CrossRef] [PubMed]
- Maftoonazad, N.; Ramaswamy, H.S.; Marcotte, M. Shelf-life extension of peaches through sodium alginate and methyl cellulose edible coatings. Int. J. Food Sci. Technol. 2008, 43, 951–957. [Google Scholar] [CrossRef]
- Dhall, R.K. Advances in edible coatings for fresh fruits and vegetables: A review. Crit. Rev. Food Sci. Nutr. 2013, 53, 435–450. [Google Scholar] [CrossRef]
- Panahirad, S.; Naghshiband-Hassani, R.; Ghanbarzadeh, B.; Zaare-Nahandi, F.; Mahna, N. Shelf life quality of plum fruits (Prunusdomestica L.) improves with carboxymethylcellulose-based edible coating. HortScience 2019, 54, 505–510. [Google Scholar] [CrossRef]
- Shah, S.; Hashmi, M.S. Chitosan–aloe vera gel coating delays postharvest decay of mango fruit. Hortic. Environ. Biotechnol. 2020, 61, 279–289. [Google Scholar] [CrossRef]
- Qin, Y. Alginate fibres: An overview of the production processes and applications in wound management. Polym. Int. 2008, 57, 171–180. [Google Scholar] [CrossRef]
- Cazón, P.; Velazquez, G.; Ramírez, J.A.; Vázquez, M. Polysaccharide-based films and coatings for food packaging: A review. Food Hydrocoll. 2017, 68, 136–148. [Google Scholar] [CrossRef]
- Li, D.; Wei, Z.; Xue, C. Alginate-based delivery systems for food bioactive ingredients: An overview of recent advances and future trends. Compr. Rev. Food Sci. Food Saf. 2021, 20, 5345–5369. [Google Scholar] [CrossRef]
- Vasilache, V.; Popa, C.; Filote, C.; Cretu, M.A.; Benta, M. Nanoparticles applications for improving the food safety and food processing. In Proceedings of the 7th International Conference on Materials Science and Engineering—BRAMAT, Bramat, Brasov, 24–26 February 2011; pp. 24–26. [Google Scholar]
- Solaiman, M.A.; Ali, M.A.; Abdel-Moein, N.M.; Mahmoud, E.A. Synthesis of Ag-NPs developed by green-chemically method and evaluation of antioxidant activities and anti-inflammatory of synthesized nanoparticles against LPS-induced NO in RAW 264.7 macrophages. Biocatal. Agric. Biotechnol. 2020, 29, 101832. [Google Scholar] [CrossRef]
- Abo-El-Yazid, Z.H.; Ahmed, O.K.; El-Tholoth, M.; Ali, M.A. Green synthesized silver nanoparticles using Cyperus rotundus L. extract as a potential antiviral agent against infectious laryngotracheitis and infectious bronchitis viruses in chickens. Chem. Biol. Technol. Agric. 2022, 9, 1–11. [Google Scholar] [CrossRef]
- Ali, M.A.; Abdel-Moein, N.M.; Owis, A.S.; Ahmed, S.E.; Hanafy, E.A. Preparation, characterization, antioxidant and antimicrobial activities of lignin and eco-friendly lignin nanoparticles from Egyptian cotton stalks. Egypt. J. Chem. 2022, 65, 703–716. [Google Scholar] [CrossRef]
- Kumar, R.; Umar, A.; Kumar, G.; Nalwa, H.S. Antimicrobial properties of ZnO nanomaterials: A review. Ceram. Int. 2017, 43, 3940–3961. [Google Scholar] [CrossRef]
- Sun, Q.; Li, J.; Le, T. Zinc oxide nanoparticle as a novel class of antifungal agents: Current advances and future perspectives. J. Agric. Food Chem. 2018, 66, 11209–11220. [Google Scholar] [CrossRef] [PubMed]
- Padmavathy, N.; Vijayaraghavan, R. Enhanced bioactivity of ZnO nanoparticles—An antimicrobial study. Sci. Technol. Adv. Mate 2008, 3, 035004. [Google Scholar] [CrossRef]
- Gammariello, D.; Conte, A.; Buonocore, G.G.; Del Nobile, M.A. Bio-based nanocomposite coating to preserve quality of Fior di latte cheese. J. Dairy Sci. 2011, 94, 5298–5304. [Google Scholar] [CrossRef]
- Hmmam, I.; Zaid, N.M.; Mamdouh, B.; Abdallatif, A.; Abd-Elfattah, M.; Ali, M. Storage Behavior of “Seddik” mango fruit coated with CMC and guar gum-based silver nanoparticles. Horticulturae 2021, 7, 44. [Google Scholar] [CrossRef]
- Bian, S.W.; Mudunkotuwa, I.A.; Rupasinghe, T.; Grassian, V.H. Aggregation and dissolution of 4 nm ZnO nanoparticles in aqueous environments: Influence of pH, ionic strength, size, and adsorption of humic acid. Langmuir 2011, 27, 6059–6068. [Google Scholar] [CrossRef]
- Shamhari, N.M.; Wee, B.S.; Chin, S.F.; Kok, K.Y. Synthesis and characterization of zinc oxide nanoparticles with small particle size distribution. Acta Chim. Slov. 2018, 65, 578–585. [Google Scholar] [CrossRef]
- Balouiri, M.; Sadiki, M.; Ibnsouda, S.K. Methods for in vitro evaluating antimicrobial activity: A review. J. Pharm. Anal. 2016, 6, 71–79. [Google Scholar] [CrossRef] [PubMed]
- Iñiguez-Moreno, M.; Ragazzo-Sánchez, J.A.; Barros-Castillo, J.C.; Sandoval-Contreras, T.; Calderón-Santoyo, M. Sodium alginate coatings added with Meyerozyma caribbica: Postharvest biocontrol of Colletotrichum gloeosporioides in avocado (Persea americana Mill. cv. Hass). Postharvest Biol. Technol. 2020, 163, 111123. [Google Scholar] [CrossRef]
- Tapia, M.S.; Rojas-Grau, M.A.; Carmona, A.; Rodriguez, F.J.; Soliva-Fortuny, R.; Martin-Belloso, O. Use of alginate- and gellan-based coatings for improving barrier, texture and nutritional properties of fresh-cut papaya. Food Hydrocoll. 2008, 22, 1493–1503. [Google Scholar] [CrossRef]
- Pristijono, P.; Golding, J.B.; Bowyer, M.C. Postharvest UV-C treatment, followed by storage in a continuous low-level ethylene atmosphere, maintains the quality of ‘Kensington pride’ mango fruit stored at 20 °C. Horticulturae 2019, 5, 1. [Google Scholar] [CrossRef]
- Liu, S.; Huang, H.; Huber, D.J.; Pan, Y.; Shi, X.; Zhang, Z. Delay of ripening and softening in ‘Guifei’ mango fruit by postharvest application of melatonin. Postharvest Biol. Technol. 2020, 163, 111136. [Google Scholar] [CrossRef]
- Islam, M.; Khan, M.Z.H.; Sarkar, M.A.R.; Absar, N.; Sarkar, S.K. Changes in acidity, TSS, and sugar content at different storage periods of the postharvest mango (Mangifera indica L.) influenced by Bavistin DF. Int. J. Food Sci. 2013, 2013, 939385. [Google Scholar] [CrossRef] [PubMed]
- Jain, A.; Jain, R.; Jain, S. Quantitative Analysis of Reducing Sugars by 3, 5-Dinitrosalicylic Acid (DNSA Method). In Basic Techniques in Biochemistry, Microbiology and Molecular Biology; Springer Protocols Handbooks; Humana: New York, NY, USA, 2020; pp. 181–183. [Google Scholar] [CrossRef]
- Nielsen, S.S. Total Carbohydrate by Phenol-Sulfuric Acid Method. In Food Analysis Laboratory Manual; Food Science Text Series; Springer: Cham, Switzerland, 2017; pp. 137–141. [Google Scholar] [CrossRef]
- Bärlocher, F.; Graça, M.A.S. Total Phenolics. In Methods to Study Litter Decomposition; Bärlocher, F., Gessner, M., Graça, M., Eds.; Springer: Cham, Switzerland, 2020; pp. 157–161. [Google Scholar] [CrossRef]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2021; Available online: https://www.R-project.org/ (accessed on 19 January 2023).
- Suganthi, K.S.; Rajan, K.S. Temperature induced changes in ZnO–water nanofluid: Zeta potential, size distribution and viscosity profiles. Int. J. Heat Mass Transf. 2012, 55, 7969–7980. [Google Scholar] [CrossRef]
- Sapsford, K.E.; Tyner, K.M.; Dair, B.J.; Deschamps, J.R.; Medintz, I.L. Analyzing nanomaterial bioconjugates: A review of current and emerging purification and characterization techniques. Anal. Chem. 2011, 83, 4453–4488. [Google Scholar] [CrossRef]
- Rasha, E.; Monerah, A.; Manal, A.; Rehab, A.; Mohammed, D.; Doaa, E. Biosynthesis of Zinc Oxide Nanoparticles from Acacia nilotica (L.) Extract to Overcome Carbapenem-Resistant Klebsiella Pneumoniae. Molecules 2021, 26, 1919. [Google Scholar] [CrossRef]
- Fakhari, S.; Jamzad, M.; Kabiri Fard, H. Green synthesis of zinc oxide nanoparticles: A comparison. Green Chem. Lett. Rev. 2019, 12, 19–24. [Google Scholar] [CrossRef]
- Umar, H.; Kavaz, D.; Rizaner, N. Biosynthesis of zinc oxide nanoparticles using Albizia lebbeck stem bark, and evaluation of its antimicrobial, antioxidant, and cytotoxic activities on human breast cancer cell lines. Int. J. Nanomed. 2019, 14, 87–100. [Google Scholar] [CrossRef] [PubMed]
- Hasnidawani, J.N.; Azlina, H.N.; Norita, H.; Bonnia, N.N.; Ratim, S.; Ali, E.S. Synthesis of ZnO nanostructures using sol-gel method. Procedia Chem. 2016, 19, 211–216. [Google Scholar] [CrossRef]
- Choy, J.H.; Kwon, Y.M.; Han, K.S.; Song, S.W.; Chang, S.H. Intra-and inter-layer structures of layered hydroxy double salts, Ni1−x Zn2x (OH)2 (CH3CO2)2x nH2O. Mater. Lett. 1998, 34, 356–363. [Google Scholar] [CrossRef]
- Sakohara, S.; Ishida, M.; Anderson, M.A. Visible luminescence and surface properties of nanosized ZnO colloids prepared by hydrolyzing zinc acetate. J. Phys. Chem. B 1998, 102, 10169–10175. [Google Scholar] [CrossRef]
- Wu, C.M.; Baltrusaitis, J.; Gillan, E.G.; Grassian, V.H. Sulfur dioxide adsorption on ZnO nanoparticles and nanorods. J. Phys. Chem. C 2011, 115, 10164–10172. [Google Scholar] [CrossRef]
- Singh, R.P.; Shukla, V.K.; Yadav, R.S.; Sharma, P.K.; Singh, P.K.; Pandey, A.C. Biological approach of zinc oxide nanoparticles formation and its characterization. Adv. Mater. Lett. 2011, 2, 313–317. [Google Scholar] [CrossRef]
- Bhuyan, T.; Mishra, K.; Khanuja, M.; Prasad, R.; Varma, A. Biosynthesis of zinc oxide nanoparticles from Azadirachta indica for antibacterial and photocatalytic applications. Mater. Sci. Semicond 2015, 32, 55–61. [Google Scholar] [CrossRef]
- Reddy, K.M.; Feris, K.; Bell, J.; Wingett, D.G.; Hanley, C.; Punnoose, A. Selective toxicity of zinc oxide nanoparticles to prokaryotic and eukaryotic systems. Appl. Phys. Lett. 2007, 90, 213902. [Google Scholar] [CrossRef]
- Zarrindokht, E.K.; Pegah, C. Antibacterial activity of ZnO nanoparticle on gram-positive and gram-negative bacteria. Afr. J. Microbiol. Res. 2011, 5, 1368–1373. [Google Scholar] [CrossRef]
- Schwartz, V.B.; Thétiot, F.; Ritz, S.; Pütz, S.; Choritz, L.; Lappas, A.; Förch, P.; Landfester, K.; Jonas, U. Antibacterial surface coatings from zinc oxide nanoparticles embedded in poly (n-isopropylacrylamide) hydrogel surface layers. Adv. Funct. Mater. 2012, 22, 2376–2386. [Google Scholar] [CrossRef]
- Wahab, R.; Siddiqui, M.A.; Saquib, Q.; Dwivedi, S.; Ahmad, J.; Musarrat, J.; Al-Khedhairy, A.A.; Shin, H.S. ZnO nanoparticles induced oxidative stress and apoptosis in HepG2 and MCF-7 cancer cells and their antibacterial activity. Colloids Surf. B Biointerfaces 2014, 117, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Xie, Y.; He, Y.; Irwin, P.L.; Jin, T.; Shi, X. Antibacterial activity and mechanism of action of zinc oxide nanoparticles against Campylobacter jejuni. Appl. Environ. Microbiol. 2011, 77, 2325–2331. [Google Scholar] [CrossRef] [PubMed]
- Shi, L.E.; Li, Z.H.; Zheng, W.; Zhao, Y.F.; Jin, Y.F.; Tang, Z.X. Synthesis, antibacterial activity, antibacterial mechanism and food applications of ZnO nanoparticles: A review. Food Addit. Contam. 2014, 31(Part A), 173–186. [Google Scholar] [CrossRef]
- Wills, R.; Golding, J. Postharvest: An Introduction to the Physiology and Handling of Fruit and Vegetables, 6th ed.; CABI: Wallingford, UK, 2016; 293p. [Google Scholar] [CrossRef]
- Ali, A.; Maqbool, M.; Ramachandran, S.; Alderson, P.G. Gum arabic as a novel edible coating for enhancing shelf-life and improving postharvest quality of tomato (Solanumlycopersicum L.) fruit. Postharvest Biol. Technol. 2010, 58, 42–47. [Google Scholar] [CrossRef]
- Kanmani, P.; Rhim, J.W. Properties and characterization of bionanocomposite films prepared with various biopolymers and ZnO nanoparticles. Carbohydr. Polym. 2014, 106, 190–199. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Aziz, M.S.; Salama, H.E. Development of alginate-based edible coatings of optimized UV-barrier properties by response surface methodology for food packaging applications. Int. J. Biol. Macromol. 2022, 212, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Saekow, M.; Naradisorn, M.; Tongdeesoontorn, W.; Hamauzu, Y. Effect of carboxymethyl cellulose coating containing ZnO-nanoparticles for prolonging shelf life of persimmon and tomato fruit. J. Food Sci. Agric. Technol. 2019, 5, 41–48. [Google Scholar]
- Meng, X.; Zhang, M.; Adhikari, B. The effects of ultrasound treatment and nano-zinc oxide coating on the physiological activities of fresh-cut kiwifruit. Food Bioproc. Technol. 2014, 7, 126–132. [Google Scholar] [CrossRef]
- Silva, G.M.C.; Silva, W.B.; Medeiros, D.B.; Salvador, A.R.; Cordeiro, M.H.M.; da Silva, N.M.; Santana, D.B.; Mizobutsi, G.P. The chitosan affects severely the carbon metabolism in mango (Mangifera indica L. cv. Palmer) fruit during storage. Food Chem. 2017, 237, 372–378. [Google Scholar] [CrossRef] [PubMed]
- Diaz-Mula, H.M.; Serrano, M.; Valero, D. Alginate coatings preserve fruit quality and bioactive compounds during storage of sweet cherry fruit. Food Bioproc. Technol. 2012, 5, 2990–2997. [Google Scholar] [CrossRef]
- Kittur, F.S.; Saroja, N.; Habibunnisa; Tharanathan, R. Polysaccharide-based composite coating formulations for shelf-life extension of fresh banana and mango. Eur. Food Res. Technol. 2001, 213, 306–311. [Google Scholar] [CrossRef]
- Yaman, Ö.; Bayoιndιrl, L. Effects of an edible coating and cold storage on shelf-life and quality of cherries. LWT-Food Sci. Technol. 2002, 35, 146–150. [Google Scholar] [CrossRef]
- Nawab, A.; Alam, F.; Hasnain, A. Mango kernel starch as a novel edible coating for enhancing shelf-life of tomato (Solanumlycopersicum) fruit. Int. J. Biol. Macromol. 2017, 103, 581–586. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Sun, Q.; Huang, H.; Duan, Y.; Xiao, G.; Le, T. Enhanced physico-mechanical, barrier and antifungal properties of soy protein isolate film by incorporating both plant-sourced cinnamaldehyde and facile synthesized zinc oxide nanosheets. Colloids Surf. B Biointerfaces 2019, 180, 31–38. [Google Scholar] [CrossRef]
- Emamifar, A.; Bavaisi, S. Nanocomposite coating based on sodium alginate and nano- ZnO for extending the storage life of fresh strawberries (Fragaria×ananassa Duch.). J Food Meas Charact. 2020, 14, 1012–1024. [Google Scholar] [CrossRef]
- Elsheshetawy, H.E.; Mossad, A.; Elhelew, W.K.; Farina, V. Comparative study on the quality characteristics of some Egyptian mango cultivars used for food processing. Ann. Agric. Sci. 2016, 61, 49–56. [Google Scholar] [CrossRef]
- Mditshwa, L.S.; Magwaza, S.Z.; Tesfay, N. Mbili Postharvest quality and composition of organically and conventionally produced fruits: A review. Sci. Hortic. 2017, 216, 148–159. [Google Scholar] [CrossRef]
- Thakur, R.; Pristijono, P.; Bowyer, M.; Singh, S.P.; Scarlett, C.J.; Stathopoulos, C.E.; Vuong, Q.V. A starch edible surface coating delays banana fruit ripening. LWT-Food Sci. Technol. 2019, 100, 341–347. [Google Scholar] [CrossRef]
- Gol, N.B.; Chaudhari, M.L.; Rao, T.V.R. Effect of edible coatings on quality and shelf life of carambola (Averrhoacarambola L.) fruit during storage. J. Food Sci. Technol. 2015, 52, 78–91. [Google Scholar] [CrossRef]
- Batista-Silva, W.; Nascimento, V.L.; Medeiros, D.B.; Nunes-Nesi, A.; Ribeiro, D.M.; Zsögön, A.; Araújo, W.L. Modifications in organic acid profiles during fruit development and ripening: Correlation or causation? Front. Plant Sci. 2018, 9, 1689. [Google Scholar] [CrossRef]
- Xu, D.; Qin, H.; Ren, D. Prolonged preservation of tangerine fruits using chitosan/montmorillonite composite coating. Postharvest Biol. Technol. 2018, 143, 50–57. [Google Scholar] [CrossRef]
- Prasanna, V.; Prabha, T.N.; Tharanathan, R.N. Fruit ripening phenomena–an overview. Crit. Rev. Food Sci. Nutr. 2007, 47, 231–258. [Google Scholar] [CrossRef]
- Cordenunsi, B.R.; Lajolo, F.M. Starch breakdown during banana ripening: Sucrose synthase and sucrose phosphate synthase. J. Agric. Food Chem. 1995, 43, 347–351. [Google Scholar] [CrossRef]
- Osorio, S.; Scossa, F.; Fernie, A. Molecular regulation of fruit ripening. Front. Plant Sci. 2013, 4, 198. [Google Scholar] [CrossRef]
- Khaliq, G.; Mohamed, M.T.M.; Ali, A.; Ding, P.; Ghazali, H.M. Effect of gum arabic coating combined with calcium chloride on physico-chemical and qualitative properties of mango (Mangifera indica L.) fruit during low temperature storage. Sci. Hortic. 2015, 190, 187–194. [Google Scholar] [CrossRef]
- Swallah, M.S.; Sun, H.; Affoh, R.; Fu, H.; Yu, H. Antioxidant potential overviews of secondary metabolites (polyphenols) in fruits. Int. J. Food Sci. 2020, 2020, 9081686. [Google Scholar] [CrossRef]
- Tanada-Palmu, P.S.; Grosso, C.R. Effect of edible wheat gluten-based films and coatings on refrigerated strawberry (Fragaria ananassa) quality. Postharvest Biol. Technol. 2005, 36, 199–208. [Google Scholar] [CrossRef]


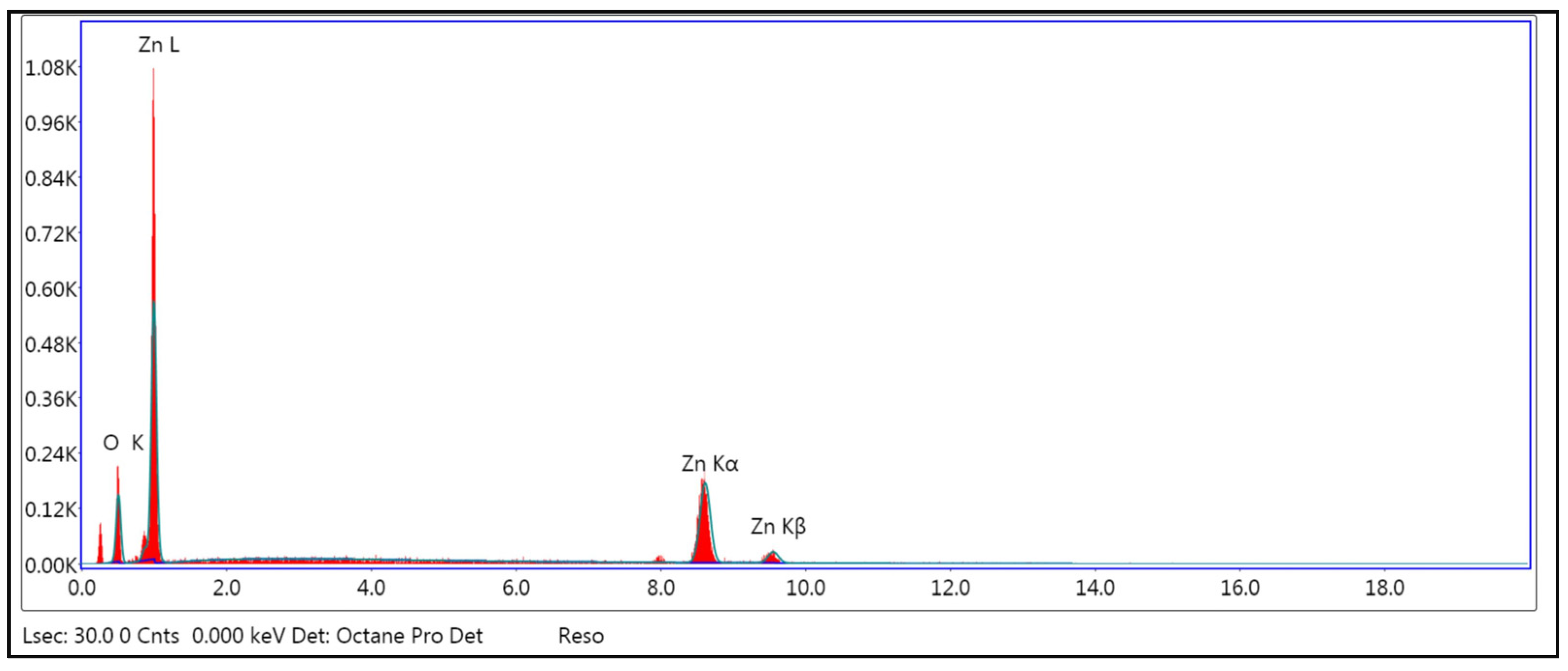





| Element | Weight % | Atomic % |
|---|---|---|
| O K | 17.52 | 46.46 |
| Zn K | 82.48 | 53.54 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hmmam, I.; Ali, M.A.-S.; Abdellatif, A. Alginate-Based Zinc Oxide Nanoparticles Coating Extends Storage Life and Maintains Quality Parameters of Mango Fruits “cv. Kiett”. Coatings 2023, 13, 362. https://doi.org/10.3390/coatings13020362
Hmmam I, Ali MA-S, Abdellatif A. Alginate-Based Zinc Oxide Nanoparticles Coating Extends Storage Life and Maintains Quality Parameters of Mango Fruits “cv. Kiett”. Coatings. 2023; 13(2):362. https://doi.org/10.3390/coatings13020362
Chicago/Turabian StyleHmmam, Ibrahim, Mohamed Abdel-Shakur Ali, and Abdou Abdellatif. 2023. "Alginate-Based Zinc Oxide Nanoparticles Coating Extends Storage Life and Maintains Quality Parameters of Mango Fruits “cv. Kiett”" Coatings 13, no. 2: 362. https://doi.org/10.3390/coatings13020362
APA StyleHmmam, I., Ali, M. A.-S., & Abdellatif, A. (2023). Alginate-Based Zinc Oxide Nanoparticles Coating Extends Storage Life and Maintains Quality Parameters of Mango Fruits “cv. Kiett”. Coatings, 13(2), 362. https://doi.org/10.3390/coatings13020362







