1. Introduction
Severe Acute Respiratory Syndrome-Coronavirus-2 (SARS-CoV-2) is an agent that causes coronavirus disease (COVID-19). Despite the available treatments against SARS-CoV-2 and vaccination, there are still about 767 million cases of daily worldwide infections, including over 6.9 million deaths, due to the COVID-19 pandemic [
1,
2]. Additionally, RNA viruses, which have a high mutation rate, like SARS-CoV-2, increase the risk of multi-resistant variant development with more virulence characteristics [
1]. This virus, which is highly communicable, is mainly transmitted though respiratory droplets and aerosols during human-to-human interactions [
3] or indirectly through physical contact, usually via the hands after being deposited on surfaces, such as packaging [
2]. The spread of virus particles has demonstrated the necessity to develop novel materials with antimicrobial properties to prevent the infection of current viruses and yet-unknown variants that could appear in the future [
1,
2,
3,
4]. Antiviral surface material treatments may influence virus particles through a reduction in their number or can directly avoid these hazards [
4]. Consequently, the use of highly efficient, low-cost, environmentally friendly antiviral coatings on surfaces or as an external packaging material layer could be a reasonable solution to mitigate the transmission of viral diseases.
The antimicrobial properties of various metals nanoparticles, such as ZnO, TiO
2, copper and silver nanoparticles, have been extensively studied and documented [
2,
5,
6,
7]. Zinc oxide nanoparticles hold antimicrobial properties for Gram-negative and Gram-positive bacteria to spores highly resistant to high temperatures and pressure environments, yeasts, mold [
6,
7,
8,
9,
10] and viruses [
1,
3]. Zinc oxide nanoparticles have been introduced into polymer matrices derived from petroleum, using conventional incorporation methods, such as melt mixing or solvent casting, as well as being added to polymer coating layers in the application of antimicrobial packaging or surfaces [
6,
7]. Zhou et al. [
11] noted that negatively charged ZnO nanoparticles may simply trap the virus particles and inhibit their attachment to the host cells. When ZnO was co-cultured with viruses and cells, the cell survival rate increased by over 50%. Additionally, an external coating of packaging or a surface covered with an active coating should be active in storage/usage, meaning it has to be able to be sufficiently resistant against the irradiation of UV, be protected against UV light by the use of an additive or be activated by UV. This means that introducing active substances, such as ZnO nanoparticles that are resistant to UV aging in a coating carriers or adding an agent with shielding properties, may not only prevent the inactivation of the external coating/surface after UV aging but can even increase its antimicrobial activity [
5,
6,
7]. It is worth mentioning that ZnO nanoparticles exhibit shielding properties [
6,
7]. Additionally, the activity of the zinc oxide nanoparticles and their shielding properties may depend on their structure/morphology. The nanoparticles can be observed as lens-like ZnO pyramidal crystals, which may grow in the form of diamonds, in which there is a hexagonal base and a conical tip. Those diamond-like ZnO structures were noted to give the nanomaterials extraordinary optical properties [
12].
Photocatalytic nanoparticles such as titanium dioxide (TiO
2) may also be used for surface coatings or packaging external coating fabrication to limit the spread of bacterial cells and viruses particles from the most commonly touched surfaces or from packed-food products by a photo-inducible antimicrobial effect [
13]. Mirzapoor et al. [
13] found that titanium dioxide or titania (TiO
2)-covered surfaces minimized microbial adhesion via altering the surface free energy. Due to the optical band gap (3.2 eV), reactivity, stability, reusability, biocompatibility, durability, crystallinity, corrosion resistance, high surface area and, most importantly, low cost, TiO
2 was found to be the best candidate as an antimicrobial agent, which could attack microorganisms with reactive oxygen species (ROS) [
14,
15]. TiO
2 nanoparticles have offered great potential for the deactivation of phage MS2, phage f2, HIV, norovirus, etc. It is also worth mentioning that UV-active TiO
2 was found to increase virus inactivation owing to its antimicrobial activity towards genetically similar SARS-CoV-2 and HCoV [
15,
16].
Comparing ZnO and TiO
2 nanoparticles as photoactive metal oxides, it was shown that both ZnO and TiO
2 as excellent semiconductors were effective against hepatitis C, polio, H1N1 influenza, herpes, SARS-CoV, SARS-CoV-2, etc. [
15,
17,
18,
19]. Many authors investigated metal doping as an effective method to extend the spectral response of TiO
2 to a visible region, as well as to decrease the electron–hole recombination rate [
14,
20,
21,
22,
23,
24]. It was reported by the authors [
14,
22] that TiO
2 doped with 10% CuCl
2 resulted in a complete inhibition of 100%
Escherichia coli growth compared to TiO
2. In a great deal of recent research, the light absorption extension of TiO
2 into the visible region has been investigated through the doping of TiO
2 with ZnO nanoparticles [
13]. The authors suggest that zinc oxide nanoparticles with a direct wide band gap of 3.3 eV may be used in an antiviral coating to improve TiO
2 photocatalyst efficiency [
13,
25,
26]. Liu et al. [
9] obtained micro-arc oxidation (MAO) coatings containing Zn, which were fabricated on Ti6Al4V alloys using EDTA-ZnNa2 electrolytes (Zn-doped coatings on Ti6Al4V alloys). The authors clearly showed that the reactive oxygen species (ROS) level of MAO samples was significantly higher than that of the uncovered Ti6Al4V. Zn-doped coatings caused the strongest oxidative stress on Gram-positive bacteria compared to uncoated alloys (due to the relatively high released Zn
2+ concentration). The results from previously carried out work by the authors [
6] demonstrated that active coatings that contained geraniol, carvacrol and nano ZnO were found to be effective against both Gram-positive and Gram-negative bacteria, confirming a synergistic effect between zinc oxide nanoparticles and geraniol or carvacrol. Against a phi 6 phage, the coatings only offered modest activity. However, the synergistic effect between ZnO nanoparticles and TiO
2, which were introduced into the coating carrier, such as a solvent gloss varnish to create an active layer, is not confirmed. Additionally, previous investigations demonstrated that UV irradiation did not decrease the antibacterial activity of the coatings containing ZnO nanoparticles into a coating carrier [
27] or into the polymer matrix after UV aging [
5]. However, the effect of Q-SUN or UV-B irradiation on the antiviral properties of the coatings containing ZnO nanoparticles was not analyzed in any previous work. These findings have led to the implication that coatings containing ZnO nanoparticles and TiO
2 could be active towards viruses, such as SARS-CoV-2, and that there is a synergistic effect between these two active agents. After Prussin II, A.J. et al. [
28] proposed that the Phi6 bacteriophage could be used as a substitute/surrogate for the SARS-CoV-2 together with influenza viruses, and based on previous research [
6], the phi 6 phage was selected to determine the antiviral properties of the coatings.
In summary, the purpose of the research was to examine the coatings’ (based on a solvent varnish contained in the ZnO nanoparticles and TiO2) antiviral activity. The goal was also to investigate the influence of accelerated UV-B and Q-SUN irradiation on the effectiveness of these active layers.
2. Materials and Methods
2.1. Materials
Both the bacterial strain and phage phi6 to be used in this experiment were acquired from a collection offered by the Leibniz Institute DSMZ (Deutsche Sammlung von Mikroorganismen und Zellkulturen, Braunschwelg, Germany). The microorganism utilized in this research was Pseudomonas syringae van Hall 1902 DSM 21482. The phi 6 phage (DSM-21518) was used as a SARS-CoV2 substitute. Polypropylene films (A4, 20 μm) were supplied by MarDruk (Andrychów, Poland). The coating carrier, which was used in these experiments, was a solvent gloss varnish 70 GU279686 (Hubergroup, Warsaw, Poland) and, in addition, zinc oxide AA 44899 (~70 nm) powder (Thermo Fisher GmbH, Kandel, Germany) and TiO2 (Grupa Azoty, Police, Poland), which were used as antiviral compounds. The 99.8% ethanol (EU-ROCHEM BGD Sp. z o.o. Tarnów, Poland) was used as a solvent. Luria-Bertani (LB) broth and Agar-Agar (Merck, Darmstadt, Germany) were taken to determine the antiviral activity of any coatings. All media were prepared in accordance with the Merck protocol (all of the media had to be weighed according to manufacturer instructions and were then suspended in 1000 mL of distilled water and autoclaved at 121 °C for 15 min).
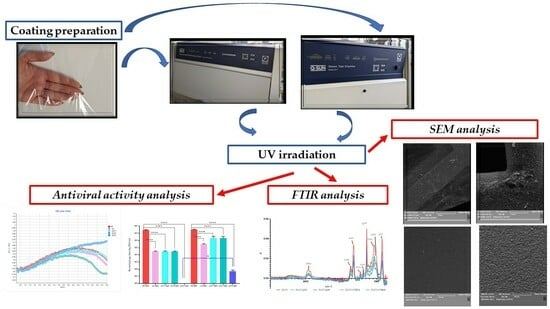
2.2. Coating Preparation
Each of the coating dispersions was prepared according to the scheme (
Figure 1). Briefly, 0.031 g of ZnO nanoparticles was added to 50 mL of 99.8% ethanol. Initially, a one-hour mixing process of dispersion took place using a magnetic stirrer (Ika, Warsaw, Poland) at 450 rpm. Next, sonication took place for 30 min (parameters: cycle: 0.5; 20% amplitude). A ZnO nanoparticle dispersion was added to 50 g of the solvent gloss varnish (coating carrier) and mixed for 10 min at 750 rpm (Zn coating). As next step, 0.031 g of ZnO nanoparticles and 0.031 g of TiO
2 (1:1) were introduced into 50 mL of 99.8% ethanol. A magnetic stirrer at 450 rpm mixed the dispersion for an hour. Next, the dispersion underwent sonication for 30 min (parameters: described above). A dispersion of ZnO and TiO
2 was added to 50 g of the coating carrier and stirred for 10 min at 750 rpm (ZnT1 coating). Consequently, step dispersions containing (a) 0.031 g of ZnO nanoparticles and 0.0405 g of TiO
2 (1:1.5) and (b) 0.031 g of ZnO nanoparticles and 0.062 g of TiO
2 (1:2) were introduced into 50 mL of 99.8% ethanol (separately). These dispersions were then stirred and sonicated as described above. Finally, the dispersions were mixed with varnish and stirred for 10 min at 750 rpm (ZnT2 and ZnT3 coatings).
Table 1 contains the content of the coating dispersions.
A Unicoater 409 (Erichsen, Hemer, Germany) was used to coat the polypropylene (PP) films at 25 °C with a 40 μm diameter roller. The coatings were dried for a period of 10 min at 50 °C. The control samples (PP) were PP films that had not been covered.
The samples of PP film and coated PP films (before and after irradiation) were then sliced into squares (3 cm × 3 cm) for an analysis of antiviral activity.
2.3. UV Irradiation
The PP films, which were non-coated, as well as the PP films covered with active layers were all cut into rectangles (23.5 × 7.0 cm and 26.0 × 2.5 cm), respectively. The aforementioned samples were introduced into (1) a weathering tester that utilized UV-B acceleration with 1.55 W/m2 (Q-LAB DEUTSCHLAND GMBH, Saarbrucken, Germany) and irradiated for 24 h at 60 °C; (2) a Q-SUN accelerated Xenon Test Chamber with 1.5 W/m2 (Model Xe-2, Q-LAB DEUTSCHLAND GMBH, Saarbrucken, Germany) and irradiated for 24 h at 47 °C (black panel) and at 39 °C (chamber air) with an RH of 40%.
2.4. Antiviral Activity Analysis
Initially, the particles of phi6 phage were refined according to Bhetwal et al. [
29]. Later, the pure bacteriophage lysate was prepared according to Bonilla et al. [
30]. The antiviral properties of the active coatings that contained nanoparticles were compared to non-covered films, with this taking place according to a modified ISO 22196-2011 standard [
31]. As a final stage, an amplification of the phage occurred through the use of a method suggested by Skaradzińska et al. [
32]. Antiviral analysis was carried out for the sample films/films covered with coatings before their irradiation and after Q-SUN and UV-B irradiation.
To analyze the real-time growth rate of the
P. syringae (phi 6 phage host) with phages, after incubation with the antiviral coating, bacteriophage lysate was incubated with the non-covered PP films (control) and with samples that were coated with active layers (with each active coating individually) according to ISO 22196-2011 [
31]. The LB broth was added to 5 BioSan bioreactors (BS-010160-A04, BioSan, Riga, Latvia).
P. syringae overnight culture was later added to 10 mL of LB broth, and this was incubated at 28° when OD = 0.2 (optical density). Five phi 6 bacteriophage lysates were amplified in their own respective host bacteria (1 lysate—after its incubation with the non-covered film (control sample), 4 lysates—after their incubation (separately) with the active coatings containing ZnO nanoparticles and TiO
2). Next, 10 µL of phage lysate (MOI = 1) was added to host cultures (OD = 0.2) and incubated at 28° (until OD for the control sample decreased). Five simultaneous tests were carried out, meaning that it was possible to analyze 4 active coatings within one experiment. An analysis of the
P. syringae growth rate in real time was performed for films/coatings before their irradiation and after Q-SUN and UV-B irradiation.
2.5. SEM Analysis
2.5.1. SEM Analysis of the Active Coatings
The PP films and PP films covered with Zn, ZnT1, ZnT2 and ZnT3 active layers were analyzed using a scanning electron microscope (SEM). As a first step, the samples before and after their Q-SUN and UV-B irradiation (for 24 h and 48 h, respectively) were placed on pin stubs with carbon tape and covered with a layer of gold in a sputter coater at 24 °C (Quorum Technologies Q150R S, Laughton, East Sussex, UK). SEM tests were performed using a Vega 3 LMU microscope (Tescan, Brno-Kohoutovice, Czech Republic). The goal of this analysis was to visually confirm that the PP films had been thoroughly and homogeneously covered with the active layers. The SEM was carried out through the use of a tungsten filament with an accelerating voltage of 10 kV.
2.5.2. P. syringae Counts Visualization
SEM analysis was carried out to visualize P. syringae counts at the same time as lysis profile experiments. P. syringae and phage phi6 after its incubation with the selected, non-irradiated active coating (ZnT3: coating which was active toward phi 6 phage) were selected for this assay to verify that the incubation of the phage lysate active layer influenced phage activity, and these phage particles did not influence the growth of the host cells. P. syringae with phage phi6 after its incubation with the non-coated PP film (control sample had no influence on the phage activity) and lysate after its incubation with the ZnT1 coating (coating which was not active against phi 6 phage) were used as lysis control. Lysis profile tests were carried out on a smaller scale in Eppendorf tubes in a 1 mL final volume. After a period of 6 h incubation at 28 °C, the P. syringae and phage phi6 samples were placed in room-temperature conditions (24 °C). Grids of carbon-coated copper (400 mesh) were then placed in the liquid samples fully immersed, and the bacterial cells were given 30 min to adhere. Later, the prepared grids were removed from Eppendorf tubes, placed on petri dishes, and sterile paper sheet was used to remove the excess liquid and they were then left to dry. For a period of 18 h at 4 °C, the samples were then fixed (2% glutaraldehyde in a 0.1 M sodium cacodylate (NaCac, pH 7.4)). Later, the samples were washed in 0.1 M sodium cacodylate and dehydrated at 1 h intervals in serial concentrations of ice-cold (−20 °C) methanol (10%, 20%, 40%, 60%, 80% and 100%). The samples were left on pin stubs where a coating of thin gold layer was added in a sputter coater at room temperature (Quorum Technologies Q150R S, Laughton, East Sussex, UK). Further on, a microscopic analysis was performed using a Vega 3 LMU (Tescan) scanning electron microscope (SEM). An analysis was performed at room temperature with a tungsten filament, and an accelerating voltage of 20 kV was used to capture SEM images for the analyzed samples. All specimens were viewed from above.
2.6. FT-IR
Fourier-transform infrared (FT-IR) spectrum of the non-coated and coated PP film (before and after UV-B and Q-SUN) irradiation was measured using FT-IR spectroscopy (Perkin Elmer Spectrophotometer, Spectrum 100), operated at a resolution of 4 cm−1 and with eight scans. The non-coated and coated PP film samples were cut into squares (3 × 3 cm) and placed directly in the ray-exposing stage. The spectrum was recorded at a wavelength of 650–4000 cm−1.
2.7. Statistical Analysis
When carrying out an analysis of variance (one-way ANOVA), statistical significance was noted, of considerably different values, where p < 0.05. The analyses were carried out by using GraphPad Prism 8 (GraphPad Software, San Diego, CA, USA).
4. Discussion
Beginning in 2019, the whole planet faced a medical emergency with the arrival of a novel, highly contagious, deadly strain, known as SARS-CoV-2 [
15]. Safety concerns led to worldwide attempts to limit COVID-19 spread through hand-to-hand contact. Coating surfaces and packaging materials could be the solution [
6,
7]. In order to overcome the inherent shortcomings associated with the low activity of natural antiviral compounds or their sensitivity to UV aging, effective nanoparticle-based agents have been added to the coating carriers to obtain layers/covers that would function or even work actively on UV irradiation [
13,
15,
17,
18,
19]. Many authors confirmed that both ZnO nanoparticles and TiO
2 demonstrated antiviral activity [
11,
15,
17,
18,
19]. These findings indicated that coating materials containing ZnO nanoparticles could actively react against the phi6 phage. A previous study [
6] showed that the addition of geraniol or of carvacrol into the coating carriers containing nanoparticles influenced their antiviral effectiveness, which confirmed synergy between these active compounds. Therefore, it was assumed that the addition of TiO
2 into the varnish with nano ZnO might significantly increase the activity of the coatings against the phi 6 bacteriophage. Mirzapoor et al. [
13] designed and synthesized an antimicrobial layer based on Se/TiO
2/ZnO/Ag nanostructures to enhance the antimicrobial activity of coating synergistically. However, our results suggest that the coatings containing ZnO nanoparticles with TiO
2 at ratios 1:1 or 1:1.5 had no antiviral activity. These findings were confirmed by SEM analysis that showed that phi6 phage particles were still active after the incubation of these coatings and inhibited bacterial cell (host) growth. Contradictory results were observed for the coating (ZnT3) containing ZnO nanoparticles and increased amount of TiO
2 (up to 1:2), which demonstrated high antiviral activity, confirming the synergistic effect between these two oxides. Additionally, SEM micrographs also revealed
P. syringae cells, which were immobilized on the surface of the copper grid, proving that bacteriophages were inactivated by the coating. Similarly, Zheng et al. [
14] confirmed the synergistic effect between two different nanoparticles. The authors prepared Cu-TiO
2 nanofibers, which exhibited high removal efficiency in the case of bacteriophage f2 under visible light, proving the antiviral properties. The coating ZnT3 was also incubated with the phi 6 phage lysate under a visible-light tool. Wang et al. [
40] designed and prepared nanomaterial based on TiO
2, showing that TiO
2 supported by single-atom nanozyme containing atomically dispersed Ag atoms (Ag-TiO
2 SAN) served as a highly efficient antiviral material that exhibited higher adsorption (99.65%) of SARS-CoV2 pseudovirus than TiO
2, which was not supported by the nanozyme. Conversely, Amiri et al. [
41] confirmed that a Ag-doped TiO
2 nanocomposite demonstrated high antimicrobial properties. Similarly, Liu et al. [
42] suggested that TiO
2 coating with nanostructural surface can be improved through UV irradiation.
According to Zheng et al. [
14], TiO
2 may be activated by lights in the near-UV region. Mirzapoor et al. [
13] suggested that when sunlight shines on the active layer containing nanoparticles, such as ZnO or/and TiO
2, the molecules of water and oxygen, along with the energy of the sun’s rays, produce active compounds called VOCs and ROCs. Due to their high strength and energy, these agents may create cavities in the bacterial cell wall or virus envelopes that cause them to be destroyed. According to these suggestions, it was assumed that active coatings based on TiO
2 and ZnO nanoparticles could be activated through UV irradiation. Our results show that coatings that contained nano ZnO or ZnO nanoparticles and TiO
2 nanoparticles at a ratio of 1:1 or 1:1.5 after 24 h irradiation using the Q-SUN Xenon Test Chamber demonstrated moderate antiviral activity, meaning that Q-SUN irradiation increased their effectiveness through the activation of the nanoparticles. It was assumed that ZnO and TiO
2 in the active layer, along with the energy of Q-SUN irradiation, could produce active compounds, such as VOCs or ROCs, which might have created cavities/holes in the Φ 6 phage lipid envelope, inactivating the virus particles. It may be mentioned that FTIR analysis demonstrated that 24 h Q-SUN and UV-B irradiation did not alter the chemical composition of active coatings, which means that ZnO nanoparticles and TiO
2 were responsible for antiviral activity. Additionally, SEM analysis of the active layers demonstrated that PP films were thoroughly and homogenously covered with antiviral coatings and that the accelerated irradiation had no influence on the coatings’ surface morphology. The small breaks and holes, which might release the active agents, were not visible on the surface of the active coatings. It also led to the suggestion that 24 h irradiation activated reactive oxygen species production, and these compounds could not be seen on SEM micrographs. An increase in irradiation time up to 48 h decreased the antiviral properties of the Zn, ZnT1 and ZnT2 coatings, although the coatings were still active. The ZnT3 layer containing ZnO nanoparticles and TiO
2 nanoparticles at a ratio of 1:2 (increased level of TiO
2) demonstrated high antiviral effectiveness towards the phi6 phage. Q-SUN irradiation of the coating for 24 h did not influence its activity, while longer irradiation times (48 h) had a negative effect on its properties. These results led to the conclusion that active coatings based on ZnO and TiO
2 nanoparticles (depending on the amount of TiO
2) could be activated by Q-SUN irradiation but for periods of time no longer than 24 h.
Analyzing the influence of one-day UV-B irradiation on layers and their antiviral properties, it should be mentioned that the Zn, ZnT2 and ZnT3 coatings showed moderate antiviral activity, confirming that UV-B irradiation activated the nanoparticles. It is worth mentioning that the longer UV-B irradiation (48 h) deteriorated the antiviral effectiveness of all analyzed coatings, clearly showing that UV-B irradiation should not exceed 24 h. Alebrahim et al. [
2] also analyzed antiviral coatings containing TiO
2, Cu
2O, TiO
2-Cu
2O and TiO
2-Al
2O
3 nanoparticles. They confirmed that TiO
2-Cu
2O and TiO
2-Al
2O
3-based coatings had higher activity than coatings with TiO
2 and Cu
2O. As confirmed by the authors, the antiviral activity of the coatings appeared to be comparable or slightly enhanced under UVA light compared to simple ambient light. This could lead to the suggestion that UVA irradiation improved the antiviral properties of active coatings. To evaluate the ability of our coatings to offer antiviral properties, HCoV-229E coronavirus (the common cold) was trialed as a surrogate for SARS-CoV-2 by these authors [
2]. Many authors have demonstrated that bacteriophages may be used as model surrogates to replicate eukaryotic viruses. The authors used phage lysates for the development of a model to assess how viruses may be distributed with airborne particles [
6,
7,
28]. Prussin II, A.J [
28] submitted the phi6 phage as a surrogate for COVID-19. This is the reason the phage was selected as SARS-CoV-2 surrogate in the experiments in this study and in our previous work [
6]. To summarize, it seems clear that the healthcare sector should develop antiviral coatings on packaging materials (as an external layer) or on surfaces against fomite transmission, which could limit super-spreading events, as in the case of SARS-CoV-2 particles and other pathogens.
5. Conclusions
The aim of the experiments in this research was to analyze the coating effectiveness of antiviral agents based on gloss varnish containing ZnO and TiO2 nanoparticles. The novelty of this work was to investigate the influence of accelerated UV-B and Q-SUN irradiation on their effectiveness (antiviral activity of the coatings). Comparing ZnO and TiO2 nanoparticles as photoactive metal oxides, it is worth mentioning that both ZnO and TiO2 are excellent semiconductors, which were found to be effective against viruses, such as SARS-CoV-2. Additionally, ZnO nanoparticles were confirmed to have additional shielding properties. On the other hand, UV irradiation may not only activate materials containing these nanoparticles but can also lead to accelerated UV aging. This is why irradiation time was considered as a vitally important parameter to research. The results of our analysis determined that only one in four of the selected coatings containing nanoparticles were highly active against the phi6 phage. It should be reiterated that Q-SUN irradiation had a positive influence on the antiviral activity of the other three coatings. After 24 h of irradiation, the coatings containing lower amounts of TiO2 than the one coating, which was found to eliminate bacteriophage particles, were noted to have moderate antiviral effectiveness, confirming that Q-SUN irradiation improved the properties of the active layers. Unfortunately, 48 h irradiation decreased the activity of the coatings, which confirmed that irradiation time should not be extended too long. Comparing the influence of the UV-B irradiation on the coating activity to the Q-SUN irradiation, it should be noted that 24 h of UV-B irradiation also improved the antiviral properties of the three coatings that were not active before irradiation. However, this decreased the activity of the coating that was shown to be active without UV-B or Q-SUN activation. Unfortunately, none of the analyzed coatings were active after 48 h of UV-B irradiation, confirming that irradiation time has to be shorter. This antiviral analysis was carried out using the phi6 phage, also known as the SARS-CoV-2 surrogate, so it could be suggested that all the coatings, which were confirmed as active against the phi6 phage, could also be effective against COVID-19 particles.