Effect of Intermittent Hypoxic Training on Selected Biochemical Indicators, Blood Rheological Properties, and Metabolic Activity of Erythrocytes in Rowers
Abstract
Simple Summary
Abstract
1. Introduction
2. Material and Methods
2.1. Study Groups
- The experimental group (n = 7) of male competitive rowers training on ergometers under normobaric hypoxia conditions (IHT) (FiO2 = 16.0%), corresponding to an altitude of approximately 2400 m above sea level. The hypoxic conditions were achieved with the use of Everest Summit II hypoxicators. During the training, the athletes breathed through special masks that were connected to generators by air tubes.
- The control group (CON) (n = 7) of male competitive rowers training on ergometers under normoxia conditions (FiO2 = 21%).
2.2. Rowing Performance and Intermittent Hypoxic Training
2.3. Blood Sample Collection
2.4. Morphological, Rheological, and Biochemical Assessments
2.5. Statistical Analysis
3. Results
4. Discussion
Limitation of the Study
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Secher, N.H. The physiology of rowing. J. Sports Sci. 1983, 1, 23–53. [Google Scholar] [CrossRef]
- De Campos Mello, F.; de Moraes Bertuzzi, R.C.; Moreno Grangeiro, P.; Franchini, E. Energy systems contributions in 2,000 m race simulation: A comparison among rowing ergometers and water. Eur. J. Appl. Physiol. 2009, 107, 615–619. [Google Scholar] [CrossRef] [PubMed]
- Hagerman, F.C. Applied physiology of rowing. Sports Med. 1984, 1, 303–326. [Google Scholar] [CrossRef] [PubMed]
- Mäestu, J.; Jürimäe, J.; Jürimäe, T. Monitoring of performance and training in rowing. Sports Med. 2005, 35, 597–617. [Google Scholar] [CrossRef]
- Secher, N.H. Rowing. In Endurance in Sport; Shephard, R.J., Åstrand, P.-O., Eds.; Blackwell Scientific Publications: Oxford, UK, 1992; pp. 563–568. [Google Scholar]
- MacKinnon, L.T. Overtraining effects on immunity and performance in athletes. Immunol. Cell Biol. 2000, 78, 502–509. [Google Scholar] [CrossRef]
- El-Sayed, M.S. Effects of exercise and training on blood rheology. Sports Med. 1998, 26, 281–292. [Google Scholar] [CrossRef]
- Connes, P.; Simmonds, M.J.; Brun, J.-F.; Baskurt, O.K. Exercise hemorheology: Classical data, recent findings and unresolved issues. Clin. Hemorheol. Microcirc. 2013, 53, 187–199. [Google Scholar] [CrossRef]
- Raberin, A.; Burtscher, J.; Connes, P.; Millet, G.P. Hypoxia and hemorheological properties in older individuals. Ageing Res. Rev. 2022, 79, 101650. [Google Scholar] [CrossRef]
- Dufour, S.P.; Ponsot, E.; Zoll, J.; Doutreleau, S.; Lonsdorfer-Wolf, E.; Geny, B.; Lampert, E.; Flück, M.; Hoppeler, H.; Billat, V.; et al. Exercise training in normobaric hypoxia in endurance runners. I. Improvements in aerobic performance capacity. J. Appl. Physiol. 2006, 100, 1238–1248. [Google Scholar] [CrossRef]
- Girard, O.; Brocherie, F.; Goods, P.S.R.; Millet, G.P. An updated panorama of “living low-training high” altitude/hypoxic methods. Front. Sports Act. Living 2020, 2, 26. [Google Scholar] [CrossRef]
- Wilber, R.L. Application of altitude/hypoxic training by elite athletes. Med. Sci. Sports Exerc. 2007, 39, 1610–1624. [Google Scholar] [CrossRef]
- Millet, G.P.; Roels, B.; Schmitt, L.; Woorons, X.; Richalet, J.P. Combining hypoxic methods for peak performance. Sports Med. 2010, 40, 55. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.-W.; Bae, S.-H.; Jeong, J.-W.; Kim, S.-H.; Kim, K.-W. Hypoxia-inducible factor (HIF-1) alpha: Its protein stability and biological functions. Exp. Mol. Med. 2004, 36, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Roels, B.; Bentley, D.J.; Coste, O.; Mercier, J.; Millet, G.P. Effects of intermittent hypoxic training on cycling performance in well-trained athletes. Eur. J. Appl. Physiol. 2007, 101, 359–368. [Google Scholar] [CrossRef] [PubMed]
- Morton, J.P.; Cable, N.T. The effects of intermittent hypoxic training on aerobic and anaerobic performance. Ergonomics 2005, 48, 1535–1546. [Google Scholar] [CrossRef] [PubMed]
- Czuba, M.; Waskiewicz, Z.; Zajac, A.; Poprzecki, S.; Cholewa, J.; Roczniok, R. The effects of intermittent hypoxic training on aerobic capacity and endurance performance in cyclists. J. Sports Sci. Med. 2011, 10, 175–183. [Google Scholar]
- Zoll, J.; Ponsot, E.; Dufour, S.; Doutreleau, S.; Ventura-Clapier, R.; Vogt, M.; Hoppeler, H.; Richard, R.; Flück, M. Exercise training in normobaric hypoxia in endurance runners. III. Muscular adjustments of selected gene transcripts. J. Appl. Physiol. 2006, 100, 1258–1266. [Google Scholar] [CrossRef]
- Czuba, M.; Wilk, R.; Karpiński, J.; Chalimoniuk, M.; Zajac, A.; Langfort, J. Intermittent hypoxic training improves anaerobic performance in competitive swimmers when implemented into a direct competition mesocycle. PLoS ONE 2017, 12, e0180380. [Google Scholar] [CrossRef] [PubMed]
- Hamlin, M.J.; Marshall, C.H.; Hellemans, J.; Ainslie, P.N.; Anglem, N. Effect of intermittent hypoxic training on 20 km time trial and 30 s anaerobic performance. Scand. J. Med. Sci. Sports 2010, 20, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Hinckson, E.A.; Hopkins, W.G.; Downey, B.M.; Smith, T.B.R.J. The effect of intermittent hypoxic training via a hypoxic inhaler on physiological and performance measures in rowers: A pilot study. J. Sci. Med. Sport 2006, 9, 177–180. [Google Scholar] [CrossRef]
- Klusiewicz, A.; Faff, J. Indirect methods of estimating maximal oxygen uptake on the rowing ergometer. Biol. Sport 2003, 20, 181–194. [Google Scholar]
- Jastrzębski, Z.; Żychowska, M. Effects of 6-week specific low-intensity training on selected aerobic capacity parameters and HSPA1A, HSPB1, and LDHb gene expression in high-level rowers. Genet. Mol. Res. 2015, 14, 7538–7547. [Google Scholar] [CrossRef] [PubMed]
- Zoladz, J.A.; Szkutnik, Z.; Majerczak, J.; Duda, K. Detection of the change point in oxygen uptake during an incremental exercise test using recursive residuals: Relationship to the plasma lactate accumulation and blood acid base balance. Eur. J. Appl. Physiol. Occup. Physiol. 1998, 78, 369–377. [Google Scholar] [CrossRef]
- Hardeman, M.R.; Goedhart, P.T.; Dobbe, J.G.G.; Lettinga, K.P. Laser-Assisted Optical Rotational Cell Analyzer (LORCA); I. A new instrument for measurement of various structural hemorheological parameters. Clin. Hemorheol. 1994, 14, 605–618. [Google Scholar]
- Hardeman, M.R.; Dobbe, J.G.G.; Ince, C. The Laser-Assisted Optical Rotational Cell Analyzer (LORCA) as red blood cell aggregometer. Clin. Hemorheol. Microcirc. 2001, 25, 1–11. [Google Scholar] [PubMed]
- Beutler, E. Red Cell Metabolism; Churchill Livingstone: Edinburgh, UK, 1986. [Google Scholar]
- Baskurt, O.K.; Hardeman, M.R.; Rampling, M.W.; Meiselman, H.J. (Eds.) Handbook of Hemorheology and Hemodynamics; IOS Press: Amsterdam, The Netherlands, 2007. [Google Scholar]
- Moon, H.-W.; Shin, S.-H.; Lee, C.-H.; Park, H.-Y.; Sunoo, S.; Nam, S.-S. Effects of various acute hypoxic conditions on the hemorheological response during exercise and recovery. Clin. Hemorheol. Microcirc. 2016, 63, 451–460. [Google Scholar] [CrossRef]
- Monnier, J.F.; Benhaddad, A.A.; Micallef, J.P.; Mercier, J.; Brun, J.F. Relationships between blood viscosity and insulin-like growth factor I status in athletes. Clin. Hemorheol. Microcirc. 2000, 22, 277–286. [Google Scholar]
- Cheng, S.K.; Yu, J.Y.; Si, B.H.; Xiao, Q.L.; Liang, Z.J. Study on the relationship of high hematocrit with changes of the blood viscosity during in the environment of altitude hypoxia. Zhongguo Ying Yong Sheng Li Xue Za Zhi 2001, 17, 231–235. [Google Scholar]
- Grau, M.; Lauten, A.; Hoeppener, S.; Goebel, B.; Brenig, J.; Jung, C.; Bloch, W.; Suhr, F. Regulation of red blood cell deformability is independent of red blood cell-nitric oxide synthase under hypoxia. Clin. Hemorheol. Microcirc. 2016, 63, 199–215. [Google Scholar] [CrossRef]
- Von Tempelhoff, G.-F.; Schelkunov, O.; Demirhan, A.; Tsikouras, P.; Rath, W.; Velten, E.; Csorba, R. Correlation between blood rheological properties and red blood cell indices (MCH, MCV, MCHC) in healthy women. Clin. Hemorheol. Microcirc. 2016, 62, 45–54. [Google Scholar] [CrossRef]
- Hauser, A.; Schmitt, L.; Troesch, S.; Saugy, J.J.; Cejuela-Anta, R.; Faiss, R.; Robinson, N.; Wehrlin, J.P.; Millet, G.P. Similar hemoglobin mass response in hypobaric and normobaric hypoxia in athletes. Med. Sci. Sports Exerc. 2016, 48, 734–741. [Google Scholar] [CrossRef] [PubMed]
- Wehrlin, J.P.; Marti, B. Live high-train low associated with increased haemoglobin mass as preparation for the 2003 World Championships in two native European world class runners. Br. J. Sports Med. 2006, 40, e3. [Google Scholar] [CrossRef] [PubMed]
- Mohanty, J.G.; Nagababu, E.; Rifkind, J.M. Red blood cell oxidative stress impairs oxygen delivery and induces red blood cell aging. Front. Physiol. 2014, 5, 84. [Google Scholar] [CrossRef] [PubMed]
- Rifkind, J.M.; Nagababu, E. Hemoglobin redox reactions and red blood cell aging. Antioxid. Redox Signal. 2013, 18, 2274–2283. [Google Scholar] [CrossRef] [PubMed]
- Pervin, M.; Hasnat, M.A.; Lee, Y.M.; Kim, D.H.; Jo, J.E.; Lim, B.O. Antioxidant activity and acetylcholinesterase inhibition of grape skin anthocyanin (GSA). Molecules 2014, 19, 9403–9418. [Google Scholar] [CrossRef] [PubMed]
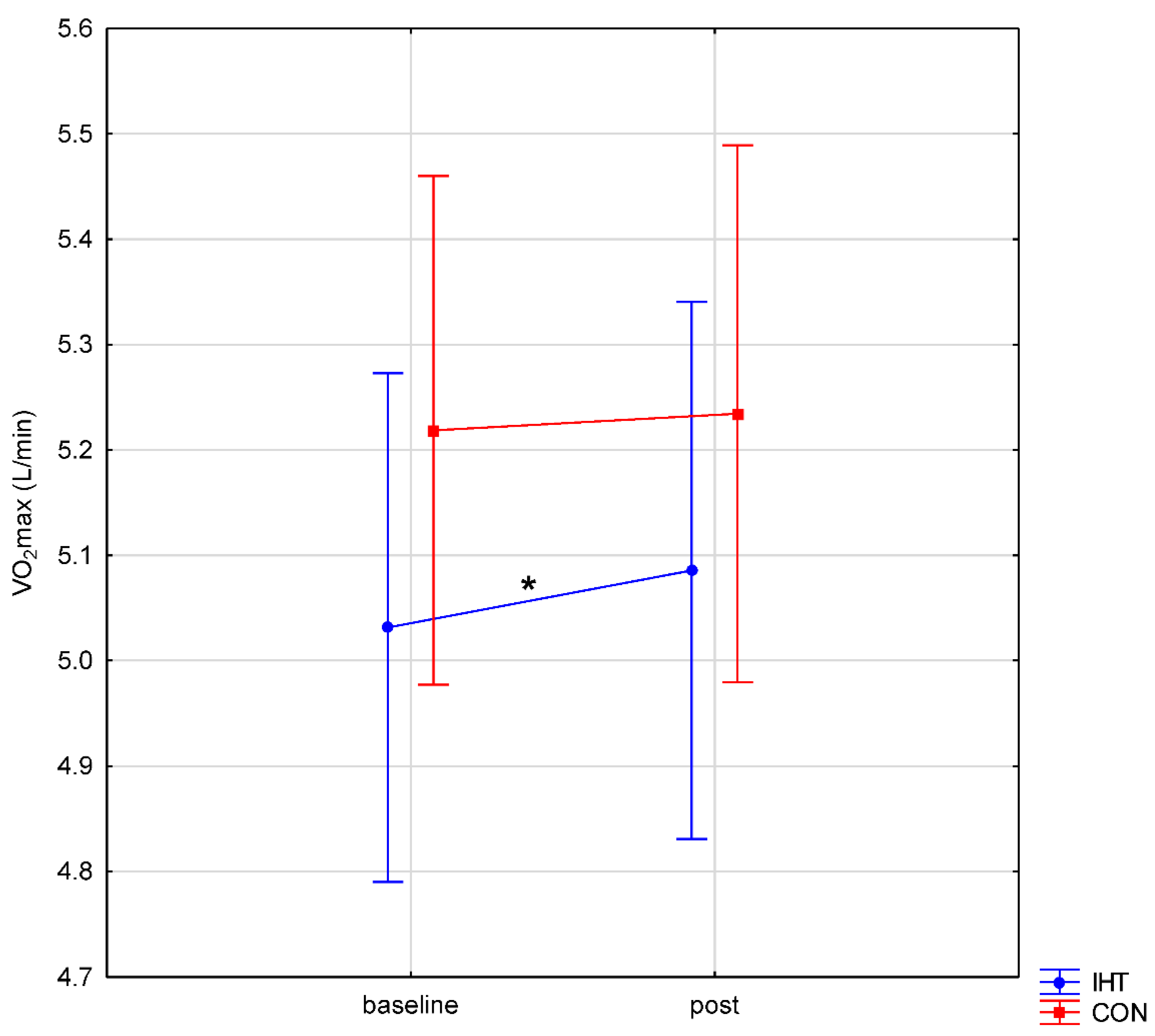

| Group | Age [Years] | BH [cm] | BM [kg] | BMI [kg/m2] | F [%] |
|---|---|---|---|---|---|
| IHT | 22.17 ± 1.83 | 185.5 ± 6.83 | 83.35 ± 9.04 | 24.18 ± 1.65 | 12.62 ± 2.96 |
| CON | 21.5 ± 1.64 | 186.83 ± 7.28 | 84.9 ± 12.14 | 24.22 ± 1.98 | 15.3 ± 2.42 |
| Day of Week | Microcycle 1 | Microcycle 2 | Microcycle 3 |
|---|---|---|---|
| 1 | Morning: rowing ergometer, continuous training, 50 min Afternoon: water training, continuous rowing, 90 min—recovery training (up to 75% HRLT) | Morning: rowing ergometer, continuous training, 50 min Afternoon: gym, strength endurance training, 90 min | Morning: rowing ergometer, continuous training, 50 min Afternoon: water training, continuous rowing, 90 min—recovery training (up to 75% HRLT) |
| 2 | Morning: off Afternoon: team games, basketball + football, 120 min | Morning: general development training, stabilization exercises, 60 min Afternoon: team games, basketball + football, 120 min | Morning: off Afternoon: team games, basketball + football, 120 min |
| 3 | Morning: rowing ergometer, 5 × 12 min Afternoon: gym, maximum strength training (10 exercises), 70 min | Morning: rowing ergometer, 5 × 12 min Afternoon: water training, speed training, 50 min | Morning: rowing ergometer, 5 × 12 min Afternoon: gym, maximum strength training (10 exercises), 70 min |
| 4 | Morning: general development training, stabilization exercises Afternoon: water training, interval training, 75 min | Morning: gym, maximum strength training (10 exercises), 70 min Afternoon: off | Morning: general development training, stabilization exercises Afternoon: water training, interval training, 75 min |
| 5 | Morning: rowing ergometer, continuous training, 50 min Afternoon: off | Morning: rowing ergometer, continuous training, 50 min Afternoon: water training, continuous rowing, 90 min—recovery training (up to 75% HRLT) | Morning: rowing ergometer, continuous training, 50 min Afternoon: off |
| 6 | Morning: cross-country running, 80 min (aerobic endurance training, 75–85% HRLT) Afternoon: strength endurance training, 90 min | Day off | Morning: cross-country running, 80 min (aerobic endurance training, 75–85% HRLT) Afternoon: strength endurance training, 90 min |
| 7 | Day off | Morning: cross-country running, 80 min (aerobic endurance training, 75–85% HRLT) | Day off |
| Variable | Group | Baseline | Post | Effect: Group F (p) | Effect: Time F (p) | Interaction F (p) | Time Change: Pre vs. Post (p) |
|---|---|---|---|---|---|---|---|
| WBC [109/L] | IHT | 8.77 ± 2.56 | 6.37 ± 0.55 | 1.20 (0.3) | 6.84 (0.02) | 2.72 (0.13) | 0.05 * |
| CON | 7.14 ± 0.79 | 6.59 ± 1.08 | 0.90 (NS) | ||||
| BD: post hoc (p) | 0.25 | 0.99 | |||||
| NEUT [109/L] | IHT | 5.06 ± 2.06 | 3.42 ± 0.28 | 2.34 (0.15) | 1.69 (0.22) | 5.18 (0.04) * | 0.11 (NS) |
| CON | 3.27 ± 0.40 | 3.72 ± 0.94 | 0.89 (NS) | ||||
| BD: post hoc (p) | 0.05 * | 0.96 | |||||
| LYM [109/L] | IHT | 3.25 ± 1.57 | 2.09 ± 0.36 | 0.42 (0.53) | 11.25 (0.007) * | 0.25 (0.62) | 0.08 (NS) |
| CON | 2.85 ± 0.41 | 1.99 ± 0.19 | 0.24 (NS) | ||||
| BD: post hoc (p) | 0.84 | 0.99 | |||||
| MONO [109/L] | IHT | 0.72 ± 0.19 | 0.64 ± 0.13 | 0.06 (0.80) | 3.02 (0.11) | 0.43 (0.52) | – |
| CON | 0.68 ± 0.08 | 0.64 ± 0.18 | – | ||||
| BD: post hoc (p) | – | – | |||||
| EOS [109/L] | IHT | 0.15 ± 0.08 | 0.17 ± 0.07 | 2.20 (0.16) | 1.75 (0.21) | 6.78 (0.02) * | 0.17 (NS) |
| CON | 0.29 ± 0.16 | 0.21 ± 0.12 | 0.07 (NS) | ||||
| BD: post hoc (p) | 0.17 | 0.92 | |||||
| BASO [109/L] | IHT | 0.05 ± 0.02 | 0.06 ± 0.02 | 0.31 (0.58) | 0.26 (0.61) | 1.45 (0.25) | – |
| CON | 0.05 ± 0.02 | 0.045 ± 0.02 | – | ||||
| BD: post hoc (p) | – | – | |||||
| RBC [1012/L] | IHT | 5.15 ± 0.2 | 5.14 ± 0.28 | 0.003 (0.95) | 3.08 (0.10) | 0.06 (0.80) | – |
| CON | 5.05 ± 0.2 | 5.07 ± 0.34 | – | ||||
| BD: post hoc (p) | – | – | |||||
| HGB [g/L] | IHT | 14.85 ± 0.59 | 14.73 ± 0.76 | 0.60 (0.45) | 0.90 (0.36) | 0.03 (0.87) | – |
| CON | 15.22 ± 0.82 | 15.05 ± 0.98 | – | ||||
| BD: post hoc (p) | – | – | |||||
| HCT [L/L] | IHT | 43.23 ± 1.72 | 42.96 ± 2.33 | 0.04 (0.83) | 1.36 (0.27) | 0.27 (0.61) | – |
| CON | 43.73 ± 2.67 | 43.03 ± 2.85 | – | ||||
| BD: post hoc (p) | – | – | |||||
| MCV [fL] | IHT | 84.02 ± 3.98 | 85.12 ± 4.34 | 0.04 (0.84) | 2.06 (0.18) | 3.16 (0.10) | – |
| CON | 85.02 ± 2.87 | 84.9 ± 2.5 | – | ||||
| BD: post hoc (p) | – | – | |||||
| MCH [fmol] | IHT | 28.87 ± 1.25 | 29.58 ± 0.75 | 1.40 (0.26) | 1.90 (0.19) | 0.68 (0.42) | – |
| CON | 29.2 ± 0.83 | 29.67 ± 0.67 | – | ||||
| BD: post hoc (p) | – | – | |||||
| MCHC [mmol/L] | IHT | 34.35 ± 0.27 | 34.35 ± 1.06 | 2.11 (0.17) | 0.14 (0.71) | 0.14 (0.71) | – |
| CON | 34.82 ± 0.47 | 35.0 ± 1.04 | – | ||||
| BD: post hoc (p) | – | – | |||||
| RDW-SD [fL] | IHT | 38.68 ± 2.56 | 40.55 ± 3.0 | 3.31 (0.09) | 1.78 (0.21) | 1.04 (0.33) | – |
| CON | 37.32 ± 2.38 | 37.57 ± 1.83 | – | ||||
| BD: post hoc (p) | – | – | |||||
| PLT [109/L] | IHT | 245.67 ± 45.48 | 240 ± 21.94 | 2.22 (0.16) | 0.21 (0.65) | 1.61 (0.23) | – |
| CON | 271.17 ± 38.49 | 283.33 ± 54.2 | – | ||||
| BD: post hoc (p) | – | – | |||||
| MPV [fL] | IHT | 10.63 ± 0.79 | 10.5 ± 0.84 | 0.002 (0.96) | 0.98 (0.34) | 0.02 (0.87) | – |
| CON | 10.63 ± 0.84 | 10.45 ± 1.16 | – | ||||
| BD: post hoc (p) | – | – | |||||
| PCT [%] | IHT | 0.25 ± 0.05 | 0.25 ± 0.05 | 3.76 (0.08) | 1.0 (0.34) | 1.0 (0.34) | – |
| CON | 0.32 ± 0.04 | 0.3 ± 0.06 | – | ||||
| BD: post hoc (p) | – | – | |||||
| PDW [fL] | IHT | 13.02 ± 2.12 | 12.52 ± 1.52 | 0.03 (0.85) | 1.47 (0.25) | 0.13 (0.71) | – |
| CON | 12.68 ± 1.73 | 12.42 ± 2.74 | – | ||||
| BD: post hoc (p) | – | – |
| Variable | Group | Baseline | Post | Effect: Group F (p) | Effect: Time F (p) | Interaction F (p) | Time Change: Pre vs. Post (p) |
|---|---|---|---|---|---|---|---|
| EI at 0.30 Pa | IHT | 0.037 ± 0.01 | 0.037 ± 0.01 | 3.17 (0.10) | 0.70 (0.42) | 0.53 (0.48) | – |
| CON | 0.053 ± 0.01 | 0.047 ± 0.01 | – | ||||
| BD: post hoc (p) | – | – | |||||
| EI at 0.58 Pa | IHT | 0.138 ± 0.01 | 0.142 ± 0.02 | 0.608 (0.45) | 0.709 (0.42) | 3.08 (0.10) | – |
| CON | 0.152 ± 0.01 | 0.143 ± 0.02 | – | ||||
| BD: post hoc (p) | – | – | |||||
| EI at 1.13 Pa | IHT | 0.227 ± 0.01 | 0.232 ± 0.01 | 0.102 (0.75) | 0.06 (0.80) | 1.98 (0.18) | – |
| CON | 0.235 ± 0.01 | 0.229 ± 0.02 | – | ||||
| BD: post hoc (p) | – | – | |||||
| EI at 2.19 Pa | IHT | 0.334 ± 0.01 | 0.341 ± 0.01 | 0.07 (0.93) | 0.98 (0.36) | 2.51 (0.14) | – |
| CON | 0.339 ± 0.01 | 0.337 ± 0.02 | – | ||||
| BD: post hoc (p) | – | – | |||||
| EI at 4.24 Pa | IHT | 0.432 ± 0.01 | 0.441 ± 0.01 | 0.41 (0.53) | 5.45 (0.04) * | 1.40 (0.26) | 0.12 (NS) |
| CON | 0.432 ± 0.01 | 0.435 ± 0.01 | 0.84 (NS) | ||||
| BD: post hoc (p) | 0.99 | 0.70 | |||||
| EI at 8.23 Pa | IHT | 0.506 ± 0.01 | 0.519 ± 0.01 | 0.43 (0.52) | 31.73 (0.0002) * | 1.31 (0.27) | 0.003 * |
| CON | 0.505 ± 0.01 | 0.514 ± 0.01 | 0.04 * | ||||
| BD: post hoc (p) | 0.99 | 0.72 | |||||
| EI at 15.95 Pa | IHT | 0.555 ± 0.01 | 0.570 ± 0.01 | 0.05 (0.82) | 65.55 (0.00001) * | 1.0 (0.34) | 0.0005 * |
| CON | 0.556 ± 0.01 | 0.567 ± 0.01 | 0.002 * | ||||
| BD: post hoc (p) | 0.99 | 0.94 | |||||
| EI at 30.94 Pa | IHT | 0.592 ± 0.01 | 0.608 ± 0.00 | 0.0 (0.88) | 131.4 (0.00) * | 5.4 (0.04) * | 0.0002 * |
| CON | 0.596 ± 0.00 | 0.606 ± 0.00 | 0.0005 * | ||||
| BD: post hoc (p) | 0.88 | 0.93 | |||||
| EI at 60.00 Pa | IHT | 0.619 ± 0.00 | 0.638 ± 0.00 | 0.1 (0.75) | 197.4 (0.00) * | 0.6 (0.47) | 0.0002 * |
| CON | 0.619 ± 0.00 | 0.637 ± 0.00 | 0.0002 * | ||||
| BD: post hoc (p) | 0.27 | 0.69 |
| Variable | Group | Baseline | Post | Effect: Group F (p) | Effect: Time F (p) | Interaction F (p) | Time Change: Pre vs. Post (p) |
|---|---|---|---|---|---|---|---|
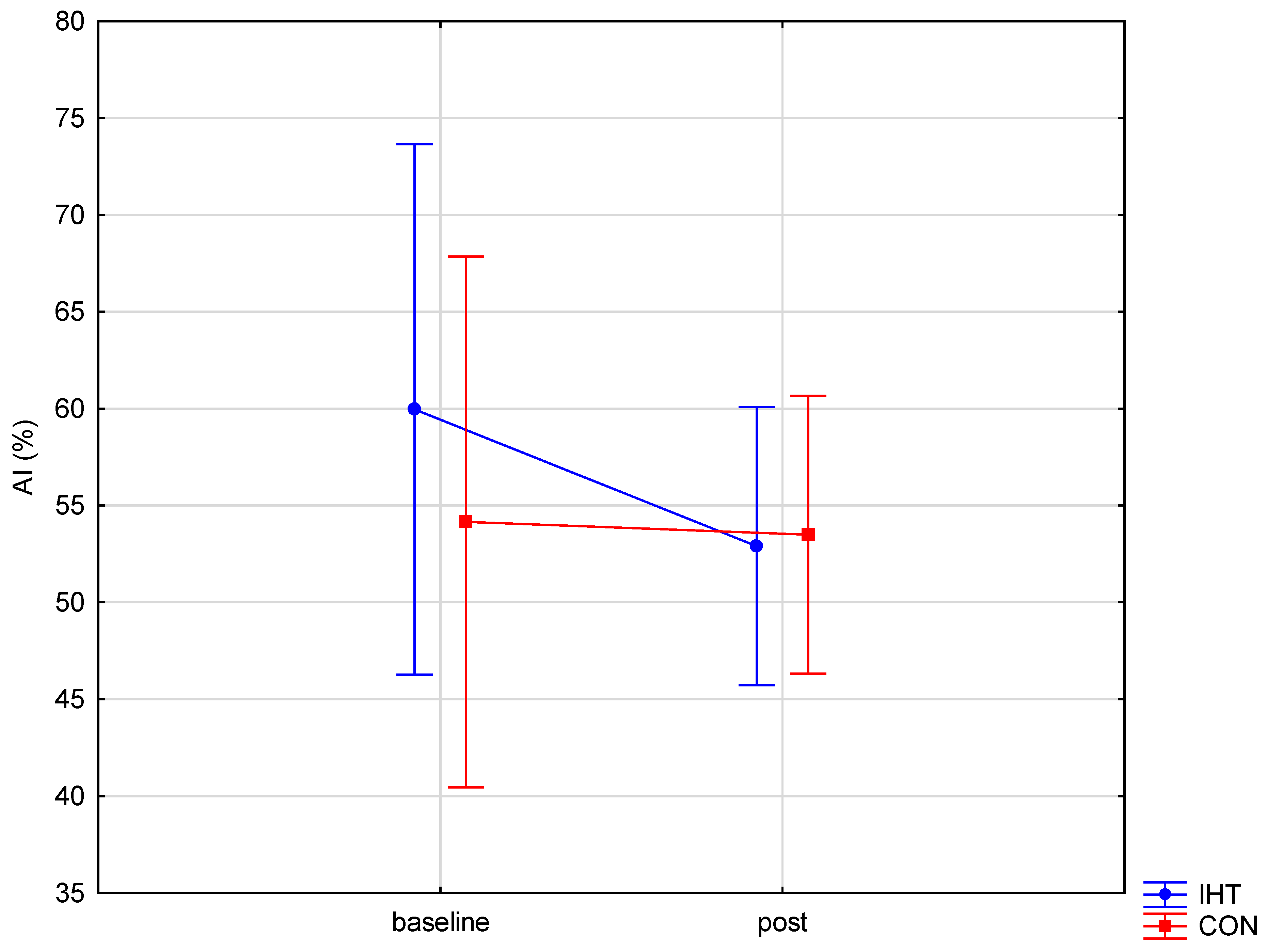
| AI [%] | IHT | 59.96 ± 20.36 | 52.89 ± 7.39 | 0.34 (0.57) | 0.52 (0.48) | 0.36 (0.55) | – |
| CON | 54.15 ± 6.23 | 53.49 ± 8.36 | – | ||||
| BD: post hoc (p) | – | – | |||||
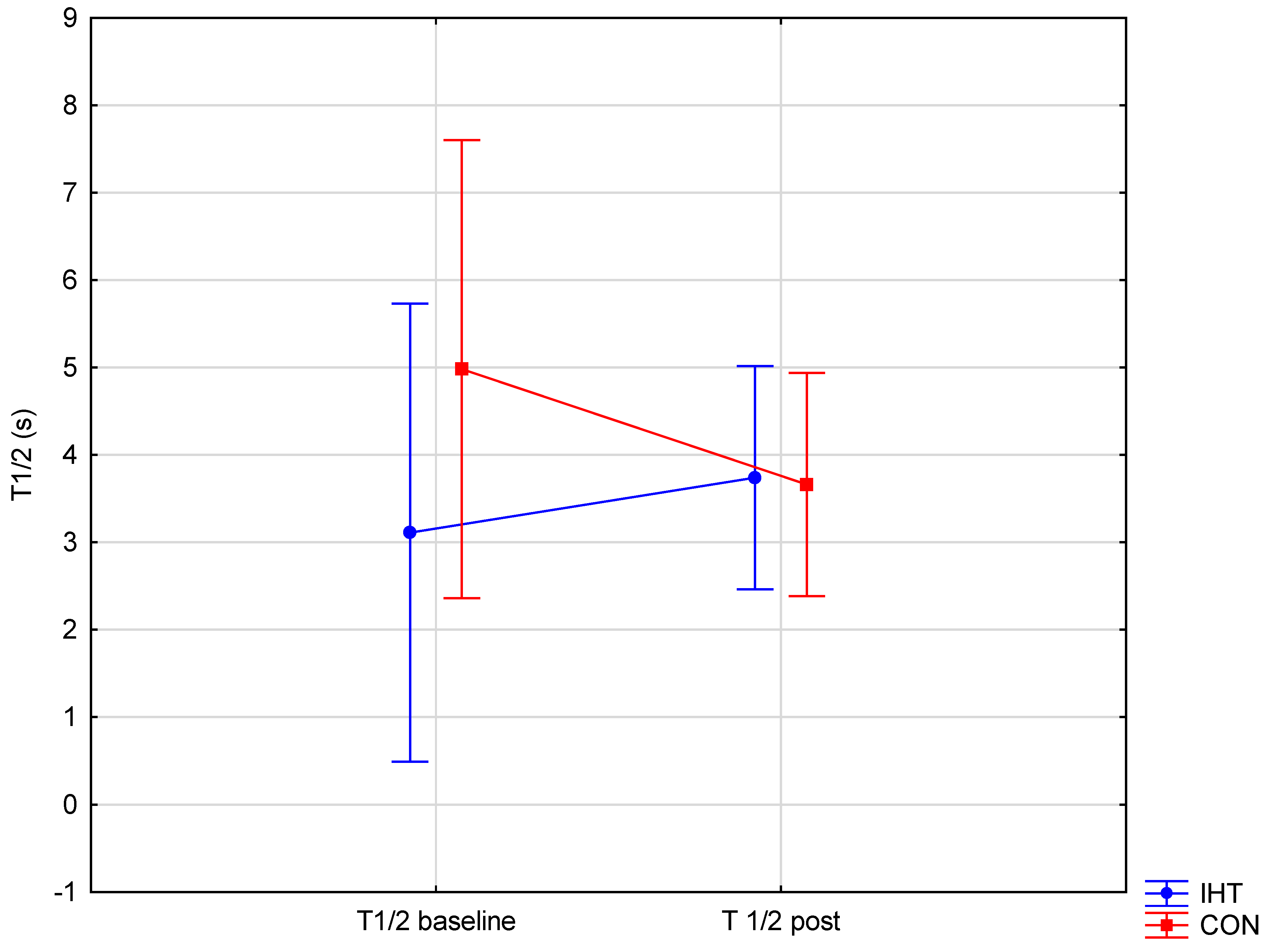
| T1/2 [s] | IHT | 3.11 ± 1.94 | 3.72 ± 1.16 | 1.27 (0.28) | 0.11 (0.74) | 0.88 (0.37) | – |
| CON | 4.98 ± 3.58 | 3.66 ± 1.61 | – | ||||
| BD: post hoc (p) | – | – | |||||
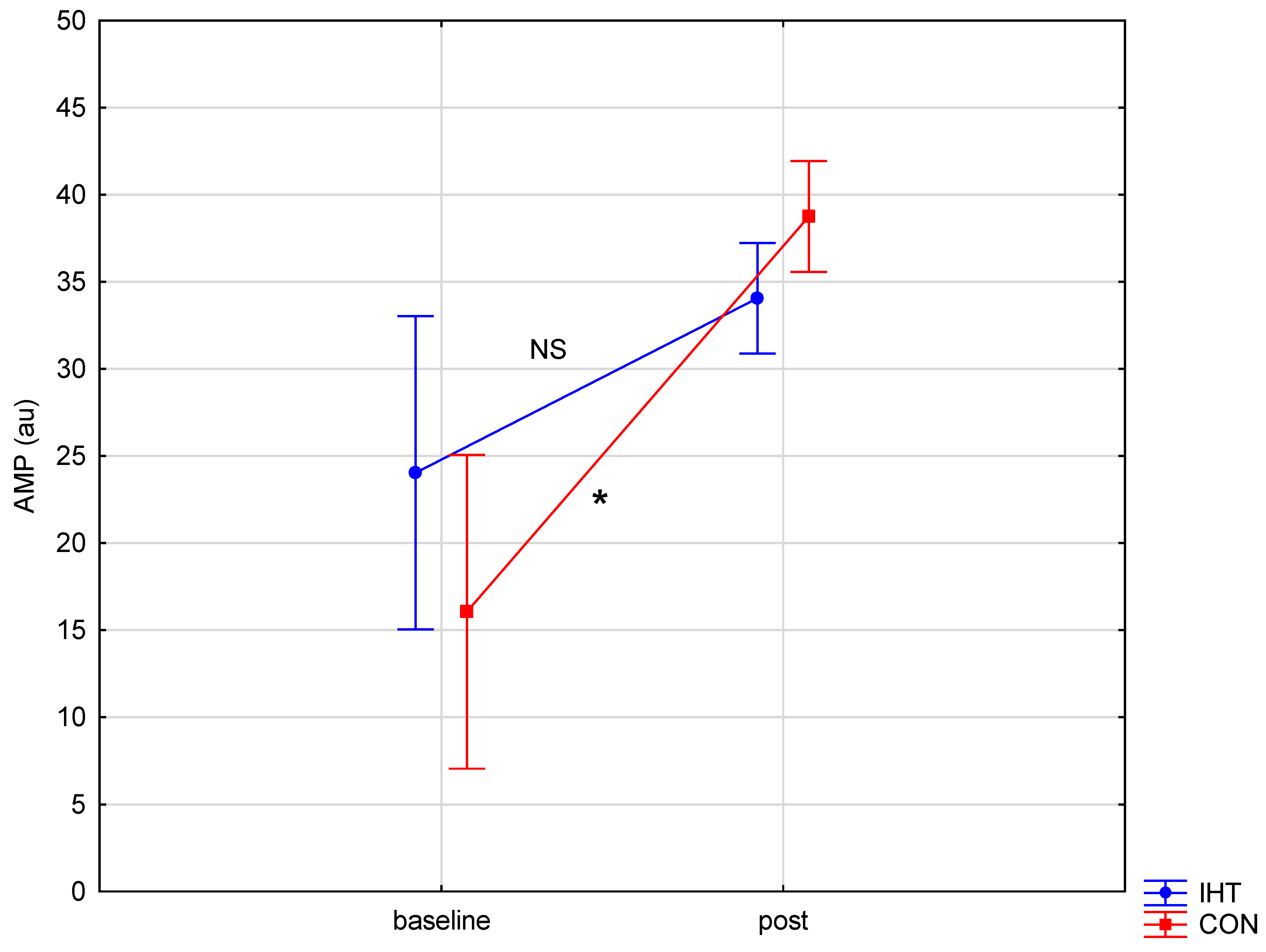
| AMP [au] | IHT | 24.03 ± 12.22 | 34.05 ± 3.57 | 0.26 (0.61) | 32.16 (0.0002) * | 4.82 (0.05) * | 0.12 (NS) |
| CON | 16.05 ± 6.82 | 38.74 ± 3.42 | 0.001 * | ||||
| BD: post hoc (p) | 0.27 | 0.69 |
| Variable | Group | Baseline | Post | Effect: Group F (p) | Effect: Time F (p) | Interaction F (p) | Time Change: Pre vs. Post (p) |
|---|---|---|---|---|---|---|---|
| Fibrinogen [g/L] | IHT | 1.95 ± 0.47 | 2.16 ± 0.24 | 2.37 (0.15) | 2.23 (0.16) | 0.009 (0.92) | – |
| CON | 2.06 ± 0.17 | 2.30 ± 0.22 | – | ||||
| BD: post hoc (p) | – | – |
| Variable | Group | Baseline | Post | Effect: Group F (p) | Effect: Time F (p) | Interaction F (p) | Time Change: Pre vs. Post (p) |
|---|---|---|---|---|---|---|---|
| AChE [IU/g HGB] | IHT | 32.03 ± 5.62 | 36.35 ± 4.04 | 0.26 (0.61) | 6.98 (0.024) * | 1.09 (0.32) | 0.10 (NS) |
| CON | 35.17 ± 1.63 | 37.03 ± 8.69 | 0.68 (NS) | ||||
| BD: post hoc (p) | 0.85 | 0.99 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teległów, A.; Mardyła, M.; Myszka, M.; Pałka, T.; Maciejczyk, M.; Bujas, P.; Mucha, D.; Ptaszek, B.; Marchewka, J. Effect of Intermittent Hypoxic Training on Selected Biochemical Indicators, Blood Rheological Properties, and Metabolic Activity of Erythrocytes in Rowers. Biology 2022, 11, 1513. https://doi.org/10.3390/biology11101513
Teległów A, Mardyła M, Myszka M, Pałka T, Maciejczyk M, Bujas P, Mucha D, Ptaszek B, Marchewka J. Effect of Intermittent Hypoxic Training on Selected Biochemical Indicators, Blood Rheological Properties, and Metabolic Activity of Erythrocytes in Rowers. Biology. 2022; 11(10):1513. https://doi.org/10.3390/biology11101513
Chicago/Turabian StyleTeległów, Aneta, Mateusz Mardyła, Michał Myszka, Tomasz Pałka, Marcin Maciejczyk, Przemysław Bujas, Dariusz Mucha, Bartłomiej Ptaszek, and Jakub Marchewka. 2022. "Effect of Intermittent Hypoxic Training on Selected Biochemical Indicators, Blood Rheological Properties, and Metabolic Activity of Erythrocytes in Rowers" Biology 11, no. 10: 1513. https://doi.org/10.3390/biology11101513
APA StyleTeległów, A., Mardyła, M., Myszka, M., Pałka, T., Maciejczyk, M., Bujas, P., Mucha, D., Ptaszek, B., & Marchewka, J. (2022). Effect of Intermittent Hypoxic Training on Selected Biochemical Indicators, Blood Rheological Properties, and Metabolic Activity of Erythrocytes in Rowers. Biology, 11(10), 1513. https://doi.org/10.3390/biology11101513










