Carboxylated ε-Poly-l-lysine Improves Post-Thaw Quality, Mitochondrial Functions and Antioxidant Defense of Goat Cryopreserved Sperm
Abstract
Simple Summary
Abstract
1. Introduction
2. Materials and Methods
2.1. Chemicals
2.2. Semen Collection, Freezing, and Thawing
2.3. Sperm Motility
2.4. Membrane Integrity and Acrosome Integrity of Sperm
2.5. Mitochondrial Membrane Potential (MMP) of Sperm
2.6. ATP Levels
2.7. Detection of Intracellular Reactive Oxygen Species (ROS)
2.8. Antioxidant and Enzyme Activity in the Thawed Sperm
2.9. Western Blotting Analysis
2.10. Statistical Analysis
3. Results
3.1. Effects of Carboxylated ε-Poly-l-lysine on Sperm Motility Parameters
3.2. Effects of Carboxylated ε-Poly-l-lysine on Sperm Membrane and Acrosome Integrity
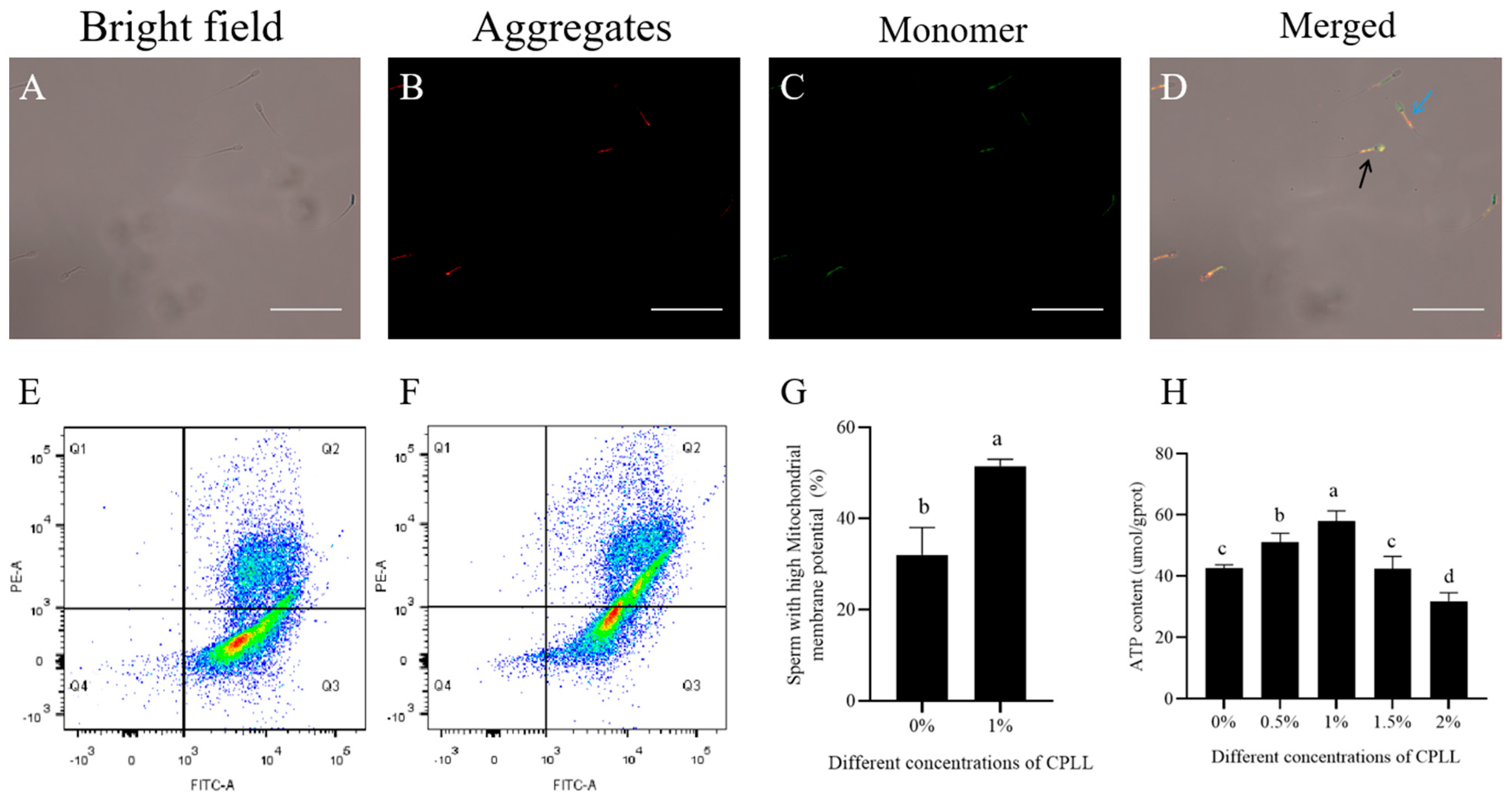
3.3. Effects of Carboxylated ε-Poly-l-lysine on Mitochondrial Membrane Potential and ATP Level
3.4. Effects of Carboxylated ε-Poly-l-lysine on Sperm ROS Level and Malondialdehyde Content
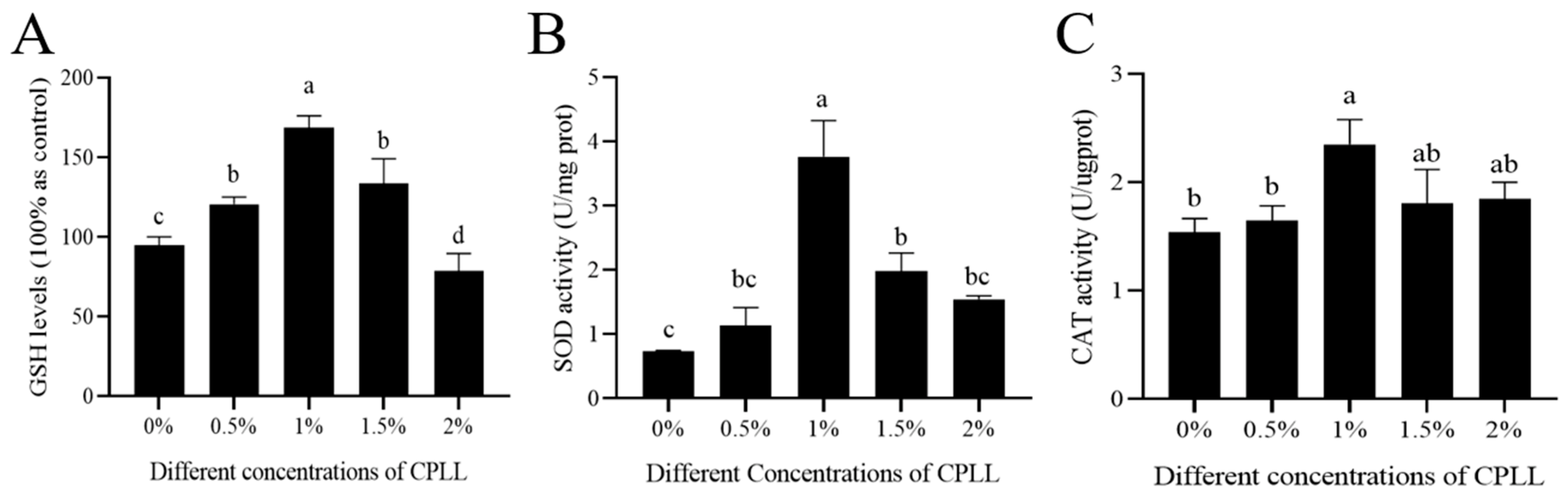
3.5. Effects of Carboxylated ε-Poly-l-lysine on Sperm GSH Levels, SOD and CAT Activity
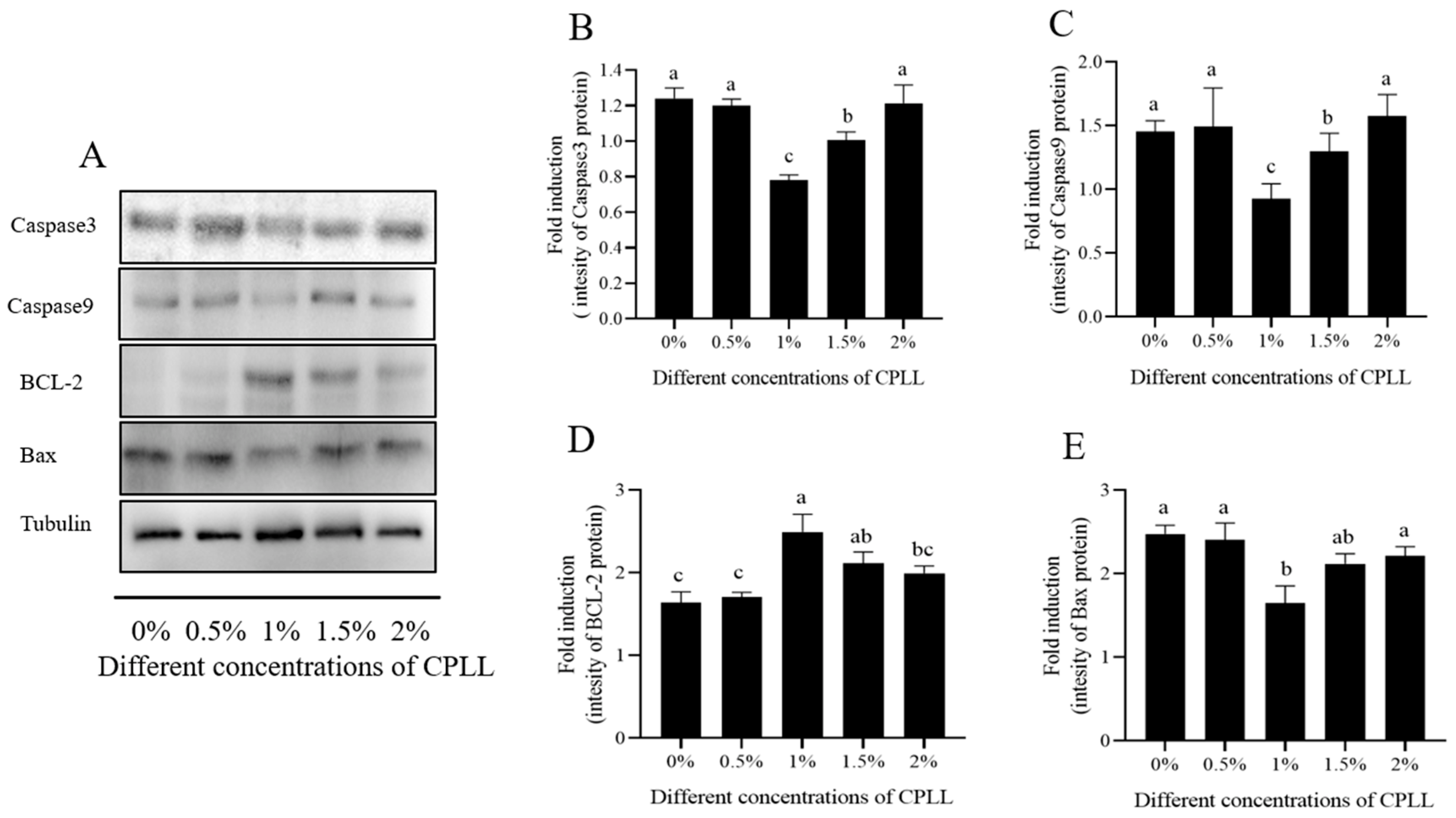
3.6. Effect of Carboxylated ε-Poly-l-lysine on Post-Thaw Sperm Apoptosis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hezavehei, M.; Sharafi, M.; Kouchesfahani, H.M.; Henkel, R.; Agarwal, A.; Esmaeili, V.; Shahverdi, A. Sperm cryopreservation: A review on current molecular cryobiology and advanced approaches. Reprod. BioMed. Online 2018, 37, 327–339. [Google Scholar] [CrossRef]
- Yeste, M. Recent Advances in Boar Sperm Cryopreservation: State of the Art and Current Perspectives. Reprod. Domest. Anim. 2015, 50 (Suppl. S2), 71–79. [Google Scholar] [CrossRef]
- Lavanya, M.; Archana, S.S.; Swathi, D.; Ramya, L.; Arangasamy, A.; Binsila, B.; Dhali, A.; Krishnaswamy, N.; Singh, S.K.; Kumar, H.; et al. Sperm preparedness and adaptation to osmotic and pH stressors relate to functional competence of sperm in Bos taurus. Sci. Rep. 2021, 11, 22563. [Google Scholar] [CrossRef]
- Ming, S.; Tian, J.; Ma, K.; Pei, C.; Li, L.; Wang, Z.; Fang, Z.; Liu, M.; Dong, H.; Li, W.; et al. Oxalate-induced apoptosis through ERS-ROS-NF-kappaB signalling pathway in renal tubular epithelial cell. Mol. Med. 2022, 28, 88. [Google Scholar] [CrossRef]
- Lin, M.; Cao, H.; Meng, Q.; Li, J.; Jiang, P. Insights into the crystallization and vitrification of cryopreserved cells. Cryobiology 2022, 106, 13–23. [Google Scholar] [CrossRef]
- Rajan, R.; Hayashi, F.; Nagashima, T.; Matsumura, K. Toward a Molecular Understanding of the Mechanism of Cryopreservation by Polyampholytes: Cell Membrane Interactions and Hydrophobicity. Biomacromolecules 2016, 17, 1882–1893. [Google Scholar] [CrossRef]
- Arciero, V.; Ammar, O.; Maggi, M.; Vignozzi, L.; Muratori, M.; Dabizzi, S. Vapour fast freezing with low semen volumes can highly improve motility and viability or DNA quality of cryopreserved human spermatozoa. Andrology 2022, 10, 1123–1133. [Google Scholar] [CrossRef]
- Oldenhof, H.; Wolkers, W.F.; Sieme, H. Cryopreservation of Semen from Domestic Livestock: Bovine, Equine, and Porcine Sperm. Methods Mol. Biol. 2021, 2180, 365–377. [Google Scholar] [CrossRef]
- Daramola, J.O.; Adekunle, E.O. Comparative effects of slow freezing and vitrification on cryosurvival of spermatozoa obtained from west African dwarf goat bucks. Cryo Lett. 2016, 37, 123–128. [Google Scholar]
- Tao, Y.; Sanger, E.; Saewu, A.; Leveille, M.C. Human sperm vitrification: The state of the art. Reprod. Biol. Endocrinol. 2020, 18, 17. [Google Scholar] [CrossRef]
- Whaley, D.; Damyar, K.; Witek, R.P.; Mendoza, A.; Alexander, M.; Lakey, J.R. Cryopreservation: An Overview of Principles and Cell-Specific Considerations. Cell Transplant. 2021, 30, 963689721999617. [Google Scholar] [CrossRef] [PubMed]
- Gilmore, J.A.; Liu, J.; Gao, D.Y.; Critser, J.K. Determination of optimal cryoprotectants and procedures for their addition and removal from human spermatozoa. Hum. Reprod. 1997, 12, 112–118. [Google Scholar] [CrossRef]
- Iwatani, M.; Ikegami, K.; Kremenska, Y.; Hattori, N.; Tanaka, S.; Yagi, S.; Shiota, K. Dimethyl sulfoxide has an impact on epigenetic profile in mouse embryoid body. Stem Cells 2006, 24, 2549–2556. [Google Scholar] [CrossRef] [PubMed]
- Shaliutina, O.; Materiienko, A.; Shaliutina-Kolešová, A.; Gazo, I. Using fish spermatozoa in in vitro toxicity tests: A potential toxicology tool. Aquaculture 2021, 539, 736647. [Google Scholar] [CrossRef]
- Rasul, Z.; Ahmed, N.; Anzar, M. Antagonist effect of DMSO on the cryoprotection ability of glycerol during cryopreservation of buffalo sperm. Theriogenology 2007, 68, 813–819. [Google Scholar] [CrossRef]
- Juanpanich, T.; Suttirojpattana, T.; Parnpai, R.; Vutyavanich, T. The relationship between reactive oxygen species, DNA fragmentation, and sperm parameters in human sperm using simplified sucrose vitrification with or without triple antioxidant supplementation. Clin. Exp. Reprod. Med. 2022, 49, 117–126. [Google Scholar] [CrossRef]
- Banihani, S.A.; Alawneh, R.F. Human Semen Samples with High Antioxidant Reservoir May Exhibit Lower Post-Cryopreservation Recovery of Sperm Motility. Biomolecules 2019, 9, 111. [Google Scholar] [CrossRef]
- Qadeer, S.; Khan, M.A.; Ansari, M.S.; Rakha, B.A.; Ejaz, R.; Iqbal, R.; Younis, M.; Ullah, N.; DeVries, A.L.; Akhter, S. Efficiency of antifreeze glycoproteins for cryopreservation of Nili-Ravi (Bubalus bubalis) buffalo bull sperm. Anim. Reprod. Sci. 2015, 157, 56–62. [Google Scholar] [CrossRef]
- Matsumura, K.; Hyon, S.-H. Polyampholytes as low toxic efficient cryoprotective agents with antifreeze protein properties. Biomaterials 2009, 30, 4842–4849. [Google Scholar] [CrossRef]
- Li, S.; Mao, Y.; Zhang, L.; Wang, M.; Meng, J.; Liu, X.; Bai, Y.; Guo, Y. Recent advances in microbial epsilon-poly-l-lysine fermentation and its diverse applications. Biotechnol. Biofuels Bioprod. 2022, 15, 65. [Google Scholar] [CrossRef]
- Nakayama, K.; Yamanaka, T.; Tamada, Y.; Hirabayashi, M.; Hochi, S. Supplementary cryoprotective effect of carboxylated epsilon-poly-l-lysine during vitrification of rat pancreatic islets. Cryobiology 2019, 88, 70–74. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Li, Y.; Zhu, Z. Carboxylated epsilon-Poly-l-Lysine Supplementation of the Freezing Extender Improves the Post-Thawing Boar Sperm Quality. Animals 2022, 12, 1726. [Google Scholar] [CrossRef] [PubMed]
- Fujikawa, T.; Ando, T.; Gen, Y.; Hyon, S.H.; Kubota, C. Cryopreservation of bovine somatic cells using antifreeze polyamino-acid (carboxylated poly-l-lysine). Cryobiology 2017, 76, 140–145. [Google Scholar] [CrossRef]
- Watanabe, H.; Kohaya, N.; Kamoshita, M.; Fujiwara, K.; Matsumura, K.; Hyon, S.H.; Ito, J.; Kashiwazaki, N. Efficient production of live offspring from mouse oocytes vitrified with a novel cryoprotective agent, carboxylated epsilon-poly-l-lysine. PLoS ONE 2013, 8, e83613. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, H.; Nishioka, M.; Maezawa, T.; Kitano, Y.; Terada-Yoshikawa, K.; Tachibana, R.; Kato, M.; Hyon, S.H.; Gen, Y.; Tanaka, K.; et al. Carboxylated Poly-l-Lysine as a Macromolecular Cryoprotective Agent Enables the Development of Defined and Xeno-Free Human Sperm Cryopreservation Reagents. Cells 2021, 10, 1435. [Google Scholar] [CrossRef] [PubMed]
- El Assal, R.; Abou-Elkacem, L.; Tocchio, A.; Pasley, S.; Matosevic, S.; Kaplan, D.L.; Zylberberg, C.; Demirci, U. Bioinspired Preservation of Natural Killer Cells for Cancer Immunotherapy. Adv. Sci. 2019, 6, 1802045. [Google Scholar] [CrossRef]
- Tariq, A.; Ahmad, M.; Iqbal, S.; Riaz, M.I.; Tahir, M.Z.; Ghafoor, A.; Riaz, A. Effect of carboxylated poly l-Lysine as a cryoprotectant on post-thaw quality and in vivo fertility of Nili Ravi buffalo (Bubalus bubalis) bull semen. Theriogenology 2020, 144, 8–15. [Google Scholar] [CrossRef]
- Kamoshita, M.; Kato, T.; Fujiwara, K.; Namiki, T.; Matsumura, K.; Hyon, S.H.; Ito, J.; Kashiwazaki, N. Successful vitrification of pronuclear-stage pig embryos with a novel cryoprotective agent, carboxylated epsilon-poly-l-lysine. PLoS ONE 2017, 12, e0176711. [Google Scholar] [CrossRef]
- Zhang, W.; Min, L.; Li, Y.; Lang, Y.; Hoque, S.A.M.; Adetunji, A.O.; Zhu, Z. Beneficial Effect of Proline Supplementation on Goat Spermatozoa Quality during Cryopreservation. Animals 2022, 12, 2626. [Google Scholar] [CrossRef]
- Zhu, Z.; Li, R.; Fan, X.; Lv, Y.; Zheng, Y.; Hoque, S.A.M.; Wu, D.; Zeng, W. Resveratrol Improves Boar Sperm Quality via 5′AMP-Activated Protein Kinase Activation during Cryopreservation. Oxidative Med. Cell. Longev. 2019, 2019, 5921503. [Google Scholar] [CrossRef]
- Feng, C.; Zhu, Z.; Bai, W.; Li, R.; Zheng, Y.; Tian, X.; Wu, D.; Lu, H.; Wang, Y.; Zeng, W. Proline Protects Boar Sperm against Oxidative Stress through Proline Dehydrogenase-Mediated Metabolism and the Amine Structure of Pyrrolidine. Animals 2020, 10, 1549. [Google Scholar] [CrossRef]
- Li, R.; Wu, X.; Zhu, Z.; Lv, Y.; Zheng, Y.; Lu, H.; Zhou, K.; Wu, D.; Zeng, W.; Dong, W.; et al. Polyamines protect boar sperm from oxidative stress in vitro. J. Anim. Sci. 2022, 100, skac069. [Google Scholar] [CrossRef]
- Zhu, Z.; Kawai, T.; Umehara, T.; Hoque, S.A.M.; Zeng, W.; Shimada, M. Negative effects of ROS generated during linear sperm motility on gene expression and ATP generation in boar sperm mitochondria. Free. Radic. Biol. Med. 2019, 141, 159–171. [Google Scholar] [CrossRef]
- Kumar, A.; Prasad, J.K.; Srivastava, N.; Ghosh, S.K. Strategies to Minimize Various Stress-Related Freeze-Thaw Damages During Conventional Cryopreservation of Mammalian Spermatozoa. Biopreserv. Biobank. 2019, 17, 603–612. [Google Scholar] [CrossRef]
- Saragusty, J.; Gacitua, H.; Rozenboim, I.; Arav, A. Do physical forces contribute to cryodamage? Biotechnol. Bioeng. 2009, 104, 719–728. [Google Scholar] [CrossRef] [PubMed]
- Grotter, L.G.; Cattaneo, L.; Marini, P.E.; Kjelland, M.E.; Ferre, L.B. Recent advances in bovine sperm cryopreservation techniques with a focus on sperm post-thaw quality optimization. Reprod. Domest. Anim. 2019, 54, 655–665. [Google Scholar] [CrossRef]
- Baskaran, A.; Kaari, M.; Venugopal, G.; Manikkam, R.; Joseph, J.; Bhaskar, P.V. Anti freeze proteins (Afp): Properties, sources and applications—A review. Int. J. Biol. Macromol. 2021, 189, 292–305. [Google Scholar] [CrossRef]
- Aitken, R.J.; Roman, S.D. Antioxidant systems and oxidative stress in the testes. Adv. Exp. Med. Biol. 2008, 636, 154–171. [Google Scholar] [CrossRef]
- de Lamirande, E.; Jiang, H.; Zini, A.; Kodama, H.; Gagnon, C. Reactive oxygen species and sperm physiology. Rev. Reprod. 1997, 2, 48–54. [Google Scholar] [CrossRef]
- Zamzami, N.; Marchetti, P.; Castedo, M.; Decaudin, D.; Macho, A.; Hirsch, T.; Susin, S.A.; Petit, P.X.; Mignotte, B.; Kroemer, G. Sequential reduction of mitochondrial transmembrane potential and generation of reactive oxygen species in early programmed cell death. J. Exp. Med. 1995, 182, 367–377. [Google Scholar] [CrossRef]
- Brentnall, M.; Rodriguez-Menocal, L.; De Guevara, R.L.; Cepero, E.; Boise, L.H. Caspase-9, caspase-3 and caspase-7 have distinct roles during intrinsic apoptosis. BMC Cell Biol. 2013, 14, 32. [Google Scholar] [CrossRef]
- Kasapoglu, M.; Ozben, T. Alterations of antioxidant enzymes and oxidative stress markers in aging. Exp. Gerontol. 2001, 36, 209–220. [Google Scholar] [CrossRef]


| Sperm Parameters | 0% | 0.5% | 1% | 1.5% | 2% |
|---|---|---|---|---|---|
| Total motility (%) | 35.85 ± 3.21 b | 39.02 ± 3.95 b | 49.33 ± 2.61 a | 42.13 ± 3.67 b | 37.00 ± 2.48 b |
| Progressive motility (%) | 24.5 ± 1.72 b | 26.98 ± 1.12 b | 35.53 ± 1.54 a | 27.5 ± 1.73 b | 23.50 ± 1.81 b |
| VCL (μm/s) | 34.37 ± 5.48 b | 46.18 ± 3.16 a | 53.8 ± 3.95 a | 47.9 ± 2.37 a | 33.46 ± 1.67 b |
| VSL (μm/s) | 13.45 ± 2.03 c | 24.64 ± 2.14 b | 35.94 ± 4.50 a | 24.85 ± 1.25 b | 16.06 ± 0.7 c |
| VAP (μm/s) | 18.49 ± 3.82 c | 30.09 ± 2.78 b | 40.42 ± 4.13 a | 30.38 ± 1.25 b | 20.84 ± 0.87 c |
| BCF (Hz) | 8.48 ± 0.58 | 8.18 ± 1.47 | 7.57 ± 1.18 | 9.03 ± 0.46 | 9.23 ± 1.01 |
| ALH (μm) | 2.99 ± 0.32 | 3.03 ± 0.22 | 2.88 ± 0.42 | 2.77 ± 0.1 | 2.88 ± 0.08 |
| STR (%) | 78.25 ± 1.05 | 77.9 ± 2.36 | 84.76 ± 3.93 | 84.18 ± 1.52 | 79.03 ± 1.86 |
| LIN (%) | 47.79 ± 1.19 b | 57.11 ± 14.96 a | 57.86 ± 0.73 a | 52.85 ± 3.06 ab | 46.23 ± 1.27 b |
| WOB (%) | 61.08 ± 1.43 b | 65.52 ± 3.36 b | 74.10 ± 1.96 a | 65.82 ± 2.41 b | 66.12 ± 2.46 b |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, W.; Cui, H.; Ding, K.; Zhou, K.; Li, Y.; Hoque, S.A.M.; Min, L.; Zhu, Z. Carboxylated ε-Poly-l-lysine Improves Post-Thaw Quality, Mitochondrial Functions and Antioxidant Defense of Goat Cryopreserved Sperm. Biology 2023, 12, 231. https://doi.org/10.3390/biology12020231
Zhang W, Cui H, Ding K, Zhou K, Li Y, Hoque SAM, Min L, Zhu Z. Carboxylated ε-Poly-l-lysine Improves Post-Thaw Quality, Mitochondrial Functions and Antioxidant Defense of Goat Cryopreserved Sperm. Biology. 2023; 12(2):231. https://doi.org/10.3390/biology12020231
Chicago/Turabian StyleZhang, Weijing, Haixiang Cui, Kexin Ding, Kaifeng Zhou, Yajing Li, S. A. Masudul Hoque, Lingjiang Min, and Zhendong Zhu. 2023. "Carboxylated ε-Poly-l-lysine Improves Post-Thaw Quality, Mitochondrial Functions and Antioxidant Defense of Goat Cryopreserved Sperm" Biology 12, no. 2: 231. https://doi.org/10.3390/biology12020231
APA StyleZhang, W., Cui, H., Ding, K., Zhou, K., Li, Y., Hoque, S. A. M., Min, L., & Zhu, Z. (2023). Carboxylated ε-Poly-l-lysine Improves Post-Thaw Quality, Mitochondrial Functions and Antioxidant Defense of Goat Cryopreserved Sperm. Biology, 12(2), 231. https://doi.org/10.3390/biology12020231






