Diatoms for Carbon Sequestration and Bio-Based Manufacturing
Abstract
:1. Introduction—The Carbon Calamity
2. Diatoms for Bio-Based Manufacturing
3. Carbon Assimilation in Diatoms
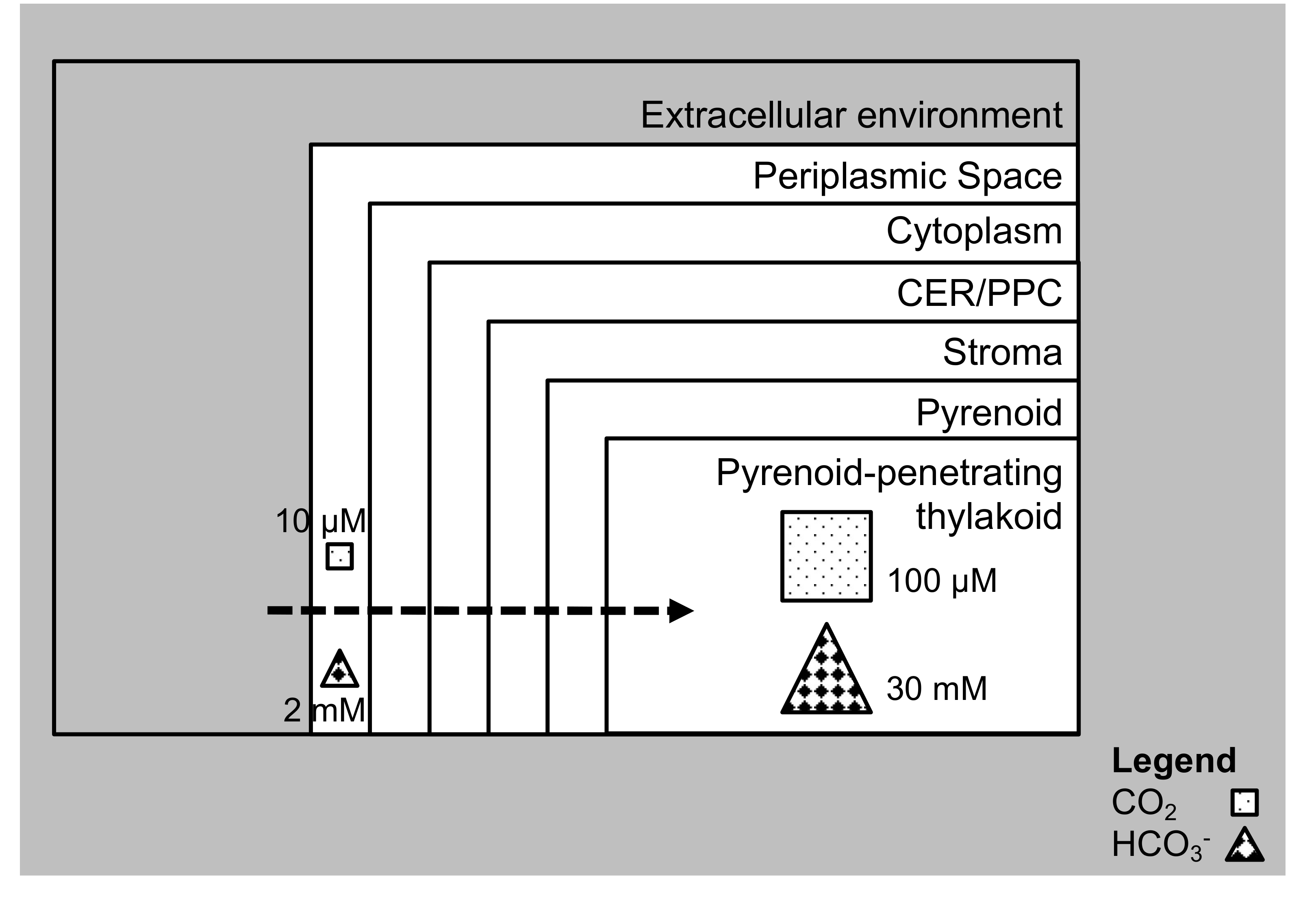
3.1. The Diatom CCM and the Chloroplast Pump Model
3.2. Carbon Transport Systems
3.3. Carbonic Anhydrase-Isoforms and Activation
3.4. Pyrenoid Matrix
3.5. RuBisCO and Its Activation: The Effect of Glycolate
3.6. Evidence of C4 Metabolism
4. Opportunities and Challenges of CO2 Sequestration by Diatoms—Direct Air Capture, Pure CO2 or Flue Gases
5. Bio-Manufacturing with CO2 Uptake
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| CO2 | carbon dioxide |
| CCM | carbon concentrating mechanism |
| CA | carbonic anhydrase |
| RuBisCO | ribulose-1,5-bisphosphate carboxylase/oxygenase |
| PBR | photobioreactor |
| TAG | triacylglycerol |
| DIC | dissolved inorganic carbon |
| SLC4 | solute carrier 4 |
| AQPs | aquaporins |
References
- Friedlingstein, P.; Jones, M.; O’Sullivan, M.; Andrew, R.; Hauck, J.; Peters, G.; Peters, W.; Pongratz, J.; Sitch, S.; Le Quéré, C.; et al. Global carbon budget 2019. Earth Syst. Sci. Data 2019, 11, 1783–1838. [Google Scholar] [CrossRef] [Green Version]
- Peters, G.P.; Andrew, R.M.; Canadell, J.G.; Friedlingstein, P.; Jackson, R.B.; Korsbakken, J.I.; Le Quéré, C.; Peregon, A. Carbon dioxide emissions continue to grow amidst slowly emerging climate policies. Nat. Clim. Chang. 2020, 10, 3–6. [Google Scholar] [CrossRef]
- Le Quéré, C.; Jackson, R.B.; Jones, M.W.; Smith, A.J.; Abernethy, S.; Andrew, R.M.; De-Gol, A.J.; Willis, D.R.; Shan, Y.; Canadell, J.G.; et al. Temporary reduction in daily global CO2 emissions during the COVID-19 forced confinement. Nat. Clim. Chang. 2020, 10, 1–7. [Google Scholar] [CrossRef]
- Jackson, R.B.; Le Quéré, C.; Andrew, R.M.; Canadell, J.G.; Korsbakken, J.I.; Liu, Z.; Peters, G.P.; Zheng, B. Global energy growth is outpacing decarbonization. Environ. Res. Lett. 2018, 13, 12. [Google Scholar] [CrossRef]
- Oelkers, E.H.; Cole, D.R. Carbon dioxide sequestration: A solution to a global problem. Elements 2008, 4, 305–310. [Google Scholar] [CrossRef]
- Blomen, E.; Hendriks, C.; Neele, F. Capture technologies: Improvements and promising developments. Energy Procedia 2009, 1, 1505–1512. [Google Scholar] [CrossRef] [Green Version]
- Luis, P. Use of Monoethanolamine (MEA) for CO2 capture in a global scenario: Consequences and alternatives. Desalination 2016, 380, 93–99. [Google Scholar] [CrossRef] [Green Version]
- Han, K.; Ahn, C.K.; Lee, M.S.; Rhee, C.H.; Kim, J.Y.; Chun, H.D. Current status and challenges of the ammonia-based CO2 capture technologies toward commercialization. Int. J. Greenh. Gas Control 2013, 14, 270–281. [Google Scholar] [CrossRef]
- Björn, L.O.; Govindjee. The evolution of photosynthesis and its environmental impact. In Photobiology: The Science of Light and Life, 3rd ed.; Springer: New York, NY, USA, 2015; pp. 207–230. [Google Scholar] [CrossRef] [Green Version]
- Formighieri, C.; Franck, F.; Bassi, R. Regulation of the pigment optical density of an algal cell: Filling the gap between photosynthetic productivity in the laboratory and in mass culture. J. Biotechnol. 2012, 162, 115–123. [Google Scholar] [CrossRef]
- Melis, A. Solar energy conversion efficiencies in photosynthesis: Minimizing the chlorophyll antennae to maximize efficiency. Plant Sci. 2009, 177, 272–280. [Google Scholar] [CrossRef]
- Escapa, C.; Coimbra, R.N.; Paniagua, S.; García, A.I.; Otero, M. Nutrients and pharmaceuticals removal from wastewater by culture and harvesting of Chlorella sorokiniana. Bioresour. Technol. 2015, 185, 276–284. [Google Scholar] [CrossRef]
- Tsai, D.D.W.; Chen, P.H.; Ramaraj, R. The potential of carbon dioxide capture and sequestration with algae. Ecol. Eng. 2017, 98, 17–23. [Google Scholar] [CrossRef]
- Field, C.B.; Behrenfeld, M.J.; Randerson, J.T.; Falkowski, P. Primary production of the biosphere: Integrating terrestrial and oceanic components. Science 1998, 281, 237–240. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Medlin, L.K. Evolution of the diatoms: Major steps in their evolution and a review of the supporting molecular and morphological evidence. Phycologia 2016, 55, 79–103. [Google Scholar] [CrossRef]
- Sorhannus, U. A nuclear-encoded small-subunit ribosomal RNA timescale for diatom evolution. Mar. Micropaleontol. 2007, 65, 1–12. [Google Scholar] [CrossRef]
- Armbrust, E.V. The Life of Diatoms in the World’s Oceans. Nature 2009, 459, 185–192. [Google Scholar] [CrossRef]
- Pančić, M.; Torres, R.R.; Almeda, R.; Kiørboe, T. Silicified cell walls as a defensive trait in diatoms. Proc. R. Soc. B Biol. Sci. 2019, 286, 20190184. [Google Scholar] [CrossRef] [Green Version]
- Taddei, L.; Stella, G.R.; Rogato, A.; Bailleul, B.; Fortunato, A.E.; Annunziata, R.; Sanges, R.; Thaler, M.; Lepetit, B.; Lavaud, J.; et al. Multisignal control of expression of the LHCX protein family in the marine diatom Phaeodactylum tricornutum. J. Exp. Bot. 2016, 67, 3939–3951. [Google Scholar] [CrossRef] [Green Version]
- Walter, B.; Peters, J.; van Beusekom, J.E. The effect of constant darkness and short light periods on the survival and physiological fitness of two phytoplankton species and their growth potential after re-illumination. Aquat. Ecol. 2017, 51, 591–603. [Google Scholar] [CrossRef]
- Bergkvist, J.; Klawonn, I.; Whitehouse, M.J.; Lavik, G.; Brüchert, V.; Ploug, H. Turbulence simultaneously stimulates small- and large-scale CO2 sequestration by chain-forming diatoms in the sea. Nat. Commun. 2018, 9, 1–10. [Google Scholar] [CrossRef]
- Huisman, J.; Sharples, J.; Stroom, J.M.; Visser, P.M.; Kardinaal, W.E.A.; Verspagen, J.M.H.; Sommeijer, B. Changes in turbulent mixing shift competition for light between phytoplankton species. Ecology 2004, 85, 2960–2970. [Google Scholar] [CrossRef] [Green Version]
- Butler, T.; Kapoore, R.V.; Vaidyanathan, S. Phaeodactylum tricornutum: A Diatom Cell Factory. Trends Biotechnol. 2020, 38, 606–622. [Google Scholar] [CrossRef]
- Granum, E.; Raven, J.A.; Leegood, R.C. How do marine diatoms fix 10 billion tonnes of inorganic carbon per year? Can. J. Bot. 2005, 83, 898–908. [Google Scholar] [CrossRef]
- Tréguer, P.J.; De La Rocha, C.L. The World Ocean Silica Cycle. Annu. Rev. Mar. Sci. 2013, 5, 477–501. [Google Scholar] [CrossRef]
- Wilhelm, C.; Büchel, C.; Fisahn, J.; Goss, R.; Jakob, T.; LaRoche, J.; Lavaud, J.; Lohr, M.; Riebesell, U.; Stehfest, K.; et al. The regulation of carbon and nutrient assimilation in diatoms is significantly different from green algae. Protist 2006, 157, 91–124. [Google Scholar] [CrossRef] [Green Version]
- Raven, J.A.; Waite, A.M. The evolution of silicification in diatoms: Inescapable sinking and sinking as escape? New Phytol. 2004, 162, 45–61. [Google Scholar] [CrossRef]
- Jensen, E.L.; Yangüez, K.; Carrière, F.; Gontero, B. Storage compound accumulation in diatoms as response to elevated CO2 concentration. Biology 2020, 9, 5. [Google Scholar] [CrossRef] [Green Version]
- Burkhardt, S.; Amoroso, G.; Riebesell, U.; Sültemeyer, D. CO2 and HCO3- uptake in marine diatoms acclimated to different CO2 concentrations. Limnol. Oceanogr. 2001, 46, 1378–1391. [Google Scholar] [CrossRef] [Green Version]
- Buono, S.; Colucci, A.; Angelini, A.; Langellotti, A.L.; Massa, M.; Martello, A.; Fogliano, V.; Dibenedetto, A. Productivity and biochemical composition of Tetradesmus obliquus and Phaeodactylum tricornutum: Effects of different cultivation approaches. J. Appl. Phycol. 2016, 28, 3179–3192. [Google Scholar] [CrossRef]
- Gérin, S.; Delhez, T.; Corato, A.; Remacle, C.; Franck, F. A novel culture medium for freshwater diatoms promotes efficient photoautotrophic batch production of biomass, fucoxanthin, and eicosapentaenoic acid. J. Appl. Phycol. 2020, 32, 1–16. [Google Scholar] [CrossRef]
- Guillard, R.R.L.; Lorenzen, C.J. Yellow-Green Algae with Chlorophyllide C 1,2. J. Phycol. 1972, 8, 10–14. [Google Scholar] [CrossRef]
- Guillard, R.R.L. Culture of Phytoplankton for Feeding Marine Invertebrates. In Culture of Marine Invertebrate Animals: Proceedings—1st Conference on Culture of Marine Invertebrate Animals Greenport; Smith, W.L., Chanley, M.H., Eds.; Springer: Boston, MA, USA, 1975; pp. 29–60. [Google Scholar] [CrossRef]
- Gagneux-Moreaux, S.; Moreau, C.; Gonzalez, J.L.; Cosson, R.P. Diatom Artificial Medium (DAM): A New Artificial Medium for the Diatom Haslea ostrearia and Other Marine Microalgae. J. Appl. Phycol. 2007, 19, 549–556. [Google Scholar] [CrossRef] [Green Version]
- Goldman, J.C.; McCarthy, J.J. Steady state growth and ammonium uptake of a fast-growing marine diatom 1. Limnol. Oceanogr. 1978, 23, 695–703. [Google Scholar] [CrossRef]
- Walne, P.R. Studies on the Food Value of Nineteen Genera of Algae to Juvenile Bivalves of the Genera Ostrea, Crassostrea, Mercenaria and Mytilus. Fish. Invest. Ser. 2 1970, 26. [Google Scholar]
- Hao, T.B.; Yang, Y.F.; Balamurugan, S.; Li, D.W.; Yang, W.D.; Li, H.Y. Enrichment of f/2 medium hyperaccumulates biomass and bioactive compounds in the diatom Phaeodactylum tricornutum. Algal Res. 2020, 47, 101872. [Google Scholar] [CrossRef]
- Yang, R.; Wei, D. Improving fucoxanthin production in mixotrophic culture of marine diatom Phaeodactylum tricornutum by LED Light shift and nitrogen supplementation. Front. Bioeng. Biotechnol. 2020, 8, 820. [Google Scholar] [CrossRef]
- Alonso, D.L.; Segura del Castillo, C.I.; Grima, E.M.; Cohen, Z. First insights into improvement of eicosapentaenoic acid content in Phaeodactylum tricornutum (bacillariophyceae) by induced mutagenesis 1. J. Phycol. 1996, 32, 339–345. [Google Scholar] [CrossRef]
- Yi, Z.; Xu, M.; Magnusdottir, M.; Zhang, Y.; Brynjolfsson, S.; Fu, W.; Martin-Jézéquel, V. Photo-Oxidative Stress-Driven Mutagenesis and Adaptive Evolution on the marine diatom Phaeodactylum tricornutum for Enhanced Carotenoid Accumulation. Mar. Drugs 2015, 13, 6138–6151. [Google Scholar] [CrossRef] [Green Version]
- Shifrin, N.S.; Chisholm, S.W. Phytoplankton lipids: Interspecific differences and effects of nitrate, silicate and light-dark cycles. J. Phycol. 1981, 17, 374–384. [Google Scholar] [CrossRef]
- Lewin, J.C. The Taxonomic Position of Phaeodactylum tricornutum. J. Gen. Microbiol. 1958, 18, 427–432. [Google Scholar] [CrossRef] [Green Version]
- Zhao, P.; Gu, W.; Wu, S.; Huang, A.; He, L.; Xie, X.; Gao, S.; Zhang, B.; Niu, J.; Peng Lin, A.; et al. Enhances the Growth of Phaeodactylum tricornutum Bohlin under Green Light and Low Temperature. Sci. Rep. 2014, 4, 3958. [Google Scholar] [CrossRef] [Green Version]
- Dismukes, G.C.; Carrieri, D.; Bennette, N.; Ananyev, G.M.; Posewitz, M.C. Aquatic Phototrophs: Efficient Alternatives to Land-Based Crops for Biofuels. Curr. Opin. Biotechnol. 2008, 19, 235–240. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Said, I.H.; Thorstenson, C.; Thomsen, C.; Ullrich, M.S.; Kuhnert, N.; Thomsen, L. Pilot-Scale Production of Antibacterial Substances by the Marine Diatom Phaeodactylum tricornutum Bohlin. Algal Res. 2018, 32, 113–120. [Google Scholar] [CrossRef]
- Wang, X.; Liu, Y.H.; Wei, W.; Zhou, X.; Yuan, W.; Balamurugan, S.; Hao, T.B.; Yang, W.D.; Liu, J.S.; Li, H.Y. Enrichment of Long-Chain Polyunsaturated Fatty Acids by Coordinated Expression of Multiple Metabolic Nodes in the Oleaginous Microalga Phaeodactylum tricornutum. J. Agric. Food Chem. 2017, 63, 7713–7720. [Google Scholar] [CrossRef]
- Suman, K.; Kiran, T.; Devi, U.K.; Sarma, N.S. Culture Medium Optimization and Lipid Profiling of Cylindrotheca, a Lipid-and Polyunsaturated Fatty Acid-Rich Pennate Diatom and Potential Source of Eicosapentaenoic Acid. Bot. Mar. 2012, 55, 289–299. [Google Scholar] [CrossRef]
- Peltomaa, E.; Hällfors, H.; Taipale, S.J. Comparison of Diatoms and Dinoflagellates from Different Habitats as Sources of PUFAs. Mar. Drugs 2019, 17, 233. [Google Scholar] [CrossRef] [Green Version]
- Krishnan, A.; Anandan, R.; Joseph, A. Culture Medium and Growth Phase Modulate the Fatty Acid Composition of the Diatom Nitzschia Palea (Kutzing) W. Smith-Potential Source for Live Feed and Biodiesel; ICAR: New Delhi, India, 2020. [Google Scholar]
- Sevilla, J.M.F.; García, M.C.C.; Mirón, A.S.; Belarbi, E.H.; Camacho, F.G.; Grima, E.M. Pilot-Plant-Scale Outdoor Mixotrophic Cultures of Phaeodactylum tricornutum Using Glycerol in Vertical Bubble Column and Airlift Photobioreactors: Studies in Fed-Batch Mode. Biotechnol. Prog. 2004, 20, 728–736. [Google Scholar] [CrossRef]
- Indrayani, I.; Moheimani, N.R.; de Boer, K.; Bahri, P.A.; Borowitzka, M.A. Temperature and Salinity Effects on Growth and Fatty Acid Composition of a Halophilic Diatom, Amphora Sp. MUR258 (Bacillariophyceae). J. Appl. Phycol. 2020, 32, 977–987. [Google Scholar] [CrossRef]
- Saxena, A.; Prakash, K.; Phogat, S.; Singh, P.K.; Tiwari, A. Inductively Coupled Plasma Nanosilica Based Growth Method for Enhanced Biomass Production in Marine Diatom Algae. Bioresour. Technol. 2020, 314, 123747. [Google Scholar] [CrossRef] [PubMed]
- Vella, F.M.; Sardo, A.; Gallo, C.; Landi, S.; Fontana, A.; D’Ippolito, G. Annual Outdoor Cultivation of the Diatom Thalassiosira weissflogii: Productivity, Limits and Perspectives. Algal Res. 2019, 42, 101553. [Google Scholar] [CrossRef]
- Indrayani, I.; Haslianti, H.; Asmariani, A.; Muskita, W.; Balubi, A.M. Growth, Biomass and Lipid Productivity of a Newly Isolated Tropical Marine Diatom, Skeletonema Sp. Uho29, under Different Light Intensities. Biodiversitas 2020, 21. [Google Scholar] [CrossRef]
- Wen, Z.Y.; Chen, F. Production Potential of Eicosapentaenoic Acid by the Diatom. Nitzschia laevis. Biotechnol. Lett. 2000, 22, 727–733. [Google Scholar] [CrossRef]
- Gao, B.; Chen, A.; Zhang, W.; Li, A.; Zhang, C. Co-Production of Lipids, Eicosapentaenoic Acid, Fucoxanthin, and Chrysolaminarin by Phaeodactylum tricornutum Cultured in a Flat-Plate Photobioreactor under Varying Nitrogen Conditions. J. Ocean Univ. China 2017, 16, 916–924. [Google Scholar] [CrossRef]
- Xia, S.; Gao, B.; Fu, J.; Xiong, J.; Zhang, C. Production of fucoxanthin, chrysolaminarin, and eicosapentaenoic acid by Odontella aurita under different nitrogen supply regimes. J. Biosci. Bioeng. 2018, 126, 723–729. [Google Scholar] [CrossRef] [PubMed]
- Hamilton, M.L.; Warwick, J.; Terry, A.; Allen, M.J.; Napier, J.A.; Sayanova, O. Towards the Industrial Production of Omega-3 Long Chain Polyunsaturated Fatty Acids from a Genetically Modified Diatom Phaeodactylum tricornutum. PLoS ONE 2015, 10, e0144054. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, T.; Yabuuchi, T.; Maeda, Y.; Nojima, D.; Matsumoto, M.; Yoshino, T. Production of Eicosapentaenoic Acid by High Cell Density Cultivation of the Marine Oleaginous Diatom Fistulifera solaris. Bioresour. Technol. 2017, 245, 567–572. [Google Scholar] [CrossRef]
- Marella, T.K.; Tiwari, A. Marine Diatom Thalassiosira weissflogii Based Biorefinery for Co-Production of Eicosapentaenoic Acid and Fucoxanthin. Bioresour. Technol. 2020, 307, 123245. [Google Scholar] [CrossRef]
- Slocombe, S.P.; Zhang, Q.; Ross, M.; Anderson, A.; Thomas, N.J.; Lapresa, Á.; Rad-Menéndez, C.; Campbell, C.N.; Black, K.D.; Stanley, M.S.; et al. Unlocking Nature’s Treasure-Chest: Screening for Oleaginous Algae. Sci. Rep. 2015, 5, 9844. [Google Scholar] [CrossRef]
- Widyaningrum, D.; Meindrawan, B. The Application of Microbial Extracellular Polymeric Substances in Food Industry. In Proceedings of the IOP Conference Series: Earth and Environmental Science, Solo, Indonesia, 13–14 November 2019. [Google Scholar] [CrossRef]
- Kim, S.M.; Kang, S.W.; Kwon, O.N.; Chung, D.; Pan, C.H. Fucoxanthin as a Major Carotenoid in Isochrysis aff. galbana: Characterization of Extraction for Commercial Application. J. Korean Soc. Appl. Biol. Chem. 2012, 55, 477–483. [Google Scholar] [CrossRef]
- Petrushkina, M.; Gusev, E.; Sorokin, B.; Zotko, N.; Mamaeva, A.; Filimonova, A.; Kulikovskiy, M.; Maltsev, Y.; Yampolsky, I.; Guglya, E.; et al. Fucoxanthin production by heterokont microalgae. Algal Res. 2017, 24, 387–393. [Google Scholar] [CrossRef]
- McClure, D.D.; Luiz, A.; Gerber, B.; Barton, G.W.; Kavanagh, J.M. An Investigation into the Effect of Culture Conditions on Fucoxanthin Production Using the Marine Microalgae Phaeodactylum tricornutum. Algal Res. 2018, 29, 41–48. [Google Scholar] [CrossRef]
- Xia, S.; Wang, K.; Wan, L.; Li, A.; Hu, Q.; Zhang, C. Production, Characterization, and Antioxidant Activity of Fucoxanthin from the Marine Diatom Odontella aurita. Mar. Drugs 2013, 11, 2667–2681. [Google Scholar] [CrossRef] [PubMed]
- Pasquet, V.; Chérouvrier, J.R.; Farhat, F.; Thiéry, V.; Piot, J.M.; Bérard, J.B.; Kaas, R.; Serive, B.; Patrice, T.; Cadoret, J.P.; et al. Study on the microalgal pigments extraction process: Performance of Microwave Assisted Extraction. Process Biochem. 2011, 46, 59–67. [Google Scholar] [CrossRef] [Green Version]
- Rodolfi, L.; Biondi, N.; Guccione, A.; Bassi, N.; D’Ottavio, M.; Arganaraz, G.; Tredici, M.R. Oil and eicosapentaenoic acid production by the diatom Phaeodactylum tricornutum cultivated outdoors in Green Wall Panel (GWP®) Reactors. Biotechnol. Bioeng. 2017, 114, 2204–2210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- D’Ippolito, G.; Sardo, A.; Paris, D.; Vella, F.M.; Adelfi, M.G.; Botte, P.; Gallo, C.; Fontana, A. Potential of lipid metabolism in marine diatoms for biofuel production. Biotechnol. Biofuels 2015, 8, 28. [Google Scholar] [CrossRef] [Green Version]
- Orcutt, D.M.; Patterson, G.W. Sterol, Fatty Acid and Elemental Composition of Diatoms Grown in Chemically Defined Media. Comp. Biochem. Physiol. Part B Biochem. 1975, 50, 579–583. [Google Scholar] [CrossRef]
- Renaud, S.M.; Thinh, L.V.; Lambrinidis, G.; Parry, D.L. Effect of temperature on growth, chemical composition and fatty acid composition of tropical australian microalgae grown in batch cultures. Aquaculture 2002, 211, 195–214. [Google Scholar] [CrossRef]
- Ruocco, N.; Albarano, L.; Esposito, R.; Zupo, V.; Costantini, M.; Ianora, A. Multiple roles of diatom-derived oxylipins within marine environments and their potential biotechnological applications. Mar. Drugs 2020, 18, 342. [Google Scholar] [CrossRef]
- Jaramillo-Madrid, A.C.; Ashworth, J.; Ralph, P.J. Levels of diatom minor sterols respond to changes in temperature and salinity. J. Mar. Sci. Eng. 2020, 8, 85. [Google Scholar] [CrossRef] [Green Version]
- Panwar, V.; Dutta, T. Diatom biogenic silica as a felicitous platform for biochemical engineering: Expanding frontiers. ACS Appl. Mater. 2019, 2, 2295–2316. [Google Scholar] [CrossRef]
- Nur, M.M.A.; Swaminathan, M.K.; Boelen, P.; Buma, A.G.J. Sulfated exopolysaccharide production and nutrient removal by the marine diatom Phaeodactylum tricornutum growing on palm oil mill effluent. J. Appl. Phycol. 2019, 31, 2335–2348. [Google Scholar] [CrossRef] [Green Version]
- Hempel, F.; Bozarth, A.S.; Lindenkamp, N.; Klingl, A.; Zauner, S.; Linne, U.; Steinbüchel, A.; Maier, U.G. Microalgae as bioreactors for bioplastic production. Microb. Cell Factories 2011, 10, 81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hempel, F.; Lau, J.; Klingl, A.; Maier, U.G. Algae as Protein Factories: Expression of a Human Antibody and the Respective Antigen in the Diatom Phaeodactylum tricornutum. PLoS ONE 2011, 6, 28424. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hempel, F.; Maier, U.G. An Engineered Diatom Acting like a Plasma Cell Secreting Human IgG Antibodies with High Efficiency. Microb. Cell Factories 2012, 11, 126. [Google Scholar] [CrossRef] [Green Version]
- Hempel, F.; Maurer, M.; Brockmann, B.; Mayer, C.; Biedenkopf, N.; Kelterbaum, A.; Becker, S.; Maier, U.G. From Hybridomas to a Robust Microalgal-Based Production Platform: Molecular Design of a Diatom Secreting Monoclonal Antibodies Directed against the Marburg Virus Nucleoprotein. Microb. Cell Factories 2017, 16, 131. [Google Scholar] [CrossRef]
- Poschenrieder, C.; Fernández, J.A.; Rubio, L.; Pérez, L.; Terés, J.; Barceló, J. Transport and Use of Bicarbonate in Plants: Current Knowledge and Challenges Ahead. Int. J. Mol. Sci. 2018, 19, 1352. [Google Scholar] [CrossRef] [Green Version]
- Battin, T.J.; Luyssaert, S.; Kaplan, L.A.; Aufdenkampe, A.K.; Richter, A.; Tranvik, L.J. The Boundless Carbon Cycle. Nat. Geosci. 2009, 2, 598–600. [Google Scholar] [CrossRef]
- Hopkinson, B.M.; Dupont, C.L.; Matsuda, Y. The Physiology and Genetics of CO2 Concentrating Mechanisms in Model Diatoms. Curr. Opin. Plant Biol. 2016, 31, 51–57. [Google Scholar] [CrossRef] [Green Version]
- Piiparinen, J.; Barth, D.; Eriksen, N.T.; Teir, S.; Spilling, K.; Wiebe, M.G. Microalgal CO2 capture at extreme PH values. Algal Res. 2018, 32, 321–328. [Google Scholar] [CrossRef]
- Meiser, A.; Schmid-Staiger, U.; Trösch, W. Optimization of Eicosapentaenoic Acid Production by Phaeodactylum tricornutum in the Flat Panel Airlift (FPA) Reactor. J. Appl. Phycol. 2004, 16, 215–225. [Google Scholar] [CrossRef]
- Negoro, M.; Shioji, N.; Miyamoto, K.; Micira, Y. Growth of microalgae in high CO2 gas and effects of SOX and NOX. Appl. Biochem. Biotechnol. 1991, 28–29, 877–886. [Google Scholar] [CrossRef] [PubMed]
- Clement, R.; Jensen, E.; Prioretti, L.; Maberly, S.C.; Gontero, B. Diversity of CO2-Concentrating Mechanisms and Responses to CO2 Concentration in Marine and Freshwater Diatoms. J. Exp. Bot. 2017, 68, 3925–3935. [Google Scholar] [CrossRef] [PubMed]
- Artamonova, E.Y.; Vasskog, T.; Eilertsen, H.C. Lipid Content and Fatty Acid Composition of Porosira Glacialis and Attheya Longicornis in Response to Carbon Dioxide (CO2) Aeration. PLoS ONE 2017, 12, e0177703. [Google Scholar] [CrossRef] [Green Version]
- Baragi, L.V.; Khandeparker, L.; Anil, A.C. Influence of elevated temperature and pCO2 on the marine periphytic diatom Navicula distans and its associated organisms in culture. Hydrobiologia 2015, 762, 127–142. [Google Scholar] [CrossRef]
- Boelen, P.; van de Poll, W.H.; van der Strate, H.J.; Neven, I.A.; Beardall, J.; Buma, A.G.J. Neither Elevated nor reduced CO2 affects the photophysiological performance of the marine antarctic diatom Chaetoceros brevis. J. Exp. Mar. Biol. Ecol. 2011, 406, 38–45. [Google Scholar] [CrossRef]
- Fettke, J.; Fernie, A.R. Intracellular and Cell-to-Apoplast Compartmentation of Carbohydrate Metabolism. Trends Plant Sci. 2015, 20, 490–497. [Google Scholar] [CrossRef]
- Smith, A.M.; Stitt, M. Coordination of carbon supply and plant growth. Plant Cell Environ. 2007, 30, 1126–1149. [Google Scholar] [CrossRef]
- Badger, M.R.; Andrews, T.J.; Whitney, S.M.; Ludwig, M.; Yellowlees, D.C.; Leggat, W.; Price, G.D. The diversity and coevolution of Rubisco, Plastids, Pyrenoids, and Chloroplast-Based CO2-concentrating mechanisms in algae. Can. J. Bot. 1998, 76, 1052–1071. [Google Scholar] [CrossRef]
- Kustka, A.B.; Milligan, A.J.; Zheng, H.; New, A.M.; Gates, C.; Bidle, K.D.; Reinfelder, J.R. Low CO2 results in a rearrangement of carbon metabolism to support C4 photosynthetic carbon assimilation in Thalassiosira pseudonana. New Phytol. 2014, 204, 507–520. [Google Scholar] [CrossRef]
- Matthijs, M.; Fabris, M.; Obata, T.; Foubert, I.; Franco-Zorrilla, J.M.; Solano, R.; Fernie, A.R.; Vyverman, W.; Goossens, A. The Transcription Factor BZIP14 regulates the TCA Cycle in the diatom Phaeodactylum tricornutum. EMBO J. 2017, 36, 1559–1576. [Google Scholar] [CrossRef] [Green Version]
- Patel, B.N.; Merrett, M.J. Inorganic-Carbon Uptake by the Marine Diatom Phaeodactylum tricornutum. Planta 1986, 169, 222–227. [Google Scholar] [CrossRef] [PubMed]
- Hopkinson, B.M. A chloroplast pump model for the CO2 Concentrating mechanism in the diatom Phaeodactylum tricornutum. Photosynth. Res. 2014, 121, 223–233. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.; Sun, J.; Wunder, T.; Tang, D.; Cousins, A.B.; Sze, S.K.; Mueller-Cajar, O.; Gao, Y.G. Structural Insights into the LCIB protein family reveals a new group of β-Carbonic anhydrases. Proc. Natl. Acad. Sci. USA 2016, 113, 14716–14721. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leegood, R.C. C4 Photosynthesis: Principles of CO2 Concentration and Prospects for Its Introduction into C3 Plants. J. Exp. Bot. 2002, 53, 581–590. [Google Scholar] [CrossRef]
- Matsuda, Y.; Hara, T.; Colman, B. Regulation of the Induction of bicarbonate uptake by Dissolved CO2 in the marine diatom, Phaeodactylum tricornutum. Plant Cell Environ. 2001, 24, 611–620. [Google Scholar] [CrossRef]
- Nakajima, K.; Tanaka, A.; Matsuda, Y. SLC4 Family transporters in a marine diatom directly pump bicarbonate from seawater. Proc. Natl. Acad. Sci. USA 2013, 110, 1767–1772. [Google Scholar] [CrossRef] [Green Version]
- Matsuda, Y.; Hopkinson, B.M.; Nakajima, K.; Dupont, C.L.; Tsuji, Y. Mechanisms of carbon dioxide acquisition and CO2 sensing in marine diatoms: A gateway to carbon metabolism. Philos. Trans. R. Soc. B Biol. Sci. 2017, 372, 20120403. [Google Scholar] [CrossRef] [Green Version]
- Hopkinson, B.M.; Dupont, C.L.; Allen, A.E.; Morel, F.M.M. Efficiency of the CO2-Concentrating mechanism of diatoms. Proc. Natl. Acad. Sci. USA 2011, 108, 3830–3837. [Google Scholar] [CrossRef] [Green Version]
- Tachibana, M.; Allen, A.E.; Kikutani, S.; Endo, Y.; Bowler, C.; Matsuda, Y. Localization of putative Carbonic anhydrases in two marine diatoms, Phaeodactylum tricornutum and Thalassiosira pseudonana. Photosynth. Res. 2011, 109, 205–221. [Google Scholar] [CrossRef]
- Samukawa, M.; Shen, C.; Hopkinson, B.M.; Matsuda, Y. Localization of Putative Carbonic Anhydrases in the Marine Diatom, Thalassiosira pseudonana. Photosynth. Res. 2014, 121, 235–249. [Google Scholar] [CrossRef]
- Liu, Y.; Yang, J.; Chen, L.M. Structure and Function of SLC4 Family HCO3- Transporters. Front. Physiol. 2015, 6, 355. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shen, C.; Dupont, C.L.; Hopkinson, B.M. The Diversity of CO2-Concentrating Mechanisms in Marine Diatoms as Inferred from Their Genetic Content. J. Exp. Bot. 2017, 68, 3937–3948. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsuji, Y.; Nakajima, K.; Matsuda, Y. Molecular Aspects of the Biophysical CO2-Concentrating Mechanism and Its Regulation in Marine Diatoms. J. Exp. Bot. 2017, 68, 3763–3772. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matsui, H.; Hopkinson, B.M.; Nakajima, K.; Matsuda, Y. Plasma Membrane-Type Aquaporins from Marine Diatoms Function as CO2/NH3 Channels and Provide Photoprotection. Plant Physiol. 2018, 178, 345–357. [Google Scholar] [CrossRef] [Green Version]
- Moroney, J.V.; Bartlett, S.G.; Samuelsson, G. Carbonic anhydrases in plants and algae. Plant Cell Environ. 2001, 24, 141–153. [Google Scholar] [CrossRef]
- Montsant, A.; Jabbari, K.; Maheswari, U.; Bowler, C. Comparative Genomics of the Pennate Diatom Phaeodactylum tricornutum. Plant Physiol. 2005, 137, 500–513. [Google Scholar] [CrossRef] [Green Version]
- Kikutani, S.; Nakajima, K.; Nagasato, C.; Tsuji, Y.; Miyatake, A.; Matsuda, Y. Thylakoid Luminal Θ-Carbonic anhydrase critical for growth and photosynthesis in the marine diatom Phaeodactylum tricornutum. Proc. Natl. Acad. Sci. USA 2016, 113, 9828–9833. [Google Scholar] [CrossRef] [Green Version]
- Alterio, V.; Langella, E.; De Simone, G.; Monti, S.M. Cadmium-containing Carbonic anhydrase CDCA1 in Marine Diatom Thalassiosira weissflogii. Mar. Drugs 2015, 13, 1688–1697. [Google Scholar] [CrossRef] [Green Version]
- Xu, Y.; Feng, L.; Jeffrey, P.D.; Shi, Y.; Morel, F.M.M. Structure and Metal Exchange in the Cadmium Carbonic Anhydrase of Marine Diatoms. Nature 2008, 452, 55–61. [Google Scholar] [CrossRef]
- Kikutani, S.; Tanaka, R.; Yamazaki, Y.; Hara, S.; Hisabori, T.; Kroth, P.G.; Matsuda, Y. Redox Regulation of Carbonic anhydrases via Thioredoxin in Chloroplast of the Marine Diatom Phaeodactylum tricornutum. J. Biol. Chem. 2012, 287, 20689–20700. [Google Scholar] [CrossRef] [Green Version]
- Lane, T.W.; Saito, M.A.; George, G.N.; Pickering, I.J.; Prince, R.C.; Morel, F.M.M. A Cadmium enzyme from a Marine Diatom. Nature 2005, 435, 42. [Google Scholar] [CrossRef] [PubMed]
- Lane, T.W.; Morel, F.M.M. A Biological Function for Cadmium in Marine Diatoms. Proc. Natl. Acad. Sci. USA 2000, 97, 4627–4631. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Roberts, S.B.; Lane, T.W.; Morel, F.M.M. Carbonic anhydrase in the Marine Diatom Thalassiosira weissflogii (Bacillariophyceae). J. Phycol. 1997, 33, 845–850. [Google Scholar] [CrossRef]
- Nonoyama, T.; Kazamia, E.; Nawaly, H.; Gao, X.; Tsuji, Y.; Matsuda, Y.; Bowler, C.; Tanaka, T.; Dorrell, R.G. Metabolic Innovations Underpinning the Origin and Diversification of the Diatom Chloroplast. Biomolecules 2019, 9, 322. [Google Scholar] [CrossRef] [PubMed]
- Harada, H.; Nakatsuma, D.; Ishida, M.; Matsuda, Y. Regulation of the Expression of Intracellular β-Carbonic anhydrase in Response to CO2 and Light in the Marine Diatom Phaeodactylum tricornutum. Plant Physiol. 2005, 139, 1041–1050. [Google Scholar] [CrossRef] [Green Version]
- Ohno, N.; Inoue, T.; Yamashiki, R.; Nakajima, K.; Kitahara, Y.; Ishibashi, M.; Matsuda, Y. CO2-CAMP-Responsive Cis-Elements Targeted by a Transcription Factor with CREB/ATF-like Basic Zipper Domain in the Marine Diatom Phaeodactylum tricornutum. Plant Physiol. 2012, 158, 499–513. [Google Scholar] [CrossRef] [Green Version]
- Tanaka, A.; Ohno, N.; Nakajima, K.; Matsuda, Y. Light and CO2 /CAMP Signal Cross Talk on the Promoter Elements of Chloroplastic β-Carbonic Anhydrase Genes in the Marine Diatom Phaeodactylum tricornutum. Plant Physiol. 2016, 170, 1105–1116. [Google Scholar] [CrossRef] [Green Version]
- Engel, B.D.; Schaffer, M.; Cuellar, L.K.; Villa, E.; Plitzko, J.M.; Baumeister, W. Native Architecture of the Chlamydomonas Chloroplast Revealed by in Situ Cryo-Electron Tomography. Elife 2015, 4, 04889. [Google Scholar] [CrossRef]
- Nayak, M.; Dhanarajan, G.; Dineshkumar, R.; Sen, R. Artificial Intelligence Driven Process Optimization for Cleaner Production of Biomass with Co-Valorization of Wastewater and Flue Gas in an Algal Biorefinery. J. Clean. Prod. 2018, 201, 1092–1100. [Google Scholar] [CrossRef]
- Mackinder, L.C.M.; Meyer, M.T.; Mettler-Altmann, T.; Chen, V.K.; Mitchell, M.C.; Caspari, O.; Rosenzweig, E.S.F.; Pallesen, L.; Reeves, G.; Itakura, A.; et al. A Repeat Protein Links Rubisco to Form the Eukaryotic Carbon-Concentrating Organelle. Proc. Natl. Acad. Sci. USA 2016, 113, 5958–5963. [Google Scholar] [CrossRef] [Green Version]
- Ewe, D.; Tachibana, M.; Kikutani, S.; Gruber, A.; Río Bártulos, C.; Konert, G.; Kaplan, A.; Matsuda, Y.; Kroth, P.G. The intracellular distribution of inorganic carbon fixing enzymes does not support the presence of a C4 pathway in the diatom Phaeodactylum tricornutum. Photosynth. Res. 2018, 137, 263–280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Young, J.N.; Heureux, A.M.C.; Sharwood, R.E.; Rickaby, R.E.M.; Morel, F.M.M.; Whitney, S.M. Large Variation in the Rubisco Kinetics of Diatoms Reveals Diversity among Their Carbon-Concentrating Mechanisms. J. Exp. Bot. 2016, 67, 3445–3456. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gonzalez-Moro, B.; Lacuesta, M.; Becerril, J.M.; Gonzalez-Murua, C.; Munoz-Rueda, A. Glycolate Accumulation Causes a Decrease of Photosynthesis by Inhibiting RUBISCO Activity in Maize. J. Plant Physiol. 1997, 150, 388–394. [Google Scholar] [CrossRef]
- South, P.F.; Cavanagh, A.P.; Liu, H.W.; Ort, D.R. Synthetic Glycolate Metabolism Pathways Stimulate Crop Growth and Productivity in the Field. Science 2019, 363, eaat9077. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Badger, M.R.; Bek, E.J. Multiple Rubisco Forms in Proteobacteria: Their Functional Significance in Relation to CO2 Acquisition by the CBB Cycle. J. Exp. Bot. 2008, 59, 1525–1541. [Google Scholar] [CrossRef] [Green Version]
- Tabita, F.R.; Satagopan, S.; Hanson, T.E.; Kreel, N.E.; Scott, S.S. Distinct Form I, II, III, and IV Rubisco proteins from the three kingdoms of life provide clues about Rubisco evolution and Structure/Function relationships. J. Exp. Bot. 2008, 59, 1515–1524. [Google Scholar] [CrossRef]
- Kroth, P.G. The biodiversity of carbon assimilation. J. Plant Physiol. 2015, 172, 76–81. [Google Scholar] [CrossRef] [Green Version]
- Loganathan, N.; Tsai, Y.C.C.; Mueller-Cajar, O. Characterization of the Heterooligomeric Red-Type Rubisco Activase from Red Algae. Proc. Natl. Acad. Sci. USA 2016, 113, 14019–14024. [Google Scholar] [CrossRef] [Green Version]
- Blayney, M.J.; Whitney, S.M.; Beck, J.L. NanoESI Mass Spectrometry of Rubisco and Rubisco Activase Structures and Their Interactions with Nucleotides and Sugar Phosphates. J. Am. Soc. Mass Spectrom. 2011, 22, 1588–1601. [Google Scholar] [CrossRef] [Green Version]
- Stotz, M.; Mueller-Cajar, O.; Ciniawsky, S.; Wendler, P.; Hartl, F.U.; Bracher, A.; Hayer-Hartl, M. Structure of Green-Type Rubisco Activase from Tobacco. Nat. Struct. Mol. Biol. 2011, 18, 1366. [Google Scholar] [CrossRef]
- Mueller-Cajar, O.; Stotz, M.; Wendler, P.; Hartl, F.U.; Bracher, A.; Hayer-Hartl, M. Structure and Function of the AAA + Protein CbbX, a Red-Type Rubisco Activase. Nature 2011, 479, 194–199. [Google Scholar] [CrossRef]
- Tabita, F.R.; Hanson, T.E.; Li, H.; Satagopan, S.; Singh, J.; Chan, S. Function, Structure, and Evolution of the RubisCO-Like Proteins and Their RubisCO Homologs. Microbiol. Mol. Biol. Rev. 2007, 71, 576–599. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whitney, S.M.; Houtz, R.L.; Alonso, H. Advancing our understanding and capacity to engineer nature’s co2-sequestering enzyme, Rubisco. Plant Physiol. 2011, 155, 27–35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carmo-Silva, E.; Scales, J.C.; Madgwick, P.J.; Parry, M.A.J. Optimizing Rubisco and its regulation for greater resource use efficiency. Plant Cell Environ. 2015, 38, 1817–1832. [Google Scholar] [CrossRef]
- Rickaby, R.E.M.; Eason Hubbard, M.R. Upper Ocean Oxygenation, Evolution of RuBisCO and the Phanerozoic Succession of Phytoplankton. Free Radic. Biol. Med. 2019, 140, 295–304. [Google Scholar] [CrossRef]
- Packer, M. Algal capture of Carbon dioxide; Biomass generation as a tool for greenhouse gas mitigation with reference to New Zealand energy strategy and policy. Energy Policy 2009, 37, 3428–3437. [Google Scholar] [CrossRef]
- Osborne, C.P.; Sack, L. Evolution of C4 Plants: A new hypothesis for an interaction of CO2 and water relations mediated by plant hydraulics. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 583–600. [Google Scholar] [CrossRef] [Green Version]
- Maier, A.; Fahnenstich, H.; von Caemmerer, S.; Engqvist, M.K.M.; Weber, A.P.M.; Flügge, U.I.; Maurino, V.G. Transgenic introduction of a Glycolate Oxidative Cycle into A. thaliana chloroplasts leads to growth improvement. Front. Plant Sci. 2012, 3, 38. [Google Scholar] [CrossRef] [Green Version]
- Valenzuela, J.; Mazurie, A.; Carlson, R.P.; Gerlach, R.; Cooksey, K.E.; Peyton, B.M.; Fields, M.W. Potential role of multiple carbon fixation pathways during lipid accumulation in Phaeodactylum tricornutum. Biotechnol. Biofuels 2012, 5, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Elzenga, J.T.M.; Prins, H.B.A.; Stefels, J. The role of extracellular carbonic anhydrase activity in inorganic carbon utilization of Phaeocystis globosa (Prymnesiophyceae): A comparison with other marine algae using the isotopic disequilibrium technique. Limnol. Oceanogr. 2000, 45, 372–380. [Google Scholar] [CrossRef]
- Yi, Z.; Su, Y.; Cherek, P.; Nelson, D.R.; Lin, J.; Rolfsson, O.; Wu, H.; Salehi-Ashtiani, K.; Brynjolfsson, S.; Fu, W. Combined Artificial High-Silicate Medium and LED Illumination Promote Carotenoid Accumulation in the Marine Diatom Phaeodactylum tricornutum. Microb. Cell Factories 2019, 18, 209. [Google Scholar] [CrossRef] [PubMed]
- Fu, W.; Chaiboonchoe, A.; Khraiwesh, B.; Sultana, M.; Jaiswal, A.; Jijakli, K.; Nelson, D.R.; Al-Hrout, A.; Baig, B.; Amin, A.; et al. Intracellular spectral recompositioning of light enhances algal photosynthetic efficiency. Sci. Adv. 2017, 3, 1603096. [Google Scholar] [CrossRef] [Green Version]
- Daboussi, F.; Leduc, S.; Maréchal, A.; Dubois, G.; Guyot, V.; Perez-Michaut, C.; Amato, A.; Falciatore, A.; Juillerat, A.; Beurdeley, M.; et al. Genome engineering empowers the diatom Phaeodactylum tricornutum for biotechnology. Nat. Commun. 2014, 5, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Olofsson, M.; Lindehoff, E.; Frick, B.; Svensson, F.; Legrand, C. Baltic sea microalgae transform cement flue gas into valuable biomass. Algal Res. 2015, 11, 227–233. [Google Scholar] [CrossRef] [Green Version]
- Ho, S.H.; Chen, C.Y.; Lee, D.J.; Chang, J.S. Perspectives on microalgal CO2-emission mitigation systems–A Review. Biotechnol. Adv. 2011, 29, 189–198. [Google Scholar] [CrossRef]
- Botte, P.; D’Ippolito, G.; Gallo, C.; Sardo, A.; Fontana, A. Combined Exploitation of CO2 and Nutrient Replenishment for Increasing Biomass and Lipid Productivity of the Marine Diatoms Thalassiosira weissflogii and Cyclotella cryptica. J. Appl. Phycol. 2018, 30, 243–251. [Google Scholar] [CrossRef]
- Zimmerman, W.; Tesar, V.; Butler, S.; Bandulasena, H. Microbubble Generation. Recent Pat. Eng. 2008, 2, 1–8. [Google Scholar] [CrossRef] [Green Version]
- García Sánchez, J.L.; Berenguel, M.; Rodríguez, F.; Fernández Sevilla, J.M.; Brindley Alias, C.; Acién Fernández, F.G. Minimization of Carbon Losses in Pilot-Scale Outdoor Photobioreactors by Model-Based Predictive Control. Biotechnol. Bioeng. 2003, 84, 533–543. [Google Scholar] [CrossRef]
- Beardall, J.; Mukerji, D.; Glover, H.E.; Morris, I. The path of carbon in photosynthesis by marine phytoplankton. J. Phycol. 1976, 12, 409–417. [Google Scholar] [CrossRef]
- Rees, T.A.V. Sodium Dependent Photosynthetic Oxygen Evolution in a Marine Diatom. J. Exp. Bot. 1984, 35, 332–337. [Google Scholar] [CrossRef]
- Zhang, D.; Wen, S.; Wu, X.; Cong, W. Effect of Culture Condition on the Growth, Biochemical Composition and EPA Production of Alkaliphilic Nitzschia plea Isolated in the Southeast of China. Bioprocess Biosyst. Eng. 2018, 41, 831–839. [Google Scholar] [CrossRef] [PubMed]
- Hu, H.; Gao, K. Impacts of CO2 enrichment on growth and photosynthesis in freshwater and marine diatoms. Chin. J. Oceanol. Limnol. 2008, 26, 407–414. [Google Scholar] [CrossRef]
- Ihnken, S.; Roberts, S.; Beardall, J. Differential responses of growth and photosynthesis in the marine diatom Chaetoceros muelleri to CO2 and light availability. Phycologia 2011, 50, 182–193. [Google Scholar] [CrossRef]
- Lines, T.; Beardall, J. Carbon acquisition characteristics of six microalgal species isolated from a subtropical reservoir: Potential implications for species succession. J. Phycol. 2018, 54, 599–607. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Gao, K. Effect of CO2 concentrations on the activity of photosynthetic CO2 fixation and extracelluar carbonic anhydrase in the marine diatom Skeletonema costatum. Chin. Sci. Bull. 2003, 48, 2616–2620. [Google Scholar] [CrossRef]
- Cvejić, J.H.; Rohmer, M. CO2 as main carbon source for isoprenoid biosynthesis via the mevalonate-independent methylerythritol 4-phosphate route in the marine diatoms Phaeodactylum tricornutum and Nitzschia ovalis. Phytochemistry 2000, 53, 21–28. [Google Scholar] [CrossRef]
- Li, W.; Xu, X.; Fujibayashi, M.; Niu, Q.; Tanaka, N.; Nishimura, O. Response of microalgae to elevated CO2 and temperature: Impact of climate change on freshwater ecosystems. Environ. Sci. Pollut. Res. 2016, 23, 19847–19860. [Google Scholar] [CrossRef]
- Ozkan, A.; Rorrer, G.L. Effects of light intensity on the selectivity of lipid and chitin nanofiber production during photobioreactor cultivation of the marine diatom Cyclotella sp. Algal Res. 2017, 25, 216–227. [Google Scholar] [CrossRef]
- Kaewpintong, K.; Shotipruk, A.; Powtongsook, S.; Pavasant, P. Photoautotrophic High-Density cultivation of vegetative cells of Haematococcus pluvialis in Airlift bioreactor. Bioresour. Technol. 2007, 98, 288–295. [Google Scholar] [CrossRef]
- Pulz, O. Photobioreactors: Production Systems for Phototrophic Microorganisms. Appl. Microbiol. Biotechnol. 2001, 57, 287–293. [Google Scholar] [CrossRef]
- Branco-Vieira, M.; San Martin, S.; Agurto, C.; Freitas, M.A.V.; Martins, A.A.; Mata, T.M.; Caetano, N.S. Biotechnological potential of Phaeodactylum tricornutum for biorefinery processes. Fuel 2020, 268, 117357. [Google Scholar] [CrossRef]
- Remmers, I.M.; Martens, D.E.; Wijffels, R.H.; Lamers, P.P. Dynamics of Triacylglycerol and EPA production in Phaeodactylum tricornutum under nitrogen starvation at different light intensities. PLoS ONE 2017, 12, e0175630. [Google Scholar] [CrossRef] [PubMed]
- Tokushima, H.; Inoue-Kashino, N.; Nakazato, Y.; Masuda, A.; Ifuku, K.; Kashino, Y. Advantageous characteristics of the diatom Chaetoceros gracilis as a sustainable biofuel producer. Biotechnol. Biofuels 2016, 9, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Araújo, S.D.C.; Garcia, V.M.T. Growth and biochemical composition of the diatom Chaetoceros Cf. wighamii Brightwell under different temperature, salinity and carbon dioxide levels. I. protein, carbohydrates and lipids. Aquaculture 2005, 246, 405–412. [Google Scholar] [CrossRef]
- Jose, S.; Suraishkumar, G.K. High Carbon (CO2) supply leads to elevated intracellular Acetyl CoA levels and increased lipid accumulation in Chlorella vulgaris. Algal Res. 2016, 19, 307–315. [Google Scholar] [CrossRef]
- Bermúdez, R.; Feng, Y.; Roleda, M.Y.; Tatters, A.O.; Hutchins, D.A.; Larsen, T.; Boyd, P.W.; Hurd, C.L.; Riebesell, U.; Winder, M. Long-Term conditioning to elevated pCO2 and warming influences the fatty and amino acid composition of the diatom Cylindrotheca fusiformis. PLoS ONE 2015, 10, 0123945. [Google Scholar] [CrossRef]
- Zhang, W.; Tang, X.; Yang, Y.; Zhang, X.; Zhang, X. Elevated pCO2 level affects the extracellular polymer metabolism of Phaeodactylum tricornutum. Front. Microbiol. 2020, 11, 339. [Google Scholar] [CrossRef] [Green Version]
- Hamm, C.E.; Merkel, R.; Springer, O.; Jurkojc, P.; Maier, C.; Prechtel, K.; Smetacek, V. Architecture and Material Properties of Diatom Shells Provide Effective Mechanical Protection. Nature 2003, 421, 841–843. [Google Scholar] [CrossRef] [Green Version]
- Borowitzka, M.A.; Volcani, B.E. The polymorphic diatom Phaeodactylum tricornutum: Ultrastructure of its morphotypes. J. Phycol. 1978, 14, 10–21. [Google Scholar] [CrossRef]
- De Martino, A.; Bartual, A.; Willis, A.; Meichenin, A.; Villazán, B.; Maheswari, U.; Bowler, C. Physiological and molecular evidence that environmental changes elicit morphological interconversion in the model diatom Phaeodactylum tricornutum. Protist 2011, 162, 462–481. [Google Scholar] [CrossRef]
- Gemmell, B.J.; Oh, G.; Buskey, E.J.; Villareal, T.A. Dynamic sinking behaviour in marine phytoplankton: Rapid changes in buoyancy may aid in nutrient uptake. Proc. R. Soc. B Biol. Sci. 2016, 283, 20161126. [Google Scholar] [CrossRef] [PubMed]



| Products for Biomanufacturing | Species | Product Yield/Productivity Reported | Reference |
|---|---|---|---|
| Antibacterial substances | Phaeodactylum tricornutum EPSAG | n/a | [45] |
| Arachidonic acid | Phaeodactylum tricornutum CCAP 1055/1 (recombinant) | 1.89% DW; 22% TFAs | [46] |
| Cylindrotheca fusiformis UTEX 2084 | 8.19% TFAs | [47] | |
| Nitzschia sp. FD397 | 0.3% DW; 2.24% TFAs | [48] | |
| Nitzchia palea | 21.61% TFAs | [49] | |
| Biomass | Phaeodactylum tricornutum UTEX 640 | 25.4 g/L; 1.7 g/L/d | [50] |
| Amphora sp. MUR258 | 0.171 g/L/d | [51] | |
| Chaetoceros sp. | 0.125 g/L/d | [52] | |
| Skeletonema sp. | 0.185 g/L/d | [52] | |
| Thalassiosira sp. | 0.312 g/L/d | [52] | |
| Thallasiosira weissflogii | 3.83 g/m2/d | [53] | |
| Skeletonema sp. UHO29 | 0.34 g/L/d | [54] | |
| Nitzchia laevis UTEX 2047 | 0.4 g/L/d | [55] | |
| Chrysolaminarin | Phaeodactylum tricornutum CAS | 94 mg/L/d; 14% DW | [56] |
| Odontella aurita SCCAP K 1251 | 161.55 mg/L/d | [57] | |
| Docosahexaenoic acid (C22:6, n-3) | Phaeodactylum tricornutum CCAP 1055/1 Pt_El05 (recombinant) | 0.64% DW | [58] |
| Eicosapentaenoic acid (C20:5, n-3) | Nitzchia laevis UTEX 2047 | 10.46 mg/L/d; 19.15% DW | [55] |
| Fistulifera solaris JPCC DA0580 | 135.7 mg/L/d; 11.7% DW; 38.6% TFAs | [59] | |
| Thalassiosira weissflogii | 33.4 mg/L/d; 24.2% TFAs | [60] | |
| Odontella aurita SCCAP K 1251 | 9.37 mg/L/d; 25.3% TFAs | [57] | |
| Cyclotella cryptica CCAP 1070/2 | 3.8 % DW; 14.4% TFAs | [61] | |
| Cylindrotheca fusiformis UTEX 2084 | 24.63% TFAs | [47] | |
| Phaeodactylum tricornutum UTEX 640 | 56 mg/L/d; 3.29% DW | [50] | |
| Extra polymeric substances | Phaeodactylum tricornutum | n/a | [62] |
| Fucoxanthin | Nitzchia sp. KMMCC-308 | 0.492% DW | [63] |
| Mallomonas SBV13 | 2.66% DW | [64] | |
| Phaeodactylum tricornutum CAS | 4.7 mg/L/d; 0.7% DW | [56] | |
| Phaeodactylum tricornutum CS-29 | 2.28 mg/L/d; 5.92% DW | [65] | |
| Odontella aurita SCCAP K 1251 | 6.01 mg/L/d; 2.33% DW | [57] | |
| Chaetoceros gracilis KMMCC-27 | 0.223% DW | [63] | |
| Thalassiosira weissflogii | 0.95% DW | [60] | |
| Odontella aurita SCCAP K-1251 | 2.17% DW | [66] | |
| Cylindrotheca closterium | 0.523% DW | [67] | |
| Triacylglycerols (TAGs) | Cylindrotheca fusiformis CCAP 1017/2 | 7.2 mg/L/d; 24.5% DW | [61] |
| Chaeotoceros muelleri CCAP 1010/3 | 5.2 mg/L/d; 23.9% DW | [61] | |
| Chaetoceros simplex CCAP 1085/3 | 5.2 mg/L/d; 19.6% DW | [61] | |
| Amphora sp. MUR258 | 62 mg/L/d (lipid); 36.26% DW | [51] | |
| Phaeodactylum tricornutum | 58.5 mg/L/d; 45% DW | [68] | |
| Thalassiosira weissflogii P09 | 3.7 mg/L/d; 15% DW | [69] | |
| Thalassiosira weissflogii CCMP 1010 | 2.58 mg/L/d; 21% DW | [69] | |
| Thalassiosira weissflogii CCMP 1336 | 1.57 mg/L/d; 11% DW | [69] | |
| Thallasiosira psuedonana CCMP 1335 | 0.33 mg/L/d; 6% DW | [69] | |
| Navicula pelliculosa | 21.4% DW | [70] | |
| Nitzschia closterium | 38.8% DW | [70] | |
| Nitzschia longissima | 25.8% DW | [70] | |
| Nitzschia ovalis | 21% DW | [70] | |
| Nitzschia frustulum | 11.8% DW | [70] | |
| Amphora exigua | 23.6% DW | [70] | |
| Amphora sp. | 18.1% DW | [70] | |
| Biddulphia aurica | 19.3% DW | [70] | |
| Fragilaria sp. | 11% DW | [70] | |
| Chaetoceros sp. | 10.2% DW | [71] | |
| Cyclotella cryptica CCAP 1070/2 | 4 mg/L/d; 23.5% DW | [61] | |
| Cyclotella cryptica CCMP 331 | 1.64 mg/L/d; 23.06% DW | [69] | |
| Oxylipins | Cocconeis scutellum parva | n/a | [72] |
| Skeletonema marinoi | n/a | [72] | |
| Skeletonema costacum | n/a | [72] | |
| Chaetoceros pseudocurvisetus | n/a | [72] | |
| Phytosterol/Sterol | Chaetoceros muelleri | 0.4% DW (fucosterol), 0.25% DW (cholesterol) | [73] |
| Phaeodactylum tricornutum | 0.5% DW (brassicasterol) | [73] | |
| Thalassiosira pseudonana | 0.25% DW (24-Methylenecholesta-5, 24(24′)-dien-3Beta-ol | [73] | |
| Silica | n/a | [74] | |
| Sulfated polysaccharides | Phaeodactylum tricornutum | 20.15 mg/L/d | [75] |
| Polyhydroxybutyrate (PHB) | Phaeodactylum tricornutum CCAP 1055/1 | 10.6% DW | [76] |
| Human igGαHBSAg | Phaeodactylum tricornutum UTEX 646 | 0.0021% DW (8.7% total soluble protein) | [77] |
| IgG1/kappa ab CL4mAb | Phaeodactylum tricornutum UTEX 646 | 2.5 mg/L (secreted) | [78] |
| Monoclonal IgG antibodies against the nucleoprotein of Marburg virus | Phaeodactylum tricornutum UTEX 646 | 2 mg/L (secreted) | [79] |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sethi, D.; Butler, T.O.; Shuhaili, F.; Vaidyanathan, S. Diatoms for Carbon Sequestration and Bio-Based Manufacturing. Biology 2020, 9, 217. https://doi.org/10.3390/biology9080217
Sethi D, Butler TO, Shuhaili F, Vaidyanathan S. Diatoms for Carbon Sequestration and Bio-Based Manufacturing. Biology. 2020; 9(8):217. https://doi.org/10.3390/biology9080217
Chicago/Turabian StyleSethi, Deepak, Thomas O. Butler, Faqih Shuhaili, and Seetharaman Vaidyanathan. 2020. "Diatoms for Carbon Sequestration and Bio-Based Manufacturing" Biology 9, no. 8: 217. https://doi.org/10.3390/biology9080217
APA StyleSethi, D., Butler, T. O., Shuhaili, F., & Vaidyanathan, S. (2020). Diatoms for Carbon Sequestration and Bio-Based Manufacturing. Biology, 9(8), 217. https://doi.org/10.3390/biology9080217





