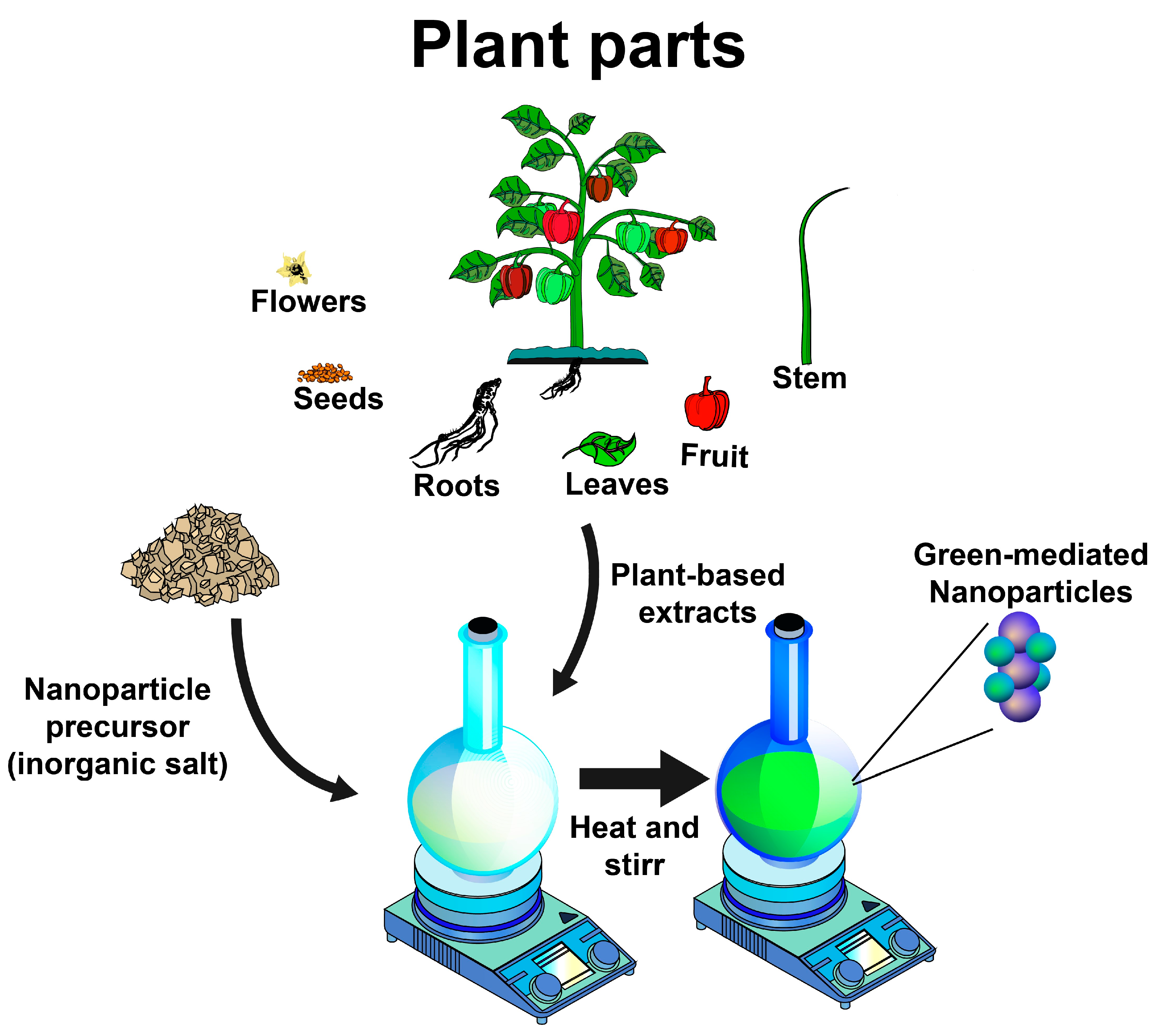
As discussed in the preceding sections, using plant-based extracts as reducing and stabilizing agents for synthesizing inorganic nanoparticles is an opportunity for developing greener synthesis approaches. Therefore, diverse inorganic nanoparticles have been synthesized using plant-based extracts, including gold, silver, titanium dioxide, zinc, copper, platinum, zirconium, iron, selenium, magnesium, nickel, sulfur, cobalt, palladium, and indium nanoparticles, which exhibit different biological activities for diverse applications (
Figure 2), as described below.
4.1. Gold Nanoparticles
Gold nanoparticles (AuNPs) have proved to be a versatile material for diverse applications [
15,
16,
17,
18]. Therefore, many routes for its synthesis have been developed involving the reduction of gold cations (Au
1+ or Au
3+) to gold-zerovalent (Au
0) [
15,
19,
20], highlighting the green synthesis route, where diverse rich bioactive compounds from plant-based extracts reduce gold precursors. Then, Au
+ ions are bound and capped by phytochemicals to form stable AuNPs [
19,
21,
22] (
Table 1). In most cases, the reduction of gold is monitored by a color change from pale pink to ruby red, or to dark violet from yellow for AuCl
3 and HAuCl
4, respectively, as AuNPs precursors [
16,
20,
21]. The green-mediated AuNPs exhibit XRD patterns of a face center cubic structure [(111), (200), (220), and (311)] [
16,
18] and UV–visible absorption peaks between 530 and 550 nm [
23], with an Eg value of 1.9 eV [
20].
In general, the methanolic and aqueous extracts from flowers, roots, seeds, fruits, and leaves of different plants have been used for the green synthesis of AuNPs with spherical shapes and sizes from 4 to 100 nm for diverse applications [
15,
18,
20,
23]. Khan et al. [
25] reported that AuNPs (5.8 nm) can be synthesized using
Clerodendrum inerme aqueous leaf extract, which exhibited antimicrobial, antioxidant, and cytotoxic activities. Similar trends were reported in AuNPs (53 nm) synthesized using licorice root extract, which exhibited antioxidant, antimicrobial, and anticancer activities in a concentration-dependent manner. These activities are associated with the bioactive compounds of the licorice root extract that includes phenolics, glycosides, organic acids, terpenes, and fatty acids [
21]. Furthermore, it has been reported that the green synthesis of AuNPs is influenced by the concentration of gold precursor [
20,
21].
Boruah et al. [
19] used
Moringa oleifera methanolic leaf extract to synthesize spherical-shaped AuNPs with size of 4 nm. They exhibited diverse biological properties, such as antimicrobial, antioxidant, and cytotoxic activities in red blood cells; moreover, they also showed photocatalytic properties against methylene blue dye. The authors suggested that the bioactive compounds of the extract contributed to the enhanced biological and technological properties of the AuNPs. Other studies have found similar trends when using green-mediated AuNPs from
Zingiber officinale root (antimicrobial, antioxidant, and cytotoxic properties) [
24],
Ricinus communis seeds [
15],
Jatropha integerrima flower [
20], and
Vitis vinifera fruit (antimicrobial activity) [
18]. It was also reported that AuNP-mediated
Pistacia chinensis exhibited analgesic and sedative properties in an acetic-acid-induced writhing model in a dose-dependent manner [
23]. Furthermore, extracts from plants such as
Phoenix dactylifera [
18],
Simarouba glauca [
22],
Salvia officinalis,
Lippia citriodora,
Pelargonium graveolens,
Punica granatum [
17], and
Centella asiatica [
16] have been investigated as reducing, capping, and stabilizing agents for the green synthesis of AuNPs.
4.2. Silver Nanoparticles
Among the metal-based nanoparticles, silver nanoparticles (AgNPs) have become very popular in the research community due to their wide range of applications, including for antimicrobial, antioxidant, anticancer, and photocatalytic dye degradation applications [
26,
27,
28,
29]. In this context, the green synthesis of AgNPs using plant-based extracts as reducing agents (Ag
+ to Ag
0) has gained significant attention in recent years [
30,
31,
32], with silver nitrate (AgNO
3) being the most used precursor for their synthesis [
29,
31,
32,
33]. After mixing AgNO
3 aqueous solution and plant extracts, the color of the reaction medium changes from yellowish green to brownish [
34,
35], suggesting the conversion of Ag ions (Ag
+) to metallic silver (Ag
0) [
31]. The green-mediated AgNPs exhibited surface plasmon resonance around 400–450 nm [
31] with a crystalline nature and a face-centered cubic structure XRD pattern of (111), (200), (220), and (331) [
35]. On the other hand, some green-synthesized AgNPs exhibited good stability (zeta potential of −15.8 to 3.31 mV) [
32]. Various plant-based extracts have been investigated to prepare spherical AgNPs with sizes < 100 nm (
Table 2).
Various plants such as
Zataria multiflora,
Brillantaisia patula,
Crossopteryx febrifuga,
Senna siamea,
Gymnema sylvestre,
Lysiloma acapulcensis, and
Achillea millefolium have been used to synthesize silver AgNPs, using extracts from different parts of the plant, such as the leaves, stems, and roots [
29,
31,
33,
34]. It was reported that factors such as the plant extract, pH, and temperature significantly influenced the green synthesis of AgNPs [
39]. These green-mediated AgNPs have exhibited antimicrobial properties against various pathogenic bacteria and fungi [
27,
28,
31,
35,
36,
37]. According to the authors, the antimicrobial activity of AgNPs is related to their ability to cause cell breakdown, which promotes changes in the cell membrane’s permeability.
It has been found that green-synthesized AgNPs prepared from aqueous extracts derived from
Annona muricata peel,
Teucrium polium leaves, and
Cynara scolymus leaf extracts exhibit anticancer properties at very low concentrations, attributed to their apoptotic properties [
30,
34,
38,
39,
40]. Additionally, green-synthesized AgNPs using
Achillea millefolium and
Annona muricata extracts exert anti-inflammatory and antioxidant activities [
28,
30]. Chand et al. [
38] reported that AgNPs synthesized using onion and tomato extracts exhibited photocatalytic properties against cationic dyes in aqueous solutions, with the effect attributed to the capability of AgNPs to produce reactive oxygen species under the UV region [
38].
In general, the plant-based extracts used for AgNPs green synthesis proceed through reducing silver ions, primarily by the capability to donate electrons. Tannins were found to play a key role in reducing and capping AgNPs in most cases [
39]. Furthermore, some plant-based extracts, such as
Centella Asiatica and Tridax, were used to synthesize silver oxide (Ag
2O) nanoparticles, which exhibited photocatalytic properties against acid orange dye [
27].
4.3. Titanium Dioxide Nanoparticles
Titanium dioxide nanoparticles (TiO
2NPs) are one of the most investigated materials due to their photocatalytic properties and chemical and thermal stability in diverse industrial uses [
41]. They have been typically synthesized by chemical routes [
42]; however, TiO
2NPs have been synthesized using green approaches using different plant-based extracts and titanium-isopropoxide or titanium dioxide solution as chemical precursors (
Table 3). In most cases, a light-green formation after mixing TiO
2 precursor and plant extracts indicates the reduction of Ti ions [
43]. The green-mediated TiO
2NPs exhibited a strong UV absorption peak around 380 to 400 nm and a crystalline structure in their anatase and rutile forms [Miller index = (101), (110), (103), (004), (112), (200), (105), and (211)] [
41] with negative zeta potential (−18.7 to −11.5 mV) and large surface area (105 m
2g
−1) [
44,
45,
46].
Thakur et al. [
41] synthesized TiO
2NPs using
Azadirachta indica aqueous leaf extract as a stabilizing and reducing agent, with anatase and rutile structures, spherical shapes, and sizes from 15 to 50 nm, which exhibited antibacterial activity against different Gram-negative bacteria. Similarly, TiO
2NPs (spherical and 20–70 nm in size) prepared by green synthesis using
Mentha arvensis leaf extracts showed antimicrobial activity in a dose-dependent manner [
45]. According to the authors, the phytochemicals such as alkaloids, terpenoids, and phenolics in
Azadirachta indica and
Mentha arvensis acted as stabilizing and reducing agents during TiO
2 synthesis. The antimicrobial effect of TiO
2 is due to its capability to interact with the cell wall of microorganisms, promoting cell death [
41,
45].
Sethy et al. [
46] reported that TiO
2NPs (anatase phase) synthesized using an aqueous leaf extract of
Syzygium cumini exhibited photocatalytic properties for removing Pb from wastewater, associated with its ability to generate OH radicals in the presence of light. On the other hand, recent research also showed that TiO
2NPs (anatase form, spherical and polygonal shape, 130 nm in size), prepared by a green synthesis approach using
Ocimum sanctum leaf extract, could improve the wound-healing efficacy of chitosan hydrogels in diabetics rats [
44]. This effect was attributed to the antimicrobial properties of TiO
2 [
45]. The authors suggested that the presence of phytochemicals in plant-based materials plays an important role in the reduction, capping, and stabilization of TiO
2 [
44,
46].
Srinivasan et al. [
43] synthesized TiO
2NPs in the anatase phase using aqueous leaf extracts of
Sesbania grandiflora, which exhibited square and spherical shapes with sizes ranging from 43 to 56 nm, completing the reduction at room temperature. Furthermore, the study revealed that TiO
2NPs exhibited toxicological effects against zebrafish embryos in a dose-dependent manner. Therefore, further studies are needed to fully evaluate the possible toxic effects of TiO
2NPs synthesized by green synthesis methods.
Additionally, the green synthesis of TiO
2NPs has been performed using different plant-based extracts, including
Moringa oleifera [
47],
Psidium guajava [
48],
Arbor tristis [
49],
Eclipta prostrata [
50], and
Ageratina altissima leaves [
51],
Vigna unguiculata seeds [
52],
Calotropis gigantea flowers [
53],
Aloe vera [
54],
Vigna radiata legumes [
55], and
Curcuma longa plant [
56]. These TiO
2NPs exhibited potential industrial, environmental, and pharmaceutical applications due to their wound healing, antimicrobial, antioxidant, dye photocatalytic degradation, acaricidal, and cytotoxic properties [
47,
49,
51,
52,
53,
55].
4.4. Zinc Nanoparticles
Zinc nanoparticles (ZnNPs) have unique features like being nontoxic, low-cost, biocompatible, multifunctional, and eco-friendly. They have been investigated for diverse applications due to their antimicrobial, antifungal, nanomedicine, antioxidant, and photocatalytic activities [
57,
58,
59]. ZnNPs have been green-synthesized using extracts from the leaves, flowers, stem bark, and fruit juice from various plant species (
Table 4), which are mostly spherical, with sizes smaller than 60 nm. The most common Zn precursors are zinc nitrate and zinc acetate [
60,
61]. After mixing the plant extracts and Zn precursors, a white or yellowish paste is observed, indicating successful formation [
62,
63]. The green-mediated ZnNPs exhibited a strong UV absorption peak around 300–400 nm [
63,
64] and a crystalline structure in their hexagonal wurtzite phase [Miller index = (100), (002), (101), (102), (110), (200), (112), (201), (004), and (202)], where each Zn
+2 ion was ordered in a tetragonal coordination with a polar symmetry throughout the hexagonal axis [
62,
65]. These ZnNPs exhibited Eg values from 2.67 to 3.37 eV and a negative zeta potential (−40 mV) [
63,
65].
Madhukara et al. [
60] used
Limonia acidissima juice for zinc ferrite (ZnFe
2O
4) nanoparticle synthesis with photocatalytic properties against Evans blue and methylene blue dyes under visible light in a concentration-dependent manner, as well as antibacterial activity against in a strain- and dose-dependent manner. It has been reported that green-synthesized ZnNPs using
Eriobutria japonica seed [
57],
Hibiscus sabdariffa [
58], or
Hydnocarpus alpina [
67] extracts are active against methylene blue dye in a dose-dependent manner; moreover, they exhibit antimicrobial activity against various bacterial strains [
57,
67]. However, ZnNPs exhibit higher antimicrobial activity against Gam-negative bacteria than Gram-positive bacteria due to differences in the thickness of the peptidoglycan layer; thus, ZnNPs can enter cells and inhibit their replication and growth. Additionally, ZnNPs synthesized using
Lippia adoensis [
61], as well as
Euphorbia hirta extracts, showed antibacterial activity in a strain- and dose-dependent manner.
Bitopan et al. [
66] studied the biocompatibility of ZnNPs using
Xanthium indicum leaf extract as a reducing and stabilizing agent and reported that the ZnNPs did not show hemolytic action at lower concentrations; however, negative effects were observed at higher concentrations (>25 mg/mL). The authors mentioned that the ZnNPs synthesized by the green route demonstrated the weakest cytotoxic effects compared with those obtained by the chemical route. Additionally, Ashwini et al. [
68] synthesized ZnNPs using
Cayratia pedate as an enzyme glucose oxidase immobilizer for biomedical applications.
4.5. Copper Nanoparticles
Copper nanoparticles (CuNPs) are gaining significant attention due to their electrical, optical, mechanical, catalytic, and antimicrobial properties [
69]. They have been green-synthesized using aqueous extracts from the leaves, flowers, and fruits from various plants for environmental and antimicrobial purposes [
3,
70,
71,
72]. The most common precursors for CuNPs are copper (II) sulfate pentahydrate [
3,
69,
71,
73], cupric nitrate trihydrate [
70], copper chloride (II) [
71,
73,
74], and copper (II) acetate [
75] (
Table 5). The formation of CuNPs is confirmed through a color change of the reaction mixture from yellowish to brownish [
71,
73], yellow to green [
3], or blue to brown [
72], depending on the Cu precursor. Green-mediated CuNPs exhibited a UV absorption peak ranging from 269 to 580 nm, which depends on the CuNPs’ energy state [
3,
69]. In most cases, CuNPs exhibited a monocyclic configuration [Miller index = (110), (111), (220), (800), and (713)] [
71]. These CuNPs are mostly spherical with sizes ranging from 2 to 80 nm [
3,
70,
71,
72,
73,
74] and a negative zeta potential (−33.98 mV) [
74].
Mali et al. [
3] used
Celastrus paniculatus aqueous leaf extract as a reducing agent to synthesize spherical CuNPs (Cu purity of 79.87%, size of 2–10 nm) with photocatalytic and antifungal properties. Similarly, it was reported that CuNPs (spherical and size of 63 nm) prepared using
Tinospora cardifolia aqueous leaf extract exhibited antibacterial activity and could be impregnated in cotton fabrics [
74]. Additionally,
Cissus vitiginea leaf extract was used to synthesize CuNPs (spherical and size of 5–20 nm) active against urinary infection pathogens [
72]. The antimicrobial properties of CuNPs are based on changes in the cell structure of microorganisms, leading to cell death [
3,
72,
74]. In general, the biomolecules present in plant extracts act as reducing and stabilizing agents during the formation of CuNPs. Particularly, flavonoids are transformed from the enol form to the keto form by releasing a reactive hydrogen atom that reduces Cu
2+, which is facilitated at pH 7 [
3].
Ismail et al. [
69] reported that zerovalent CuNPs synthesized using
Duranta erecta aqueous fruit extract showed photocatalytic activity against anionic dyes in a dose-dependent manner. Similarly, CuNPs with photocatalytic properties against anionic and cationic dyes could be synthesized using
Jatropha curcas leaf extract [
71].
Additionally, Chowdhury et al. [
75] synthesized copper oxide nanoparticles (CuONPs) with
Lantana camara flower extract in an alkaline hydrolysis process. The resulting rod-shaped nanoparticles (15–23 nm in size) exhibited catalytic properties against acrylonitrile and aniline. Similarly, CuONPs synthesized using
Calotropis procera leaf extract exhibited antimicrobial activity. Furthermore, Cu-based nanoparticles have also been doped with inorganic materials to enhance their physicochemical properties. Green-synthesized Cu-doped MoO
3 (Cu-MoO
3) nanoparticles prepared using
Genus Santalum aqueous leaf extract enhanced the photocatalytic properties in degrading hazardous organic pollutants [
70]. Cu-doped silver (Cu-Ag) nanoparticles could remove dyes from aqueous solutions [
76], while copper–nickel hybrid nanoparticles synthesized using extracts from
Zingiber officinale rhizomes showed photocatalytic activity against crystal violet dye [
77].
4.6. Platinum Nanoparticles
Over the past few years, researchers have been exploring the use of aqueous extracts from various plant materials, including
Cordyceps militaris,
Nymphaea tetragona,
Atriplex halimus, olive, Saudi’s dates, and tea polyphenols, along with their parts, to synthesis platinum nanoparticles (PtNPs) (
Table 6). For this purpose, hexachloroplatinic acid (H
2PtCl
6) is commonly used as precursor to produce spherical-shaped PtNPs ranging from 1 to 13 nm [
78,
79,
80]. After mixing plant extracts and H
2PtCl
6, the color of the reaction mixture gradually changed from pale yellow to brown or black, indicating the reduction to Pt
0 [
79,
80,
81,
82,
83]. The green-mediated PtNPs exhibited surface plasmon resonance around 230 to 295 nm and a cubic structure XRD pattern of (111), (200), (220), and (311) [
81,
82] with a zeta potential of −17.28 to −0.0536 mV [
80,
84,
85].
PtNPs have shown promising antimicrobial activity against pathogenic bacteria [
79,
80,
82], as well as antioxidant capacity [
80,
81,
82]. Additionally, PtNPs have been found to possess anticancer properties [
79] and inhibit serum aspartate aminotransferase in serum levels of patients with chronic liver disease [
83]. PtNPs have also demonstrated antiaging effects/skin protection by promoting collagen I biosynthesis in HFF-1 cells and inhibiting tyrosinase activity in A375 cells [
81]. On the other hand, PtNPs with polycrystalline structures were used to develop sensors for hydrogen peroxide detection [
78]. All authors agreed that bioactive compounds in plant-based extracts exerted capping and reducing effects, both associated with metal reduction and contribute as a stabilizing agent to forming PtNPs, avoiding their agglomeration [
78,
79,
82]. Furthermore, green synthesis of PtNPs has been performed using hyacinth plant extracts [
85] and seaweed
Padina gymnospora [
82] with potential biomedical applications.
4.7. Zirconium Nanoparticles
Zirconium (ZrNPs) nanoparticles can be synthesized by green chemistry methods with high purity using plant-based extracts for diverse applications, such as antimicrobial and dye photocatalytic degradation [
86]. They can be prepared from various precursors, including zirconium isopropoxide [
87], zirconylchloride octahydrate [
88], zirconyl nitrate [
84], and zirconyl chloride [
89]. After mixing a Zr precursor and plant extract, color changes from yellow to brown [
89] or milky white formation indicates the formation of ZrNPs [
88]. Green-mediated ZrNPs exhibited monocyclic structure of baddeleyite [XRD (111), (002), (022), (031), and (131)] [
90], a UV absorption peak around 275 nm, an Eg value of 3.7 eV [
91], and a zeta potential of −32.8 mV [
88,
92]. The ZrNPs had cubic, spherical, triangular, and oval shapes with sizes from 10 to <200 nm [
87,
88] (
Table 7).
Kazi et al. [
86] used aqueous leaf extracts from
Sphagneticola trilobata to synthesize ZrNPs of various shapes and sizes (20 to 100 nm). These nanomaterials showed antimicrobial properties and antimalarial activity. Similarly, spherical ZrNPs (121 nm) synthesized using
Phyllanthus niruri aqueous leaf extracts, cubic ZrNPs obtained from
Laurus nobilis (20 to 100 nm), and neem-gum nanoparticles displayed antimicrobial activity. In all cases, the antimicrobial effects of ZrNPs were dose-dependent. According to the authors, the antimicrobial properties of ZrNPs are related to their surface energy and surface-related interactions, leading to cell death by disrupting cell membranes and altering membrane fluidity [
87,
88,
89].
Regarding environmental applications, ZrNPs synthesized by green methods with extracts from the pericarp of
Sapindus mukorossias and leaves of
Phyllanthus niruri exhibited photocatalytic properties for different dyes. These effects were attributed to the photocatalytic and adsorptive properties of ZrNPs [
89,
93].
The authors agreed that the presence of biomolecules in the plant-based extracts played a crucial role in reducing, capping, chelating, and stabilizing the conversion to ZrNPs due to their antioxidant properties [
86,
87,
89,
93]. These phytochemicals also helped to form and stabilize the octahedral complex of Zr
2+ phytochemicals [
94]. Moreover, zirconium oxide (ZrO
2) nanoparticles can be synthesized using different plant-based extracts, including
Euclea natalensis roots [
95],
Salvia Rosmarinus leaves [
96],
Helianthus annuus seeds [
91],
Nephelium lappaceum fruit [
90], and
Nyctanthes arbortristis flowers [
92] for diverse industrial applications.
4.8. Iron Nanoparticles
Iron oxide nanoparticles (Fe
2O
3NPs) have gained significant attention due to their unique physicochemical and catalytic properties for environmental and biomedical applications [
97,
98], mainly those synthesized by green routes using plant-based extracts. Fe
2O
3NPs can be prepared from various precursors, including ferric chloride hexahydrate (FeCl
3·6H
2O) [
97], ferric nitrate (Fe(NO
3)
3·9H
2O) [
99], and ferrous sulfate (FeSO
4) [
100]. The formation of a black precipitate ensures the formation of α-Fe
2O
3 nanoparticles [
101] or Fe
3O
4 [
102], which are corroborated by the XRD patterns [
101]. The nanoparticles exhibited spherical, semispherical, and cubic shapes with sizes ranging from 4 to 200 nm [
97,
100,
103] (
Table 8).
Generally, the most common form of green-synthesized iron nanoparticles is iron oxide (α-Fe
2O
3), which has been investigated for dye degradation, antimicrobial, and antioxidant purposes [
103,
104,
105]. In this context, Lohrasbi et al. [
97] used an aqueous extract of
Plantago major leaves to prepare spherical Fe
2O
3NPs with sizes from 4 of 30 nm for environmental applications, while
Ficus carica fruit extract was used in core–shell form with an average size of 9 nm. Additionally, Fe
2O
3 or Fe
3O
4 iron nanoparticles synthesized using
Phoenix dactylifera aqueous leaf extract were found to exhibit antioxidant properties [
102]. Nas et al. [
98] reported that Fe
2O
3NPs synthesized using
Rhus punjabensis extract can be used for antimicrobial, antioxidant, and anticancer applications. This effect is associated with the functional groups of the phenolics and flavonoids on the surface of the nanoparticles [
98].
Devi et al. [
99] reported that
Platanus orientalis leaf extract can be used for the green synthesis of spherical Fe
2O
3NPs with an average size of 38 nm. These nanoparticles exhibited antifungal activity in a dose-dependent manner, attributed to the capability of Fe
2O
3NPs to disrupt microbial cell walls. Similarly, it was reported that Fe
2O
3NPs with irregular shapes and an average size of 21 nm can be synthesized using
Carica papaya leaf extract, exhibiting moderate antimicrobial and photocatalytic properties in a dose-dependent manner [
101]. During the green synthesis of Fe
2O
3NPs, the phytochemicals of plant extracts can reduce metallic salts to nanoparticles and act as stabilizing agents, preventing the aggregation of nanoparticles. The bioactive compounds reacted with the iron ions to give Fe
2O
3NPs, as the first-row transition metals are oxidation-prone. However, it is possible that the phytochemicals are not able to reduce Fe
3+ to Fe
0 [
99,
101].
Furthermore,
Withania coagulans extract was used as a reducing agent to prepare α-Fe
2O
3 nanorods (16 nm) with photocatalytic and antimicrobial activities [
105]. Similarly, it was reported that using pomegranate seed extract, semispherical Fe
2O
3NPs (25–55 nm) exhibited the catalytic degradation of reactive blue dyes under UV light [
103]. On the other hand, Fe-based nanoparticles doped with other inorganic materials showed enhanced physicochemical properties. Younas et al. [
100] found that green-synthesized Fe–Cu bimetallic nanoparticles using
Ixora finlaysonian leaf extract with rectangular and cubic shapes and sizes from 50 to 200 nm exhibited antioxidant and photocatalytic dye-degradation activities.
4.9. Selenium Nanoparticles
Selenium nanoparticles (SeNPs) are biocompatible compounds that exhibit low toxicity and high biological activities, which can be synthesized by green chemistry using plant-based extracts from fruits, leaves, peel, and plants such as
Crataegus monogyna [
106],
Melia azedarach [
107],
Rosmarinus officinali [
108], orange [
109],
Cleistocalyx operculatus [
110],
Portulaca oleracea [
111],
Urtica dioica [
112],
Abelmoschus esculentus [
113],
Cordia myxa [
114],
and Withania somnífera [
115], with sodium selenite (Na
2SeO
3), selenium dioxide (SeO
2), and selenious acid (H
2SeO
3, as SeNPs precursors [
106,
110,
115]. The formation of SeNPs can be identified by a color change from light green to brick red [
107], colorless to light pink or red [
106,
114], or from pale yellow to deep red [
106,
109]. SeNPs exhibited a UV absorption peak ranging from 200 to 302 nm with monocyclic and trigonal phases [
106,
107,
108,
109,
110] and a zeta potential of −64 mV [
113]. These nanoparticles are spherical with sizes ranging from 2 to 200 nm [
110,
111] and exhibit antioxidant, antimicrobial, antiviral, anticancer, and photocatalytic activities [
106,
107,
111,
115] (
Table 9).
Barzegarparay et al. [
106] reported that green-synthesized SeNPs using
Crataegus monogyna methanolic fruit extract exhibited anticancer activity; using an ethanolic leaf extract from
Withania somnifera and
Cordia myxa aqueous fruit extract, SeNPs showed antiproliferative properties [
114,
115]. This effect was attributed to the apoptotic properties of SeNPs, which were able to arrest the C2/M cell cycle [
106], as well as to their antioxidant activity [
106,
115].
Additionally, SeNPs prepared using
Melia azedarach aqueous leaf extract exhibited antifungal properties [
107], while those synthesized using
Rosmarinus officinali,
Abelmoschus esculentus,
Cleistocalys operculants leaves, and orange peel extracts exhibit antimicrobial activity [
108,
109,
110,
113].
Portulaca oleracea-based green SeNPs exhibited antimicrobial, antiviral, and mosquitocidal properties [
111], while
Urtica dioica-mediated SeNPs exerted antifungal activity [
112]. Additionally, it was reported that SeNPs synthesized using combinations of plant extracts (
Allium cepa,
Malpighia emarginata, and
Gymnanthemum amygdalinum) exhibited antimicrobial activity [
116]. Moreover, green-synthesized SeNPs (
Withania somnifera leaf extract) have been explored for the photocatalytic degradation of methylene blue dye [
115].
4.10. Magnesium Nanoparticles
Magnesium oxide nanoparticles (MgONPs) are an attractive material for antimicrobial purposes [
117]. Moreover, they have been investigated for other potential applications, including dye degradation, antioxidant, cytotoxic, and antiaging applications [
118,
119,
120]. The green synthesis of MgONPs has been performed using aqueous extracts from the leaves, flowers, and barks of different plants [
118,
121,
122], when magnesium nitrate (Mg(NO
3)
2) [
121] and magnesium chloride (MgCl
2) [
123] have been used as precursors (
Table 10). The formation of MgONPs can be identified by a color change from pale green to brown [
121], brownish to dark brownish-red [
119], and colorless to dark brown [
117,
123]. Moreover, it has been reported that MgONPs exhibited a UV absorption peak around 280–290 nm and a hexagonal or cubic structure [
117,
118,
119,
122,
123].
Vergheese and Vishal [
119] demonstrated that spherical MgONPs (13 nm) synthesized using
Trigonella foenum-graecum,
Xanthomonas oryzae pv.
Oryzae, and
Dalbergia sissoo aqueous leaf extract exhibited antibacterial activity [
118]. In general, bioactive compounds such as alkaloids, saponins, flavonoids, phenolics, and terpenoids act as a capping and stabilizing agent during synthesis [
120]. However, it must be noted that the pH of the reaction solution may affect the reduction ability of bioactive compounds, mainly owing to the concentration of hydroxyl ions in the medium [
118].
Younis et al. [
118] found that MgONPs (polyhedral and size of 35–55 nm) synthesized using flower extracts of
Rosa floribunda charisma exhibited antioxidant, antiaging, and antibacterial activities in a dose-dependent manner against skin pathogens. Additionally, the bark and leaf extracts of
Moringa oleifera have been investigated for the synthesis of MgONPs with antioxidant and antimicrobial activities [
117,
123]. Recently, it has been reported that MgONPs with irregular shapes and sizes < 100 nm synthesized by a green approach (
Abrus precatorius aqueous bark extract) exhibited antioxidant, antibacterial, and cytotoxic activities without toxic effects on zebrafish embryos; moreover, MgONPs exhibited the photocatalytic degradation of methylene blue dye [
119].
4.11. Nickel Nanoparticles
Nickel/nickel oxide nanoparticles (NiNPs/NiONPs) have been green-synthesized using aqueous extracts from the leaves, seeds, and flowers of various plants for environmental, antioxidant, antimicrobial, anticancer, and antileishmanial applications [
76,
124,
125,
126]. In the process, nickel nitrate (Ni(NO
3)
2) [
127], nickel chloride (NiCl
2) [
128], and nickel sulfate (NiSO
4) [
129] are the most commonly used precursors (
Table 11). During the green synthesis of NiNPs, the color of the reaction mixture changes from green to dark brown [
77]. NiNPs were characterized by XRD [Miller index = (110), (111), (200), (220), and (311)], and UV–Vis (surface resonance plasmon of 341 nm and bandgap energy of 1.57 eV) [
126,
128].
Yuan et al. [
129] synthesized NiNPs (spherical, 20–36 nm) using
Alhagi maurorum leaf aqueous extract that exhibited cytotoxic and anti-human ovarian cancer activity in a dose-dependent manner [
125,
128]. Similarly, it was reported that nickel ferrite (NiFe
2O
4) nanoparticles (spherical shape, size of 19 nm) synthesized using
Terminalia catappa aqueous leaf extract exhibited anticancer activity in a dose-dependent manner, possibly through an apoptosis mechanism. The average crystallite size of the Ni nanoparticles was reduced by increasing the volume of the plant extract used [
124].
Additionally,
Rhamnus virgata leaf aqueous extract was used as stabilizing, reducing, and chelating agent during the formation of NiONPs, showing anticancer, antileishmanial, and antimicrobial activities without toxicological effects in human RBCs and macrophages [
125]. Ali et al. [
126] reported that green-synthesized spherical Ni/NiONPs using the seed extract of
Lactuca serriola exhibited antimicrobial properties in a strain- and dose-dependent manner due to the capability of the nanoparticles to modify the cell membrane and block the transport channels. Moreover,
Senna auriculata-mediated and
Hammada scoparia-mediated NiONPs have exerted antimicrobial properties [
76,
130].
Furthermore, the seed extract of
Hordeum vulgare was used to synthesize NiNPs and NiONPs (<100 nm) and was able to degrade methylene blue dye, which exhibited first-order kinetics [
127]. Similarly, it was reported that NiONPs green-synthesized using
Syzygium cumini leaf extract with a spherical shape and size of 10 nm exhibited photocatalytic activity against methylene blue and Congo red dyes [
128]. Moreover, other plant-based extracts from the seeds, flowers, and leaves from
Lactuca Serriola [
126],
Senna auriculata [
130], and
Hammada scoparia [
76], respectively, have been used for the green synthesis of Ni and NiO nanoparticles for the photocatalytic degradation of crystal violet, methylene blue, and malachite green [
76,
126,
130].
4.12. Sulfur Nanoparticles
Sulfur nanoparticles (SNPs) are biocompatible compounds with a high surface area and catalytic activity, which have great potential for diverse biomedical and agricultural applications [
131]. These SNPs have been green-synthesized using aqueous extracts from various plant materials and their parts, including
Rosmarinus officinalis [
132],
Citrus limon [
131],
Aloe vera [
133],
Allium sativum [
134], and
Cinnamomum zeylanicum [
135]. Sodium thiosulfate pentahydrate is commonly used as a precursor for spherical SNPs, ranging from 40 to 69 nm in size [
131,
132,
133,
134,
135]. After mixing the plant extract and sulfur precursor, a yellow color indicates the formation of SNPs [
135]. These nanoparticles agreed with the standard XRD orthorhombic sulfur pattern [
133] and exhibited a UV absorption peak of around 245 to 295 nm [
132,
133] with a zeta potential of −10.4 mV [
134].
These SNPs have been used as plant-growth-promoting and nematicidal agents [
132,
134,
135] and are effective antimicrobial agents [
131]. Furthermore, SNPs were embedded in a chitosan nanohydrogel for wound-healing applications [
131].
Table 12 lists some green-synthesized sulfiur SNPs and their applications.
4.13. Other Nanoparticles Synthesized by Green Methods
The other nanoparticles synthesized by the green approach using plant-based extracts as capping, reducing, and stabilizing agents include cobalt [
136,
137], palladium [
138], and indium [
139,
140]. Gingasu et al. [
137] used an aqueous extract of ginger roots and cardamom seeds to synthesize cobalt ferrite nanoparticles (CoFe
2O
4) with irregular forms and sizes smaller than 100 nm. The authors reported that the Co
2+ cation distribution was higher than that of Fe
3+, possibly associated with the nature of the plant extracts. Similarly, cobalt oxide (Co
3O
4) nanoparticles were green-synthesized using
Populus cilata aqueous leaf extracts, which exhibited antimicrobial activity [
136].
Vinodhini et al. [
138] used
Allium fistulous,
Basella alba, and
Tabernaemontana divaricate aqueous leaf extracts to synthesize palladium nanoparticles with photocatalytic activity for Congo red dye degradation. Moreover, palladium nanoparticles exhibited antioxidant and antimicrobial properties in a concentration-dependent manner. Furthermore, palladium nanoparticles prepared with
Tabernaemontana divaricate leaf extract showed antidiabetic activity in vitro inhibiting the α-amylase enzyme. According to the authors, polyphenol-rich plant-based extracts played an important role in reducing the metal ions and stabilizing the inorganic nanoparticle formation, showing diverse potential applications [
137,
138]. On the other hand,
Aloe vera plant extract [
140] and
Astragalus gummifer gums [
139] have been used in green synthesis of indium oxide (In
2O
3) nanoparticles, which exhibited good optical properties for further applications [
139,
140].