Presence and Distribution of Fluoride Ions in Groundwater for Human in a Semiconfined Volcanic Aquifer
Abstract
:1. Introduction
2. Materials and Methods
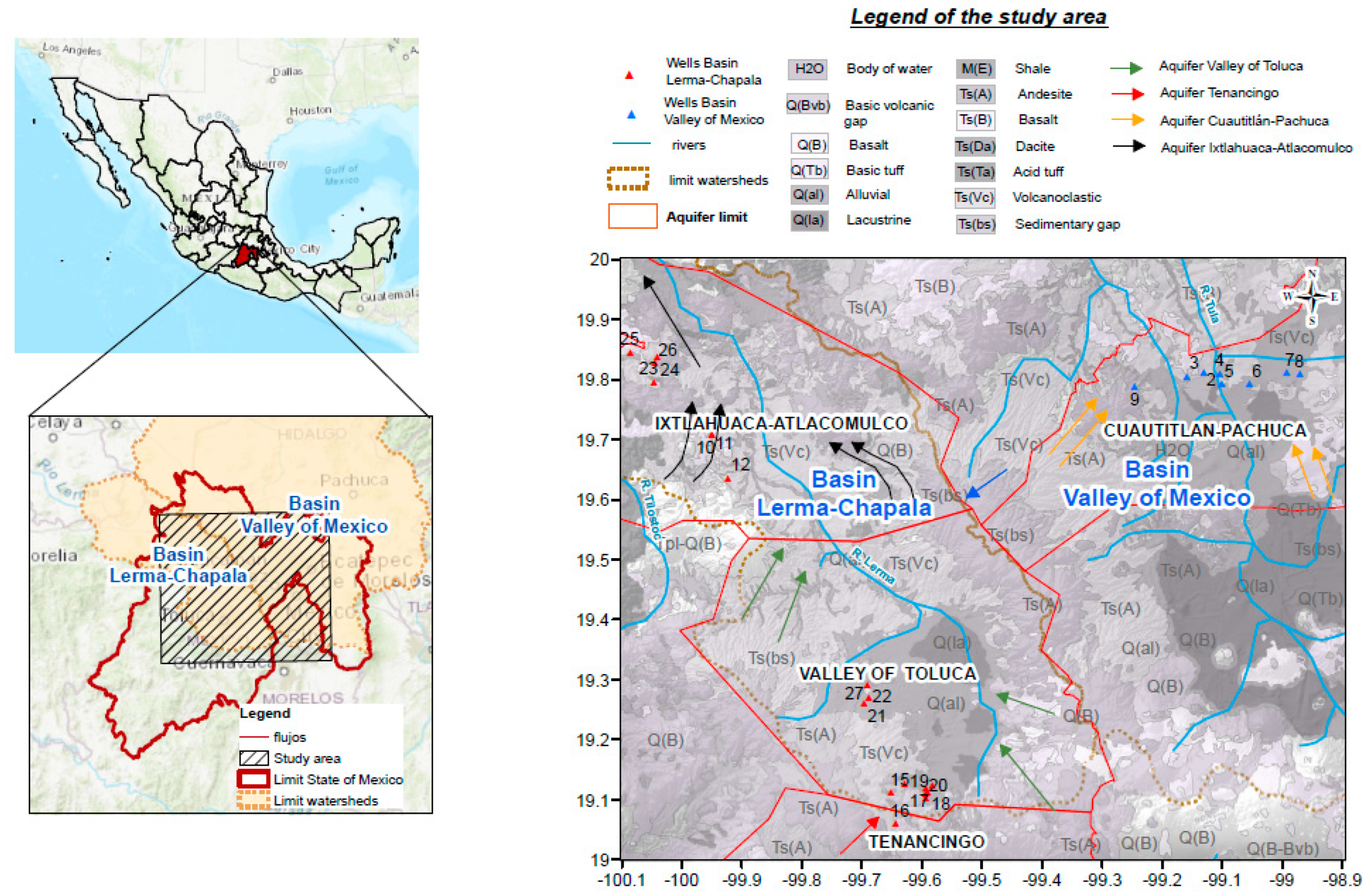
2.1. Study Area
2.2. Sampling
3. Results and Discussion
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Carrillo, J.; Cardona, A.; Edmunds, W. Use of abstraction regime and knowledge of hydrogeological conditions to control high-fluoride concentration in abstracted groundwater: San Luis Potosı basin, Mexico. J. Hydrol. 2002, 261, 24–47. [Google Scholar] [CrossRef]
- Mohapatra, M.; Anand, S.; Mishra, B.K.; Giles, D.E.; Singh, P. Review of fluoride removal from drinking water. J. Environ. Manag. 2009, 91, 67–77. [Google Scholar] [CrossRef] [PubMed]
- Edmunds, W.M.; Smedley, P.L. Fluoride in Natural Waters Springer. In Essentials of Medical Geology; Elsevier Academic Press: Schmallenberg, Germany, 2005; p. 832. [Google Scholar]
- Ozsvath, D.L. Fluoride and environmental health: A review. Rev. Environ. Sci. Bio/Technol. 2009, 8, 59–79. [Google Scholar] [CrossRef]
- World Health Organization (WHO). Fluoride in Drinking-Water Background Document for Development of WHO Guidelines for Drinking-Water Quality; WHO: Geneva, Switzerland, 2004. [Google Scholar]
- Harrison, P. Fluoride in water: A UK perspective. J. Fluor. Chem. 2005, 126, 1448–1456. [Google Scholar] [CrossRef]
- Fan, X.; Parker, D.J.; Smith, M.D. Adsorption kinetics of fluoride on low cost materials. Water Res. 2003, 37, 4929–4937. [Google Scholar] [CrossRef] [PubMed]
- Bhatnagar, A.; Kumar, E.; Sillanpää, M. Fluoride removal from water by adsorption—A review. Chem. Eng. J. 2011, 171, 811–840. [Google Scholar] [CrossRef]
- Ruiz, A.; Ortiz, M.; Duarte, M. Determination of fluoride in drinking water and in urine of adolescents living in three counties in Northern Chihuahua Mexico using a fluoride ion selective electrode. Microchem. J. 2005, 81, 19–22. [Google Scholar] [CrossRef]
- Mandinic, Z.; Curcic, M.; Antonijevic, B.; Carevic, M.; Mandic, J.; Djukic-Cosic, D.; Lekic, C.P. Fluoride in drinking water and dental fluorosis. Sci. Total Environ. 2010, 408, 3507–3512. [Google Scholar] [CrossRef] [PubMed]
- Pontigo, A.; Medina, C.; Borges, A.; Patiño, N.; Islas, A.; Maupome, G. Prevalence and severity of dental caries in adolescents aged 12 and 15 living in communities with various fluoride concentrations. J. Public Health Dent. 2007, 67, 8–13. [Google Scholar] [CrossRef]
- OMS. OMS|Guías Para la Calidad del Agua Potable, Tercera Edición; WHO: Geneva, Switzerland, 2006; Volume 3, pp. 300–301. [Google Scholar]
- Wang, S.; Wang, Z.; Cheng, X.; Li, J.; Sang, Z.; Zhang, X.; Han, L.; Qiao, X.; Wu, Z.; Wang, Z. Arsenic and Fluoride Exposure in Drinking Water: Children’s IQ and Growth in Shanyin County, Shanxi Province, China. Environ. Health Perspect. 2007, 115, 643–647. [Google Scholar] [CrossRef] [PubMed]
- De la Cruz, D.; Chaires, I.; Mejía, M.; Sandoval, A.; Bolaños, P. Análisis de la concentración de fluoruro en aguas embotelladas de diferentes entidades federativas de la República Mexicana Analysis of the concentration of fluoride in bottled water in different Mexican states. Rev. ADM 2013, 70, 81–90. [Google Scholar]
- Valenzuela, L.; Ramírez, J.; Sol, J.; Reyes, J. Alternativas para la eliminación doméstica de fluor en el agua de consumo humano. Inf. Tecnol. 2011, 22, 23–32. [Google Scholar] [CrossRef]
- Betancourt, A.; Irigoyen, M.E.; Mejía, A.; Zepeda, M.; Sánchez, L. Prevalencia de fluorosis dental en localidades Mexicanas ubicadas en 27 estados y el D.F. A seis años de la publicación de la Norma Oficial Mexicana para la fluoruración de la sal. Rev. Investig. Clin. 2013, 65, 237–247. [Google Scholar]
- Porter, T.A.; Jóhannesson, G.; Moskalenko, I.V. The FRaNKIE code: A tool for calculating multi-wavelength interstellar emissions in galaxies. In Proceedings of the 34th International Cosmic Ray Conference (ICRC2015), The Hague, The Netherlands, 30 July–6 August 2015. [Google Scholar]
- La, D.E. Real academia nacional de medicina. Real Acad. Nac. Med. 1980, XCVII, 429–462. [Google Scholar]
- Instituto Nacional de Ecología (INE). Diagnóstico Bio-Físico y Socio-Económico de la Cuenca Lerma-Chapala; INE: Mexico City, México, 2003; Volume 58, pp. 8–19. [Google Scholar]
- CONAGUA. Determinación de la Disponibilidad de Agua en el Acuífero Valle de Toluca, Estado de México; Comision Nacional del Agua: Mexico City, Mexico, 2002. [Google Scholar]
- Sánchez Hernández, J.M. Redes de Monitoreo del Agua Subterránea en el Acuífero del Valle de Toluca. Propuesta de su Distribución Espacial Mediante el Manejo de Factores Hidrogeológicos, Ambientales y Socioeconómicos. Master’s Thesis, Universidad Autónoma del Estado de Morelos, Cuernavaca, Mexico, 2013. [Google Scholar]
- Del Campo, M.M.; Hirata, R.; Esteller, M.; Expósito, J. Dinámica De Niveles Piezométricos en un Acuífero Multicapa Sometido a Explotación Intensiva: Acuífero Del Valle De Toluca, México; Rev. Latino-Americana de Hidrogeologia: Mexico, Mexico, 2012; pp. 88–99. [Google Scholar]
- Brunett, E.; Baró, J.; Cadena, E.; Esteller, M. Pago por servicios ambientales hidrológicos: Caso de estudio Parque Nacional del Nevado de Toluca, México. CIENCIA Ergo-Sum 2010, 17, 286–295. [Google Scholar]
- Fuentes-Rivas, R.M.; Ramos-Leal, J.A.; Jiménez-Moleón, M.D.; Esparza-Soto, M. Caracterización de la materia orgánica disuelta en agua subterránea del Valle de Toluca mediante espectrofotometría de fluorescencia 3D. Rev. Int. Contam. Ambient. 2015, 31, 253–264. [Google Scholar]
- American Public Health Association (APHA). Standard Methods for Examination of Water and Wastewater (Standard Methods for the Examination of Water and Wastewater); APHA: Washington, DC, USA, 2012. [Google Scholar]
- Millán, F.; Mathison, J.; Alvares, M.; Jarbouh, W. Estudio comparativo de la dureza del agua en el estado Mérida y algunas localidades del centro y occidente de Venezuela. Cienc. Ing. 2003, 24, 39–46. [Google Scholar]
- Frape, S.K.; Blyth, A.; Blomqvist, R.; McNutt, R.H.; Gascoyne, M. Deep Fluids in the Continents: II. Crystalline Rocks. Treatise Geochem. 2003, 5, 605. [Google Scholar]
- Fonseca, R. Comportamiento fisicoquímico e hidrogeoquímico del agua del acuífero del Valle de Toluca y su impacto en la calidad de agua de consumo. Ph.D. Thesis, Universidad Autónoma de Nuevo León, San Nicolás de los Garza, N.L., Mexico, 2019. [Google Scholar]
- Fakharany, E.; Gomma, M.; Gado, H.; Eissa, M. Relation of groundwater composition to weathering and alteration of silicate minerals in Basement aquifer at Wadi Hodein Basin, southeastern Desert, Egypt. Available online: https://www.researchgate.net/publication/306055906_Relation_of_groundwater_composition_to_weathering_and_alteration_of_silicate_minerals_in_Basement_aquifer_at_Wadi_Hodein_Basin_southeastern_Desert_Egypt (accessed on 18 June 2019).
- Hem, J. Study and Interpretation of the Chemical Characteristics of Natural Water. Engineering 1989, 3, 248. [Google Scholar]
- Saha, D.; Kumar, S.; Udayabhanu, G.; Venkatesh, A.S.; Singh, R. Geochemical signatures and isotopic systematics constraining dynamics of fluoride contamination in groundwater across Jamui district, Indo-Gangetic alluvial plains, India. Chemosphere 2018, 205, 493–505. [Google Scholar]
- Alanís, J. Estudio bacterológico y contenido de fluoruro del agua de consumo en 858 localidades del Estado de México, tercera época. In Coordinación General de Investigación y Estudios Avanzados; Cuaderno de investigación, Facultad de Odontología, UAEMEX: Toluca, Edo. De México, Mexico, 1987; pp. 7–45. [Google Scholar]
- Daesslé, L.; Ruíz, L.; Tobschall, H.; Chandrajith, R.; Camacho, V.; Mendoza, L.; Quintanilla, A.; Lugo, K. Fluoride, nitrate and water hardness in groundwater supplied to the rural communities of Ensenada County, Baja California, Mexico. Environ. Geol. 2009, 58, 419–429. [Google Scholar] [CrossRef]
- Navarro, O.; González, J.; Júnez-Ferreira, H.E.; Bautista, C.F.; Cardona, A. Correlation of Arsenic and Fluoride in the Groundwater for Human Consumption in a Semiarid Region of Mexico. Procedia Eng. 2017, 186, 333–340. [Google Scholar] [CrossRef]
- Yamanaka, M.; Bottrell, S.H.; Wu, J.; Kumagai, Y.; Mori, K.; Satake, H. Chlorine stable isotope evidence for salinization processes of confined groundwater in southwestern Nobi Plain aquifer system, central Japan. J. Hydrol. 2014, 519, 295–306. [Google Scholar] [CrossRef]
- Jiménez-Moleón, M.D.; Fuentes-Rivas, R.M.; Mota-González, M.T.; Islas-Espinoza, M. Groundwater hydrochemistry of an overexploited aquifer: The case of the Toluca Valley, Mexico. WIT Trans. Ecol. Environ. 2010, 135, 27–37. [Google Scholar]
- Alvarez, C.; Martínez, V.; Solache, M.; Linares, I.; Teutli, A.; Vázquez, G. Drinking water characterization and removal of manganese. Removal of manganese from water. J. Environ. Chem. Eng. 2018, 6, 2119–2125. [Google Scholar] [CrossRef]
- Ortega, A.M. Presencia, distribución, hidrogeoquímica y origen de arsénico, fluoruro y otros elementos traza disueltos en agua subterránea, a escala de cuenca hidrológica tributaria de Lerma-Chapala, México. Rev. Mex. Cienc. Geol. 2009, 26, 143–161. [Google Scholar]






| ID | Depth | T | pH | EC | F− | Cl− | Alkalinity | Cl2 | Hardness | HCO3− | TDS |
|---|---|---|---|---|---|---|---|---|---|---|---|
| (m) | (°C) | (µS/cm) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | ||
| MPL | 6.5–8.5 | 1.5 | 250 | 300 | 0.2–1.5 | 500 | N.E | 1000 | |||
| 1 | - | 19.9 | 7.4 | 284.0 | 0.17 | 25.4 | 181.0 | S/C | 159.0 | 221.0 | 181.6 |
| 10 | 96 | 25.5 | 7.7 | 391.0 | 0.26 | 0.0 | 290.0 | S/C | 159.0 | 354.0 | 249.9 |
| 11 | 72 | 21.2 | 7.4 | 609.0 | 0.49 | 7.8 | 387.0 | S/C | 249.0 | 472.0 | 390.0 |
| 12 | - | 23.1 | 7.9 | 281.0 | 0.17 | 3.9 | 146.0 | S/C | 149.0 | 178.0 | 179.9 |
| 13 | 137 | 19.1 | 7.2 | 217.0 | 0.23 | 7.8 | 130.0 | S/C | 119.0 | 159.0 | 138.9 |
| 14 | - | 18.0 | 7.0 | 258.0 | 0.12 | 0.1 | 130.0 | S/C | 133.0 | 159.0 | 165.3 |
| 15 | - | 18.0 | 7.2 | 157.0 | 0.18 | 3.9 | 125.0 | S/C | 87.0 | 152.0 | 100.5 |
| 16 | 300 | 18.0 | 7.3 | 99.0 | 0.13 | 7.8 | 103.0 | 1.0 | 103.0 | 126.0 | 63.14 |
| 17 | - | 20.0 | 8.0 | 307.0 | 0.16 | 11.7 | 146.0 | S/C | 189.0 | 178.0 | 196.7 |
| 18 | 100 | 16.2 | 7.3 | 167.0 | 0.29 | 11.7 | 144.0 | 0.3 | 77.0 | 176.0 | 106.6 |
| 19 | 300 | 16.5 | 7.8 | 162.0 | 0.23 | 7.8 | 139.0 | 0.3 | 129.0 | 169.0 | 103.7 |
| 20 | 200 | 13.8 | 7.9 | 264.0 | 1.31 | 7.8 | 114.0 | 0.5 | 70.0 | 139.0 | 169.1 |
| 21 | - | 18.8 | 7.5 | 287.0 | 0.21 | 16.9 | 118.0 | 0.8 | 101.0 | 143.0 | 183.9 |
| 22 | 250 | 19.3 | 7.5 | 300.0 | 0.06 | 25.7 | 102.0 | 1.0 | 172.0 | 124.0 | 192 |
| 23 | 115 | 25.4 | 7.4 | 206.0 | 0.06 | 10.3 | 87.0 | 1.3 | 87.0 | 106.0 | 132.0 |
| 24 | 140 | 24.4 | 7.6 | 307.0 | 0.32 | 12.5 | 132.0 | 1.0 | 117.0 | 161.0 | 196.6 |
| 25 | 200 | 25.6 | 7.6 | 340.0 | 0.36 | 24.8 | 132.0 | 1.0 | 127.0 | 161.0 | 217.6 |
| 26 | 200 | 21.9 | 7.7 | 423.0 | 0.21 | 27.9 | 166.0 | 1.3 | 160.0 | 202.0 | 270.9 |
| 27 | 200 | 19.9 | 7.5 | 244.0 | 0.019 | 2.3 | 156.0 | 0.0 | 98.0 | 191.0 | 155.9 |
| max | 300 | 25.6 | 8.0 | 609.0 | 1.31 | 27.9 | 387.0 | 1.3 | 249.0 | 472.0 | 390.0 |
| min | 72 | 13.8 | 7.0 | 99.0 | 0.02 | 0.0 | 87.0 | 0.0 | 70.0 | 106.0 | 63.1 |
| average | 20.20 ± 3.36 | 7.51 ± 0.28 | 269.1 ± 113.67 | 0.30 ± 0.28 | 11.4 ± 8.83 | 154 ± 70.62 | 0.80 ± 0.51 | 130.8 ± 44.31 | 198.5 ± 89.89 | 178.7 ± 72.8 |
| ID | Depth | T | pH | EC | F− | Cl− | Alkalinity | Cl2 | Hardness | HCO3− | TDS |
|---|---|---|---|---|---|---|---|---|---|---|---|
| (m) | (°C) | (µS/cm) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | ||
| MPL | 6.5–8.5 | 1.5 | 250 | 300 | 0.2–1.5 | 500 | N.E | 1000 | |||
| 2 | 110 | 23.9 | 7.0 | 1388 | 1.06 | 79.9 | 538 | S/C | 129 | 656 | 888.3 |
| 3 | 82 | 25.1 | 7.6 | 1237 | 1.01 | 62.4 | 574 | 0.7 | 179 | 700 | 791.7 |
| 4 | 90 | 24.5 | 7.8 | 1230 | 0.79 | 79.9 | 452 | S/C | 204 | 551 | 786.2 |
| 5 | 120 | 27.2 | 7.5 | 1642 | 0.70 | 124.8 | 600 | S/C | 244 | 732 | 1050.9 |
| 6 | - | 30.2 | 7.8 | 1053 | 0.51 | 99.4 | 372 | S/C | 149 | 454 | 673.9 |
| 7 | - | 23.1 | 7.8 | 1193 | 0.39 | 60.4 | 413 | S/C | 269 | 504 | 763.5 |
| 8 | - | 26.2 | 7.3 | 1346 | 0.29 | 36.6 | 641 | S/C | 458 | 782 | 861.4 |
| 9 | - | 21.8 | 7.7 | 492 | 0.36 | 13.7 | 335 | S/C | 189 | 408 | 315.1 |
| max | 120 | 30.2 | 7.8 | 1642 | 1.06 | 124.8 | 641 | 0.7 | 458 | 782 | 1050.9 |
| min | 82 | 21.8 | 7.0 | 492 | 0.29 | 13.7 | 335 | 0.7 | 129 | 408 | 315.1 |
| average | 24.94 ± 2.31 | 7.57 ± 0.23 | 904.75 ± 509.8 | 0.51 ± 0.31 | 52.72 ± 37.65 | 370.17 ± 200.2 | 1.06 ± 0.56 | 192.67 ± 98.82 | 421.36 ± 231.78 | 766.4 ± 213.2 |
| ID | Ca2+ | Mg2+ | Na+ | K+ | SO42− | NO3− |
|---|---|---|---|---|---|---|
| (mg/L) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | |
| 2 | 14.81 | 18.71 | 31.00 | 6.05 | 71.59 | 6.3 |
| 3 | 19.04 | 11.77 | 19.40 | 3.74 | 43.63 | 7.8 |
| 4 | 19.04 | 96.85 | 161.53 | 31.47 | 67.92 | 9.2 |
| 5 | 10.58 | 48.76 | 81.19 | 15.79 | 108.66 | 4.2 |
| max | 19.04 | 96.85 | 161.53 | 31.47 | 108.66 | 9.2 |
| min | 10.58 | 11.77 | 31.00 | 6.05 | 43.63 | 4.2 |
| average | 15.87 ± 4.05 | 44.02 ± 38.71 | 73.28 ± 64.66 | 14.25 ± 12.61 | 72.95 ± 26.85 | 6.88 ± 2.14 |
| ID | Ca2+ | Mg2+ | Na+ | K+ | SO42− | NO3− |
|---|---|---|---|---|---|---|
| (mg/L) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | (mg/L) | |
| 16 | 8 | 2.43 | 3.8 | 0.701 | 6.62 | 6.2 |
| 18 | 10 | 4.13 | 6.65 | 1.25 | 13.42 | 1.4 |
| 19 | 10 | 5.59 | 9.08 | 1.73 | 7.07 | 7.8 |
| 20 | 22 | 3.89 | 6.24 | 1.18 | 27.67 | 12 |
| 23 | 14 | 6.32 | 10.3 | 1.97 | 8.13 | 6.6 |
| 24 | 18 | 8.75 | 14.36 | 2.76 | 6.01 | 2.2 |
| 25 | 20 | 12.88 | 21.27 | 4.11 | 9.1 | 5.6 |
| 26 | 20 | 18.23 | 30.2 | 5.85 | 26.07 | 2.5 |
| max | 22 | 18.23 | 30.2 | 5.85 | 27.67 | 12 |
| min | 8 | 2.43 | 3.8 | 0.7 | 6.01 | 1.4 |
| average | 16.14 ± 5.76 | 7.24 ± 5.24 | 11.84 ± 8.75 | 2.27 ± 1.71 | 13.01 ± 8.86 | 5.54 ± 3.50 |
| F− | pH | EC | T | Cl− | Alkal | Cl2 | Hard | HCO3− | STD | |
|---|---|---|---|---|---|---|---|---|---|---|
| F− | 1.0000 | |||||||||
| pH | 0.0833 | 1.0000 | ||||||||
| CE | 0.5933 | 0.2389 | 1.0000 | |||||||
| T | 0.1909 | 0.3172 | 0.4726 | 1.0000 | ||||||
| Cl− | −0.0208 | 0.1609 | 0.2091 | 0.1175 | 1.0000 | |||||
| Alcal | 0.6924 | 0.1061 | 0.7881 | 0.2245 | −0.2051 | 1.0000 | ||||
| Cl2 | −0.3520 | −0.1225 | 0.4659 | 0.7574 | 0.4242 | −0.2310 | 1.0000 | |||
| Hard | 0.3367 | 0.3257 | 0.7778 | 0.2026 | 0.0947 | 0.7159 | 0.4018 | 1.0000 | ||
| HCO3− | 0.6924 | 0.1061 | 0.7881 | 0.2245 | −0.2051 | 1.0000 | −0.2310 | 0.7159 | 1.0000 | |
| STD | 0.5933 | 0.2389 | 1.0000 | 0.4726 | 0.2091 | 0.7881 | 0.4659 | 0.7779 | 0.7881 | 1.0000 |
| F− | T | pH | CE | Cl− | Alkalinity | Hardness | HCO3− | STD | |
|---|---|---|---|---|---|---|---|---|---|
| F− | 1.0000 | ||||||||
| T | 0.2247 | 1.0000 | |||||||
| pH | −0.4236 | 0.0624 | 1.0000 | ||||||
| CE | 0.5493 | 0.4594 | −0.3998 | 1.0000 | |||||
| Cl− | 0.3525 | 0.8033 | −0.2157 | 0.8593 | 1.0000 | ||||
| Alkalinity | 0.7884 | 0.2220 | −0.7585 | 0.8272 | 0.5695 | 1.0000 | |||
| Hardness | −0.2153 | −0.4211 | −0.2490 | 0.4326 | 0.1138 | 0.2706 | 1.0000 | ||
| HCO3− | 0.7884 | 0.2220 | −0.7585 | 0.8272 | 0.5699 | 1.0000 | 0.2706 | 1.0000 | |
| STD | 0.5493 | 0.4594 | −0.3998 | 1.0000 | 0.8593 | 0.8272 | 0.4326 | 0.8272 | 1.0000 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Valdez-Alegría, C.J.; Fuentes-Rivas, R.M.; García-Rivas, J.L.; Fonseca-Montes de Oca, R.M.G.; García-Gaitán, B. Presence and Distribution of Fluoride Ions in Groundwater for Human in a Semiconfined Volcanic Aquifer. Resources 2019, 8, 116. https://doi.org/10.3390/resources8020116
Valdez-Alegría CJ, Fuentes-Rivas RM, García-Rivas JL, Fonseca-Montes de Oca RMG, García-Gaitán B. Presence and Distribution of Fluoride Ions in Groundwater for Human in a Semiconfined Volcanic Aquifer. Resources. 2019; 8(2):116. https://doi.org/10.3390/resources8020116
Chicago/Turabian StyleValdez-Alegría, Cecilia Judith, Rosa María Fuentes-Rivas, José Luis García-Rivas, Reyna María Guadalupe Fonseca-Montes de Oca, and Beatriz García-Gaitán. 2019. "Presence and Distribution of Fluoride Ions in Groundwater for Human in a Semiconfined Volcanic Aquifer" Resources 8, no. 2: 116. https://doi.org/10.3390/resources8020116
APA StyleValdez-Alegría, C. J., Fuentes-Rivas, R. M., García-Rivas, J. L., Fonseca-Montes de Oca, R. M. G., & García-Gaitán, B. (2019). Presence and Distribution of Fluoride Ions in Groundwater for Human in a Semiconfined Volcanic Aquifer. Resources, 8(2), 116. https://doi.org/10.3390/resources8020116





