1. Introduction
Prevalence of sensitive skin worldwide is said to have steadily increased over time. A review of the prevalence of sensitive skin communicated figures of ≈60–70% among women and ≈50–60% among men [
1]. A recent large-scale survey conducted on 10,743 volunteers from Brazil, China, France Russia, and the United States, with an even distribution of men and women, reported a rate of 48.2% of self-reported sensitive skin [
2].
Skin barrier defect is a frequently cited attribute of sensitive skin [
3], notably as a more permeable skin would be more prone to react to external stimuli. Levels of inflammatory biomarkers such as cytokines, proinflammatory PUFAs and prostaglandin E2 are hallmarks of sensitive skin [
4,
5]. There is nowadays a greater emphasis on the skin’s nociception capabilities, or transduction of noxious stimuli to nerve fibre free endings located in the epidermis, to explain the pathophysiology of sensitive skin. The superfamily of Transient Receptor Protein (TRP), expressed in both neuronal cells and keratinocytes, can be elicited by stimuli of chemical or physical nature, to signal pain and itching. Vanilloid and Ankyrin families of TRP have been the foci of various investigations, notably TRPV1 and TRPA1 [
6,
7]. Descriptions of perceived discomfort may vary across ethnic groups as shown previously, with Caucasians relying more on visual cues (e.g., redness, swelling) than pain descriptors (e.g., stinging, burning), compared to Asians or African Americans [
1].
Skin discomfort in response to climatic and environmental factors have been previously highlighted in a Korean population [
8]. Pollution was reported as a key factor in another study among 286 Mexican women, particularly in the cohort based in Mexico City [
9]. Temperature shift, sun exposure and stress have been cited in multiple countries. Moreover, newer insights from a lifestyle perspective have been uncovered, with greater instances of sleep disorders, fatigue, sweating or dust and food or tobacco consumption in people with sensitive skin, over people that do not report sensitive skin. A hormonal component was also suggested from these results, with a higher incidence of sensitive skin among pregnant women, women with painful menstruation or those using contraceptive pills [
2].
Thus, skin sensitivity has become a dynamic skin care segment with targeted solutions. Products aimed at alleviating this burden can be found on the market and in the literature, with, for instance, a facial gel containing niacinamide,
Hamamelis virginiana (witch hazel) and
Avena sativa (oat) kernel extract, albeit without anti-ageing endpoints in vivo [
10]. A recent review has compiled a thorough list of technologies addressing sensitive skin, launched over the last few years. Mentioned among the recurring mechanisms were: blockage of TRPV1 receptors, targeting key inflammatory pathways (NF-κB), modulation of inflammatory biomarker levels such as IL-1α/IL-1-/IL-8/CGRP/PEG2, NGF reduction, substance P reduction, inhibition of mast cells degranulation and binding to µ-opioid receptors [
11]. We previously disclosed data on the cumulative anti-ageing effect of using a comprehensive skin care regimen in comparison to a simpler daily routine (e.g., cleanser and day cream only) [
12]. Because extensive skin care routines may be met with reticence by people with reactive skin, we aimed at developing a 4-step routine that combines anti-ageing performance, desirable tolerability and ability to improve the burden of sensitive skin. In the present article, we disclose the approach to designing anti-ageing skin care routines suitable for sensitive skin from a formulation, safety and clinical efficacy standpoint.
4. Discussion
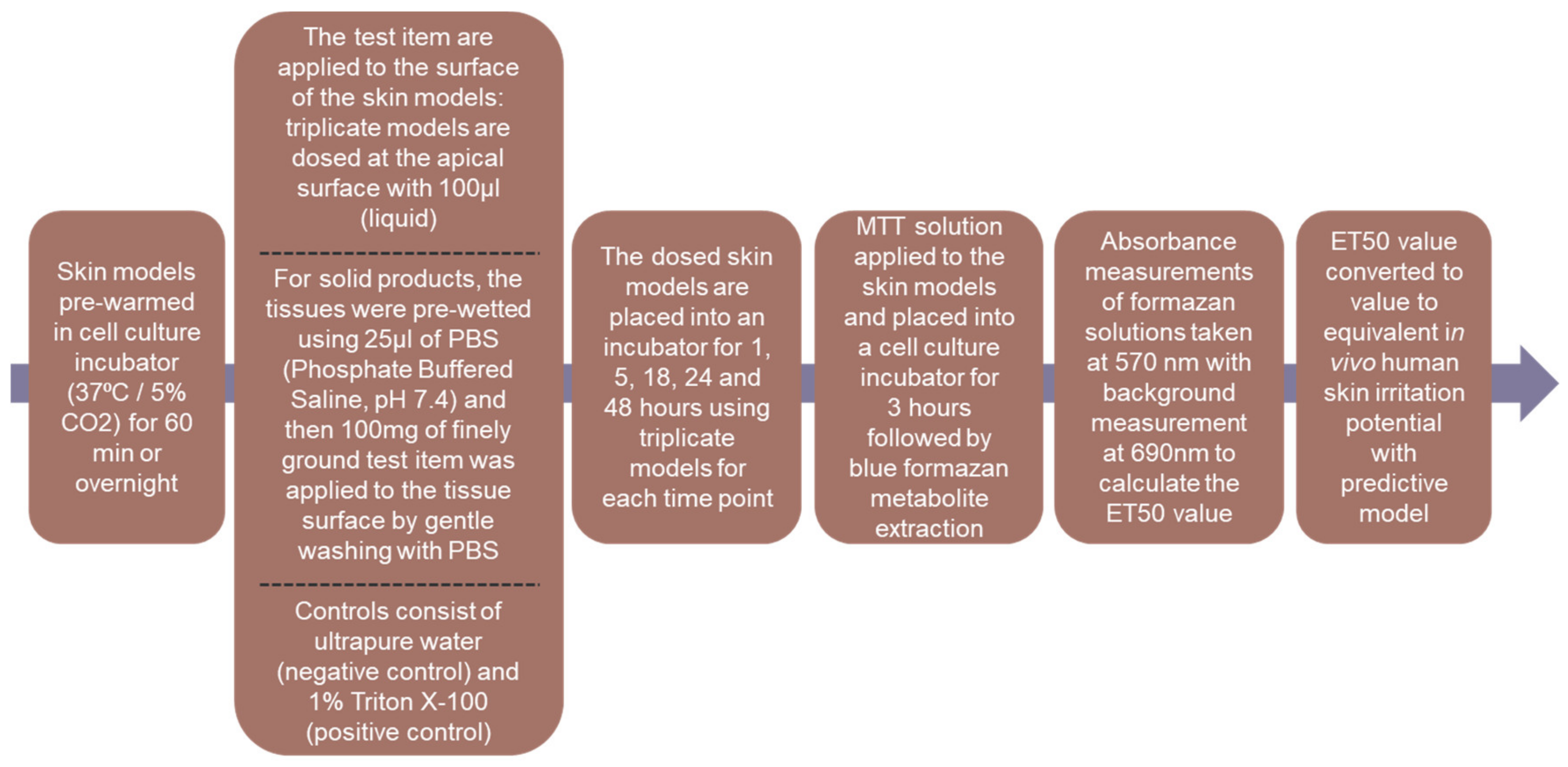
The in vitro assessment of the skin irritation potential following the XtraMild skin mildness test was performed, using reconstructed human epidermis method (ET50). This method provided a pre-screen, prior to human volunteer studies, driving key decisions in formulation development. The XtraMild testing was performed on nine items to define their ET50 values and correlate the results to the expected in vivo skin irritancy from applying the relevant product on the skin. Cleansers A and C are very similar in terms of formulation and their respective ET50 values were both higher than Cleanser B with an expected ‘Moderate to Mild’ in vivo skin irritancy. Ultimately, Cleanser C was chosen as Cleanser for the sensitive skin care range. Irrespective of the product tested, all scored at least ‘Moderate to Mild’ and were chosen to continue with clinical testing and product development. The serum exhibited—by far—the best mildness of all products screened in vitro. With regards to its composition, this formula stands out with much lower levels of tensioactive ingredients compared to the other products and may explain this excellent tolerability. Overall, there was a moderate correlation between the level of tensioactive ingredients, irrespective of function (e.g., surfactants, solubilizer, emulsifier) and the mildness score of the products (
Figure S2), and this further highlights the need to optimize the levels of this category of ingredient.
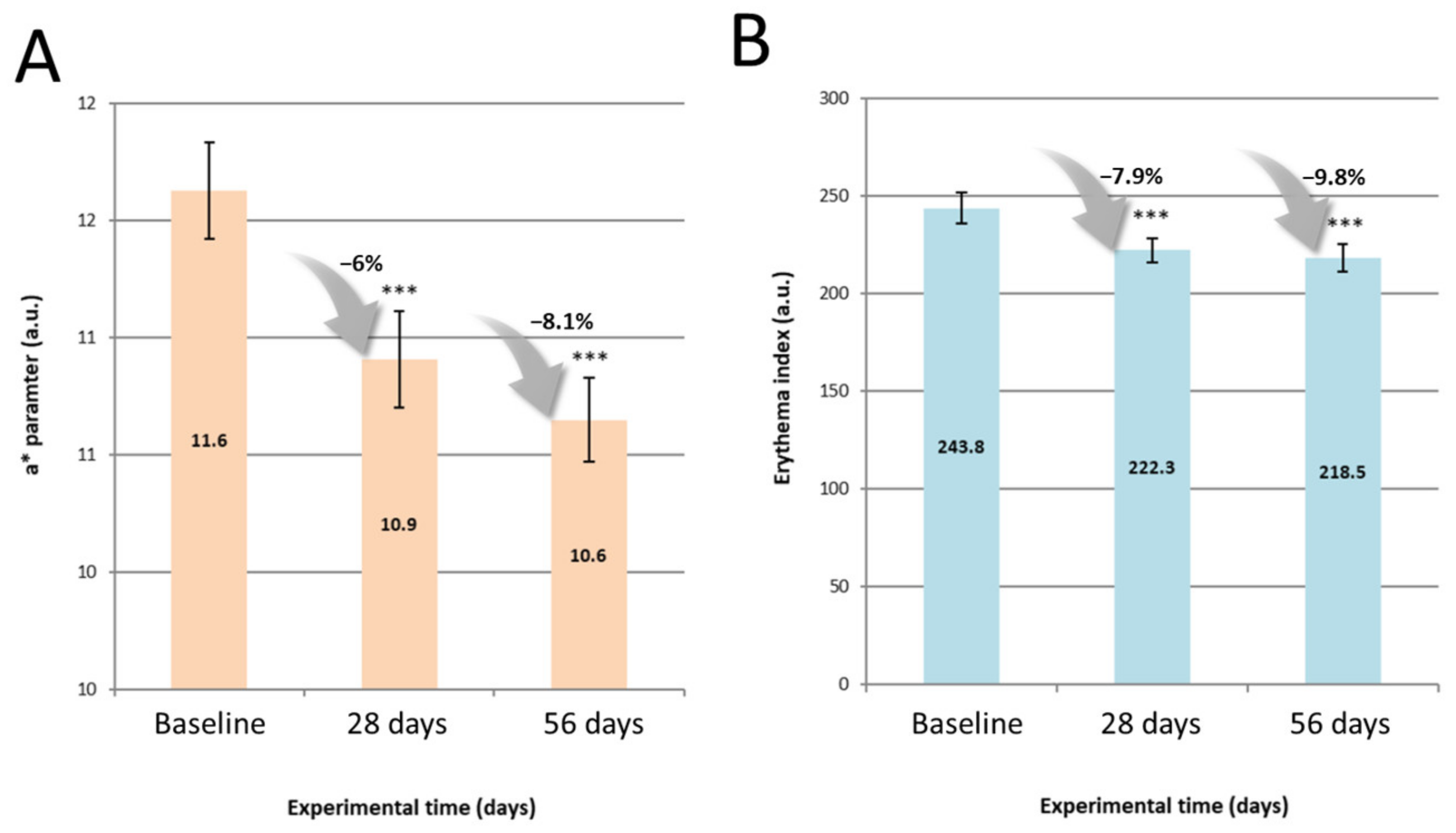
Following the pre-screening in vitro, the benefits of a 4-step routine at improving signs of ageing and skin discomfort were assessed by means of instrumental and clinical evaluation. Topographic features such as wrinkles and roughness were improved as early as 28 days of treatment and sustained until 56 days. Similarly, biomechanical properties of the skin were enhanced at all timepoints, with a greater capability to withstand stretch and to return to its initial state following deformation. Both topographic features and skin biomechanical properties inevitably decline with age, have been extensively discussed in the literature among various ethnic groups [
25,
26] and, thus, were deemed ideal parameters to showcase anti-ageing benefits. In addition, clinical evaluation evidenced further benefits in terms of smoothness, suppleness and radiance after 56 days. Altogether, we can speculate that the choice of actives featured in the serum, conjugated with the layering effect of the routine yielded these positive outcomes. The full skincare range was well-tolerated by a carefully selected panel of sensitive skin volunteers, as evidenced with a lack of functional (e.g., stinging) and physical (e.g., erythema) signs throughout the study, thus confirming the relevance of the XtraMild ET50 in vitro approach used for mildness prediction prior to the clinical stage. Performance and gentleness to the skin should not be antinomic, and the present study features elements that invalidate a misconception popularized by widespread retinol use: skin reactions happen because the product is working on the skin [
27]. Beyond being suitable for sensitive skin in terms of tolerability, the range was found to improve skin tolerance towards a chemical stimulus such as lactic acid, thus alleviating discomfort mediated by keratinocyte–nerve fibre interactions. Thus, following the guidelines described in the method for formulating a range of products enables us to seamlessly combine performance and tolerability. The decision to conduct this study with an Asian panel was mostly because Asian skin is prone to greater reactivity to cosmetic products, lactic acid, capsaicin and SLS, compared to Caucasian skin [
28].
Ultimately, volunteers reported an improvement in their dermatological quality of life. Although significant, improvement in DLQI appeared to be of modest amplitude. It must be borne in mind that the burden of sensitive skin is nonetheless lower than known skin dermatoses, such as acne or psoriasis [
29], thus with a reduced margin for improvement. From our findings, improvement in TEWL seemed to be the most apparent parameter to explain DLQI improvements, a sensible outcome since a less permeable skin would prevent most external stimuli (e.g., chemical, physical) to elicit a neurosensory response.
There are nonetheless opportunities to further collect evidence in the context of skincare solutions to manage sensitive skin burden. Assessing the mildness of similar product types for the rest of the product in the skincare range would be beneficial, as was done for the cleansers in this study. From a microbiome perspective, it appears that a shift in abundance of specific Proteobacteria types (Neisseriaceae and Neisseria), concomitantly with a lower diversity of species, are a footprint of sensitive skin, as reported in a recent investigation in women from Guangzhou [
30]. Assessing the rebalancing effect of skincare routines is worth considering, bearing in mind the overlap of several preservative systems within a routine. More recently, cytokines and polyunsaturated fatty acid (PUFA) sampled in vivo, have been shown to be relevant inflammatory markers of sensitive skin in a paediatric cohort [
4]. The lactic acid test used in this study remains a relevant trigger, as seen with its larger effect upon brain activity for people with sensitive skin [
31], and provides evidence of a stronger resilience of the skin towards chemical stimuli. The additional investigation with regards to mask wearing relief hints at potential novel application of sensitive skin care and would deserve further focus as it was conducted with a very modest panel. On that note, it would be of interest to design protocols including other known skin discomfort triggers reported in the literature, such as temperature shift or pollution [
2,
8,
9,
32]. Inclusion of a Neurometer or Quantitative Sensory Testing is also relevant in in vivo instrumental approaches to directly assess the threshold needed to elicit itching/stinging in unmyelinated C-fibres and thickly myelinated Aδ-fibres [
32,
33].
A relevant skincare routine can improve both signs of ageing and skin resilience to external stimuli, thus reducing the likelihood of sensitive skin occurrence. Beyond TRPA1 Receptor potential channels (TRP), other targets can be considered for sensitive skin management, namely: G protein-coupled receptors (GPCRs), release of pruritogenic molecules involved in itching (e.g., Substance P, neutrophins) or TRP indirect activation mediated by GPCRs [
34].