Ternary Polymer Solar Cells: Impact of Non-Fullerene Acceptors on Optical and Morphological Properties
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Impact of the Third Component on Absorption Spectra
3.2. Impact of the Third Component on Solar Cell Efficiency
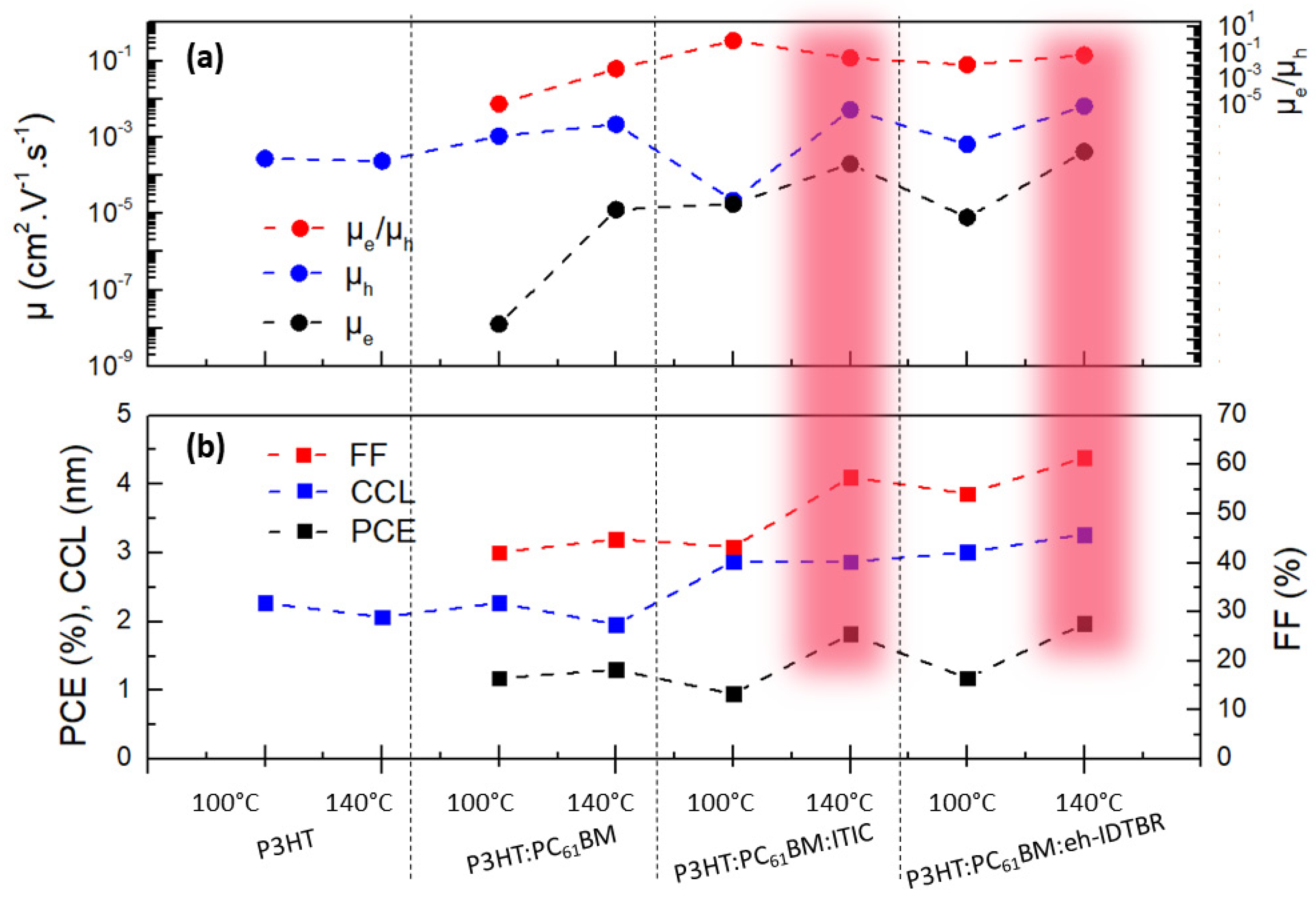
3.3. Impact of the Third Component on Charge Transport Properties
3.4. Impact of the Third Component on Molecular Ordering
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Scharber, M.C.; Sariciftci, N.S. Efficiency of bulk-heterojunction organic solar cells. Prog. Polym. Sci. 2013, 38, 1929–1940. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Zheng, T.; Wu, Q.; Schneider, A.M.; Zhao, D.; Yu, L. Recent advances in bulk heterojunction polymer solar cells. Chem. Rev. 2015, 115, 12666–12731. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Kramer, E.J.; Heeger, A.J.; Bazan, G.C. Bulk heterojunction solar cells: Morphology and performance relationships. Chem. Rev. 2014, 114, 7006–7043. [Google Scholar] [CrossRef] [PubMed]
- Nelson, J. Polymer:fullerene bulk heterojunction solar cells. Mater. Today 2011, 14, 462–470. [Google Scholar] [CrossRef]
- Kim, H.; Shin, M.; Kim, Y. Distinct annealing temperature in polymer: Fullerene: Polymer ternary blend solar cells. J. Phys. Chem. C 2009, 113, 1620–1623. [Google Scholar] [CrossRef]
- Gasparini, N.; Salleo, A.; McCulloch, I.; Baran, D. The role of the third component in ternary organic solar cells. Nat. Rev. Mater. 2019, 4, 229–242. [Google Scholar] [CrossRef]
- Doumon, N.Y.; Yang, L.; Rosei, F. Ternary organic solar cells: A review of the role of the third element. Nano Energy 2022, 94, 106915. [Google Scholar] [CrossRef]
- Ameri, T.; Khoram, P.; Min, J.; Brabec, C.J. Organic ternary solar cells: A review. Adv. Mater. 2013, 25, 4245–4266. [Google Scholar] [CrossRef]
- Bi, P.; Hao, X. Versatile ternary approach for novel organic solar cells: A review. Sol. RRL 2019, 3, 1800263. [Google Scholar] [CrossRef]
- Zhang, Y.; Li, G. Functional third components in nonfullerene acceptor-based ternary organic solar cells. Acc. Mater. Res. 2020, 1, 158–171. [Google Scholar] [CrossRef]
- Zhan, L.; Li, S.; Li, Y.; Sun, R.; Min, J.; Chen, Y.; Fang, J.; Ma, C.-Q.; Zhou, G.; Zhu, H.; et al. Manipulating charge transfer and transport via intermediary zlectron acceptor channels enables 19.3% efficiency organic photovoltaics. Adv. Energy Mater. 2022, 12, 2201076. [Google Scholar] [CrossRef]
- Ma, R.; Yan, C.; Fong, P.W.-K.; Yu, J.; Liu, H.; Yin, J.; Huang, J.; Lu, X.; Yan, H.; Li, G. In situ and ex situ investigations on ternary strategy and co-solvent effects towards high-efficiency organic solar cells. Energy Environ. Sci. 2022, 15, 2479–2488. [Google Scholar] [CrossRef]
- Kim, C.S.; Tinker, L.L.; Di Salle, B.F.; Gomez, E.D.; Lee, S.; Bernhard, S.; Loo, Y.-L. Altering the thermodynamics of phase separation in inverted bulk-heterojunction organic solar cells. Adv. Mater. 2009, 21, 3110–3115. [Google Scholar] [CrossRef]
- Xu, X.; Zhang, G.; Yu, L.; Li, R.; Peng, Q. P3HT-based polymer solar cells with 8.25% efficiency enabled by a matched molecular acceptor and smart green-solvent processing technology. Adv. Mater. 2019, 31, 1906045. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Wang, J.; Zhang, Z.-G.; Bai, H.; Li, Y.; Zhu, D.; Zhan, X. An Electron Acceptor Challenging Fullerenes for Efficient Polymer Solar Cells. Adv. Mater. 2015, 27, 1170–1174. [Google Scholar] [CrossRef] [PubMed]
- Yuan, J.; Zhang, Y.; Zhou, L.; Zhang, G.; Yip, H.-L.; Lau, T.-K.; Lu, X.; Zhu, C.; Peng, H.; Johnson, P.A.; et al. Single-Junction Organic Solar Cell with over 15% Efficiency Using Fused-Ring Acceptor with Electron-Deficient Core. Joule 2019, 3, 1140–1151. [Google Scholar] [CrossRef]
- Zhang, G.; Zhao, J.; Chow, P.C.Y.; Jiang, K.; Zhang, J.; Zhu, Z.; Zhang, J.; Huang, F.; Yan, H. Nonfullerene acceptor molecules for bulk heterojunction organic solar cells. Chem. Rev. 2018, 118, 3447–3507. [Google Scholar] [CrossRef] [PubMed]
- Holliday, S.; Ashraf, R.S.; Wadsworth, A.; Baran, D.; Yousaf, S.A.; Nielsen, C.B.; Tan, C.H.; Dimitrov, S.D.; Shang, Z.; Gasparini, N.; et al. High-efficiency and air-stable P3HT-based polymer solar cells with a new non-fullerene acceptor. Nat. Commun. 2016, 7, 11585. [Google Scholar] [CrossRef] [PubMed]
- Ben Dkhil, S.; Perkhun, P.; Luo, C.; Müller, D.; Alkarsifi, R.; Barulina, E.; Avalos Quiroz, Y.A.; Margeat, O.; Dubas, S.T.; Koganezawa, T.; et al. Direct correlation of nanoscale morphology and device performance to study photocurrent generation in donor-enriched phases of polymer solar cells. ACS Appl. Mater. Interfaces 2020, 12, 28404–28415. [Google Scholar] [CrossRef]
- Eynaud, Q.; Avalos Quiroz, Y.A.; Koganezawa, T.; Sato, R.; Yoshimoto, N.; Margeat, O.; Ruiz, C.M.; Ackermann, J.; Videlot-Ackermann, C. Towards efficient NFA-based selective near-infrared organic photodetectors: Impact of thermal annealing of polymer blends. J. Mater. Chem. C 2023, 11, 9657–9669. [Google Scholar] [CrossRef]
- Eynaud, Q.; El Amine Kramdi, M.; Kannampalli, V.; Koganezawa, T.; Yoshimoto, N.; Santinacci, L.; Ackermann, J.; Videlot-Ackermann, C. Towards air-stability of efficient filter-free band-selective organic photodetectors based on bulk heterojunction: Avoiding environmental degradation with atomic layer deposition encapsulation. Adv. Energy Sustain. Res. 2024, 2300262. [Google Scholar] [CrossRef]
- Blakesley, C.; Castro, F.A.; Kylberg, W.; Dibb, G.F.A.; Arantes, C.; Valaski, R.; Cremona, M.; Kim, J.S.; Kim, J.-S. Towards reliable charge-mobility benchmark measurements for organic semiconductors. Org. Electron. 2014, 15, 1263–1272. [Google Scholar] [CrossRef]
- Fei, Z.; Boufflet, P.; Wood, S.; Wade, J.; Moriarty, J.; Gann, E.; Ratcliff, E.L.; McNeill, C.R.; Sirringhaus, H.; Kim, J.-S.; et al. Influence of backbone fluorination in regioregular poly(3-alkyl-4- fluoro)thiophenes. J. Am. Chem. Soc. 2015, 137, 6866–6879. [Google Scholar] [CrossRef] [PubMed]
- Liu, H.-W.; Chang, D.-Y.; Chiu, W.-Y.; Rwei, S.-P.; Wang, L. Fullerene bisadduct as an effective phase-separation inhibitor in preparing poly(3-hexylthiophene)–[6,6]-phenyl-C61-butyric acid methyl ester blends with highly stable morphology. J. Mater. Chem. 2012, 22, 15586–15591. [Google Scholar] [CrossRef]
- Szymanski, R.; Henry, R.; Stuard, S.; Vongsaysyn, U.; Courtel, S.; Vellutini, L.; Bertrand, M.; Ade, H.; Chambon, S.; Wantz, G. Balanced charge transport optimizes industry-relevant ternary polymer solar cells. Sol. RRL 2020, 4, 2000538. [Google Scholar] [CrossRef]
- Ameri, T.; Min, J.; Li, N.; Machui, F.; Baran, D.; Forster, M.; Schottler, K.J.; Dolfen, D.; Scherf, U.; Brabec, C.J. Performance enhancement of the P3HT/PCBM solar cells through NIR sensitization using a small-bangap polymer. Adv. Energy Mater. 2012, 2, 1198–1202. [Google Scholar] [CrossRef]
- Bristow, H.; White, A.J.P.; Wadsworth, A.; Babics, M.; Hamid, Z.; Panidi, J.; McCulloch, I. Impact of nonfullerene acceptor side chain variation on transistor mobility. Adv. Electon. Mater. 2019, 5, 1900344. [Google Scholar] [CrossRef]
- Sun, Q.; Zhang, F.; Wang, J.; An, Q.; Zhao, C.; Li, L.; Teng, F.; Hu, B. A two-step strategy to clarify the roles of a solution processed PFN interfacial layer in highly efficient polymer solar cells. J. Mater. Chem. A 2015, 3, 18432–18441. [Google Scholar] [CrossRef]
- Lv, R.; Chen, D.; Liao, X.; Chen, L.; Chen, Y. A thermally tetrafluorinated nonfullerene acceptor for well-performing alloy ternary solar cells. Adv. Funct. Mater. 2019, 29, 1805872. [Google Scholar] [CrossRef]
- Pascual-San-José, R.; Rodríguez-Martínez, X.; Adel-Abdelaleim, R.; Stella, M.; Martínez-Ferrero, E.; Campoy-Quiles, M. Blade coated P3HT:non-fullerene acceptor solar cells: A high-throughput parameter study with a focus on up-scalability. J. Mater. Chem. C 2019, 7, 20369–20382. [Google Scholar] [CrossRef]
- Li, C.; Li, M.; Ni, Z.; Guan, Q.; Blackman, B.R.K.; Saiz, E. Stimuli-responsive surfaces for switchable wettability and adhesion. J. R. Soc. Interface 2021, 18, 20210162. [Google Scholar] [CrossRef] [PubMed]
- Avalos-Quiroz, Y.A.; Koganezawa, T.; Perkhun, P.; Barulina, E.; Ruiz, C.M.; Ackermann, J.; Yoshimoto, N.; Videlot-Ackermann, C. Exploring charge transport in high temperature polymorphism of ITIC derivatives in simple processed unipolar bottom contact organic field-effect transistor. Adv. Electon. Mater. 2022, 8, 2100743. [Google Scholar] [CrossRef]
- Koppe, M.; Egelhaaf, H.J.; Dennler, G.; Scharber, M.C.; Brabec, C.J.; Schilinsky, P.; Hoth, C.N. Near IR sensitization of organic bulk heterojunction solar cells: Towards optimization of the spectral response of organic solar cells. Adv. Funct. Mater. 2010, 20, 338–346. [Google Scholar] [CrossRef]
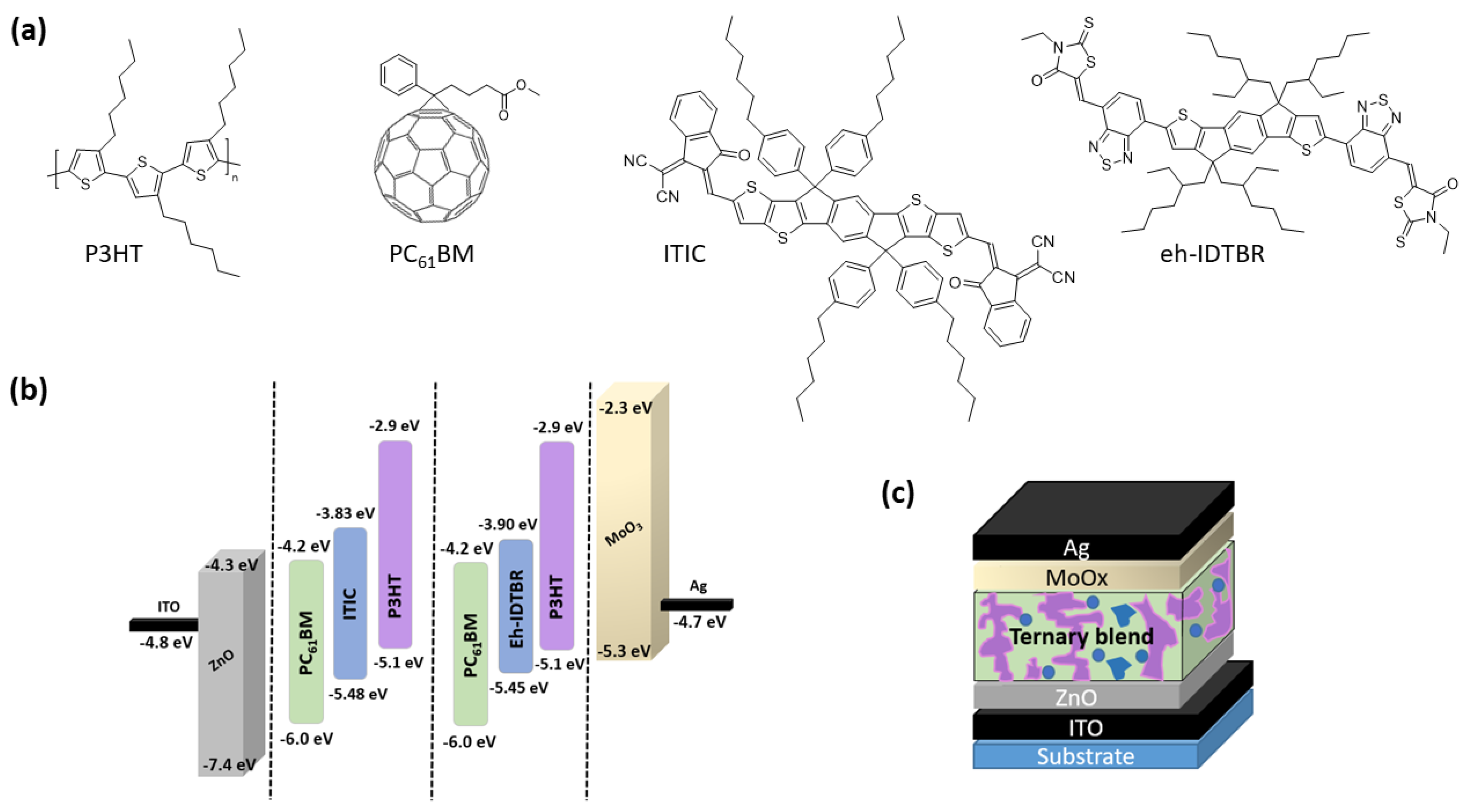

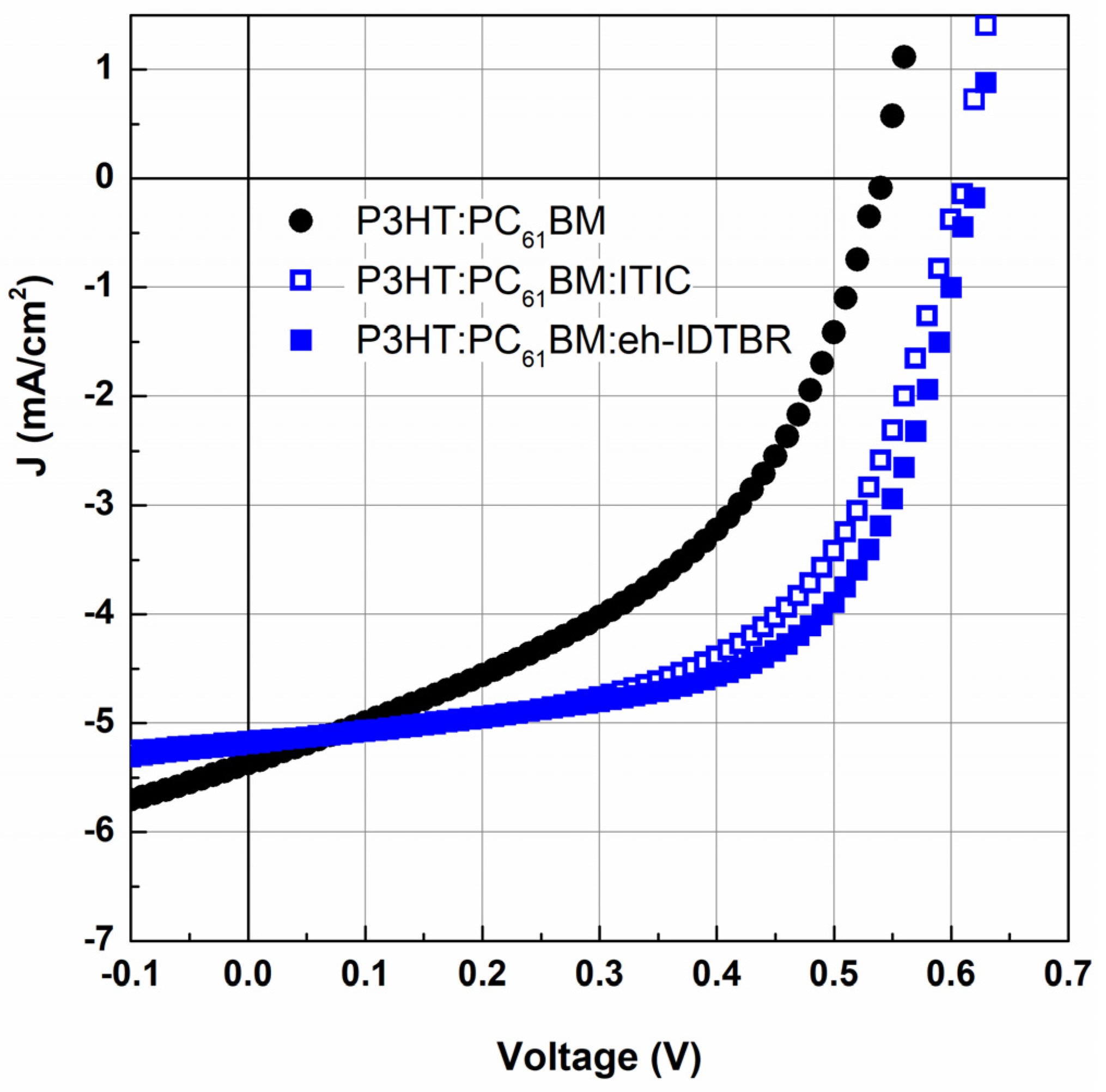
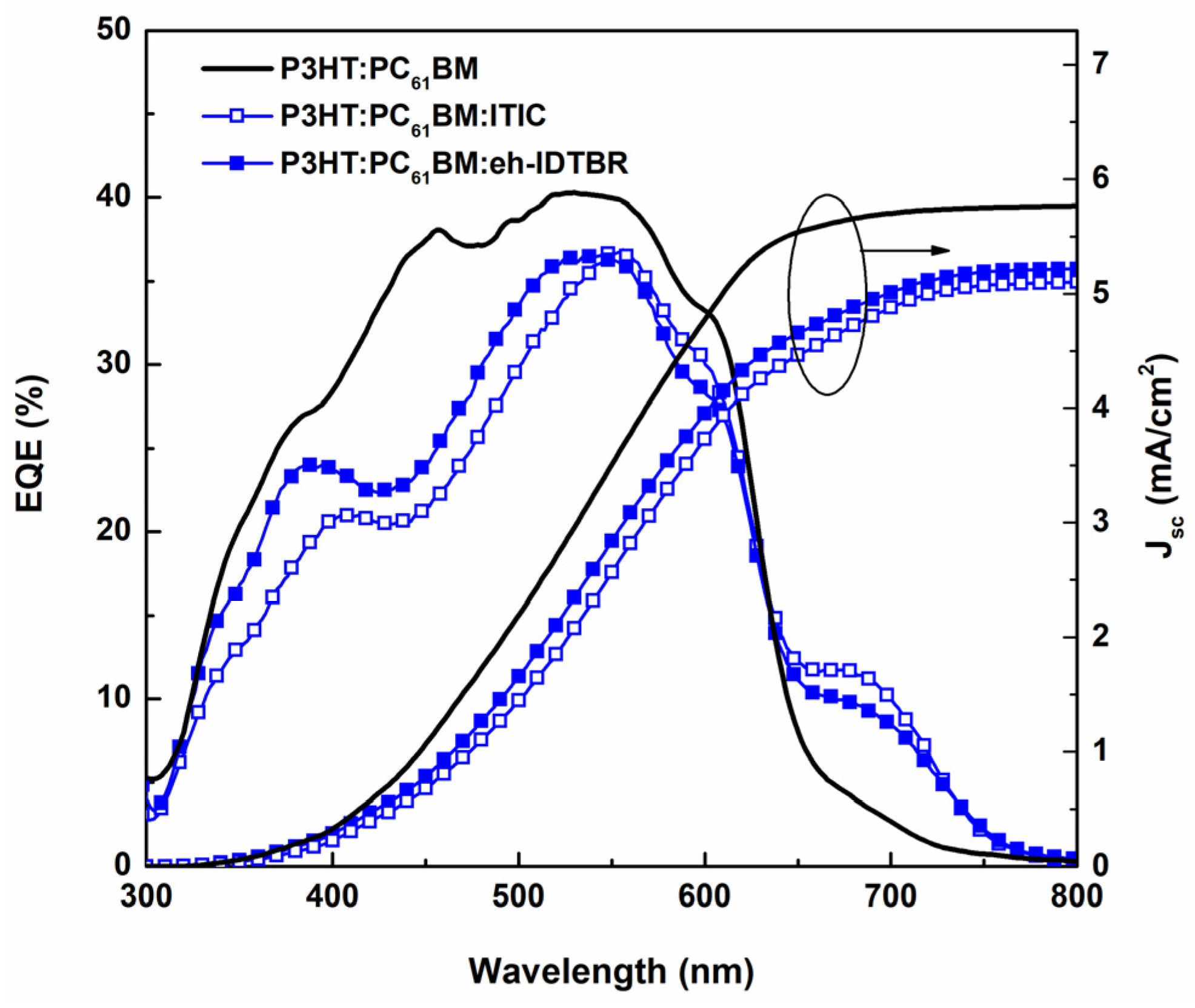


| Layer | Ratio | Temp. (°C) | Voc (V) | Jsc (mA.cm−2) | FF (%) | PCE (%) | Integrated Jsc (mA.cm−2) |
|---|---|---|---|---|---|---|---|
| P3HT:PC61BM | 1:1 | 100 | 0.52 | 5.41 | 42.07 | 1.18 | |
| 140 | 0.54 | 5.37 | 44.83 | 1.30 | 5.77 | ||
| P3HT:PC61BM:ITIC | 0.85:1:0.15 | 100 | 0.62 | 3.55 | 43.05 | 0.95 | |
| 140 | 0.61 | 5.17 | 57.55 | 1.82 | 5.11 | ||
| P3HT:PC61BM:eh-IDTBR | 0.85:1:0.15 | 100 | 0.60 | 3.6 | 54.72 | 1.18 | |
| 140 | 0.62 | 5.19 | 61.27 | 1.97 | 5.22 |
| Layer | Ratio | T (°C) | rms (nm) | WCA (°) |
|---|---|---|---|---|
| P3HT | 100 | 3.35 | 101.2 | |
| 140 | 3.30 | 103.0 | ||
| PC61BM | 100 | 0.34 | 77.7 | |
| 140 | 0.43 | 74.4 | ||
| ITIC | 100 | 0.47 | 94.4 | |
| 140 | 0.86 | 92.2 | ||
| eh-IDTBR | 100 | 0.40 | 90.4 | |
| 140 | 1.16 | 49.5 | ||
| P3HT:PC61BM | 1:1 | 100 | 0.76 | 92.7 |
| 140 | 0.87 | 92.0 | ||
| P3HT:PC61BM:ITIC | 0.85:1:0.15 | 100 | 1.31 | 90.9 |
| 140 | 1.34 | 92.9 | ||
| P3HT:PC61BM:eh-IDTBR | 0.85:1:0.15 | 100 | 0.99 | 103.5 |
| 140 | 1.13 | 103.2 |
| Layer | Ratio | T (°C) | qz (Å−1) | d (nm) | CCL (nm) |
|---|---|---|---|---|---|
| P3HT | 100 | 0.39 | 1.61 | 2.24 | |
| 140 | 0.39 | 2.07 | |||
| P3HT:PC61BM | 1:1 | 100 | 0.39 | 1.61 | 2.27 |
| 140 | 0.39 | 1.96 | |||
| P3HT:PC61BM:ITIC | 0.85:1:0.15 | 100 | 0.38 | 1.62 | 2.88 |
| 140 | 0.38 | 2.87 | |||
| P3HT:PC61BM:eh-IDTBR | 0.85:1:0.15 | 100 | 0.38 | 1.62 | 3.01 |
| 140 | 0.38 | 3.27 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Eynaud, Q.; Koganezawa, T.; Sekimoto, H.; Kramdi, M.e.A.; Quéléver, G.; Margeat, O.; Ackermann, J.; Yoshimoto, N.; Videlot-Ackermann, C. Ternary Polymer Solar Cells: Impact of Non-Fullerene Acceptors on Optical and Morphological Properties. Electronics 2024, 13, 1752. https://doi.org/10.3390/electronics13091752
Eynaud Q, Koganezawa T, Sekimoto H, Kramdi MeA, Quéléver G, Margeat O, Ackermann J, Yoshimoto N, Videlot-Ackermann C. Ternary Polymer Solar Cells: Impact of Non-Fullerene Acceptors on Optical and Morphological Properties. Electronics. 2024; 13(9):1752. https://doi.org/10.3390/electronics13091752
Chicago/Turabian StyleEynaud, Quentin, Tomoyuki Koganezawa, Hidehiro Sekimoto, Mohamed el Amine Kramdi, Gilles Quéléver, Olivier Margeat, Jörg Ackermann, Noriyuki Yoshimoto, and Christine Videlot-Ackermann. 2024. "Ternary Polymer Solar Cells: Impact of Non-Fullerene Acceptors on Optical and Morphological Properties" Electronics 13, no. 9: 1752. https://doi.org/10.3390/electronics13091752
APA StyleEynaud, Q., Koganezawa, T., Sekimoto, H., Kramdi, M. e. A., Quéléver, G., Margeat, O., Ackermann, J., Yoshimoto, N., & Videlot-Ackermann, C. (2024). Ternary Polymer Solar Cells: Impact of Non-Fullerene Acceptors on Optical and Morphological Properties. Electronics, 13(9), 1752. https://doi.org/10.3390/electronics13091752






