1. Introduction
COVID-19 (SARS-CoV-2) is a devastating disease that has spread around the world since the last quarter of 2019. Although the exact origin of the virus is still unknown, the first officially recognized outbreak was reported in Wuhan, China in November 2019. This disease is a deadly pandemic that is transmitted from an animal (host) to another one (intermediate host) or from an intermediate host to humans [
1]. Human-to-human transmission occurs mainly through respiratory droplets and aerosolization when a person breathes in the same enclosed space or in close proximity to other people. Transmission increases in poorly ventilated indoors and when the infected person coughs, sneezes, talks, or sings [
2,
3]. A sudden loss of smell (anosmia), whether or not associated with a loss of taste (ageusia), is a relatively frequent manifestation and the revealing origin of SARS-CoV-2 infection. Other common symptoms may be fever, cough, the difficulty of breathing, chills, muscle aches or sore throat. Symptoms may occur from the second day after the contamination to the 14th one. The novel coronavirus might cause a moderate or severe form of infection. The severe form involves complications such as pneumonia or death of the infected person. A specific category known as “highly comorbid individuals” is considered as the class of individuals with high risk of having the severe form of COVID-19. This category concerns people of all ages with underlying health problems, especially if those problems are poorly controlled, including people with chronic lung disease [
4] or moderate to severe asthma, heart disease, weakened immune system, severe obesity (body mass index of 40 or more), diabetes, chronic kidney disease, especially with dialysis, and liver disease.
The spread of COVID-19 is a very complex phenomenon that draws the attention of many researchers. To overcome the pandemic of COVID-19, it is advisable to study and model it as a complex system that requires a solution in the form of modeling simulation for its understanding. In the literature, complex systems have been mainly studied by means of equation-based models (EBM) and agent-based models (ABM).
The first point of view (EBM) is based on mathematical modeling. In this direction, ref [
5] presented a general model of epidemic spread to understand the timing of transmission. In [
6], the authors studied, with the aid of mathematical models, the infection force of the hepatitis C virus among drug users in France. In [
7], a mathematical model of the COVID-19 outbreak with three forms of infections (benign, respiratory, and reanimation forms) is proposed. In [
8], complex systems, such as the spread of tuberculosis, are solved by means of hybrid stochastic modeling and computer simulations. In their study, ref [
9] established a model of differential equations with piecewise constant arguments in order to explore the spread of COVID-19. A formulation of a stochastic susceptible-infected-recovered model and the determination of sufficient conditions for extinction and persistence of COVID-19 are carried out in [
10].
The second point of view (ABM) is based on artificial intelligence. Many researchers have dealt with such problems using ABM. For example, in [
11] an agent-based simulation is proposed to understand the tuberculosis timing of transmission. In [
12], the authors proposed a study wherein the spread of tuberculosis is controlled by means of a stochastic agent-based model and simulation.
Obviously, both EBM and ABM have strengths and weaknesses, and it would be favorable to try to choose one approach over the other. In this paper, we compare the above-mentioned modeling approaches and apply them to the dynamics of the COVID-19 pandemic. Based on the results of our experiments, we explain the reasons for choosing one approach over another in terms of computing time and memory requirements.
This paper is structured as follows: First, we introduce some key concepts (see
Section 2), then we describe the differences between ABM and EBM (see
Section 3). Third, we apply these two approaches in case studies and discuss the obtained results with numerical simulations (see
Section 4,
Section 5 and
Section 6), and finally, we conclude the study (see
Section 7).
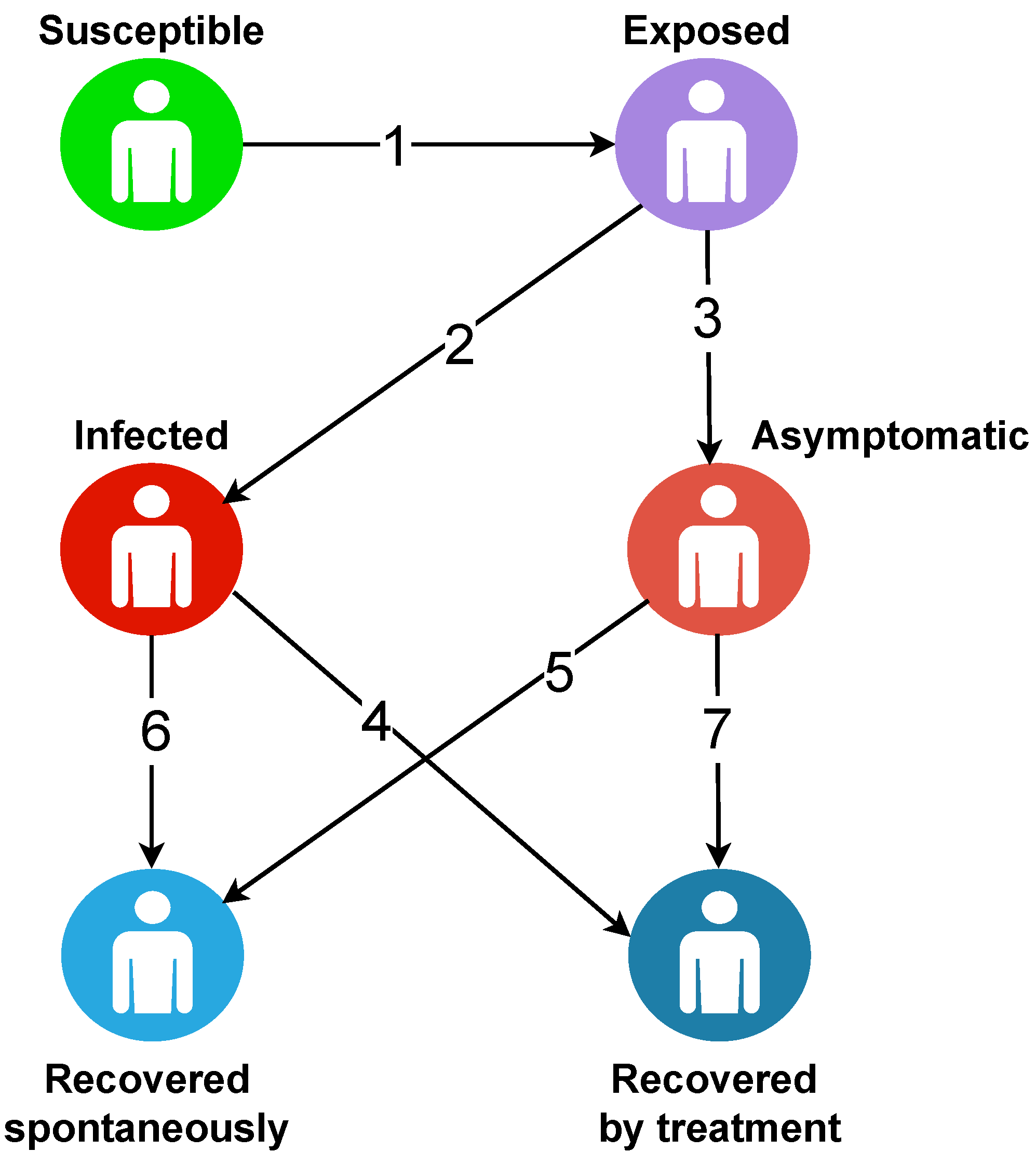
5. Case Study 2: Agent-Based Modeling (ABM) for the Spread of COVID-19
For this case study, we consider autonomous agents that represent the whole population with different statuses.
S: Susceptible individuals,
E: Latent or Exposed individuals,
I: Infected individuals,
A: Asymptomatic individuals,
: Recovered individuals, and
: Recovered spontaneously individuals.
Figure 2 presents the transfer diagram of the multi-agent model.
With:
Exposition.
Visible infection.
Hidden infection.
Recovered of visible infection by treatment.
Recovered of hidden infection without treatment.
Recovered of visible infection without treatment.
Recovered of hidden infection by treatment.
There is a strong case for addressing the different links that may exist between different agents. In the ABM, agents are not static either. They can undergo changes of state by contact or by the impulse of certain parameters. Further, this is why in this model, we have provided particular algorithms for each agent guiding the passage from one state to another.
Description of the Model Using ODD
To avoid any problems with the model results, we used the ODD (The Overview, Design concepts, and Details) protocol as a consistent, logical, and readable account of the ABM structures and dynamics [
22].
Overview
Objective: the interest of the model was to predict over a long period of time the incidental effects (evolution, treatment, management, brief scientific data) of the COVID-19 pandemic, and to understand the impact of the contact links between individuals in a precise contamination radius (environmental configuration) of the population.
Entities, state variables, and scales:
- -
Agent or Individual: in the model we consider only one type of agent. Each agent has state variables and characteristics.
- -
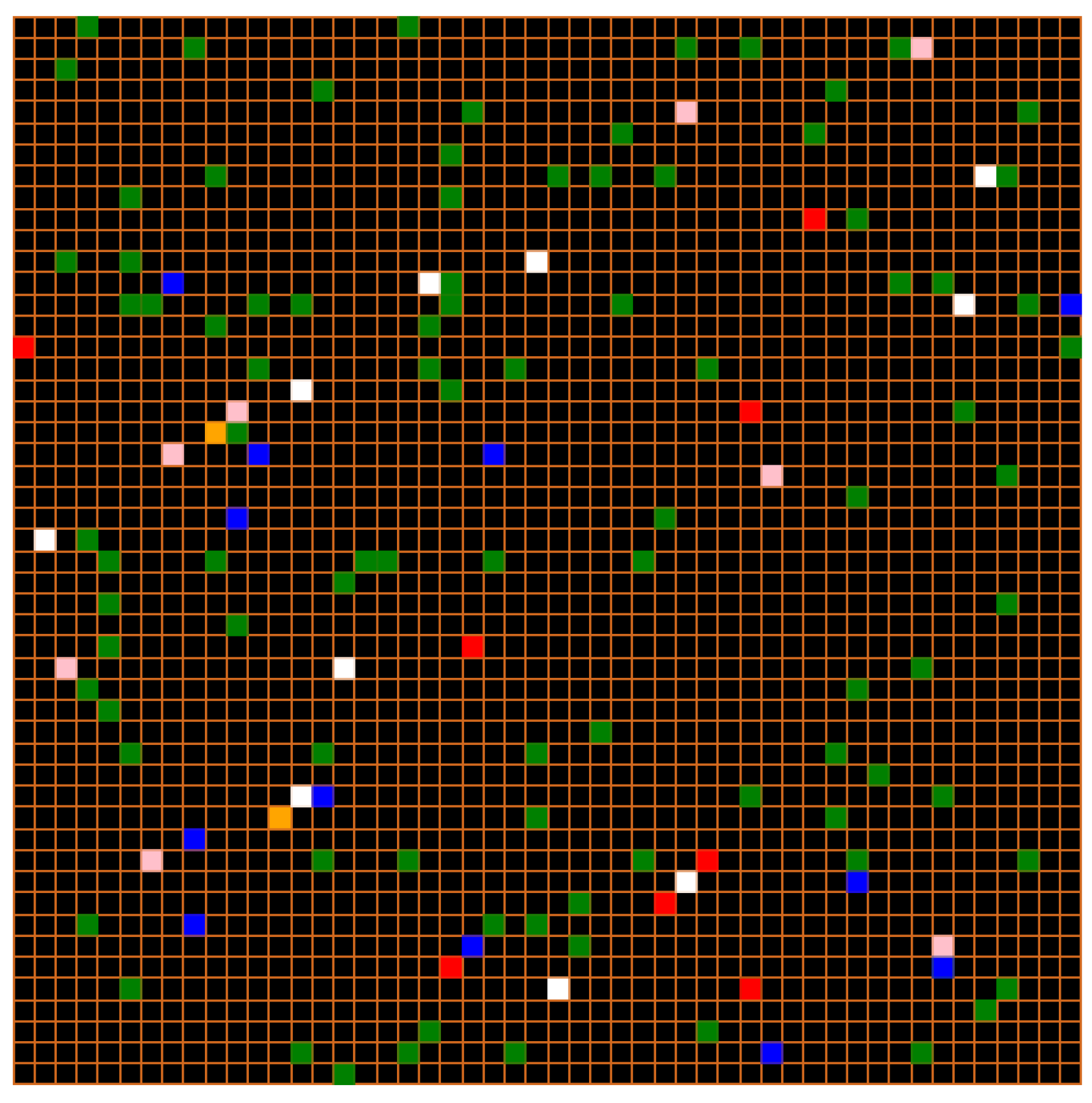
Environment: For a better-mixed population structural representation, we started from an environment in the form of a 50 × 50 mesh. An environment in which agents move randomly from one cell to another.
Figure 3 shows this environment:
Process overview and scheduling: The model presents a process in discrete time steps.
At the beginning of the simulation, all individual agents in the model are in any state (susceptible, exposed, infectious, asymptomatic, recovered, or recovered spontaneously). Each agent can move from one state to another according to a probability.
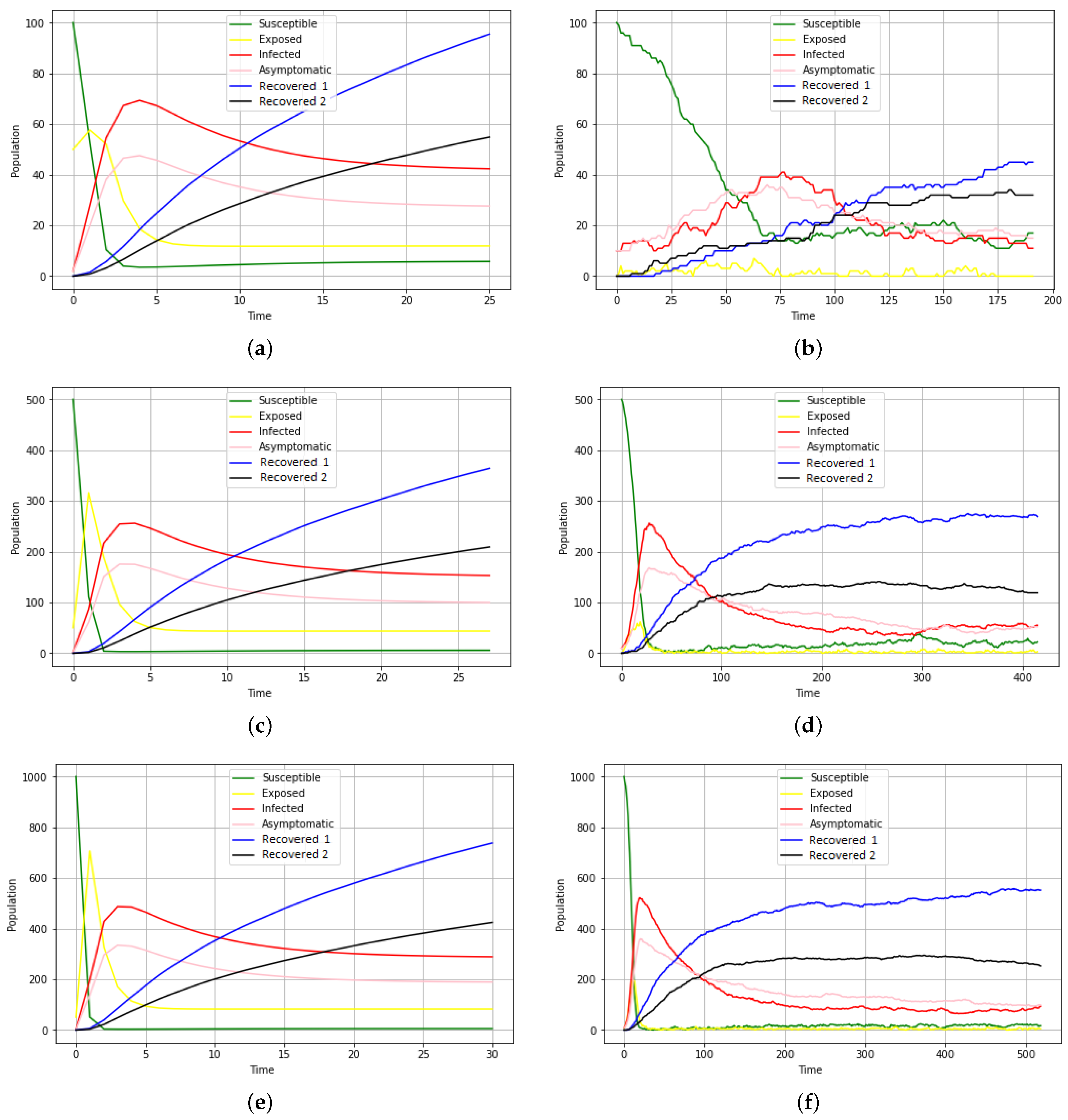
Figure 4 displays the output of the model simulation at a given time
t.
Design Concepts
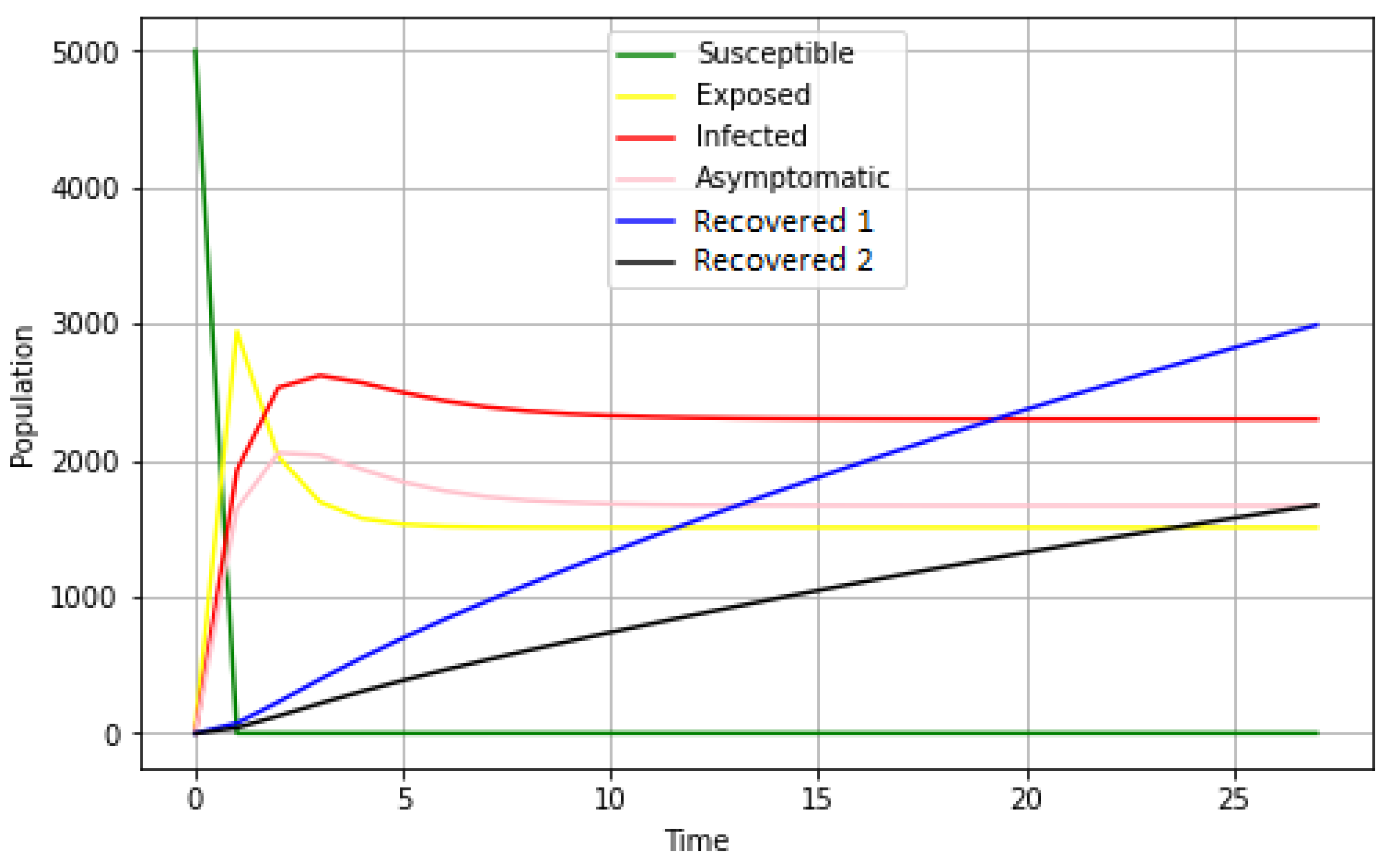
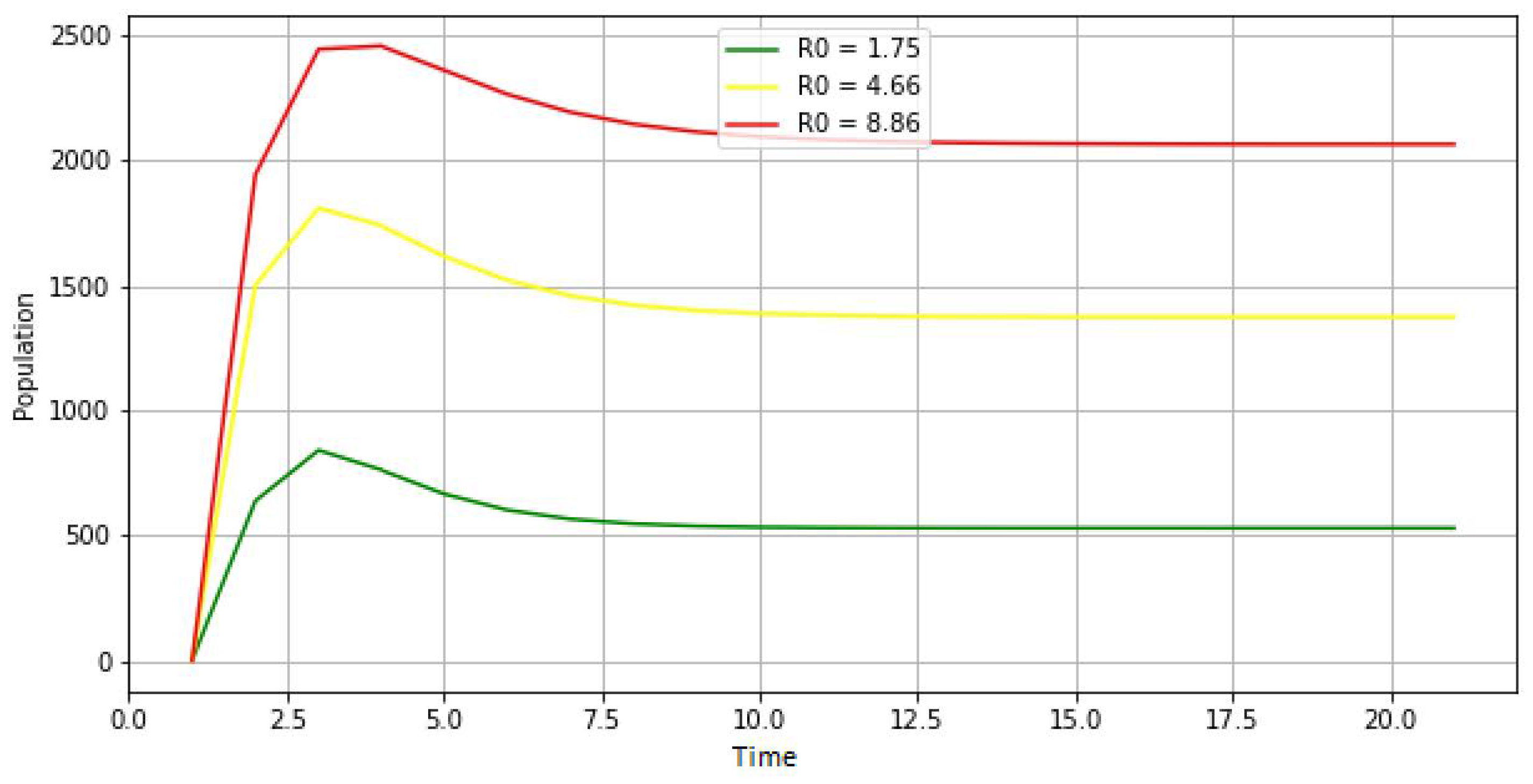
Basic principle: it is a propagation of the disease following a compartmental model containing six groups of individuals (susceptible, exposed, infected, asymptomatic, spontaneously recovered, and recovered with treatment).
The idea here is that, initially, a susceptible individual comes into contact with an infectious individual. This contact gives the agent the opportunity to become either exposed or directly infected or asymptomatic. As a result, a newly exposed individual could also become either infected or asymptomatic. In the process, the infected or asymptomatic individual may either recover or succumb to the disease. There is also a prediction of natural death, which is an irrefutable factor.
Emerging: The emergence of the system is justified in the evolution of COVID-19 infection in the population. In fact, this emergence actually depends on several evolutionary factors that come into play: the type of agents that are initially infectious, other agents that come into contact with them along a contamination radius, the duration of contact but also the frequency of contact. However, there are some factors that surely have an impact on the readjustment of data.
Adaptation: Algorithms are designed to make agents able to reproduce different behaviors they observe in the environment. For example, in the mixed population structure, if an agent is already recovered, they will adapt their behavior by avoiding contact with infectious agents.
Detection: As infectious agents move through the environment, they can detect susceptible agents and recovered ones. Once recovered agents detect infectious agents, they avoid contact with them.
Interaction: In this model, we assume that agents in the same radius defined in the code interact with each other and with their environment. For example, if a patient agent is in the same radius as an infectious agent, it is possible for the patient agent to be contaminated.
Stochasticity: In a model, agents’ movements are random. With their specific states, agents’ movements are stochastic. Likewise, the choice of destinations for all agents is random. Stochasticity is also observed in COVID-19 contamination. If a susceptible agent comes into contact with an infectious agent, there is a certain likelihood that determines whether they are exposed, asymptomatic, or directly infectious. Additionally, the length of time an agent remains in a state (susceptible, exposed, infectious, asymptomatic, spontaneously recovered, or recovered after treatment) is chosen at random. So, in short, we are talking about a random pathway.
Observation: Data are collected at each model run on individual agents according to their states (susceptible, exposed, infectious, asymptomatic, recovered with treatment, or spontaneously).
Details
Initialization: For the mixed population structure, agents are randomly placed in cells. At the start of the simulation, the environment contains only a given number of susceptible agents and a few infectious agents (chosen by the user).
Sub-models:
- -
Timer: this sub-model manages the time in the system.
- -
Death: death is considered here under both aspects, either natural or by disease also containing a drug failure. Infectious agents lose their lives with a certain probability.
- -
Update of global variables: all global variables are updated at the end of each time step. At the same time, the number and percentage of susceptible, exposed, infected, asymptomatic, and recovered (spontaneously and after treatment) agents are all calculated.
7. Concluding Remarks
In this paper, we have noted a major problem shaking our society: “the context of precarious life impacted by the COVID-19 pandemic”. To address this problem, we explored two approaches: equation-based modeling and agent-based modeling on the spread of diseases, especially during the COVID-19 pandemic. Then, we proposed a compartmental model of SEIAR-type describing the transfer diagram of COVID-19 dynamics. Based on the results obtained, it seems, on the one hand, that EBM is synthetic, formalized, homogeneous, and faster. Moreover, modeling with equations tends to describe reality at the macroscopic level. The model is far from the biological reality of the studied phenomenon. On the other hand, ABM is modular, incremental, heterogeneous, and close to the biological reality of the studied phenomenon since the description is made at the individual level. A few drawbacks were raised with ABM: the model is not formalized and requires many parameters, high runtime, and high memory capacity, as shown in
Table 5.
Both approaches can be used to model the problem studied in this paper. However, it would be advisable to combine them to understand the reality, since none of them seem to outrank the other on the desirable criteria for a modeling approach.