COVID-19 Disease Leading to Chronic Spontaneous Urticaria Exacerbation: A Romanian Retrospective Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Patients and Clinical Evaluation
2.3. COVID-19 Disease
2.4. Biological Evaluation
2.5. Statistical Analysis
3. Results
3.1. Patients’ Demographic Data
3.2. The Treatment in Our Group with CSU
3.3. CSU and COVID-19 Disease
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Zuberbier, T.; Aberer, W.; Asero, R.; Bindslev-Jensen, C.; Brzoza, Z.; Canonica, G.W.; Church, M.K.; Ensina, L.F.; Giménez-Arnau, A.; Godse, K.; et al. The EAACI/GA2LEN/EDF/WAO guideline for the definition, classification, diagnosis and management of urticaria. Allergy 2018, 73, 1393–1414. [Google Scholar] [CrossRef]
- Deleanu, D.; Nedelea, I.; Petricau, C.; Leru, P.; Dumitrascu, D.; Muntean, A. Clinical impact of omalizumab in refractory chronic urticaria: One centre experience. Exp. Ther. Med. 2019, 18, 5078–5081. [Google Scholar] [CrossRef] [Green Version]
- Guillén-Aguinaga, S.; Jáuregui Presa, I.; Aguinaga-Ontoso, E.; Guillén-Grima, F.; Ferrer, M. Updosing nonsedating antihista-mines in patients with chronic spontaneous urticaria: A systematic review and meta-analysis. Br. J. Dermatol. 2016, 175, 1153–1165. [Google Scholar] [CrossRef] [PubMed]
- Theoharides, T.C. Potential association of mast cells with coronavirus disease 2019. Ann. Allergy Asthma Immunol. 2020, 126, 217–218. [Google Scholar] [CrossRef]
- Marshall, J.S.; Portales-Cervantes, L.; Leong, E. Mast Cell Responses to Viruses and Pathogen Products. Int. J. Mol. Sci. 2019, 20, 4241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Criado, P.R.; Pagliari, C.; Criado, R.F.J.; Marques, G.F.; Belda, W., Jr. What the physicians should know about mast cells, dendritic cells, urticaria, and omalizumab during COVID-19 or asymptomatic infections due to SARS-CoV-2? Dermatol. Ther. 2020, 33, e14068. [Google Scholar] [CrossRef]
- Kempuraj, D.; Selvakumar, G.P.; Ahmed, M.E.; Raikwar, S.P.; Thangavel, R.; Khan, A.; Zaheer, S.A.; Iyer, S.S.; Burton, C.; James, D.; et al. COVID-19, Mast Cells, Cytokine Storm, Psychological Stress, and Neuroinflammation. Neuroscientist 2020, 26, 402–414. [Google Scholar] [CrossRef]
- Kritas, S.K.; Ronconi, G.; Caraffa, A.; Gallenga, C.E.; Ross, R.; Conti, P. Mast cells contribute to coronavirus-induced inflammation: New anti-inflammatory strategy. J. Biol. Regul. Homeost. Agents 2020, 34, 9–14. [Google Scholar]
- Theoharides, T.C. Covid-19, pulmonary mast cells, cytokine storms, and beneficial actions of luteolin. Biofactors 2020, 46, 306–308. [Google Scholar] [CrossRef] [PubMed]
- Kocatürk, E.; Salman, A.; Cherrez-Ojeda, I.; Criado, P.R.; Peter, J.; Comert-Ozer, E.; Abuzakouk, M.; Agondi, R.C.; Al-Ahmad, M.; Al-trichter, S.; et al. The global impact of the COVID-19 pandemic on the management and course of chronic urticaria. Allergy 2021, 76, 816–830. [Google Scholar] [CrossRef]
- Paquet, C.; Babes, V.T.; Drucker, J.; Sénémaud, B.; Dobrescu, A. Viral hepatitis in Bucharest. Bull. World Health Organ. 1993, 71, 781–786. [Google Scholar] [PubMed]
- Azkur, A.K.; Akdis, M.; Azkur, D.; Sokolowska, M.; van de Veen, W.; Bruggen, M.C. Immune response to SARS-CoV-2 and mecha-nisms of immunopathological changes in COVID-19. Allergy 2020, 75, 1564–1581. [Google Scholar] [CrossRef]
- Afrin, L.B.; Weinstock, L.B.; Molderings, G.J. Covid-19 hyperinflammation and post-Covid-19 illness may be rooted in mast cell activation syndrome. Int. J. Infect. Dis. 2020, 100, 327–332. [Google Scholar] [CrossRef] [PubMed]
- Hermans, M.A.W.; Van Lennep, J.E.R.; Van Daele, P.L.A.; Bot, I. Mast Cells in Cardiovascular Disease: From Bench to Bedside. Int. J. Mol. Sci. 2019, 20, 3395. [Google Scholar] [CrossRef] [Green Version]
- Feldstein, L.R.; Rose, E.B.; Horwitz, S.M. Multisystem inflammatory syndrome in U.S. children and adolescents. N. Engl. J. Med. 2020, 383, 334–346. [Google Scholar] [CrossRef] [PubMed]
- Akin, C.; Valent, P.; Metcalfe, D.D. Faculty Opinions recommendation of Mast cell activation syndrome: Proposed diagnostic criteria. J. Allergy Clin. Immunol. 2010, 126, 1099–1104. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maurer, M.; Staubach, P.; Raap, U.; Richter-Huhn, G.; Bauer, A.; Ruëff, F.; Jakob, T.; Yazdi, A.S.; Mahler, V.; Wagner, N.; et al. H1-antihistamine-refractory chronic spontaneous ur-ticaria: It’s worse than we thought—First results of the multicenter real-life AWARE study. Clin. Exp. Allergy 2017, 47, 684–692. [Google Scholar] [CrossRef]
- Ayhan, E.; Öztürk, M.; An, I.; Bekçibaşi, M. COVID-19 infection under omalizumab therapy for chronic spontaneous urticaria: Three cases. Int. J. Dermatol. 2020, 60, 253–254. [Google Scholar] [CrossRef]
- Msc, A.A.; Goldust, M.; Vestita, M. Comment on “Chronic spontaneous urticaria exacerbation in a patient with COVID-19, rapid and excellent response to omalizumab”. Int. J. Dermatol. 2020, 59, 1417–1418. [Google Scholar] [CrossRef]
- Criado, P.R.; Criado, R.F.J.; Pincelli, T.P.; Yoshimoto, T.A.; Naufal, G.G.A.; Abdalla, B.M.Z. Chronic spontaneous urticaria exacerbation in a patient with COVID-19, rapid and excellent response to omalizumab. Int. J. Dermatol. 2020, 59, 1294–1295. [Google Scholar] [CrossRef]
- Muntean, I.A.; Bocsan, I.C.; Miron, N.; Buzoianu, A.D.; Deleanu, D. How Could We Influence Systemic Inflammation in Allergic Rhinitis? The Role of H1 Antihistamines. Oxidative Med. Cell. Longev. 2018, 2018, 1–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chilamakuri, R.; Agarwal, S. COVID-19, Characteristics and Therapeutics. Cells 2021, 10, 206. [Google Scholar] [CrossRef] [PubMed]
- Bumbăcea, R.; Deaconu, C.; Berghea, E.C. Management problems in severe chronic inducible urticaria: Two case reports. Exp. Ther. Med. 2019, 18, 960–963. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Passante, M.; Napolitano, M.; Dastoli, S.; Bennardo, L.; Fabbrocini, G.; Nisticò, S.P.; Patruno, C. Safety of Omalizumab Treatment in Patients with Chronic Spontaneous Urticaria and Covid-19. Dermatol. Ther. 2021, in press. [Google Scholar] [CrossRef]
- Bousquet, J.; Bedbrook, A.; Czarlewski, W.; Onorato, G.L.; Arnavielhe, S.; Laune, D.; Mathieu-Dupas, E.; Fonseca, J.; Costa, E.; Lourenço, O.; et al. Guidance to 2018 good practice: ARIA digitally-enabled, integrated, person-centred care for rhinitis and asthma. Clin. Transl. Allergy 2019, 9, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Erdem, Y.; Ekinci, A.P.; Altunay, I.K.; Sivaz, O.; Inal, S.; Gokalp, M.O.; Pehlivan, G.; Özkaya, E. The impact of COVID-19 pandemic on the management of patients with chronic urticaria: An observational two-center study from Turkey. Dermatol. Ther. 2020, 34, e14652. [Google Scholar] [CrossRef]
- Deza, G.; Ricketti, P.A.; Giménez-Arnau, A.M.; Casale, T.B. Emerging Biomarkers and Therapeutic Pipelines for Chronic Sponta-neous Urticaria. J. Allergy Clin. Immunol. Pract. 2018, 6, 1108–1117. [Google Scholar] [CrossRef]
| Parameter | Mild CSU (n = 69) | Moderate/Severe CSU (n = 149) | p | |
|---|---|---|---|---|
| Age * | 38.05 ± 6.2 | 59.89 ± 9.7 | 0.01 | |
| Sex | male | 34.7% (24) | 20.1% (30) | 0.2 |
| female | 65.3% (65) | 79.9% (99) | ||
| Living area | urban | 78.2% (54) | 68.4% (102) | 0.7 |
| rural | 21.8% (15) | 31.6% (27) | ||
| CSU onset (months) | 124 (3–160) | 186 (4.5–268) | 0.5 | |
| CSU scores | UAS7 | ≤15 | ≥16 | 0.01 |
| VAS | ≤6 | ≥6 | 0.01 | |
| Treatment | AH1 | 10% (7) | 2.6% (4) | 0.05 |
| 2xAH1 | 37.6% (26) | 5.9% (10) | 0.8 | |
| 4xAH1 | 37.6% (26) | 36.2% (54) | 0.05 | |
| * Omalizumab | 0% (0) | 16.7% (25) | 0.01 | |
| * Cyclosporine | 0% (0) | 2% (3) | 0.01 | |
| * Prednisone | 0% (0) | 22.1% (33) | 0.01 | |
| CSU in remission | No treatment | 21.8% (15) | 16.7% (25) | 0.6 |
| COVID 19 infection | Not present | 69.5% (48) | 65.7% (98) | 0.7 |
| Present | 31.5% (21) | 34.3% (51) | 0.8 | |
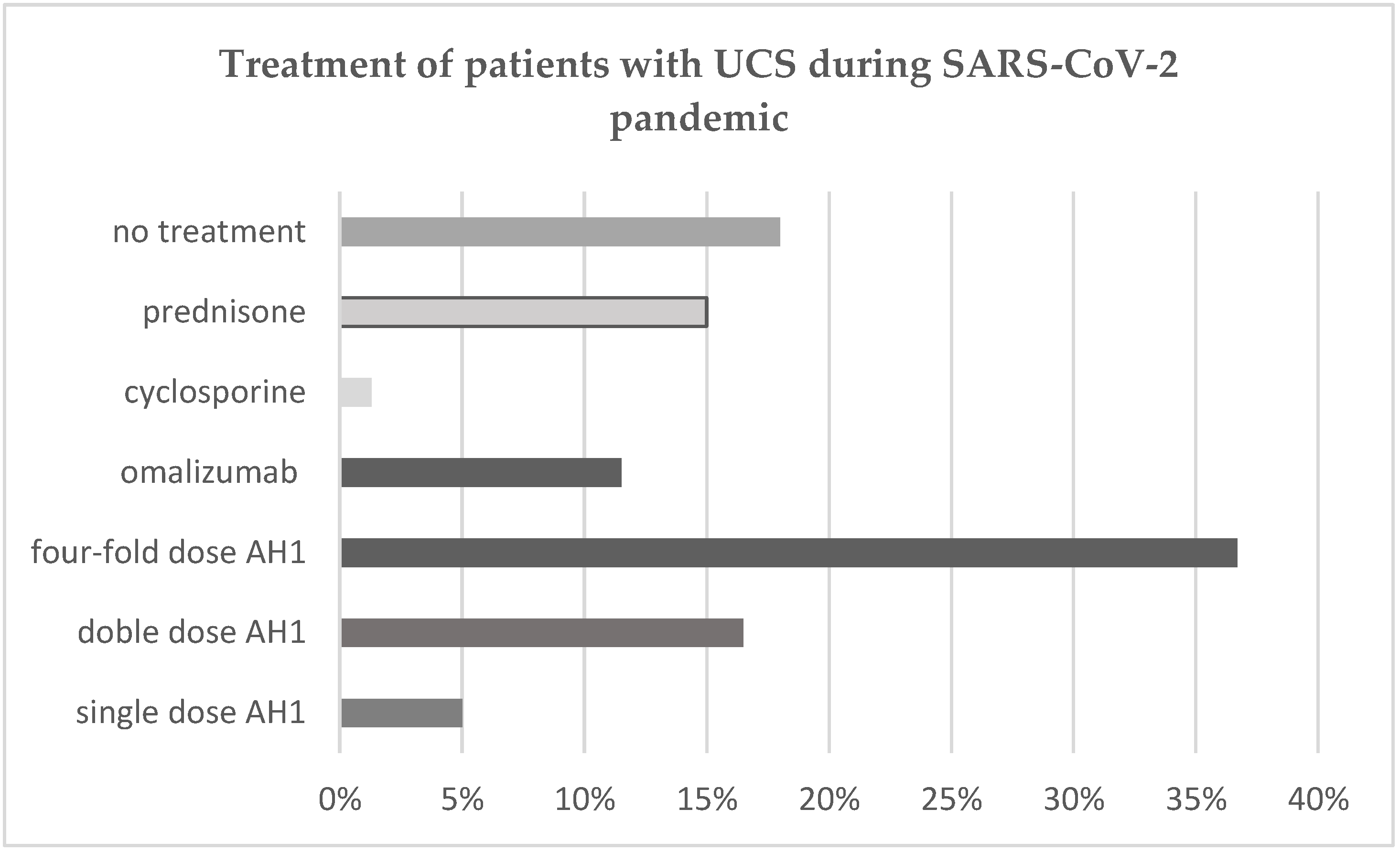
| CSU Treatment | Mild COVID-19 Infection (n = 21) | Moderate/Severe COVID-19 Infection (n = 51) |
|---|---|---|
| Single dose AH1 | 14.2% (3) | 1.9% (1) |
| Double dose AH1 | 42.8% (9) | 29.4% (15) |
| Four-fold dose AH1 | 33.3% (7) | 39.1% (20) |
| Omalizumab | 0% (0) | 19.6% (10) |
| Cyclosporine | 0% (0) | 0% (0) |
| Prednisone | 0% (0) | 3.9% (2) |
| No treatment | 9.7% (2) | 9.1% (5) |
| CSU Course | Mild COVID-19 Infection (n = 21) | Moderate/Severe COVID-19 Infection (n = 51) |
|---|---|---|
| Better | 4.7% (1) | 9.8% (5) |
| No change | 61.9% (13) | 43.1% (22) |
| Worse | 33.4% (7) | 47% (24) |
| Parameter | Mild COVID-19 Infection | Moderate/Severe COVID-19 Infection | p |
|---|---|---|---|
| Leucocytes * 103/μL | 7.54 (±9.7) | 3.89 (±2.9) | 0.03 |
| CRP * (mg/dL) | 2.9 (±6.1) | 49.8 (±52.5) | 0.01 |
| Troponin * (μg/L) | 0.1 (±0.2) | 2.3 (±1.8) | 0.02 |
| LDH * (U/L) | 438 (±195.6) | 657 (±185.8) | 0.05 |
| UAS7 | 23 (±5.2) | 38 (±3.8) | 0.03 |
| VAS | 7 (±1.4) | 9 (±0.8) | 0.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Muntean, I.A.; Pintea, I.; Bocsan, I.C.; Dobrican, C.T.; Deleanu, D. COVID-19 Disease Leading to Chronic Spontaneous Urticaria Exacerbation: A Romanian Retrospective Study. Healthcare 2021, 9, 1144. https://doi.org/10.3390/healthcare9091144
Muntean IA, Pintea I, Bocsan IC, Dobrican CT, Deleanu D. COVID-19 Disease Leading to Chronic Spontaneous Urticaria Exacerbation: A Romanian Retrospective Study. Healthcare. 2021; 9(9):1144. https://doi.org/10.3390/healthcare9091144
Chicago/Turabian StyleMuntean, Ioana Adriana, Irena Pintea, Ioana Corina Bocsan, Carmen Teodora Dobrican, and Diana Deleanu. 2021. "COVID-19 Disease Leading to Chronic Spontaneous Urticaria Exacerbation: A Romanian Retrospective Study" Healthcare 9, no. 9: 1144. https://doi.org/10.3390/healthcare9091144
APA StyleMuntean, I. A., Pintea, I., Bocsan, I. C., Dobrican, C. T., & Deleanu, D. (2021). COVID-19 Disease Leading to Chronic Spontaneous Urticaria Exacerbation: A Romanian Retrospective Study. Healthcare, 9(9), 1144. https://doi.org/10.3390/healthcare9091144