Pneumatic Cell Stretching Chip to Generate Uniaxial Strain Using an Elastomeric Membrane with Ridge Structure
Abstract
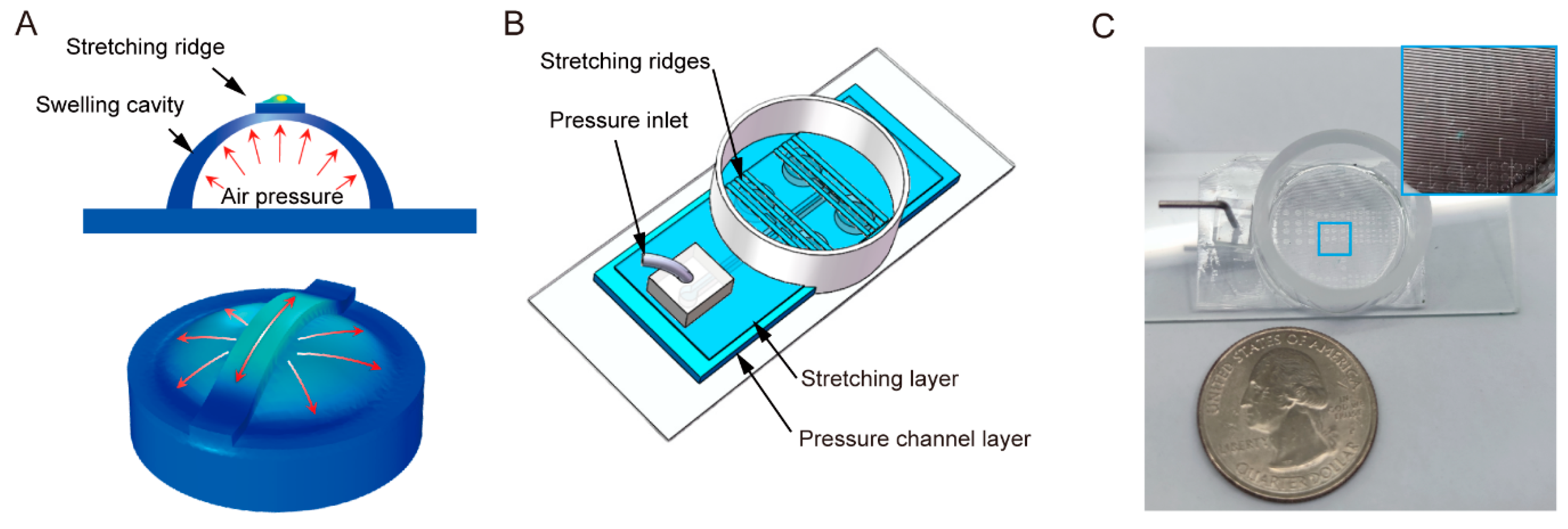
:1. Introduction
2. Materials and Methods
2.1. Fabrication of Cell Stretching Chip
2.2. Characterization of Pressure Control Layer
2.3. FEM Simulation and Experimental Measurement for Strain
2.4. Cell Culture and Stretching
2.5. Cytoskeleton Staining
2.6. Measurement of Cell Orientation Index
2.7. Statistical Analysis
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Kamble, H.; Barton, M.J.; Jun, M.; Park, S.; Nguyen, N.T. Cell stretching devices as research tools: Engineering and biological considerations. Lab Chip 2016, 16, 3193–3203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riehl, B.D.; Park, J.H.; Kwon, I.K.; Lim, J.Y. Mechanical stretching for tissue engineering: Two-dimensional and three-dimensional constructs. Tissue Eng. Part B Rev. 2012, 18, 288–300. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cui, Y.; Hameed, F.M.; Yang, B.; Lee, K.; Pan, C.Q.; Park, S.; Sheetz, M. Cyclic stretching of soft substrates induces spreading and growth. Nat. Commun. 2015, 6, 6333. [Google Scholar] [CrossRef] [PubMed]
- Ghibaudo, M.; Trichet, L.; Le Digabel, J.; Richert, A.; Hersen, P.; Ladoux, B. Substrate topography induces a crossover from 2D to 3D behavior in fibroblast migration. Biophys. J. 2009, 97, 357–368. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Heo, S.J.; Driscoll, T.P.; Thorpe, S.D.; Nerurkar, N.L.; Baker, B.M.; Yang, M.T.; Chen, C.S.; Lee, D.A.; Mauck, R.L. Differentiation alters stem cell nuclear architecture, mechanics, and mechano-sensitivity. eLife 2016, 5, e18207. [Google Scholar] [CrossRef]
- Kong, M.; Lee, J.; Yazdi, I.K.; Miri, A.K.; Lin, Y.D.; Seo, J.; Zhang, Y.S.; Khademhosseini, A.; Shin, S.R. Cardiac Fibrotic Remodeling on a Chip with Dynamic Mechanical Stimulation. Adv. Healthc. Mater. 2019, 8, e1801146. [Google Scholar] [CrossRef]
- Boudou, T.; Andersen, T.; Balland, M. On the spatiotemporal regulation of cell tensional state. Exp. Cell Res. 2019, 378, 113–117. [Google Scholar] [CrossRef] [PubMed]
- Zhang, W.; Huang, G.; Xu, F. Engineering Biomaterials and Approaches for Mechanical Stretching of Cells in Three Dimensions. Front. Bioeng. Biotechnol. 2020, 8, 589590. [Google Scholar] [CrossRef]
- Costa, J.; Ahluwalia, A. Advances and Current Challenges in Intestinal in vitro Model Engineering: A Digest. Front. Bioeng. Biotechnol. 2019, 7, 144. [Google Scholar] [CrossRef] [Green Version]
- Guenat, O.T.; Berthiaume, F. Incorporating mechanical strain in organs-on-a-chip: Lung and skin. Biomicrofluidics 2018, 12, 042207. [Google Scholar] [CrossRef] [Green Version]
- Zheng, W.; Jiang, B.; Wang, D.; Zhang, W.; Wang, Z.; Jiang, X. A microfluidic flow-stretch chip for investigating blood vessel biomechanics. Lab Chip 2012, 12, 3441–3450. [Google Scholar] [CrossRef] [PubMed]
- Sunuwar, L.; Yin, J.; Kasendra, M.; Karalis, K.; Kaper, J.; Fleckenstein, J.; Donowitz, M. Mechanical Stimuli Affect Escherichia coli Heat-Stable Enterotoxin-Cyclic GMP Signaling in a Human Enteroid Intestine-Chip Model. Infect. Immun. 2020, 88, e00866-19. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Wang, Z.; Chu, Q.; Jiang, K.; Li, J.; Tang, N. The Strength of Mechanical Forces Determines the Differentiation of Alveolar Epithelial Cells. Dev. Cell 2018, 44, 297–312. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wu, H.; Yu, Y.; Huang, H.; Hu, Y.; Fu, S.; Wang, Z.; Shi, M.; Zhao, X.; Yuan, J.; Li, J.; et al. Progressive Pulmonary Fibrosis Is Caused by Elevated Mechanical Tension on Alveolar Stem Cells. Cell 2020, 180, 107–121. [Google Scholar] [CrossRef]
- Michielin, F.; Serena, E.; Pavan, P.; Elvassore, N. Microfluidic-assisted cyclic mechanical stimulation affects cellular membrane integrity in a human muscular dystrophy in vitro model. RSC Adv. 2015, 5, 98429–98439. [Google Scholar] [CrossRef]
- Dolle, J.P.; Morrison, B.; Schloss, R.S.; Yarmush, M.L. An organotypic uniaxial strain model using microfluidics. Lab Chip 2013, 13, 432–442. [Google Scholar] [CrossRef] [PubMed]
- Gaio, N.; van Meer, B.; Quiros Solano, W.; Bergers, L.; van de Stolpe, A.; Mummery, C.; Sarro, P.M.; Dekker, R. Cytostretch, an Organ-on-Chip Platform. Micromachines 2016, 7, 120. [Google Scholar] [CrossRef] [Green Version]
- Schurmann, S.; Wagner, S.; Herlitze, S.; Fischer, C.; Gumbrecht, S.; Wirth-Hucking, A.; Prolss, G.; Lautscham, L.A.; Fabry, B.; Goldmann, W.H.; et al. The IsoStretcher: An isotropic cell stretch device to study mechanical biosensor pathways in living cells. Biosens. Bioelectron. 2016, 81, 363–372. [Google Scholar] [CrossRef]
- Seriani, S.; Del Favero, G.; Mahaffey, J.; Marko, D.; Gallina, P.; Long, C.S.; Mestroni, L.; Sbaizero, O. The cell-stretcher: A novel device for the mechanical stimulation of cell populations. Rev. Sci. Instrum. 2016, 87, 084301. [Google Scholar] [CrossRef] [PubMed]
- Kamble, H.; Barton, M.J.; Nguyen, N.-T. Modelling of an uniaxial single-sided magnetically actuated cell-stretching device. Sens. Actuators A Phys. 2016, 252, 174–179. [Google Scholar] [CrossRef] [Green Version]
- Kamble, H.; Vadivelu, R.; Barton, M.; Boriachek, K.; Munaz, A.; Park, S.; Shiddiky, M.J.A.; Nguyen, N.T. An Electromagnetically Actuated Double-Sided Cell-Stretching Device for Mechanobiology Research. Micromachines 2017, 8, 256. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- He, Y.; Mao, T.; Gu, Y.; Yang, Y.; Ding, J. A simplified yet enhanced and versatile microfluidic platform for cyclic cell stretching on an elastic polymer. Biofabrication 2020, 12, 045032. [Google Scholar] [CrossRef] [PubMed]
- Kamble, H.; Vadivelu, R.; Barton, M.; Shiddiky, M.J.A.; Nguyen, N.T. Pneumatically actuated cell-stretching array platform for engineering cell patterns in vitro. Lab Chip 2018, 18, 765–774. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ao, M.; Brewer, B.M.; Yang, L.; Franco Coronel, O.E.; Hayward, S.W.; Webb, D.J.; Li, D. Stretching fibroblasts remodels fibronectin and alters cancer cell migration. Sci. Rep. 2015, 5, 8334. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prevedello, L.; Michielin, F.; Balcon, M.; Savio, E.; Pavan, P.; Elvassore, N. A Novel Microfluidic Platform for Biomechano-Stimulations on a Chip. Ann. Biomed. Eng. 2019, 47, 231–242. [Google Scholar] [CrossRef] [PubMed]
- Walker, M.; Godin, M.; Pelling, A.E. A vacuum-actuated microtissue stretcher for long-term exposure to oscillatory strain within a 3D matrix. Biomed. Microdevices 2018, 20, 43. [Google Scholar] [CrossRef]
- Xue, X.; Sun, Y.; Resto-Irizarry, A.M.; Yuan, Y.; Aw Yong, K.M.; Zheng, Y.; Weng, S.; Shao, Y.; Chai, Y.; Studer, L.; et al. Mechanics-guided embryonic patterning of neuroectoderm tissue from human pluripotent stem cells. Nat. Mater. 2018, 17, 633–641. [Google Scholar] [CrossRef]
- Chen, K.; Rong, N.; Wang, S.; Luo, C. Corrigendum to: A novel two-layer-integrated microfluidic device for high-throughput yeast proteomic dynamics analysis at the single-cell level. Integr. Biol. 2021, 13, 258. [Google Scholar] [CrossRef]
- Li, X.; Shi, J.; Gao, Z.; Xu, J.; Wang, S.; Li, X.; Ouyang, Q.; Luo, C. Biophysical studies of cancer cells’ traverse-vessel behaviors under different pressures revealed cells’ motion state transition. Sci. Rep. 2022, 12, 7392. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Shen, W.; Cai, Z.; Chen, K.; Ouyang, Q.; Wei, P.; Yang, W.; Luo, C. Microfluidic-Enabled Multi-Cell-Densities-Patterning and Culture Device for Characterization of Yeast Strains’ Growth Rates under Mating Pheromone. Chemosensors 2022, 10, 141. [Google Scholar] [CrossRef]
- Sorba, F.; Poulin, A.; Ischer, R.; Shea, H.; Martin-Olmos, C. Integrated elastomer-based device for measuring the mechanics of adherent cell monolayers. Lab Chip 2019, 19, 2138–2146. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, X.; Chu, J.; Wang, A.; Zhu, Y.; Chu, W.K.; Yang, L.; Li, S. Uniaxial mechanical strain modulates the differentiation of neural crest stem cells into smooth muscle lineage on micropatterned surfaces. PLoS ONE 2011, 6, e26029. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chagnon-Lessard, S.; Godin, M.; Pelling, A.E. Time dependence of cellular responses to dynamic and complex strain fields. Integr. Biol. 2019, 11, 4–15. [Google Scholar] [CrossRef] [PubMed]
- Esfahani, A.M.; Rosenbohm, J.; Safa, B.T.; Lavrik, N.V.; Minnick, G.; Zhou, Q.; Kong, F.; Jin, X.; Kim, E.; Liu, Y.; et al. Characterization of the strain-rate-dependent mechanical response of single cell-cell junctions. Proc. Natl. Acad. Sci. USA 2021, 118, e2019347118. [Google Scholar] [CrossRef]
- Su, W.T.; Liao, Y.F.; Chu, I.M. Observation of fibroblast motility on a micro-grooved hydrophobic elastomer substrate with different geometric characteristics. Micron 2007, 38, 278–285. [Google Scholar] [CrossRef] [PubMed]
- Hsu, H.J.; Lee, C.F.; Kaunas, R. A dynamic stochastic model of frequency-dependent stress fiber alignment induced by cyclic stretch. PLoS ONE 2009, 4, e4853. [Google Scholar] [CrossRef] [Green Version]
- Kaunas, R.; Usami, S.; Chien, S. Regulation of stretch-induced JNK activation by stress fiber orientation. Cell. Signal. 2006, 18, 1924–1931. [Google Scholar] [CrossRef]





Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fang, X.; Wang, P.; Lin, F.; Huang, J.; Fang, J.; Xiong, C. Pneumatic Cell Stretching Chip to Generate Uniaxial Strain Using an Elastomeric Membrane with Ridge Structure. Chemosensors 2022, 10, 302. https://doi.org/10.3390/chemosensors10080302
Fang X, Wang P, Lin F, Huang J, Fang J, Xiong C. Pneumatic Cell Stretching Chip to Generate Uniaxial Strain Using an Elastomeric Membrane with Ridge Structure. Chemosensors. 2022; 10(8):302. https://doi.org/10.3390/chemosensors10080302
Chicago/Turabian StyleFang, Xu, Pudi Wang, Feng Lin, Jianyong Huang, Jing Fang, and Chunyang Xiong. 2022. "Pneumatic Cell Stretching Chip to Generate Uniaxial Strain Using an Elastomeric Membrane with Ridge Structure" Chemosensors 10, no. 8: 302. https://doi.org/10.3390/chemosensors10080302







