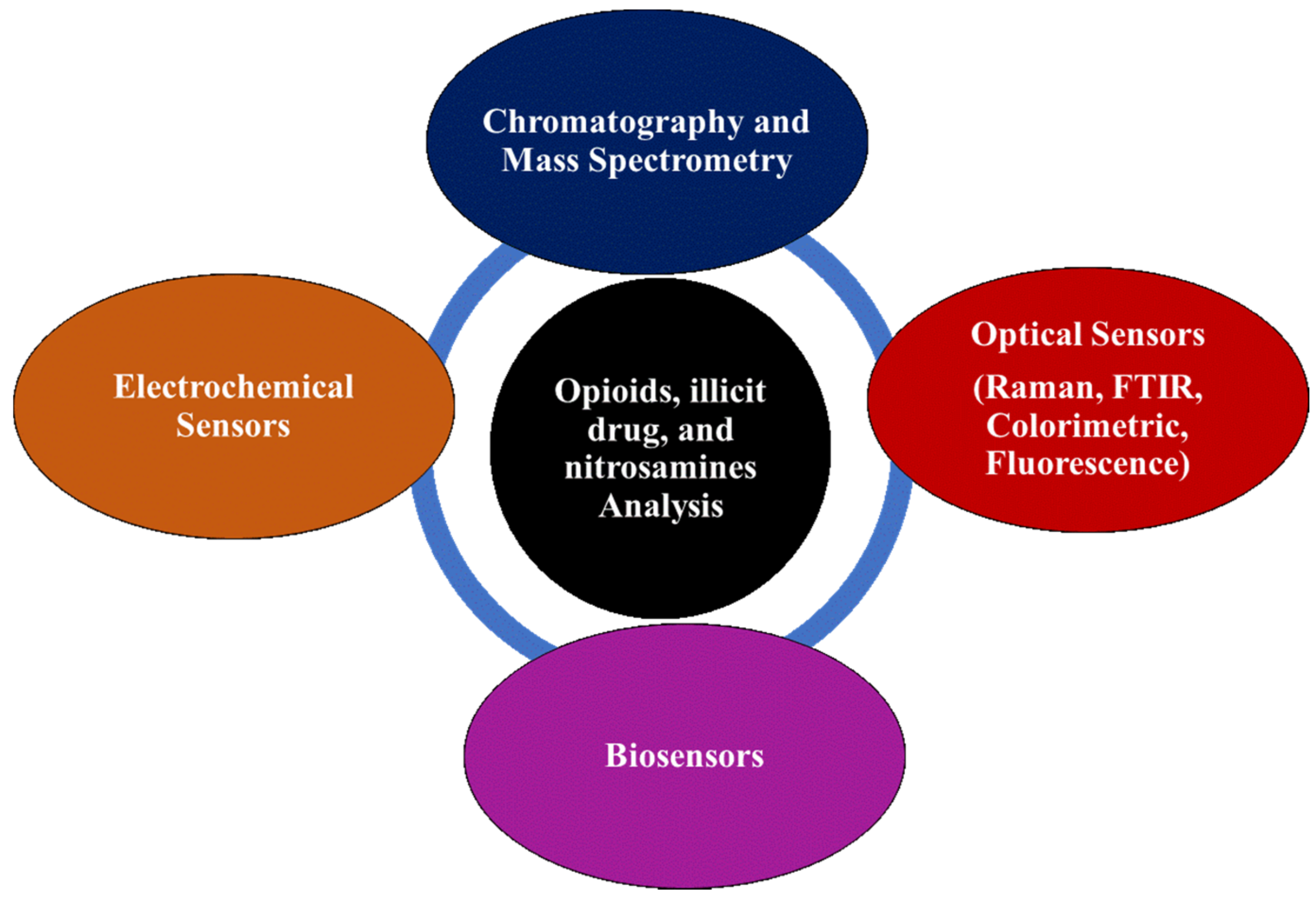
Electrochemical Sensors, Biosensors, and Optical Sensors for the Detection of Opioids and Their Analogs: Pharmaceutical, Clinical, and Forensic Applications
Abstract
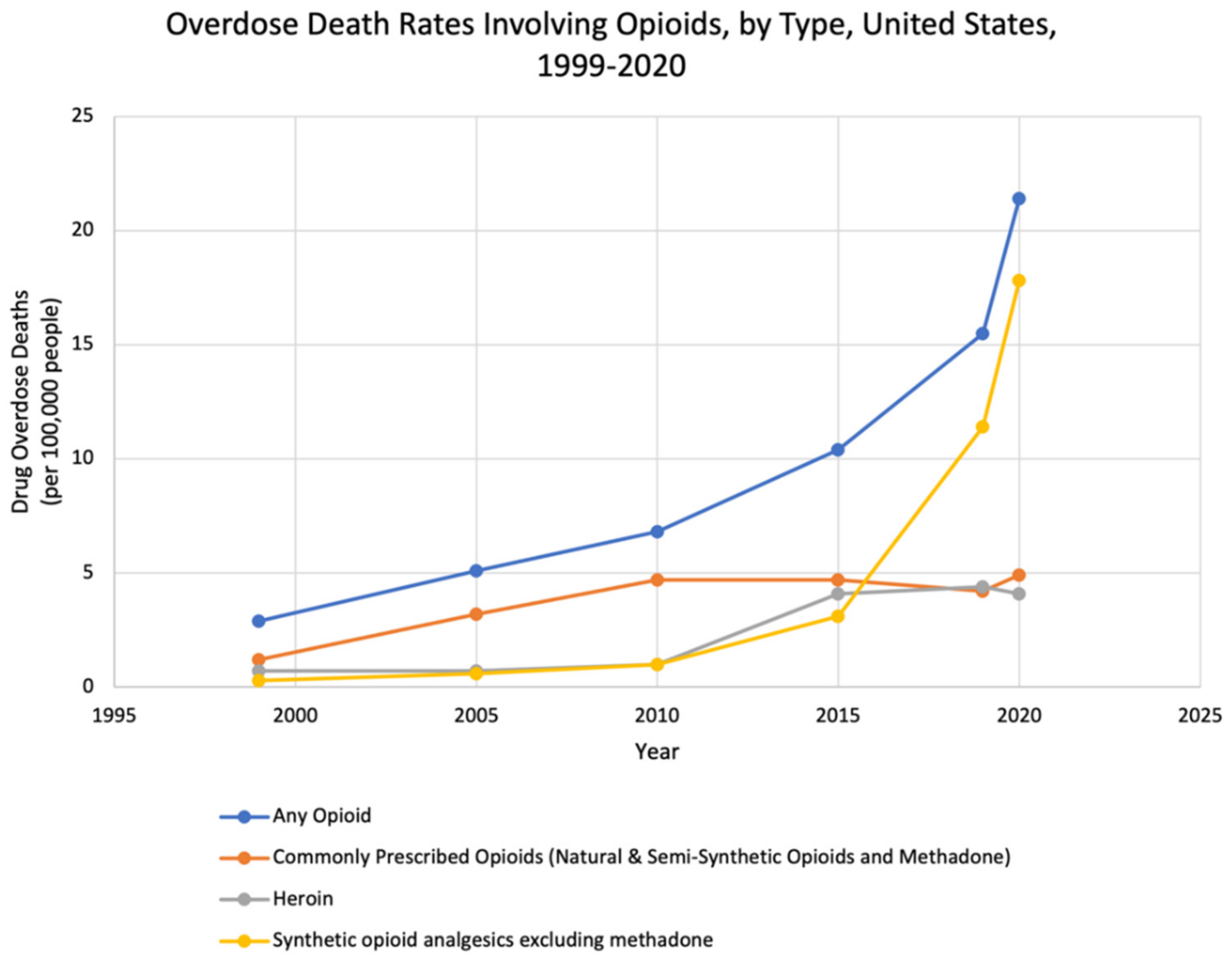
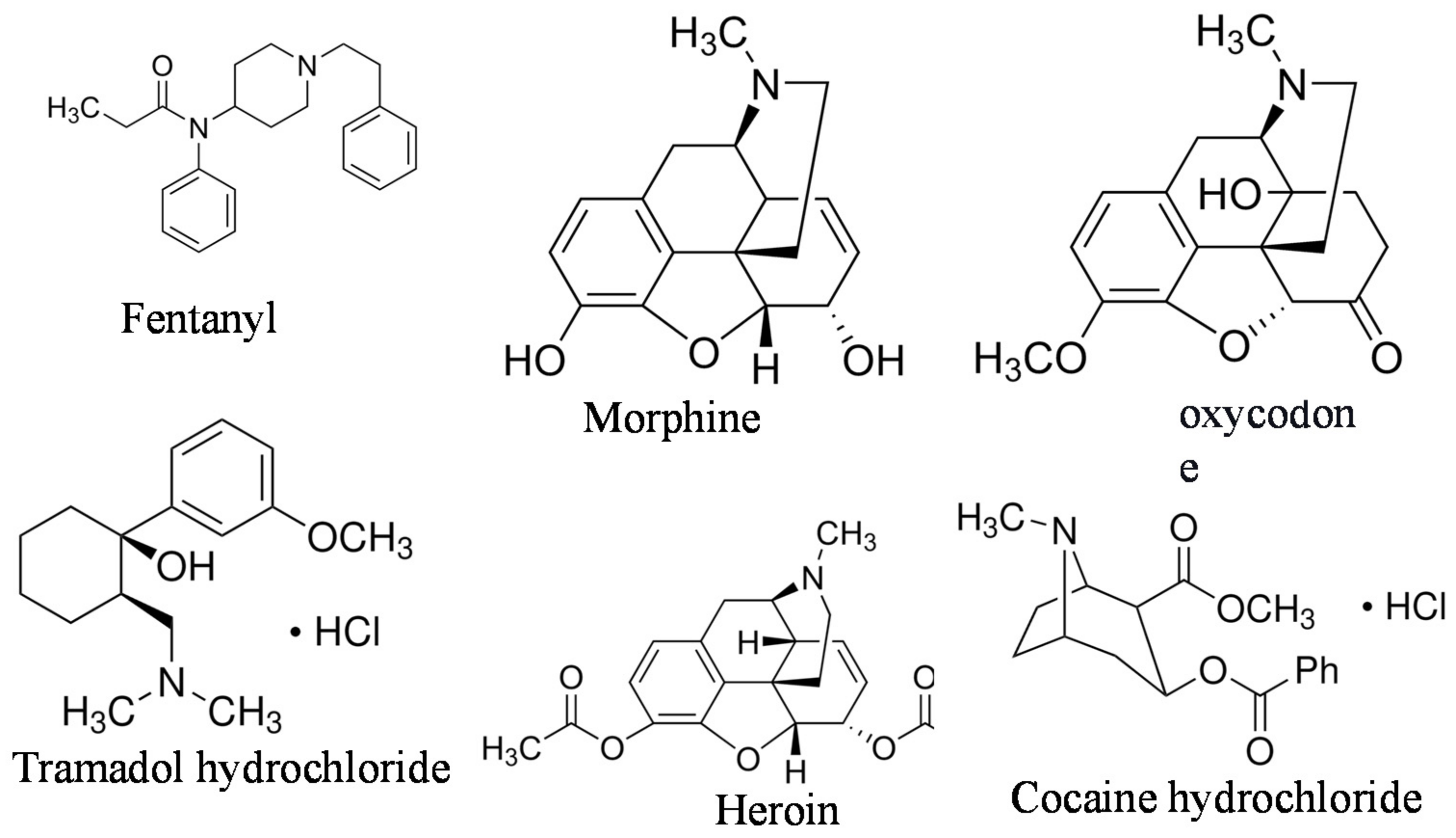
:1. Introduction
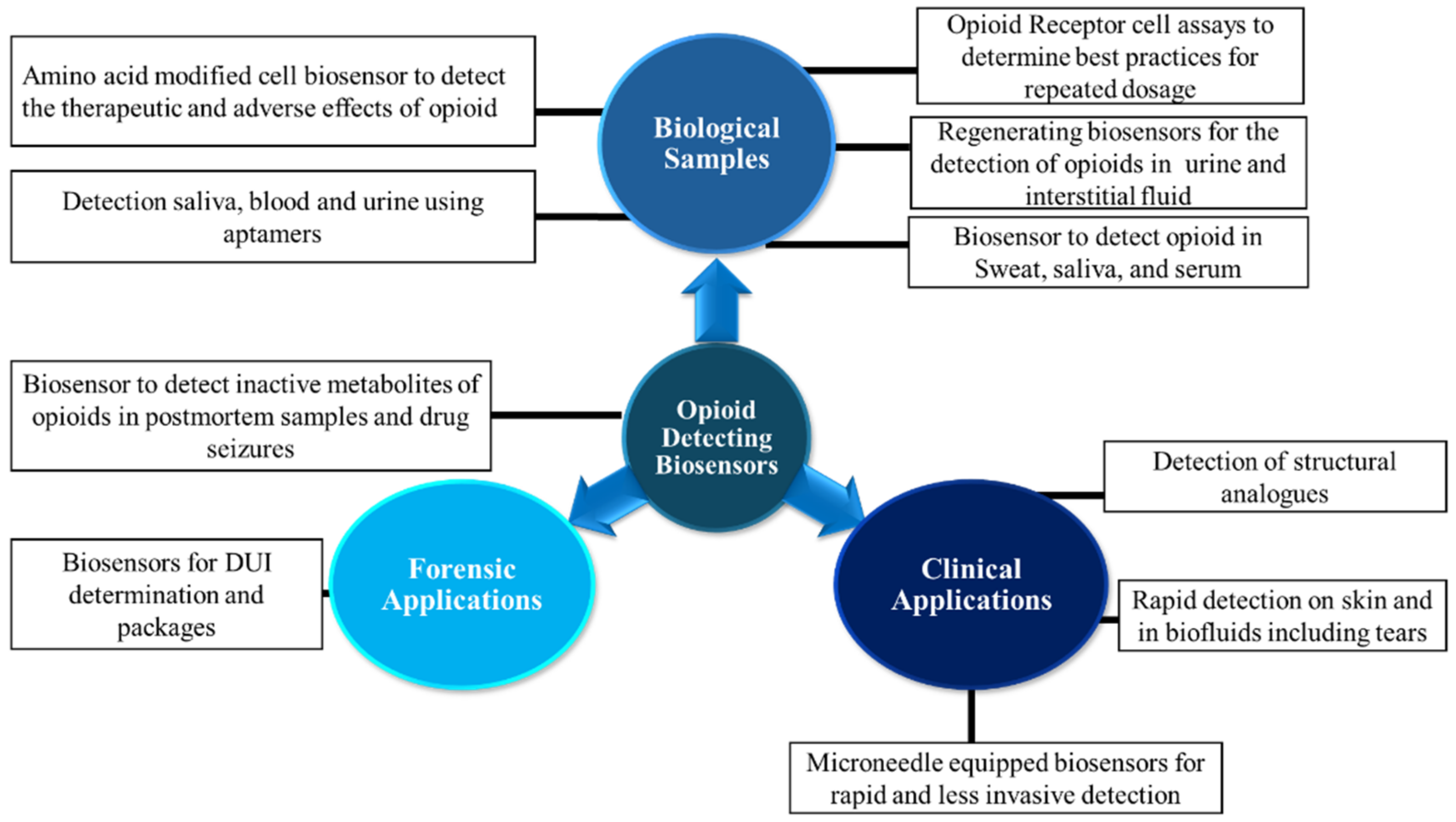
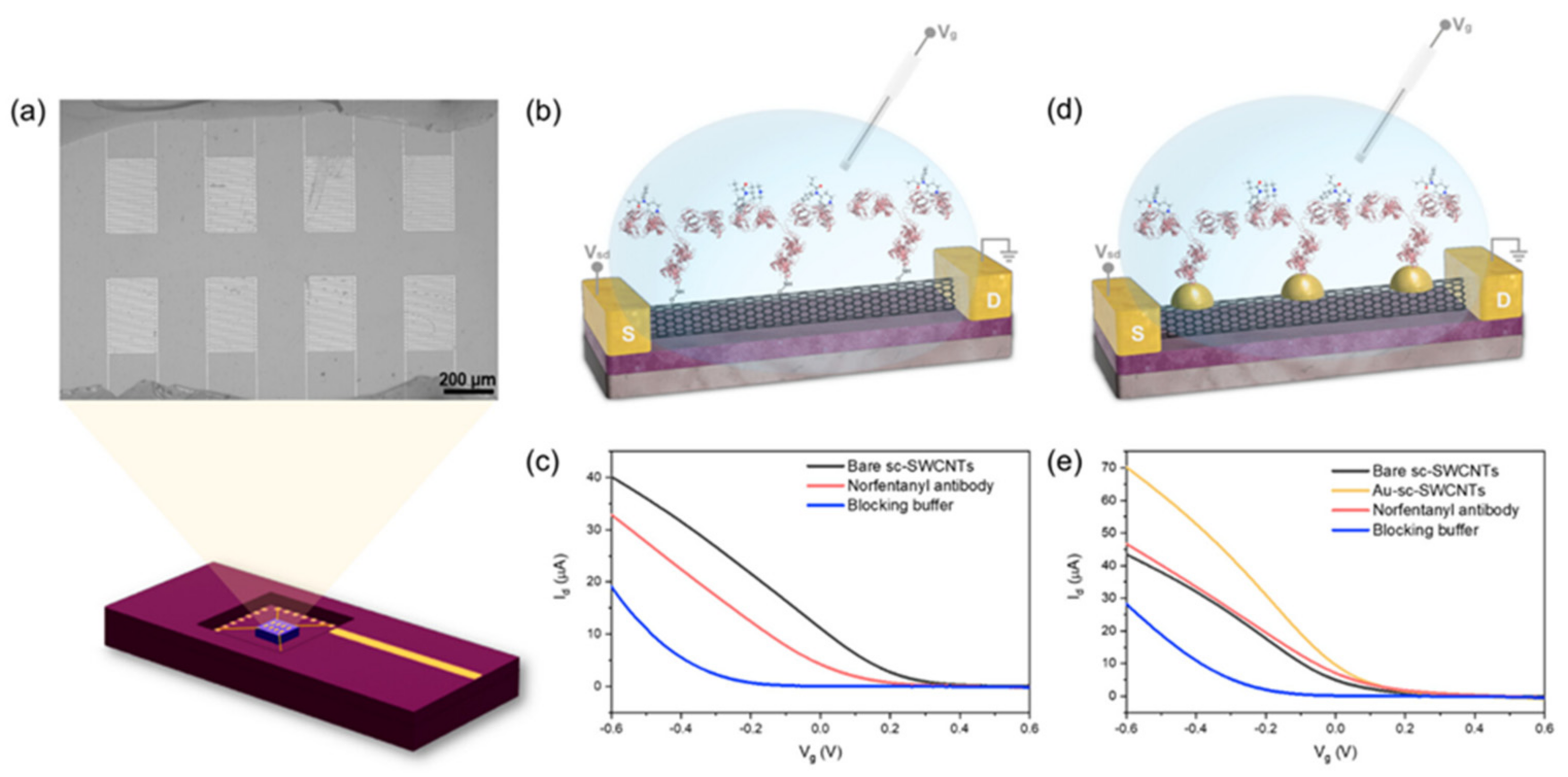
2. Biosensors Detection
2.1. Bisosensors Detection of Pharmaceutical Opioids, Illicit Opioids, and Analogs in Biological Samples
2.2. Biosensors: Clinical Applications
2.3. Biosensors: Forensic Applications
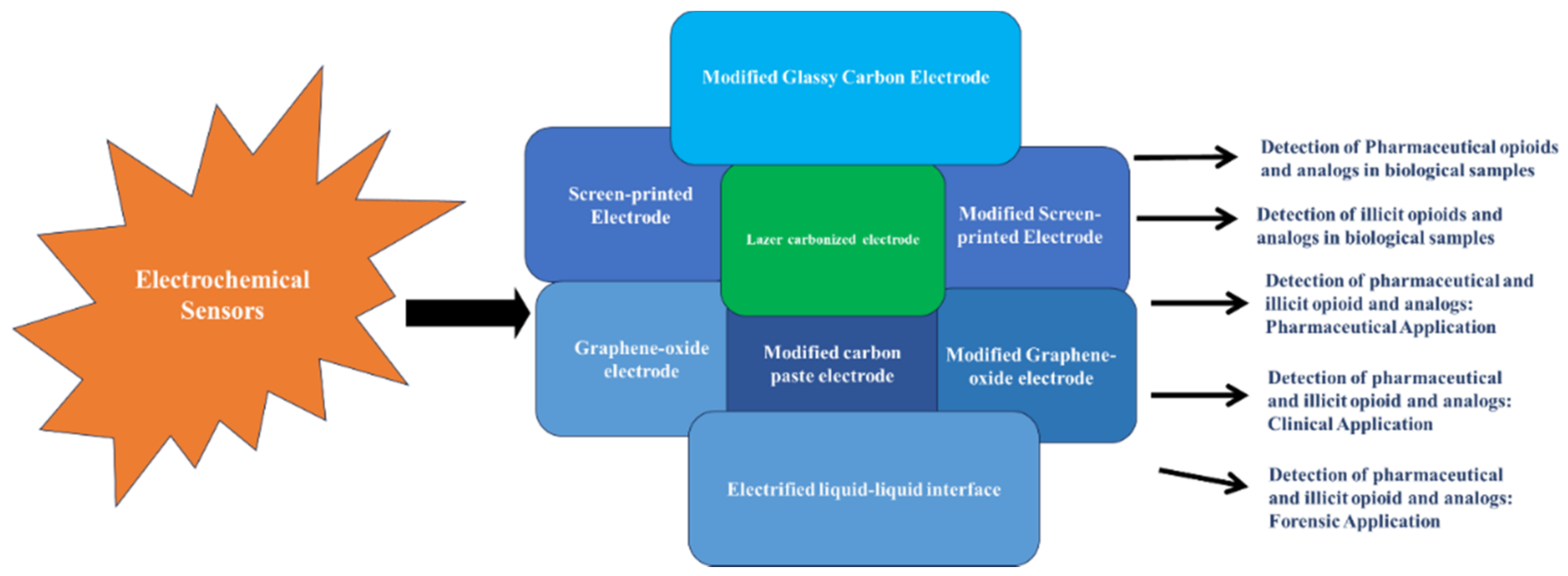
3. Electrochemical Detection
3.1. Electrochemical Sensors Detection of Pharmaceutical Opioids, Illicit Opioids, and Analogs in Biological Samples
3.2. Electrochemical Sensors: Pharmaceutical Applications
3.3. Electrochemical Sensors: Clinical Applications
3.4. Electrochemical Sensors: Forensic Applications
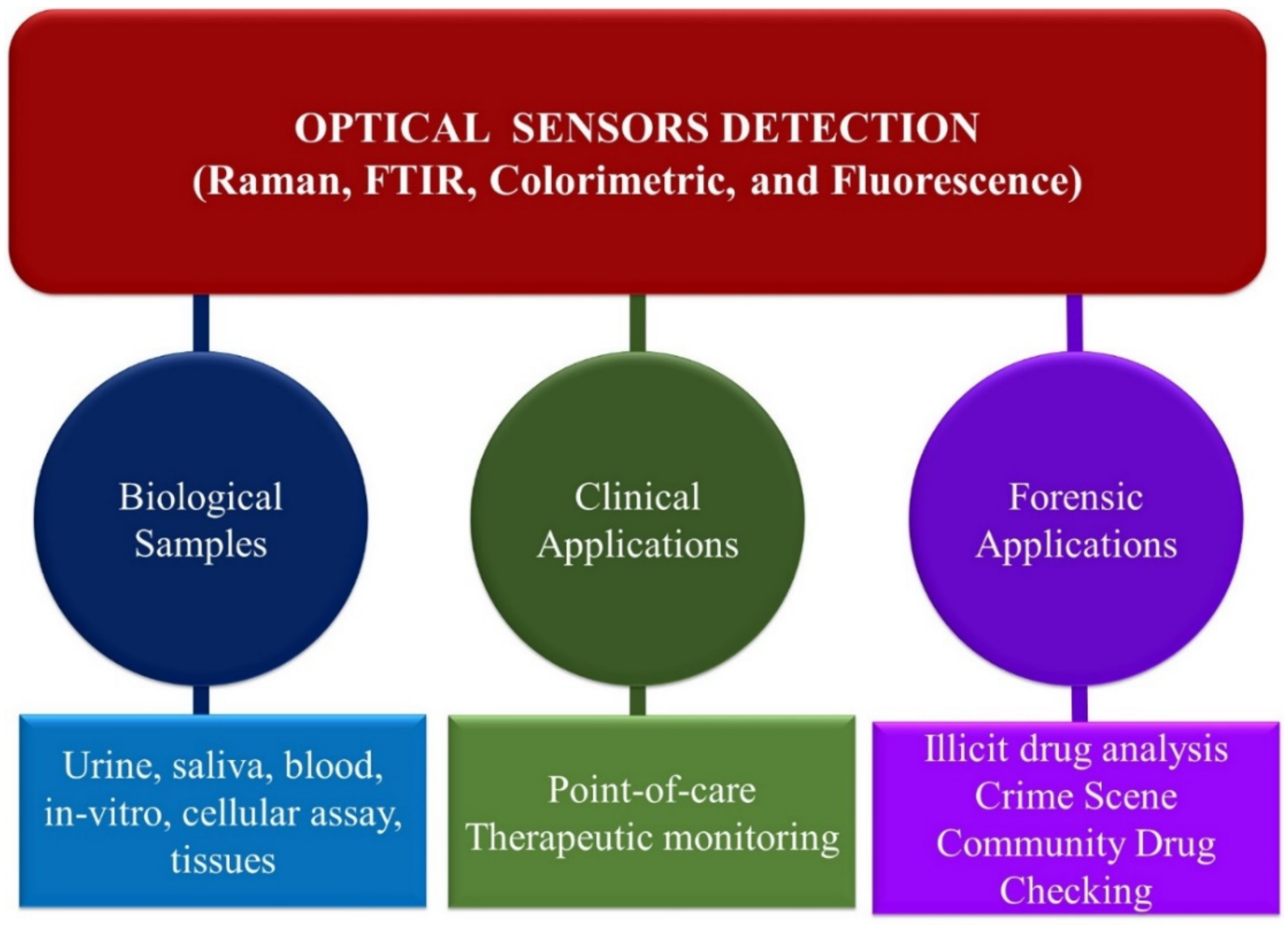
4. Main Optical Spectroscopy Sensors Detection Methods
4.1. FTIR Detection Methods
4.2. Raman Detection Methods
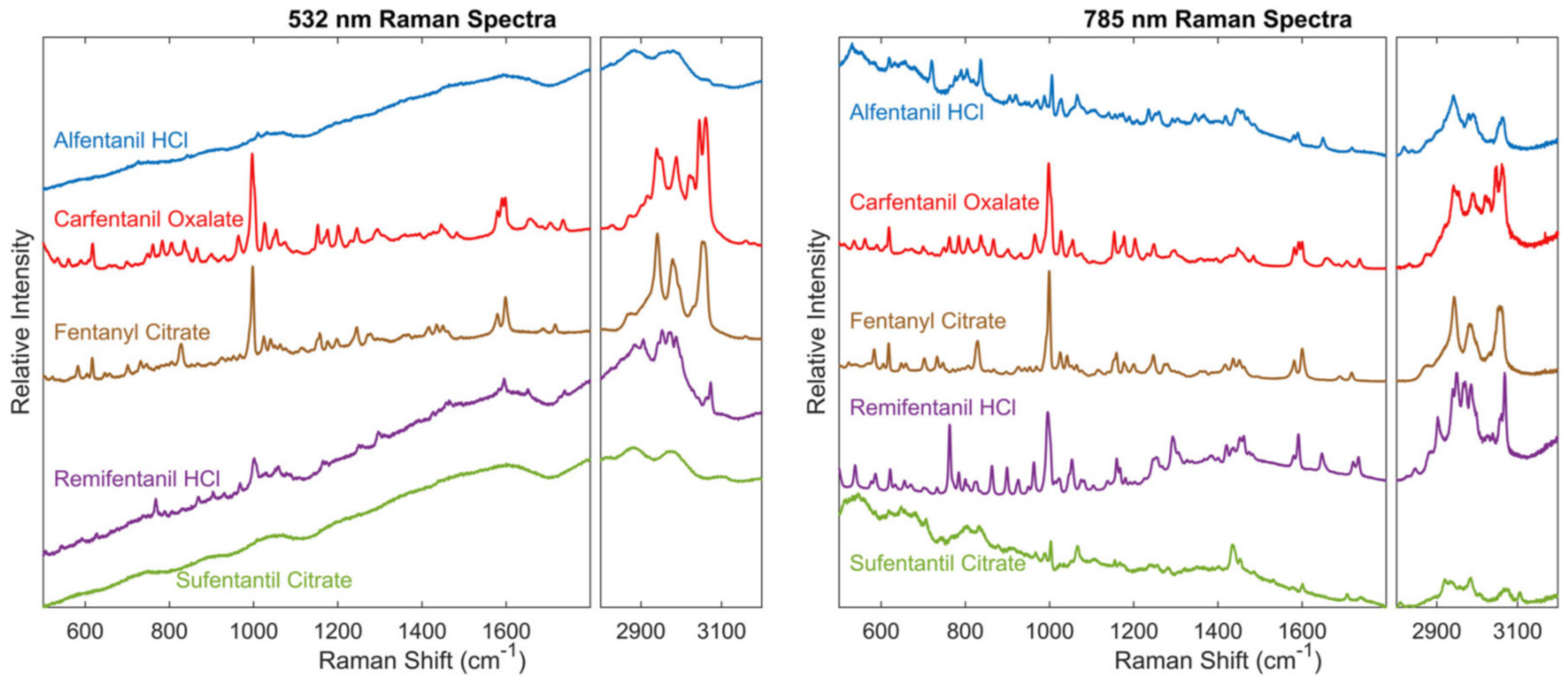
4.2.1. Raman Spectroscopic Detection of Pharmaceutical Opioids, Illicit Opioids, and Analogs in Biological Samples
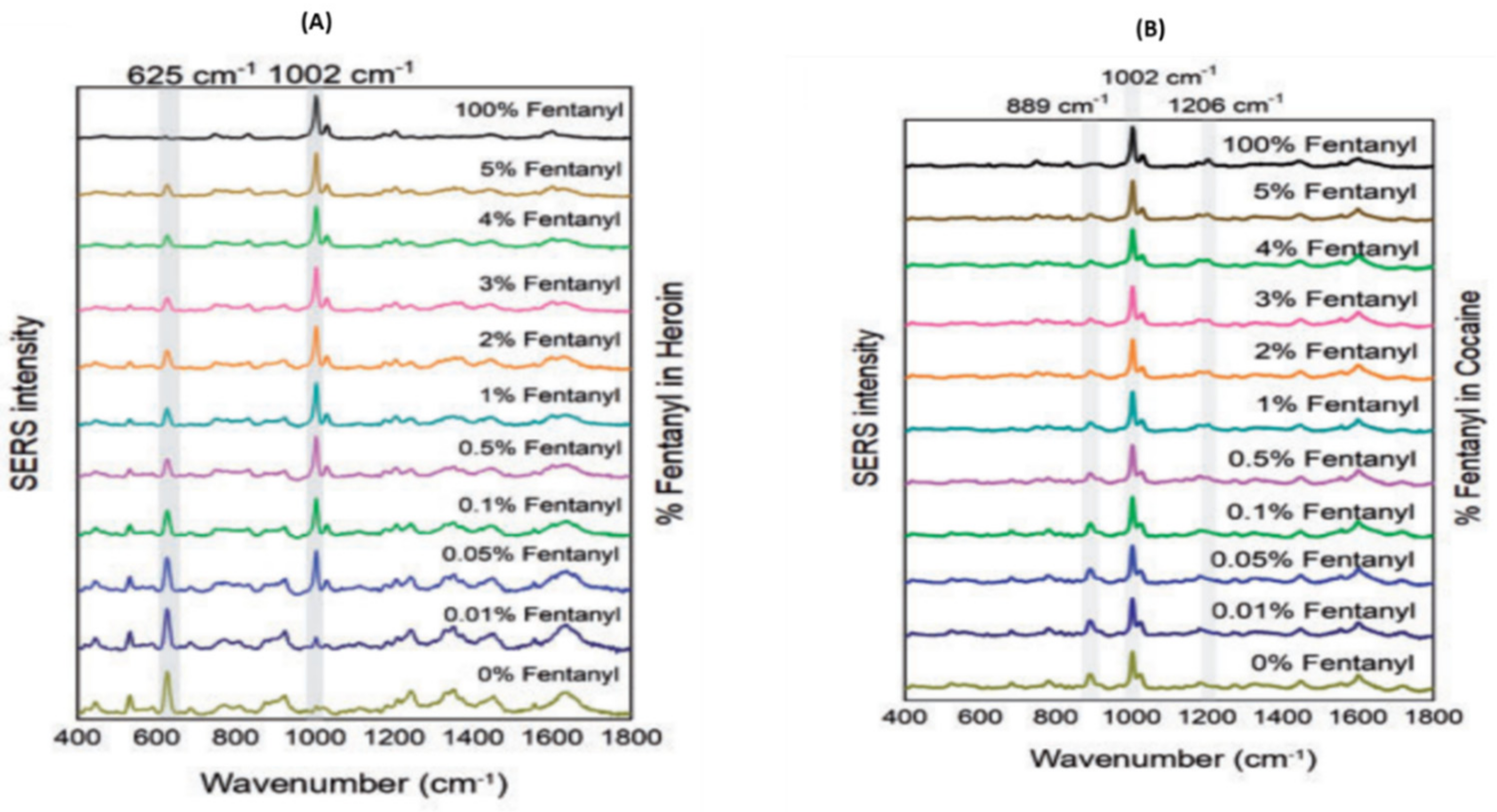
4.2.2. Raman Spectroscopy: Forensic Application
4.2.3. Raman Spectroscopy: Clinical Applications
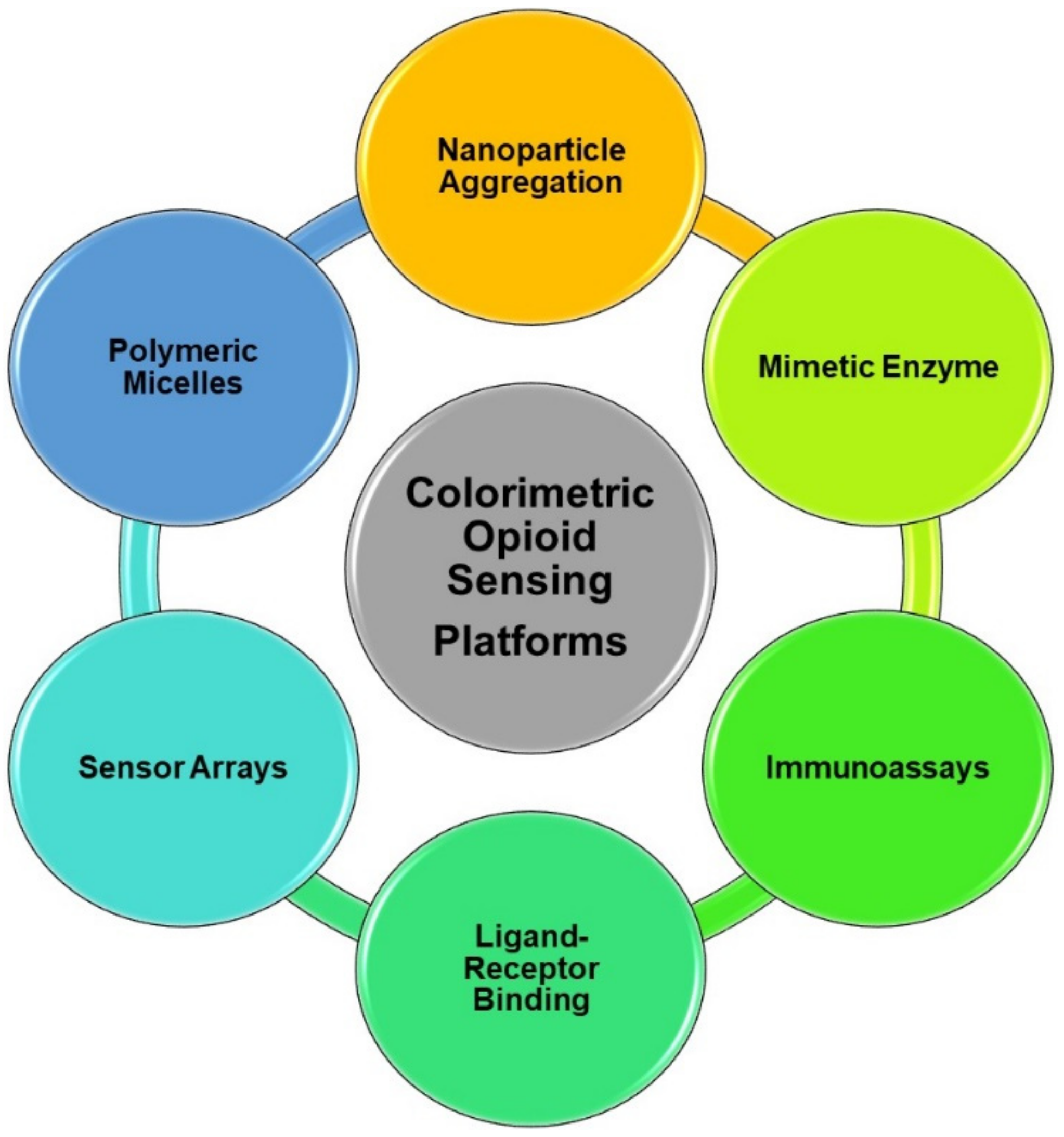
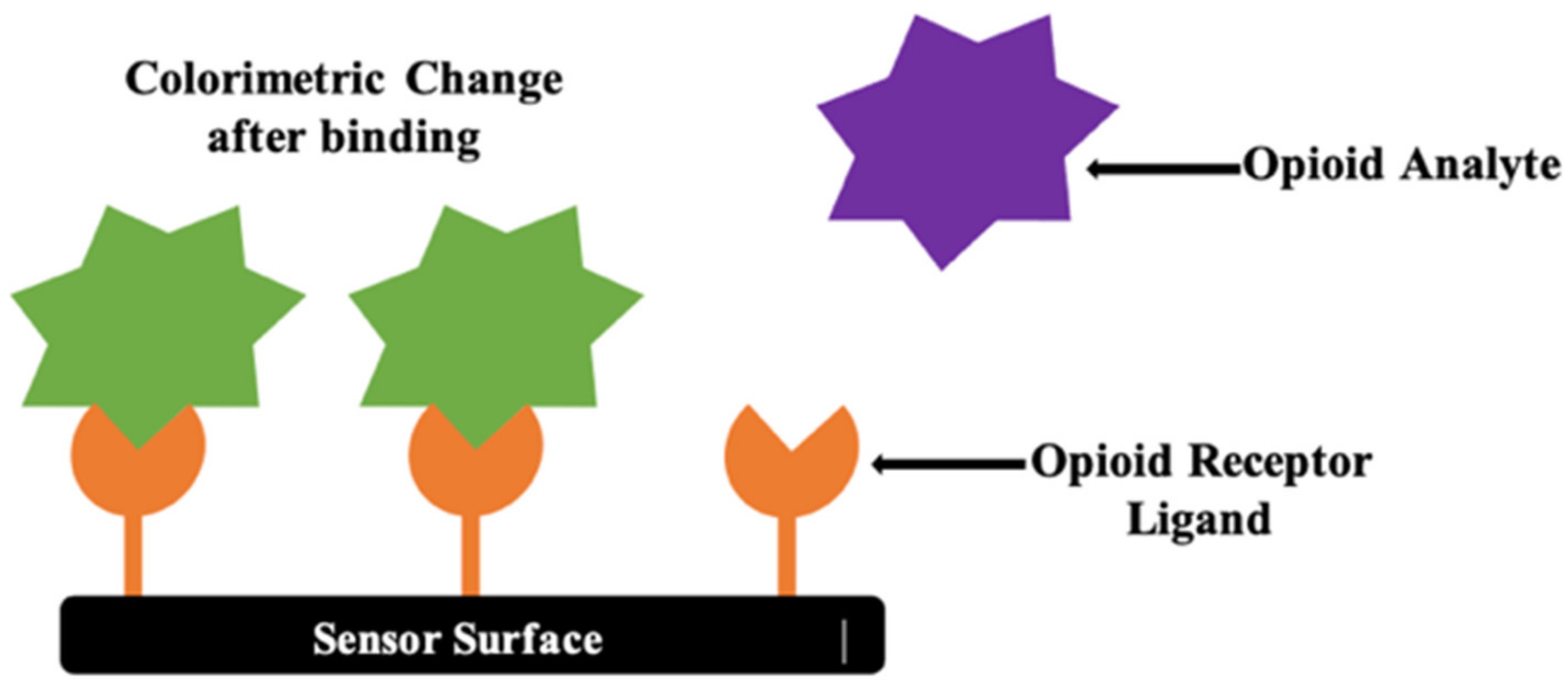
4.3. Colorimetric Detection
4.3.1. Colorimetric Detection of Pharmaceutical Opioids, Illicit Opioids, and Analogs in Biological Samples
4.3.2. Colorimetry Sensors: Pharmaceutical Applications
4.3.3. Colorimetry Sensors: Clinical Applications
4.3.4. Colorimetry Sensors: Forensic Applications
4.4. Fluorescence Detection
4.4.1. Fluorescence Sensors: Detection of Pharmaceutical Opioids, Illicit Opioids, and Analogs in Biological Samples
4.4.2. Fluorescence Sensors: Pharmaceutical Applications
4.4.3. Fluorescence Sensors: Clinical Applications
4.4.4. Fluorescence Sensors: Forensic Applications
5. Conclusions and Prospects
Author Contributions
Funding
Conflicts of Interest
References
- Breivik, H.; Collett, B.; Ventafridda, V.; Cohen, R.; Gallacher, D. Survey of chronic pain in Europe: Prevalence, impact on daily life, and treatment. Eur. J. Pain 2006, 10, 287–333. [Google Scholar] [CrossRef] [PubMed]
- Eccleston, C.; Jordan, A.L.; Crombez, G. The impact of chronic pain on adolescents: A review of previously used measures. J. Pediatr. Psychol. 2006, 31, 684–697. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.D.; McLellan, A.T. Opioid Abuse in Chronic Pain—Misconceptions and Mitigation Strategies. N. Engl. J. Med. 2016, 374, 1253–1263. [Google Scholar] [CrossRef] [PubMed]
- An Hecke, O.; Torrance, N.; Smith, B.H. Chronic pain epidemiology and its clinical relevance. Br. J. Anaesth. 2013, 111, 13–18. [Google Scholar] [CrossRef] [PubMed]
- Yong, R.J.; Mullins, P.M.; Bhattacharyya, N. Prevalence of chronic pain among adults in the United States. Pain 2022, 163, e328–e332. [Google Scholar] [CrossRef]
- Singh, A.; Menéndez-Perdomo, I.M.; Facchini, P.J. Benzylisoquinoline Alkaloid Biosynthesis in Opium Poppy: An Update. Phytochem. Rev. 2019, 18, 1457–1482. [Google Scholar] [CrossRef]
- World Health Organization. WHO Model List of Essential Medicines—22nd List, 2021; WHO: Geneva, Switzerland, 2021. [Google Scholar]
- Chou, R.; Fanciullo, G.J.; Fine, P.G.; Adler, J.A.; Ballantyne, J.C.; Davies, P.; Donovan, M.I.; Fishbain, D.A.; Foley, K.M.; Fudin, J. Clinical guidelines for the use of chronic opioid therapy in chronic noncancer pain. J. Pain 2009, 10, 113–130.e22. [Google Scholar]
- Fowler, T.O.; Durham, C.O.; Planton, J.; Edlund, B.J. Use of nonsteroidal anti-inflammatory drugs in the older adult. J. Am. Assoc. Nurse Pract. 2014, 26, 414–423. [Google Scholar] [CrossRef]
- Reduce Overdose Deaths Involving Opioids—IVP-20. Available online: https://health.gov/healthypeople/objectives-and-data/browse-objectives/injury-prevention/reduce-overdose-deaths-involving-opioids-ivp-20 (accessed on 16 December 2023).
- National Institutes on Drug Abuse. Drug Overdose Death Rates. Available online: https://nida.nih.gov/research-topics/trends-statistics/overdose-death-rates (accessed on 16 December 2023).
- Centers for Disease Control and Prevention. Drug Overdose Deaths in the U.S Top 100,000. Available online: https://www.cdc.gov/drugoverdose/deaths/index.html (accessed on 16 December 2023).
- Lombardi, A.R.; Arya, R.; Rosen, J.G.; Thompson, E.; Welwean, R.; Tardif, J.; Rich, J.D.; Park, J.N. Overdose Detection Technologies to Reduce Solitary Overdose Deaths: A Literature Review. Int. J. Environ. Res. Public Health 2023, 20, 1230. [Google Scholar] [CrossRef] [PubMed]
- Centers for Disease Control and Prevention; National Center for Health Statistics. Underlying Cause of Death 1999–2015 on CDC WONDER Online Database, Released 2017. Data are from the Multiple Cause of Death Files, 1999–2016, as Compiled from Data Provided by the 57 Vital Statistics Jurisdictions through the Vital Statistics Cooperative Program. 2017. Available online: http://wonder.cdc.gov/ucd-icd10.html (accessed on 16 December 2023).
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; American Psychiatric Association: Washington, DC, USA, 2013. [Google Scholar]
- Campbell, C.I.; Bahorik, A.L.; VanVeldhuisen, P.; Weisner, C.; Rubinstein, A.L.; Ray, G.T. Use of a prescription opioid registry to examine opioid misuse and overdose in an integrated health system. Prev. Med. 2018, 110, 31–37. [Google Scholar] [CrossRef]
- Centers for Disease Control and Prevention, Opioid Overdose: Opioid Data Analysis and Resources. Available online: https://www.cdc.gov/drugoverdose/data/OD-death-data.html (accessed on 3 February 2024).
- Nuamah, J.K.; Sasangohar, F.; Erraguntla, M.; Mehta, R.K. The past, present and future opioid withdrawal assessment: A scoping review of scales and technologies. BMC Med. Inform. Decis. Mak. 2019, 19, 113. [Google Scholar] [CrossRef] [PubMed]
- Birnbaum, H.G.; White, A.G.; Schiller, M.; Waldman, T.; Cleveland, J.M.; Roland, C.L. Societal costs of prescription opioid abuse, dependence, and misuse in the United States. Pain Med. 2011, 12, 657–667. [Google Scholar] [CrossRef] [PubMed]
- Florence, C.S.; Zhou, C.; Luo, F.; Xu, L. The economic burden of prescription opioid overdose, abuse, and dependence in the United States, 2013. Med. Care 2016, 54, 901–906. [Google Scholar] [CrossRef] [PubMed]
- McAdam-Marx, C.; Roland, C.L.; Cleveland, J.; Oderda, G.M. Costs of opioid abuse and misuse determined from a Medicaid database. J. Pain Palliat. Care Pharmacother. 2010, 24, 5–18. [Google Scholar] [CrossRef] [PubMed]
- Krueger, A.B. Where have all the workers gone? An inquiry into the decline of the U.S. labor force participation rate. Brook. Pap. Econ. Act. 2017, 2017, 1–87. [Google Scholar]
- Unfinished Business: Bipartisan Help for Child Victims of the Opioid Crisis. Retrieved September 17, 2018. Available online: https://www.brookings.edu/opinions/unfinished-business-bipartisan-help-for-child-victims-of-the-opioid-crisis/ (accessed on 16 December 2023).
- Strang, J.; Volkow, N.D.; Degenhardt, L.; Hickman, M.; Johnson, K.; Koob, G.F.; Marshall, B.D.L.; Walsh, S.L. Opioid use disorder. Nat. Rev. Dis. Primers 2020, 6, 4. [Google Scholar] [CrossRef] [PubMed]
- Jones, C.M.; Baldwin, G.T.; Compton, W.M. Recent Increases in Cocaine-Related Overdose Deaths and the Role of Opioids. AJPH Res. 2017, 107, 3. [Google Scholar]
- BC Coroners Service. Illicit Drug Overdose Deaths in BC: January 1, 2008–December 31, 2018; Ministry of Public Safety & Solicitor General: Victoria, BC, Canada, 2019. [Google Scholar]
- Ahmad, F.B.; Rossen, L.M.; Spencer, M.R.; Warner, M.; Sutton, P. Provisional Drug Overdose Death Counts; National Center for Health Statistics: Centers for Disease Control and Prevention. Available online: https://www.cdc.gov/nchs/nvss/vsrr/drug-overdose-data.htm (accessed on 16 December 2023).
- PHAC. Opioid- and Stimulant-Related Harms in Canada. Public Health Agency of Canada. 2022. Available online: https://health-infobase.canada.ca/substance-related-harms/opioids-stimulants/ (accessed on 16 December 2023).
- British Columbia Coroner’s Service. Distribution of illicit Drug Toxicity Deaths (2015-2024): British Columbia Coroner’s Service. Available online: http://www.bccdc.ca/resource-gallery/Documents/Statistics%20and%20Research/Statistics%20and%20Reports/Overdose/Illicit%20Drug%20Toxicity%20Deaths%20by%20HSDA%20BC%20-%20No%20Counts.pdf (accessed on 4 April 2024).
- BC Coroners Service. Illicit Drug Toxicity Deaths in BC; BC Coroners Service: Victoria, BC, Canada, 2022. [Google Scholar]
- Special Advisory Committee on the Epidemic of Opioid Overdoses. National Report: Apparent Opioid-Related Deaths in Canada (January 2016 to December 2018); Web Based Report; Public Health Agency of Canada: Ottawa, ON, Canada, 2019. [Google Scholar]
- Rudd, R.A.; Aleshire, N.; Zibbell, J.E.; Gladden, R.M. Increases in drug and opioid overdose deaths—United States, 2000-2014. MMWR Morb. Mortal Wkly. Rep. 2016, 64, 1378–1382. [Google Scholar] [CrossRef] [PubMed]
- Peterson, A.B.; Gladden, R.M.; Delcher, C.; Spies, E.; Garcia-Williams, A.; Wang, Y.; Halpin, J.; Zibbell, J.; McCarthy, C.L.; DeFiore-Hyrmer, J.; et al. Increases in fentanyl-related overdose deaths—Florida and Ohio, 2013–2015. MMWR Morb. Mortal Wkly. Rep. 2016, 65, 844–849. [Google Scholar] [CrossRef] [PubMed]
- American Medical Association. Physicians’ Progress toward Ending the Nation’s Drug Overdose Epidemic. 2022. Available online: https://www.ama-assn.org/delivering-care/overdose-epidemic/physicians-progress-toward-ending-nation-s-drug-overdose-epidemic (accessed on 11 December 2023).
- Irvine, M.A.; Kuo, M.; Buxton, J.A.; Balshaw, R.; Otterstatter, M.; Macdougall, L.; Milloy, M.J.; Bharmal, A.; Henry, B.; Tyndall, M. Modelling the combined impact of interventions in averting deaths during a synthetic-opioid overdose epidemic. Addiction 2019, 114, 1602–1613. [Google Scholar] [CrossRef] [PubMed]
- Wallace, B.; van Roode, T.; Pagan, F.; Phillips, P.; Wagner, H.; Calder, S.; Aasen, J.; Pauly, B.; Hore, D. What is needed for implementing drug checking services in the context of the overdose crisis? A qualitative study to explore perspectives of potential service users. Harm Reduct. J. 2020, 17, 29. [Google Scholar] [CrossRef] [PubMed]
- Maghsoudi, N.; McDonald, K.; Stefan, C.; Beriault, D.R.; Mason, K.; Barnaby, L. Evaluating networked drug checking services in Toronto, Ontario: Study protocol and rationale. Harm Reduct. J. 2020, 17, 9. [Google Scholar] [CrossRef] [PubMed]
- Larnder, A.; Saatchi, A.; Borden, S.A.; Moa, B.; Gill, C.G.; Wallace, B.; Hore, D. Variability in the unregulated opioid market in the context of extreme rates of overdose. Drug Alcohol Depend. 2022, 235, 109427. [Google Scholar] [CrossRef] [PubMed]
- Beaulieu, T.; Wood, E.; Tobias, S.; Lysyshyn, M.; Patel, P.; Mathews, J.; Ti, L. Is expected substance type associated with timing of drug checking service utilization? A cross-sectional study. Harm Reduct. J. 2021, 18, 66. [Google Scholar] [CrossRef] [PubMed]
- Tupper, K.W.; McCrae, K.; Garber, I.; Lysyshyn, M.; Wood, E. Initial results of a drug checking pilot program to detect fentanyl adulteration in a Canadian setting. Drug Alcohol Depend. 2018, 1, 242–245. [Google Scholar] [CrossRef] [PubMed]
- Wallace, B.; van Roode, T.; Burek, P.; Hore, D.; Pauly, B. Everywhere and for everyone: Proportionate universalism as a framework for equitable access to community drug checking. Harm Reduct. J. 2022, 19, 143. [Google Scholar] [CrossRef] [PubMed]
- Gozdzialski, L.; Wallace, B.; Hore, D. Point-of-care community drug checking technologies: An insider look at the scientific principles and practical considerations. Harm Reduct. J. 2023, 20, 39. [Google Scholar] [CrossRef] [PubMed]
- Harper, L.; Powell, J.; Pijl, E.M. An overview of forensic drug testing methods and their suitability for harm reduction point-of-care services. Harm Reduct. J. 2017, 14, 52. [Google Scholar] [CrossRef] [PubMed]
- Barratt, M.J.; Kowalski, M.; Maier, L.J.; Ritter, A. Global Review of Drug Checking Services Operating in 2017; National Drug and Alcohol Research Centre: Syndey, QLD, Australia, 2018. [Google Scholar]
- Pratiwi, R.; Noviana, E.; Fauziati, R.; Carrão, D.B.; Gandhi, F.A.; Majid, M.A.; Saputri, F.A. A Review of Analytical Methods for Codeine Determination. Molecules 2021, 26, 800. [Google Scholar] [CrossRef] [PubMed]
- Shan, X.; Cao, C.; Yang, B. Analytical Approaches for the Determination of Buprenorphine, Methadone and Their Metabolites in Biological Matrices. Molecules 2022, 27, 5211. [Google Scholar] [CrossRef]
- Musiał, J.; Czarny, J.; Gadzała-Kopciuch, R. Overview of analytical methods for determining novel psychoactive substances, drugs and their metabolites in biological samples. Crit. Rev. Toxicol. 2022, 52, 239–258. [Google Scholar] [CrossRef] [PubMed]
- Guerrieri, D.; Kjellqvist, F.; Kronstrand, R.; Gréen, H. Validation and cross-reactivity data for fentanyl analogs with the immunalysis fentanyl ELISA. J. Anal. Toxicol. 2019, 43, 18–24. [Google Scholar] [CrossRef] [PubMed]
- Abbott, D.L.; Limoges, J.F.; Virkler, K.J.; Tracy, S.J.; Sarris, G.G. ELISA screens for fentanyl in urine are susceptible to false-positives in high concentration methamphetamine samples. J. Anal. Toxicol. 2022, 46, 457–459. [Google Scholar] [CrossRef] [PubMed]
- European Network of OMCLs, OMCLs Release. Determination of NDMA in Valsartan Active Substances and Finished Products by HPLC/UV. Available online: https://www.google.com.hk/url?sa=t&source=web&rct=j&opi=89978449&url=https://www.edqm.eu/documents/52006/71923/Ad-hoc-projects-OMCL-Network-ansm-powdered-tablets-valsartan.pdf/a9bf0d81-f8da-9531-6de0-0268ab1614ad%3Ft%3D1628667816207&ved=2ahUKEwjYvPCM7ayFAxWSslYBHcYaD9AQFnoECB0QAQ&usg=AOvVaw2ft9Q3ITY49RCEAytE1dNV (accessed on 22 February 2024).
- French National Agency for Medicines and Health Products Safety; European Network of OMCLs, OMCLs Release. Test Method for the Determination of NDMA by LC/MS/MS in Valsartan Finished Products; Chemisches und Veterinäruntersuchungsamt: Karlsruhe, Germany, 2018. [Google Scholar]
- Chang, S.; Chang, C.; Wang, L.; Chen, W.; Fan, S.; Zang, C.; Hsu, Y.; Lin, M.; Tseng, S.; Wang, D. A multi-analyte LC-MS/MS method for screening and quantification of nitrosamines in sartans. J. Food Drug Anal. 2020, 28, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Marzan, T.A.; Ye, W.; Sommers, C.D.; Rodriguez, J.D.; Keire, D.A. A Cautionary Tale: Quantitative LC-HRMS Analytical Procedures for the Analysis of N-Nitrosodimethylamine in Metformin. AAPS J. 2020, 22, 89. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, S.; Sharma, A.; Chandrasekar, M.P.; Kotikalapudi, S. SCIEX, Gurgaon, India. LC-MS/MS Quantitative Analysis of NDMA in Ranitidine Active Pharmaceutical Ingredient (API) and Drug Product using the SCIEX 4500 QTRAP. Phenomenex India. Available online: https://www.phenomenex.com/c68401a7-a7d2-4274-9dc1-0e1c88a9cb92 (accessed on 16 December 2023).
- Guo, L.; Long, Z.; Leng, X.; Turner, J.; Rapid Analysis of Genotoxic Nitrosamines by HPLC-MS/MS. SCIEX APAC Applications Support Center, Beijing, China. Available online: https://www.phenomenex.com/c04c6179-3a07-4864-bc03-0abdc16cc57c (accessed on 16 December 2023).
- Wichitnithad, W.; Sudtanon, O.; Srisunak, P.; Cheewatanakornkool, K.; Nantaphol, S.; Rojsitthisak, P. Development of a Sensitive Headspace Gas Chromatography–Mass Spectrometry Method for the Simultaneous Determination of Nitrosamines in Losartan Active Pharmaceutical Ingredients ACS Omega 2021, 6, 11048–11058. [CrossRef]
- Parr, M.K.; Joseph, J.F. NDMA Impurity in Valsartan and Other Pharmaceutical Products: Analytical Methods for the Determination of N-Nitrosamines. J. Pharm. Biomed. Anal. 2019, 164, 536–549. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Xie, B.; Mai, B.; Cai, Q.; He, R.; Guo, D.; Zhang, Z.; Fan, J.; Zhang, W. Development of a Sensitive and Stable GC-MS/MS Method for Simultaneous Determination of Four N-Nitrosamine Genotoxic Impurities in Sartan Substances. J. Anal. Sci. Technol. 2021, 12, 3. [Google Scholar] [CrossRef]
- US Food and Drug Administration. Combined Headspace N-Nitrosodimethylamine (NDMA), N-Nitrosodiethylamine (NDEA), N-Nitrosoethylisopropylamine (NEIPA), and N-Nitrosodiisopropylamine (NDIPA) Impurity Assay by GC-MS/MS; US Food and Drug Administration: Silver Spring, MD, USA, 2019; pp. 1–7. [Google Scholar]
- Tsutsumi, T.; Akiyama, H.; Demizu, Y.; Uchiyama, N.; Masada, S.; Tsuji, G.; Arai, R.; Abe, Y.; Hakamatsuka, T.; Izutsu, K.; et al. Analysis of an Impurity, N-Nitrosodimethylamine, in Valsartan Drug Substances and Associated Products Using GC-MS. Biol. Pharm. Bull. 2019, 42, 547–551. [Google Scholar] [CrossRef] [PubMed]
- New OMCL Method for Simultaneous Determination of NDMA and NDEA in Sartan. Available online: https://www.edqm.eu/documents/52006/71923/Ad-hoc-projects-OMCL-Network-ranitidine.pdf/f775fbf3-705e-ce82-d1e1-8eb3c06002d4?t=1628667875462 (accessed on 16 December 2023).
- Lee, D.H.; Hwang, S.H.; Park, S.; Lee, J.; Oh, H.B.; Han, S.B.; Liu, K.-H.; Lee, Y.-M.; Pyo, H.S.; Hong, J. A Solvent-Free Headspace GC/MS Method for Sensitive Screening of N-Nitrosodimethylamine in Drug Products. Anal. Methods 2021, 13, 3402–3409. [Google Scholar] [CrossRef] [PubMed]
- Fakayode, S.O.; Walgama, C.; Fernand-Narcisse, V.E.; Grant, C. Electrochemical and Optical Nanosensors for Detection of Heavy Metal Ions: A Review. Sensors 2023, 23, 9080. [Google Scholar] [CrossRef] [PubMed]
- Fakayode, S.O.; Lisse, C.; Medawala, W.; Brady, P.M.; Bwambok, D.; Anum, D.; Alonge, T.; Taylor, M.E.; Baker, G.A.; Mehari, T.F.; et al. Fluorescent Chemical Sensors: Applications in Analytical, Environmental, Forensic, Pharmaceutical, Biological, and Biomedical Sample Measurement and Clinical Diagnosis. Appl. Spectrosc. Rev. 2023, 59, 1–89. [Google Scholar] [CrossRef]
- Siraj, N.; Brady, N.P.; Elzey, B.; Taylor, T.; Baker, G.; Bwambok, D.; Bashiru, M.; Macchi, S.; Jalihal, A.; Denmark, I.; et al. Raman Spectroscopy and Multivariate Regression Analysis in Biomedical Research, Medical Diagnosis, and Clinical Analysis. Appl. Spectrosc. Rev. 2021, 56, 615–672. [Google Scholar] [CrossRef]
- Fakayode, S.O.; Bwambok, D.; Suraj, N.; Baker, G.; Rodriguez, J.; Banerjee, S.; Perez, R.; Ayala, C.E.; Walgama, C.; Elzy, B.; et al. QCM Sensor Arrays, Electroanalytical Techniques and NIR Spectroscopy Coupled to Multivariate Analysis for Quality Assessment of Food Products, Raw Materials, and Ingredients and Foodborne Pathogen Detection: Challenges and Breakthroughs. Sensors 2020, 20, 6982. [Google Scholar] [CrossRef] [PubMed]
- Uezono, E.; Mizobuchi, Y.; Miyano, K.; Ohbuchi, K.; Murata, H.; Komatsu, A.; Manabe, S.; Nonaka, M.; Hirokawa, T.; Yamaguchi, K.; et al. Distinct profiles of desensitization of µ-opioid receptors caused by remifentanil or fentanyl: In vitro assay with cells and three-dimensional structural analyses. Int. J. Mol. Sci. 2023, 24, 8369. [Google Scholar] [CrossRef] [PubMed]
- Kroning, K.E.; Wang, W. Designing a single protein-chain reporter for opioid detection at cellular resolution. Angew. Chem. Int. Ed. Engl. 2021, 60, 13358–13365. [Google Scholar] [CrossRef] [PubMed]
- Kroning, K.E.; Li, M.; Petrescu, D.I.; Wang, W. A genetically encoded sensor with improved fluorescence intensity for opioid detection at cellular resolution. Chem. Commun. 2021, 57, 10560–10563. [Google Scholar] [CrossRef] [PubMed]
- Canoura, J.; Liu, Y.; Alkhamis, O.; Xiao, Y. Aptamer-based fentanyl detection in biological fluids. Anal. Chem. 2023, 95, 18258–18267. [Google Scholar] [CrossRef] [PubMed]
- Canoura, J.; Liu, Y.; Perry, J.; Willis, C.; Xiao, Y. Suite of aptamer-based sensors for the detection of fentanyl and its analogues. ACS Sens. 2023, 8, 1901–1911. [Google Scholar] [CrossRef] [PubMed]
- Nah, S.H.; Unsihuay, D.; Wang, P.; Yang, S. A highly sensitive and specific photonic crystal-based opioid sensor with rapid regeneration. ACS Appl. Mater. Inter. 2023, 15, 27647–27657. [Google Scholar] [CrossRef] [PubMed]
- Muthusamy, A.K.; Kim, C.H.; Virgil, S.C.; Knox, H.J.; Marvin, J.S.; Nichols, A.L.; Cohen, B.N.; Dougherty, D.A.; Looger, L.L.; Lester, H.A. Three mutations convert the selectivity of a protein sensor from nicotinic agonists to s-methadone for use in cells, organelles, and biofluids. J. Am. Chem. Soc. 2022, 144, 8480–8486. [Google Scholar] [CrossRef] [PubMed]
- González-Hernández, J.; Moya-Alvarado, G.; Alvarado-Gámez, A.L.; Urcuyo, R.; Barquero-Quirós, M.; Arcos-Martínez, M.J. Electrochemical biosensor for quantitative determination of fentanyl based on immobilized cytochrome c on multi-walled carbon nanotubes modified screen-printed carbon electrodes. Mikrochim. Acta 2022, 189, 483–495. [Google Scholar] [CrossRef] [PubMed]
- Cortade, D.L.; Wang, S.X. Quantitative and rapid detection of morphine and hydromorphone at the point of care by an automated giant magnetoresistive nanosensor platform. Anal. Bioanal. Chem. 2022, 414, 7211–7221. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Grillo, F.; Canfarotta, F.; Whitcombe, M.; Morgan, S.P.; Piletsky, S.; Correia, R.; He, C.; Norris, A.; Korposh, S. Carboxyl-fentanyl detection using optical fibre grating-based sensors functionalised with molecularly imprinted nanoparticles. Biosens. Bioelectron. 2021, 177, 113002–113011. [Google Scholar] [CrossRef]
- Wang, Z.; Nautiyal, A.; Alexopoulos, C.; Aqrawi, R.; Huang, X.; Ali, A.; Lawson, K.E.; Riley, K.; Adamczyk, A.J.; Dong, P.; et al. Fentanyl assay derived from intermolecular interaction-enabled small molecule recognition (imsr) with differential impedance analysis for point-of-care testing. Anal. Chem. 2022, 94, 9242–9251. [Google Scholar] [CrossRef] [PubMed]
- Joshi, P.; Riley, P.R.; Mishra, R.; Azizi Machekposhti, S.; Narayan, R. Transdermal polymeric microneedle sensing platform for fentanyl detection in biofluid. Biosensors 2022, 12, 198. [Google Scholar] [CrossRef] [PubMed]
- Shao, W.; Zeng, Z.; Star, A. An ultrasensitive norfentanyl sensor based on a carbon nanotube-based field-effect transistor for the detection of fentanyl exposure. ACS Appl. Mater. Interfaces 2023, 15, 37784–37793. [Google Scholar] [CrossRef] [PubMed]
- Angelini, D.J.; Biggs, T.D.; Prugh, A.M.; Smith, J.A.; Hanburger, J.A.; Llano, B.; Avelar, R.; Ellis, A.; Lusk, B.; Malik Naanaa, A.; et al. The use of lateral flow immunoassays for the detection of fentanyl in seized drug samples and postmortem urine. J. Foren. Sci. 2021, 66, 758–765. [Google Scholar] [CrossRef] [PubMed]
- Khosropour, H.; Rezaei, B.; Alinajafi, H.A.; Ensafi, A.A. Electrochemical sensor based on glassy carbon electrode modified by polymelamine formaldehyde/graphene oxide nanocomposite for ultrasensitive detection of oxycodone. Mikrochim. Acta 2021, 188, 1. [Google Scholar] [CrossRef] [PubMed]
- Baghayeri, M.; Nabavi, S.; Hasheminejad, E.; Ebrahimi, V. Introducing an electrochemical sensor based on two layers of Ag nanoparticles decorated graphene for rapid determination of methadone in human blood serum. Top Catal. 2022, 65, 623–632. [Google Scholar] [CrossRef]
- Habibi, M.M.; Ghasemi, J.B.; Badiei, A.; Narouzi, P. Simultaneous electrochemical determination of morphine and methadone by using CMK-5 mesoporous carbon and multivariate calibration. Sci. Rep. 2022, 12, 8270. [Google Scholar] [CrossRef] [PubMed]
- Abraham, P.; Renjini, S.; Mary Nancy, T.E.; Kumary, V.A. Electrochemical synthesis of thin-layered graphene oxide poly-(CTAB) composite for detection of morphine. J. App. Electrochem. 2020, 50, 41–50. [Google Scholar] [CrossRef]
- Beitollahi, H.; Nejad, F.G. Magnetic core-shell graphene oxide/Fe3O4@SiO2 nanocomposite for sensitive and selective electrochemical detection of morphine using modified graphite screen printed electrode. J. Anal. Chem. 2020, 75, 127–134. [Google Scholar] [CrossRef]
- Beitollhi, H.; Nejad, G.F.; Tajik, S.; Di Bartolomeo, A. Screen printed graphite electrode modified with graphene Co3O4 nanocomposite: Voltametric assay of morphine in the presence of diclofenac in pharmaceutical and biological samples. Nanomaterials 2022, 12, 3454. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, S.Z.; Jafari, M.; Jahani, M.P.; Ahmadi, S.A.; Mashayekh, R. Electrochemical determination of tramadol using modified screen-printed electrode. J. Electrochem. Sci. Eng. 2022, 12, 127–135. [Google Scholar] [CrossRef]
- Atta, N.F.; Abdo, G.G.; Elzatahry, A.; Galal, A.; Hassan, S.H. Designed electrochemical sensor based on metallocene modified conducting polymer composite for effective determination of tramadol in real samples. Can. J. Chem. 2021, 99, 437–446. [Google Scholar] [CrossRef]
- Mishra, K.R.; Krishnakumar, A.; Zareei, A.; Heredia-Rivera, U.; Rahimi, R. Electrochemical sensor for the rapid detection of fentanyl using laser-induced porous carbon-electrodes. Microchim. Acta 2022, 189, 198. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Wang, H. Fabrication of an electrochemical sensor based on a nanocomposite of CoO@f-CNTs for determination of tramadol narcotic drug in urine of athlete volunteers. Int. J. Electrochem. Sci. 2022, 17, 221018. [Google Scholar] [CrossRef]
- Abd-Rabboh, H.S.M.; Amr, A.E.-G.E.; Almehizia, A.A.; Kamel, A.H. All-solid-ctate potentiometric ion-sensors based tailored imprinted polymers for Pholcodine determination. Polymers 2021, 13, 1192. [Google Scholar] [CrossRef] [PubMed]
- Najafi, M.; Sohouli, E.; Mousavi, F. An electrochemical sensor for fentanyl detection based on multi-walled carbon nanotubes as electrocatalyst and the electrooxidation mechanism. J. Anal. Chem. 2021, 75, 1209–1217. [Google Scholar] [CrossRef]
- Hojjati-Najafabadi, A.; Salmanpour, S.; Sen, F.; Asrami, P.N.; Mahdavian, M.; Khalilzadeh, M.A. A Tramadol drug electrochemical sensor amplified by biosynthesized Au nanoparticle using Mentha aquatic extract and ionic liquid. Top. Catal. 2022, 65, 587–594. [Google Scholar] [CrossRef]
- Garkani Nejad, F.; Beitollahi, H.; Sheikhshoaie, I. A UiO-66-NH2 MOF/PAMAM Dendrimer Nanocomposite for Electrochemical Detection of Tramadol in the Presence of Acetaminophen in Pharmaceutical Formulations. Biosensors 2023, 13, 514. [Google Scholar] [CrossRef] [PubMed]
- Khairy, M.; Banks, C.E. A screen-printed electrochemical sensing platform surface modified with nanostructured ytterbium oxide nanoplates facilitating the electroanalytical sensing of the analgesic drugs acetaminophen and tramadol. Microchim. Acta 2020, 187, 126. [Google Scholar] [CrossRef] [PubMed]
- Kozak, J.; Tyszczuk-Rotko, K.; Wojciak, M.; Sowa, I. Electrochemically activated screen-printed carbon sensor modified with anionic surfactant (a SPCE/SDS) for simultaneous determination of Paracetamol, Diclofenac, and Tramadol. Materials 2021, 14, 3581. [Google Scholar] [CrossRef] [PubMed]
- Keskin, E.; Allahverdiyeva, S.; Seyho, E.; Yardim, Y. Determination of tramadol in pharmaceutical forms and urine samples using a boron-doped diamond electrode. J. Serb. Chem. Soc. 2020, 85, 933–937. [Google Scholar] [CrossRef]
- Solis, E., Jr.; Cameron-Burr, K.T.; Kivatkin, E.A. Rapid physiological fluctuations in nucleus accumbens oxygen levels induced by arousing stimuli: Relationships with changes in brain glucose and metabolic neural activation. Front. Integr. Neurosci. 2017, 11, 9. [Google Scholar] [CrossRef] [PubMed]
- Mohammadi, S.; Taher, M.A.; Beitollahi, H. A hierarchical 3D camellia-like molybdenum tungsten disulfide architectures for the determination of morphine and tramadol. Mikrochim. Acta 2020, 187, 312. [Google Scholar] [CrossRef] [PubMed]
- Razavi, R.; Amiri, M.; Divsalar, K.; Foroumadi, A. CuONPs/MWCNTs/carbon paste modified electrode for determination of tramadol: Theoretical and experimental investigation. Sci. Rep. 2023, 13, 7999. [Google Scholar] [CrossRef] [PubMed]
- Borgul, P.; Sobczak, K.; Sipa, K.; Rudnicki, K.; Skrzypek, S.; Trynda, A.; Poltorak, L. Heroin detection in a droplet hosted in a 3D printed support at the miniaturized electrified liquid-liquid interface. Sci. Rep. 2022, 12, 18615. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.R.; Bochner, F.; Somogyi, A. Pharmacokinetics of pholcodine in healthy volunteers: Single and chronic dosing studies. Br. J. Clin. Pharmacol. 1988, 26, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Thomas, S.A.; Curay, C.M.; Kiyatkin, E.A. Relationships between oxygen changes in the brain and periphery following physiological activation and the actions of heroin and cocaine. Sci. Rep. 2021, 11, 6355. [Google Scholar] [CrossRef] [PubMed]
- Rosendo, L.M.; Antunes, M.; Simão, A.Y.; Brinca, A.T.; Catarro, G.; Pelixo, R.; Martinho, J.; Pires, B.; Soares, S.; Cascalheira, J.F.; et al. Sensors in the Detection of Abused Substances in Forensic Contexts: A Comprehensive Review. Micromachines 2023, 14, 2249. [Google Scholar] [CrossRef] [PubMed]
- Mccrae, K.; Tobias, S.; Grant, C.; Lysyshyn, M.; Laing, R.; Wood, E.; Ti, L. Assessing the limit of detection of Fourier-transform infrared spectroscopy and immunoassay strips for fentanyl in a real-world setting. Drug Alcohol Rev. 2020, 39, 98–102. [Google Scholar] [CrossRef] [PubMed]
- United Nations Office on Drugs and Crime. United States: Rising Number of Deaths Involving Fentanyl and Fentanyl Analogues, Including Carfentanil. United Nations Office on Drugs and Crime. 2018. Available online: https://www.unodc.org/LSS/Announcement/Details/b9ed5cee-8ab2-4c4c-a1d1-b34069eddfe3 (accessed on 27 January 2024).
- Misailidi, N.; Papoutsis, I.; Nikolaou, P.; Dona, A.; Spiliopoulou, C.; Athanaselis, S. Fentanyls continue to replace heroin in the drug arena: The cases of ocfentanil and carfentanil. Forensic Toxicol. 2018, 36, 12–32. [Google Scholar] [CrossRef] [PubMed]
- Ti, L.; Tobias, S.; Lysyshyn, M.; Laing, R.; Nosova, E.; Choi, J.; Arredondo, J.; McCrae, K.; Tupper, K.; Wood, E. Detecting fentanyl using point-of-care drug checking technologies: A validation study. Drug Alcohol Depend. 2020, 212, 108006. [Google Scholar] [CrossRef] [PubMed]
- Deconinck, E.; Duchateau, C.; Balcaen, M.; Gremeaux, L.; Courselle, P. Chemometrics and infrared spectroscopy—A winning team for the analysis of illicit drug products. Rev. Anal. Chem. 2022, 41, 228–255. [Google Scholar] [CrossRef]
- Che, Z.; St Sauver, J.; Liu, H.; Liu, Y. Deep Learning Solutions for Classifying Patients on Opioid Use. AMIA Annu. Symp. Proc. 2018, 2017, 525–534. [Google Scholar] [PubMed]
- Marcelo, M.C.A.; Mariotti, K.C.; Ferrão, M.F.; Ortiz, R.S. Profiling cocaine by ATR–FTIR. Forensic Sci. Int. 2015, 246, 65–71. [Google Scholar] [CrossRef]
- Hespanhol, M.C.; Pasquini, C.; Maldaner, A. O Evaluation of a low-cost portable near-infrared spectrophotometer for in situ cocaine profiling. Talanta 2019, 200, 553–561. [Google Scholar] [CrossRef] [PubMed]
- Ti, L.; Grant, C.J.; Tobias, S.; Hore, D.K.; Laing, R.; Marshall, B.D.L. Development of a neural network model to predict the presence of fentanyl in community drug samples. PLoS ONE 2023, 18, e0288656. [Google Scholar] [CrossRef]
- He, X.; Wang, J.; Niu, F.; Fan, L.; Teng, X.; Zhang, C.; He, X. Characterization of Heroin and Its Additives by Attenuated Total Reflection (ATR)—Fourier Transform Infrared Spectroscopy (FTIR) and Multivariate Analysis. Anal. Lett. 2020, 53, 2656–2670. [Google Scholar] [CrossRef]
- Kranenburg, R.F.; Verduin, J.; Weesepoel, Y.; Alewijn, M.; Heerschop, M.; Koomen, G.; Keizers, P.; Bakker, F.; Wallace, F.; van Esch, A.; et al. Rapid and robust on-scene detection of cocaine in street samples using a handheld near-infrared spectrometer and machine learning algorithms. Drug Test Anal. 2020, 12, 1404–1418. [Google Scholar] [CrossRef] [PubMed]
- Farquharson, S.; Shende, C.; Newcomb, J.; Petrakis, I.L.; Arias, A.J. Analysis of drugs in saliva of US military veterans treated for substance use disorders using supported liquid extraction and surface-enhanced Raman spectral analysis. Molecules 2023, 28, 2010. [Google Scholar] [CrossRef] [PubMed]
- Markina, N.E.; Goryacheva, I.Y.; Markin, A.V. Surface-enhanced Raman spectroscopy for the determination of medical and narcotic drugs in human biofluids. J. Anal. Chem. 2022, 77, 930–947. [Google Scholar] [CrossRef]
- Akcan, R.; Yildirim, M.S.; Ilhan, H.; Guven, B.; Tamer, U.; Saglam, N. Surface enhanced Raman spectroscopy as a novel tool for rapid quantification of heroin and metabolites in saliva. Turk. J. Med. Sci. 2020, 50, 1470–1479. [Google Scholar] [CrossRef] [PubMed]
- Azimi, S.; Docoslis, A. Recent advances in the use of surface–enhanced Raman scattering for illicit drug detection. Sensors 2022, 22, 3887. [Google Scholar] [CrossRef]
- Haddad, A.; Green, O.; Lombardi, J.R. Detection of fentanyl in binary mixtures with cocaine by use of surface-enhanced Raman spectroscopy. Spectrosc. Lett. 2019, 52, 462–472. [Google Scholar] [CrossRef]
- Wang, L.; Vendrell-Dones, M.O.; Deriu, C.; Dogruer, S.; Harrington, P.B.; McCord, B. Multivariate analysis aided surface-enhanced Raman spectroscopy (MVA-SERS) multiplex quantitative detection of trace fentanyl in illicit drug mixtures using a handheld Raman spectrometer. Appl. Spectrosc. 2021, 75, 1225–1236. [Google Scholar] [CrossRef] [PubMed]
- Wilcox, P.G.; Emmons, E.D.; Pardoe, I.J.; Kline, N.D.; Guicheteau, J.A. Quantitative Raman cross-sections and band assignments for fentanyl and fentanyl analogs. Appl. Spectrosc. 2023, 77, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Shende, C.; Farquharson, A.; Brouillette, C.; Smith, W.; Farquharson, S. Quantitative measurements of codeine and fentanyl on a surface-enhanced Raman-active pad. Molecules 2019, 24, 2578. [Google Scholar] [CrossRef] [PubMed]
- Qin, Y.; Mo, F.; Yao, S.; Wu, Y.; He, Y.; Yao, W. Facile synthesis of porous Ag crystals as SERS sensor for detection of five methamphetamine analogs. Molecule 2022, 27, 3939. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Sheng, W.; Liu, X.; Guo, J.; Zhang, X.; Qi, X.; Zou, M.; Wang, C. The detection of 27 fentanyl compounds in solid and liquid drugs based on differential Raman spectroscopy. Chemosensors 2023, 11, 561. [Google Scholar] [CrossRef]
- Grover, C.; How, J.; Close, R. Fentanyl, etizolam, and beyond: A feasibility study of a community partnership using handheld Raman spectrometry to identify substances in the local illicit drug supply. J. Public Health Res. 2023, 12, 22799036231166313. [Google Scholar] [CrossRef] [PubMed]
- Mullin, A.; Scott, M.; Vaccaro, G.; Gittins, R.; Ferla, S.; Schifano, F.; Guirguis, A. Handheld Raman spectroscopy in the first UK home office licensed pharmacist-led community drug checking service. Int. J. Environ. Res. Public Health 2023, 20, 4793. [Google Scholar] [CrossRef] [PubMed]
- Gerace, E.; Seganti, F.; Luciano, C.; Lombardo, T.; Di Corcia, D.; Teifel, H.; Vincenti, M.; Salomone, A. On-site identification of psychoactive drugs by portable Raman spectroscopy during drug-checking service in electronic music events. Drug Alcohol Rev. 2019, 38, 50–56. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Ren, S.; Huang, H.; Duan, G.; Liu, K.; Liu, J. Fluorescent and Colorimetric Sensors Based on the Oxidation of o-Phenylenediamine. ACS Omega 2020, 5, 20698–20706. [Google Scholar] [CrossRef] [PubMed]
- Du, X.; Hao, H.; Qin, A.; Tang, B.Z. Highly sensitive chemosensor for detection of methamphetamine by the combination of AIE luminogen and cucurbit [7] uril. Dye Pigment. 2020, 180, 108413. [Google Scholar] [CrossRef]
- Zhang, K.; Chen, T.; Zhang, L.; Ma, S.; Shen, Y.; Feng, C.; Nie, P.; Yang, Z.; Zhu, C. The selective colorimetric probe based on a macrocyclic Sm(III) complex for detecting lithium ion and its performance in the psychiatric drug. Dye Pigment. Dye Pigment. 2020, 174, 108027, ISSN 0143-7208. [Google Scholar] [CrossRef]
- Jornet-Martínez, N.; Campíns-Falcó, P.; Herráez-Hernández, R. A Colorimetric Membrane-Based Sensor with Improved Selectivity towards Amphetamine. Molecules 2021, 26, 6713. [Google Scholar] [CrossRef] [PubMed]
- Sun, J.; Lu, Y.; He, L.; Pang, J.; Yang, F.; Liu, Y. Colorimetric sensor array based on gold nanoparticles: Design principles and recent advances. TrAC Trends Anal. Chem. 2020, 122, 115754, ISSN 0165-9936. [Google Scholar] [CrossRef]
- Alberti, G.; Zanoni, C.; Magnaghi, L.R.; Biesuz, R. Gold and Silver Nanoparticle-Based Colorimetric Sensors: New Trends and Applications. Chemosensors 2021, 9, 305. [Google Scholar] [CrossRef]
- Bastami, R.; Mansour Bayat, T.; Paolesse, R. Naked-eye detection of morphine by Au@ Ag nanoparticles-based colorimetric chemosensors. Sensors 2022, 22, 2072. [Google Scholar] [CrossRef] [PubMed]
- Yen, Y.; Lin, Y.; Chen, T.; Chyueh, S.; Chang, H. A Carbon-Dot Sensing Probe for Screening of Date Rape Drugs: Nitro-containing Benzodiazepines. Sens. Actuators B Chem. 2020, 305, 127441, ISSN 0925-4005. [Google Scholar] [CrossRef]
- Wei, S.; Li, Y.; Liang, H.; Yen, Y.; Lin, Y.; Chang, H. Photoluminescent carbon nanomaterials for sensing of illicit drugs: Focus. Anal. Sci. 2022, 38, 247–260. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Zhang, J.; Wang, M.; Su, X. Silicon quantum dots based dual-mode fluorometric and colorimetric sensing of D-penicillamine. Talanta 2021, 224, 121886, ISSN 0039-9140. [Google Scholar] [CrossRef]
- Ye, M.; Zhu, Y.; Lu, Y.; Gan, L.; Zhang, Y.; Zhao, Y. Magnetic nanomaterials with unique nanozymes-like characteristics for colorimetric sensors: A review. Talanta 2021, 230, 122299, ISSN 0039-9140. [Google Scholar] [CrossRef]
- Shaban, S.; Moon, B.; Kim, D. Selective and sensitive colorimetric detection of p-aminophenol in human urine and paracetamol drugs based on seed-mediated growth of silver nanoparticles. Environ. Technol. Innov. 2021, 22, 101517, ISSN 2352-1864. [Google Scholar] [CrossRef]
- Nishan, U.; Gul, R.; Muhammad, N.; Asad, M.; Rahim, A.; Shah, M.; Iqbal, J.; Uddin, J.; Shah, A.; Shujah, S. Colorimetric based sensing of dopamine using ionic liquid functionalized drug mediated silver nanostructures. Microchem. J. 2020, 159, 105382, ISSN 0026-265X. [Google Scholar] [CrossRef]
- Crowley, R.; Riley, A.; Alder, A.; Anderson III, R.; Luo, D.; Kaska, S.; Maynez, P.; Kivell, B.; Prisinzano, T. Synthetic Studies of Neoclerodane Diterpenes from Salvia divinorum: Design, Synthesis, and Evaluation of Analogues with Improved Potency and G-protein Activation Bias at the μ-Opioid Receptor. ACS Chem. Neurosci. 2020, 11, 1781–1790. [Google Scholar] [CrossRef] [PubMed]
- Soni, S.; Jain, U.; Burke, D.; Chauhan, N. Recent trends and emerging strategies for aptasensing technologies for illicit drugs detection. J. Electroanal. Chem. 2022, 910, 116128, ISSN 1572-6657. [Google Scholar] [CrossRef]
- Xie, M.; Zhao, F.; Zhang, Y.; Xiong, Y.; Han, S. Recent advances in aptamer-based optical and electrochemical biosensors for detection of pesticides and veterinary drugs. Food Control 2022, 131, 108399, ISSN 0956-7135. [Google Scholar] [CrossRef]
- Shkembi, X.; Botero, M.; Skouridou, V.; Jauset-Rubio, M.; Svobodova, M.; Ballester, P.; Bashammakh, A.; El-Shahawi, M.; Alyoubi, A.; O’Sullivan, C. Novel nandrolone aptamer for rapid colorimetric detection of anabolic steroids. Anal. Biochem. 2022, 658, 114937, ISSN 0003-2697. [Google Scholar] [CrossRef]
- Zhao, J.; Kan, Y.; Chen, Z.; Li, H.; Zhang, W. MOFs-Modified Electrochemical Sensors and the Application in the Detection of Opioids. Biosensors 2023, 13, 284. [Google Scholar] [CrossRef] [PubMed]
- D’Aurelio, R.; Chianella, I.; Goode, J.A.; Tothill, I.E. Molecularly Imprinted Nanoparticles Based Sensor for Cocaine Detection. Biosensors 2020, 10, 22. [Google Scholar] [CrossRef] [PubMed]
- Akhoundian, M.; Alizadeh, T. Enzyme-free colorimetric sensor based on molecularly imprinted polymer and ninhydrin for methamphetamine detection. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 285, 121866, ISSN 1386-1425. [Google Scholar] [CrossRef]
- Völlmecke, K.; Afroz, R.; Bierbach, S.; Brenker, L.J.; Frücht, S.; Glass, A.; Giebelhaus, R.; Hoppe, A.; Kanemaru, K.; Lazarek, M.; et al. Hydrogel-Based Biosensors. Gels 2022, 8, 768. [Google Scholar] [CrossRef] [PubMed]
- Akgönüllü, S.; Denizli, A. Plasmonic nanosensors for pharmaceutical and biomedical analysis. J. Pharm. Biomed. Anal. 2023, 236, 115671, ISSN 0731-7085. [Google Scholar] [CrossRef]
- Clifford, A.; Das, J.; Yousefi, H.; Mahmud, A.; Chen, J.; Kelley, S. Strategies for Biomolecular Analysis and Continuous Physiological Monitoring. J. Am. Chem. Soc. 2021, 143, 5281–5294. [Google Scholar] [CrossRef] [PubMed]
- Ghorbanizamani, F.; Moulahoum, H.; Timur, S. Noninvasive Optical Sensor for the Detection of Cocaine and Methamphetamine in Saliva Using Rhodamine B-Labeled Polymersomes. IEEE Sens. J. 2022, 22, 1146–1153. [Google Scholar] [CrossRef]
- Cho, S.; Kim, Y. Donor–acceptor Stenhouse adduct formation for the simple and rapid colorimetric detection of amphetamine-type stimulants. Sens. Actuators B Chem. 2022, 355, 131274, ISSN 0925-4005. [Google Scholar] [CrossRef]
- Ramadan, H.; Salam, R.; Hadad, G.; Belal, F.; Salim, M. Eco-friendly simultaneous multi-spectrophotometric estimation of the newly approved drug combination of celecoxib and tramadol hydrochloride tablets in its dosage form. Sci. Rep. 2023, 13, 11716. [Google Scholar] [CrossRef] [PubMed]
- Crocombe, R.; Giuntini, G.; Schiering, D.; Profeta, L.; Hargreaves, M.; Leary, P. Field-portable detection of fentanyl and its analogs: A review. J. Forensic Sci. 2023, 68, 1570–1600. [Google Scholar] [CrossRef] [PubMed]
- Alonzo, M.; Alder, R.; Clancy, L.; Fu, S. Portable testing techniques for the analysis of drug materials. Wiley Interdiscip. Rev. Forensic Sci. 2022, 4, e1461. [Google Scholar] [CrossRef]
- Bui, T.H.; Thangavel, B.; Sharipov, M.; Chen, K.; Shin, J.H. Smartphone-Based Portable Bio-Chemical Sensors: Exploring Recent Advancements. Chemosensors 2023, 11, 468. [Google Scholar] [CrossRef]
- Lantam, A.; Limbut, W.; Thiagchanya, A.; Phonchai, A. A portable optical colorimetric sensor for the determination of promethazine in lean cocktail and pharmaceutical doses. Microchem. J. 2020, 159, 105519, ISSN 0026-265X. [Google Scholar] [CrossRef]
- Kang, C.; Shrestha, K.L.; Kwon, S.; Park, S.; Kim, J.; Kwon, Y. Intein-Mediated Protein Engineering for Cell-Based Biosensors. Biosensors 2022, 12, 283. [Google Scholar] [CrossRef]
- Anzar, N.; Suleman, S.; Parvez, S.; Narang, J. A review on Illicit drugs and biosensing advances for its rapid detection. Process Biochem. 2022, 113, 113–124, ISSN 1359-5113. [Google Scholar] [CrossRef]
- Truta, F.; Florea, A.; Cernat, A.; Tertis, M.; Hosu, O.; De Wael, K.; Cristea, C. Tackling the Problem of Sensing Commonly Abused Drugs Through Nanomaterials and (Bio)Recognition Approaches. Front. Chem. 2020, 8, 561638. [Google Scholar] [CrossRef]
- Kim, K.; Stoll, S.; Singh, R.; Lee, W.; Hwang, J. Recent advances in illicit drug detection sensor technology in water. TrAC Trends Anal. Chem. 2023, 168, 117295, ISSN 0165-9936. [Google Scholar] [CrossRef]
- Ahmed, S.; Chand, R.; Kumar, S.; Mittal, N.; Srinivasan, S.; Rajabzadeh, A. Recent biosensing advances in the rapid detection of illicit drugs. TrAC Trends Anal. Chem. 2020, 131, 116006, ISSN 0165-9936. [Google Scholar] [CrossRef]
- Bruni, A.T.; Rodrigues, C.H.P.; dos Santos, C.; de Castro, J.S.; Mariotto, L.S.; Sinhorini, L.F.C. Analytical Challenges for Identification of New Psychoactive Substances. Braz. J. Anal. Chem. 2020, 9, 52–78. [Google Scholar] [CrossRef]
- Cao, F.; Xu, J.; Yan, S.; Yuan, X.; Yang, F.; Hou, L.; Zhao, L.; Zeng, L.; Liu, W.; Zhu, L.; et al. A surface plasmon resonance-based inhibition immunoassay for forensic determination of methamphetamine in human serum. Forensic Chem. 2018, 8, 21–27, ISSN 2468-1709. [Google Scholar] [CrossRef]
- Dagar, M.; Yadav, S.; Sai, V.; Satija, J.; Bhatia, H. Emerging trends in point-of-care sensors for illicit drugs analysis. Talanta 2022, 238, 123048, ISSN 0039-9140. [Google Scholar] [CrossRef]
- Goli-Malekabadi, Z.; Salarizadeh, N.; Dianatkhah, M.; Amoo, M.; Shayeh, J. Chapter 11—Biosensors for drug detection. In Advanced Sensor Technology; Elsevier: Amsterdam, The Netherlands, 2023; pp. 383–412. ISBN 9780323902229. [Google Scholar] [CrossRef]
- Dashtian, K.; Amourizi, F.; Shahbazi, N.; Mousavi, A.; Saboorizadeh, B.; Astaraei, S.; Zare-Dorabei, R. Chapter 5—Biosensors for drug of abuse detection. In Advanced Sensor Technology; Elsevier: Amsterdam, The Netherlands, 2023; pp. 125–172. ISBN 9780323902229. [Google Scholar] [CrossRef]
- Theyagarajan, K.; Sruthi, V.; Mohanapriya, D.; Thenmozhi, K.; Senthilkumar, S. Chapter 7—Biosensor platform for testing active pharmaceutical ingredients. In Health and Environmental Applications of Biosensing Technologies; Elsevier: Amsterdam, the Netherlands, 2024; pp. 129–152. ISBN 9780443190391. [Google Scholar] [CrossRef]
- Abdelazim, A.; Abourehab, M.; Abd Elhalim, L.; Almrasy, A.; Ramzy, S. Green adherent spectrophotometric determination of molnupiravir based on computational calculations; application to a recently FDA-approved pharmaceutical dosage form. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2023, 285, 121911, ISSN 1386-1425. [Google Scholar] [CrossRef]
- Pallavi, M.; Kumar, S.; Baggi, T. Analytical methods for tramadol in pharmaceutical and forensic context—A review. Int. J. Med. Toxicol. Leg. Med. 2023, 26, 122–147. [Google Scholar] [CrossRef]
- Chen, C.; Wang, C.; Ko, P.; Chen, Y. Nanomaterial-based adsorbents and optical sensors for illicit drug analysis. J. Food Drug Anal. 2020, 28, 654–676. [Google Scholar] [CrossRef] [PubMed]
- Jang, S.; Uk Son, S.; Kang, B.; Kim, J.; Lim, J.; Seo, S.; Kang, T.; Jung, J.; Lee, K.; Kim, H.; et al. Electrospun Nanofibrous Membrane-Based Colorimetric Device for Rapid and Simple Screening of Amphetamine-Type Stimulants in Drinks. Anal. Chem. 2022, 94, 3535–3542. [Google Scholar] [CrossRef] [PubMed]
- Akula, G.; Sapavatu, S.; Jadi, R.; Battineni, J.; Boggula, N. Analytical method development and validation for the estimation of tramadol in bulk and its formulations by UV-spectroscopy. J. Adv. Sci. Res. 2021, 12, 77–83. [Google Scholar] [CrossRef]
- Razlansari, M.; Ulucan-Karnak, F.; Kahrizi, M.; Mirinejad, S.; Sargazi, S.; Mishra, S.; Rahdar, A.; Diez-Pascual, A. Nanobiosensors for detection of opioids: A review of latest advancements. Eur. J. Pharm. Biopharm. 2022, 179, 79–94, ISSN 0939-6411. [Google Scholar] [CrossRef]
- Siddiqui, J.; Taheri, M.; Alam, A.U.; Deen, M.J. Nanomaterials in Smart Packaging Applications: A Review. Small 2022, 18, 2101171. [Google Scholar] [CrossRef] [PubMed]
- Mohammadpour, Z.; Naghib, S. Smart nanosensors for intelligent packaging. In Nanosensors for Smart Manufacturing; Elsevier: Amsterdam, The Netherlands, 2021; pp. 323–346. ISBN 9780128233580. [Google Scholar] [CrossRef]
- Tan, Z.; Zhu, C.; Han, L.; Liao, X.; Wang, C. SERS and dark-field scattering dual-mode detection of intracellular hydrogen peroxide using biocompatible Au@ COF nanosensor. Sens. Actuators B Chem. 2022, 373, 132770, ISSN 0925-4005. [Google Scholar] [CrossRef]
- Negut, I.; Bita, B. Polymeric Micellar Systems—A Special Emphasis on “Smart” Drug Delivery. Pharmaceutics 2023, 15, 976. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wang, P. Point-of-care drug of abuse testing in the opioid epidemic. Arch. Pathol. Lab. Med. 2020, 144, 1325–1334. [Google Scholar] [CrossRef] [PubMed]
- Singh, N.K.; Sidhu, G.K.; Gupta, K. Current and Future Perspective of Devices and Diagnostics for Opioid and OIRD. Biomedicines 2022, 10, 743. [Google Scholar] [CrossRef] [PubMed]
- Canfield, J.R.; Agarwal, S.; Fortener, S.K.; Sprague, J.E. Fentanyl Detection Using Eosin Y Paper Assays. J. Forensic Sci. 2020, 65, 1432–1442. [Google Scholar] [CrossRef] [PubMed]
- Lin, Y.; Sun, J.; Tang, M.; Zhang, G.; Yu, L.; Zhao, X.; He, Y. Synergistic recognition-triggered charge transfer enables rapid visual colorimetric detection of fentanyl. Anal. Chem. 2020, 93, 6544–6550. [Google Scholar] [CrossRef] [PubMed]
- Qu, F.; Lin, L.; Nie, P.; Xia, Z. High-Precision Automatic Identification of Fentanyl-Related Drugs by Terahertz Spectroscopy with Molecular Dynamics Simulation and Spectral Similarity Mapping. Int. J. Mol. Sci. 2022, 23, 10321. [Google Scholar] [CrossRef] [PubMed]
- Purohit, S.; Pandey, G.; Tharmavaram, M.; Rawtani, D.; Mustansar Hussain, C. Sensors for the Detection of Illicit Drugs. Technol. Forensic Sci. Sampl. Anal. Data Regul. 2020, 199, 21–238. [Google Scholar] [CrossRef]
- Usman, M.; Baig, Y.; Nardiello, D.; Quinto, M. How new nanotechnologies are changing the opioid analysis scenery? A comparison with classical analytical methods. Forensic Sci. Res. 2024, 9, owae001. [Google Scholar] [CrossRef]
- Wu, Y.; Feng, J.; Hu, G.; Zhang, E.; Yu, H.-H. Colorimetric Sensors for Chemical and Biological Sensing Applications. Sensors 2023, 23, 2749. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, C.H.P.; de Mascarenhas, R.O.; Bruni, A.T. In Silico Infrared Spectroscopy as a Benchmark for Identifying Seized Samples Suspected of Being N-Ethylpentylone. Psychoactives 2023, 2, 1–22. [Google Scholar] [CrossRef]
- Shin, Y.-H.; Wing-Gutierrez, M.T.; Choi, J.W. Review- Recent Progress in Portable Fluorescence Sensors. J. Electrochem. Soc. 2021, 168, 017502. [Google Scholar] [CrossRef]
- Ghose, R.; Forati, A.M.; Mantsch, J.R. Impact of the COVID-19 Pandemic on Opioid Overdose Deaths: A Spatiotemporal Analysis. J. Urban Health 2022, 99, 316–327. [Google Scholar] [CrossRef] [PubMed]
- Richards, N.; Fried, M.; Svirsky, L.; Thomas, N.; Zettler, P.J.; Howards, D. Clinicians Perspectives on Opioid Treatment Agreements: A Qualitative Analysis of Focus Groups. AJOB Empir. Bioeth. 2023, 2023, 2274606. [Google Scholar] [CrossRef] [PubMed]
- Meng, L.F.; Wu, H.Z. Carbon Quantum Dots from Carrot as Fluorescence Probes for High-Sensitivity Detection of Lidocaine. Appl. Ecol. Environ. Res. 2023, 21, 4531–4544. [Google Scholar] [CrossRef]
- Garcia de Arquer, P.; Talapin, D.V.; Klimov, V.I.; Arakawa, Y.; Bayer, M.; Sargent, E.H. Semiconductor quantum dots: Technological progress and future challenges. Science 2021, 373, eaaz8541. [Google Scholar] [CrossRef]
- Bahar, E.; Yoon, H. Lidocaine: A Local Anesthetic, Its Adverse Effects and Management. Medicina 2021, 57, 782. [Google Scholar] [CrossRef] [PubMed]
- Gan, Y.X.; Jayatissa, A.H.; Yu, Z.; Chen, X.; Li, M. Hydrothermal Synthesis of Nanomaterials. J. Nanomater. 2020, 2020, 891703. [Google Scholar] [CrossRef]
- Sarigul, N.; Korkmaz, F.; Kurultak, I. A New Artificial Urine Protocol to Better Imitate Human Urine. Sci. Rep. 2019, 9, 20159. [Google Scholar] [CrossRef] [PubMed]
- Shang, Y.F.; Shen, Y.Y.; Zhang, M.C.; Lv, C.M.; Wang, T.Y.; Chen, X.Q.; Lin, J. Progress in salivary glands: Endocrine glands with immune functions. Sec. Exp. Endocrinol. 2023, 14, 1061235. [Google Scholar] [CrossRef] [PubMed]
- Aydin, B.E.; Aydin, M.; Sezgintük, M.K. Chapter One—Biosensors for Saliva Biomarkers. Adv. Clin. Chem. 2023, 113, 1–41. [Google Scholar] [PubMed]
- U.S. Government. Part 40 Final Rule—Dot Summary of Changes. U.S. Department of Transportation. Available online: https://www.transportation.gov/odapc/Notice_Summary_May_2023 (accessed on 10 February 2024).
- Kanter, K.; Gallagher, R.; Eweje, F.; Lee, A.; Gordon, D.; Landy, S.; Gasior, J.; Soto-Calderon, H.; Cronholm, P.F.; Cocchiaro, B.; et al. Willingness to use a wearable device capable of detecting and reversing overdose among people who use opioids in Philadelphia. Harm Reduct. J. 2021, 18, 75. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Cao, X.; Deng, Y.; Ji, X.; Sun, W.; Xia, S.; Wan, S.; Zhang, H.; Xing, R.; Ding, J.; et al. An overview on recent advances of reversible fluorescent probes and their biological applications. Talanta 2024, 268, 125275. [Google Scholar] [CrossRef]
- Spencer, R.M.; Garnett, M.F.; Miniño, A.M. Urban-Rural Differences in Drug Overdoses Death Rates, 2020. NCHS Data Brief No. 440 2022, 440. [Google Scholar]
- Loch, A.S.; Burn, P.L.; Shaw, P.E. Fluorescent sensors for the detection of free-base illicit drugs—Effect of tuning the electronic properties. Sens. Actuators B Chem. 2023, 387, 133766. [Google Scholar] [CrossRef]
- Lawson, H.D.; Walton, P.S.; Chan, C. Metal-Organic Frameworks for Drug Delivery: A Design Perspective. ACS Appl. Mater. Interfaces 2021, 13, 7004–7020. [Google Scholar] [CrossRef] [PubMed]
- Heinz, K.; Rogge, S.M.J.; Kalytta-Mewes, A.; Volkmer, D.; Bunzen, H. MOFs for long-term gas storage: Exploiting kinetic trapping in ZIF-8 for on-demand and stimuli-controlled gas release. Inor. Chem. Front. 2023, 10, 4763–4772. [Google Scholar] [CrossRef]
- Qian, Q.; Asinger, P.A.; Lee, M.J.; Ha, G.; Rodriguez, K.M.; Lin, S.; Benedetti, F.M.; Wu, A.X.; Chi, W.S.; Smith, Z.P. MOF-Bases Membranes for Gas Separations. Chem. Rev. 2020, 120, 8161–8266. [Google Scholar] [CrossRef] [PubMed]
- Rosen, A.S.; Fung, V.; Huck, P.; O’Donnell, C.T.; Horton, M.K.; Truhlar, D.G.; Persson, K.A.; Notestein, J.M.; Snurr, R.Q. High-throughput predictions of metal-organic framework electronic properties: Theoretical challenges, graph neural networks, and data exploration. Comput. Mater. 2022, 8, 112. [Google Scholar] [CrossRef]
- Wu, Z.; Zhou, H.; Han, Q.; Lin, X.; Han, D.; Li, X. A cost-effective fluorescence biosensor for cocaine based on a “mix-and-detect” strategy. Analyst 2020, 145, 4664–4670. [Google Scholar] [CrossRef] [PubMed]
- Ju, L.; Lyuy, A.; Hao, H.; Shen, W.; Cui, H. Deep Learning-Assisted Three-Dimensional Fluorescence Difference Spectroscopy for Identification and Semiquantification of Illicit Drugs in Biofluids. Anal. Chem. 2019, 91, 9343–9347. [Google Scholar] [CrossRef]
- Judd, D.; King, C.R.; Galke, C. The Opioid Epidemic: A Review of the Contributing Factors, Negative Consequences, and Best Practices. Cureus 2023, 15, e41621. [Google Scholar] [CrossRef] [PubMed]
- Beatty, Z.G.; Muthusamy, A.K.; Unger, E.K.; Doubhberty, D.A.; Tian, L.; Looger, L.L.; Shivange, A.V.; Bera, K.; Lester, H.A.; Nichols, A.L. Fluorescence Screens for Identifying Central Nervous System-Acting Drug-Biosensor Pairs for Subcellular and Supracellular Pharmacokinetics. Bio-Protocol 2022, 12, e4551. [Google Scholar] [CrossRef]
- Rappleye, M.; Wait, S.J.; Lee, D.J.; Siebart, J.C.; Torp, L.; Smith, N.; Muster, J.; Matreyek, K.A.; Fowler, D.M.; Brendt, A. Optogenetics Microwell Array Screening System: A High-Throughput Engineering Platform for Genetically Encoded Fluorescent Indicators. ACS Sens. 2023, 8, 4233–4244. [Google Scholar] [CrossRef] [PubMed]
- Itai, D.; Vanle, B.; Groningen, V.; Ishak, W.; Nuckols, T. Opioid Overdose in the Hospital Setting: A systematic review. J. Addict. Med. 2020, 14, 39–47. [Google Scholar] [CrossRef]
- Hoots, B.E. Opioid Overdose Surveillance: Improving Data to Inform Action. Public Health Rep. 2021, 136 (Suppl. 1), 5S. [Google Scholar] [CrossRef]
- Wilke, C. Portable Spectrometers Give On-Site Drug Testing a Boost. ACS Centr. Sci. 2023, 9, 124–127. [Google Scholar] [CrossRef] [PubMed]
- D’Orsogna, M.R.; Böttcher, L.; Chou, T. Fentanyl-driven acceleration of racial, gender and geographical disparities in drug overdose deaths in the United States. PLoS Glob. Public Health 2023, 3, e0000769. [Google Scholar] [CrossRef] [PubMed]
- Hassan, M.H.; Khan, R.; Andreescu, S. Advances in electrochemical detection methods for measuring contaminants of emerging concerns. Electrochem. Sci. Adv. 2022, 2, e2100184. [Google Scholar] [CrossRef]
- Shi, Y.; Wu, W. Multimodal non-invasive non-pharmacological therapies for chronic pain: Mechanisms and progress. BMC Med. 2023, 21, 372. [Google Scholar] [CrossRef] [PubMed]



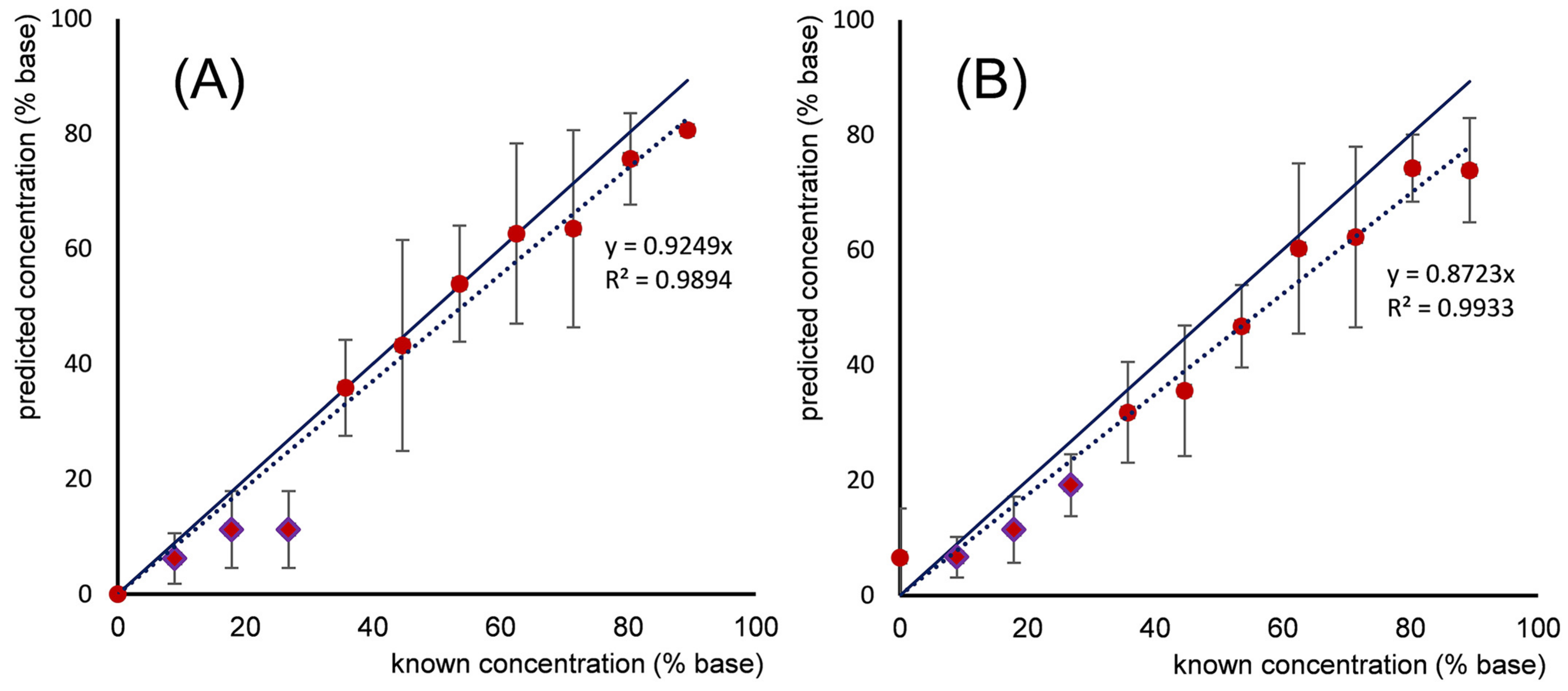

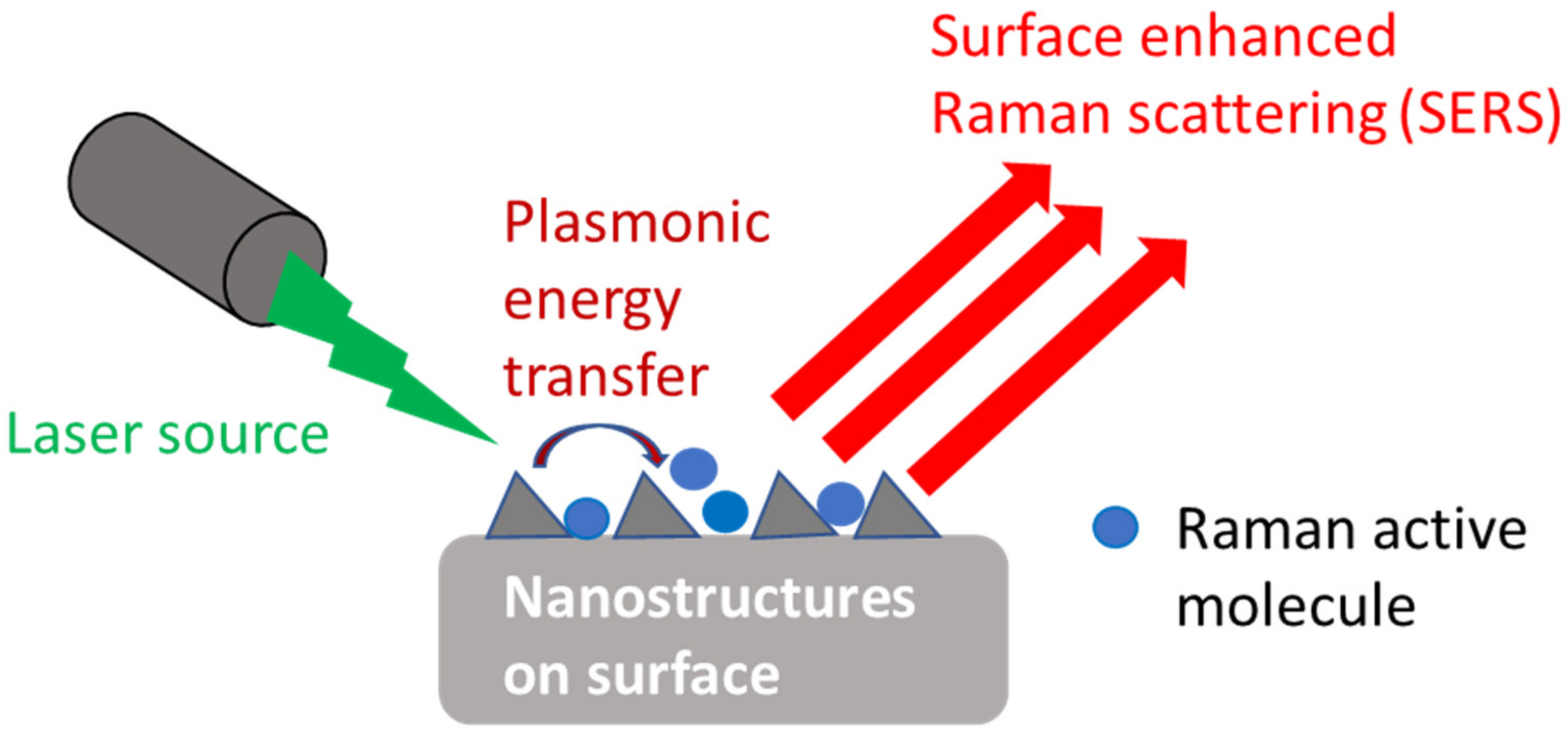

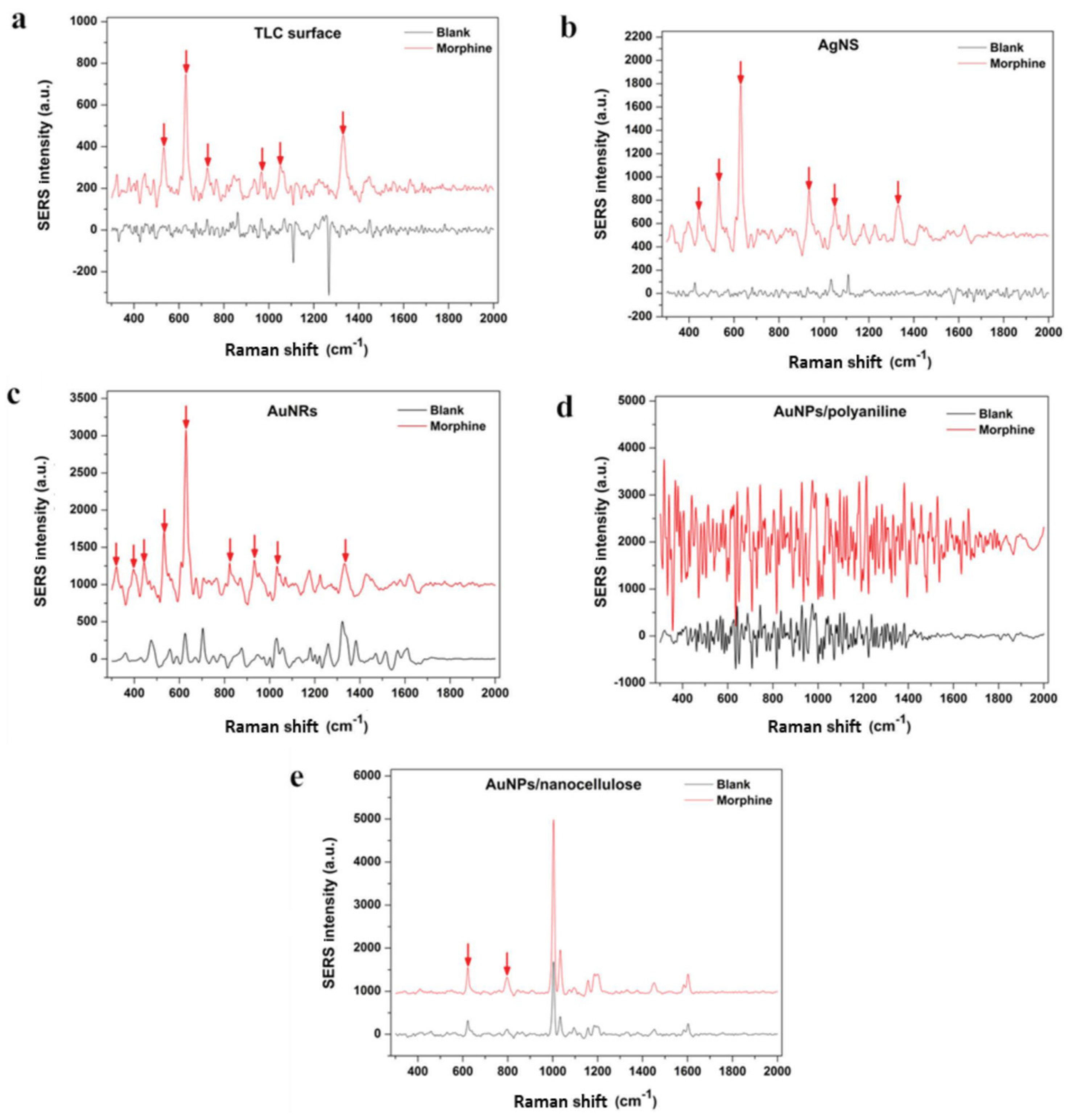
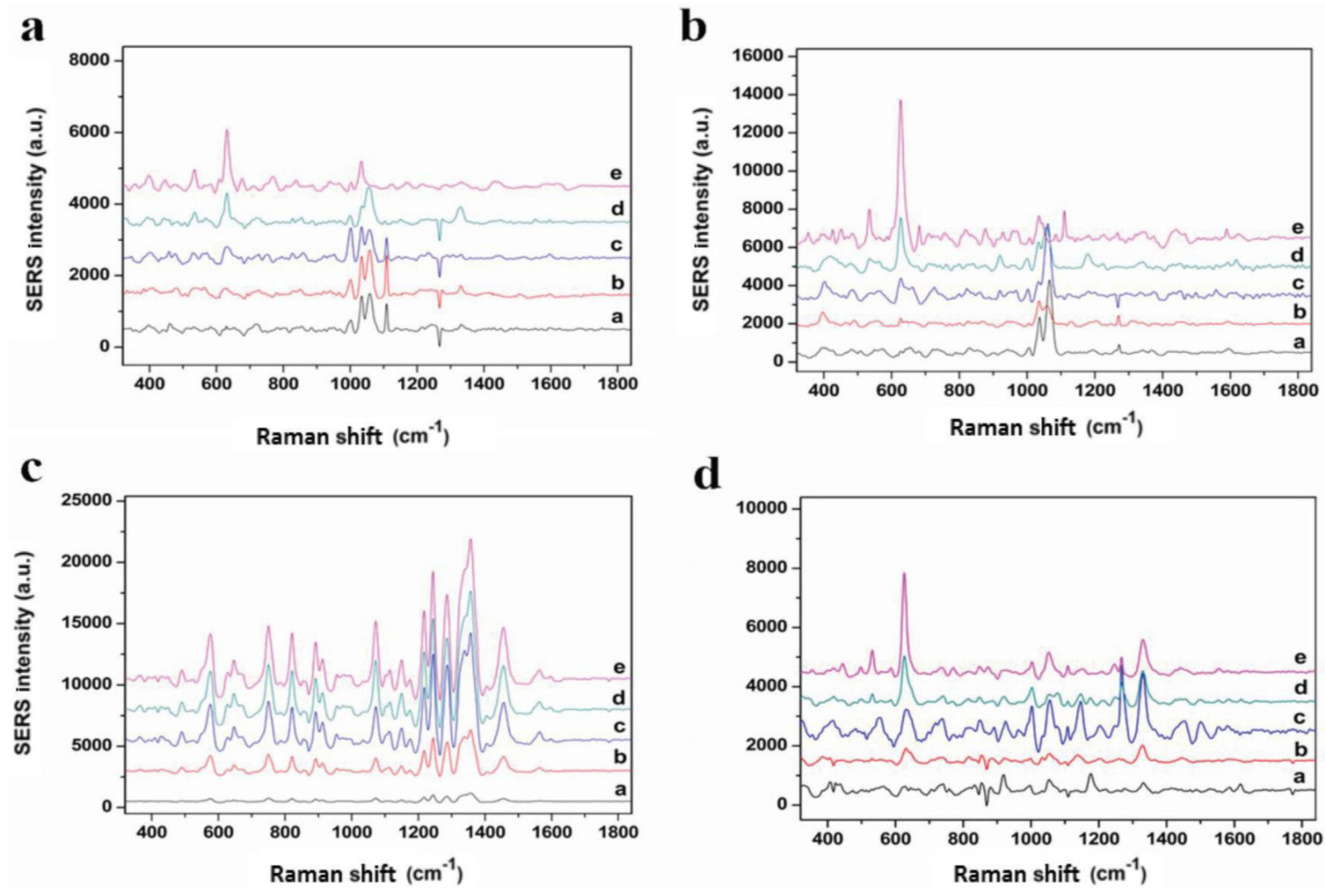


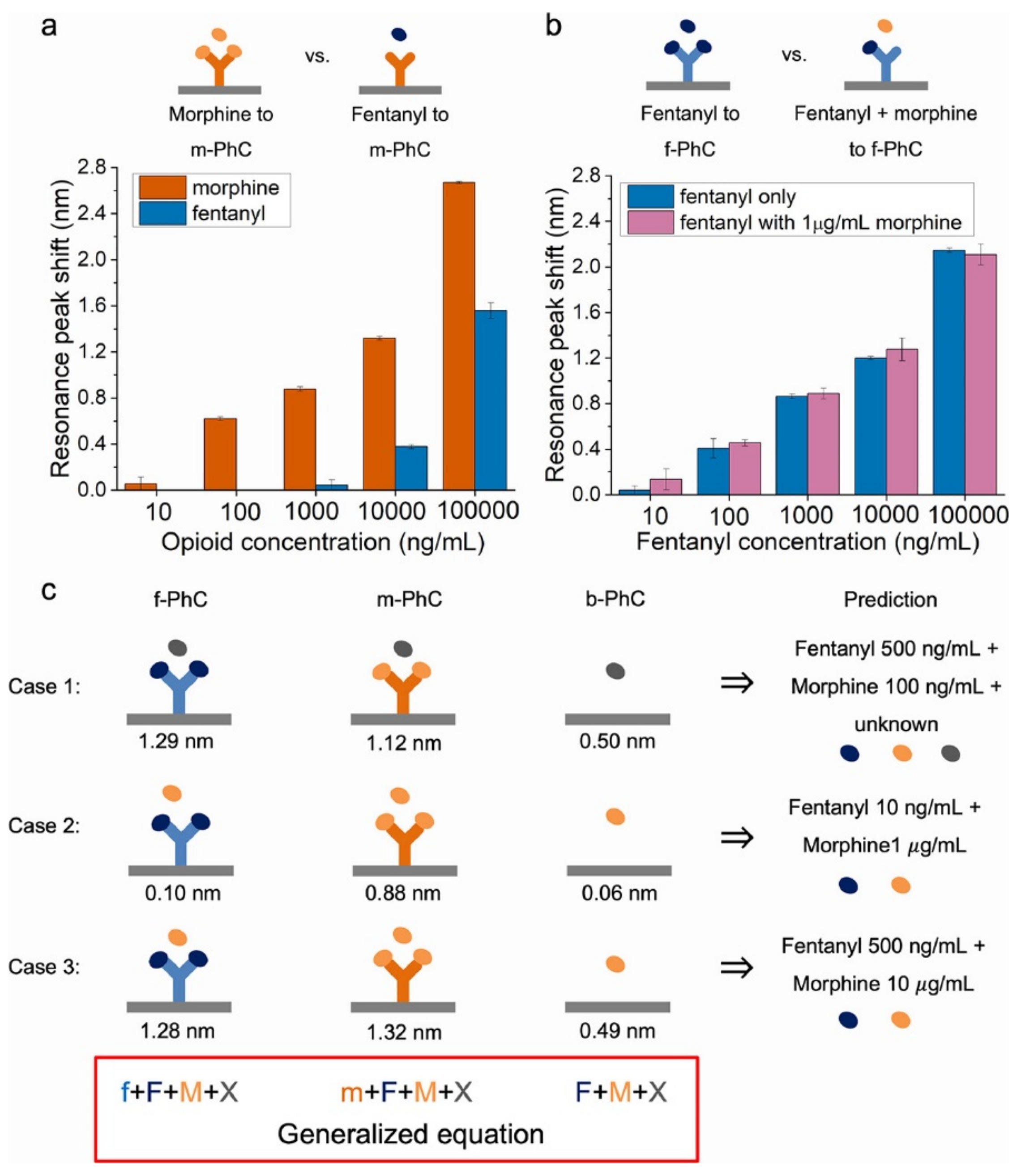
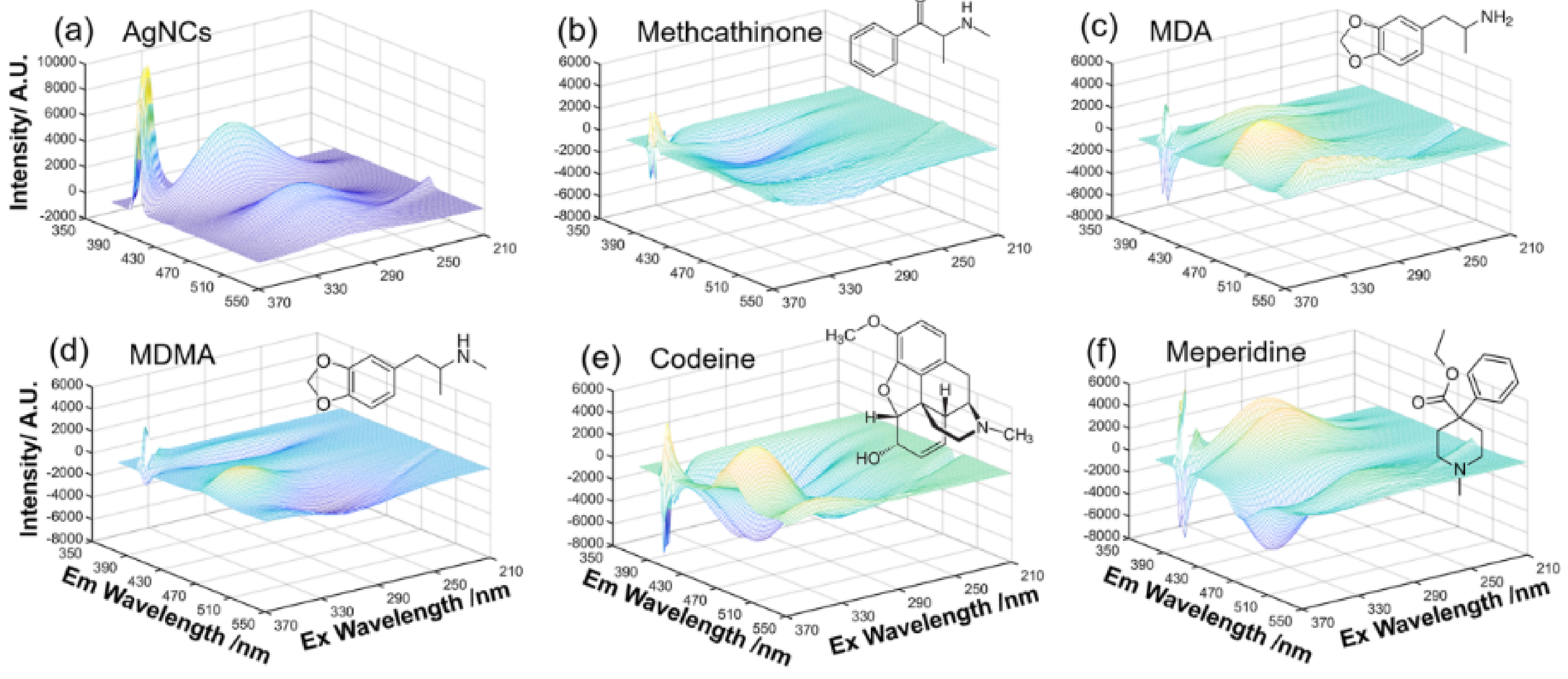
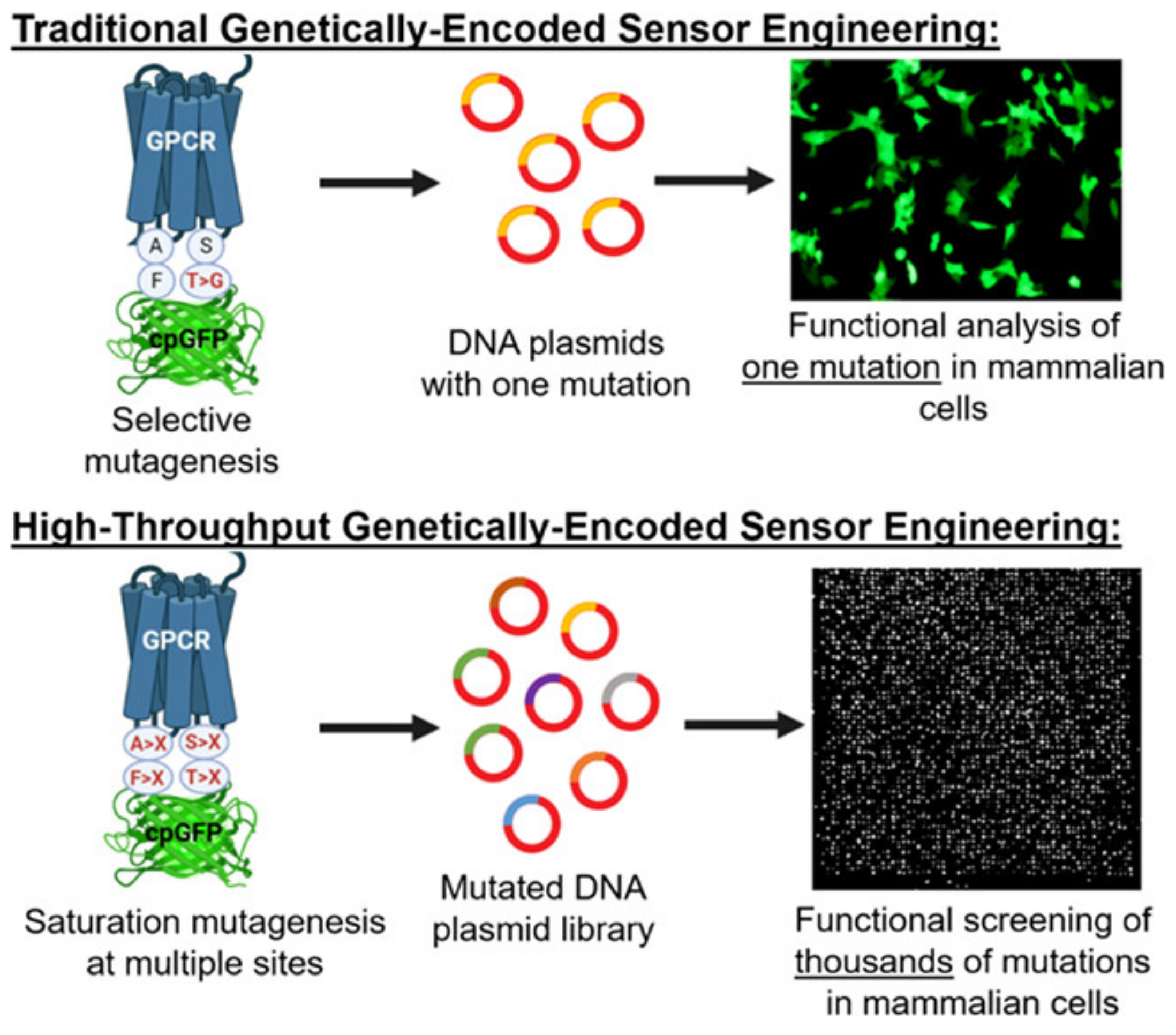
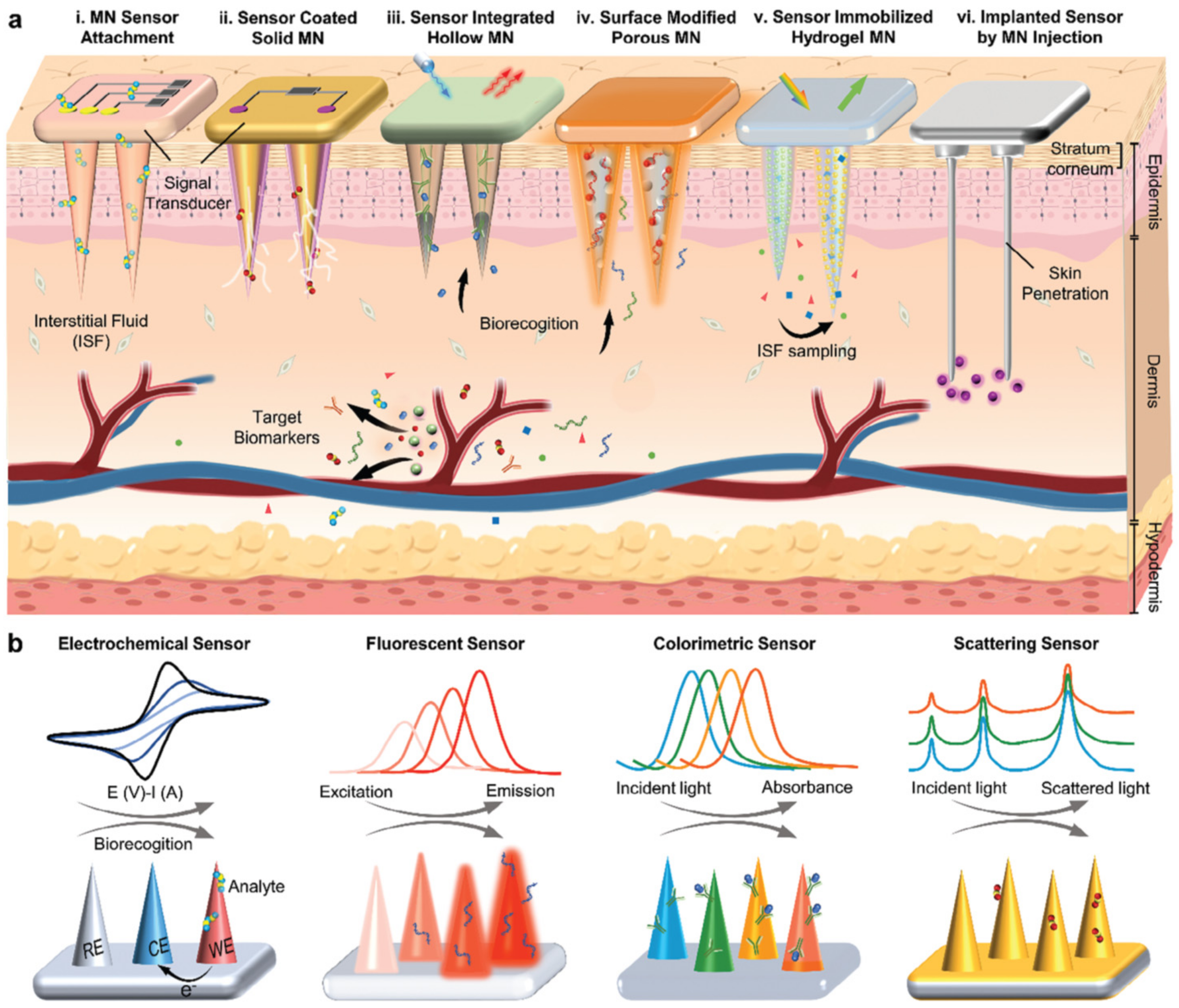
| Electrode Type | Electrode Modification | Electrochemical Method | Target Analyte | Linear Range | LOD | R2 | Ref. |
|---|---|---|---|---|---|---|---|
| Biological Applications | |||||||
| Glassy carbon | PMF/GO nanocomposite | Differential pulse voltammetry | Oxycodone | 0.01–45 µmol·L−1 | 2.0 nmol·L−1 | 0.994 | [81] |
| Glassy carbon | (Gr/AgNPs)2/GCE nanocomposite | Differential pulse voltammetry | Methadone | 1.0–200 µM | 0.18 µM | 0.998 | [82] |
| Glassy carbon | GCE/CMK-5 | Square wave voltammetry | Methadone and morphine | 0.1 to 4 µM | 0.027 µM | 0.948 and 0.990 | [83] |
| Glassy carbon | Poly(CTAB)/GO | Differential pulse voltammetry | Morphine | 50 nM–60 µM | 0.36 µM | 0.990 | [84] |
| Graphite screen printed | GO/Fe3O4@SiO2 | Differential pulse voltammetry | Morphine | 1.0–100 µM | 7.5 × 10−7 M | 0.9986 | [85] |
| Screen printed glassy electrode | Graphene–Co3O4 nanocomposite | Differential pulse voltammetry | Morphine and diclofenac | 2.5–550 µM and 2.5–700 µM | 0.007 µM | [86] | |
| Screen printed electrode | La3+/ZnO nanoflowers and MWCNTs | Differential pulse voltammetry | Tramadol | 0.5–800 µM | 0.08 µM | 0.9992 | [87] |
| Platinum (Pt) electrode | Pt/PEDOT/FC1/PEDOT…SDS | Cyclic voltammetry | Tramadol | 7–300 µM 5–280 µM | 18.6 nM 16 nM | [88] | |
| Laser carbonized polyimide electrode (LCE) | None | Square-wave voltammetry | Fentanyl | 20–200 µM | 1 µM | [89] | |
| Glassy carbon | CoO@f-CNTs nanocomposite | Amperometry | Tramadol | 1–300 mM | 6 nM | 0.9998 | [90] |
| Pharmaceutical Applications | |||||||
| Screen-printed | MWCNTs | Potentiometric | Pholcodine | 5.5 × 10−6 to 1.0 × 10−2 M | 2.5 × 10−7 M | 0.9980 | [91] |
| Glassy carbon electrode | MWCNTs | Differential pulse adsorptive stripping voltammetry | Fentanyl | 5 × 10−7 to 1 × 10−4 M | 1 × 10−7 M | [92] | |
| Carbon paste electrode | Au/NPs/BMTCB/CPE | Square-wave voltammetry | Tramadol | 0.01–400 µM | 6 nM | 0.993 | [93] |
| Glass carbon electrode | UiO-66-NH2MOF/G3-PAMAM nanocomposite | Differential pulse voltammetry | Tramadol in acetaminophen | 0.5–500 µM | 0.2 µM | 0.9997 | [94] |
| Screen-printed electrode | Yb2O3 nanoplate | Cyclic voltammetry and differential pulse voltammetry | Acetaminophen and tramadol—mixed and separate | 0.25–654 µmol·L−1 and 0.50–115 µmol·L−1 | 55 and 87 nmol·L−1 | [95] | |
| Activated screen-printed electrode | aSPCE/SDS | Voltammetry | Paracetamol (PA), diclofenac (DF), and tramadol (TR) | 5.0 × 0−8–2.0 × 10−5 mol·L−1 for PA; 1.0 × 10−9–2.0 × 10−7 mol·L−1 for DF; 1.0 × 10−8–2.0 × 10−7 mol·L−1 and 2.0 × 10−7–2.0 × 10−6 mol·L−1 for TR | 14.87, 0.21, and 1.71 nmol·L−1 | [96] | |
| Boron-doped diamond (BDD) electrode | None | Square wave voltammetry | Tramadol | 0.25–50 µg·mL−1 | 0.072 µg.mL−1 | [97] | |
| Clinical Applications | |||||||
| Microneedle sensor array platform | 4-(3-butyl-1-imidazolio)-1-butanesulfonate | Square wave voltammetry | Fentanyl | 0–160 µM | 27.8 µM | [78] | |
| Disc electrode with a Pt-Ir wire | None | Amperometry | Heroin and cocaine | 0–20 µM | Not reported | [98] | |
| Forensic Applications | |||||||
| Carbon screen-printed electrode | MoWS2 nanopetals | Differential pulse voltammetry | Morphine in tramadol | 4.8 × 10−8 M–5.05 × 10−4 M | 1.44 × 10−8 M | [99] | |
| Carbon paste electrode | CuO nanostructures and MWCNTs | Square wave voltammetry | Tramadol | 0.05–200 µM | 0.025 µM | [100] | |
| 3D-printed support | None | Electrified liquid–liquid interface (eLLI) miniaturization | Heroin (by itself and with cutting agents) | 0–50 µM | 1.3 µM | [101] | |
| Fentanyl Immunoassay Strip Result | ||
|---|---|---|
| Positive (n = 6099) | Negative (n = 6585) | |
| Year | ||
| 2018 | 519 (8.5) | 394 (6.0) |
| 2019 | 2473 (40.5) | 1718 (26.1) |
| 2020 | 2723 (44.6) | 3960 (60.1) |
| 2021 | 384 (6.3) | 513 (7.8) |
| Health Authority Region | ||
| Fraser | 533 (8.7) | 194 (2.9) |
| Interior | 359 (5.9) | 445 (6.8) |
| Vancouver Coastal | 5161 (84.6) | 5931 (90.1) |
| Vancouver Island | 46 (0.8) | 15 (0.2) |
| Color | ||
| Black | 72 (1.2 | 28 (0.4) |
| Blue | 413 (6.8) | 138 (2.1) |
| Brown | 1173 (19.2) | 769 (11.7) |
| Colorless | 25 (0.4) | 1009 (15.3) |
| Green | 1229 (20.2) | 143 (2.2) |
| Grey | 292 (4.8) | 195 (3.0) |
| Orange | 217 (3.6) | 116 (1.8) |
| Pink | 368 (6.0) | 175 (2.7) |
| Purple | 1400 (23.0) | 167 (2.5) |
| Red | 140 (6.2) | 34 (0.5) |
| White | 380 (5.8) | 3552 (53.9) |
| Yellow | 351 (5.8) | 227 (3.4) |
| Other | 5 (0.1) | 0 (0.0) |
| Not reported | 34 (0.6) | 32 (0.5) |
| Texture | ||
| Chunk | 1471 (24.1) | 567 (8.6) |
| Crystal | 48 (0.8) | 2264 (34.4) |
| Flake | 18 (0.3) | 68 (1.0) |
| Granules | 533 (8.7) | 151 (2.3) |
| Liquid | 20 (0.3) | 110 (1.7) |
| Paste | 145 (2.4) | 49 (0.7) |
| Pebble | 3010 (49.4) | 271 (4.1) |
| Powder | 704 (11.5) | 2483 (37.7) |
| Pressed tablet | 7 (0.1) | 170 (2.6) |
| Residue | 65 (1.1) | 19 (0.3) |
| Tablet (pharmaceutical) | 65 (0.4) | 368 (5.6) |
| Other | 3 (0.05) | 28 (0.4) |
| Not reported | 23 (0.9) 37 | 37 (0.6) |
| F1 Score | Accuracy | Precision | Recall | |
|---|---|---|---|---|
| Neural network model | 96.4 | 96.4 | 95.7 | 97.1 |
| % (95%CI) | (95.6–97.1) | (95.6–97.1) | (94.5–96.7) | (96.1–98.0) |
| Drug-checking technician | 78.4 | 82.4 | 99.4 | 64.7 |
| % (95%CI) | (76.4–80.3) | (80.9–83.9) | (98.8–99.9) | (62.1–67.3) |
| CI: confidence interval | ||||
| Type | Color Test Name | Target Compounds |
|---|---|---|
| General Screening | Marquis | Alkaloids and other compounds (e.g., opiates, amphetamines, and phenthylamines) |
| Liebermann’s | Alkaloids and other compounds (e.g., amphetamines, and some synthetic cathinones) | |
| Mandelin’s | Alkaloids and other compounds (e.g., amphetamines, some phenthylamines, and ketamine) | |
| Mecke | Alkaloids and other compounds (e.g., amphetamines, and some synthetic cathinones) | |
| Froehde’s | Alkaloids and other compounds (e.g., opiates, and some synthetic cathinones) | |
| Drug Class Selective | Duquenois–Levine | Δ- 9Tetrahydrocannabinol (THC) and other cannabinoids |
| Scott’s | Cocaine | |
| Dille–Koppanyi | Barbiturates | |
| Chen-Kao | Ephedrine | |
| Ehrlich’s | Ergot alkaloids, indoles, aromatic amines, and LSD | |
| Fast Blue B salt | Δ-9 Tetrahydrocannabinol (THC) and other cannabinoids | |
| Zwikker | Barbiturates | |
| Functional Group Selective | Simon’s | Secondary amines (e.g., methamphetamine) |
| Zimmermann | Ketones (e.g., synthetic cathinones and benzodiazepines) |
| Illicit Drugs | LC-MS Analysis: Concentration (µg/mL) | FL-DL Analysis: Concentration (µg/mL) |
|---|---|---|
| Codeine | 3.12 ± 0.10 | 2.95 ± 0.81 |
| MDA | 2.55 ± 0.05 | 2.13 ± 1.15 |
| MDMA | 6.96 ± 0.11 | 8.91 ± 4.47 |
| Meperidine | 2.13 ± 0.08 | 3.24 ± 1.39 |
| Methcathinone | 8.36 ± 0.11 | 11.22 ± 3.36 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fakayode, S.O.; Brady, P.N.; Grant, C.; Fernand Narcisse, V.; Rosado Flores, P.; Lisse, C.H.; Bwambok, D.K. Electrochemical Sensors, Biosensors, and Optical Sensors for the Detection of Opioids and Their Analogs: Pharmaceutical, Clinical, and Forensic Applications. Chemosensors 2024, 12, 58. https://doi.org/10.3390/chemosensors12040058
Fakayode SO, Brady PN, Grant C, Fernand Narcisse V, Rosado Flores P, Lisse CH, Bwambok DK. Electrochemical Sensors, Biosensors, and Optical Sensors for the Detection of Opioids and Their Analogs: Pharmaceutical, Clinical, and Forensic Applications. Chemosensors. 2024; 12(4):58. https://doi.org/10.3390/chemosensors12040058
Chicago/Turabian StyleFakayode, Sayo O., Pamela Nicole Brady, Cidya Grant, Vivian Fernand Narcisse, Peter Rosado Flores, Catrena Higginbothan Lisse, and David K. Bwambok. 2024. "Electrochemical Sensors, Biosensors, and Optical Sensors for the Detection of Opioids and Their Analogs: Pharmaceutical, Clinical, and Forensic Applications" Chemosensors 12, no. 4: 58. https://doi.org/10.3390/chemosensors12040058
APA StyleFakayode, S. O., Brady, P. N., Grant, C., Fernand Narcisse, V., Rosado Flores, P., Lisse, C. H., & Bwambok, D. K. (2024). Electrochemical Sensors, Biosensors, and Optical Sensors for the Detection of Opioids and Their Analogs: Pharmaceutical, Clinical, and Forensic Applications. Chemosensors, 12(4), 58. https://doi.org/10.3390/chemosensors12040058






