Pathophysiological Role of Chymase-Activated Matrix Metalloproteinase-9
Abstract
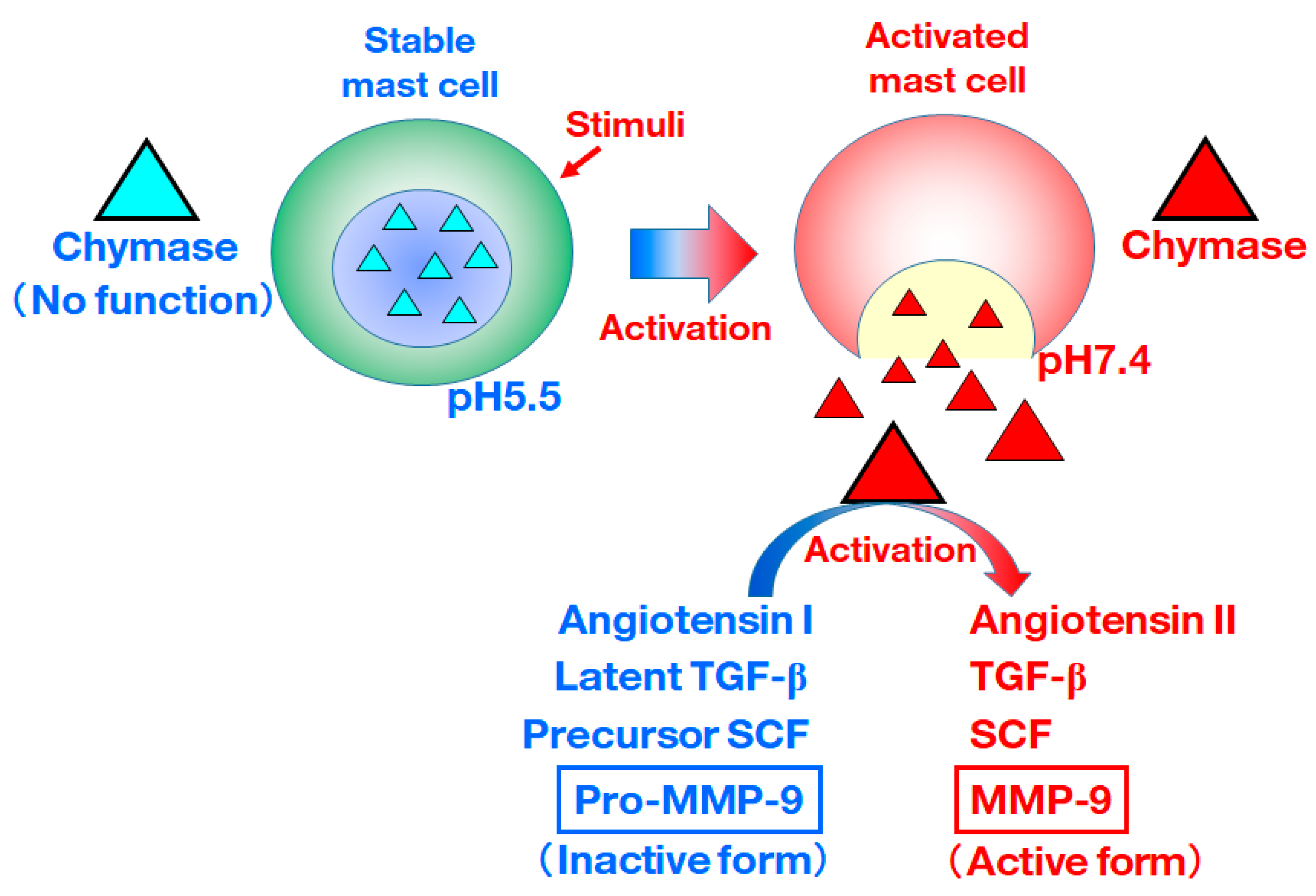
:1. Introduction
2. Relationship between MMP-9 and Various Diseases
3. Cardiovascular Diseases
3.1. Aneurysm
3.2. Myocardial Infarction
4. Inflammatory Diseases
4.1. Inflammatory Gastrointestinal Diseases
4.2. Inflammatory Hepatic Diseases
4.3. Acute Pancreatic Failure
4.4. Atopic Diseases
4.5. Rheumatoid Arthritis
5. Cancer
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- De Young, M.B.; Nemeth, E.F.; Scarpa, A. Measurement of the internal pH of mast cell granules using microvolumetric fluorescence and isotopic techniques. Arch. Biochem. Biophys. 1987, 254, 222–233. [Google Scholar] [CrossRef]
- Takai, S.; Shiota, N.; Yamamoto, D.; Okunishi, H.; Miyazaki, M. Purification and characterization of angiotensin II-generating chymase from hamster cheek pouch. Life Sci. 1996, 58, 591–597. [Google Scholar] [CrossRef]
- Fang, K.C.; Raymond, W.W.; Blount, J.L.; Caughey, G.H. Dog mast cell alpha-chymase activates progelatinase B by cleaving the Phe88-Gln89 and Phe91-Glu92 bonds of the catalytic domain J. Biol. Chem. 1997, 272, 25628–25635. [Google Scholar] [CrossRef] [Green Version]
- Furubayashi, K.; Takai, S.; Jin, D.; Miyazaki, M.; Katsumata, T.; Inagaki, S.; Kimura, M.; Tanaka, K.; Nishimoto, M.; Fukumoto, H. Chymase activates promatrix metalloproteinase-9 in human abdominal aortic aneurysm. Clin. Chim. Acta 2008, 388, 214–216. [Google Scholar] [CrossRef] [PubMed]
- Tchougounova, E.; Lundequist, A.; Fajardo, I.; Winberg, J.O.; Abrink, M.; Pejler, G. A key role for mast cell chymase in the activation of pro-matrix metalloprotease-9 and pro-matrix metalloprotease-2. J. Biol. Chem. 2005, 280, 9291–9296. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rosenkranz, S. TGF-β1 and angiotensin networking in cardiac remodeling. Cardiovasc. Res. 2004, 63, 423–432. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Longley, B.J.; Tyrrell, L.; Lu, S.; Ma, Y.; Klump, V.; Murphy, G.F. Chronically KIT-stimulated clonally-derived human mast cells show heterogeneity in different tissue microenvironments. J. Investig. Dermatol. 1997, 108, 792–796. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rabkin, S.W. The role matrix metalloproteinases in the production of aortic aneurysm. Prog. Mol. Biol. Transl. Sci. 2017, 147, 239–265. [Google Scholar] [PubMed]
- Pyo, R.; Lee, J.K.; Shipley, J.M.; Curci, J.A.; Mao, D.; Ziporin, S.J.; Ennis, T.L.; Shapiro, S.D.; Senior, R.M.; Thompson, R.W. Targeted gene disruption of matrix metalloproteinase-9 (gelatinase B) suppresses development of experimental abdominal aortic aneurysms. J. Clin. Investig. 2000, 105, 1641–1649. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nishimoto, M.; Takai, S.; Fukumoto, H.; Tsunemi, K.; Yuda, A.; Sawada, Y.; Yamada, M.; Jin, D.; Sakaguchi, M.; Nishimoto, Y.; et al. Increased local angiotensin II formation in aneurysmal aorta. Life Sci. 2002, 71, 2195–2205. [Google Scholar] [CrossRef]
- Furubayashi, K.; Takai, S.; Jin, D.; Muramatsu, M.; Ibaraki, T.; Nishimoto, M.; Fukumoto, H.; Katsumata, T.; Miyazaki, M. The significance of chymase in the progression of abdominal aortic aneurysms in dogs. Hypertens. Res. 2007, 30, 349–357. [Google Scholar] [CrossRef] [Green Version]
- Inoue, N.; Muramatsu, M.; Jin, D.; Takai, S.; Hayashi, T.; Katayama, H.; Kitaura, Y.; Tamai, H.; Miyazaki, M. Effects of chymase inhibitor on angiotensin II-induced abdominal aortic aneurysm development in apolipoprotein E-deficient mice. Atherosclerosis 2009, 204, 359–364. [Google Scholar] [CrossRef]
- Tomimori, Y.; Manno, A.; Tanaka, T.; Futamura-Takahashi, J.; Muto, T.; Nagahira, K. ASB17061, a novel chymase inhibitor, prevented the development of angiotensin II-induced abdominal aortic aneurysm in apolipoprotein E-deficient mice. Eur. J. Pharmacol. 2019, 856, 172403. [Google Scholar] [CrossRef] [PubMed]
- Furukawa, H.; Wada, K.; Tada, Y.; Kuwabara, A.; Sato, H.; Ai, J.; Lawton, M.T.; Hashimoto, T. Mast cell promotes the development of intracranial aneurysm rupture. Stroke 2020, 51, 3332–3339. [Google Scholar] [CrossRef] [PubMed]
- Bederson, J.B.; Awad, I.A.; Wiebers, D.O.; Piepgras, D.; Haley, E.C., Jr.; Brott, T.; Hademenos, G.; Chyatte, D.; Rosenwasser, R.; Caroselli, C. Recommendations for the management of patients with unruptured intracranial aneurysms: A Statement for healthcare professionals from the Stroke Council of the American Heart Association. Stroke 2000, 31, 2742–2750. [Google Scholar] [CrossRef] [Green Version]
- Rojas, H.A.; Fernandes, K.S.D.S.; Ottone, M.R.; Magalhães, K.C.S.F.; Albuquerque, L.A.F.; Pereira, J.L.B.; Vieira-Júnior, G.; Sousa-Filho, J.L.; Costa, B.S.; Sandrim, V.C.; et al. Levels of MMP-9 in patients with intracranial aneurysm: Relation with risk factors, size and clinical presentation. Clin. Biochem. 2018, 55, 63–68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kushamae, M.; Miyata, H.; Shirai, M.; Shimizu, K.; Oka, M.; Koseki, H.; Abekura, Y.; Ono, I.; Nozaki, K.; Mizutani, T.; et al. Involvement of neutrophils in machineries underlying the rupture of intracranial aneurysms in rats. Sci. Rep. 2020, 10, 20004. [Google Scholar] [CrossRef] [PubMed]
- Simões, G.; Pereira, T.; Caseiro, A. Matrix metaloproteinases in vascular pathology. Microvasc. Res. 2022, 143, 104398. [Google Scholar] [CrossRef]
- Gisterå, A.; Hansson, G.K. The immunology of atherosclerosis. Nat. Rev. Nephrol. 2017, 13, 368–380. [Google Scholar] [CrossRef] [PubMed]
- Kaartinen, M.; van der Wal, A.C.; van der Loos, C.M.; Piek, J.J.; Koch, K.T.; Becker, A.E.; Kovanen, P.T. Mast cell infiltration in acute coronary syndromes: Implications for plaque rupture. J. Am. Coll. Cardiol. 1998, 32, 606–612. [Google Scholar] [CrossRef]
- Kai, H.; Ikeda, H.; Yasukawa, H.; Kai, M.; Seki, Y.; Kuwahara, F.; Ueno, T.; Sugi, K.; Imaizumi, T. Peripheral blood levels of matrix metalloproteases-2 and -9 are elevated in patients with acute coronary syndromes. J. Am. Coll. Cardiol. 1998, 32, 368–372. [Google Scholar] [CrossRef] [Green Version]
- Takai, S.; Jin, D.; Inagaki, S.; Yamamoto, D.; Tanaka, K.; Miyazaki, M. Significance of matrix metalloproteinase-9 in cardiac dysfunction during the very acute phase after myocardial infarction in hamsters. Eur. J. Pharmacol. 2007, 572, 57–60. [Google Scholar] [CrossRef]
- Jin, D.; Takai, S.; Nonaka, Y.; Yamazaki, S.; Fujiwara, M.; Nakamura, Y. A chymase inhibitory RNA aptamer improves cardiac function and survival after myocardial infarction. Mol. Ther. Nucleic Acids 2019, 14, 41–51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lindsey, M.L.; Escobar, G.P.; Dobrucki, L.W.; Goshorn, D.K.; Bouges, S.; Mingoia, J.T.; McClister, D.M., Jr.; Su, H.; Gannon, J.; MacGillivray, C.; et al. Matrix metalloproteinase-9 gene deletion facilitates angiogenesis after myocardial infarction. Am. J. Physiol. Heart Circ. Physiol. 2006, 290, H232–H239. [Google Scholar] [CrossRef] [Green Version]
- Oyamada, S.; Bianchi, C.; Takai, S.; Chu, L.M.; Sellke, F.W. Chymase inhibition reduces infarction and matrix metalloproteinase-9 activation and attenuates inflammation and fibrosis after acute myocardial ischemia/reperfusion. J. Pharmacol. Exp. Ther. 2011, 339, 143–151. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Liu, C.L.; Fang, W.; Zhang, X.; Yang, C.; Li, J.; Liu, J.; Sukhova, G.K.; Gurish, M.F.; Libby, P.; et al. Deficiency of mouse mast cell protease 4 mitigates cardiac dysfunctions in mice after myocardium infarction. Biochim. Biophys. Acta Mol. Basis Dis. 2019, 1865, 1170–1181. [Google Scholar] [CrossRef] [PubMed]
- Houde, M.; Schwertani, A.; Touil, H.; Desbiens, L.; Sarrhini, O.; Lecomte, R.; Lepage, M.; Gagnon, H.; Takai, S.; Pejler, G.; et al. Mouse mast cell protease 4 deletion protects heart function and survival after permanent myocardial infarction. Front. Pharmacol. 2018, 9, 868. [Google Scholar] [CrossRef] [Green Version]
- Kanemitsu, H.; Takai, S.; Tsuneyoshi, H.; Nishina, T.; Yoshikawa, K.; Miyazaki, M.; Ikeda, T.; Komeda, M. Chymase inhibition prevents cardiac fibrosis and dysfunction after myocardial infarction in rats. Hypertens. Res. 2006, 29, 57–64. [Google Scholar] [CrossRef] [Green Version]
- Juliano, G.R.; Skaf, M.F.; Ramalho, L.S.; Juliano, G.R.; Torquato, B.G.S.; Oliveira, M.S.; Oliveira, F.A.; Espíndula, A.P.; Cavellani, C.L.; Teixeira, V.P.A.; et al. Analysis of mast cells and myocardial fibrosis in autopsied patients with hypertensive heart disease. Rev. Port. Cardiol. 2020, 39, 89–96. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, T.; Wada, A.; Tsutamoto, T.; Ohnishi, M.; Isono, T.; Kinoshita, M. Chymase inhibition prevents cardiac fibrosis and improves diastolic dysfunction in the progression of heart failure. Circulation 2003, 107, 2555–2558. [Google Scholar] [CrossRef]
- Mori, N.; Sato, H.; Hayashibara, T.; Senba, M.; Geleziunas, R.; Wada, A.; Hirayama, T.; Yamamoto, N. Helicobacter pylori induces matrix metalloproteinase-9 through activation of nuclear factor κB. Gastroenterology 2003, 124, 983–992. [Google Scholar] [CrossRef] [PubMed]
- Matsuo, T.; Ikura, Y.; Ohsawa, M.; Ogami, M.; Kayo, S.; Yoshimi, N.; Hai, E.; Naruko, T.; Ohishi, M.; Higuchi, K.; et al. Mast cell chymase expression in Helicobacter pylori-associated gastritis. Histopathology 2003, 43, 538–549. [Google Scholar] [CrossRef] [PubMed]
- Fagundes, F.L.; Pereira, Q.C.; Zarricueta, M.L.; Dos Santos, R.C. Malvidin Protects against and Repairs Peptic Ulcers in Mice by Alleviating Oxidative Stress and Inflammation. Nutrients 2021, 13, 3312. [Google Scholar] [CrossRef] [PubMed]
- Chao, G.; Hong, X.; Zhang, S. Effects of mast cells induced by NSAIDs impair intestinal epithelial barrier function in vivo and in vitro. Inflammation 2021, 44, 1396–1404. [Google Scholar] [CrossRef]
- Kakimoto, K.; Takai, S.; Murano, M.; Ishida, K.; Yoda, Y.; Inoue, T.; Jin, D.; Umegaki, E.; Higuchi, K. Significance of chymase-dependent matrix metalloproteinase-9 activation on indomethacin-induced small intestinal damages in rats. J. Pharmacol. Exp. Ther. 2010, 332, 684–689. [Google Scholar] [CrossRef] [Green Version]
- Nakov, R. New markers in ulcerative colitis. Clin. Chim. Acta 2019, 497, 141–146. [Google Scholar] [CrossRef]
- Baugh, M.D.; Perry, M.J.; Hollander, A.P.; Davies, D.R.; Cross, S.S.; Lobo, A.J.; Taylor, C.J.; Evans, G.S. Matrix metalloproteinase levels are elevated in inflammatory bowel disease. Gastroenterology 1999, 117, 814–822. [Google Scholar] [CrossRef]
- Tarlton, J.F.; Whiting, C.V.; Tunmore, D.; Bregenholt, S.; Reimann, J.; Claesson, M.H.; Bland, P.W. The role of up-regulated serine proteases and matrix metalloproteinases in the pathogenesis of a murine model of colitis. Am. J. Pathol. 2000, 157, 1927–1935. [Google Scholar] [CrossRef] [Green Version]
- Castaneda, F.E.; Walia, B.; Vijay-Kumar, M.; Patel, N.R.; Roser, S.; Kolachala, V.L.; Rojas, M.; Wang, L.; Oprea, G.; Garg, P.; et al. Targeted deletion of metalloproteinase 9 attenuates experimental colitis in mice: Central role of epithelial-derived MMP. Gastroenterology 2005, 129, 1991–2008. [Google Scholar] [CrossRef]
- Garg, P.; Rojas, M.; Ravi, A.; Bockbrader, K.; Epstein, S.; Vijay-Kumar, M.; Gewirtz, A.T.; Merlin, D.; Sitaraman, S.V. Selective ablation of matrix metalloproteinase-2 exacerbates experimental colitis: Contrasting role of gelatinases in the pathogenesis of colitis. J. Immunol. 2006, 177, 4103–4112. [Google Scholar] [CrossRef]
- Ishida, K.; Takai, S.; Murano, M.; Nishikawa, T.; Inoue, T.; Murano, N.; Inoue, N.; Jin, D.; Umegaki, E.; Higuchi, K.; et al. Role of chymase-dependent matrix metalloproteinase-9 activation in mice with dextran sodium sulfate-induced colitis. J. Pharmacol. Exp. Ther. 2008, 324, 422–426. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Decker, K.; Keppler, D. Galactosamine hepatitis: Key role of the nucleotide deficiency period in the pathogenesis of cell injury and cell death. Rev Physiol. Biochem. Pharmacol. 1974, 71, 77–106. [Google Scholar]
- Leist, M.; Gantner, F.; Bohlinger, I.; Tiegs, G.; Germann, P.G.; Wendel, A. Tumor necrosis factor-induced hepatocyte apoptosis precedes liver failure in experimental murine shock models. Am. J. Pathol. 1995, 146, 1220–1234. [Google Scholar]
- Wielockx, B.; Lannoy, K.; Shapiro, S.D.; Itoh, T.; Itohara, S.; Vandekerckhove, J.; Libert, C. Inhibition of matrix metalloproteinases blocks lethal hepatitis and apoptosis induced by tumor necrosis factor and allows safe antitumor therapy. Nat. Med. 2001, 7, 1202–1208. [Google Scholar] [CrossRef]
- Yan, C.; Zhou, L.; Han, Y.P. Contribution of hepatic stellate cells and matrix metalloproteinase 9 in acute liver failure. Liver Int. 2008, 28, 959–971. [Google Scholar] [CrossRef] [PubMed]
- Imai, Y.; Takai, S.; Jin, D.; Komeda, K.; Tashiro, K.; Li, Z.L.; Otsuki, Y.; Okamura, H.; Hayashi, M.; Uchiyama, K. Chymase inhibition attenuates lipopolysaccharide/d-galactosamine-induced acute liver failure in hamsters. Pharmacology 2014, 93, 47–56. [Google Scholar] [CrossRef]
- Hashimoto, E.; Tokushige, K. Prevalence, gender, ethnic variations, and prognosis of NASH. J. Gastroenterol. 2011, 46 (Suppl. S1), 63–69. [Google Scholar] [CrossRef]
- Ekstedt, M.; Nasr, P.; Kechagias, S. Natural history of NAFLD/NASH. Curr. Hepatol. Rep. 2017, 16, 391–397. [Google Scholar] [CrossRef] [Green Version]
- Georgescu, E.F.; Ionescu, R.; Niculescu, M.; Mogoanta, L.; Vancica, L. Angiotensin receptor blockers as therapy for mild-to-moderate hypertension-associated non-alcoholic steatohepatitis. World J. Gastroenterol. 2009, 15, 942–954. [Google Scholar] [CrossRef]
- Park, H.; Hasegawa, G.; Shima, T.; Fukui, M.; Nakamura, N.; Yamaguchi, K.; Mitsuyoshi, H.; Minami, M.; Yasui, K.; Itoh, Y.; et al. The fatty acid composition of plasma cholesteryl esters and estimated desaturase activities in patients with nonalcoholic fatty liver disease and the effect of long-term ezetimibe therapy on these levels. Clin. Chim. Acta 2010, 411, 1735–1740. [Google Scholar] [CrossRef]
- Mahady, S.E.; Webster, A.C.; Walker, S.; Sanyal, A.; George, J. The role of thiazolidinediones in non-alcoholic steatohepatitis e a systematic review and meta analysis. J. Hepatol. 2010, 55, 1383–1390. [Google Scholar] [CrossRef] [PubMed]
- Tiniakos, D.G.; Vos, M.B.; Brunt, E.M. Nonalcoholic fatty liver disease: Pathology and pathogenesis. Annu. Rev. Pathol. 2010, 5, 145–171. [Google Scholar] [CrossRef] [PubMed]
- Miyaoka, Y.; Jin, D.; Tashiro, K.; Komeda, K.; Masubuchi, S.; Hirokawa, F.; Hayashi, M.; Takai, S.; Uchiyama, K. Chymase inhibitor prevents the development and progression of non-alcoholic steatohepatitis in rats fed a high-fat and high-cholesterol diet. J. Pharmacol. Sci. 2017, 134, 139–146. [Google Scholar] [CrossRef] [PubMed]
- Vege, S.S.; DiMagno, M.J.; Forsmark, C.E.; Martel, M.; Barkun, A.N. Initial Medical Treatment of Acute Pancreatitis: American Gastroenterological Association Institute Technical Review. Gastroenterology 2018, 154, 1103–1139. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crockett, S.D.; Wani, S.; Gardner, T.B.; Falck-Ytter, Y.; Barkun, A.N.; Crockett, S.; Falck-Ytter, Y.; Feuerstein, J.; Flamm, S.; Gellad, Z.; et al. American Gastroenterological Association Institute Guideline on Initial Management of Acute Pancreatitis. Gastroenterology 2018, 154, 1096–1101. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lopez-Font, I.; Gea-Sorlí, S.; de-Madaria, E.; Gutiérrez, L.M.; Pérez-Mateo, M.; Closa, D. Pancreatic and pulmonary mast cells activation during experimental acute pancreatitis. World J. Gastroenterol. 2010, 16, 3411–3417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yönetçi, N.; Oruç, N.; Ozütemiz, A.O.; Celik, H.A.; Yüce, G. Effects of mast-cell stabilization in cerulein-induced acute pancreatitis in rats. Int. J. Pancreatol. 2001, 29, 163–171. [Google Scholar] [CrossRef]
- Awla, D.; Abdulla, A.; Syk, I.; Jeppsson, B.; Regnér, S.; Thorlacius, H. Neutrophil-derived matrix metalloproteinase-9 is a potent activator of trypsinogen in acinar cells in acute pancreatitis. J. Leukoc. Biol. 2012, 91, 711–719. [Google Scholar] [CrossRef]
- Kuramoto, T.; Jin, D.; Komeda, K.; Taniguchi, K.; Hirokawa, F.; Takai, S.; Uchiyama, K. Chymase as a novel therapeutic target in acute pancreatitis. Int. J. Mol. Sci. 2021, 22, 12313. [Google Scholar] [CrossRef]
- Badertscher, K.; Brönnimann, M.; Karlen, S.; Braathen, L.R.; Yawalkar, N. Mast cell chymase is increased in chronic atopic dermatitis but not in psoriasis. Arch. Dermatol. Res. 2005, 296, 503–506. [Google Scholar] [CrossRef]
- He, S.; Walls, A.F. Human mast cell chymase induces the accumulation of neutrophils, eosinophils and other inflammatory cells in vivo. Br. J. Pharmacol. 1998, 125, 1491–1500. [Google Scholar] [CrossRef] [Green Version]
- Terakawa, M.; Tomimori, Y.; Goto, M.; Fukuda, Y. Mast cell chymase induces expression of chemokines for neutrophils in eosinophilic EoL-1 cells and mouse peritonitis eosinophils. Eur. J. Pharmacol. 2006, 538, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, N.; Tomimori, Y.; Terakawa, M.; Ishiwata, K.; Wada, A.; Muto, T.; Tanaka, T.; Maruoka, H.; Nagahira, K.; Nakatsuka, T.; et al. Oral administration of chymase inhibitor improves dermatitis in NC/Nga mice. J. Investig. Dermatol. 2007, 127, 971–973. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Terakawa, M.; Fujieda, Y.; Tomimori, Y.; Muto, T.; Tanaka, T.; Maruoka, H.; Nagahira, K.; Ogata, A.; Nakatsuka, T.; Fukuda, Y. Oral chymase inhibitor SUN13834 ameliorates skin inflammation as well as pruritus in mouse model for atopic dermatitis. Eur. J. Pharmacol. 2008, 601, 186–191. [Google Scholar] [CrossRef] [PubMed]
- Rikken, G.; Gertner, J. SUN13834 in the treatment of subjects with atopic dermatitis. J. Investig. Dermatol. 2010, 130, S69. [Google Scholar]
- Ebihara, N.; Funaki, T.; Takai, S.; Miyazaki, M.; Fujiki, K.; Murakami, A. Tear chymase in vernal keratoconjunctivitis. Curr. Eye Res. 2004, 28, 417–420. [Google Scholar] [CrossRef]
- Leonardi, A.; Brun, P.; Abatangelo, G.; Plebani, M.; Secchi, A.G. Tear levels and activity of matrix metalloproteinase (MMP)-1 and MMP-9 in vernal keratoconjunctivitis. Investig. Ophthalmol. Vis. Sci. 2003, 44, 3052–3058. [Google Scholar] [CrossRef] [Green Version]
- Nabe, T.; Kijitani, Y.; Kitagawa, Y.; Sakano, E.; Ueno, T.; Fujii, M.; Nakao, S.; Sakai, M.; Takai, S. Involvement of chymase in allergic conjunctivitis of guinea pigs. Exp. Eye Res. 2013, 113, 74–79. [Google Scholar] [CrossRef]
- Ahrens, D.; Koch, A.E.; Pope, R.M.; Stein-Picarella, M.; Niedbala, M.J. Expression of matrix metalloproteinase 9 (96-kd gelatinase B) in human rheumatoid arthritis. Arthritis Rheum. 1996, 39, 1576–1587. [Google Scholar] [CrossRef]
- Ishiguro, N.; Ito, T.; Oguchi, T.; Kojima, T.; Iwata, H.; Ionescu, M.; Poole, A.R. Relationships of matrix metalloproteinases and their inhibitors to cartilage proteoglycan and collagen turnover and inflammation as revealed by analyses of synovial fluids from patients with rheumatoid arthritis. Arthritis Rheum. 2001, 44, 2503–2511. [Google Scholar] [CrossRef]
- Guo, Y.; Wei, T.; Hu, N.; Zhou, X. Disrupted homeostasis of synovial hyaluronic acid and its associations with synovial mast cell proteases of rheumatoid arthritis patients and collagen-induced arthritis rats. Immunol. Res. 2021, 69, 584–593. [Google Scholar] [CrossRef]
- Kneilling, M.; Hültner, L.; Pichler, B.J.; Mailhammer, R.; Morawietz, L.; Solomon, S.; Eichner, M.; Sabatino, J.; Biedermann, T.; Krenn, V.; et al. Targeted mast cell silencing protects against joint destruction and angiogenesis in experimental arthritis in mice. Arthritis Rheum. 2007, 56, 1806–1816. [Google Scholar] [CrossRef]
- Chu, Y.; Wang, J.; Zhou, X. Mast cell chymase in synovial fluid of collagen-induced-arthritis rats regulates gelatinase release and promotes synovial fibroblasts proliferation via FAK/p21 signaling pathway. Biochem. Biophys. Res. Commun. 2019, 514, 336–343. [Google Scholar] [CrossRef] [PubMed]
- Starkey, J.R.; Crowle, P.K.; Taubenberger, S. Mast-cell-deficient W/Wv mice exhibit a decreased rate of tumor angiogenesis. Int. J. Cancer 1988, 42, 48–52. [Google Scholar] [CrossRef]
- Lee, J.H.; Jeon, Y.D.; Xin, M.; Lim, J.Y.; Lee, Y.M.; Kim, D.K. Mast cell modulates tumorigenesis caused by repeated bowel inflammation condition in azoxymethane/dextran sodium sulfate-induced colon cancer mouse model. Biochem. Biophys. Rep. 2022, 30, 101253. [Google Scholar] [CrossRef]
- Ibaraki, T.; Muramatsu, M.; Takai, S.; Jin, D.; Maruyama, H.; Orino, T.; Katsumata, T.; Miyazaki, M. The relationship of tryptase- and chymase-positive mast cells to angiogenesis in stage I non-small cell lung cancer. Eur. J. Cardiothorac. Surg. 2005, 28, 617–621. [Google Scholar] [CrossRef] [Green Version]
- Laface, C.; Laforgia, M.; Zito, A.F.; Loisi, D.; Zizzo, N.; Tamma, R.; Gadaleta, C.D.; Porcelli, M.; Currò, G.; Ammendola, M.; et al. Chymase-positive Mast cells correlate with tumor angiogenesis: First report in pancreatic cancer patients. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 6862–6873. [Google Scholar]
- Kondo. K.; Muramatsu, M.; Okamoto, Y.; Jin, D.; Takai, S.; Tanigawa, N.; Miyazaki, M. Expression of chymase-positive cells in gastric cancer and its correlation with the angiogenesis. J. Surg. Oncol. 2006, 93, 36–42. [Google Scholar] [CrossRef]
- Katada, J.; Muramatsu, M.; Hayashi, I.; Tsutsumi, M.; Konishi, Y.; Majima, M. Significance of vascular endothelial cell growth factor up-regulation mediated via a chymase-angiotensin-dependent pathway during angiogenesis in hamster sponge granulomas. J. Pharmacol. Exp. Ther. 2002, 302, 949–956. [Google Scholar] [CrossRef] [Green Version]
- Muramatsu, M.; Yamada, M.; Takai, S.; Miyazaki, M. Suppression of basic fibroblast growth factor-induced angiogenesis by a specific chymase inhibitor, BCEAB, through the chymase-angiotensin-dependent pathway in hamster sponge granulomas. Br. J. Pharmacol. 2002, 137, 554–560. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sammarco, G.; Varricchi, G.; Ferraro, V.; Ammendola, M.; De Fazio, M.; Altomare, D.F.; Luposella, M.; Maltese, L.; Currò, G.; Marone, G.; et al. Mast Cells, Angiogenesis and Lymphangiogenesis in Human Gastric Cancer. Int. J. Mol. Sci. 2019, 20, 2106. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deryugina, E.I.; Zajac, E.; Juncker-Jensen, A.; Kupriyanova, T.A.; Welter, L.; Quigley, J.P. Tissue-infiltrating neutrophils constitute the major in vivo source of angiogenesis-inducing MMP-9 in the tumor microenvironment. Neoplasia 2014, 16, 771–788. [Google Scholar] [CrossRef] [Green Version]
- Jodele, S.; Chantrain, C.F.; Blavier, L.; Lutzko, C.; Crooks, G.M.; Shimada, H.; Coussens, L.M.; Declerck, Y.A. The contribution of bone marrow-derived cells to the tumor vasculature in neuroblastoma is matrix metalloproteinase-9 dependent. Cancer Res. 2005, 65, 3200–3208. [Google Scholar] [CrossRef] [Green Version]
- Duengen, H.D.; Kim, R.J.; Zahger, D.; Orvin, K.; Kornowski, R.; Admon, D.; Kettner, J.; Shimony, A.; Otto, C.; Becka, M.; et al. Effects of the chymase inhibitor fulacimstat on adverse cardiac remodeling after acute myocardial infarction-Results of the Chymase Inhibitor in Adverse Remodeling after Myocardial Infarction (CHIARA MIA) 2 trial. Am. Heart J. 2020, 224, 129–137. [Google Scholar] [CrossRef]
- Rossing, P.; Strand, J.; Avogaro, A.; Becka, M.; Kanefendt, F.; Otto, C. Effects of the chymase inhibitor fulacimstat in diabetic kidney disease-results from the CADA DIA trial. Nephrol. Dial. Transplant. 2021, 36, 2263–2273. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Takai, S.; Jin, D. Pathophysiological Role of Chymase-Activated Matrix Metalloproteinase-9. Biomedicines 2022, 10, 2499. https://doi.org/10.3390/biomedicines10102499
Takai S, Jin D. Pathophysiological Role of Chymase-Activated Matrix Metalloproteinase-9. Biomedicines. 2022; 10(10):2499. https://doi.org/10.3390/biomedicines10102499
Chicago/Turabian StyleTakai, Shinji, and Denan Jin. 2022. "Pathophysiological Role of Chymase-Activated Matrix Metalloproteinase-9" Biomedicines 10, no. 10: 2499. https://doi.org/10.3390/biomedicines10102499




