The Effect of Heat Treatment of β-Tricalcium Phosphate-Containing Silica-Based Bioactive Aerogels on the Cellular Metabolism and Proliferation of MG63 Cells
Abstract
:1. Introduction
2. Materials and Methods
2.1. Synthesis of β-TCP-Modified Aerogels
2.2. Nitrogen Adsorption–Desorption Porosimetry
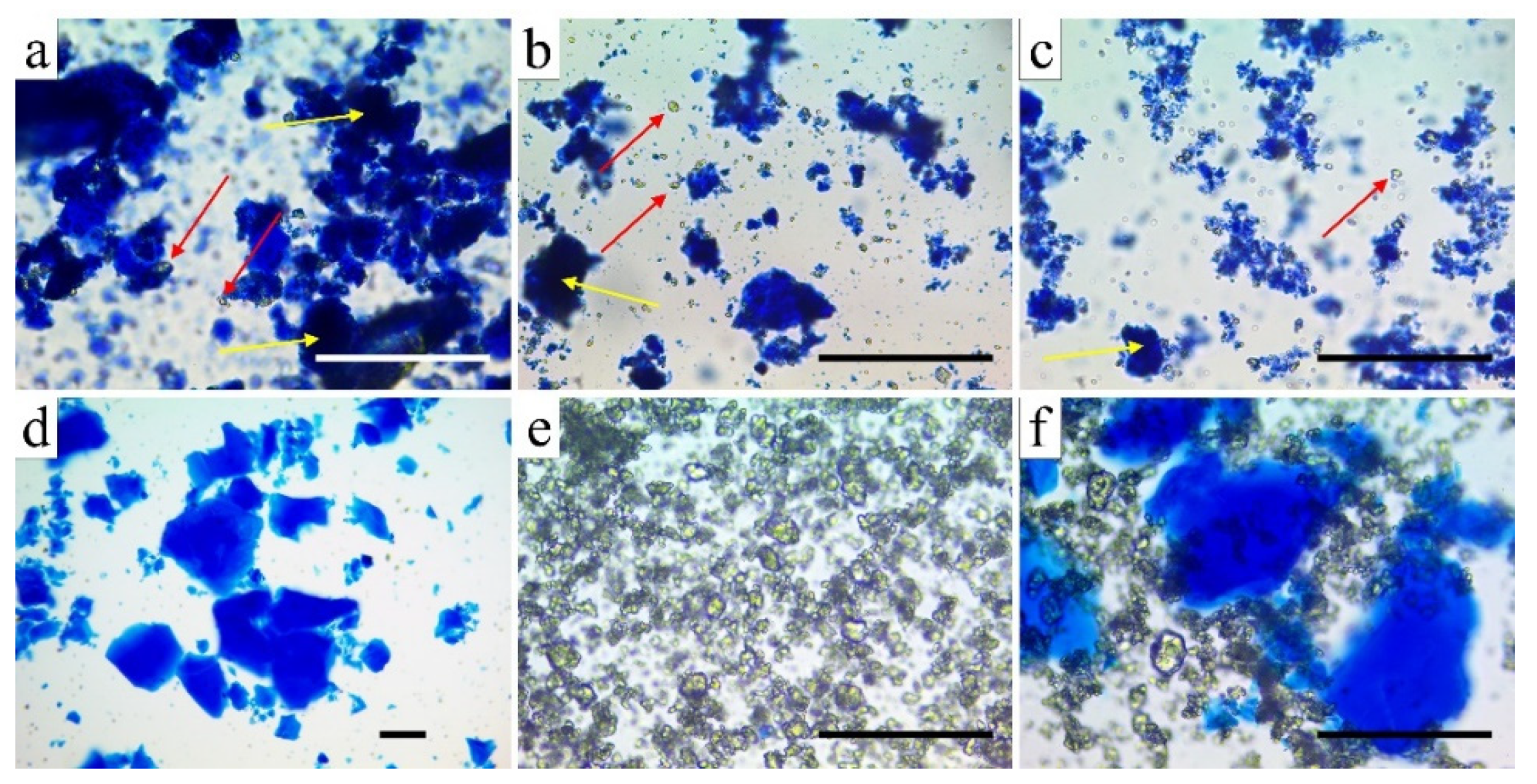
2.3. Grinding and Microscopic Study of β-TCP-Modified Aerogels (Aerogel Composites)
2.4. Determination of the Compressive Strength Values
2.5. Structural Study of as-Prepared and Heat-Treated Samples
2.6. Dissolution Dynamics and Ion-Release Properties of the Heat-Treated Composites
2.7. Cell Culture Studies
2.8. Scanning Electron Microscopy (SEM) Analysis of Structural Properties and Cell Growth
2.9. Cell Viability Assay
2.10. Alkaline Phosphatase Activity Assay
2.11. Gene Expression Analysis
2.12. Statistical Analysis
3. Results
3.1. Structural Studies
3.2. Cell Viability Assay
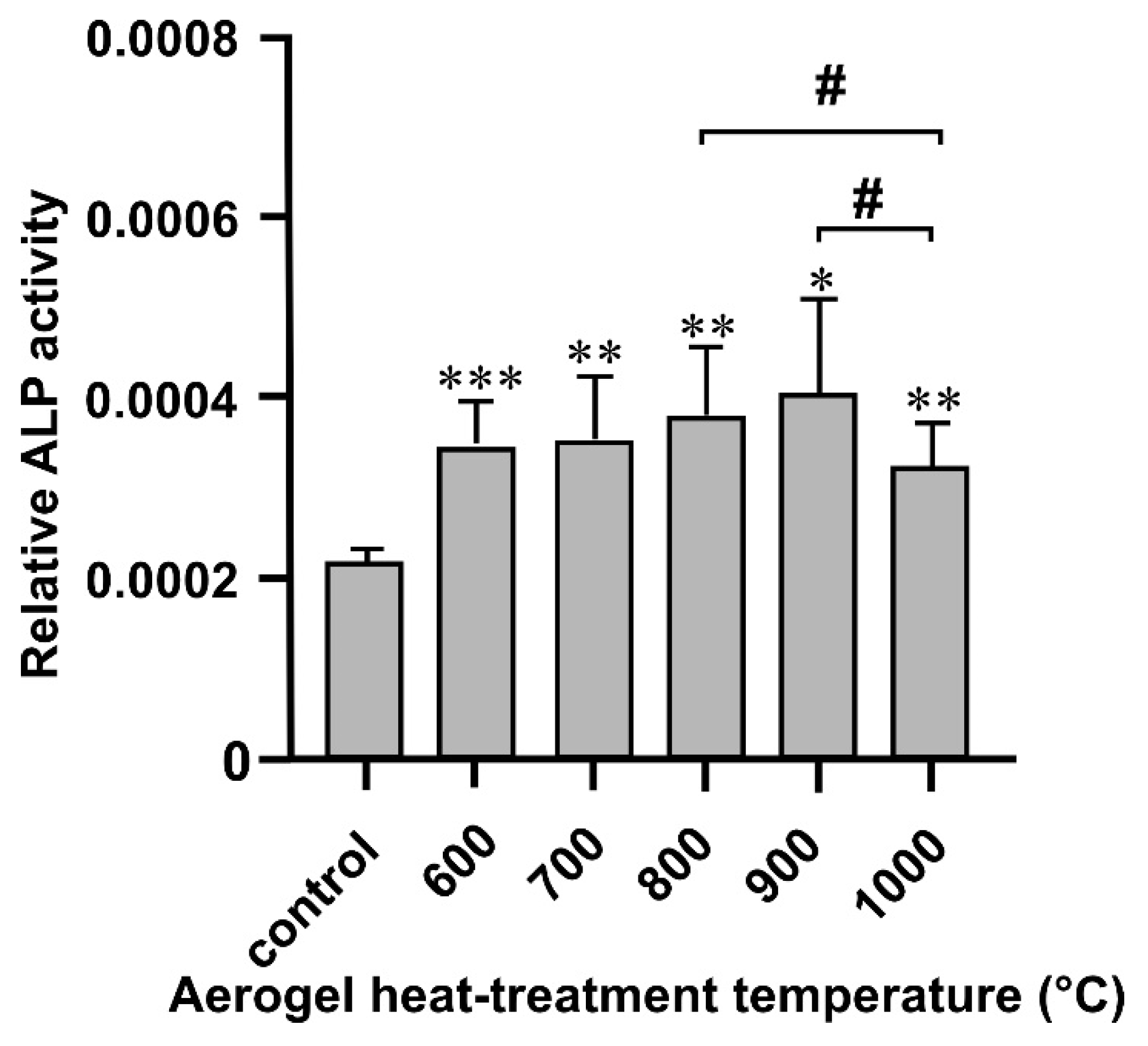
3.3. Alkaline Phosphatase Activity Assay
3.4. Gene Expression Analysis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Aegerter, M.A.; Leventis, N.; Koebel, M.M. (Eds.) Aerogels Handbook; Springer: New York, NY, USA, 2011; ISBN 978-1-4419-7477-8. [Google Scholar]
- Akimov, Y.K. Fields of Application of Aerogels. Instrum. Exp. Tech. 2003, 46, 287–299. [Google Scholar] [CrossRef]
- Maleki, H.; Durães, L.; García-González, C.A.; del Gaudio, P.; Portugal, A.; Mahmoudi, M. Synthesis and biomedical applications of aerogels: Possibilities and challenges. Adv. Colloid Interface Sci. 2016, 236, 1–27. [Google Scholar] [CrossRef] [PubMed]
- Zheng, L.; Zhang, S.; Ying, Z.; Liu, J.; Zhou, Y.; Chen, F. Engineering of aerogel-based biomaterials for biomedical applications. Int. J. Nanomed. 2020, 15, 2363–2378. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Stergar, J.; Maver, U. Review of aerogel-based materials in biomedical applications. J. Sol-Gel. Sci. Technol. 2016, 77, 738–752. [Google Scholar] [CrossRef]
- Soleimani Dorcheh, A.; Abbasi, M.H. Silica aerogel; Synthesis, properties and characterization. J. Mater. Process. Technol. 2008, 199, 10–26. [Google Scholar] [CrossRef]
- Hüsing, N.; Schubert, U. Aerogels-airy materials: Chemistry, structure, and properties. Angew. Chem. Int. Ed. 1998, 37, 22–45. [Google Scholar] [CrossRef]
- Maleki, H.; Durães, L.; Portugal, A. An overview on silica aerogels synthesis and different mechanical reinforcing strategies. J. Non-Cryst. Solids 2014, 385, 55–74. [Google Scholar] [CrossRef] [Green Version]
- Ge, J.; Li, M.; Zhang, Q.; Yang, C.Z.; Wooley, P.H.; Chen, X.; Yang, S.-Y. Silica aerogel improves the biocompatibility in a poly-ε-caprolactone composite used as a tissue engineering scaffold. Int. J. Polym. Sci. 2013, 2013, 402859. [Google Scholar] [CrossRef] [Green Version]
- Reséndiz-Hernández, P.J.; Cortés-Hernández, D.A.; Méndez Nonell, J.; Escobedo-Bocardo, J.C. Bioactive and biocompatible silica/pseudowollastonite aerogels. Adv. Sci. Technol. 2014, 96, 21–26. [Google Scholar]
- Alnaief, M.; Alzaitoun, M.A.; García-González, C.A.; Smirnova, I. Preparation of biodegradable nanoporous microspherical aerogel based on alginate. Carbohydr. Polym. 2011, 84, 1011–1018. [Google Scholar] [CrossRef]
- Veronovski, A.; Tkalec, G.; Knez, Ž.; Novak, Z. Characterisation of biodegradable pectin aerogels and their potential use as drug carriers. Carbohydr. Polym. 2014, 113, 272–278. [Google Scholar] [CrossRef] [PubMed]
- Ulker, Z.; Erkey, C. An Emerging platform for drug delivery: Aerogel based systems. J. Control. Release 2014, 177, 51–63. [Google Scholar] [CrossRef] [PubMed]
- García-González, C.A.; Alnaief, M.; Smirnova, I. Polysaccharide-based aerogels—Promising Biodegradable carriers for drug delivery systems. Carbohydr. Polym. 2011, 86, 1425–1438. [Google Scholar] [CrossRef]
- García-González, C.A.; Jin, M.; Gerth, J.; Alvarez-Lorenzo, C.; Smirnova, I. Polysaccharide-based aerogel microspheres for oral drug delivery. Carbohydr. Polym. 2015, 117, 797–806. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Prokopowicz, M.; Szewczyk, A.; Skwira, A.; Sądej, R.; Walker, G. Biphasic composite of calcium phosphate-based mesoporous silica as a novel bone drug delivery system. Drug Deliv. Transl. Res. 2020, 10, 455–470. [Google Scholar] [CrossRef] [Green Version]
- Kuttor, A.; Szalóki, M.; Rente, T.; Kerényi, F.; Bakó, J.; Fábián, I.; Lázár, I.; Jenei, A.; Hegedüs, C. Preparation and application of highly porous aerogel-based bioactive materials in dentistry. Front. Mater. Sci. 2014, 8, 46–52. [Google Scholar] [CrossRef]
- Lázár, I.; Manó, S.; Jónás, Z.; Kiss, L.; Fábián, I.; Csernátony, Z. Mesoporous silica-calcium phosphate composites for experimental bone substitution. Biomech. Hung. 2010, 3, 151–158. [Google Scholar] [CrossRef]
- Veres, P.; Kéri, M.; Bányai, I.; Lázár, I.; Fábián, I.; Domingo, C.; Kalmár, J. Mechanism of drug release from silica-gelatin aerogel—Relationship between matrix structure and release kinetics. Colloids Surf. B Biointerfaces 2017, 152, 229–237. [Google Scholar] [CrossRef] [PubMed]
- Nagy, G.; Király, G.; Veres, P.; Lázár, I.; Fábián, I.; Bánfalvi, G.; Juhász, I.; Kalmár, J. controlled release of methotrexate from functionalized silica-gelatin aerogel microparticles applied against tumor cell growth. Int. J. Pharm. 2019, 558, 396–403. [Google Scholar] [CrossRef]
- Lázár, I.; Bereczki, H.F.; Manó, S.; Daróczi, L.; Deák, G.; Fábián, I.; Csernátony, Z. Synthesis and study of new functionalized silica aerogel poly(methyl methacrylate) composites for biomedical use. Polym. Compos. 2015, 36, 348–358. [Google Scholar] [CrossRef] [Green Version]
- Szabó, B.A.; Kiss, L.; Manó, S.; Jónás, Z.; Lázár, I.; Fábián, I.; Dezső, B.; Csernátony, Z. The examination of aerogel composite artificial bone substitutes in animal models. Biomech. Hung. 2013, 6, 52–63. [Google Scholar] [CrossRef]
- Martin, K.R. The chemistry of silica and its potential health benefits. J. Nutr. Health Aging 2007, 11, 94–97. [Google Scholar] [PubMed]
- De Godoy, R.F.; Hutchens, S.; Campion, C.; Blunn, G. Silicate-substituted calcium phosphate with enhanced strut porosity stimulates osteogenic differentiation of human mesenchymal stem cells. J. Mater. Sci. Mater. Med. 2015, 26, 54. [Google Scholar] [CrossRef] [PubMed]
- Feng, J.; Yan, W.; Gou, Z.; Weng, W.; Yang, D. Stimulating Effect of silica-containing nanospheres on proliferation of osteoblast-like cells. J. Mater. Sci. Mater. Med. 2007, 18, 2167–2172. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Wu, X.; Wei, J.; Lu, X.; Zhang, S.; Shi, J.; Liu, C. Stimulated osteoblastic proliferation by mesoporous silica xerogel with high specific surface area. J. Mater. Sci. Mater. Med. 2011, 22, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Obata, A.; Kasuga, T. Stimulation of human mesenchymal stem cells and osteoblasts activities in vitro on silicon-releasable scaffolds. J. Biomed. Mater. Res. 2009, 91A, 11–17. [Google Scholar] [CrossRef] [PubMed]
- Anderson, S.I.; Downes, S.; Perry, C.C.; Caballero, A.M. Evaluation of the osteoblast response to a silica gel in vitro. J. Mater. Sci. Mater. Med. 1998, 9, 731–735. [Google Scholar] [CrossRef]
- Uribe, P.; Johansson, A.; Jugdaohsingh, R.; Powell, J.J.; Magnusson, C.; Davila, M.; Westerlund, A.; Ransjö, M. Soluble silica stimulates osteogenic differentiation and gap junction communication in human dental follicle cells. Sci. Rep. 2020, 10, 9923. [Google Scholar] [CrossRef]
- Zou, S.; Ireland, D.; Brooks, R.A.; Rushton, N.; Best, S. The effects of silicate ions on human osteoblast adhesion, proliferation, and differentiation. J. Biomed. Mater. Res. 2008, 90B, 123–130. [Google Scholar] [CrossRef]
- Xynos, I.D.; Edgar, A.J.; Buttery, L.D.K.; Hench, L.L.; Polak, J.M. Ionic products of bioactive glass dissolution increase proliferation of human osteoblasts and induce insulin-like growth factor II MRNA expression and protein synthesis. Biochem. Biophys. Res. Commun. 2000, 276, 461–465. [Google Scholar] [CrossRef]
- Shie, M.-Y.; Ding, S.-J.; Chang, H.-C. The role of silicon in osteoblast-like cell proliferation and apoptosis. Acta Biomater. 2011, 7, 2604–2614. [Google Scholar] [CrossRef] [PubMed]
- Kim, E.-J.; Bu, S.-Y.; Sung, M.-K.; Choi, M.-K. Effects of silicon on osteoblast activity and bone mineralization of MC3T3-E1 cells. Biol. Trace Elem. Res. 2013, 152, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Ha, S.-W.; Weitzmann, M.N.; Beck, G.R. Bioactive silica nanoparticles promote osteoblast differentiation through stimulation of autophagy and direct association with LC3 and P62. ACS Nano 2014, 8, 5898–5910. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, M.; Jiao, G.; Liu, H.; Wu, W.; Li, S.; Wang, Q.; Xu, D.; Li, X.; Liu, H.; Chen, Y. Biological Silicon stimulates collagen type 1 and osteocalcin synthesis in human osteoblast-like cells through the BMP-2/Smad/RUNX2 signaling pathway. Biol. Trace Elem. Res. 2016, 173, 306–315. [Google Scholar] [CrossRef] [PubMed]
- Prins, H.-J.; Braat, A.K.; Gawlitta, D.; Dhert, W.J.A.; Egan, D.A.; Tijssen-Slump, E.; Yuan, H.; Coffer, P.J.; Rozemuller, H.; Martens, A.C. In vitro induction of alkaline phosphatase levels predicts in vivo bone forming capacity of human bone marrow stromal cells. Stem. Cell Res. 2014, 12, 428–440. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gough, J.E.; Jones, J.R.; Hench, L.L. Nodule formation and mineralisation of human primary osteoblasts cultured on a porous bioactive glass scaffold. Biomaterials 2004, 25, 2039–2046. [Google Scholar] [CrossRef] [PubMed]
- Kayongo-Male, H.; Julson, J.L. Effects of high levels of dietary silicon on bone development of growing rats and turkeys fed semi-purified diets. Biol. Trace Elem. Res. 2008, 123, 191–201. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.-H.; Bae, Y.-J.; Choi, M.-K.; Chung, Y.-S. Silicon supplementation improves the bone mineral density of calcium-deficient ovariectomized rats by reducing bone resorption. Biol. Trace Elem. Res. 2009, 128, 239–247. [Google Scholar] [CrossRef]
- Sadeghzade, S.; Emadi, R.; Tavangarian, F.; Naderi, M. Fabrication and Evaluation of silica-based ceramic scaffolds for hard tissue engineering applications. Mater. Sci. Eng. C 2017, 71, 431–438. [Google Scholar] [CrossRef] [PubMed]
- Szurkowska, K.; Kolmas, J. Hydroxyapatites enriched in silicon—Bioceramic Materials for biomedical and pharmaceutical applications. Prog. Nat. Sci. Mater. Int. 2017, 27, 401–409. [Google Scholar] [CrossRef]
- Al-Harbi, N.; Mohammed, H.; Al-Hadeethi, Y.; Bakry, A.S.; Umar, A.; Hussein, M.A.; Abbassy, M.A.; Vaidya, K.G.; Al Berakdar, G.; Mkawi, E.M.; et al. Silica-based bioactive glasses and their applications in hard tissue regeneration: A review. Pharmaceuticals 2021, 14, 75. [Google Scholar] [CrossRef] [PubMed]
- Schatkoski, V.M.; do Amaral Montanheiro, L.T.; Canuto de Menezes, B.R.; Pereira, R.M.; Rodrigues, K.F.; Ribas, R.G.; Morais da Silva, D.; Thim, G.P. Current advances concerning the most cited metal ions doped bioceramics and silicate-based bioactive glasses for bone tissue engineering. Ceram. Int. 2021, 47, 2999–3012. [Google Scholar] [CrossRef]
- Heimann, R.B. Silicon nitride, a close to ideal ceramic material for medical application. Ceramics 2021, 4, 208–223. [Google Scholar] [CrossRef]
- Awad, K.R.; Ahuja, N.; Shah, A.; Tran, H.; Aswath, P.B.; Brotto, M.; Varanasi, V. Silicon nitride enhances osteoprogenitor cell growth and differentiation via increased surface energy and formation of amide and nanocrystalline HA for craniofacial reconstruction. Med. Devices Sens. 2019, 2, e10032. [Google Scholar] [CrossRef]
- Mirkhalaf, M.; Goldsmith, J.; Ren, J.; Dao, A.; Newman, P.; Schindeler, A.; Woodruff, M.A.; Dunstan, C.R.; Zreiqat, H. Highly substituted calcium silicates 3D printed with complex architectures to produce stiff, strong and bioactive scaffolds for bone regeneration. Appl. Mater. Today 2021, 25, 101230. [Google Scholar] [CrossRef]
- Devi, K.B.; Lee, B.; Roy, A.; Kumta, P.N.; Roy, M. Effect of Zinc oxide doping on in vitro degradation of magnesium silicate bioceramics. Mater. Lett. 2017, 207, 100–103. [Google Scholar] [CrossRef]
- Qian, G.; Lu, T.; Zhang, J.; Liu, R.; Wang, Z.; Yu, B.; Li, H.; Shi, H.; Ye, J. Promoting bone regeneration of calcium phosphate cement by addition of PLGA Microspheres and zinc silicate via synergistic effect of in-situ pore generation, bioactive ion stimulation and macrophage immunomodulation. Appl. Mater. Today 2020, 19, 100615. [Google Scholar] [CrossRef]
- Eltohamy, M.; Kundu, B.; Moon, J.; Lee, H.-Y.; Kim, H.-W. Anti-Bacterial zinc-doped calcium silicate cements: Bone filler. Ceram. Int. 2018, 44, 13031–13038. [Google Scholar] [CrossRef]
- Surmenev, R.A.; Shkarina, S.; Syromotina, D.S.; Melnik, E.V.; Shkarin, R.; Selezneva, I.I.; Ermakov, A.M.; Ivlev, S.I.; Cecilia, A.; Weinhardt, V.; et al. Characterization of biomimetic silicate- and strontium-containing hydroxyapatite microparticles embedded in biodegradable electrospun polycaprolactone scaffolds for bone regeneration. Eur. Polym. J. 2019, 113, 67–77. [Google Scholar] [CrossRef]
- Ressler, A.; Žužić, A.; Ivanišević, I.; Kamboj, N.; Ivanković, H. Ionic substituted hydroxyapatite for bone regeneration applications: A review. Open Ceram. 2021, 6, 100122. [Google Scholar] [CrossRef]
- Jeong, J.; Kim, J.H.; Shim, J.H.; Hwang, N.S.; Heo, C.Y. Bioactive calcium phosphate materials and applications in bone regeneration. Biomater. Res. 2019, 23, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Metsger, D.S.; Driskell, T.D.; Paulsrud, J.R. Tricalcium phosphate ceramic—A resorbable bone implant: Review and current status. J. Am. Dent. Assoc. 1982, 105, 1035–1038. [Google Scholar] [CrossRef] [PubMed]
- Sakai, S.; Anada, T.; Tsuchiya, K.; Yamazaki, H.; Margolis, H.C.; Suzuki, O. Comparative Study on the resorbability and dissolution behavior of octacalcium phosphate, β-tricalcium phosphate, and hydroxyapatite under physiological conditions. Dent. Mater. J. 2016, 35, 216–224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kotani, S.; Fujita, Y.; Kitsugi, T.; Nakamura, T.; Yamamuro, T.; Ohtsuki, C.; Kokubo, T. Bone bonding mechanism of [Beta]-tricalcium phosphate. J. Biomed. Mater. Res. 1991, 25, 1303–1315. [Google Scholar] [CrossRef] [PubMed]
- Fujita, R.; Yokoyama, A.; Nodasaka, Y.; Kohgo, T.; Kawasaki, T. Ultrastructure of ceramic-bone interface using hydroxyapatite and β-tricalcium phosphate ceramics and replacement mechanism of β-tricalcium phosphate in bone. Tissue Cell 2003, 35, 427–440. [Google Scholar] [CrossRef]
- Tzaphlidou, M.; Zaichick, V. Calcium, phosphorus, calcium-phosphorus ratio in rib bone of healthy humans. Biol. Trace Elem. Res. 2003, 93, 63–74. [Google Scholar] [CrossRef]
- Zaichick, V.; Tzaphlidou, M. Calcium and phosphorus concentrations and the calcium/phosphorus ratio in trabecular bone from the femoral neck of healthy humans as determined by neutron activation analysis. Appl. Radiat. Isot. 2003, 58, 623–627. [Google Scholar] [CrossRef]
- Tzaphlidou, M.; Zaichick, V. Sex and age related Ca/P ratio in cortical bone of iliac crest of healthy humans. J. Radioanal. Nucl. Chem. 2004, 259, 347–349. [Google Scholar] [CrossRef]
- Kwon, S.-H.; Jun, Y.-K.; Hong, S.-H.; Kim, H.-E. Synthesis and dissolution behavior of β-TCP and HA/β-TCP composite powders. J. Eur. Ceram. Soc. 2003, 23, 1039–1045. [Google Scholar] [CrossRef]
- Chamary, S.; Grenho, L.; Fernandes, M.H.; Bouchart, F.; Monteiro, F.J.; Hornez, J.C. Influence of a Macroporous β-TCP Structure on human mesenchymal stem cell proliferation and differentiation in vitro. Open Ceram. 2021, 7, 100141. [Google Scholar] [CrossRef]
- Guo, X.; Zheng, Q.; Kulbatski, I.; Yuan, Q.; Yang, S.; Shao, Z.; Wang, H.; Xiao, B.; Pan, Z.; Tang, S. Bone regeneration with active angiogenesis by basic fibroblast growth factor gene transfected mesenchymal stem cells seeded on porous β-tcp ceramic scaffolds. Biomed. Mater. 2006, 1, 93–99. [Google Scholar] [CrossRef] [PubMed]
- Ding, S.-J.; Shie, M.-Y.; Hoshiba, T.; Kawazoe, N.; Chen, G.; Chang, H.-C. osteogenic differentiation and immune response of human bone-marrow-derived mesenchymal stem cells on injectable calcium-silicate-based bone grafts. Tissue Eng. Part A 2010, 16, 2343–2354. [Google Scholar] [CrossRef] [PubMed]
- Gao, P.; Zhang, H.; Liu, Y.; Fan, B.; Li, X.; Xiao, X.; Lan, P.; Li, M.; Geng, L.; Liu, D.; et al. Beta-Tricalcium phosphate granules improve osteogenesis in vitro and establish innovative osteo-regenerators for bone tissue engineering in vivo. Sci. Rep. 2016, 6, 23367. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hegedűs, V.; Kerényi, F.; Boda, R.; Horváth, D.; Lázár, I.; Tóth-Győri, E.; Dezső, B.; Hegedus, C. β-tricalcium phosphate silica aerogel as an alternative bioactive ceramic for the potential use in dentistry. Adv. Appl. Ceram. 2018, 117, 476–484. [Google Scholar] [CrossRef]
- Matsumoto, N.; Yoshida, K.; Hashimoto, K.; Toda, Y. Thermal Stability of β-tricalcium phosphate doped with monovalent metal ions. Mater. Res. Bull. 2009, 44, 1889–1894. [Google Scholar] [CrossRef]
- Gurav, J.L.; Jung, I.-K.; Park, H.-H.; Kang, E.S.; Nadargi, D.Y. Silica aerogel: Synthesis and applications. J. Nanomater. 2010, 2010, 409310. [Google Scholar] [CrossRef] [Green Version]
- Woignier, T.; Primera, J.; Alaoui, A.; Etienne, P.; Despestis, F.; Calas-Etienne, S. Mechanical Properties and Brittle Behavior of Silica Aerogels. Gels 2015, 1, 256–275. [Google Scholar] [CrossRef] [Green Version]
- Warring, S.L.; Beattie, D.A.; McQuillan, A.J. Surficial siloxane-to-silanol interconversion during room-temperature hydration/dehydration of amorphous silica films observed by ATR-IR and TIR-raman spectroscopy. Langmuir 2016, 32, 1568–1576. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, A.S.; Pantano, C.G. Hydroxylation and dehydroxylation behavior of silica glass fracture surfaces. J. Am. Ceram. Soc. 2002, 85, 1499–1504. [Google Scholar] [CrossRef]
- Dixit, S.; van Cappellen, P. Surface chemistry and reactivity of biogenic silica. Geochim. Cosmochim. Acta 2002, 66, 2559–2568. [Google Scholar] [CrossRef]
- Schrader, A.M.; Monroe, J.I.; Sheil, R.; Dobbs, H.A.; Keller, T.J.; Li, Y.; Jain, S.; Shell, M.S.; Israelachvili, J.N.; Han, S. Surface chemical heterogeneity modulates silica surface hydration. Proc. Natl. Acad. Sci. USA 2018, 115, 2890–2895. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kerisit, S.; Mahadevan, T.; Du, J. Patchy particle model of hydrated amorphous silica. J. Non-Cryst. Solids 2021, 556, 120555. [Google Scholar] [CrossRef]
- Rimstidt, J.D.; Zhang, Y.; Zhu, C. Rate equations for sodium catalyzed amorphous silica dissolution. Geochim. Cosmochim. Acta 2016, 195, 120–125. [Google Scholar] [CrossRef] [Green Version]
- Icenhower, J.P.; Dove, P.M. The dissolution kinetics of amorphous silica into sodium chloride solutions: Effects of temperature and ionic strength. Geochim. Cosmochim. Acta 2000, 64, 4193–4203. [Google Scholar] [CrossRef]
- Daniele, P.G.; De Robertis, A.; De Stefano, C.; Gianguzza, A.; Sammartano, S. Salt effects on the protonation of ortho-phosphate between 10 and 50 °C in aqueous solution. A complex formation model. J. Solut. Chem. 1991, 20, 495–515. [Google Scholar] [CrossRef]
- Liu, H.; Yazici, H.; Ergun, C.; Webster, T.J.; Bermek, H. An in vitro evaluation of the Ca/P ratio for the cytocompatibility of nano-to-micron particulate calcium phosphates for bone regeneration. Acta Biomater. 2008, 4, 1472–1479. [Google Scholar] [CrossRef] [PubMed]
- Kaur, D.; Reddy, M.S.; Pandey, O.P. In-vitro bioactivity of silicate-phosphate glasses using agriculture biomass silica. J. Mater. Sci. Mater. Med. 2020, 31, 65. [Google Scholar] [CrossRef] [PubMed]
- Szumera, M.; Wacławska, I. Spectroscopic and thermal studies of silicate-phosphate glass. J. Anal. Calorim. 2007, 88, 151–156. [Google Scholar] [CrossRef]









| Temperature | BET Surface Area (m2/g) | C-Constant | Average Pore Diameter (nm) | Pore Volume (cm3/g) | Micropore Volume (cm3/g) |
|---|---|---|---|---|---|
| 500 | 400.0 | 82.46 | 26.34 | 3.397 | 0.026 |
| 600 | 415.0 | 56.33 | 33.98 | 3.254 | 0.021 |
| 700 | 382.8 | 64.81 | 33.97 | 3.251 | 0.000 |
| 800 | 384.9 | 59.40 | 31.95 | 3.074 | 0.021 |
| 900 | 277.0 | 51.18 | 46.73 | 3.237 | 0.005 |
| 1000 | 184.4 | 52.03 | 29.59 | 1.363 | 0.010 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hegedűs, C.; Czibulya, Z.; Tóth, F.; Dezső, B.; Hegedűs, V.; Boda, R.; Horváth, D.; Csík, A.; Fábián, I.; Tóth-Győri, E.; et al. The Effect of Heat Treatment of β-Tricalcium Phosphate-Containing Silica-Based Bioactive Aerogels on the Cellular Metabolism and Proliferation of MG63 Cells. Biomedicines 2022, 10, 662. https://doi.org/10.3390/biomedicines10030662
Hegedűs C, Czibulya Z, Tóth F, Dezső B, Hegedűs V, Boda R, Horváth D, Csík A, Fábián I, Tóth-Győri E, et al. The Effect of Heat Treatment of β-Tricalcium Phosphate-Containing Silica-Based Bioactive Aerogels on the Cellular Metabolism and Proliferation of MG63 Cells. Biomedicines. 2022; 10(3):662. https://doi.org/10.3390/biomedicines10030662
Chicago/Turabian StyleHegedűs, Csaba, Zsuzsanna Czibulya, Ferenc Tóth, Balázs Dezső, Viktória Hegedűs, Róbert Boda, Dóra Horváth, Attila Csík, István Fábián, Enikő Tóth-Győri, and et al. 2022. "The Effect of Heat Treatment of β-Tricalcium Phosphate-Containing Silica-Based Bioactive Aerogels on the Cellular Metabolism and Proliferation of MG63 Cells" Biomedicines 10, no. 3: 662. https://doi.org/10.3390/biomedicines10030662
APA StyleHegedűs, C., Czibulya, Z., Tóth, F., Dezső, B., Hegedűs, V., Boda, R., Horváth, D., Csík, A., Fábián, I., Tóth-Győri, E., Sajtos, Z., & Lázár, I. (2022). The Effect of Heat Treatment of β-Tricalcium Phosphate-Containing Silica-Based Bioactive Aerogels on the Cellular Metabolism and Proliferation of MG63 Cells. Biomedicines, 10(3), 662. https://doi.org/10.3390/biomedicines10030662







