Perspective: Chicken Models for Studying the Ontogenetic Origin of Neuropsychiatric Disorders
Abstract
:1. The Pregnancy-Specific Environment and Neuropsychiatric Dysfunction
1.1. Ontogenetic Origin of Neuropsychiatric Dysfunction
1.2. The Maternal–Fetal Transmission in Neurodevelopmental Disorders and Neuropsychiatric Dysfunction
1.3. The Microbial Barrier for Investigating Fetal Exposure on Neuropsychiatric Development
1.4. Ethical Issues in Investigating Xenobiotics Deposition in Fetus
2. Thinking Chickens
2.1. Chicken Embryo as a Potential Model for Nonclinical Studies
2.2. Techniques for Experimental Manipulation and Characterization in the Chicken Embryo
2.3. Social Predisposition in Chickens
2.4. Chicken Neuroendocrine Development
3. Chicken Embryo as an Ontogenetic Model for Investigating Neuropsychiatric Disorders and the Underlying Mechanisms
3.1. Embryonic Valproic Acid Exposure Impairs Social Predispositions
3.2. The Dosage Effect of Embryonic Serotonin Exposure in Neuroendocrinological Development
3.3. Embryonic Exposure to Tryptophan Alters the Microbiota–Gut–Brain Axis in Offspring
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ball, H.A.; Arseneault, L.; Taylor, A.; Maughan, B.; Caspi, A.; Moffitt, T.E. Genetic and environmental influences on victims, bullies and bully-victims in childhood. J. Child Psychol. Psychiatry 2008, 49, 104–112. [Google Scholar] [CrossRef] [PubMed]
- Han, V.X.; Patel, S.; Jones, H.F.; Nielsen, T.C.; Mohammad, S.S.; Hofer, M.J.; Gold, W.; Brilot, F.; Lain, S.J.; Nassar, N. Maternal acute and chronic inflammation in pregnancy is associated with common neurodevelopmental disorders: A systematic review. Transl. Psychiatry 2021, 11, 71. [Google Scholar] [CrossRef]
- Mairesse, J.; Lesage, J.; Breton, C.; Bréant, B.; Hahn, T.; Darnaudéry, M.; Dickson, S.L.; Seckl, J.; Blondeau, B.; Vieau, D. Maternal stress alters endocrine function of the feto-placental unit in rats. Am. J. Physiol. Endocrinol. Metab. 2007, 292, E1526–E1533. [Google Scholar] [CrossRef]
- Hay, D.; Mundy, L.; Roberts, S.; Carta, R.; Waters, C.; Perra, O.; Jones, R.; Jones, I.; Goodyer, I.; Harold, G.; et al. Known risk factors for violence predict 12-month-old infants’ aggressiveness with peers. Psychol. Sci. 2011, 22, 1205–1211. [Google Scholar] [CrossRef]
- Zhang, Z.; Imperial, M.Z.; Patilea-Vrana, G.I.; Wedagedera, J.; Gaohua, L.; Unadkat, J.D. Development of a novel maternal-fetal physiologically based pharmacokinetic model I: Insights into factors that determine fetal drug exposure through simulations and sensitivity analyses. Drug Metab. Dispos. 2017, 45, 920–938. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koren, G. Maternal-Fetal Toxicology: A Clinician’s Guide; Marcel Dekker Inc.: New York, NY, USA, 2001. [Google Scholar]
- De La Rochebrochard, E.; Joshi, H. Children born after unplanned pregnancies and cognitive development at 3 years: Social differentials in the United Kingdom Millennium Cohort. Am. J. Epidemiol. 2013, 178, 910–920. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carson, C.; Redshaw, M.; Sacker, A.; Kelly, Y.; Kurinczuk, J.J.; Quigley, M.A. Effects of pregnancy planning, fertility, and assisted reproductive treatment on child behavioral problems at 5 and 7 years: Evidence from the Millennium Cohort Study. Fertil. Steril. 2013, 99, 456–463. [Google Scholar] [CrossRef] [PubMed]
- Hayatbakhsh, M.R.; Najman, J.M.; Khatun, M.; Al Mamun, A.; Bor, W.; Clavarino, A. A longitudinal study of child mental health and problem behaviours at 14 years of age following unplanned pregnancy. Psychiatry Res. 2011, 185, 200–204. [Google Scholar] [CrossRef]
- Mitchell, A.A.; Gilboa, S.M.; Werler, M.M.; Kelley, K.E.; Louik, C.; Hernández-Díaz, S.; Study, N.B.D.P. Medication use during pregnancy, with particular focus on prescription drugs: 1976–2008. Am. J. Obstet. Gynecol. 2011, 205, 51.e51–51.e58. [Google Scholar] [CrossRef] [Green Version]
- Dashraath, P.; Wong, J.L.J.; Lim, M.X.K.; Lim, L.M.; Li, S.; Biswas, A.; Choolani, M.; Mattar, C.; Su, L.L. Coronavirus disease 2019 (COVID-19) pandemic and pregnancy. Am. J. Obstet. Gynecol. 2020, 222, 521–531. [Google Scholar] [CrossRef]
- Son, M.; Gallagher, K.; Lo, J.Y.; Lindgren, E.; Burris, H.H.; Dysart, K.; Greenspan, J.; Culhane, J.F.; Handley, S.C. Coronavirus disease 2019 (COVID-19) pandemic and pregnancy outcomes in a US population. Obstet. Gynecol. 2021, 138, 542. [Google Scholar] [CrossRef]
- Rosenfeld, C.S. Placental serotonin signaling, pregnancy outcomes, and regulation of fetal brain development. Biol. Reprod. 2020, 102, 532–538. [Google Scholar] [CrossRef]
- Montgomery, A.K.; Shuffrey, L.C.; Guter, S.J.; Anderson, G.M.; Jacob, S.; Mosconi, M.W.; Sweeney, J.A.; Turner, J.B.; Sutcliffe, J.S.; Cook, E.H., Jr. Maternal serotonin levels are associated with cognitive ability and core symptoms in autism spectrum disorder. J. Am. Acad. Child Adolesc. Psychiatry 2018, 57, 867–875. [Google Scholar] [CrossRef]
- De Angelis, M.; Piccolo, M.; Vannini, L.; Siragusa, S.; De Giacomo, A.; Serrazzanetti, D.I.; Cristofori, F.; Guerzoni, M.E.; Gobbetti, M.; Francavilla, R. Fecal microbiota and metabolome of children with autism and pervasive developmental disorder not otherwise specified. PLoS ONE 2013, 8, e76993. [Google Scholar] [CrossRef] [Green Version]
- Bonnin, A.; Levitt, P. Fetal, maternal, and placental sources of serotonin and new implications for developmental programming of the brain. Neuroscience 2011, 197, 1–7. [Google Scholar] [CrossRef] [Green Version]
- Zhou, F.C.; Sari, Y.; Zhang, J.; Goodlett, C.R.; Li, T.-K. Prenatal alcohol exposure retards the migration and development of serotonin neurons in fetal C57BL mice. Dev. Brain Res. 2001, 126, 147–155. [Google Scholar] [CrossRef]
- Zhou, F.C.; Sari, Y.; Powrozek, T.A. Fetal alcohol exposure reduces serotonin innervation and compromises development of the forebrain along the serotonergic pathway. Alcohol. Clin. Exp. Res. 2005, 29, 141–149. [Google Scholar] [CrossRef]
- Martinez-Morga, M.; Quesada-Rico, M.; Bueno, C.; Martinez, S. Neurobiological bases of autistic spectrum disorder and attention deficit hyperactivity disorder: Neural differentiation and synaptogenesis. Rev. Neurol. 2018, 66, S97–S102. [Google Scholar]
- Grizenko, N.; Fortier, M.-E.; Zadorozny, C.; Thakur, G.; Schmitz, N.; Duval, R.; Joober, R. Maternal stress during pregnancy, ADHD symptomatology in children and genotype: Gene-environment interaction. J. Can. Acad. Child Adolesc. Psychiatry 2012, 21, 9. [Google Scholar]
- Kwok, J.; Hall, H.A.; Murray, A.L.; Auyeung, B. The association between analgesic drug use in pregnancy and neurodevelopmental disorders: Protocol for an umbrella review. Syst. Rev. 2020, 9, 202. [Google Scholar] [CrossRef]
- Salisbury, A.L.; Papandonatos, G.D.; Stroud, L.R.; Smith, A.K.; Brennan, P.A. Prenatal antidepressant exposures and gastrointestinal complaints in childhood: A gut–brain axis connection? Dev. Psychobiol. 2020, 62, 816–828. [Google Scholar] [CrossRef] [PubMed]
- Coste, J.; Blotiere, P.-O.; Miranda, S.; Mikaeloff, Y.; Peyre, H.; Ramus, F.; Zureik, M.; Weill, A.; Dray-Spira, R. Risk of early neurodevelopmental disorders associated with in utero exposure to valproate and other antiepileptic drugs: A nationwide cohort study in France. Sci. Rep. 2020, 10, 17362. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-W.; Choi, C.S.; Kim, K.C.; Park, J.H.; Seung, H.; Joo, S.H.; Yang, S.M.; Shin, C.Y.; Park, S.H. Gastrointestinal tract abnormalities induced by prenatal valproic Acid exposure in rat offspring. Toxicol. Res. 2013, 29, 173–179. [Google Scholar] [CrossRef] [PubMed]
- Wise, L.D. The ICH S5 (R2) guideline for the testing of medicinal agents. In Teratogenicity Testing; Springer: Berlin/Heidelberg, Germany, 2013; pp. 1–11. [Google Scholar]
- Alcock, J.; Maley, C.C.; Aktipis, C.A. Is eating behavior manipulated by the gastrointestinal microbiota? Evolutionary pressures and potential mechanisms. Bioessays 2014, 36, 940–949. [Google Scholar] [CrossRef]
- Yap, C.X.; Henders, A.K.; Alvares, G.A.; Wood, D.L.; Krause, L.; Tyson, G.W.; Restuadi, R.; Wallace, L.; McLaren, T.; Hansell, N.K. Autism-related dietary preferences mediate autism-gut microbiome associations. Cell 2021, 184, 5916–5931.e17. [Google Scholar] [CrossRef]
- Xiao, L.; Yan, J.; Yang, T.; Zhu, J.; Li, T.; Wei, H.; Chen, J. Fecal Microbiome Transplantation from Children with Autism Spectrum Disorder Modulates Tryptophan and Serotonergic Synapse Metabolism and Induces Altered Behaviors in Germ-Free Mice. mSystems 2021, 6, e01343-20. [Google Scholar] [CrossRef]
- Kong, Q.; Wang, B.; Tian, P.; Li, X.; Zhao, J.; Zhang, H.; Chen, W.; Wang, G. Daily intake of Lactobacillus alleviates autistic-like behaviors by ameliorating the 5-hydroxytryptamine metabolic disorder in VPA-treated rats during weaning and sexual maturation. Food Funct. 2021, 12, 2591–2604. [Google Scholar] [CrossRef]
- Suri, D.; Teixeira, C.M.; Cagliostro, M.K.C.; Mahadevia, D.; Ansorge, M.S. Monoamine-sensitive developmental periods impacting adult emotional and cognitive behaviors. Neuropsychopharmacology 2015, 40, 88–112. [Google Scholar] [CrossRef] [Green Version]
- Bailey, M.T.; Coe, C.L. Maternal separation disrupts the integrity of the intestinal microflora in infant rhesus monkeys. Dev. Psychobiol. 1999, 35, 146–155. [Google Scholar] [CrossRef]
- Ming, X.; Chen, N.; Ray, C.; Brewer, G.; Kornitzer, J.; Steer, R.A. A gut feeling: A hypothesis of the role of the microbiome in attention-deficit/hyperactivity disorders. Child Neurol. Open 2018, 5, 2329048X18786799. [Google Scholar] [CrossRef] [Green Version]
- Roussin, L.; Prince, N.; Perez-Pardo, P.; Kraneveld, A.D.; Rabot, S.; Naudon, L. Role of the gut microbiota in the pathophysiology of autism spectrum disorder: Clinical and preclinical evidence. Microorganisms 2020, 8, 1369. [Google Scholar] [CrossRef]
- Shankar, R.; Perera, B.; Thomas, R.H. Epilepsy, an orphan disorder within the neurodevelopmental family. J. Neurol. Neurosurg. Psychiatry 2020, 91, 1245–1247. [Google Scholar] [CrossRef]
- Bozzi, Y.; Casarosa, S.; Caleo, M. Epilepsy as a neurodevelopmental disorder. Front. Psychiatry 2012, 3, 19. [Google Scholar] [CrossRef] [Green Version]
- Usui, N.; Matsuzaki, H.; Shimada, S. Characterization of Early Life Stress-Affected Gut Microbiota. Brain Sci. 2021, 11, 913. [Google Scholar] [CrossRef]
- Rincel, M.; Aubert, P.; Chevalier, J.; Grohard, P.-A.; Basso, L.; de Oliveira, C.M.; Helbling, J.C.; Lévy, É.; Chevalier, G.; Leboyer, M. Multi-hit early life adversity affects gut microbiota, brain and behavior in a sex-dependent manner. Brain Behav. Immun. 2019, 80, 179–192. [Google Scholar] [CrossRef]
- Luna, R.A.; Foster, J.A. Gut brain axis: Diet microbiota interactions and implications for modulation of anxiety and depression. Curr. Opin. Biotechnol. 2015, 32, 35–41. [Google Scholar] [CrossRef]
- Sarnyai, Z.; Kraeuter, A.-K.; Palmer, C.M. Ketogenic diet for schizophrenia: Clinical implication. Curr. Opin. Psychiatry 2019, 32, 394–401. [Google Scholar] [CrossRef]
- Lindefeldt, M.; Eng, A.; Darban, H.; Bjerkner, A.; Zetterström, C.K.; Allander, T.; Andersson, B.; Borenstein, E.; Dahlin, M.; Prast-Nielsen, S. The ketogenic diet influences taxonomic and functional composition of the gut microbiota in children with severe epilepsy. NPJ Biofilms Microbiomes 2019, 5, 5. [Google Scholar] [CrossRef]
- Geda, Y.E.; Ragossnig, M.; Roberts, L.A.; Roberts, R.O.; Pankratz, V.S.; Christianson, T.J.; Mielke, M.M.; Levine, J.A.; Boeve, B.F.; Sochor, O. Caloric intake, aging, and mild cognitive impairment: A population-based study. J. Alzheimer’s Dis. 2013, 34, 501–507. [Google Scholar] [CrossRef]
- Barker, D.J. The fetal origins of adult disease. Fetal Matern. Med. Rev. 1994, 6, 71–80. [Google Scholar] [CrossRef]
- Gedeon, C.; Nava-Ocampo, A.A.; Koren, G. Ethical issues in pharmacologic research in women undergoing pregnancy termination: A systemic review and survey of researchers. Obstet. Gynecol. Int. 2012, 2012, 724591. [Google Scholar] [CrossRef]
- Cates, W.; Grimes, D.A.; Schulz, K.F. The public health impact of legal abortion: 30 years later. Perspect. Sex. Reprod. Health 2003, 35, 25–28. [Google Scholar] [CrossRef]
- Cheluvappa, R.; Scowen, P.; Eri, R. Ethics of animal research in human disease remediation, its institutional teaching; and alternatives to animal experimentation. Pharmacol. Res. Perspect. 2017, 5, e00332. [Google Scholar] [CrossRef]
- Statista. Number of Chickens Worldwide from 1990 to 2020. Available online: https://www.statista.com/statistics/263962/number-of-chickens-worldwide-since-1990/ (accessed on 21 January 2022).
- Lee, S.; La, T.-M.; Lee, H.-J.; Choi, I.-S.; Song, C.-S.; Park, S.-Y.; Lee, J.-B.; Lee, S.-W. Characterization of microbial communities in the chicken oviduct and the origin of chicken embryo gut microbiota. Sci. Rep. 2019, 9, 6838. [Google Scholar] [CrossRef] [PubMed]
- Zosen, D.; Hadera, M.G.; Lumor, J.S.; Andersen, J.M.; Paulsen, R.E. Chicken embryo as animal model to study drug distribution to the developing brain. J. Pharmacol. Toxicol. Methods 2021, 112, 107105. [Google Scholar] [CrossRef]
- Kue, C.S.; Tan, K.Y.; LaM, M.L.; Lee, H.B. Chick embryo chorioallantoic membrane (CAM): An alternative predictive model in acute toxicological studies for anti-cancer drugs. Exp. Anim. 2015, 64, 129–138. [Google Scholar] [CrossRef] [Green Version]
- Vargas, A.; Zeisser-Labouèbe, M.; Lange, N.; Gurny, R.; Delie, F. The chick embryo and its chorioallantoic membrane (CAM) for the in vivo evaluation of drug delivery systems. Adv. Drug Deliv. Rev. 2007, 59, 1162–1176. [Google Scholar] [CrossRef]
- Zohn, I.E.; Sarkar, A.A. The visceral yolk sac endoderm provides for absorption of nutrients to the embryo during neurulation. Birth Defects Res. Part A Clin. Mol. Teratol. 2010, 88, 593–600. [Google Scholar] [CrossRef] [PubMed]
- Bjørnstad, S.; Austdal, L.P.E.; Roald, B.; Glover, J.C.; Paulsen, R.E. Cracking the egg: Potential of the developing chicken as a model system for nonclinical safety studies of pharmaceuticals. J. Pharmacol. Exp. Ther. 2015, 355, 386–396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guideline, I.H. ICH Guideline S11 on Nonclinical Safety Testing in Support of Development of Paediatric Pharmaceuticals—Step 5. Available online: https://www.ema.europa.eu/en/ich-guideline-s11-nonclinical-safety-testing-support-development-paediatric-pharmaceuticals-step-5 (accessed on 31 March 2020).
- Aden, P.; Goverud, I.; Liestøl, K.; Løberg, E.M.; Paulsen, R.E.; Mæhlen, J.; Lømo, J. Low-potency glucocorticoid hydrocortisone has similar neurotoxic effects as high-potency glucocorticoid dexamethasone on neurons in the immature chicken cerebellum. Brain Res. 2008, 1236, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Aden, P.; Paulsen, R.E.; Mæhlen, J.; Løberg, E.M.; Goverud, I.L.; Liestøl, K.; Lømo, J. Glucocorticoids dexamethasone and hydrocortisone inhibit proliferation and accelerate maturation of chicken cerebellar granule neurons. Brain Res. 2011, 1418, 32–41. [Google Scholar] [CrossRef]
- Mathisen, G.H.; Yazdani, M.; Rakkestad, K.E.; Aden, P.K.; Bodin, J.; Samuelsen, M.; Nygaard, U.C.; Goverud, I.L.; Gaarder, M.; Løberg, E.M. Prenatal exposure to bisphenol A interferes with the development of cerebellar granule neurons in mice and chicken. Int. J. Dev. Neurosci. 2013, 31, 762–769. [Google Scholar] [CrossRef]
- Guy, N.T.; Fadlallah, N.; Naquet, R.; Batini, C. Development of epileptic activity in embryos and newly hatched chicks of the Fayoumi mutant chicken. Epilepsia 1995, 36, 101–107. [Google Scholar] [CrossRef]
- Batini, C.; Telliet, M.; Naquet, R. An avian model of genetic reflex epilepsy. Arch. Ital. Biol. 2004, 142, 297–312. [Google Scholar]
- Douaud, M.; Feve, K.; Pituello, F.; Gourichon, D.; Boitard, S.; Leguern, E.; Coquerelle, G.; Vieaud, A.; Batini, C.; Naquet, R. Epilepsy caused by an abnormal alternative splicing with dosage effect of the SV2A gene in a chicken model. PLoS ONE 2011, 6, e26932. [Google Scholar] [CrossRef]
- Di Pascoli, S.; Puntin, D.; Pinciaroli, A.; Balaban, E.; Pompeiano, M. Design and implementation of a wireless in-ovo EEG/EMG recorder. IEEE Trans. Biomed. Circuits Syst. 2013, 7, 832–840. [Google Scholar] [CrossRef]
- Paonessa, S.; Di Pascoli, S.; Balaban, E.; Vaquero, J.J. Design and development of a wireless infrared EEG recorder for chicken embryos. In Proceedings of the 2016 IEEE International Symposium on Medical Measurements and Applications (MeMeA), Benevento, Italy, 15–18 May 2016; pp. 1–6. [Google Scholar]
- Zhou, Z.; Chen, Z.; Shan, J.; Ma, W.; Li, L.; Zu, J.; Xu, J. Monitoring brain development of chick embryos in vivo using 3.0 T MRI: Subdivision volume change and preliminary structural quantification using DTI. BMC Dev. Biol. 2015, 15, 29. [Google Scholar] [CrossRef] [Green Version]
- Fabene, P.; Sbarbati, A. In vivo MRI in different models of experimental epilepsy. Curr. Drug Targets 2004, 5, 629–636. [Google Scholar] [CrossRef]
- Wang, Y.; Seidel, J.; Tsui, B.M.; Vaquero, J.J.; Pomper, M.G. Performance evaluation of the GE healthcare eXplore VISTA dual-ring small-animal PET scanner. J. Nucl. Med. 2006, 47, 1891–1900. [Google Scholar]
- Balaban, E.; Desco, M.; Vaquero, J.-J. Waking-like brain function in embryos. Curr. Biol. 2012, 22, 852–861. [Google Scholar] [CrossRef] [Green Version]
- Rosa-Salva, O.; Mayer, U.; Versace, E.; Hébert, M.; Lemaire, B.S.; Vallortigara, G. Sensitive periods for social development: Interactions between predisposed and learned mechanisms. Cognition 2021, 213, 104552. [Google Scholar] [CrossRef] [PubMed]
- De Haas, E.N.; Newberry, R.C.; Edgar, J.; Riber, A.B.; Estevez, I.; Ferrante, V.; Hernandez, C.E.; Kjaer, J.B.; Ozkan, S.; Dimitrov, I. Prenatal and Early Postnatal Behavioural Programming in Laying Hens, with Possible Implications for the Development of Injurious Pecking. Front. Vet. Sci. 2021, 8, 693. [Google Scholar] [CrossRef] [PubMed]
- Marino, L. Thinking chickens: A review of cognition, emotion, and behavior in the domestic chicken. Anim. Cogn. 2017, 20, 127–147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jarvis, E.D.; Güntürkün, O.; Bruce, L.; Csillag, A.; Karten, H.; Kuenzel, W.; Medina, L.; Paxinos, G.; Perkel, D.J.; Shimizu, T. Avian brains and a new understanding of vertebrate brain evolution. Nat. Rev. Neurosci. 2005, 6, 151–159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Groef, B.; Grommen, S.; Darras, V. The chicken embryo as a model for developmental endocrinology: Development of the thyrotropic, corticotropic, and somatotropic axes. Mol. Cell. Endocrinol. 2008, 293, 17–24. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Himmel, H.M. Safety pharmacology assessment of central nervous system function in juvenile and adult rats: Effects of pharmacological reference compounds. J. Pharmacol. Toxicol. Methods 2008, 58, 129–146. [Google Scholar] [CrossRef] [PubMed]
- Walther, A.; Rice, T.; Kufert, Y.; Ehlert, U. Neuroendocrinology of a male-specific pattern for depression linked to alcohol use disorder and suicidal behavior. Front. Psychiatry 2017, 7, 206. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Kuang, S.; Applegate, T.J.; Lin, T.-L.; Cheng, H.-W. The development of the serotonergic and dopaminergic systems during chicken mid-late embryogenesis. Mol. Cell. Endocrinol. 2019, 493, 110472. [Google Scholar] [CrossRef]
- Cassiers, L.L.; Niemegeers, P.; Fransen, E.; Morrens, M.; De Boer, P.; Van Nueten, L.; Claes, S.; Sabbe, B.G.; Van Den Eede, F. Neuroendocrine and inflammatory effects of childhood trauma following psychosocial and inflammatory stress in women with remitted major depressive disorder. Brain Sci. 2019, 9, 375. [Google Scholar] [CrossRef] [Green Version]
- Blier, P.; El Mansari, M. Serotonin and beyond: Therapeutics for major depression. Philos. Trans. R. Soc. B Biol. Sci. 2013, 368, 20120536. [Google Scholar] [CrossRef]
- Wise, P.M.; Frye, B. Functional development of the hypothalamo-hypophyseal-adrenal cortex axis in the chick embryo, Gallus domesticus. J. Exp. Zool. 1973, 185, 277–291. [Google Scholar] [CrossRef] [Green Version]
- Jenkins, S.; Porter, T. Ontogeny of the hypothalamo–pituitary–adrenocortical axis in the chicken embryo: A review. Domest. Anim. Endocrinol. 2004, 26, 267–275. [Google Scholar] [CrossRef]
- Gastón, M.S.; Cid, M.P.; Salvatierra, N.A. Bicuculline, a GABAA-receptor antagonist, blocked HPA axis activation induced by ghrelin under an acute stress. Behav. Brain Res. 2017, 320, 464–472. [Google Scholar] [CrossRef]
- Gualtieri, F.; Armstrong, E.; Longmoor, G.; D’eath, R.; Sandilands, V.; Boswell, T.; Smulders, T. Unpredictable chronic mild stress suppresses the incorporation of new neurons at the caudal pole of the chicken hippocampal formation. Sci. Rep. 2019, 9, 7129. [Google Scholar] [CrossRef] [Green Version]
- Smulders, T.V. The avian hippocampal formation and the stress response. Brain Behav. Evol. 2017, 90, 81–91. [Google Scholar] [CrossRef]
- Smulders, T.V. Telencephalic regulation of the HPA axis in birds. Neurobiol. Stress 2021, 15, 100351. [Google Scholar] [CrossRef]
- Christensen, J.; Grønborg, T.K.; Sørensen, M.J.; Schendel, D.; Parner, E.T.; Pedersen, L.H.; Vestergaard, M. Prenatal valproate exposure and risk of autism spectrum disorders and childhood autism. JAMA 2013, 309, 1696–1703. [Google Scholar] [CrossRef] [Green Version]
- Nicolini, C.; Fahnestock, M. The valproic acid-induced rodent model of autism. Exp. Neurol. 2018, 299, 217–227. [Google Scholar] [CrossRef]
- Nishigori, H.; Kagami, K.; Takahashi, A.; Tezuka, Y.; Sanbe, A.; Nishigori, H. Impaired social behavior in chicks exposed to sodium valproate during the last week of embryogenesis. Psychopharmacology 2013, 227, 393–402. [Google Scholar] [CrossRef]
- Lorenzi, E.; Pross, A.; Rosa-Salva, O.; Versace, E.; Sgadò, P.; Vallortigara, G. Embryonic Exposure to Valproic Acid Affects Social Predispositions for Dynamic Cues of Animate Motion in Newly-Hatched Chicks. Front. Physiol. 2019, 10, 501. [Google Scholar] [CrossRef] [Green Version]
- Zachar, G.; Tóth, A.; Gerecsei, L.; Zsebők, S.; Ádám, Á.; Csillag, A. Valproate Exposure in ovo Attenuates the Acquisition of Social Preferences of Young Post-hatch Domestic Chicks. Front. Physiol. 2019, 10, 881. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sgadò, P.; Rosa-Salva, O.; Versace, E.; Vallortigara, G. Embryonic Exposure to Valproic Acid Impairs Social Predispositions of Newly-Hatched Chicks. Sci. Rep. 2018, 8, 5919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Anderson, K.N.; Lind, J.N.; Simeone, R.M.; Bobo, W.V.; Mitchell, A.A.; Riehle-Colarusso, T.; Polen, K.N.; Reefhuis, J. Maternal use of specific antidepressant medications during early pregnancy and the risk of selected birth defects. JAMA Psychiatry 2020, 77, 1246–1255. [Google Scholar] [CrossRef] [PubMed]
- Castelbaum, L.; Sylvester, C.M.; Zhang, Y.; Yu, Q.; Constantino, J.N. On the nature of monozygotic twin concordance and discordance for autistic trait severity: A quantitative analysis. Behav. Genet. 2019, 50, 263–272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fitzpatrick, S.E.; Srivorakiat, L.; Wink, L.K.; Pedapati, E.V.; Erickson, C.A. Aggression in autism spectrum disorder: Presentation and treatment options. Neuropsychiatr. Dis. Treat. 2016, 12, 1525–1538. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duclot, F.; Kabbaj, M. Epigenetics of Aggression; Springer: Berlin/Heidelberg, Germany, 2021. [Google Scholar]
- Lopez-Duran, N.L.; Olson, S.L.; Hajal, N.J.; Felt, B.T.; Vazquez, D.M. Hypothalamic pituitary adrenal axis functioning in reactive and proactive aggression in children. J. Abnorm. Child Psychol. 2009, 37, 169–182. [Google Scholar] [CrossRef]
- Yan, H.; Boutet, A.; Mithani, K.; Germann, J.; Elias, G.J.; Yau, I.; Go, C.; Kalia, S.K.; Lozano, A.M.; Fasano, A. Aggressiveness after centromedian nucleus stimulation engages prefrontal thalamocortical circuitry. Brain Stimul. 2020, 13, 357–359. [Google Scholar] [CrossRef] [Green Version]
- Dennis, R.L.; Fahey, A.G.; Cheng, H.-W. Alterations to embryonic serotonin change aggression and fearfulness. Aggress. Behav. 2013, 39, 91–98. [Google Scholar] [CrossRef]
- Dennis, R.; Cheng, H.-W. The dopaminergic system and aggression in laying hens. Poult. Sci. 2011, 90, 2440–2448. [Google Scholar] [CrossRef]
- Cheng, H.-W.; Muir, W. Chronic social stress differentially regulates neuroendocrine responses in laying hens: II. Genetic basis of adrenal responses under three different social conditions. Psychoneuroendocrinology 2004, 29, 961–971. [Google Scholar] [CrossRef]
- Huang, X.; Kuang, S.; Applegate, T.J.; Lin, T.-L.; Cheng, H.-W. Prenatal serotonin fluctuation affects serotoninergic development and related neural circuits in chicken embryos. Neuroscience 2021, 473, 66–80. [Google Scholar] [CrossRef]
- Höglund, E.; Øverli, Ø.; Winberg, S. Tryptophan metabolic pathways and brain serotonergic activity: A comparative review. Front. Endocrinol. 2019, 10, 158. [Google Scholar] [CrossRef]
- Hoshino, Y.; Yamamoto, T.; Kaneko, M.; Kumashiro, H. Plasma free tryptophan concentration in autistic children. Brain Dev. 1986, 8, 424–427. [Google Scholar] [CrossRef]
- Daigle, C.L.; Rodenburg, T.B.; Bolhuis, J.E.; Swanson, J.C.; Siegford, J.M. Individual consistency of feather pecking behavior in laying hens: Once a feather pecker always a feather pecker? Front. Vet. Sci. 2015, 2, 6. [Google Scholar] [CrossRef] [Green Version]
- Huang, X.; Hu, J.; Peng, H.; Cheng, H.-W. Embryonic exposure to tryptophan yields bullying victimization via reprogramming the microbiota-gut-brain axis. Nutrients 2022, 14, 661. [Google Scholar] [CrossRef]
- Saffarian, A.; Mulet, C.; Regnault, B.; Amiot, A.; Tran-Van-Nhieu, J.; Ravel, J.; Sobhani, I.; Sansonetti, P.J.; Pedron, T. Crypt-and mucosa-associated core microbiotas in humans and their alteration in colon cancer patients. mBio 2019, 10, e01315–e01319. [Google Scholar] [CrossRef] [Green Version]


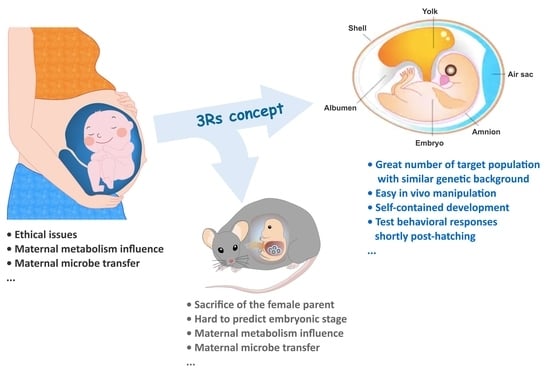
| Species | Human | Rodent | Chicken |
|---|---|---|---|
| Advantages |
|
|
|
| Disadvantages |
|
|
|
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Huang, X.; Cheng, H.-w. Perspective: Chicken Models for Studying the Ontogenetic Origin of Neuropsychiatric Disorders. Biomedicines 2022, 10, 1155. https://doi.org/10.3390/biomedicines10051155
Huang X, Cheng H-w. Perspective: Chicken Models for Studying the Ontogenetic Origin of Neuropsychiatric Disorders. Biomedicines. 2022; 10(5):1155. https://doi.org/10.3390/biomedicines10051155
Chicago/Turabian StyleHuang, Xiaohong, and Heng-wei Cheng. 2022. "Perspective: Chicken Models for Studying the Ontogenetic Origin of Neuropsychiatric Disorders" Biomedicines 10, no. 5: 1155. https://doi.org/10.3390/biomedicines10051155