Effect of Vitamin D on Graft-versus-Host Disease
Abstract
:1. Introduction
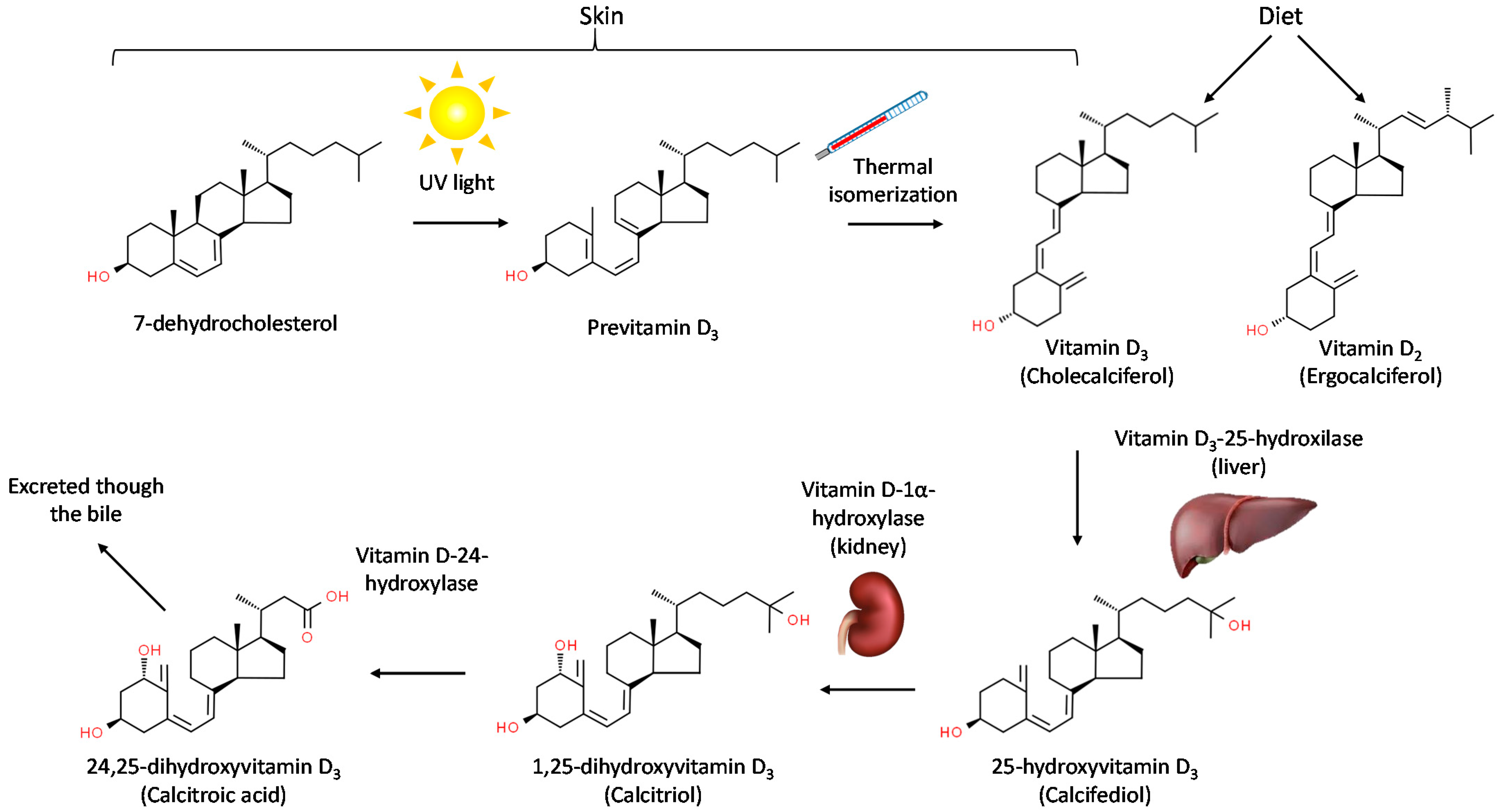
2. Chemical Structure, Synthesis and Metabolism of Vitamin D
3. Classical and Non-Classical Effects of Vitamin D
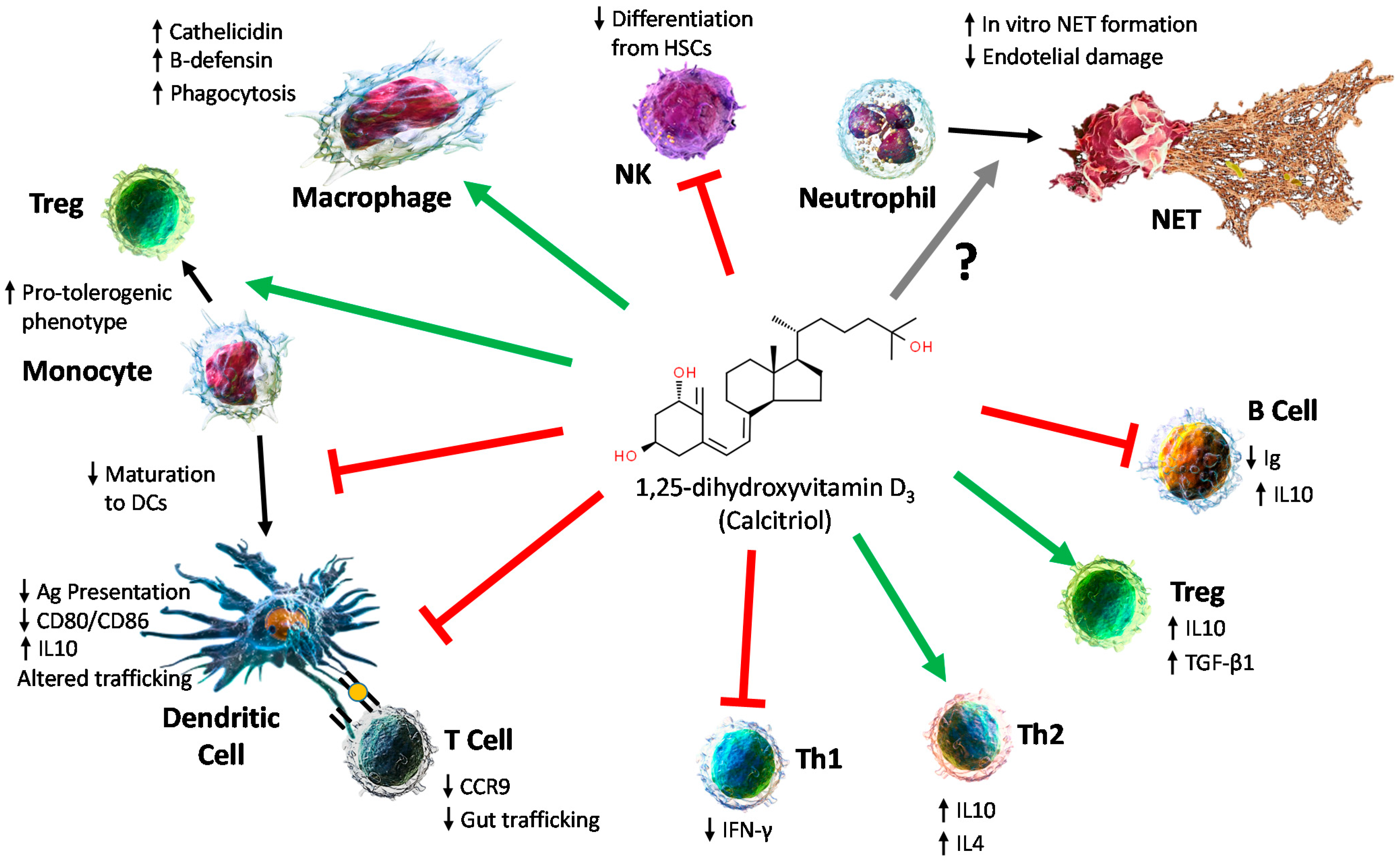
4. Effects on the Immune System
4.1. Effect on Innate Immune Cells
4.2. Effect on Adaptative Immunity
4.3. Preclinical Models of Vitamin D in Immune Diseases and Solid Organ Transplantation
5. Vitamin D in the Clinical Setting
Vitamin D Compounds Available in the Clinical Setting
6. Vitamin D and Hematologic Malignancies
7. Vitamin D and Allogeneic Stem-Cell Transplantation: Effect on Graft-Versus-Host Disease (GvHD)
7.1. Vitamin D Levels: Impacts on Allogenic HSCT Outcomes
7.2. Studies Evaluating the Efficacy of Vitamin D Administration
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Feldman, D.; Pike, W.J.; Glorieux, F.H. Vitamin D. Vitam. D 2005, 1–2, 1489–1510. [Google Scholar] [CrossRef]
- Sarafin, K.; Durazo-Arvizu, R.; Tian, L.; Phinney, K.W.; Tai, S.; Camara, J.E.; Merkel, J.; Green, E.; Sempos, C.T.; Brooks, S.P.J. Standardizing 25-hydroxyvitamin D values from the Canadian Health Measures Survey. Am. J. Clin. Nutr. 2015, 102, 1044–1050. [Google Scholar] [CrossRef] [Green Version]
- Schleicher, R.L.; Sternberg, M.R.; Looker, A.C.; Yetley, E.A.; Lacher, D.A.; Sempos, C.T.; Taylor, C.L.; Durazo-Arvizu, R.A.; Maw, K.L.; Chaudhary-Webb, M.; et al. National Estimates of Serum Total 25-Hydroxyvitamin D and Metabolite Concentrations Measured by Liquid Chromatography–Tandem Mass Spectrometry in the US Population during 2007–2010. J. Nutr. 2016, 146, 1051–1061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cashman, K.D. Vitamin D Deficiency: Defining, Prevalence, Causes, and Strategies of Addressing. Calcif. Tissue Int. 2020, 106, 14–29. [Google Scholar] [CrossRef]
- Cashman, K.D.; Dowling, K.G.; Škrabáková, Z.; Gonzalez-Gross, M.; Valtueña, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amrein, K.; Scherkl, M.; Hoffmann, M.; Neuwersch-Sommeregger, S.; Köstenberger, M.; Tmava Berisha, A.; Martucci, G.; Pilz, S.; Malle, O. Vitamin D deficiency 2.0: An update on the current status worldwide. Eur. J. Clin. Nutr. 2020, 74, 1498–1513. [Google Scholar] [CrossRef]
- Dusso, A.S.; Brown, A.J.; Slatopolsky, E. Vitamin D. Am. J. Physiol.-Ren. Physiol. 2005, 289, 8–28. [Google Scholar] [CrossRef]
- Mellanby Discussion on the Importance of Accessory Food Factors (Vitamines) in the Feeding of Infants. Proc. R. Soc. Med. 1920, 13, 95–98. [CrossRef]
- Lawson, D.E.M.; Fraser, D.R.; Kodicek, E.; Morris, H.R.; Williams, D.H. Identification of 1,25-dihydroxycholecalciferol, a new kidney hormone controlling calcium metabolism. Nature 1971, 230, 228–230. [Google Scholar] [CrossRef]
- Holick, M.F.; Schnoes, H.K.; DeLuca, H.F.; Suda, T.; Cousins, R.J. Isolation and Identification of 1,25-Dihydroxycholecalciferol. a Metabolite of Vitamin D Active in Intestine. Biochemistry 1971, 10, 2799–2804. [Google Scholar] [CrossRef] [PubMed]
- Norman, A.W.; Myrtle, J.F.; Midgett, R.J.; Nowicki, H.G.; Williams, V.; Popják, G. 1,25-Dihydroxy cholecalciferol: Identification of the proposed active form of vitamin D3 in the intestine. Science 1971, 173, 51–54. [Google Scholar] [CrossRef] [PubMed]
- Fu, G.K.; Lin, D.; Zhang, M.Y.H.; Bikle, D.D.; Shackleton, C.H.L.; Miller, W.L.; Portale, A.A. Cloning of human 25-hydroxyvitamin D-1 alpha-hydroxylase and mutations causing vitamin D-dependent rickets type 1. Mol. Endocrinol. 1997, 11, 1961–1970. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Monkawa, T.; Yoshida, T.; Wakino, S.; Shinki, T.; Anazawa, H.; DeLuca, H.F.; Suda, T.; Hayashi, M.; Saruta, T. Molecular cloning of cDNA and genomic DNA for human 25-hydroxyvitamin D3 1 alpha-hydroxylase. Biochem. Biophys. Res. Commun. 1997, 239, 527–533. [Google Scholar] [CrossRef]
- Jones, G.; Prosser, D.E.; Kaufmann, M. 25-Hydroxyvitamin D-24-hydroxylase (CYP24A1): Its important role in the degradation of vitamin D. Arch. Biochem. Biophys. 2012, 523, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Haussler, M.R.; Whitfield, G.K.; Kaneko, I.; Haussler, C.A.; Hsieh, D.; Hsieh, J.-C.; Jurutka, P.W. Molecular mechanisms of vitamin D action. Calcif. Tissue Int. 2013, 92, 77–98. [Google Scholar] [CrossRef] [PubMed]
- Brown, A.J.; Dusso, A.; Slatopolsky, E. Vitamin D. Am. J. Physiol.-Ren. Physiol. 1999, 277, 157–175. [Google Scholar] [CrossRef]
- Cheskis, B.; Freedman, L.P. Ligand modulates the conversion of DNA-bound vitamin D3 receptor (VDR) homodimers into VDR-retinoid X receptor heterodimers. Mol. Cell. Biol. 1994, 14, 3329–3338. [Google Scholar] [CrossRef] [PubMed]
- Umesono, K.; Murakami, K.K.; Thompson, C.C.; Evans, R.M. Direct repeats as selective response elements for the thyroid hormone, retinoic acid, and vitamin D3 receptors. Cell 1991, 65, 1255–1266. [Google Scholar] [CrossRef]
- Bikle, D.D. Ligand-Independent Actions of the Vitamin D Receptor: More Questions Than Answers. JBMR Plus 2021, 5. [Google Scholar] [CrossRef] [PubMed]
- Skorija, K.; Cox, M.; Sisk, J.M.; Dowd, D.R.; MacDonald, P.N.; Thompson, C.C.; Demay, M.B. Ligand-Independent Actions of the Vitamin D Receptor Maintain Hair Follicle Homeostasis. Mol. Endocrinol. 2005, 19, 855–862. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ellison, T.I.; Smith, M.K.; Gilliam, A.C.; MacDonald, P.N. Inactivation of the Vitamin D Receptor Enhances Susceptibility of Murine Skin to UV-Induced Tumorigenesis. J. Investig. Dermatol. 2008, 128, 2508–2517. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Harris, S.S.; Eccleshall, T.R.; Gross, C.; Dawson-Hughes, B.; Feldman, D. The vitamin D receptor start codon polymorphism (FokI) and bone mineral density in premenopausal American black and white women. J. Bone Miner. Res. 1997, 12, 1043–1048. [Google Scholar] [CrossRef]
- Faraco, J.H.; Morrison, N.A.; Baker, A.; Shine, J.; Frossard, P.M. ApaI dimorphism at the human vitamin D receptor gene locus. Nucleic Acids Res. 1989, 17, 2150. [Google Scholar] [CrossRef]
- Morrison, N.A.; Yeoman, R.; Kelly, P.J.; Eisman, J.A. Contribution of trans-acting factor alleles to normal physiological variability: Vitamin D receptor gene polymorphism and circulating osteocalcin. Proc. Natl. Acad. Sci. USA 1992, 89, 6665–6669. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Morrison, N.A.; Qi, J.C.; Tokita, A.; Kelly, P.J.; Crofts, L.; Nguyen, T.V.; Sambrook, P.N.; Eisman, J.A. Prediction of bone density from vitamin D receptor alleles. Nature 1994, 367, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Uitterlinden, A.G.; Fang, Y.; van Meurs, J.B.J.; Pols, H.A.P.; van Leeuwen, J.P.T.M. Genetics and biology of vitamin D receptor polymorphisms. Gene 2004, 338, 143–156. [Google Scholar] [CrossRef] [Green Version]
- Uitterlinden, A.G.; Pols, H.A.P.; Burger, H.; Huang, Q.; Van Daele, P.L.A.; Van Duijn, C.M.; Hofman, A.; Birkenhäger, J.C.; Van Leeuwen, J.P.T.M. A large-scale population-based study of the association of vitamin D receptor gene polymorphisms with bone mineral density. J. Bone Miner. Res. 1996, 11, 1241–1248. [Google Scholar] [CrossRef] [Green Version]
- Van Etten, E.; Verlinden, L.; Giulietti, A.; Ramos-Lopez, E.; Branisteanu, D.D.; Ferreira, G.B.; Overbergh, L.; Verstuyf, A.; Bouillon, R.; Roep, B.O.; et al. The vitamin D receptor gene FokI polymorphism: Functional impact on the immune system. Eur. J. Immunol. 2007, 37, 395–405. [Google Scholar] [CrossRef]
- Bikle, D. Nonclassic actions of vitamin D. J. Clin. Endocrinol. Metab. 2009, 94, 26–34. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Provvedini, D.M.; Tsoukas, C.D.; Deftos, L.J.; Manolagas, S.C. 1,25-Dihydroxyvitamin D3 Receptors in Human Leukocytes. Science 1983, 221, 1181–1183. [Google Scholar] [CrossRef]
- Chen, W.C.; Vayuvegula, B.; Gupta, S. 1,25-Dihydroxyvitamin D3-mediated inhibition of human B cell differentiation. Clin. Exp. Immunol. 1987, 69, 639–646. [Google Scholar]
- Ohta, M.; Okabe, T.; Ozawa, K.; Urabe, A.; Takaku, F. 1α,25-Dihydroxy vitamin D3 (calcitriol) stimulates proliferation of human circulating monocytes in vitro. FEBS Lett. 1985, 185, 9–13. [Google Scholar] [CrossRef] [Green Version]
- Hodler, B.; EvÊquoz, V.; Trechsel, U.; Fleisch, H.; Stadler, B. Influence of Vitamin D3 Metabolites on the Production of Interleukins 1, 2 and 3. Immunobiology 1985, 170, 256–269. [Google Scholar] [CrossRef]
- Lemire, J.M.; Adams, J.S.; Kermani-Arab, V.; Bakke, A.C.; Sakai, R.; Jordan, S.C. 1,25-Dihydroxyvitamin D3 suppresses human T helper/inducer lymphocyte activity in vitro. J. Immunol. 1985, 134, 3032–3035. [Google Scholar]
- Lemire, J.M.; Adams, J.S.; Sakai, R.; Jordan, S.C. 1 alpha,25-dihydroxyvitamin D3 suppresses proliferation and immunoglobulin production by normal human peripheral blood mononuclear cells. J. Clin. Investig. 1984, 74, 657–661. [Google Scholar] [CrossRef] [Green Version]
- Abe, E.; Miyaura, C.; Sakagami, H.; Takeda, M.; Konno, K.; Yamazaki, T.; Yoshiki, S.; Suda, T. Differentiation of mouse myeloid leukemia cells induced by 1 alpha,25-dihydroxyvitamin D3. Proc. Natl. Acad. Sci. USA 1981, 78, 4990–4994. [Google Scholar] [CrossRef] [Green Version]
- Davies, P.D.O. A possible link between vitamin d deficiency and impaired host defence to mycobacterium tuberculosis. Tubercle 1985, 66, 301–306. [Google Scholar] [CrossRef]
- Green, M. Cod liver oil and tuberculosis. BMJ 2011, 343, d7505. [Google Scholar] [CrossRef]
- Rolf, L.; Muris, A.H.; Hupperts, R.; Damoiseaux, J. Vitamin D effects on B cell function in autoimmunity. Ann. N. Y. Acad. Sci. 2014, 1317, 84–91. [Google Scholar] [CrossRef]
- Veldman, C.M.; Cantorna, M.T.; DeLuca, H.F. Expression of 1,25-dihydroxyvitamin D(3) receptor in the immune system. Arch. Biochem. Biophys. 2000, 374, 334–338. [Google Scholar] [CrossRef] [PubMed]
- Edfeldt, K.; Liu, P.T.; Chun, R.; Fabri, M.; Schenk, M.; Wheelwright, M.; Keegan, C.; Krutzik, S.R.; Adams, J.S.; Hewison, M.; et al. T-cell cytokines differentially control human monocyte antimicrobial responses by regulating vitamin D metabolism. Proc. Natl. Acad. Sci. USA 2010, 107, 22593–22598. [Google Scholar] [CrossRef] [Green Version]
- Baeke, F.; Korf, H.; Overbergh, L.; van Etten, E.; Verstuyf, A.; Gysemans, C.; Mathieu, C. Human T lymphocytes are direct targets of 1,25-dihydroxyvitamin D3 in the immune system. J. Steroid Biochem. Mol. Biol. 2010, 121, 221–227. [Google Scholar] [CrossRef]
- Baeke, F.; Takiishi, T.; Korf, H.; Gysemans, C.; Mathieu, C. Vitamin D: Modulator of the immune system. Curr. Opin. Pharmacol. 2010, 10, 482–496. [Google Scholar] [CrossRef]
- Hewison, M. An update on vitamin D and human immunity. Clin. Endocrinol. 2012, 76, 315–325. [Google Scholar] [CrossRef] [PubMed]
- Liu, P.T.; Stenger, S.; Li, H.; Wenzel, L.; Tan, B.H.; Krutzik, S.R.; Ochoa, M.T.; Schauber, J.; Wu, K.; Meinken, C.; et al. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science 2006, 311, 1770–1773. [Google Scholar] [CrossRef] [PubMed]
- Gombart, A.F.; Borregaard, N.; Koeffler, H.P. Human cathelicidin antimicrobial peptide (CAMP) gene is a direct target of the vitamin D receptor and is strongly up-regulated in myeloid cells by 1,25-dihydroxyvitamin D3. FASEB J. 2005, 19, 1067–1077. [Google Scholar] [CrossRef] [Green Version]
- Wang, T.-T.; Nestel, F.P.; Bourdeau, V.; Nagai, Y.; Wang, Q.; Liao, J.; Tavera-Mendoza, L.; Lin, R.; Hanrahan, J.W.; Mader, S.; et al. Cutting Edge: 1,25-Dihydroxyvitamin D3 Is a Direct Inducer of Antimicrobial Peptide Gene Expression. J. Immunol. 2004, 173, 2909–2912. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Leung, D.Y.M.; Richers, B.N.; Liu, Y.; Remigio, L.K.; Riches, D.W.; Goleva, E. Vitamin D Inhibits Monocyte/Macrophage Proinflammatory Cytokine Production by Targeting MAPK Phosphatase-1. J. Immunol. 2012, 188, 2127–2135. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fabri, M.; Stenger, S.; Shin, D.M.; Yuk, J.M.; Liu, P.T.; Realegeno, S.; Lee, H.M.; Krutzik, S.R.; Schenk, M.; Sieling, P.A.; et al. Vitamin D is required for IFN-γ-mediated antimicrobial activity of human macrophages. Sci. Transl. Med. 2011, 3, 104ra102. [Google Scholar] [CrossRef] [Green Version]
- Gauzzi, M.C.; Purificato, C.; Donato, K.; Jin, Y.; Wang, L.; Daniel, K.C.; Maghazachi, A.A.; Belardelli, F.; Adorini, L.; Gessani, S. Suppressive Effect of 1α,25-Dihydroxyvitamin D 3 on Type I IFN-Mediated Monocyte Differentiation into Dendritic Cells: Impairment of Functional Activities and Chemotaxis. J. Immunol. 2005, 174, 270–276. [Google Scholar] [CrossRef] [Green Version]
- Small, A.G.; Harvey, S.; Kaur, J.; Putty, T.; Quach, A.; Munawara, U.; Perveen, K.; McPhee, A.; Hii, C.S.; Ferrante, A. Vitamin D upregulates the macrophage complement receptor immunoglobulin in innate immunity to microbial pathogens. Commun. Biol. 2021, 4, 401. [Google Scholar] [CrossRef] [PubMed]
- Merad, M.; Sathe, P.; Helft, J.; Miller, J.; Mortha, A. The Dendritic Cell Lineage: Ontogeny and Function of Dendritic Cells and Their Subsets in the Steady State and the Inflamed Setting. Annu. Rev. Immunol. 2013, 31, 563–604. [Google Scholar] [CrossRef] [Green Version]
- Bscheider, M.; Butcher, E.C. Vitamin D immunoregulation through dendritic cells. Immunology 2016, 148, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Penna, G.; Adorini, L. 1α,25-Dihydroxyvitamin D 3 Inhibits Differentiation, Maturation, Activation, and Survival of Dendritic Cells Leading to Impaired Alloreactive T Cell Activation. J. Immunol. 2000, 164, 2405–2411. [Google Scholar] [CrossRef] [Green Version]
- Griffin, M.D.; Lutz, W.; Phan, V.A.; Bachman, L.A.; McKean, D.J.; Kumar, R. Dendritic cell modulation by 1α,25 dihydroxyvitamin D3 and its analogs: A vitamin D receptor-dependent pathway that promotes a persistent state of immaturity in vitro and in vivo. Proc. Natl. Acad. Sci. USA 2001, 98, 6800–6805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferreira, G.B.; Vanherwegen, A.S.; Eelen, G.; Gutiérrez, A.C.F.; VanLommel, L.; Marchal, K.; Verlinden, L.; Verstuyf, A.; Nogueira, T.; Georgiadou, M.; et al. Vitamin D3 Induces Tolerance in Human Dendritic Cells by Activation of Intracellular Metabolic Pathways. Cell Rep. 2015, 10, 711–725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Enioutina, E.Y.; Bareyan, D.; Daynes, R.A. Vitamin D3-mediated alterations to myeloid dendritic cell trafficking in vivo expand the scope of their antigen presenting properties. Vaccine 2007, 25, 1236–1249. [Google Scholar] [CrossRef] [PubMed]
- Sigmundsdottir, H.; Pan, J.; Debes, G.F.; Alt, C.; Habtezion, A.; Soler, D.; Butcher, E.C. DCs metabolize sunlight-induced vitamin D3 to “program” T cell attraction to the epidermal chemokine CCL27. Nat. Immunol. 2007, 8, 285–293. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, K.; Nakayama, Y.; Horiuchi, H.; Ohta, T.; Komoriya, K.; Ohmori, H.; Kamimura, T. Human neutrophils express messenger RNA of vitamin D receptor and respond to 1alpha,25-dihydroxyvitamin D3. Immunopharmacol. Immunotoxicol. 2002, 24, 335–347. [Google Scholar] [CrossRef]
- Boeltz, S.; Amini, P.; Anders, H.J.; Andrade, F.; Bilyy, R.; Chatfield, S.; Cichon, I.; Clancy, D.M.; Desai, J.; Dumych, T.; et al. To NET or not to NET:current opinions and state of the science regarding the formation of neutrophil extracellular traps. Cell Death Differ. 2019, 26, 395–408. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nat. Rev. Immunol. 2018, 18, 134–147. [Google Scholar] [CrossRef]
- Agraz-Cibrian, J.M.; Giraldo, D.M.; Urcuqui-Inchima, S. 1,25-Dihydroxyvitamin D3 induces formation of neutrophil extracellular trap-like structures and modulates the transcription of genes whose products are neutrophil extracellular trap-associated proteins: A pilot study. Steroids 2019, 141, 14–22. [Google Scholar] [CrossRef]
- Weeres, M.A.; Robien, K.; Ahn, Y.-O.; Neulen, M.-L.; Bergerson, R.; Miller, J.S.; Verneris, M.R. The Effects of 1,25-Dihydroxyvitamin D3 on In Vitro Human NK Cell Development from Hematopoietic Stem Cells. J. Immunol. 2014, 193, 3456–3462. [Google Scholar] [CrossRef] [Green Version]
- Joseph, R.W.; Bayraktar, U.D.; Kim, T.K.; John, S.L.S.; Popat, U.; Khalili, J.; Molldrem, J.J.; Wieder, E.D.; Komanduri, K.V. Vitamin D receptor upregulation in alloreactive human T cells. Hum. Immunol. 2012, 73, 693–698. [Google Scholar] [CrossRef]
- Hayes, C.E.; Hubler, S.L.; Moore, J.R.; Barta, L.E.; Praska, C.E.; Nashold, F.E. Vitamin D actions on CD4+ T cells in autoimmune disease. Front. Immunol. 2015, 6, 100. [Google Scholar] [CrossRef] [PubMed]
- O’Kelly, J.; Hisatake, J.; Hisatake, Y.; Bishop, J.; Norman, A.; Koeffler, H.P.; O’Kelly, J.; Hisatake, J.; Hisatake, Y.; Bishop, J.; et al. Normal myelopoiesis but abnormal T lymphocyte responses in vitamin D receptor knockout mice. J. Clin. Investig. 2002, 109, 1091–1099. [Google Scholar] [CrossRef]
- Yu, S.; Bruce, D.; Froicu, M.; Weaver, V.; Cantorna, M.T. Failure of T cell homing, reduced CD4/CD8αα intraepithelial lymphocytes, and inflammation in the gut of vitamin D receptor KO mice. Proc. Natl. Acad. Sci. USA 2008, 105, 20834–20839. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lemire, J.M.; Archer, D.C.; Beck, L.; Spiegelberg, H.L. Immunosuppressive actions of 1,25-dihydroxyvitamin D3: Preferential inhibition of Th1 functions. J. Nutr. 1995, 125, 1704S–1708S. [Google Scholar] [CrossRef] [PubMed]
- Von Essen, M.R.; Kongsbak, M.; Schjerling, P.; Olgaard, K.; Ødum, N.; Geisler, C. Vitamin D controls T cell antigen receptor signaling and activation of human T cells. Nat. Immunol. 2010, 11, 344–349. [Google Scholar] [CrossRef]
- Chambers, E.S.; Hawrylowicz, C.M. The impact of vitamin D on regulatory T cells. Curr. Allergy Asthma Rep. 2011, 11, 29–36. [Google Scholar] [CrossRef]
- Unger, W.W.J.J.; Laban, S.; Kleijwegt, F.S.; Van Der Slik, A.R.; Roep, B.O. Induction of Treg by monocyte-derived DC modulated by vitamin D3 or dexamethasone: Differential role for PD-L1. Eur. J. Immunol. 2009, 39, 3147–3159. [Google Scholar] [CrossRef]
- Gregori, S.; Casorati, M.; Amuchastegui, S.; Smiroldo, S.; Davalli, A.M.; Adorini, L. Regulatory T Cells Induced by 1α,25-Dihydroxyvitamin D 3 and Mycophenolate Mofetil Treatment Mediate Transplantation Tolerance. J. Immunol. 2001, 167, 1945–1953. [Google Scholar] [CrossRef] [Green Version]
- Urry, Z.; Chambers, E.S.; Xystrakis, E.; Dimeloe, S.; Richards, D.F.; Gabryšová, L.; Christensen, J.; Gupta, A.; Saglani, S.; Bush, A.; et al. The role of 1α,25-dihydroxyvitamin D3 and cytokines in the promotion of distinct Foxp3+and IL-10+ CD4+ T cells. Eur. J. Immunol. 2012, 42, 2697–2708. [Google Scholar] [CrossRef] [Green Version]
- Zhou, Q.; Qin, S.; Zhang, J.; Zhon, L.; Pen, Z.; Xing, T. 1,25(OH)2D3 induces regulatory T cell differentiation by influencing the VDR/PLC-γ1/TGF-β1/pathway. Mol. Immunol. 2017, 91, 156–164. [Google Scholar] [CrossRef]
- Chen, S.S.S.; Sims, G.P.; Chen, X.X.; Gu, Y.Y.; Chen, S.S.S.; Lipsky, P.E. Modulatory effects of 1,25-dihydroxyvitamin D3 on human B cell differentiation. J. Immunol. 2007, 179, 1634–1647. [Google Scholar] [CrossRef] [Green Version]
- Provvedini, D.M.; Tsoukas, C.D.; Deftos, L.J.; Manolagas, S.C. 1 alpha,25-Dihydroxyvitamin D3-binding macromolecules in human B lymphocytes: Effects on immunoglobulin production. J. Immunol. 1986, 136, 2734–2740. [Google Scholar] [PubMed]
- Iho, S.; Takahashi, T.; Kura, F.; Sugiyama, H.; Hoshino, T. The effect of 1,25-dihydroxyvitamin D3 on in vitro immunoglobulin production in human B cells. J. Immunol. 1986, 136, 4427–4431. [Google Scholar]
- Hartmann, B.; Heine, G.; Babina, M.; Steinmeyer, A.; Zügel, U.; Radbruch, A.; Worm, M. Targeting the vitamin D receptor inhibits the B cell-dependent allergic immune response. Allergy Eur. J. Allergy Clin. Immunol. 2011, 66, 540–548. [Google Scholar] [CrossRef]
- Heine, G.; Niesner, U.; Chang, H.D.; Steinmeyer, A.; Zügel, U.; Zuberbier, T.; Radbruch, A.; Worm, M. 1,25-dihydroxyvitamin D3 promotes IL-10 production in human B cells. Eur. J. Immunol. 2008, 38, 2210–2218. [Google Scholar] [CrossRef] [PubMed]
- Matsumoto, M.; Baba, A.; Yokota, T.; Nishikawa, H.; Ohkawa, Y.; Kayama, H.; Kallies, A.; Nutt, S.L.; Sakaguchi, S.; Takeda, K.; et al. Interleukin-10-producing plasmablasts exert regulatory function in autoimmune inflammation. Immunity 2014, 41, 1040–1051. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rolf, L.; Muris, A.H.; Hupperts, R.; Damoiseaux, J. Illuminating vitamin D effects on B cells—The multiple sclerosis perspective. Immunology 2016, 147, 275–284. [Google Scholar] [CrossRef]
- Ao, T.; Kikuta, J.; Ishii, M. The Effects of Vitamin D on Immune System and Inflammatory Diseases. Biomolecules 2021, 11, 1624. [Google Scholar] [CrossRef] [PubMed]
- Ghelani, D.; Alesi, S.; Mousa, A. Vitamin D and COVID-19: An Overview of Recent Evidence. Int. J. Mol. Sci. 2021, 22, 10559. [Google Scholar] [CrossRef] [PubMed]
- Soto, J.R.; Anthias, C.; Madrigal, A.; Snowden, J.A. Insights Into the Role of Vitamin D as a Biomarker in Stem Cell Transplantation. Front. Immunol. 2020, 11, 966. [Google Scholar] [CrossRef] [PubMed]
- Hong, S.; Ferraro, C.S.; Hamilton, B.K.; Majhail, N.S. To D or not to D: Vitamin D in hematopoietic cell transplantation. Bone Marrow Transplant. 2020, 55, 2060–2070. [Google Scholar] [CrossRef]
- Medrano, M.; Carrillo-Cruz, E.; Montero, I.; Perez-Simon, J.A. Vitamin D: Effect on Haematopoiesis and Immune System and Clinical Applications. Int. J. Mol. Sci. 2018, 19, 2663. [Google Scholar] [CrossRef] [Green Version]
- Jeon, S.M.; Shin, E.A. Exploring vitamin D metabolism and function in cancer. Exp. Mol. Med. 2018, 50, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Adorini, L.; Penna, G.; Giarratana, N.; Uskokovic, M. Tolerogenic dendritic cells induced by vitamin D receptor ligands enhance regulatory T cells inhibiting allograft rejection and autoimmune diseases. J. Cell. Biochem. 2003, 88, 227–233. [Google Scholar] [CrossRef]
- Xi, Y.; Ma, Y.; Xie, B.; Di, A.; Xu, S.; Luo, X.; Wang, C.; Dai, H.; Yan, G.; Qi, Z. Vitamin D3 combined with antibody agents suppresses alloreactive memory T-cell responses to induce heart allograft long-term survival. Transpl. Immunol. 2021, 66, 101374. [Google Scholar] [CrossRef] [PubMed]
- Infante, M.; Ricordi, C.; Padilla, N.; Alvarez, A.; Linetsky, E.; Lanzoni, G.; Mattina, A.; Bertuzzi, F.; Fabbri, A.; Baidal, D.; et al. The Role of Vitamin D and Omega-3 PUFAs in Islet Transplantation. Nutrients 2019, 11, 2937. [Google Scholar] [CrossRef] [Green Version]
- Gregori, S.; Giarratana, N.; Smiroldo, S.; Uskokovic, M.; Adorini, L. A 1α,25-dihydroxyvitamin D3 analog enhances regulatory T-cells and arrests autoimmune diabetes in NOD mice. Diabetes 2002, 51, 1367–1374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giarratana, N.; Penna, G.; Amuchastegui, S.; Mariani, R.; Daniel, K.C.; Adorini, L. A Vitamin D Analog Down-Regulates Proinflammatory Chemokine Production by Pancreatic Islets Inhibiting T Cell Recruitment and Type 1 Diabetes Development. J. Immunol. 2004, 173, 2280–2287. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gysemans, C.; Van Etten, E.; Overbergh, L.; Giulietti, A.; Eelen, G.; Waer, M.; Verstuyf, A.; Bouillon, R.; Mathieu, C. Unaltered Diabetes Presentation in NOD Mice Lacking the Vitamin D Receptor. Diabetes 2008, 57, 269–275. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Spach, K.M.; Nashold, F.E.; Dittel, B.N.; Hayes, C.E. IL-10 Signaling Is Essential for 1,25-Dihydroxyvitamin D 3 -Mediated Inhibition of Experimental Autoimmune Encephalomyelitis. J. Immunol. 2006, 177, 6030–6037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pedersen, L.B.; Nashold, F.E.; Spach, K.M.; Hayes, C.E. 1,25-Dihydroxyvitamin D3 reverses experimental autoimmune encephalomyelitis by inhibiting chemokine synthesis and monocyte trafficking. J. Neurosci. Res. 2007, 85, 2480–2490. [Google Scholar] [CrossRef]
- Nashold, F.E.; Hoag, K.A.; Goverman, J.; Hayes, C.E. Rag-1-dependent cells are necessary for 1,25-dihydroxyvitamin D3 prevention of experimental autoimmune encephalomyelitis. J. Neuroimmunol. 2001, 119, 16–29. [Google Scholar] [CrossRef]
- Meehan, T.F.; DeLuca, H.F. CD8+ T cells are not necessary for 1α,25-dihydroxy-vitamin D3 to suppress experimental autoimmune encephalomyelitis in mice. Proc. Natl. Acad. Sci. USA 2002, 99, 5557–5560. [Google Scholar] [CrossRef] [Green Version]
- Mayne, C.G.; Spanier, J.A.; Relland, L.M.; Williams, C.B.; Hayes, C.E. 1,25-Dihydroxyvitamin D3 acts directly on the T lymphocyte vitamin D receptor to inhibit experimental autoimmune encephalomyelitis. Eur. J. Immunol. 2011, 41, 822–832. [Google Scholar] [CrossRef]
- Lemire, J.M.; Ince, A.; Takashima, M. 1,25-Dihydroxyvitamin D3 Attenuates of Expression of Experimental Murine Lupus of MRL/1 Mice. Autoimmunity 2009, 12, 143–148. [Google Scholar] [CrossRef]
- Reeves, W.H.; Lee, P.Y.; Weinstein, J.S.; Satoh, M.; Lu, L. Induction of autoimmunity by pristane and other naturally occurring hydrocarbons. Trends Immunol. 2009, 30, 455–464. [Google Scholar] [CrossRef] [Green Version]
- Correa Freitas, E.; Evelyn Karnopp, T.; de Souza Silva, J.M.; Cavalheiro do Espírito Santo, R.; da Rosa, T.H.; de Oliveira, M.S.; da Costa Gonçalves, F.; de Oliveira, F.H.; Guilherme Schaefer, P.; André Monticielo, O. Vitamin D supplementation ameliorates arthritis but does not alleviates renal injury in pristane-induced lupus model. Autoimmunity 2019, 52, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.; Mahon, B.D.; Froicu, M.; Cantorna, M.T. Calcium and 1alpha,25-dihydroxyvitamin D3 target the TNF-alpha pathway to suppress experimental inflammatory bowel disease. Eur. J. Immunol. 2005, 35, 217–224. [Google Scholar] [CrossRef]
- Froicu, M.; Cantorna, M.T. Vitamin D and the vitamin D receptor are critical for control of the innate immune response to colonic injury. BMC Immunol. 2007, 8, 5. [Google Scholar] [CrossRef] [Green Version]
- Lagishetty, V.; Misharin, A.V.; Liu, N.Q.; Lisse, T.S.; Chun, R.F.; Ouyang, Y.; McLachlan, S.M.; Adams, J.S.; Hewison, M. Vitamin D Deficiency in Mice Impairs Colonic Antibacterial Activity and Predisposes to Colitis. Endocrinology 2010, 151, 2423–2432. [Google Scholar] [CrossRef] [Green Version]
- Pakkala, I.; Taskinen, E.; Pakkala, S.; Räisänen-Sokolowski, A. MC1288, a vitamin D analog, prevents acute graft-versus-host disease in rat bone marrow transplantation. Bone Marrow Transplant. 2001, 27, 863–867. [Google Scholar] [CrossRef] [Green Version]
- Taylor, B.; Du, J.; Dodge, J.; Li, Y.C.; Chen, X. Targeting Intestinal Vitamin D Receptor Signaling to Mitigate Graft-Versus-Host Disease. Blood 2018, 132, 4515. [Google Scholar] [CrossRef]
- Tripkovic, L.; Lambert, H.; Hart, K.; Smith, C.P.; Bucca, G.; Penson, S.; Chope, G.; Hyppönen, E.; Berry, J.; Vieth, R.; et al. Comparison of vitamin D2 and vitamin D3 supplementation in raising serum 25-hydroxyvitamin D status: A systematic review and meta-analysis. Am. J. Clin. Nutr. 2012, 95, 1357–1364. [Google Scholar] [CrossRef] [Green Version]
- Bischoff-Ferrari, H.A.; Dawson-Hughes, B.; Stöcklin, E.; Sidelnikov, E.; Willett, W.C.; Edel, J.O.; Stähelin, H.B.; Wolfram, S.; Jetter, A.; Schwager, J.; et al. Oral supplementation with 25(OH)D 3 versus vitamin D 3: Effects on 25(OH)D levels, lower extremity function, blood pressure, and markers of innate immunity. J. Bone Miner. Res. 2012, 27, 160–169. [Google Scholar] [CrossRef]
- Mazzaferro, S.; Goldsmith, D.; Larsson, T.E.; Massy, Z.A.; Cozzolino, M. Vitamin D Metabolites and/or Analogs: Which D for Which Patient? Curr. Vasc. Pharmacol. 2014, 12, 339–349. [Google Scholar] [CrossRef] [PubMed]
- Zand, L.; Kumar, R. The Use of Vitamin D Metabolites and Analogues in the Treatment of Chronic Kidney Disease. Endocrinol. Metab. Clin. N. Am. 2017, 46, 983–1007. [Google Scholar] [CrossRef]
- Hansen, D.; Rasmussen, K.; Danielsen, H.; Meyer-Hofmann, H.; Bacevicius, E.; Lauridsen, T.G.; Madsen, J.K.; Tougaard, B.G.; Marckmann, P.; Thye-Roenn, P.; et al. No difference between alfacalcidol and paricalcitol in the treatment of secondary hyperparathyroidism in hemodialysis patients: A randomized crossover trial. Kidney Int. 2011, 80, 841–850. [Google Scholar] [CrossRef] [Green Version]
- Ross, A.C.; Manson, J.E.; Abrams, S.A.; Aloia, J.F.; Brannon, P.M.; Clinton, S.K.; Durazo-Arvizu, R.A.; Gallagher, J.C.; Gallo, R.L.; Jones, G.; et al. The 2011 Report on Dietary Reference Intakes for Calcium and Vitamin D from the Institute of Medicine: What Clinicians Need to Know. J. Clin. Endocrinol. Metab. 2011, 96, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Dawson-Hughes, B.; Mithal, A.; Bonjour, J.-P.; Boonen, S.; Burckhardt, P.; Fuleihan, G.E.-H.; Josse, R.G.; Lips, P.; Morales-Torres, J.; Yoshimura, N. IOF position statement: Vitamin D recommendations for older adults. Osteoporos. Int. 2010, 21, 1151–1154. [Google Scholar] [CrossRef]
- Lips, P. Vitamin D Deficiency and Secondary Hyperparathyroidism in the Elderly: Consequences for Bone Loss and Fractures and Therapeutic Implications. Endocr. Rev. 2001, 22, 477–501. [Google Scholar] [CrossRef]
- Giustina, A.; Adler, R.A.; Binkley, N.; Bouillon, R.; Ebeling, P.R.; Lazaretti-Castro, M.; Marcocci, C.; Rizzoli, R.; Sempos, C.T.; Bilezikian, J.P. Controversies in Vitamin D: Summary Statement From an International Conference. J. Clin. Endocrinol. Metab. 2019, 104, 234–240. [Google Scholar] [CrossRef] [Green Version]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Evaluation, Treatment, and Prevention of Vitamin D Deficiency: An Endocrine Society Clinical Practice Guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [Green Version]
- Heaney, R.P.; Davies, K.M.; Chen, T.C.; Holick, M.F.; Barger-Lux, M.J. Human serum 25-hydroxycholecalciferol response to extended oral dosing with cholecalciferol. Am. J. Clin. Nutr. 2003, 77, 204–210. [Google Scholar] [CrossRef] [Green Version]
- Varsavsky, M.; Rozas Moreno, P.; Becerra Fernández, A.; Luque Fernández, I.; Quesada Gómez, J.M.; Ávila Rubio, V.; García Martín, A.; Cortés Berdonces, M.; Naf Cortés, S.; Romero Muñoz, M.; et al. Recomendaciones de vitamina D para la población general. Endocrinol. Diabetes Y Nutr. 2017, 64, 7–14. [Google Scholar] [CrossRef]
- Ish-Shalom, S.; Segal, E.; Salganik, T.; Raz, B.; Bromberg, I.L.; Vieth, R. Comparison of Daily, Weekly, and Monthly Vitamin D3 in Ethanol Dosing Protocols for Two Months in Elderly Hip Fracture Patients. J. Clin. Endocrinol. Metab. 2008, 93, 3430–3435. [Google Scholar] [CrossRef] [Green Version]
- Manson, J.E.; Cook, N.R.; Lee, I.-M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N. Engl. J. Med. 2019, 380, 33–44. [Google Scholar] [CrossRef]
- Miyaura, C.; Abe, E.; Kuribayashi, T.; Tanaka, H.; Konno, K.; Nishii, Y.; Suda, T. 1α,25-Dihydroxyvitamin D3 induces differentiation of human myeloid leukemia cells. Biochem. Biophys. Res. Commun. 1981, 102, 937–943. [Google Scholar] [CrossRef]
- Munker, R.; Norman, A.; Koeffler, H.P. Vitamin D compounds. Effect on clonal proliferation and differentiation of human myeloid cells. J. Clin. Investig. 1986, 78, 424–430. [Google Scholar] [CrossRef]
- Camagna, A.; Testa, U.; Masciulli, R.; Barberi, T.; Samoggia, P.; Tritarelli, E.; Pustorino, E.; Cipollone, L.; Ciancio, L.; del Duca, P.; et al. The synergistic effect of simultaneous addition of retinoic acid and vitamin D3 on the in-vitro differentiation of human promyelocytic leukemia cell lines could be efficiently transposed in vivo. Med. Hypotheses 1998, 50, 253–257. [Google Scholar] [CrossRef]
- Makishima, M.; Shudo, K.; Honma, Y. Greater synergism of retinoic acid receptor (RAR) agonists with vitamin D3 than that of retinoid X receptor (RXR) agonists with regard to growth inhibition and differentiation induction in monoblastic leukemia cells. Biochem. Pharmacol. 1999, 57, 521–529. [Google Scholar] [CrossRef]
- Elstner, E.; Linker-Israeli, M.; Umiel, T.; Le, J.; Grillier, I.; Said, J.; Shintaku, I.P.; Krajewski, S.; Reed, J.C.; Binderup, L.; et al. Combination of a potent 20-epi-vitamin D3 analogue (KH 1060) with 9-cis-retinoic acid irreversibly inhibits clonal growth, decreases bcl-2 expression, and induces apoptosis in HL-60 leukemic cells. Cancer Res. 1996, 56, 3570–3576. [Google Scholar] [PubMed]
- Elstner, E.; Linker-Israeli, M.; Le, J.; Umiel, T.; Michl, P.; Said, J.W.; Binderup, L.; Reed, J.C.; Koeffler, H.P. Synergistic decrease of clonal proliferation, induction of differentiation, and apoptosis of acute promyelocytic leukemia cells after combined treatment with novel 20-epi vitamin D3 analogs and 9-cis retinoic acid. J. Clin. Investig. 1997, 99, 349–360. [Google Scholar] [CrossRef]
- Hisatake, J.; O’Kelly, J.; Uskokovic, M.R.; Tomoyasu, S.; Koeffler, H.P. Novel vitamin D3 analog, 21-(3-methyl-3-hydroxy-butyl)-19-nor D3, that modulates cell growth, differentiation, apoptosis, cell cycle, and induction of PTEN in leukemic cells. Blood 2001, 97, 2427–2433. [Google Scholar] [CrossRef] [Green Version]
- Kumagai, T.; Shih, L.-Y.; Hughes, S.V.; Desmond, J.C.; O’Kelly, J.; Hewison, M.; Koeffler, H.P. 19-Nor-1,25(OH) 2 D 2 (a Novel, Noncalcemic Vitamin D Analogue), Combined with Arsenic Trioxide, Has Potent Antitumor Activity against Myeloid Leukemia. Cancer Res. 2005, 65, 2488–2497. [Google Scholar] [CrossRef] [Green Version]
- Müller-Thomas, C.; Tüchler, H.; Rudelius, M.; Schneider, H.; Pfefferkorn, S.; Götze, K.S. Serum Vitamin D Levels in Patients with Myelodysplastic Syndromes: A Retrospective Single-Center Analysis. Acta Haematol. 2019, 141, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Pardanani, A.; Drake, M.T.; Finke, C.; Lasho, T.L.; Rozell, S.A.; Jimma, T.; Tefferi, A. Vitamin D insufficiency in myeloproliferative neoplasms and myelodysplastic syndromes: Clinical correlates and prognostic studies. Am. J. Hematol. 2011, 86, 1013–1016. [Google Scholar] [CrossRef]
- Radujkovic, A.; Schnitzler, P.; Ho, A.D.; Dreger, P.; Luft, T. Low serum vitamin D levels are associated with shorter survival after first-line azacitidine treatment in patients with myelodysplastic syndrome and secondary oligoblastic acute myeloid leukemia. Clin. Nutr. 2017, 36, 542–551. [Google Scholar] [CrossRef]
- Lee, H.J.; Muindi, J.R.; Tan, W.; Hu, Q.; Wang, D.; Liu, S.; Wilding, G.E.; Ford, L.A.; Sait, S.N.J.; Block, A.W.; et al. Low 25(OH) vitamin D 3 levels are associated with adverse outcome in newly diagnosed, intensively treated adult acute myeloid leukemia. Cancer 2014, 120, 521–529. [Google Scholar] [CrossRef] [Green Version]
- Koeffler, H.P.; Hirji, K.; Itri, L. 1,25-Dihydroxyvitamin D3: In vivo and in vitro effects on human preleukemic and leukemic cells. Cancer Treat. Rep. 1985, 69, 1399–1407. [Google Scholar]
- Mellibovsky; Díez; Pérez-Vila; Serrano; Nacher; AubÍa; Supervía; Recker, R.R. Vitamin D treatment in myelodysplastic syndromes. Br. J. Haematol. 1998, 100, 516–520. [Google Scholar] [CrossRef] [PubMed]
- Koeffler, H.P.; Aslanian, N.; O’Kelly, J. Vitamin D2 analog (Paricalcitol; Zemplar) for treatment of myelodysplastic syndrome. Leuk. Res. 2005, 29, 1259–1262. [Google Scholar] [CrossRef]
- Petrich, A.; Kahl, B.; Bailey, H.; Kim, K.; Turman, N.; Juckett, M. Phase II study of doxercalciferol for the treatment of myelodysplastic syndrome. Leuk. Lymphoma 2008, 49, 57–61. [Google Scholar] [CrossRef] [Green Version]
- Motomura, S.; Kanamori, H.; Ohkubo, T.; Maruta, A.; Kodama, F. The effect of 1-hydroxyvitamin D3 for prolongation of leukemic transformation-free survival in myelodysplastic syndromes. Am. J. Hematol. 1991, 38, 67–68. [Google Scholar] [CrossRef] [PubMed]
- Akiyama, N.; Miyazawa, K.; Kanda, Y.; Tohyama, K.; Omine, M.; Mitani, K.; Ohyashiki, K. Multicenter phase II trial of vitamin K2 monotherapy and vitamin K2 plus 1α-hydroxyvitamin D3 combination therapy for low-risk myelodysplastic syndromes. Leuk. Res. 2010, 34, 1151–1157. [Google Scholar] [CrossRef]
- Hellström, E.; Robert, K.-H.; Samuelsson, J.; Lindemalm, C.; Grimfors, G.; Kimby, E.; Öberg, G.; Winqvist, I.; Billström, R.; Carneskog, J.; et al. Treatment of myelodysplastic syndromes with retinoic acid and 1α-hydroxy-vitamin D3 in combination with low-dose ara-C is not superior to ara-C alone. Results from a randomized study. Eur. J. Haematol. 2009, 45, 255–261. [Google Scholar] [CrossRef] [PubMed]
- Ferrero, D.; Darbesio, A.; Giai, V.; Genuardi, M.; Dellacasa, C.M.; Sorasio, R.; Bertini, M.; Boccadoro, M. Efficacy of a combination of human recombinant erythropoietin + 13- cis -retinoic acid and dihydroxylated vitamin D3 to improve moderate to severe anaemia in low/intermediate risk myelodysplastic syndromes. Br. J. Haematol. 2009, 144, 342–349. [Google Scholar] [CrossRef]
- Slapak, C.A.; Desforges, J.F.; Fogaren, T.; Miller, K.B. Treatment of acute myeloid leukemia in the elderly with low-dose cytarabine, hydroxyurea, and calcitriol. Am. J. Hematol. 1992, 41, 178–183. [Google Scholar] [CrossRef]
- Tracy, S.I.; Maurer, M.J.; Witzig, T.E.; Drake, M.T.; Ansell, S.M.; Nowakowski, G.S.; Thompson, C.A.; Inwards, D.J.; Johnston, P.B.; Micallef, I.N.; et al. Vitamin D insufficiency is associated with an increased risk of early clinical failure in follicular lymphoma. Blood Cancer J. 2017, 7, e595. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kelly, J.L.; Salles, G.; Goldman, B.; Fisher, R.I.; Brice, P.; Press, O.; Casasnovas, O.; Maloney, D.G.; Soubeyran, P.; Rimsza, L.; et al. Low Serum Vitamin D Levels Are Associated With Inferior Survival in Follicular Lymphoma: A Prospective Evaluation in SWOG and LYSA Studies. J. Clin. Oncol. 2015, 33, 1482–1490. [Google Scholar] [CrossRef] [PubMed]
- Drake, M.T.; Maurer, M.J.; Link, B.K.; Habermann, T.M.; Ansell, S.M.; Micallef, I.N.; Kelly, J.L.; Macon, W.R.; Nowakowski, G.S.; Inwards, D.J.; et al. Vitamin D Insufficiency and Prognosis in Non-Hodgkin’s Lymphoma. J. Clin. Oncol. 2010, 28, 4191–4198. [Google Scholar] [CrossRef] [Green Version]
- Park, H.Y.; Hong, Y.-C.; Lee, K.; Koh, J. Vitamin D status and risk of non-Hodgkin lymphoma: An updated meta-analysis. PLoS ONE 2019, 14, e0216284. [Google Scholar] [CrossRef] [PubMed]
- Xu, D.; Liang, J.; Wang, L.; Zhu, H.-Y.; Xia, Y.; Fan, L.; Li, J.-Y.; Xu, W. 25-Hydroxy vitamin D deficiency predicts inferior prognosis in mantle cell lymphoma. J. Cancer Res. Clin. Oncol. 2020, 146, 1003–1009. [Google Scholar] [CrossRef]
- Qin, J.-Q.; Yin, H.; Wu, J.-Z.; Chen, R.-Z.; Xia, Y.; Wang, L.; Zhu, H.-Y.; Fan, L.; Li, J.-Y.; Liang, J.-H.; et al. 25-Hydroxy vitamin D deficiency predicts inferior prognosis in Hodgkin lymphoma. Leuk. Res. 2021, 105, 106580. [Google Scholar] [CrossRef]
- Hohaus, S.; Tisi, M.C.; Bellesi, S.; Maiolo, E.; Alma, E.; Tartaglia, G.; Corrente, F.; Cuccaro, A.; D’Alo’, F.; Basile, U.; et al. Vitamin D deficiency and supplementation in patients with aggressive B-cell lymphomas treated with immunochemotherapy. Cancer Med. 2018, 7, 270–281. [Google Scholar] [CrossRef]
- Ammann, E.M.; Drake, M.T.; Haraldsson, B.; Wallace, R.B.; Johnson, K.C.; Desai, P.; Lin, E.M.; Link, B.K. Incidence of hematologic malignancy and cause-specific mortality in the Women’s Health Initiative randomized controlled trial of calcium and vitamin D supplementation. Cancer 2017, 123, 4168–4177. [Google Scholar] [CrossRef]
- Sfeir, J.G.; Drake, M.T.; LaPlant, B.R.; Maurer, M.J.; Link, B.K.; Berndt, T.J.; Shanafelt, T.D.; Cerhan, J.R.; Habermann, T.M.; Feldman, A.L.; et al. Validation of a vitamin D replacement strategy in vitamin D-insufficient patients with lymphoma or chronic lymphocytic leukemia. Blood Cancer J. 2017, 7, e526. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Siitonen, T.; Timonen, T.; Juvonen, E.; Terava, V.; Kutila, A.; Honkanen, T.; Mikkola, M.; Hallman, H.; Kauppila, M.; Nylanden, P.; et al. Valproic acid combined with 13-cis retinoic acid and 1,25-dihydroxyvitamin D3 in the treatment of patients with myelodysplastic syndromes. Haematologica 2007, 92, 1119–1122. [Google Scholar] [CrossRef] [Green Version]
- Puthier, D.; Bataille, R.; Barillé, S.; Mellerin, M.P.; Harousseau, J.L.; Ponzio, A.; Robillard, N.; Wijdenes, J.; Amiot, M. Myeloma cell growth arrest, apoptosis, and interleukin-6 receptor modulation induced by EB1089, a vitamin D3 derivative, alone or in association with dexamethasone. Blood 1996, 88, 4659–4666. [Google Scholar] [CrossRef] [Green Version]
- Park, W.H.; Seol, J.G.; Kim, E.S.; Binderup, L.; Koeffler, H.P.; Kim, B.K.; Lee, Y.Y. The induction of apoptosis by a combined 1,25(OH)2D3 analog, EB1089 and TGF-beta1 in NCI-H929 multiple myeloma cells. Int. J. Oncol. 2002, 20, 533–542. [Google Scholar]
- Burwick, N. Vitamin D and plasma cell dyscrasias: Reviewing the significance. Ann. Hematol. 2017, 96, 1271–1277. [Google Scholar] [CrossRef] [PubMed]
- Simmons, J.; Sheedy, C.; Lee, H.; Koh, S.; Alvarez, J.; Koyama, T.; Friedman, D. Prevalence of 25-hydroxyvitamin D deficiency in child and adolescent patients undergoing hematopoietic cell transplantation compared to a healthy population. Pediatr. Blood Cancer 2013, 60, 2025–2030. [Google Scholar] [CrossRef]
- Kenny, S.A.; Collum, K.; Featherstone, C.A.; Farooki, A.; Jakubowski, A. Impact of a Replacement Algorithm for Vitamin D Deficiency in Adult Hematopoietic Stem Cell Transplant Patients. J. Adv. Pract. Oncol. 2019, 10, 109–118. [Google Scholar]
- Martinez-Cibrian, N.; Zeiser, R.; Perez-Simon, J.A. Graft-versus-host disease prophylaxis: Pathophysiology-based review on current approaches and future directions. Blood Rev. 2020, 48, 100792. [Google Scholar] [CrossRef] [PubMed]
- MacDonald, K.P.A.A.; Hill, G.R.; Blazar, B.R. Chronic graft-versus-host disease: Biological insights from preclinical and clinical studies. Blood 2017, 129, 13–21. [Google Scholar] [CrossRef]
- Cooke, K.R.; Luznik, L.; Sarantopoulos, S.; Hakim, F.T.; Jagasia, M.; Fowler, D.H.; van den Brink, M.R.M.; Hansen, J.A.; Parkman, R.; Miklos, D.B.; et al. The Biology of Chronic Graft-versus-Host Disease: A Task Force Report from the National Institutes of Health Consensus Development Project on Criteria for Clinical Trials in Chronic Graft-versus-Host Disease. Biol. Blood Marrow Transplant. 2017, 23, 211–234. [Google Scholar] [CrossRef] [Green Version]
- Zeiser, R.; Blazar, B.R. Pathophysiology of chronic graft-versus-host disease and therapeutic targets. N. Engl. J. Med. 2017, 377, 2565–2579. [Google Scholar] [CrossRef]
- Urbain, P.; Ihorst, G.; Biesalski, H.-K.; Bertz, H. Course of serum 25-hydroxyvitamin D3 status and its influencing factors in adults undergoing allogeneic hematopoietic cell transplantation. Ann. Hematol. 2012, 91, 759–766. [Google Scholar] [CrossRef]
- Wallace, G.; Jodele, S.; Howell, J.; Myers, K.C.; Teusink, A.; Zhao, X.; Setchell, K.; Holtzapfel, C.; Lane, A.; Taggart, C.; et al. Vitamin D Deficiency and Survival in Children after Hematopoietic Stem Cell Transplant. Biol. Blood Marrow Transplant. 2015, 21, 1627–1631. [Google Scholar] [CrossRef] [Green Version]
- Dahir, K.; Perry, B.; Jagasia, S. Post-Transplantation Bone Disease: Prevalence, Monitoring, Prevention, and Management Guidelines. In Blood and Marrow Transplantation Long-Term Management; John Wiley & Sons, Ltd.: Oxford, UK, 2013; pp. 151–161. [Google Scholar]
- Jabbour, J.; Manana, B.; Zahreddine, A.; Al-Shaar, L.; Bazarbachi, A.; Blaise, D.; El-Cheikh, J. Vitamins and minerals intake adequacy in hematopoietic stem cell transplant: Results of a randomized controlled trial. Bone Marrow Transplant. 2021, 56, 1106–1115. [Google Scholar] [CrossRef]
- Kreutz, M.; Eissner, G.; Hahn, J.; Andreesen, R.; Drobnik, W.; Holler, E. Variations in 1α,25-dihydroxyvitamin D3 and 25-hydroxyvitamin D3 serum levels during allogeneic bone marrow transplantation. Bone Marrow Transplant. 2004, 33, 871–873. [Google Scholar] [CrossRef]
- Glotzbecker, B.; Ho, V.T.; Aldridge, J.; Kim, H.T.; Horowitz, G.; Ritz, J.; Soiffer, R.; Avigan, D.; Rosenblatt, J. Low levels of 25-hydroxyvitamin D before allogeneic hematopoietic SCT correlate with the development of chronic GVHD. Bone Marrow Transplant. 2013, 48, 593–597. [Google Scholar] [CrossRef] [Green Version]
- Ganetsky, A.; Richman, L.P.; Frey, N.V.; Vonderheide, R.H.; Porter, D.L.; Reshef, R. Vitamin D Deficiency Predicts Acute Cutaneous Graft-Versus-Host Disease in Reduced-Intensity Allogeneic Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2014, 20, S267–S268. [Google Scholar] [CrossRef] [Green Version]
- Hansson, M.E.A.; Norlin, A.-C.; Omazic, B.; Wikström, A.-C.; Bergman, P.; Winiarski, J.; Remberger, M.; Sundin, M. Vitamin D Levels Affect Outcome in Pediatric Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2014, 20, 1537–1543. [Google Scholar] [CrossRef] [Green Version]
- Von Bahr, L.; Blennow, O.; Alm, J.; Björklund, A.; Malmberg, K.-J.; Mougiakakos, D.; Le Blanc, A.; Oefner, P.J.; Labopin, M.; Ljungman, P.; et al. Increased incidence of chronic GvHD and CMV disease in patients with vitamin D deficiency before allogeneic stem cell transplantation. Bone Marrow Transplant. 2015, 50, 1217–1223. [Google Scholar] [CrossRef] [Green Version]
- Campos, D.J.; Biagini, G.L.K.; Funke, V.A.M.; Bonfim, C.M.S.; Boguszewski, C.L.; Borba, V.Z.C. Vitamin D deficiency in children and adolescents submitted to hematopoietic stem cell transplantation. Rev. Bras. Hematol. Hemoter. 2014, 36, 126–131. [Google Scholar] [CrossRef] [Green Version]
- Beebe, K.; Magee, K.; McNulty, A.; Stahlecker, J.; Salzberg, D.; Miller, H.; Mirea, L.; Adams, R.; Ngwube, A. Vitamin D deficiency and outcomes in pediatric hematopoietic stem cell transplantation. Pediatr. Blood Cancer 2018, 65, e26817. [Google Scholar] [CrossRef]
- Robien, K.; Strayer, L.G.; Majhail, N.; Lazovich, D.; Baker, K.S.; Smith, A.R.; Mulrooney, D.A.; Burns, L.J. Vitamin D status among long-term survivors of hematopoietic cell transplantation. Bone Marrow Transplant. 2011, 46, 1472–1479. [Google Scholar] [CrossRef] [Green Version]
- Bajwa, R.P.S.; Taylor, K.; Hoyt, A.; Kamboj, M.K.; Stanek, J.; Mahadeo, K.M.; Alsaedi, H.; Abdel-Azim, H.; O’Kane, S.; Martin, P.L.; et al. Vitamin D has no impact on outcomes after HSCT in children—A retrospective study. Pediatr. Transplant. 2021, 25. [Google Scholar] [CrossRef]
- Katić, M.; Pirsl, F.; Steinberg, S.M.; Dobbin, M.; Curtis, L.M.; Pulanić, D.; Desnica, L.; Titarenko, I.; Pavletic, S.Z. Vitamin D levels and their associations with survival and major disease outcomes in a large cohort of patients with chronic graft-vs-host disease. Croat. Med. J. 2016, 57, 276–286. [Google Scholar] [CrossRef]
- Bhandari, R.; Malvar, J.; Sacapano, A.; Aguayo-Hiraldo, P.; Jodele, S.; Orgel, E. Association between Vitamin D and Risk for Early and Late Post-Transplant Complications. Biol. Blood Marrow Transplant. 2020, 26, 343–350. [Google Scholar] [CrossRef]
- Perera, T.; Ming Lim, A.B.; Mason, K.; Szer, J.; Ritchie, D.S. The Relationship Between Pre-Transplant 25-Hydroxy-Vitamin D Levels, Survival and Graft-Versus-Host Disease, in Allogeneic Haematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2015, 21, S303. [Google Scholar] [CrossRef] [Green Version]
- Radujkovic, A.; Kordelas, L.; Krzykalla, J.; Beelen, D.W.; Benner, A.; Lehners, N.; Schmidt, K.; Dreger, P.; Luft, T. Pretransplant Vitamin D Deficiency Is Associated With Higher Relapse Rates in Patients Allografted for Myeloid Malignancies. J. Clin. Oncol. 2017, 35, 3143–3152. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peter, K.; Siska, P.J.; Roider, T.; Matos, C.; Bruns, H.; Renner, K.; Singer, K.; Weber, D.; Güllstorf, M.; Kröger, N.; et al. 1,25-dihydroxyvitamin-D3 but not the clinically applied marker 25-hydroxyvitamin-D3 predicts survival after stem cell transplantation. Bone Marrow Transplant. 2021, 56, 419–433. [Google Scholar] [CrossRef]
- ITO, Y.; Honda, A.; Kurokawa, M. Impact of vitamin D level at diagnosis and transplantation on the prognosis of hematological malignancy: A meta-analysis. Blood Adv. 2021, 6, 1499–1511. [Google Scholar] [CrossRef]
- Chiengthong, K.; Cheungpasitporn, W.; Thongprayoon, C.; Lertjitbanjong, P.; Cato, L.D.; Bathini, T.; Ungprasert, P.; Mao, M.A.; Chokesuwattanaskul, R. Vitamin D deficiency is not associated with graft versus host disease after hematopoietic stem cell transplantation: A meta-analysis. J. Evid. Based. Med. 2020, 13, 183–191. [Google Scholar] [CrossRef] [PubMed]
- Gjærde, L.K.; Ostrowski, S.R.; Andersen, N.S.; Friis, L.S.; Kornblit, B.; Petersen, S.L.; Schjødt, I.; Sengeløv, H. Pre-transplantation plasma vitamin D levels and acute graft-versus-host disease after myeloablative hematopoietic cell transplantation in adults. Transpl. Immunol. 2021, 68, 101437. [Google Scholar] [CrossRef] [PubMed]
- Ros-Soto, J.; Anthias, C.; Madrigal, A.; Snowden, J.A. Vitamin D: Is it important in haematopoietic stem cell transplantation? A review. Bone Marrow Transplant. 2019, 54, 810–820. [Google Scholar] [CrossRef] [PubMed]
- Duncan, C.N.; Vrooman, L.; Apfelbaum, E.M.; Whitley, K.; Bechard, L.; Lehmann, L.E. 25-Hydroxy Vitamin D Deficiency Following Pediatric Hematopoietic Stem Cell Transplant. Biol. Blood Marrow Transplant. 2011, 17, 749–753. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wallace, G.; Jodele, S.; Myers, K.C.; Dandoy, C.E.; El-Bietar, J.; Nelson, A.; Teusink-Cross, A.; Khandelwal, P.; Taggart, C.; Gordon, C.M.; et al. Single Ultra-High-Dose Cholecalciferol to Prevent Vitamin D Deficiency in Pediatric Hematopoietic Stem Cell Transplantation. Biol. Blood Marrow Transplant. 2018, 24, 1856–1860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Silva, F.; Pérez-Simón, J.A.; Caballero-Velazquez, T.; Sánchez-Guijo, F.M.; Villanueva-Gomez, F.; Vazquez, L.; del Cañizo, C.; Caballero, D.; San Miguel, J. Effect of vitamin D treatment in chronic GVHD. Bone Marrow Transplant. 2011, 46, 1395–1397. [Google Scholar] [CrossRef] [PubMed]
- Bhandari, R.; Aguayo-Hiraldo, P.; Malvar, J.; Cheng, K.; Sacapano, A.; Abdel-Azim, H.; Chi, Y.-Y.; Wallace, G.; Asgharzadeh, S.; Jodele, S.; et al. Ultra-High Dose Vitamin D in Pediatric Hematopoietic Stem Cell Transplantation: A Nonrandomized Controlled Trial. Transplant. Cell. Ther. 2021, 27, 1001.e1–1001.e9. [Google Scholar] [CrossRef] [PubMed]
- Wallace, G.; Jodele, S.; Myers, K.C.; Dandoy, C.E.; El-Bietar, J.; Nelson, A.; Taggart, C.B.; Daniels, P.; Lane, A.; Howell, J.; et al. Vitamin D Deficiency in Pediatric Hematopoietic Stem Cell Transplantation Patients Despite Both Standard and Aggressive Supplementation. Biol. Blood Marrow Transplant. 2016, 22, 1271–1274. [Google Scholar] [CrossRef] [Green Version]
- Carrillo-Cruz, E.; García-Lozano, J.R.; Márquez-Malaver, F.J.; Sánchez-Guijo, F.M.; Montero Cuadrado, I.; Ferra i Coll, C.; Valcárcel, D.; López-Godino, O.; Cuesta, M.; Parody, R.; et al. Vitamin D Modifies the Incidence of Graft-versus-Host Disease after Allogeneic Stem Cell Transplantation Depending on the Vitamin D Receptor (VDR) Polymorphisms. Clin. Cancer Res. 2019, 25, 4616–4623. [Google Scholar] [CrossRef]
- Caballero-Velazquez, T.; Montero, I.; Sanchez-Guijo, F.; Parody, R.; Saldana, R.; Valcarcel, D.; Lopez-Godino, O.; Ferra i Coll, C.; Cuesta, M.; Carrillo-Vico, A.; et al. Immunomodulatory Effect of Vitamin D after Allogeneic Stem Cell Transplantation: Results of a Prospective Multicenter Clinical Trial. Clin. Cancer Res. 2016, 22, 5673–5681. [Google Scholar] [CrossRef] [Green Version]
| Study | N | Intervention | Vit. D Levels Median/Range | Endpoints | |
|---|---|---|---|---|---|
| MDSs Koeffler et al., 1985 [133] | NR | 18 | Calcitriol (>2 mcg) | --- | MR and PR 44% (8/18) |
| MDSs Motomura et al., 1991 [137] | Phase II | 30 | Alfacalcidol (4–6 mcg/day) vs. no therapy | --- | Progression to AML Alfacalcidol: 6% (1/15) No therapy: 46.6% (7/15) |
| MDS low and high IPSSs Koeffler et al., 2005 [135] | NR | 12 | Paricalcitol (8 μg/day and increments of 8 μg/day every 2 weeks) | --- | OR: 0%; 1/12 patients’ platelet counts achieved normal range for 5 weeks |
| MDSs and CMML low-int1 IPSSs Mellibovsky et al., 2001 [134] | NR | 19 | Calcifediol (266 mcg 3 times a week; n = 5) or calcitriol (0.25–0.75 mcg/d; n = 14) | Increased from 9.4 ± 4.6 ng/ mL to 37.5 ± 44.2 (p = 0.003) | OR: 57% (11/19); No hypercalcemia |
| MDSs and CMML Petrich et al., 2008 [136] | Phase II | 15 | Doxercalciferol (12.5 mcg/day for 12 weeks) | --- | No responses |
| MDSs with low IPSSs and int-1 Akiyama et al., 2010 [138] | Phase II | 20 | Alfacalcidol (0.75 mcg/day) + menatetrenone (45 mg for 1 year if response) | --- | ORR: 30% (6/20) |
| MDSs and AML Hellström et al., 2009 [139] | Phase III | 63 MDS 15 AML | Arm 1 Ld ara-C vs. arm 2 Ld ara-C + 13-CRA and alfacalcidol | --- | Similar OS, ORR and DOR; progressed from MDSs to AML: 44% vs. 20% (p = 0.0527) |
| MDSs low-int-2 IPSSs Ferrero et al., 2008 [140] | Phase II | 63 | EPO + 13-CRA + calcitriol | --- | RAEB1 OS 14 months; non-RAEB1 OS 55 months; erythroid response 60% (93% in low-risk patients) |
| MDSs and CMML Siitonen et al., 2007 [151] | NR | 19 | Valproic acid (dose adjusted by levels) + 13-CRA (10 mg/12 h) + calcitriol (1 mcg) | --- | Blood improvement: 3/19 patients (16%); 8/19 discontinued (side effects, no hypercalcemia) |
| AML (elderly) Slapak et al., 1992 [141] | NR | 29 | - Ld Ara-C + hydroxyurea + calcitriol (0.5 μg/12 h) | --- | ORR: 79%; CR: 45%/PR: 34%; DOR: 9.8 months |
| MDSs—IPSSs 0/I NCT00068276 Ongoing | Phase II | 36 | Cholecalciferol (doses not specified) | Safety and efficacy | |
| CLL NCT01518959 Ongoing | Phase III | 31 | Cholecalciferol (180.000 IU monthly) vs. placebo | 5 years OS, PFS and TTF 5-year lymphocyte count | |
| Aggressive NHL Hohaus et al., 2018 [148] | NR | 155 | Cholecalciferol loading phase (25,000 IU daily) and maintenance phase (25,000 IU weekly) | Vitamin D pre-treatment 14 ± 1.4 ng/mL and post-treatment 33 ± 1.4 (n = 81; p < 0.0001) | Independent prognostic parameters for EFS 25(OH)D levels < 20 ng/mL; HR of 2.88; p < 0.02 IPI HR of 2.97; p < 0.002 No hypercalcemia |
| NHL and CLL Sfeir et al., 2017 [150] NCT01787409 | Phase I/II | 158 | Cholecalciferol (50.000 IU weekly for 12 weeks; if <30 ng/mL: 50.000 IU twice weekly; when ≥30 ng/mL: 50.000 IU/month) | Vitamin D deficiency 45% (n = 71); mean ± SEM 17 ± 5 ng/mL | 97% vitamin D insufficient group reached ≥30 ng/mL prior to follow-up period of 3 years, during which these levels were maintained |
| NHL and CLL NCT01787409 Ongoing | Phase I/II | 713 | Cholecalciferol PO (once weekly for 12 weeks and then once monthly for a total of 36 months) | --- | 12 months EFS; 36 months treatment free; 5 years ORR and OS; 5 years TTF (CLL patients) |
| NHL and CLL NCT02553447 Ongoing | Early phase I | 370 | - Arm I: high-dose cholecalciferol PO daily - Arm II: low-dose cholecalciferol PO daily - Arm III (control) | --- | 3 years PFS; 3 years OS |
| Indolent NHL (ILyAD clinical trial) NCT03078855 Ongoing | Phase III | 210 | Weekly rituximab (4 weeks + - Arm 1: Cholecalciferol (2.000 IU daily) or - Arm 2: placebo | --- | 3 years PFS and OS; response to rituximab (reduction of lymphoma burden by at least 50%) |
| Diffuse large B cell lymphoma—65 years and older (FIL_PREVID) NCT04442412 Ongoing | Phase III | 430 | - Arm A: 7 days of prephase oral prednisone - Arm B: 7 days of prephase oral prednisone and cholecalciferol (25.000 IU/day), then 25.000 IU/week - Both followed by six courses of R-CHOP R-miniCHOP/21 days | --- | 54 months PFS, OS and EFS; 54 months RR and EDR; 54-month rate of ECOG changed after prephase; rate of patients with 25(OH)D levels corrected at Cycle 2; time-to-deterioration physical functioning and fatigue at Cycle 2 |
| Untreated early-stage CLL (or SLL) Ongoing | Phase II | 35 | Curcumin + oral daily cholecalciferol (on days 1–28 for six cycles; if PR, treatment up to 2 years) | --- | ORR and TTNT; 2 years PFS and OS; 2 years DOR |
| Study | N | Vitamin D Levels Mean ± 2 S.D OR Median (Range) | Impact on GvHD | Survival and Other Endpoints | ||
|---|---|---|---|---|---|---|
| Pre-Allo | Post-Allo | |||||
| Kreutz et al., 2004 [165] | Prospective | 48, UP | 36.4 ± 2.2 nmol/L | ↓ compared to pre-allo (27.8 ± 1.3 nmol/L) | In patients with grades 3–4 GvHD, serum levels remained low/dropped (p = 0.031) | |
| Glotzbecker et al., 2013 [166] | Retrospective | 53, AP | 21.9 ng/mL (7.8–45.7); vitamin D cutoff 25 ng/mL | cGVHD at 2 y 63.8% vs. 23.8% (p = 0.009); extensive cGVHD at 2 y 54.5% vs. 14.3% (p = 0.005) | OS: 53% vs. 50% (p = 0.57); PFS: 51% vs. 47% (p = 0.61) | |
| Ganetsky et al., 2014 [167] | Retrospective | 54, AP | D + 30 20 ng/mL (6–50) | D30 levels inversely correlate with risk of skin aGvHD in patients undergoing RIC (p < 0.001) | ||
| Campos et al., 2014 [170] | Prospective | 66, PP | 25.7 ± 12.3 ng/mL vs. controls 31.9 (p = 0.01); deficiency prevalence 32% vs. 8% (p = 0.01) | D + 30 22.7 ± 10.7 ng/mL; D + 180 20.9 ± 10.9 ng/mL (p = 0.01) | No association with GvHD | No effect on survival |
| Beebe et al., 2018 [171] | Retrospective | 72, PP | 26 ng/mL (19–34 ng/mL); deficiency 35% | Pre-HSCT and D + 100 similar at 1 year (p = 0.01); 35 ± 16 vs. 27 ± 10 | No association with GvHD | 1-year OS significantly lower among patients with vitamin D deficit (p = 0.001) |
| Robien, et al., 2011 [172] | Retrospective | 95, PAP | 65% had ≥ 75 nmol/L; 24% had low levels (50–75); 11% had < 50 nmol/L | No association with GvHD | ||
| Urbain, et al., 2012 [161] | Prospective | 102, AP | 16.4 ± 8.9 ng/mL; 89.2% had < 30 ng/mL and 23.5% < 10 ng/mL | D + 30 15.5 ± 8.7 ng/mL; D + 100 14.9 ± 7.5 ng/mL | Trend toward higher risk of grade 2–4 aGvHD among patients with lower vitamin D levels (p = 0.066) | |
| Gjærde, et al.,2021 [181] | Retrospective | 116, AP | 64 nmol/L; 29% had < 50 nmol/L and 8% had < 25 nmol/L | Pre-HSCT > 85 nmol/L had 1.5 times higher odds of grade II–IV aGvHD than < 47 nmol/L (CI: 0.84–2.7) | ||
| Bajwa et al., 2021 [173] | Retrospective | 233, PP | 24.24 ng/mL All patients had vitamin D insufficiency | D + 30 24.76 ng/mL vs. D + 100 29.89 ng/mL All normal thereafter | No statistical difference in acute or chronic GvHD | No significant influence on OS |
| Hansson et al., 2014 [168] | Retrospective | 123, PP | Insufficient-level group (33 nmol/L; 13–49); sufficient level group (63 nmol/L; 50–97) | Vitamin D at 6 months 23 nmol/L (18–24) in moderate/severe cGvHD vs. 37 nmol/L (10–80) in no cGVHD (p = 0.004) | Grades 2–4 aGvHD 47% in low vitamin D levels vs. 30% in sufficient (p = 0.05) | OS: 87% vs. 50% (p = 0.01) for insufficient vs. sufficient level; relapse for insufficient vs. sufficient level groups: 33% vs. 4% (p = 0.03) |
| Wallace et al., 2015 [162] | Prospective | 134, PP | 70% insufficient levels (<30 ng/mL); 33% deficient levels (<20 ng/mL) | 68% D + 100 insufficient (<30 ng/mL); 31% deficient (<20 ng/mL). | No significant impact on acute or chronic GvHD (p = 0.8). | Vitamin D < 20 ng/mL at D + 100 was associated with ↓ OS (70% vs. 84.1%; p = 0.044); no impact pre-allo |
| Von Bahr et al., 2015 [169] | Retrospective | 166, AP | 42 nmol/L (10–118; 53% insufficient levels; 11% deficient); healthy controls (66.5 nmol/L; 21–104; p < 0.001) | 39 nmol/L (10–116) at 6 months. | No significant impact on aGvHD; in 2-year cGvHD (moderate/severe), deficient vit. D level 56%, insufficient vit. D level 31% and sufficient vit. D level 21% (p = 0.01) | 2-y OS according to vit. D levels; 63% deficient, 69% insufficient, 76% normal; p = 0.24; aa p = 0.02; Significant ↑ in CMV disease if deficient vit. D (p = 0.005) and ↑ in antibiotics (p = 0.011) |
| Katic et al., 2016 [174] | Prospective | 310, PAP | Only patients with GvHD; 30 ng/mL (22–42); 77.7% had >20 ng/mL and 22.3% had ≤20 ng/mL | No association between vit. D levels and major cGvHD characteristics | ↓ OS in patients with vitamin D ≤20 ng/mL vs. >20 ng/mL | |
| Perera et al., 2015 [176] | Retrospective | 492, UP | No significant differences in acute/chronic GvHD | Higher mortality in vitamin-D-deficient cohort vs. replete cohort (HR of 1.5; CI of 1.1–2.0; p = 0.013); no PFS or relapse differences | ||
| Radujkovic et al., 2017 [177] | Retrospective | 492, AP | 11.8 ng/mL (4.0–46.3); vitamin D deficiency in training cohort 80%; in validation cohort 87% | No significant impact on cumulative incidences of acute and chronic GvHD | ↓ OS in vitamin D deficiency (HR of 1.78; p = 0.007) due to higher risk of relapse (HR of 1.96; p = 0.006) | |
| Peter et al., 2021 [178] | Prospective | 143 + 365, AP | All patients tested for 1,25-dihydroxyvitamin-D3 and 25-hydroxyvitamin-D3 from day −16 to −6 before allo-HSCT | 25-hydroxyvitamin-D3 showed a steady increase; 1,25-dihydroxyvitamin-D3 peaked around the time of allo-HSCT | No significant association between vitamin D levels and severe GvHD | ↓ 25-hydroxyvitamin-D3 during follow-up or ↓ peritransplant 1,25-dihydroxyvitamin-D3 was associated with increased TRM (p = 0.002 and p = 0.001). |
| Study | N | Vitamin D2 or D3 and Dose | Vitamin D Levels | Impact on GvHD NRM and Survival | |||
|---|---|---|---|---|---|---|---|
| Pre-Allo | Post-Allo | ||||||
| Wallace et al., 2018 [184] | Prospective | 10, PP | Cholecalciferol: single enteral dose (maximum 600,000 IU) based on weight and pre-transplantation vitamin D levels | Mean pre-transplantation 25-OH vitamin D level 28.9 ± 13.1 ng/mL | All patients achieved a therapeutic vitamin D level (>30 ng/mL) sustained at or above 8 weeks | ||
| Silva et al., 2011 [185] | Retrospective | 12, AP | Cholecalciferol: 1000 IU per day (orally) plus calcium carbonate (1250 mg; one pill daily) after HSCT for at least 6 months in patients with osteopenia | All patients had active cGvHD; At 6 months after treatment, 5 patients obtained complete response, 6 patients obtained partial response and 1 patient had no response | |||
| Duncan et al., 2011 [183] | Prospective | 22, PP | Ergocalciferol: 50,000 IU once weekly for 6 weeks | Mean pre-transplantation 22.8 ng/mL (7–42.6); vitamin D deficiency 37.3% (CI of 25.8–50%). | Mean increase following supplementation 18.8 (SD = 11.3; 8–42); 4.5% remained deficient | ||
| Bhandari et al., 2021 [186] | Prospective/historical cohort comparison | 33, PP | Cholecalciferol: one-time oral Stoss * dose of cholecalciferol in 5000 IU/mL liquid formulation, 5000 IU/capsule or 50,000 IU/capsule vs. standard dose 14 days before conditioning) | Mean pre-transplantation 27.7 ng/mL (SD 10.8); 59% were vitamin D insufficient vs. 61% in the historical cohort | * Mean level (p < 0.001) post Stoss of 72.2 ng/mL vs. standard dose of 35.8 ng/mL; * 97% of Stoss cohort vs. 67% of standard-dose cohort vitamin D sufficient | No association with acute GvHD, veno-occlusive disease or transplant-associated thrombotic microangiopathy | |
| Wallace et al., 2016 [187] | Prospective | 60, PP | Cholecalciferol. * control cohort (1) treated according NKF ^ guidelines; * intervention cohort (2): high doses of vitamin D based on body weight (15,000–100,000 IU weekly) | 51% (18 of 35 patients) in control cohort and 48% (12 of 25 patients) in the intervention cohort were vitamin-D-insufficient at the time of transplant | Outcomes improved in Cohort 2 but only 64% achieved therapeutic level despite receiving > 200 IU/kg/day | ||
| Kenny et al., 2019 [156] | Prospective | 144, AP | Cholecalciferol: The dose was guided by vitamin D levels; max. 50,000 IU orally once weekly) | 72.9% vitamin-D-deficient before HSCTs; mean pre-transplantation 21 ng/mL | 26.4% were vitamin D deficient before HSCTs; mean 6 month post-transplant level 36 ng/mL After treatment, significant difference between Vit D levels pretrans-plant vs. posttransplant (p < 0.001) | ||
| Caballero-Velázquez et al., 2016 [188] | Prospective | 150, AP | Cholecalciferol in three groups: control (CG; no vitamin), low-dose (LdD; 1000 IU/day) and high-dose (HdD; 5000 IU/day) | Plasma levels of 25-OH vitamin D3 were measured on days −5, +1, +7, +14 and +21 | Significantly higher levels among patients receiving high doses compared with the control group beyond day +7 | ↓ overall and moderate + severe cGvHD at 1 year (LdD 37.5% and 19.5% and HdD 42.4% and 27% compared with CG 67.5% and 44.7%; p < 0.05); in multivariable analysis, vitamin D ↓ the risks of overall cGvHD and moderate and severe cGvHD (p ≤ 0.01); similar relapse and survival rates | |
| Carrillo-Cruz et al., 2019 [189] | Prospective | 107, AP | Cholecalciferol in three groups: D3 control (CG; no vitamin D), low-dose (LdD; 1000 IU/day) and high-dose (HdD; 5000 IU/day | Incidences of overall cGvHD varied depending on the VDR genotype among patients with FokI CT genotype, (22.5% vs. 80%; p = 0.0004 and among patients treated with vitamin D compared with CG (HR of 0.143; p < 0.001) and patients w/o BsmI/ApaI/TaqI ATC haplotype (22.2% vs. 68.8%; p = 0.0005) | |||
| Bhandari, 2020 [175] | Prospective | 314, PP | Cholecalciferol | Obtained in 94 patients; mean levels of vitamin D with supplementation 33.67 ng/mL vs. 29.16 ng/mL without supplementation (p = 0.11) | 31.85 ng/mL in patients with aGvHD vs. 31.42 ng/mL in those w/o aGvHD (p = 0.91) | Vitamin D levels correlated with OS; for every 10 ng/mL increase, there was a 28% decreased risk of death (p = 0.01), but no difference for levels before HSCT; malignant diagnoses were associated (multivar. analysis) with EFS (p < 0.01). | |
| N | Vitamin D | Dose | Main Objective | |
|---|---|---|---|---|
| Cincinnati Children’s Hospital Medical Center, 2018 | 100 | Single large dose of vitamin D “Stoss therapy” with a placebo vs. single large doses of both vitamins D and A | Investigate incidences of acute GI GvHD at day +100 after transplant | |
| Cincinnati Children’s Hospital Medical Center, 2021 | 20 | Cholecalciferol | Vitamin D OTF weekly for a maximum of 12 weeks. The dose may be increased or decreased based on the dosing schema | Investigate efficacy of OTF D3 replacements by measuring vitamin D levels |
| Children’s Hospital Los Angeles, 2018 | 33 | Cholecalciferol | Single ultra-high dose of vitamin D | Investigate incidences of GvHD, veno-occlusive disease and thrombotic microangiopathy at day +100 after transplant |
| Cincinnati Children’s Hospital Medical Center, 2016 | 10 | Cholecalciferol | One oral vitamin D dose (based on vitamin D status and rounded to 5000 IU) <2 weeks prior to HSCT | Investigate vitamin D sufficiency following Stoss dosing prior to transplant |
| University of British Columbia, 2018 | 84 | Cholecalciferol | Intervention group: loading dose of 100,000 IU of vitamin D3 after 2000 IU of vitamin D3 daily. | Test efficacy and safety of high-dose vitamin D therapy by measuring serum 25-OH vitamin D levels weekly for 8 weeks |
| Seoul National University Hospital, 2017 | 88 | Cholecalciferol | Control group: 2000 IU vitamin D3 daily | Assess efficacy (in patients achieving sufficient serum 25-OH vitamin D3 levels on day +100 post-aHSCT) of 100.000 IU of vitamin D3 prior to aHSCT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rodríguez-Gil, A.; Carrillo-Cruz, E.; Marrero-Cepeda, C.; Rodríguez, G.; Pérez-Simón, J.A. Effect of Vitamin D on Graft-versus-Host Disease. Biomedicines 2022, 10, 987. https://doi.org/10.3390/biomedicines10050987
Rodríguez-Gil A, Carrillo-Cruz E, Marrero-Cepeda C, Rodríguez G, Pérez-Simón JA. Effect of Vitamin D on Graft-versus-Host Disease. Biomedicines. 2022; 10(5):987. https://doi.org/10.3390/biomedicines10050987
Chicago/Turabian StyleRodríguez-Gil, Alfonso, Estrella Carrillo-Cruz, Cristina Marrero-Cepeda, Guillermo Rodríguez, and José A. Pérez-Simón. 2022. "Effect of Vitamin D on Graft-versus-Host Disease" Biomedicines 10, no. 5: 987. https://doi.org/10.3390/biomedicines10050987






