Magnetoliposomes Containing Multicore Nanoparticles and a New Antitumor Thienopyridine Compound with Potential Application in Chemo/Thermotherapy
Abstract
:1. Introduction
2. Materials and Methods
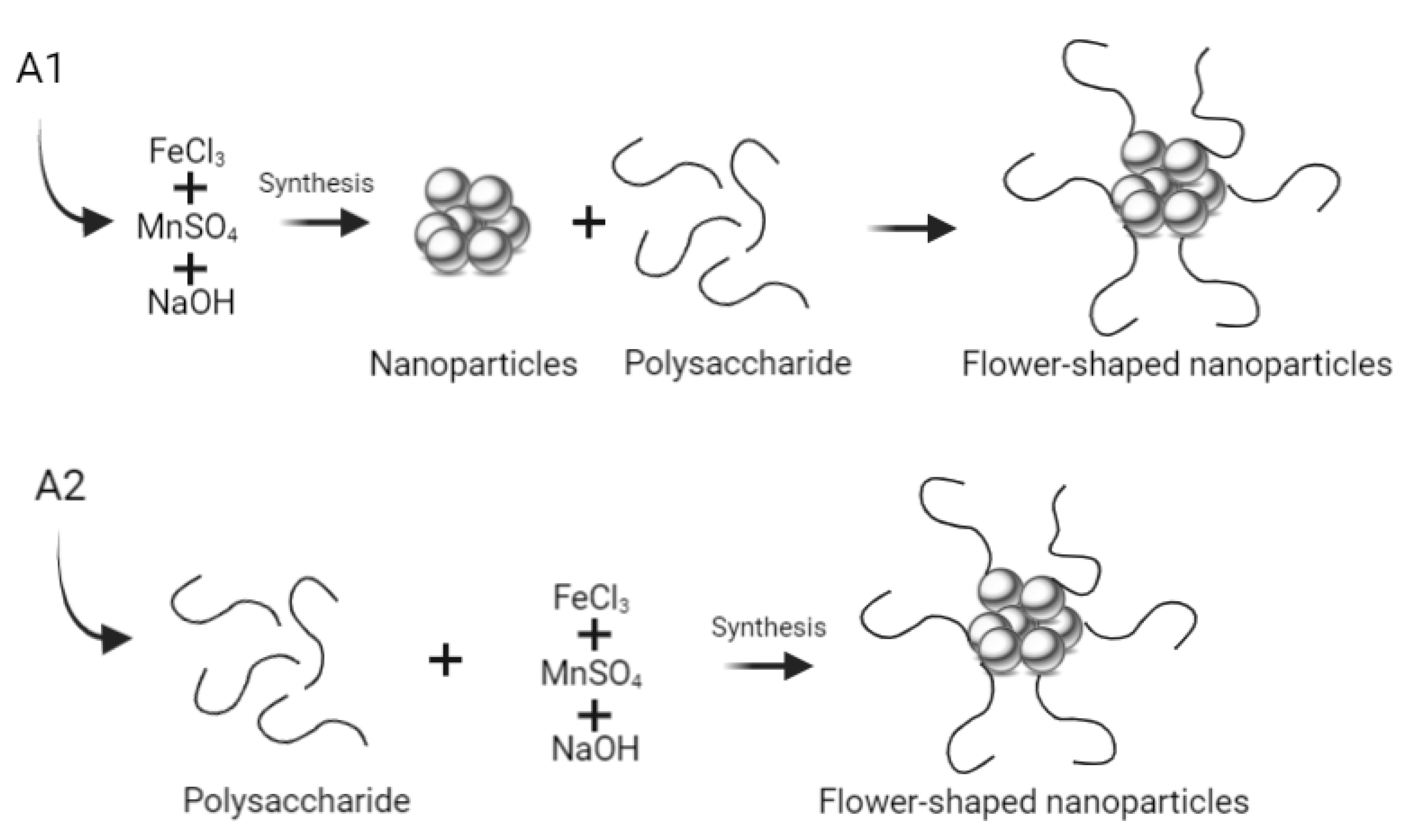
2.1. Synthesis of Multicore Manganese Ferrite NPs
2.1.1. Synthesis Using Carboxymethyl Dextran
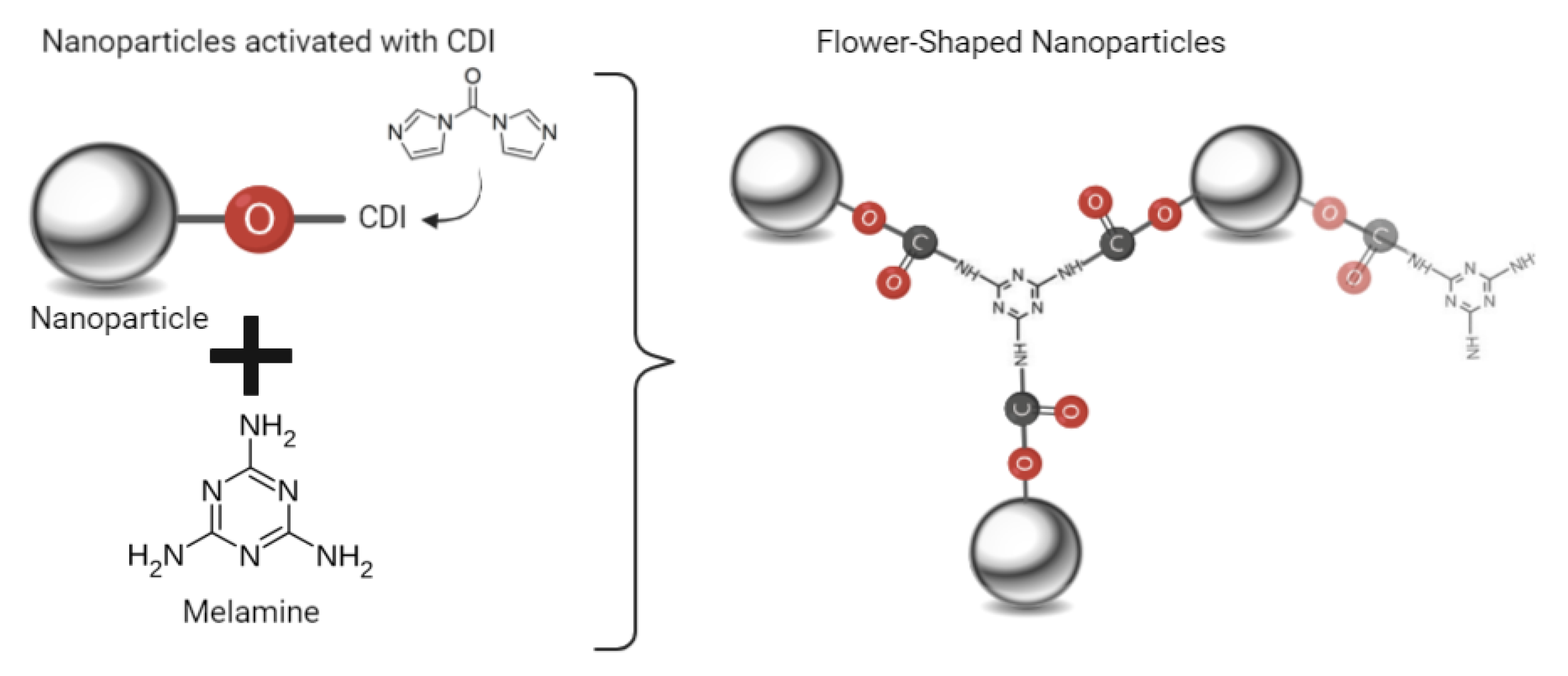
2.1.2. Synthesis Using Melamine
2.2. Magnetoliposomes Preparation
2.3. Sedimentation Curves of the Nanoparticles
2.4. Techniques for Sample Characterization
2.5. Photophysical Study of the Antitumor Compound in Solution
2.6. Encapsulation Efficiency of the Compound in the Magnetoliposomes
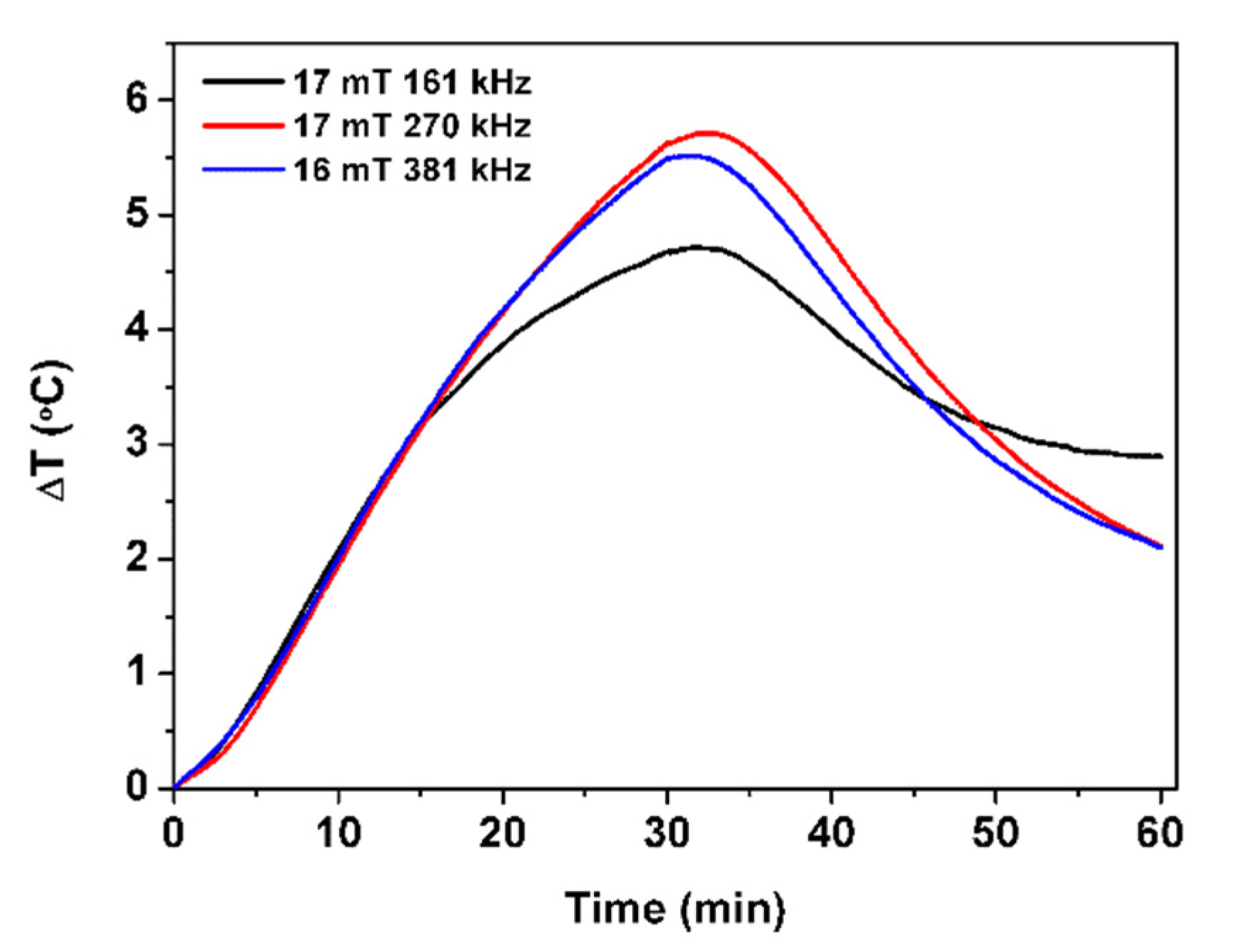
2.7. Magnetic Hyperthermia
3. Results and Discussion
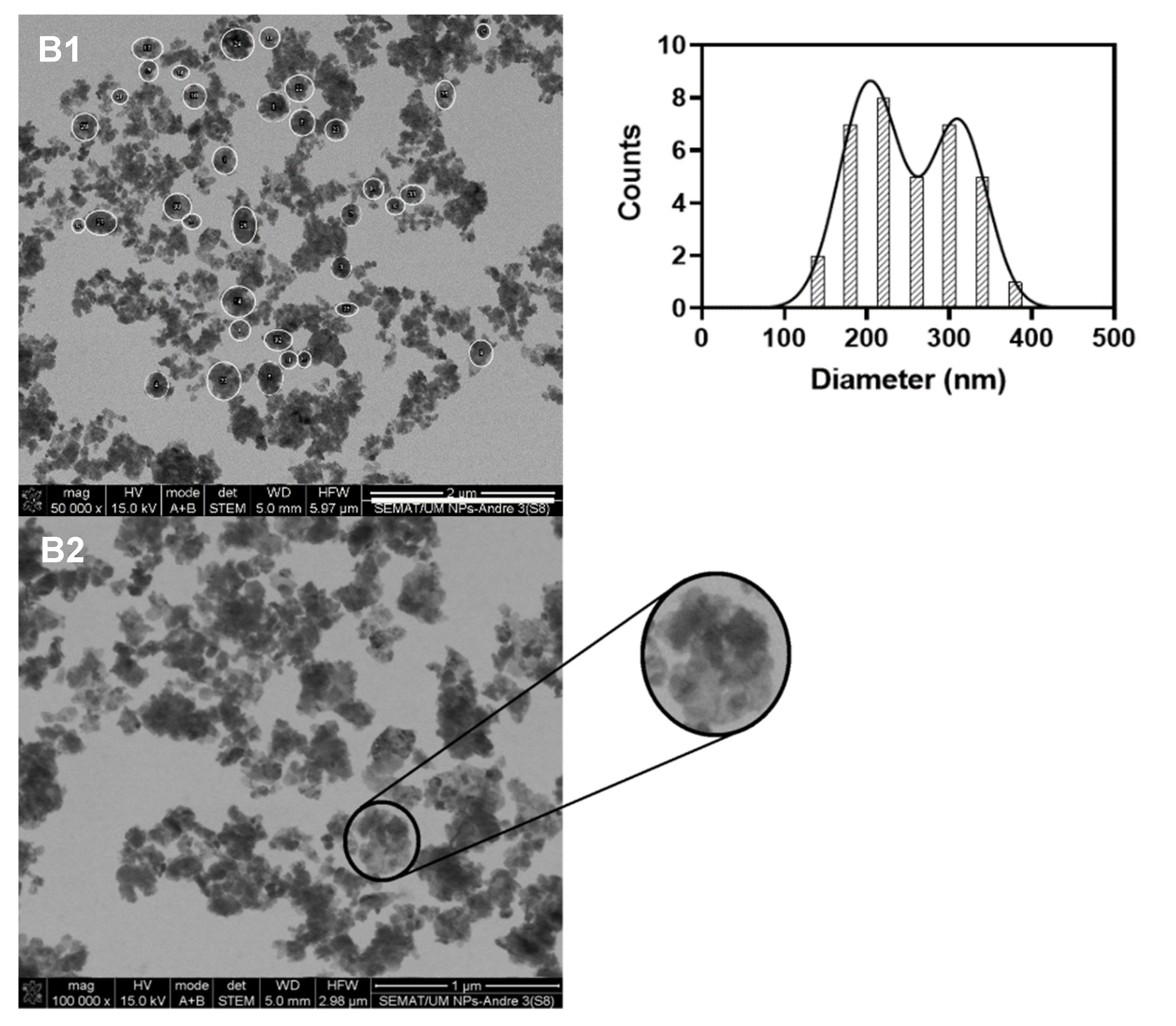
3.1. Characterization of the Multicore Nanoparticles
3.2. Magnetoliposomes Characterization
3.3. Drug-Loaded Magnetoliposomes
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, H.; Wang, H.; Wen, C. Effects of iron oxide nanoparticles as T2-MRI contrast agents on reproductive system in male mice. J. Nanobiotechnol. 2022, 20, 98. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; Zhang, C.; Shi, X.; Hu, H.; Du, X.; Fang, Y.; Ma, Y.; Wu, H.; Yang, S. Water-Soluble Superparamagnetic Manganese Ferrite NPs for Magnetic Resonance Imaging. Biomaterials 2010, 31, 3667–3673. [Google Scholar] [CrossRef] [PubMed]
- Pradhan, P.; Giri, J.; Banerjee, R.; Bellare, J.; Bahadur, D. Preparation and Characterization of Manganese Ferrite-Based Magnetic Liposomes for Hyperthermia Treatment of Cancer. J. Magn. Magn. Mater. 2007, 311, 208–215. [Google Scholar] [CrossRef]
- Mahmoudi, K.; Bouras, A.; Bozec, D.; Ivkov, R.; Hadjipanayis, C. Magnetic hyperthermia therapy for the treatment of glioblastoma: A review of the therapy’s history, efficacy and application in humans. Int. J. Hyperth. 2018, 34, 1316–1328. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gavilán, H.; Kowalski, A.; Heinke, D.; Sugunan, A.; Sommertune, J.; Varón, M.; Bogart, L.K.; Posth, O.; Zeng, L.; González-Alonso, D.; et al. Colloidal Flower-Shaped Iron Oxide NPs: Synthesis Strategies and Coatings. Part. Part. Syst. Charact. 2017, 34, 1700094. [Google Scholar] [CrossRef] [Green Version]
- Dutz, S. Are Magnetic Multicore NPs Promising Candidates for Biomedical Applications? IEEE Trans. Magn. 2016, 52, 0200103. [Google Scholar] [CrossRef]
- Bender, P.; Honecker, D.; Baruin, L.F. Supraferromagnetic correlations in clusters of magnetic nanoflowers. Appl. Phys. Lett. 2019, 115, 132406. [Google Scholar] [CrossRef]
- Storozhuk, L.; Besenhard, M.O.; Mourdikoudis, S.; LaGrow, A.P.; Lees, M.R.; Tung, L.D.; Gavriilidis, A.; Thanh, N.T.K. Stable Iron Oxide Nanoflowers with Exceptional Magnetic Heating Efficiency: Simple and Fast Polyol Synthesis. ACS Appl. Mater. Interfaces 2021, 13, 45870–45880. [Google Scholar] [CrossRef]
- Rodrigues, A.R.O.; Ramos, J.M.F.; Gomes, I.T.; Almeida, B.G.; Araújo, J.P.; Queiroz, M.-J.R.P.; Coutinho, P.J.G.; Castanheira, E.M.S. Magnetoliposomes based on manganese ferrite NPs as nanocarriers for antitumor drugs. RSC Adv. 2016, 6, 17302–17313; [Google Scholar] [CrossRef] [Green Version]
- Shubitidze, F.; Kekalo, K.; Stigliano, R.; Baker, I. Magnetic NPs with high specific absorption rate of electromagnetic energy at low field strength for hyperthermia therapy. J. Appl. Phys. 2015, 117, 094302. [Google Scholar] [CrossRef]
- Hu, F.; MacRenaris, K.W.; Waters, E.A.; Schultz-Sikma, E.A.; Eckermann, A.L.; Meade, T.J. Highly Dispersible, Superparamagnetic Magnetite Nanoflowers for Magnetic Resonance Imaging. Chem. Commun. 2010, 46, 73–75. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Bose, A.; Bothun, G.D. Controlled Release from Bilayer-Decorated Magnetoliposomes via Electromagnetic Heating. ACS Nano 2010, 4, 3215–3221. [Google Scholar] [CrossRef] [PubMed]
- Nappini, S.; Bonini, M.; Bombelli, F.B.; Pineider, F.; Sangregorio, C.; Baglioni, P.; Nordèn, B. Controlled Drug Release under a Low Frequency Magnetic Field: Effect of the Citrate Coating on Magnetoliposomes Stability. Soft Matter 2011, 7, 1025–1037. [Google Scholar] [CrossRef]
- Peetla, C.; Stine, A.; Labhasetwar, V. Biophysical Interactions with Model Lipid Membranes: Applications in Drug Discovery and Drug Delivery. Mol. Pharm. 2009, 6, 1264–1276. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, A.R.O.; Almeida, B.G.; Rodrigues, J.M.; Queiroz, M.-J.R.P.; Calhelha, R.C.; Ferreira, I.C.F.R.; Pires, A.; Pereira, A.M.; Araújo, J.P.; Coutinho, P.J.G.; et al. Magnetoliposomes as carriers for promising antitumor thieno[3,2-b]pyridin-7-arylamines: Photophysical and biological studies. RSC Adv. 2017, 7, 15352–15361. [Google Scholar] [CrossRef] [Green Version]
- Islam, K.; Haque, M.; Kumar, A.; Hoq, A.; Hyder, F.; Hoque, S.M. Manganese Ferrite Nanoparticles (MnFe2O4): Size Dependence for Hyperthermia and Negative/Positive Contrast Enhancement in MRI. Nanomaterials 2020, 10, 2297. [Google Scholar] [CrossRef]
- Rodrigues, J.M.; Calhelha, R.C.; Ferreira, I.C.F.R.; Queiroz, M.-J.R.P. Application of PEG400 in the one-pot synthesis of 7-[4-alkyl- or (hetero)aryl-1H-1,2,3-triazol-1-yl]thieno[3,2-b]pyridines via SNAr and Cu(I)-catalyzed Azide-Alkyne Cycloaddition and preliminary evaluation of their anti-tumour activity. Tetrahedron Lett. 2020, 61, 151900. [Google Scholar] [CrossRef]
- Han, D.; Han, S.-Y.; Lee, N.S.; Shin, J.; Jeong, Y.G.; Park, H.-W.; Kim, D.K. Magnetofluorescent Nanocomposite Comprised of Carboxymethyl Dextran Coated Superparamagnetic Iron Oxide NPs and β-Diketon Coordinated Europium Complexes. Nanomaterials 2019, 9, 62. [Google Scholar] [CrossRef] [Green Version]
- Woodman, E.K.; Chaffey, J.G.; Hopes, P.A.; Hose, D.R.; Gilday, J.P. N,N′-Carbonyldiimidazole-mediated amide coupling: Significant rate enhancement achieved by acid catalysis with imidazole·HCl. Org. Process Res. Dev. 2008, 13, 106–113. [Google Scholar] [CrossRef]
- Padiya, K.J.; Gavade, S.; Kardile, B.; Tiwari, M.; Bajare, S.; Mane, M.; Gaware, V.; Varghese, S.; Harel, D.; Kurhade, S. Unprecedented “in water” Imidazole Carbonylation: Paradigm Shift for Preparation of Urea and Carbamate. Org. Lett. 2012, 14, 2814–2817. [Google Scholar] [CrossRef]
- Rio, I.S.R.; Rodrigues, A.R.O.; Rodrigues, J.M.; Queiroz, M.-J.R.P.; Calhelha, R.C.; Ferreira, I.C.F.R.; Almeida, B.G.; Pires, A.; Pereira, A.M.; Araújo, J.P.; et al. Magnetoliposomes based on magnetic/plasmonic NPs loaded with tricyclic lactones for combined cancer therapy. Pharmaceutics 2021, 13, 1905. [Google Scholar] [CrossRef]
- Valeur, B. Molecular Fluorescence—Principles and Applications; Wiley-VCH: Weinheim, Germany, 2001. [Google Scholar]
- Tamba, Y.; Terashima, H.; Yamazaki, M. A Membrane Filtering Method for the Purification of Giant Unilamellar Vesicles. Chem. Phys. Lipids 2011, 164, 351–358. [Google Scholar] [CrossRef] [Green Version]
- Foderà, V.; Donald, A. Tracking the heterogeneous distribution of amyloid spherulites and their population balance with free fibrils. Eur. Phys. J. E 2010, 33, 273–282. [Google Scholar] [CrossRef]
- Eastman, J.W. Quantitative spectrofluorimetry—The Fluorescence quantum yield of quinine sulfate. Photochem. Photobiol. 1967, 6, 55–72. [Google Scholar] [CrossRef]
- Crosby, G.A.; Demas, J.N. Measurement of Photoluminescence Quantum Yields. Rev. J. Phys. Chem. 1971, 75, 991–1024. [Google Scholar] [CrossRef]
- Fery-Forgues, S.; Lavabre, D. Are Fluorescence Quantum Yields so Tricky to Measure? A Demonstration Using Familiar Stationery Products. J. Chem. Educ. 1999, 76, 1260. [Google Scholar] [CrossRef]
- Dash, S.; Murthy, P.N.; Nath, L.; Chowdhury, P. Kinetic modelling on drug release from controlled drug delivery systems. Acta Pol. Pharm. 2010, 67, 217–223. [Google Scholar] [PubMed]
- Kallumadil, M.; Tada, M.; Nakagawa, T.; Abe, M.; Southern, P.; Pankhurst, Q.A. Suitability of commercial colloids for magnetic hyperthermia. J. Magn. Magn. Mater. 2009, 321, 1509–1513. [Google Scholar] [CrossRef]
- Kang, H.; Rho, S.; Stiles, W.R.; Hu, S.; Baek, Y.; Hwang, D.W.; Kashiwagi, S.; Kim, M.S.; Choi, H.S. Size-Dependent EPR Effect of Polymeric Nanoparticles on Tumor Targeting. Adv. Healthc. Mater. 2020, 9, e1901223. [Google Scholar] [CrossRef]
- Cardoso, B.D.; Rodrigues, A.R.; Almeida, B.G.; Amorim, C.O.; Amaral, V.S.; Castanheira, E.M.S.; Coutinho, P.J.G. Stealth Magnetoliposomes Based on Calcium-Substituted Magnesium Ferrite NPs for Curcumin Transport and Release. Int. J. Mol. Sci. 2020, 21, 3641. [Google Scholar] [CrossRef]
- Bender, P.; Bogart, L.; Posth, O. Structural and magnetic properties of multi-core nanoparticles analysed using a generalised numerical inversion method. Sci. Rep. 2017, 7, 45990. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ilg, P. Equilibrium magnetization and magnetization relaxation of multicore magnetic nanoparticles. Phys. Rev. B 2017, 95, 214427. [Google Scholar] [CrossRef]
- Kekalo, K.; Baker, I.; Meyers, R.; Shyong, J. Magnetic Nanoparticles with High Specific Absorption Rate at Low Alternating Magnetic Field. Nano Life 2015, 5, 1550002. [Google Scholar] [CrossRef] [PubMed]
- Gupta, R.; Tomar, R.; Chakraverty, S.; Sharma, D. Effect of Manganese Doping on the Hyperthermic Profile of Ferrite NPs Using Response Surface Methodology. RSC Adv. 2021, 11, 16942–16954. [Google Scholar] [CrossRef]
- Liu, P.; Chen, G.; Zhang, J. A Review of Liposomes as a Drug Delivery System: Current Status of Approved Products, Regulatory Environments, and Future Perspectives. Molecules 2022, 27, 1372. [Google Scholar] [CrossRef]
- Queiroz, M.-J.R.P.; Dias, S.; Peixoto, D.; Rodrigues, A.R.O.; Oliveira, A.D.S.; Coutinho, P.J.G.; Vale-Silva, L.A.; Pinto, E.; Castanheira, E.M.S. New Potential Antitumoral Di(Hetero)Arylether Derivatives in the Thieno[3,2-b]Pyridine Series: Synthesis and Fluorescence Studies in Solution and in Nanoliposomes. J. Photochem. Photobiol. A Chem. 2012, 238, 71–80. [Google Scholar] [CrossRef] [Green Version]
- Queiroz, M.-J.R.P.; Calhelha, R.C.; Vale-Silva, L.; Pinto, E.; Nascimento, M.S.-J. Novel [6-(hetero)arylamino]thieno[3,2-b] pyridines: Synthesis and antitumoral activities. Eur. J. Med. Chem. 2010, 45, 5732–5738. [Google Scholar] [CrossRef]
- Machado, V.A.; Peixoto, D.; Costa, R.; Froufe, H.J.C.; Calhelha, R.C.; Abreu, R.M.V.; Ferreira, I.C.F.R.; Soares, R.; Queiroz, M.-J.R.P. Synthesis, antiangiogenesis evaluation and molecular docking studies of 1-aryl-3-[(thieno[3,2-b]pyridin- 7-ylthio)phenyl]ureas: Discovery of a new substitution pattern for type II VEGFR-2 Tyr kinase inhibitors. Bioorg. Med. Chem. 2015, 23, 6497–6509. [Google Scholar] [CrossRef] [Green Version]
- Machado, V.A.; Peixoto, D.; Queiroz, M.-J.R.P.; Soares, R. Antiangiogenic 1-aryl-3-[3-(thieno[3,2-b]pyridin- 7-ylthio)phenyl]ureas inhibit MCF-7 and MDA-MB-231 human breast cancer cell lines through PI3K/Akt and MAPK/Erk pathways. J. Cell. Biochem. 2016, 117, 2791–2799. [Google Scholar] [CrossRef]
- Costa, C.N.C.; Hortelão, A.C.L.; Ramos, J.M.F.; Oliveira, A.D.S.; Calhelha, R.C.; Queiroz, M.-J.R.P.; Coutinho, P.J.G.; Castanheira, E.M.S. A new antitumoral heteroarylaminothieno[3,2-b]pyridine derivative: Incorporation in liposomes and interaction with proteins monitored by fluorescence. Photochem. Photobiol. Sci. 2014, 13, 1730–1740. [Google Scholar] [CrossRef] [Green Version]








| Concentration (% m/v) | k (min−1) | ||
|---|---|---|---|
| NPs A1 | NPs A2 | NPs B | |
| 0.025 | 0.0011 | 0.0008 | 0.0017 |
| 0.05 | 0.0016 | 0.0008 | 0.0017 |
| 0.2 | 0.0034 | 0.0009 | 0.0021 |
| Coercivity (Oe) | Remnant Magnetization (emu/g) | Saturation Magnetization (emu/g) | Mr/Ms | |
|---|---|---|---|---|
| NPs B | 16.23 | 0.11 | 6.16 | 0.02 |
| 17 mT, 161 kHz | 17 mT, 270 kHz | 16 mT, 381 kHz | |
|---|---|---|---|
| SAR (W/g) | 0.12 | 0.10 | 0.14 |
| ILP (nH.m2/kg) | 0.46 | 0.20 | 0.22 |
| ΦFRET | R0 (Å) | r (Å) | |
|---|---|---|---|
| MLs | 0.68 | 13.6 | 12 |
| Hydrodynamic Diameter (nm) | PDI | Zeta Potential (mV) | |
|---|---|---|---|
| MLs NPs B | 388 ± 22 | 0.2 ± 0.11 | −2.4 ± 7.4 |
| Solvent | λabs/nm (ε/104 M−1 cm−1) | λem (nm) | ΦF |
|---|---|---|---|
| Ethyl acetate | 288 (1.1) | 333 | 0.04 |
| Acetonitrile | 301 (0.7) | 353 | 0.03 |
| Ethanol | 305 (0.7) | 357 | 0.03 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lopes, F.A.C.; Fernandes, A.V.F.; Rodrigues, J.M.; Queiroz, M.-J.R.P.; Almeida, B.G.; Pires, A.; Pereira, A.M.; Araújo, J.P.; Castanheira, E.M.S.; Rodrigues, A.R.O.; et al. Magnetoliposomes Containing Multicore Nanoparticles and a New Antitumor Thienopyridine Compound with Potential Application in Chemo/Thermotherapy. Biomedicines 2022, 10, 1547. https://doi.org/10.3390/biomedicines10071547
Lopes FAC, Fernandes AVF, Rodrigues JM, Queiroz M-JRP, Almeida BG, Pires A, Pereira AM, Araújo JP, Castanheira EMS, Rodrigues ARO, et al. Magnetoliposomes Containing Multicore Nanoparticles and a New Antitumor Thienopyridine Compound with Potential Application in Chemo/Thermotherapy. Biomedicines. 2022; 10(7):1547. https://doi.org/10.3390/biomedicines10071547
Chicago/Turabian StyleLopes, Fábio A. C., André V. F. Fernandes, Juliana M. Rodrigues, Maria-João R. P. Queiroz, Bernardo G. Almeida, Ana Pires, André M. Pereira, João P. Araújo, Elisabete M. S. Castanheira, Ana Rita O. Rodrigues, and et al. 2022. "Magnetoliposomes Containing Multicore Nanoparticles and a New Antitumor Thienopyridine Compound with Potential Application in Chemo/Thermotherapy" Biomedicines 10, no. 7: 1547. https://doi.org/10.3390/biomedicines10071547
APA StyleLopes, F. A. C., Fernandes, A. V. F., Rodrigues, J. M., Queiroz, M.-J. R. P., Almeida, B. G., Pires, A., Pereira, A. M., Araújo, J. P., Castanheira, E. M. S., Rodrigues, A. R. O., & Coutinho, P. J. G. (2022). Magnetoliposomes Containing Multicore Nanoparticles and a New Antitumor Thienopyridine Compound with Potential Application in Chemo/Thermotherapy. Biomedicines, 10(7), 1547. https://doi.org/10.3390/biomedicines10071547







