Effect of Lauric vs. Oleic Acid-Enriched Diets on Leptin Autoparacrine Signalling in Male Mice
Abstract
1. Introduction
2. Material and Methods
2.1. Animals and Diets
2.2. Determination of Subcutaneous and Visceral White Adipose Tissue Composition
2.3. Hematoxylin/Eosin Staining and Quantification of Adipocyte Size
2.4. Leptin Resistance Assessment in White Adipose Tissue
2.5. Western Blotting
2.6. RNA Preparation and Quantitative Real-Time PCR
2.7. Statistics
3. Results
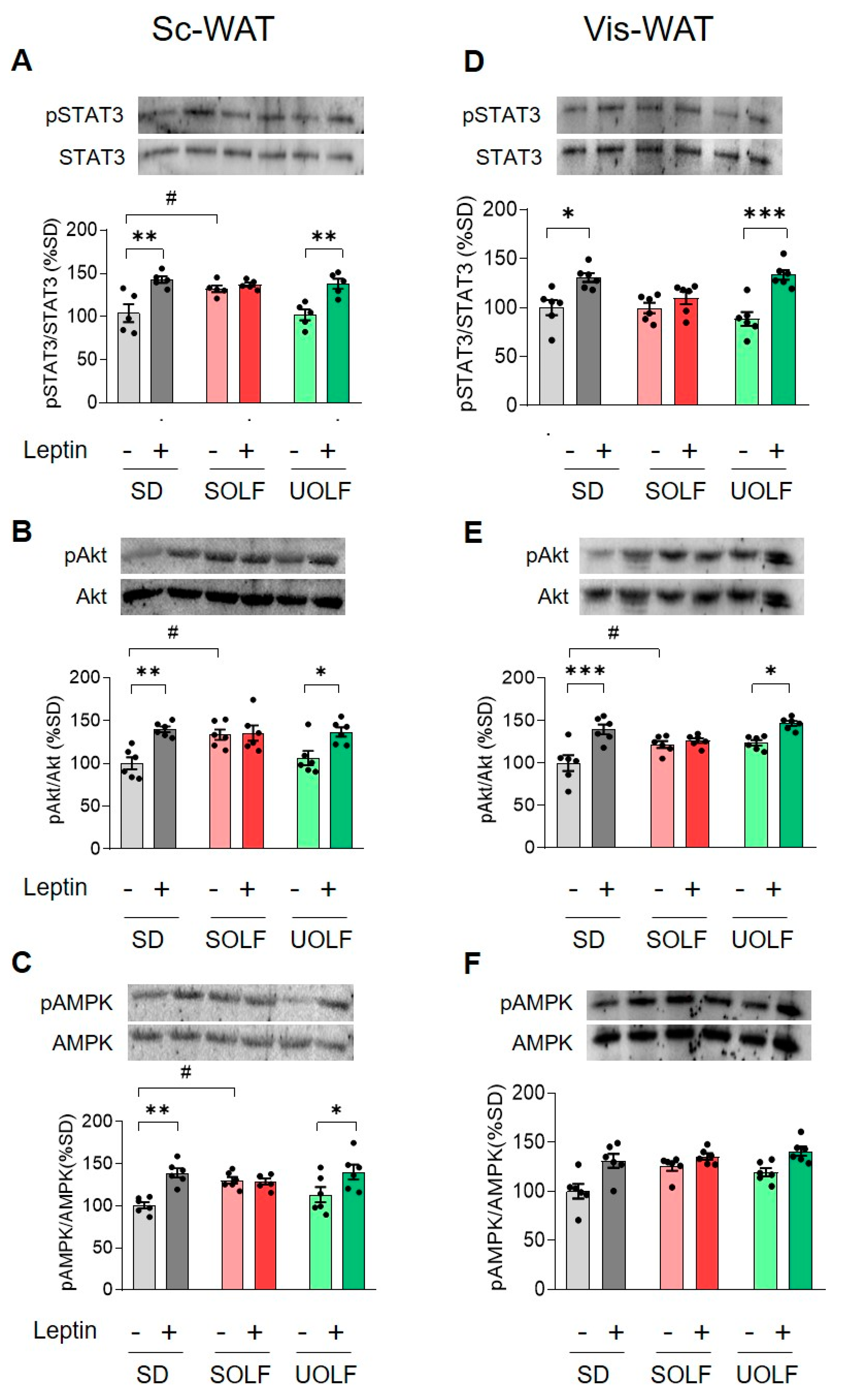
3.1. UOLF, but Not SOLF, Preserved the Effect of Exogenous Leptin on STAT3, Akt and AMPK Phosphorylation Both in Subcutaneous and Visceral WAT
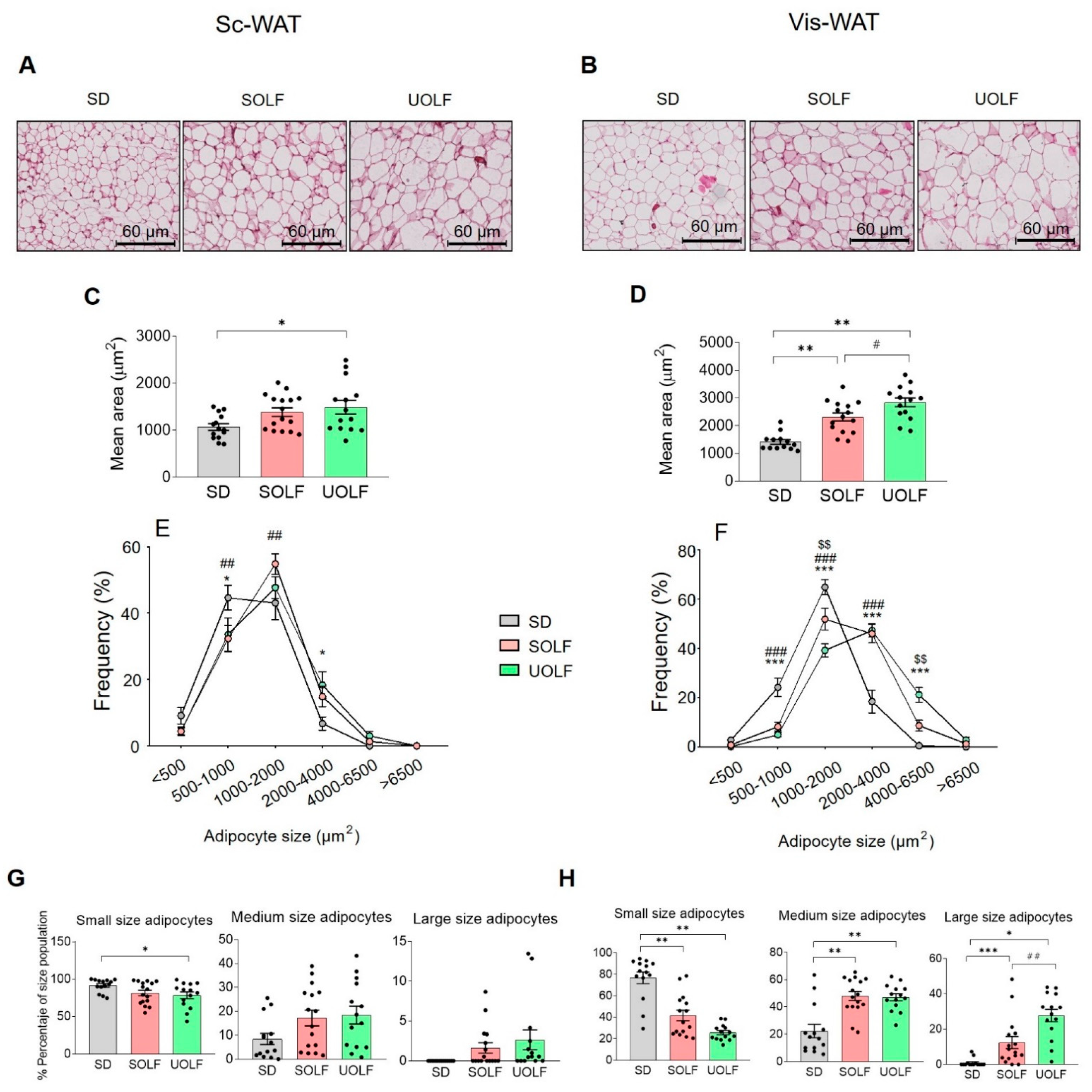
3.2. SOLF and UOLF Increased Adipocyte Size Both in Subcutaneous and Visceral WAT
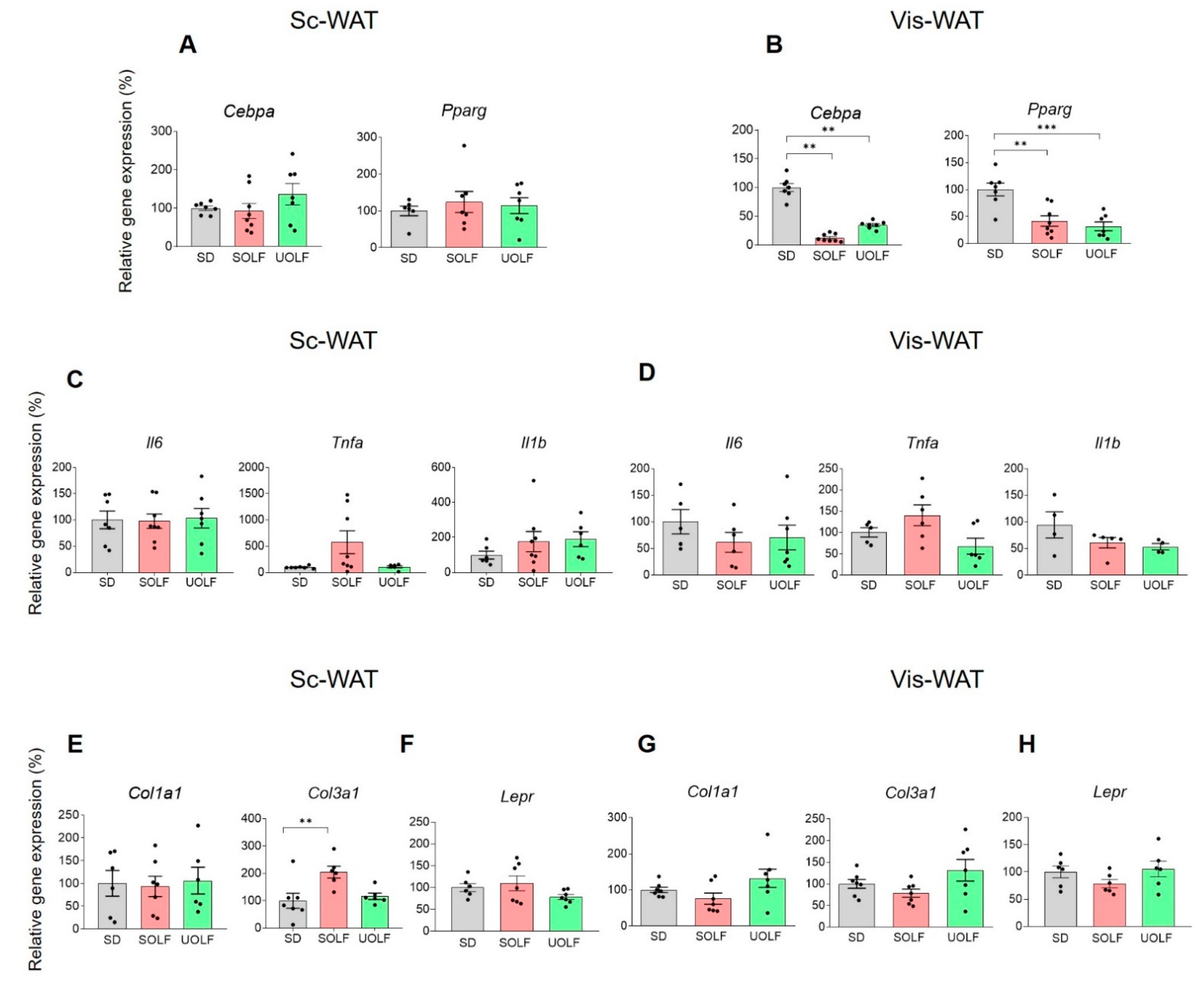
3.3. UOLF and SOLF Inhibited the Expression of Genes Involved in White Adipocyte Differentiation but Neither the Expression of Pro-Inflammatory and Pro-Fibrotic Factors nor Leptin Receptor Genes Were Affected
3.4. SOLF and UOLF Differently Affect the Composition of Subcutaneous and Visceral WAT
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Grundy, S.M. Adipose tissue and metabolic syndrome: Too much, too little or neither. Eur. J. Clin. Investig. 2015, 45, 1209–1217. [Google Scholar] [CrossRef] [PubMed]
- Wronska, A.; Kmiec, Z. Structural and biochemical characteristics of various white adipose tissue depots. Acta Physiol. 2012, 205, 194–208. [Google Scholar] [CrossRef] [PubMed]
- Romacho, T.; Elsen, M.; Röhrborn, D.; Eckel, J. Adipose tissue and its role in organ crosstalk. Acta Physiol. 2014, 210, 733–753. [Google Scholar] [CrossRef] [PubMed]
- Trayhurn, P.; Wood, I.S. Adipokines: Inflammation and the pleiotropic role of white adipose tissue. Br. J. Nutr. 2004, 92, 347–355. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Glazier, B.J.; Hinkel, B.C.; Cao, J.; Liu, L.; Liang, C.; Shi, H. Neuroendocrine Regulation of Energy Metabolism Involving Different Types of Adipose Tissues. Int. J. Mol. Sci. 2019, 20, 2707. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Sánchez, N. There and Back Again: Leptin Actions in White Adipose Tissue. Int. J. Mol. Sci. 2020, 21, 6039. [Google Scholar] [CrossRef]
- Considine, R.V.; Sinha, M.K.; Heiman, M.L.; Kriauciunas, A.; Stephens, T.W.; Nyce, M.R.; Ohannesian, J.P.; Marco, C.C.; McKee, L.J.; Bauer, T.L.; et al. Serum Immunoreactive-Leptin Concentrations in Normal-Weight and Obese Humans. N. Engl. J. Med. 1996, 334, 292–295. [Google Scholar] [CrossRef]
- Machinal-Quélin, F.; Dieudonné, M.N.; Leneveu, M.C.; Pecquery, R.; Giudicelli, Y. Proadipogenic effect of leptin on rat preadipocytes in vitro: Activation of MAPK and STAT3 signaling pathways. Am. J. Physiol. Physiol. 2002, 282, C853–C863. [Google Scholar] [CrossRef]
- Rhee, S.D.; Sung, Y.-Y.; Jung, W.H.; Cheon, H.G. Leptin inhibits rosiglitazone-induced adipogenesis in murine primary adipocytes. Mol. Cell. Endocrinol. 2008, 294, 61–69. [Google Scholar] [CrossRef]
- Guzmán-Ruiz, R.; Stucchi, P.; Ramos, M.P.; Sevillano, J.; Somoza, B.; Fernández-Alfonso, M.; Ruiz-Gayo, M. Leptin drives fat distribution during diet-induced obesity in mice. Endocrinol. Nutr. 2012, 59, 354–361. [Google Scholar] [CrossRef]
- Guzmán-Ruiz, R.; Somoza, B.; Gil-Ortega, M.; Merino, B.; Cano, V.; Attané, C.; Castan-Laurell, I.; Valet, P.; Fernández-Alfonso, M.S.; Ruiz-Gayo, M. Sensitivity of Cardiac Carnitine Palmitoyltransferase to Malonyl-CoA Is Regulated by Leptin: Similarities with a Model of Endogenous Hyperleptinemia. Endocrinology 2010, 151, 1010–1018. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Plaza, A.; Antonazzi, M.; Blanco-Urgoiti, J.; Del Olmo, N.; Ruiz-Gayo, M. Potential Role of Leptin in Cardiac Steatosis Induced by Highly Saturated Fat Intake during Adolescence. Mol. Nutr. Food Res. 2019, 63, 1900110. [Google Scholar] [CrossRef] [PubMed]
- Stucchi, P.; Guzmán-Ruiz, R.; Gil-Ortega, M.; Merino, B.; Somoza, B.; Cano, V.; de Castro, J.; Sevillano, J.; Ramos, M.P.; Fernández-Alfonso, M.S.; et al. Leptin resistance develops spontaneously in mice during adult life in a tissue-specific manner. Consequences for hepatic steatosis. Biochimie 2011, 93, 1779–1785. [Google Scholar] [CrossRef] [PubMed]
- Münzberg, H.; Flier, J.S.; Bjørbæk, C. Region-Specific Leptin Resistance within the Hypothalamus of Diet-Induced Obese Mice. Endocrinology 2004, 145, 4880–4889. [Google Scholar] [CrossRef]
- Rahmouni, K.; Morgan, D.A.; Morgan, G.M.; Mark, A.L.; Haynes, W.G. Role of Selective Leptin Resistance in Diet-Induced Obesity Hypertension. Diabetes 2005, 54, 2012–2018. [Google Scholar] [CrossRef]
- Wang, M.-Y.; Orci, L.; Ravazzola, M.; Unger, R.H. Fat storage in adipocytes requires inactivation of leptin’s paracrine activity: Implications for treatment of human obesity. Proc. Natl. Acad. Sci. USA 2005, 102, 18011–18016. [Google Scholar] [CrossRef]
- Unger, R.H. Lipotoxic Diseases. Annu. Rev. Med. 2002, 53, 319–336. [Google Scholar] [CrossRef]
- Unger, R.H.; Zhou, Y.-T.; Orci, L. Regulation of fatty acid homeostasis in cells: Novel role of leptin. Proc. Natl. Acad. Sci. USA 1999, 96, 2327–2332. [Google Scholar] [CrossRef] [PubMed]
- Wang, M.-Y.; Grayburn, P.; Chen, S.; Ravazzola, M.; Orci, L.; Unger, R.H. Adipogenic capacity and the susceptibility to type 2 diabetes and metabolic syndrome. Proc. Natl. Acad. Sci. USA 2008, 105, 6139–6144. [Google Scholar] [CrossRef]
- Couillard, C.; Mauriège, P.; Imbeault, P.; Prud’homme, D.; Nadeau, A.; Tremblay, A.; Bouchard, C.; Després, J.-P. Hyperleptinemia is more closely associated with adipose cell hypertrophy than with adipose tissue hyperplasia. Int. J. Obes. 2000, 24, 782–788. [Google Scholar] [CrossRef]
- Lee, Y.; Yu, X.; Gonzales, F.; Mangelsdorf, D.J.; Wang, M.-Y.; Richardson, C.; Witters, L.A.; Unger, R.H. PPARα is necessary for the lipopenic action of hyperleptinemia on white adipose and liver tissue. Proc. Natl. Acad. Sci. USA 2002, 99, 11848–11853. [Google Scholar] [CrossRef] [PubMed]
- Herrera, E.; Ayanz, A. Calculation of lipolysis and esterification from glycerol metabolism in rat adipose tissue. J. Lipid Res. 1972, 13, 802–809. [Google Scholar] [CrossRef]
- Giraudeau, P.; Baguet, E. Improvement of the inverse-gated-decoupling sequence for a faster quantitative analysis of various samples by 13C NMR spectroscopy. J. Magn. Reson. 2006, 180, 110–117. [Google Scholar] [CrossRef] [PubMed]
- Verboven, K.; Wouters, K.; Gaens, K.; Hansen, D.; Bijnen, M.; Wetzels, S.; Stehouwer, C.D.; Goossens, G.; Schalkwijk, C.G.; Blaak, E.E.; et al. Abdominal subcutaneous and visceral adipocyte size, lipolysis and inflammation relate to insulin resistance in male obese humans. Sci. Rep. 2018, 8, 4677. [Google Scholar] [CrossRef] [PubMed]
- Livak, K.J.; Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2−ΔΔCT Method. Methods 2001, 25, 402–408. [Google Scholar] [CrossRef]
- Ahima, R.S.; Flier, J.S. Adipose Tissue as an Endocrine Organ. Trends Endocrinol. Metab. 2000, 11, 327–332. [Google Scholar] [CrossRef]
- Dunmore, S.J.; Brown, J.E.P. The role of adipokines in β-cell failure of type 2 diabetes. J. Endocrinol. 2012, 216, T37–T45. [Google Scholar] [CrossRef]
- Stern, J.H.; Rutkowski, J.M.; Scherer, P.E. Adiponectin, Leptin, and Fatty Acids in the Maintenance of Metabolic Homeostasis through Adipose Tissue Crosstalk. Cell Metab. 2016, 23, 770–784. [Google Scholar] [CrossRef]
- Fernández-Felipe, J.; Merino, B.; Sanz-Martos, A.; Plaza, A.; Contreras, A.; Naranjo, V.; Morales, L.; Chowen, J.; Cano, V.; Ruiz-Gayo, M.; et al. Saturated and unsaturated fat diets impair hippocampal glutamatergic transmission in adolescent mice. Psychoneuroendocrinology 2021, 133, 105429. [Google Scholar] [CrossRef]
- Zhang, K.; Guo, W.; Yang, Y.; Wu, J. JAK2/STAT3 pathway is involved in the early stage of adipogenesis through regulatingC/EBPβtranscription. J. Cell. Biochem. 2011, 112, 488–497. [Google Scholar] [CrossRef]
- Harris, R.B. Direct and indirect effects of leptin on adipocyte metabolism. Biochim. Biophys. Acta 2013, 1842, 414–423. [Google Scholar] [CrossRef] [PubMed]
- Deng, J.; Hua, K.; Lesser, S.S.; Harp, J.B. Activation of Signal Transducer and Activator of Transcription-3 during Proliferative Phases of 3T3-L1 Adipogenesis. Endocrinology 2000, 141, 2370–2376. [Google Scholar] [CrossRef] [PubMed]
- Richard, A.J.; Stephens, J.M. The role of JAK–STAT signaling in adipose tissue function. Biochim. Biophys. Acta 2013, 1842, 431–439. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, C.M.; Emanuelli, B.; Kahn, C.R. Critical nodes in signalling pathways: Insights into insulin action. Nat. Rev. Mol. Cell Biol. 2006, 7, 85–96. [Google Scholar] [CrossRef]
- Habinowski, S.A.; Witters, L.A. The Effects of AICAR on Adipocyte Differentiation of 3T3-L1 Cells. Biochem. Biophys. Res. Commun. 2001, 286, 852–856. [Google Scholar] [CrossRef]
- Villena, J.A.; Viollet, B.; Andreelli, F.; Kahn, A.; Vaulont, S.; Sul, H.S. Induced Adiposity and Adipocyte Hypertrophy in Mice Lacking the AMP-Activated Protein Kinase-α2 Subunit. Diabetes 2004, 53, 2242–2249. [Google Scholar] [CrossRef] [PubMed]
- Fritsche, K.L. The Science of Fatty Acids and Inflammation. Adv. Nutr. Int. Rev. J. 2015, 6, 293S–301S. [Google Scholar] [CrossRef]
- Al Hasan, M.; Martin, P.; Shu, X.; Patterson, S.; Bartholomew, C. Type III Collagen is Required for Adipogenesis and Actin Stress Fibre Formation in 3T3-L1 Preadipocytes. Biomolecules 2021, 11, 156. [Google Scholar] [CrossRef]
- Hajri, T.; Hall, A.M.; Jensen, D.R.; Pietka, T.A.; Drover, V.A.; Tao, H.; Eckel, R.; Abumrad, N.A. CD36-Facilitated Fatty Acid Uptake Inhibits Leptin Production and Signaling in Adipose Tissue. Diabetes 2007, 56, 1872–1880. [Google Scholar] [CrossRef]
- Cheng, L.; Yu, Y.; Szabo, A.; Wu, Y.; Wang, H.; Camer, D.; Huang, X.-F. Palmitic acid induces central leptin resistance and impairs hepatic glucose and lipid metabolism in male mice. J. Nutr. Biochem. 2015, 26, 541–548. [Google Scholar] [CrossRef]
- Saraswathi, V.; Kumar, N.; Gopal, T.; Bhatt, S.; Ai, W.; Ma, C.; Talmon, G.; Desouza, C. Lauric Acid versus Palmitic Acid: Effects on Adipose Tissue Inflammation, Insulin Resistance, and Non-Alcoholic Fatty Liver Disease in Obesity. Biology 2020, 9, 346. [Google Scholar] [CrossRef] [PubMed]
- Farmer, S.R. Transcriptional control of adipocyte formation. Cell Metab. 2006, 4, 263–273. [Google Scholar] [CrossRef] [PubMed]
- Vishvanath, L.; Gupta, R.K. Contribution of adipogenesis to healthy adipose tissue expansion in obesity. J. Clin. Investig. 2019, 129, 4022–4031. [Google Scholar] [CrossRef] [PubMed]
- Plaza, A.; Merino, B.; Cano, V.; Domínguez, G.; Pérez-Castells, J.; Fernandez-Alfonso, M.S.; Sengenes, C.; Chowen, J.A.; Ruiz-Gayo, M. Cholecystokinin is involved in triglyceride fatty acid uptake by rat adipose tissue. J. Endocrinol. 2018, 236, 137–150. [Google Scholar] [CrossRef] [PubMed]
- Shoham, N.; Girshovitz, P.; Katzengold, R.; Shaked, N.T.; Benayahu, D.; Gefen, A. Adipocyte Stiffness Increases with Accumulation of Lipid Droplets. Biophys. J. 2014, 106, 1421–1431. [Google Scholar] [CrossRef]
- Shoham, N.; Gefen, A. Mechanotransduction in adipocytes. J. Biomech. 2011, 45, 1–8. [Google Scholar] [CrossRef]
- Hsieh, C.-M.; Liu, P.Y.; Chin, L.K.; Zhang, J.B.; Wang, K.; Sung, K.-B.; Ser, W.; Bourouina, T.; Leprince-Wang, Y.; Liu, A.-Q. Regulation of lipid droplets in live preadipocytes using optical diffraction tomography and Raman spectroscopy. Opt. Express 2019, 27, 22994–23008. [Google Scholar] [CrossRef]
- Hossain, G.; Iwata, T.; Mizusawa, N.; Shima, S.W.N.; Okutsu, T.; Ishimoto, K.; Yoshimoto, K. Compressive force inhibits adipogenesis through COX-2-mediated down-regulation of PPARγ2 and C/EBPα. J. Biosci. Bioeng. 2010, 109, 297–303. [Google Scholar] [CrossRef]
- Maddalozzo, G.F.; Iwaniec, U.T.; Turner, R.T.; Rosen, C.J.; Widrick, J.J. Whole-body vibration slows the acquisition of fat in mature female rats. Int. J. Obes. 2008, 32, 1348–1354. [Google Scholar] [CrossRef]
- Phillips, R.; Ursell, T.; Wiggins, P.; Sens, P. Emerging roles for lipids in shaping membrane-protein function. Nature 2009, 459, 379–385. [Google Scholar] [CrossRef] [PubMed]
- Abbott, S.K.; Else, P.L.; Atkins, T.A.; Hulbert, A. Fatty acid composition of membrane bilayers: Importance of diet polyunsaturated fat balance. Biochim. Biophys. Acta 2012, 1818, 1309–1317. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.J.; Miller, L.J. Changes in the plasma membrane in metabolic disease: Impact of the membrane environment on G protein-coupled receptor structure and function. Br. J. Pharmacol. 2017, 175, 4009–4025. [Google Scholar] [CrossRef] [PubMed]
- Ge, G.; Wu, J.; Lin, Q. Effect of Membrane Fluidity on Tyrosine Kinase Activity of Reconstituted Epidermal Growth Factor Receptor. Biochem. Biophys. Res. Commun. 2001, 282, 511–514. [Google Scholar] [CrossRef] [PubMed]
- Sellem, F.; Pesando, D.; Bodennec, G.; Girard, J.-P.; Simopoulos, A.P. An Increase in the Omega-6/Omega-3 Fatty Acid Ratio Increases the Risk for Obesity. Nutrients 2016, 8, 128. [Google Scholar] [CrossRef]
| Antigen | Manufacturer | Ref. | Host and Molecular Weight | Dilution |
|---|---|---|---|---|
| STAT3 | Santa Cruz Biotechnology | Sc-483 | Rabbit polyclonal, 86 kDa | 1/250 |
| pSTAT3 (Y705) | Cell Signalling | #9131 | Rabbit polyclonal, 86 kDa | 1/250 |
| Akt | Cell Signalling | #9272 | Rabbit polyclonal, 60 kDa | 1/500 |
| pAkt (S473) | Cell Signalling | #9271 | Rabbit polyclonal, 60 kDa | 1/500 |
| AMPK | Cell Signalling | #2532 | Rabbit polyclonal, 62 kDa | 1/500 |
| pAMPK (T172) | Cell Signalling | #2531 | Rabbit polyclonal, 62 kDa | 1/500 |
| Anti-rabbit IgG | Santa Cruz Biotechnology | Sc-2357 | Mouse monoclonal, HRP | 1/2000 |
| mRNA | Forward (5′→3′) | Reverse (5′→3′) |
|---|---|---|
| Cebpa | CCGATGAGCAGTCACCTCC | AGGAACTCGTCGTTGAAGGC |
| Col1a1 | ACTGCCCTCCTGACGCAT | AGAAAGCACAGCACTCGCC |
| Col3a1 | CCCATGACTGTCCCACGTAAGCAC | TGGCCTGATCCATATAGGCAATACTG |
| Il1b | TGCCACCTTTTGACAGTGATG | TGATACTGCCTGCCTGAAGC |
| Il6 | TACCACTTCACAAGTCGGAGGC | CTGCAAGTGCATCATCGTTGTTC |
| Pparg | CATGGTTGACACAGAGATGCCATTCTG | TTGATCGCACTTTGGTATTCTTGGAGC |
| Tnfa | GGTGCCTATGTCTCAGCCTC | GCTCCTCCACTTGGTGGTTT |
| Lepr | GCAGCAAAAGGAAGCATTGGA | GGTGAGGAGCAAGAGACTGG |
| 18s | GGGAGCCTGAGAAACGGC | GGGTCGGGAGTGGGTAATTT |
| Actb | TGGTGGGAATGGGTCAGAAGGACTC | CATGGCTGGGGTGTTGAAGGTCTCA |
| F Value | |||
|---|---|---|---|
| Protein | Leptin Effect | Diet Effect | Interaction Leptin × Diet |
| pSTAT3/STAT3 | F(1,30) = 42.89; p < 0.001 | F(2,30) = 6.890; p < 0.01 | F(2,30) = 4.393; p < 0.05 |
| pAkt/Akt | F(1,30) = 18.92; p < 0.001 | F(2,30) = 2.785; p = 0.07 | F(2,30) = 4.214; p < 0.05 |
| pAMPK/AMPK | F(1,30) = 17.86; p < 0.001 | F(2,30) = 1.321; n.s. | F(2,30) = 5.466; p < 0.01 |
| F Value | |||
|---|---|---|---|
| Protein | Leptin Effect | Diet Effect | Interaction Leptin × Diet |
| pSTAT3/STAT3 | F(1,30) = 32.93; p < 0.001 | F(2,30) = 1.558; n.s. | F(2,30) = 4.114; p < 0.05 |
| pAkt/Akt | F(1,30) = 27.75; p < 0.001 | F(2,30) = 4.705; p < 0.05 | F(2,30) = 5.727; p < 0.01 |
| pAMPK/AMPK | F(1,30) = 22.64; p < 0.001 | F(2,30) = 4.996; p < 0.05 | F(2,30) = 1.914; n.s. |
| Vis-WAT | ||||||
|---|---|---|---|---|---|---|
| Standard Diet | SOLF | UOLF | ||||
| FA | Mol% | Mass% | Mol% | Mass% | Mol% | Mass% |
| Oleic | 45.4 ± 2.3 | 47.9 ± 2.4 | 36.5 ± 1.8 | 42.3 ± 2.1 | 87.7 ± 4.4 | 89.0 ± 4.4 |
| Palmitic | 54.5 ± 2.7 | 52.1 ± 2.6 | 10.2 ± 0.5 | 10.8 ± 0.5 | 12.2 ± 0.6 | 11.0 ± 0.5 |
| Myristic | ND | ND | 26.6 ± 1.3 | 25.0 ± 1.2 | ND | ND |
| Lauric | ND | ND | 26.6 ± 1.3 | 21.9 ± 1.1 | ND | ND |
| Linoleic | ND | ND | ND | ND | ND | ND |
| Sc-WAT | ||||||
| Oleic | 35.6 ± 1.8 | 36.8 ± 1.8 | 37.7 ± 1.9 | 41.6 ± 2.1 | 81.9 ± 4.1 | 82.7 ± 4.1 |
| Palmitic | 33.1 ± 1.6 | 31.1 ± 1.5 | 20.0 ± 1.0 | 20.0 ± 1.0 | 8.1 ± 0.4 | 7.5 ± 0.4 |
| Myristic | ND | ND | 9.8 ± 0.5 | 8.7 ± 0.4 | ND | ND |
| Lauric | ND | ND | 18.5 ± 0.9 | 14.4 ± 0.7 | ND | ND |
| Linoleic | 31.3 ± 1.6 | 32.1 ± 1.6 | 13.9 ± 0.7 | 15.5 ± 0.8 | 9.8 ± 0.5 | 9.8 ± 0.5 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fernández-Felipe, J.; Plaza, A.; Domínguez, G.; Pérez-Castells, J.; Cano, V.; Cioni, F.; Del Olmo, N.; Ruiz-Gayo, M.; Merino, B. Effect of Lauric vs. Oleic Acid-Enriched Diets on Leptin Autoparacrine Signalling in Male Mice. Biomedicines 2022, 10, 1864. https://doi.org/10.3390/biomedicines10081864
Fernández-Felipe J, Plaza A, Domínguez G, Pérez-Castells J, Cano V, Cioni F, Del Olmo N, Ruiz-Gayo M, Merino B. Effect of Lauric vs. Oleic Acid-Enriched Diets on Leptin Autoparacrine Signalling in Male Mice. Biomedicines. 2022; 10(8):1864. https://doi.org/10.3390/biomedicines10081864
Chicago/Turabian StyleFernández-Felipe, Jesús, Adrián Plaza, Gema Domínguez, Javier Pérez-Castells, Victoria Cano, Francesco Cioni, Nuria Del Olmo, Mariano Ruiz-Gayo, and Beatriz Merino. 2022. "Effect of Lauric vs. Oleic Acid-Enriched Diets on Leptin Autoparacrine Signalling in Male Mice" Biomedicines 10, no. 8: 1864. https://doi.org/10.3390/biomedicines10081864
APA StyleFernández-Felipe, J., Plaza, A., Domínguez, G., Pérez-Castells, J., Cano, V., Cioni, F., Del Olmo, N., Ruiz-Gayo, M., & Merino, B. (2022). Effect of Lauric vs. Oleic Acid-Enriched Diets on Leptin Autoparacrine Signalling in Male Mice. Biomedicines, 10(8), 1864. https://doi.org/10.3390/biomedicines10081864









