Immunological Phenotyping of Mice with a Point Mutation in Cdk4
Abstract
:1. Introduction
2. Materials and Methods
2.1. Mice
2.2. Cell Preparation and Flow Cytometry
2.3. Bromodeoxyuridine (BrdU) Labelling
2.4. Intracellular Cytokine Production
2.5. Generation of Bone Marrow Chimeras
2.6. Statistical Analysis
3. Results
3.1. The Effect of the CDK4wnch Mutation on Immune Cell Subsets
3.2. Proliferation Capacity of Cdk4wnch/wnch Cells Is Normal
3.3. Cytokine Production by Cdk4wnch/wnch T Cells Is Normal
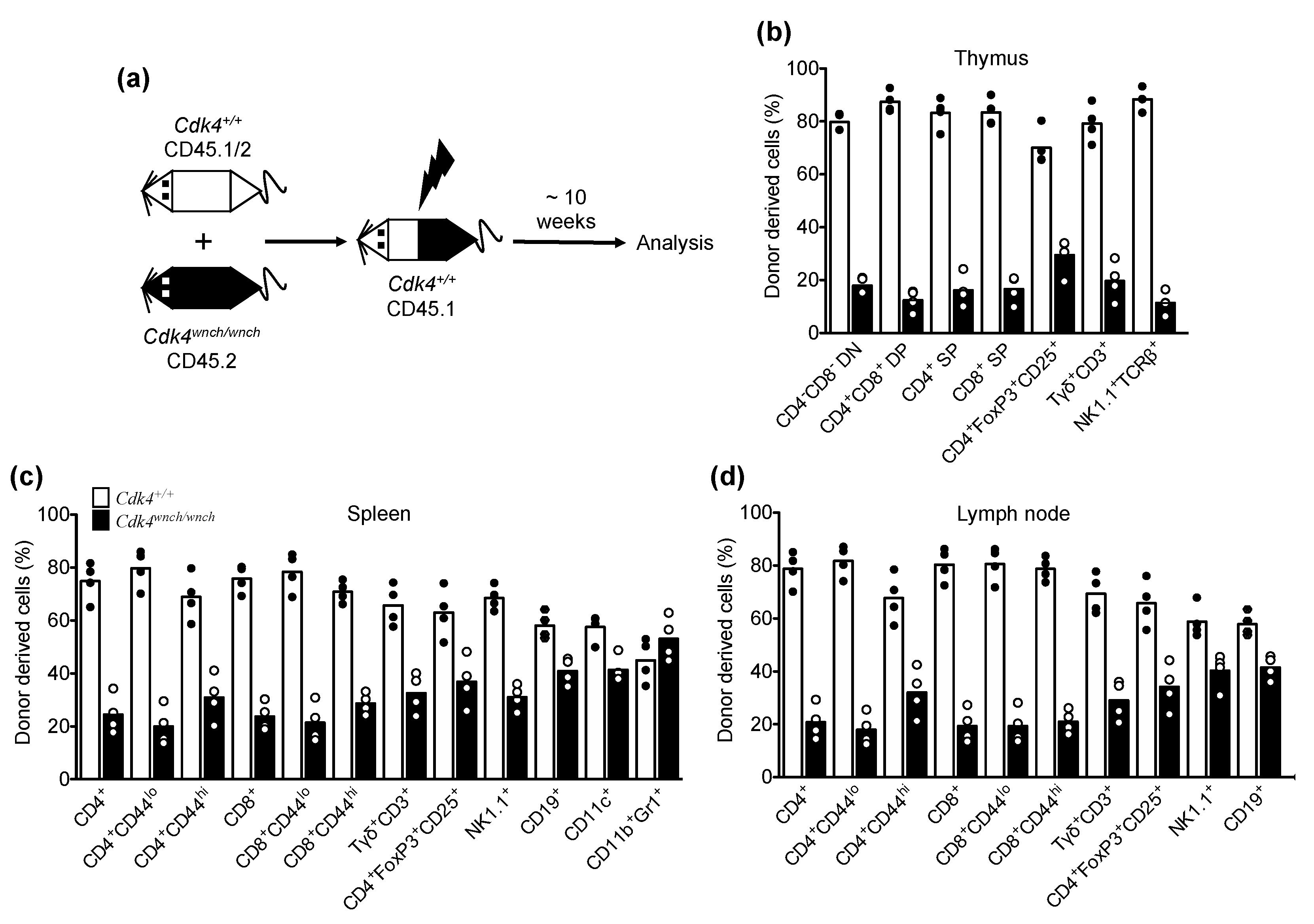
3.4. Reduced Cdk4wnch/wnch-Derived T Cell Subsets in Mixed Bone Marrow Chimeric Recipients
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Swaffer, M.P.; Jones, A.W.; Flynn, H.R.; Snijders, A.P.; Nurse, P. CDK Substrate Phosphorylation and Ordering the Cell Cycle. Cell 2016, 167, 1750–1761.e1716. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.; Kaldis, P. Cdks, cyclins and CKIs: Roles beyond cell cycle regulation. Development 2013, 140, 3079–3093. [Google Scholar] [CrossRef] [PubMed]
- Dyson, N. The regulation of E2F by pRB-family proteins. Genes Dev. 1998, 12, 2245–2262. [Google Scholar] [CrossRef] [PubMed]
- Hatakeyama, M.; Brill, J.A.; Fink, G.R.; Weinberg, R.A. Collaboration of G1 cyclins in the functional inactivation of the retinoblastoma protein. Genes Dev. 1994, 8, 1759–1771. [Google Scholar] [CrossRef] [PubMed]
- Lundberg, A.S.; Weinberg, R.A. Functional inactivation of the retinoblastoma protein requires sequential modification by at least two distinct cyclin-cdk complexes. Mol. Cell. Biol. 1998, 18, 753–761. [Google Scholar] [CrossRef]
- Stevaux, O.; Dyson, N.J. A revised picture of the E2F transcriptional network and RB function. Curr. Opin. Cell Biol. 2002, 14, 684–691. [Google Scholar] [CrossRef]
- Muljo, S.A.; Schlissel, M.S. Pre-B and pre-T-cell receptors: Conservation of strategies in regulating early lymphocyte development. Immunol. Rev. 2000, 175, 80–93. [Google Scholar] [CrossRef]
- Cooper, A.B.; Sawai, C.M.; Sicinska, E.; Powers, S.E.; Sicinski, P.; Clark, M.R.; Aifantis, I. A unique function for cyclin D3 in early B cell development. Nat. Immunol. 2006, 7, 489–497. [Google Scholar] [CrossRef]
- Sawai, C.M.; Freund, J.; Oh, P.; Ndiaye-Lobry, D.; Bretz, J.C.; Strikoudis, A.; Genesca, L.; Trimarchi, T.; Kelliher, M.A.; Clark, M.; et al. Therapeutic targeting of the cyclin D3:CDK4/6 complex in T cell leukemia. Cancer Cell 2012, 22, 452–465. [Google Scholar] [CrossRef]
- Sicinska, E.; Aifantis, I.; Le Cam, L.; Swat, W.; Borowski, C.; Yu, Q.; Ferrando, A.A.; Levin, S.D.; Geng, Y.; von Boehmer, H.; et al. Requirement for cyclin D3 in lymphocyte development and T cell leukemias. Cancer Cell 2003, 4, 451–461. [Google Scholar] [CrossRef]
- Glassford, J.; Vigorito, E.; Soeiro, I.; Madureira, P.A.; Zoumpoulidou, G.; Brosens, J.J.; Turner, M.; Lam, E.W. Phosphatidylinositol 3-kinase is required for the transcriptional activation of cyclin D2 in BCR activated primary mouse B lymphocytes. Eur. J. Immunol. 2005, 35, 2748–2761. [Google Scholar] [CrossRef] [PubMed]
- Barata, J.T.; Cardoso, A.A.; Nadler, L.M.; Boussiotis, V.A. Interleukin-7 promotes survival and cell cycle progression of T-cell acute lymphoblastic leukemia cells by down-regulating the cyclin-dependent kinase inhibitor p27(kip1). Blood 2001, 98, 1524–1531. [Google Scholar] [CrossRef] [PubMed]
- Di Santo, J.P.; Aifantis, I.; Rosmaraki, E.; Garcia, C.; Feinberg, J.; Fehling, H.J.; Fischer, A.; von Boehmer, H.; Rocha, B. The common cytokine receptor gamma chain and the pre-T cell receptor provide independent but critically overlapping signals in early alpha/beta T cell development. J. Exp. Med. 1999, 189, 563–574. [Google Scholar] [CrossRef] [PubMed]
- Parrish-Novak, J.; Dillon, S.R.; Nelson, A.; Hammond, A.; Sprecher, C.; Gross, J.A.; Johnston, J.; Madden, K.; Xu, W.; West, J.; et al. Interleukin 21 and its receptor are involved in NK cell expansion and regulation of lymphocyte function. Nature 2000, 408, 57–63. [Google Scholar] [CrossRef]
- Schneider, P.; MacKay, F.; Steiner, V.; Hofmann, K.; Bodmer, J.L.; Holler, N.; Ambrose, C.; Lawton, P.; Bixler, S.; Acha-Orbea, H.; et al. BAFF, a novel ligand of the tumor necrosis factor family, stimulates B cell growth. J. Exp. Med. 1999, 189, 1747–1756. [Google Scholar] [CrossRef]
- Lucas, J.J.; Szepesi, A.; Domenico, J.; Tordai, A.; Terada, N.; Gelfand, E.W. Differential regulation of the synthesis and activity of the major cyclin-dependent kinases, p34cdc2, p33cdk2, and p34cdk4, during cell cycle entry and progression in normal human T lymphocytes. J. Cell. Physiol. 1995, 165, 406–416. [Google Scholar] [CrossRef]
- Modiano, J.F.; Domenico, J.; Szepesi, A.; Lucas, J.J.; Gelfand, E.W. Differential requirements for interleukin-2 distinguish the expression and activity of the cyclin-dependent kinases Cdk4 and Cdk2 in human T cells. J. Biol. Chem. 1994, 269, 32972–32978. [Google Scholar] [CrossRef]
- Modiano, J.F.; Mayor, J.; Ball, C.; Fuentes, M.K.; Linthicum, D.S. CDK4 expression and activity are required for cytokine responsiveness in T cells. J. Immunol. 2000, 165, 6693–6702. [Google Scholar] [CrossRef]
- Brorson, K.; Brunswick, M.; Ezhevsky, S.; Wei, D.G.; Berg, R.; Scott, D.; Stein, K.E. xid affects events leading to B cell cycle entry. J. Immunol. 1997, 159, 135–143. [Google Scholar] [CrossRef]
- Solvason, N.; Wu, W.W.; Kabra, N.; Wu, X.; Lees, E.; Howard, M.C. Induction of cell cycle regulatory proteins in anti-immunoglobulin-stimulated mature B lymphocytes. J. Exp. Med. 1996, 184, 407–417. [Google Scholar] [CrossRef]
- Tanguay, D.A.; Colarusso, T.P.; Doughty, C.; Pavlovic-Ewers, S.; Rothstein, T.L.; Chiles, T.C. Cutting edge: Differential signaling requirements for activation of assembled cyclin D3-cdk4 complexes in B-1 and B-2 lymphocyte subsets. J. Immunol. 2001, 166, 4273–4277. [Google Scholar] [CrossRef]
- Rane, S.G.; Dubus, P.; Mettus, R.V.; Galbreath, E.J.; Boden, G.; Reddy, E.P.; Barbacid, M. Loss of Cdk4 expression causes insulin-deficient diabetes and Cdk4 activation results in beta-islet cell hyperplasia. Nat. Genet. 1999, 22, 44–52. [Google Scholar] [CrossRef] [PubMed]
- Tsutsui, T.; Hesabi, B.; Moons, D.S.; Pandolfi, P.P.; Hansel, K.S.; Koff, A.; Kiyokawa, H. Targeted disruption of CDK4 delays cell cycle entry with enhanced p27(Kip1) activity. Mol. Cell. Biol. 1999, 19, 7011–7019. [Google Scholar] [CrossRef] [PubMed]
- Jirawatnotai, S.; Aziyu, A.; Osmundson, E.C.; Moons, D.S.; Zou, X.; Kineman, R.D.; Kiyokawa, H. Cdk4 is indispensable for postnatal proliferation of the anterior pituitary. J. Biol. Chem. 2004, 279, 51100–51106. [Google Scholar] [CrossRef] [PubMed]
- Moons, D.S.; Jirawatnotai, S.; Parlow, A.F.; Gibori, G.; Kineman, R.D.; Kiyokawa, H. Pituitary hypoplasia and lactotroph dysfunction in mice deficient for cyclin-dependent kinase-4. Endocrinology 2002, 143, 3001–3008. [Google Scholar] [CrossRef]
- Chow, Y.H.; Zhu, X.D.; Liu, L.; Schwartz, B.R.; Huang, X.Z.; Harlan, J.M.; Schnapp, L.M. Role of Cdk4 in lymphocyte function and allergen response. Cell Cycle 2010, 9, 4922–4930. [Google Scholar] [CrossRef] [PubMed]
- Hoyne, G.F.; Goodnow, C.C. The use of genomewide ENU mutagenesis screens to unravel complex mammalian traits: Identifying genes that regulate organ-specific and systemic autoimmunity. Immunol. Rev. 2006, 210, 27–39. [Google Scholar] [CrossRef]
- Nelms, K.A.; Goodnow, C.C. Genome-wide ENU mutagenesis to reveal immune regulators. Immunity 2001, 15, 409–418. [Google Scholar] [CrossRef]
- Hoyne, G.F.; Flening, E.; Yabas, M.; Teh, C.; Altin, J.A.; Randall, K.; Thien, C.B.; Langdon, W.Y.; Goodnow, C.C. Visualizing the role of Cbl-b in control of islet-reactive CD4 T cells and susceptibility to type 1 diabetes. J. Immunol. 2011, 186, 2024–2032. [Google Scholar] [CrossRef]
- Ciofani, M.; Zuniga-Pflucker, J.C. The thymus as an inductive site for T lymphopoiesis. Annu. Rev. Cell Dev. Biol. 2007, 23, 463–493. [Google Scholar] [CrossRef]
- Hayday, A.C.; Pennington, D.J. Key factors in the organized chaos of early T cell development. Nat. Immunol. 2007, 8, 137–144. [Google Scholar] [CrossRef]
- Hayday, A.C. Gammadelta T cells and the lymphoid stress-surveillance response. Immunity 2009, 31, 184–196. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, S. Naturally arising CD4+ regulatory t cells for immunologic self-tolerance and negative control of immune responses. Annu. Rev. Immunol. 2004, 22, 531–562. [Google Scholar] [CrossRef] [PubMed]
- Carman, J.A.; Wechsler-Reya, R.J.; Monroe, J.G. Immature stage B cells enter but do not progress beyond the early G1 phase of the cell cycle in response to antigen receptor signaling. J. Immunol. 1996, 156, 4562–4569. [Google Scholar] [CrossRef] [PubMed]
- Tanguay, D.A.; Chiles, T.C. Regulation of the catalytic subunit (p34PSK-J3/cdk4) for the major D-type cyclin in mature B lymphocytes. J. Immunol. 1996, 156, 539–548. [Google Scholar] [CrossRef]
- Berthet, C.; Rodriguez-Galan, M.C.; Hodge, D.L.; Gooya, J.; Pascal, V.; Young, H.A.; Keller, J.; Bosselut, R.; Kaldis, P. Hematopoiesis and thymic apoptosis are not affected by the loss of Cdk2. Mol. Cell. Biol. 2007, 27, 5079–5089. [Google Scholar] [CrossRef]
- Hu, M.G.; Deshpande, A.; Enos, M.; Mao, D.; Hinds, E.A.; Hu, G.F.; Chang, R.; Guo, Z.; Dose, M.; Mao, C.; et al. A requirement for cyclin-dependent kinase 6 in thymocyte development and tumorigenesis. Cancer Res. 2009, 69, 810–818. [Google Scholar] [CrossRef]
- Liao, W.; Lin, J.X.; Leonard, W.J. IL-2 family cytokines: New insights into the complex roles of IL-2 as a broad regulator of T helper cell differentiation. Curr. Opin. Immunol. 2011, 23, 598–604. [Google Scholar] [CrossRef]
- Brocker, T. Survival of mature CD4 T lymphocytes is dependent on major histocompatibility complex class II-expressing dendritic cells. J. Exp. Med. 1997, 186, 1223–1232. [Google Scholar] [CrossRef]
- Ernst, B.; Lee, D.S.; Chang, J.M.; Sprent, J.; Surh, C.D. The peptide ligands mediating positive selection in the thymus control T cell survival and homeostatic proliferation in the periphery. Immunity 1999, 11, 173–181. [Google Scholar] [CrossRef]
- Goldrath, A.W.; Bevan, M.J. Low-affinity ligands for the TCR drive proliferation of mature CD8+ T cells in lymphopenic hosts. Immunity 1999, 11, 183–190. [Google Scholar] [CrossRef]
- Kieper, W.C.; Jameson, S.C. Homeostatic expansion and phenotypic conversion of naïve T cells in response to self peptide/MHC ligands. Proc. Natl. Acad. Sci. USA 1999, 96, 13306–13311. [Google Scholar] [CrossRef]
- Tan, J.T.; Dudl, E.; LeRoy, E.; Murray, R.; Sprent, J.; Weinberg, K.I.; Surh, C.D. IL-7 is critical for homeostatic proliferation and survival of naive T cells. Proc. Natl. Acad. Sci. USA 2001, 98, 8732–8737. [Google Scholar] [CrossRef] [PubMed]
- Kündig, T.M.; Schorle, H.; Bachmann, M.F.; Hengartner, H.; Zinkernagel, R.M.; Horak, I. Immune responses in interleukin-2-deficient mice. Science 1993, 262, 1059–1061. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, H.; Duncan, G.S.; Takimoto, H.; Mak, T.W. Abnormal development of intestinal intraepithelial lymphocytes and peripheral natural killer cells in mice lacking the IL-2 receptor beta chain. J. Exp. Med. 1997, 185, 499–505. [Google Scholar] [CrossRef]
- Bhandoola, A.; von Boehmer, H.; Petrie, H.T.; Zuniga-Pflucker, J.C. Commitment and developmental potential of extrathymic and intrathymic T cell precursors: Plenty to choose from. Immunity 2007, 26, 678–689. [Google Scholar] [CrossRef] [PubMed]
- Di Santo, J.P.; Radtke, F.; Rodewald, H.R. To be or not to be a pro-T? Curr. Opin. Immunol. 2000, 12, 159–165. [Google Scholar] [CrossRef]
- Yasunaga, M.; Wang, F.; Kunisada, T.; Nishikawa, S.; Nishikawa, S. Cell cycle control of c-kit+IL-7R+ B precursor cells by two distinct signals derived from IL-7 receptor and c-kit in a fully defined medium. J. Exp. Med. 1995, 182, 315–323. [Google Scholar] [CrossRef]
- Fehling, H.J.; Krotkova, A.; Saint-Ruf, C.; von Boehmer, H. Crucial role of the pre-T-cell receptor alpha gene in development of alpha beta but not gamma delta T cells. Nature 1995, 375, 795–798. [Google Scholar] [CrossRef]
- Hoffman, E.S.; Passoni, L.; Crompton, T.; Leu, T.M.; Schatz, D.G.; Koff, A.; Owen, M.J.; Hayday, A.C. Productive T-cell receptor beta-chain gene rearrangement: Coincident regulation of cell cycle and clonality during development in vivo. Genes Dev. 1996, 10, 948–962. [Google Scholar] [CrossRef]
- Bruno, L.; von Boehmer, H.; Kirberg, J. Cell division in the compartment of naive and memory T lymphocytes. Eur. J. Immunol. 1996, 26, 3179–3184. [Google Scholar] [CrossRef] [PubMed]
- Tough, D.F.; Sprent, J. Turnover of naive- and memory-phenotype T cells. J. Exp. Med. 1994, 179, 1127–1135. [Google Scholar] [CrossRef] [PubMed]
- Zimmerman, C.; Brduscha-Riem, K.; Blaser, C.; Zinkernagel, R.M.; Pircher, H. Visualization, characterization, and turnover of CD8+ memory T cells in virus-infected hosts. J. Exp. Med. 1996, 183, 1367–1375. [Google Scholar] [CrossRef] [PubMed]
- Miller, J.P.; Izon, D.; DeMuth, W.; Gerstein, R.; Bhandoola, A.; Allman, D. The earliest step in B lineage differentiation from common lymphoid progenitors is critically dependent upon interleukin 7. J. Exp. Med. 2002, 196, 705–711. [Google Scholar] [CrossRef] [PubMed]




Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yabas, M.; Hoyne, G.F. Immunological Phenotyping of Mice with a Point Mutation in Cdk4. Biomedicines 2023, 11, 2847. https://doi.org/10.3390/biomedicines11102847
Yabas M, Hoyne GF. Immunological Phenotyping of Mice with a Point Mutation in Cdk4. Biomedicines. 2023; 11(10):2847. https://doi.org/10.3390/biomedicines11102847
Chicago/Turabian StyleYabas, Mehmet, and Gerard F. Hoyne. 2023. "Immunological Phenotyping of Mice with a Point Mutation in Cdk4" Biomedicines 11, no. 10: 2847. https://doi.org/10.3390/biomedicines11102847
APA StyleYabas, M., & Hoyne, G. F. (2023). Immunological Phenotyping of Mice with a Point Mutation in Cdk4. Biomedicines, 11(10), 2847. https://doi.org/10.3390/biomedicines11102847





