Clinicopathological Significance of Cancer Stem Cell Markers (OCT-3/4 and SOX-2) in Oral Submucous Fibrosis and Oral Squamous Cell Carcinoma
Abstract
:1. Introduction
2. Materials and Methods
2.1. Institutional Ethics Approval
2.2. Sample Collection
2.3. Immunohistochemistry
2.4. Evaluation of Immunoexpression
2.5. Homogenization of Tissue Samples
2.6. RNA Isolation from the Tissues
2.7. cDNA Conversion
2.8. Real-Time PCR
2.9. Statistical Analysis
3. Results
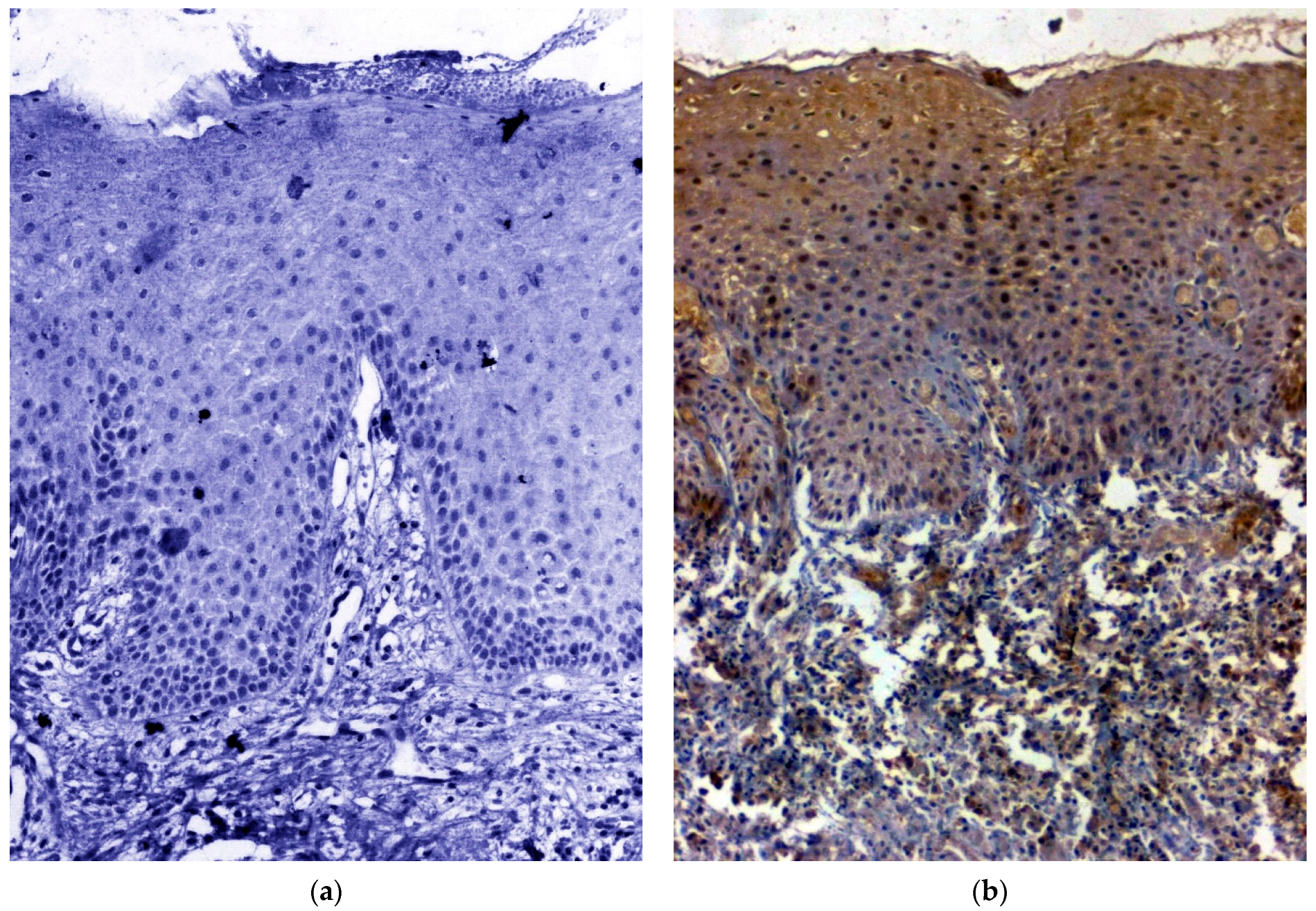
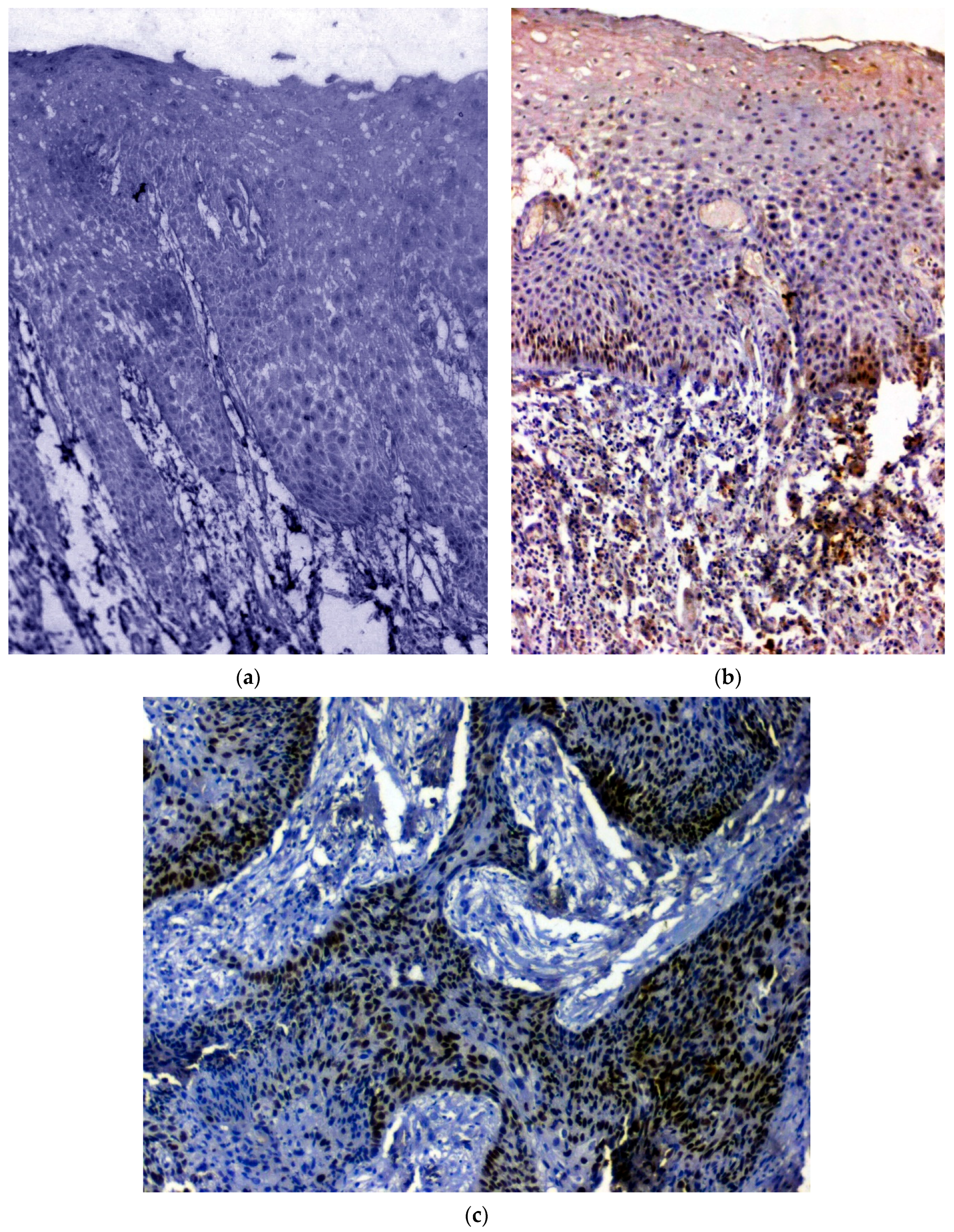
3.1. IHC Using Clinical Samples for OCT 3/4
3.2. IHC Using Clinical Samples for SOX 2
3.3. Real-Time PCR
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Ekanayaka, R.P.; Tilakaratne, W.M. Oral Submucous Fibrosis: Review on Mechanisms of Malignant Transformation. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2016, 122, 192–199. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Tail, Y.-H.; Wang, W.-C.; Chen, C.-Y.; Kao, Y.-H.; Chen, Y.-K.; Chen, C.-H. Malignant Transformation in 5071 Southern Taiwanese Patients with Potentially Malignant Oral Mucosal Disorders. BMC Oral Health 2014, 14, 99. [Google Scholar] [CrossRef] [PubMed]
- Hu, Y.; Jian, X.; Peng, J.; Jiang, X.; Li, N.; Zhou, S. Gene Expression Profiling of Oral Submucous Fibrosis Using Oligonucleotide Microarray. Oncol. Rep. 2008, 20, 287–294. [Google Scholar] [PubMed] [Green Version]
- Tilakaratne, W.M.; Iqbal, Z.; Teh, M.T.; Ariyawardana, A.; Pitiyage, G.; Cruchley, A.; Stewart, J.E.; Hagi-Pavli, E.; Lalli, A.; Waseem, A.; et al. Upregulation of HIF-1alpha in Malignant Transformation of Oral Submucous Fibrosis. J. Oral Pathol. Med. 2008, 37, 372–377. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.-C.; Cheng, A.-J.; Lee, L.-Y.; Huang, Y.-C.; Chang, J.T.-C. Multifaceted Mechanisms of Areca Nuts in Oral Carcinogenesis: The Molecular Pathology from Precancerous Condition to Malignant Transformation. J. Cancer 2019, 10, 4054. [Google Scholar] [CrossRef] [Green Version]
- Sadeghi, H.; Saffar, H.; Taheri, P.; Yazdani, F.; Etebarian, A. Prognostic Significance of Cancer Stem Cell Markers in Patients With Salivary Gland Carcinomas. Appl. Immunohistochem. Mol. Morphol. 2022, 30, 284–290. [Google Scholar] [CrossRef]
- Razmi, M.; Ghods, R.; Vafaei, S.; Sahlolbei, M.; Zanjani, L.S.; Madjd, Z. Clinical and Prognostic Significances of Cancer Stem Cell Markers in Gastric Cancer Patients: A Systematic Review and Meta-Analysis. Cancer Cell Int. 2021, 21, 139. [Google Scholar] [CrossRef]
- Major, A.G.; Pitty, L.P.; Farah, C.S. Cancer Stem Cell Markers in Head and Neck Squamous Cell Carcinoma. Stem Cells Int. 2013, 2013, 319489. [Google Scholar] [CrossRef] [Green Version]
- Vaiphei, K.; Sinha, S.K.; Kochhar, R. Comparative Analysis of Oct4 in Different Histological Subtypes of Esophageal Squamous Cell Carcinomas in Different Clinical Conditions. Asia Pac. J. Cancer Prev. 2014, 15, 3519–3524. [Google Scholar] [CrossRef] [Green Version]
- Tsai, L.-L.; Hu, F.-W.; Lee, S.-S.; Yu, C.-H.; Yu, C.-C.; Chang, Y.-C. Oct4 Mediates Tumor Initiating Properties in Oral Squamous Cell Carcinomas through the Regulation of Epithelial-Mesenchymal Transition. PLoS ONE 2014, 9, e87207. [Google Scholar] [CrossRef]
- Fu, T.-Y.; Hsieh, I.-C.; Cheng, J.-T.; Tsai, M.-H.; Hou, Y.-Y.; Lee, J.-H.; Liou, H.-H.; Huang, S.-F.; Chen, H.-C.; Yen, L.-M.; et al. Association of OCT4, SOX2, and NANOG Expression with Oral Squamous Cell Carcinoma Progression. J. Oral Pathol. Med. 2016, 45, 89–95. [Google Scholar] [CrossRef]
- Sharma, M.; Fonseca, F.P.; Hunter, K.D.; Radhakrishnan, R. Loss of Oral Mucosal Stem Cell Markers in Oral Submucous Fibrosis and Their Reactivation in Malignant Transformation. Int. J. Oral. Sci. 2020, 12, 23. [Google Scholar] [CrossRef]
- Khanna, J.N.; Andrade, N.N. Oral Submucous Fibrosis: A New Concept in Surgical Management. Report of 100 Cases. Int. J. Oral Maxillofac. Surg. 1995, 24, 433–439. [Google Scholar] [CrossRef] [PubMed]
- Akhter, M.; Hossain, S.; Rahman, Q.B.; Molla, M.R. A Study on Histological Grading of Oral Squamous Cell Carcinoma and Its Co-Relationship with Regional Metastasis. J. Oral Maxillofac. Pathol. 2011, 15, 168–176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Raungrut, P.; Petjaroen, P.; Geater, S.L.; Keeratichananont, W.; Phukaoloun, M.; Suwiwat, S.; Thongsuksai, P. Methylation of 14-3-3σ Gene and Prognostic Significance of 14-3-3σ Expression in Non-Small Cell Lung Cancer. Oncol. Lett. 2017, 14, 5257–5264. [Google Scholar] [CrossRef] [Green Version]
- Tavassol, F.; Starke, O.F.; Kokemüller, H.; Wegener, G.; Müller-Tavassol, C.C.M.; Gellrich, N.-C.; Eckardt, A. Prognostic Significance of Heat Shock Protein 70 (HSP70) in Patients with Oral Cancer. Head Neck Oncol. 2011, 3, 10. [Google Scholar] [CrossRef] [Green Version]
- Venugopal, D.C.; Ravindran, S.; Shyamsundar, V.; Sankarapandian, S.; Krishnamurthy, A.; Sivagnanam, A.; Madhavan, Y.; Ramshankar, V. Integrated Proteomics Based on 2D Gel Electrophoresis and Mass Spectrometry with Validations: Identification of a Biomarker Compendium for Oral Submucous Fibrosis—An Indian Study. J. Pers. Med. 2022, 12, 208. [Google Scholar] [CrossRef]
- Vidhale, R.G.; Pereira, T.; Lalai, M.N.; Anjali, A.K.; Jain, A.; Pereira, C. Qualitative Expression of Sox2 and Oct4 in Oral Submucous Fibrosis: An Immunohistochemical Study. J. Pharm. Res. Int. 2022, 34, 34–42. [Google Scholar] [CrossRef]
- Ren, Z.-H.; Zhang, C.-P.; Ji, T. Expression of SOX2 in Oral Squamous Cell Carcinoma and the Association with Lymph Node Metastasis. Oncol. Lett. 2016, 11, 1973–1979. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Smith, B.; Li, N.; Andersen, A.S.; Slotved, H.C.; Krogfelt, K.A. Optimising Bacterial DNA Extraction from Faecal Samples: Comparison of Three Methods. Open Microbiol. J. 2011, 5, 14–17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- RNA Extraction. Available online: https://www.usbio.net/protocols/rna-extraction (accessed on 10 June 2022).
- Picard-Meyer, E.; Peytavin de Garam, C.; Schereffer, J.L.; Marchal, C.; Robardet, E.; Cliquet, F. Cross-Platform Evaluation of Commercial Real-Time SYBR Green RT-PCR Kits for Sensitive and Rapid Detection of European Bat Lyssavirus Type 1. BioMed Res. Int. 2015, 2015, 839518. [Google Scholar] [CrossRef] [Green Version]
- Yip, C.C.-Y.; Ho, C.-C.; Chan, J.F.-W.; To, K.K.-W.; Chan, H.S.-Y.; Wong, S.C.-Y.; Leung, K.-H.; Fung, A.Y.-F.; Ng, A.C.-K.; Zou, Z.; et al. Development of a Novel, Genome Subtraction-Derived, SARS-CoV-2-Specific COVID-19-Nsp2 Real-Time RT-PCR Assay and Its Evaluation Using Clinical Specimens. Int. J. Mol. Sci. 2020, 21, E2574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- How to Cite IBM SPSS Statistics or Earlier Versions of SPSS. Available online: https://www.ibm.com/support/pages/how-cite-ibm-spss-statistics-or-earlier-versions-spss (accessed on 10 June 2022).
- Chen, C.; Méndez, E.; Houck, J.; Fan, W.; Lohavanichbutr, P.; Doody, D.; Yueh, B.; Futran, N.D.; Upton, M.; Farwell, D.G.; et al. Gene Expression Profiling Identifies Genes Predictive of Oral Squamous Cell Carcinoma. Cancer Epidemiol. Biomark. Prev. 2008, 17, 2152–2162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, S.; Zhu, Y.; He, Z.; Zhang, D.; Guo, F.; Jian, X.; Zhang, C. Long Non-Coding RNA Expression Profile Associated with Malignant Progression of Oral Submucous Fibrosis. J. Oncol. 2019, 2019, 6835176. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baek, K.-H.; Choi, J.; Pei, C.-Z. Cellular Functions of OCT-3/4 Regulated by Ubiquitination in Proliferating Cells. Cancers 2020, 12, 663. [Google Scholar] [CrossRef] [Green Version]
- Liu, K.; Xie, F.; Zhao, T.; Zhang, R.; Gao, A.; Chen, Y.; Li, H.; Zhang, S.; Xiao, Z.; Li, J.; et al. Targeting SOX2 Protein with Peptide Aptamers for Therapeutic Gains against Esophageal Squamous Cell Carcinoma. Mol. Ther. 2020, 28, 901–913. [Google Scholar] [CrossRef] [PubMed]
- Mithani, S.K.; Mydlarz, W.K.; Grumbine, F.L.; Smith, I.M.; Califano, J.A. Molecular Genetics of Premalignant Oral Lesions. Oral Dis. 2007, 13, 126–133. [Google Scholar] [CrossRef]
- Habu, N.; Imanishi, Y.; Kameyama, K.; Shimoda, M.; Tokumaru, Y.; Sakamoto, K.; Fujii, R.; Shigetomi, S.; Otsuka, K.; Sato, Y.; et al. Expression of Oct3/4 and Nanog in the Head and Neck Squamous Carcinoma Cells and Its Clinical Implications for Delayed Neck Metastasis in Stage I/II Oral Tongue Squamous Cell Carcinoma. BMC Cancer 2015, 15, 730. [Google Scholar] [CrossRef] [Green Version]
- de Vicente, J.C.; Donate-Pérez Del Molino, P.; Rodrigo, J.P.; Allonca, E.; Hermida-Prado, F.; Granda-Díaz, R.; Rodríguez Santamarta, T.; García-Pedrero, J.M. SOX2 Expression Is an Independent Predictor of Oral Cancer Progression. J. Clin. Med. 2019, 8, E1744. [Google Scholar] [CrossRef] [Green Version]
- Anura, A.; Kazi, A.; Pal, M.; Paul, R.R.; Sengupta, S.; Chatterjee, J. Endorsing Cellular Competitiveness in Aberrant Epithelium of Oral Submucous Fibrosis Progression: Neighbourhood Analysis of Immunohistochemical Attributes. Histochem. Cell Biol. 2018, 150, 61–75. [Google Scholar] [CrossRef]
- Sachdeva, M.; Zhu, S.; Wu, F.; Wu, H.; Walia, V.; Kumar, S.; Elble, R.; Watabe, K.; Mo, Y.-Y. P53 Represses C-Myc through Induction of the Tumor Suppressor MiR-145. Proc. Natl. Acad. Sci. USA 2009, 106, 3207–3212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, T.Y.; Peng, C.-Y.; Lee, S.-S.; Chou, M.-Y.; Yu, C.-C.; Chang, Y.-C. Acquisition Cancer Stemness, Mesenchymal Transdifferentiation, and Chemoresistance Properties by Chronic Exposure of Oral Epithelial Cells to Arecoline. Oncotarget 2016, 7, 84072–84081. [Google Scholar] [CrossRef]
- Abeyasinghe, W.; Tennakoon, P.; Jayasooriya, P. Evaluation of cancer stem cell marker, BMI-1 expression in oral submucous fibrosis with and without malignant transformation. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2021, 132, e1–e2. [Google Scholar] [CrossRef]
- Xie, C.; Feng, H.; Zhong, L.; Shi, Y.; Wei, Z.; Hua, Y.; Ji, N.; Li, J.; Tang, Z.; Chen, Q. Proliferative Ability and Accumulation of Cancer Stem Cells in Oral Submucous Fibrosis Epithelium. Oral Dis. 2020, 26, 1255–1264. [Google Scholar] [CrossRef]
- Čēma, I.; Dzudzilo, M.; Kleina, R.; Franckevica, I.; Svirskis, Š. Correlation of Soluble CD44 Expression in Saliva and CD44 Protein in Oral Leukoplakia Tissues. Cancers 2021, 13, 5739. [Google Scholar] [CrossRef] [PubMed]



| Gene Name | Forward Sequence | Reverse Sequence |
|---|---|---|
| OCT 3/4 | 5′-GACAGGGGGAGGGGAGGAGCTAGG-3′ | 5′-CTTCCCTCCAACCAGTTGCCCCAAAC-3′ |
| SOX 2 | 5′-GGGAAATGGGAGGGGTGCAAAAGAGG-3′ | 5′-TTGCGTGAGTGTGGATGGGATTGGTG-3′ |
| Criteria | Total (n = 115) | OCT ¾ Negative (n = 45) | OCT ¾ Positive (n = 70) |
|---|---|---|---|
| Gender | |||
| Male | 83 | 32 (36.8%) | 51 (58.6%) |
| Female | 32 | 13 (39.4%) | 19 (57.6%) |
| Age | |||
| >47 years | 60 | 15 (25%) | 45 (75%) |
| <47 years | 55 | 30 (54.5%) | 25 (45.5%) |
| p value = 0.001; χ2 = 10.517 | |||
| Diagnosis | |||
| Healthy control | 17 | 15 (88.2%) | 2 (11.8%) |
| OSMF | 44 | 14 (31.8%) | 30 (68.2%) |
| OSCC | 54 | 16 (29.6%) | 38 (70.4%) |
| p value = 0.000; χ2 = 20.244 | |||
| Epithelial Nature | |||
| Healthy control | 9 | 9(100%) | 0 (0%) |
| Hypertrophic | 16 | 8 (50%) | 8 (50%) |
| Atrophic | 33 | 12(36.4%) | 21 (63.6%) |
| Dysplasia | 3 | 0 (0%) | 3 (100%) |
| OSCC | 54 | 16 (29.6%) | 38 (70.4%) |
| p value = 0.001; χ2 = 18.875 | |||
| Habits (OSMF n = 44) | |||
| Pan | 27 | 9 (33.3%) | 18 (66.7%) |
| Betel Nut | 6 | 3 (50%) | 3 (50%) |
| Maava | 9 | 2 (22.2%) | 7 (77.8%) |
| Gutka | 2 | 0 (0%) | 2 (100%) |
| Inflammation (OSMF n = 44) | |||
| Mild | 15 | 5 (33.3%) | 10 (66.7%) |
| Moderate | 15 | 4 (26.7%) | 11 (73.3%) |
| Severe | 14 | 5 (35.7%) | 9 (64.3%) |
| Fibrosis (OSMF n = 44) | |||
| Mild | 10 | 1 (10%) | 9 (90%) |
| Moderate | 17 | 6 (35.3%) | 11 (64.7%) |
| Severe | 17 | 7 (41.2%) | 10 (58.8%) |
| Vascularity (OSMF n = 44) | |||
| Healthy Control | 7 | 1 (14.3%) | 6 (85.7%) |
| Reduced | 22 | 8 (36.4%) | 14 (63.6%) |
| Enlarged and increased | 15 | 5 (33.3%) | 10(66.7%) |
| OSMF Clinical Stage (OSMF n = 44) | |||
| Stage I | 8 | 3 (37.5%) | 5 (62.5%) |
| Stage II | 18 | 8 (44.4%) | 10 (55.6%) |
| Stage III | 15 | 2 (13.3%) | 13 (86.7%) |
| Stage IV | 3 | 1(33.3%) | 2(66.7%) |
| OSCC Histological Stage (OSCC n = 54) | |||
| WDSCC | 27 | 9 (33.3%) | 18 (66.7%) |
| MDSCC | 24 | 5 (20.8%) | 19 (79.2%) |
| PDSCC | 3 | 2 (66.7%) | 1 (33.3%) |
| OSCC Clinical Stage (OSCC n = 54) | |||
| Stage I | 8 | 3 (37.5%) | 5 (62.5%) |
| Stage II | 16 | 5 (31.2%) | 11 (68.8%) |
| Stage III | 15 | 6 (40.0%) | 9 (60.0%) |
| Stage IV | 15 | 2 (13.3%) | 13 (86.7%) |
| Criteria | Total (n = 113) | SOX 2 Negative (n = 69) | SOX 2 Positive (n = 44) |
|---|---|---|---|
| Gender | |||
| Male | 81 | 49 (60.5%) | 32 (39.5%) |
| Female | 32 | 20 (62.5%) | 12 (37.5%) |
| Age | |||
| >47 years | 60 | 26 (43.3%) | 34 (56.7%) |
| <47 years | 53 | 43 (81.1%) | 10 (18.9%) |
| p value = 0.000; χ2 = 16.911 | |||
| Diagnosis | |||
| Healthy Control | 17 | 16 (94.1%) | 1 (5.9%) |
| OSMF | 43 | 26 (60.5%) | 17 (39.5%) |
| OSCC | 53 | 27 (50.9%) | 26 (49.1%) |
| p value = 0.006; χ2 = 10.101 | |||
| Epithelial Nature | |||
| Healthy Control | 9 | 8 (88.9%) | 1 (11.1%) |
| Hypertrophic | 16 | 27 (87.5%) | 2 (12.5%) |
| Atrophic | 32 | 19(59.4%) | 13 (40.6%) |
| Dysplasia | 3 | 1 (33.3%) | 2 (66.7%) |
| OSCC | 53 | 27 (50.9%) | 26 (49.1%) |
| p value = 0.027; χ2 = 10.925 | |||
| Habits (OSMF n = 43) | |||
| Pan | 27 | 19 (70.4%) | 8 (29.6%) |
| Betel Nut | 6 | 3 (50.0%) | 3 (50.0%) |
| Maava | 9 | 4 (44.4%) | 5 (55.6%) |
| Gutka | 1 | 0 (0%) | 1 (100%) |
| Inflammation (OSMF n = 43) | |||
| Mild | 14 | 8 (57.1%) | 6 (42.9%) |
| Moderate | 15 | 10 (66.7%) | 5 (33.3%) |
| Severe | 14 | 8 (57.1%) | 6 (42.9%) |
| Fibrosis (OSMF n = 43) | |||
| Mild | 9 | 6 (66.7%) | 3 (33.3%) |
| Moderate | 17 | 11 (64.7%) | 6 (35.3%) |
| Severe | 17 | 9 (52.9%) | 8 (47.1%) |
| Vascularity (OSMF n = 43) | |||
| Healthy Control | 6 | 4 (66.7%) | 2 (33.3%) |
| Reduced | 22 | 13 (59.1%) | 9 (40.9%) |
| Enlarged and increased | 15 | 9 (60.0%) | 6(40.0%) |
| OSMF Clinical Stage (OSMF n = 43) | |||
| Stage I | 7 | 3 (42.9%) | 4 (57.1%) |
| Stage II | 18 | 14 (77.8%) | 4 (22.2%) |
| Stage III | 15 | 8 (53.3%) | 7 (46.7%) |
| Stage IV | 3 | 1(33.3%) | 2(66.7%) |
| OSCC Histological Stage (OSCC n = 53) | |||
| WDSCC | 27 | 15 (55.6%) | 12 (44.4%) |
| MDSCC | 24 | 10 (41.7%) | 14 (58.3%) |
| PDSCC | 2 | 2 (100%) | 0 (0%) |
| OSCC Clinical Stage (OSCC n = 53) | |||
| Stage I | 8 | 5 (62.5%) | 3 (37.5%) |
| Stage II | 15 | 5 (33.3%) | 10 (66.7%) |
| Stage III | 15 | 7 (46.7%) | 8 (53.3%) |
| Stage IV | 15 | 10 (66.7%) | 5 (33.3%) |
| Patient Sample Type | Log 2 RQ |
|---|---|
| Healthy Control | 0.004444 |
| OSCC | −0.2368 |
| OSMF | 3.721 |
| Patient Sample Type | Log 2 RQ |
|---|---|
| Healthy Control | 1.18 × 10−15 |
| OSCC | 0.6524 |
| OSMF | 3.34 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Venugopal, D.C.; Caleb, C.L.; Kirupakaran, N.P.; Shyamsundar, V.; Ravindran, S.; Yasasve, M.; Krishnamurthy, A.; Harikrishnan, T.; Sankarapandian, S.; Ramshankar, V. Clinicopathological Significance of Cancer Stem Cell Markers (OCT-3/4 and SOX-2) in Oral Submucous Fibrosis and Oral Squamous Cell Carcinoma. Biomedicines 2023, 11, 1040. https://doi.org/10.3390/biomedicines11041040
Venugopal DC, Caleb CL, Kirupakaran NP, Shyamsundar V, Ravindran S, Yasasve M, Krishnamurthy A, Harikrishnan T, Sankarapandian S, Ramshankar V. Clinicopathological Significance of Cancer Stem Cell Markers (OCT-3/4 and SOX-2) in Oral Submucous Fibrosis and Oral Squamous Cell Carcinoma. Biomedicines. 2023; 11(4):1040. https://doi.org/10.3390/biomedicines11041040
Chicago/Turabian StyleVenugopal, Divyambika Catakapatri, Cynthia Leslie Caleb, Nandhini Priyadarshini Kirupakaran, Vidyarani Shyamsundar, Soundharya Ravindran, Madhavan Yasasve, Arvind Krishnamurthy, Thamizhchelvan Harikrishnan, Sathasivasubramanian Sankarapandian, and Vijayalakshmi Ramshankar. 2023. "Clinicopathological Significance of Cancer Stem Cell Markers (OCT-3/4 and SOX-2) in Oral Submucous Fibrosis and Oral Squamous Cell Carcinoma" Biomedicines 11, no. 4: 1040. https://doi.org/10.3390/biomedicines11041040
APA StyleVenugopal, D. C., Caleb, C. L., Kirupakaran, N. P., Shyamsundar, V., Ravindran, S., Yasasve, M., Krishnamurthy, A., Harikrishnan, T., Sankarapandian, S., & Ramshankar, V. (2023). Clinicopathological Significance of Cancer Stem Cell Markers (OCT-3/4 and SOX-2) in Oral Submucous Fibrosis and Oral Squamous Cell Carcinoma. Biomedicines, 11(4), 1040. https://doi.org/10.3390/biomedicines11041040







