2,3,5,4′-Tetrahydroxystilbene (TG1), a Novel Compound Derived from 2,3,5,4′-Tetrahydroxystilbene-2-O-β-D-glucoside (THSG), Inhibits Colorectal Cancer Progression by Inducing Ferroptosis, Apoptosis, and Autophagy
Abstract
:1. Introduction
2. Materials and Methods
2.1. Production of TG1 (2,3,5,4′-Tetrahydroxystilbene)
2.2. Chemicals, Reagents, and Cell Culture
2.3. Examination of Cell Viability
2.4. Annexin V-FITC/Propidium Iodide (PI) Assay
2.5. Animal Model
2.6. Autophagy Assay by the FlexiCyte Protocol
2.7. Reverse Transcription Polymerase Chain Reaction (RT-PCR) and Quantitative RT-PCR Analysis
2.8. Protein Extraction and Western Blot Analysis
2.9. RNA-Sequencing Analysis
2.10. Statistical Analysis
3. Results
3.1. TG1 Possesses Dose-Dependent Cytotoxicity on Colorectal Cancer Cells
3.2. TG1 Treatment Suppresses CRC Progression in a Xenograft Mouse Model
3.3. CRC Cells Apoptosis Is Induced by TG1
3.4. TG1 Treatment Causes an Induction of Autophagy Process
3.5. Analysis of Gene Expression Changes in TG1 Treatment
3.6. TG1 Treatment Regulates Expression of Ferroptosis-Related Genes
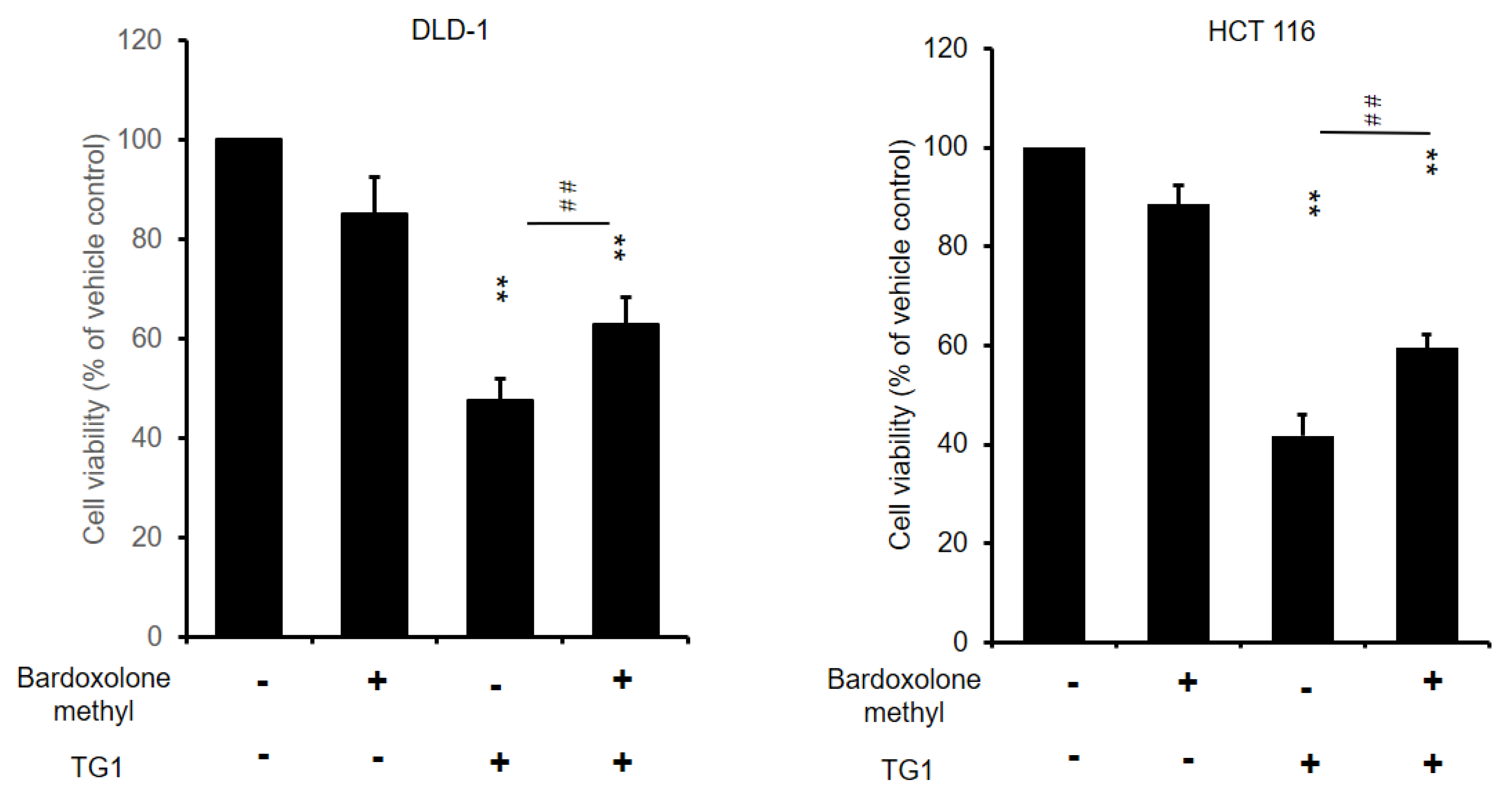
3.7. Prevention of TG1-Induced Cancer Cell Death by Inhibiting Ferroptosis
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Emons, G.; Spitzner, M.; Reineke, S.; Möller, J.; Auslander, N.; Kramer, F.; Hu, Y.; Beissbarth, T.; Wolff, H.A.; Rave-Fränk, M. Chemoradiotherapy resistance in colorectal cancer cells is mediated by Wnt/β-catenin signaling. Mol. Cancer Res. 2017, 15, 1481–1490. [Google Scholar] [PubMed] [Green Version]
- Arnold, M.; Sierra, M.S.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global patterns and trends in colorectal cancer incidence and mortality. Gut 2017, 66, 683–691. [Google Scholar] [PubMed] [Green Version]
- Siegel, R.; DeSantis, C.; Virgo, K.; Stein, K.; Mariotto, A.; Smith, T.; Cooper, D.; Gansler, T.; Lerro, C.; Fedewa, S.; et al. Cancer treatment and survivorship statistics, 2012. CA Cancer J. Clin. 2012, 62, 220–241. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sankaranarayanan, R.; Swaminathan, R.; Brenner, H.; Chen, K.; Chia, K.S.; Chen, J.G.; Law, S.C.; Ahn, Y.O.; Xiang, Y.B.; Yeole, B.B.; et al. Cancer survival in Africa, Asia, and Central America: A population-based study. Lancet Oncol. 2010, 11, 165–173. [Google Scholar] [CrossRef] [PubMed]
- Brenner, H.; Bouvier, A.M.; Foschi, R.; Hackl, M.; Larsen, I.K.; Lemmens, V.; Mangone, L.; Francisci, S.; Group, E.W. Progress in colorectal cancer survival in Europe from the late 1980s to the early 21st century: The EUROCARE study. Int. J. Cancer 2012, 131, 1649–1658. [Google Scholar] [CrossRef]
- Marques, R.P.; Duarte, G.S.; Sterrantino, C.; Pais, H.L.; Quintela, A.; Martins, A.P.; Costa, J. Triplet (FOLFOXIRI) versus doublet (FOLFOX or FOLFIRI) backbone chemotherapy as first-line treatment of metastatic colorectal cancer: A systematic review and meta-analysis. Crit. Rev. Oncol. Hematol. 2017, 118, 54–62. [Google Scholar] [CrossRef]
- Geng, F.; Wang, Z.; Yin, H.; Yu, J.; Cao, B. Molecular Targeted Drugs and Treatment of Colorectal Cancer: Recent Progress and Future Perspectives. Cancer Biother. Radiopharm. 2017, 32, 149–160. [Google Scholar] [CrossRef]
- Goldstein, D.A.; Zeichner, S.B.; Bartnik, C.M.; Neustadter, E.; Flowers, C.R. Metastatic Colorectal Cancer: A Systematic Review of the Value of Current Therapies. Clin. Color. Cancer 2016, 15, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Normanno, N.; Tejpar, S.; Morgillo, F.; De Luca, A.; Van Cutsem, E.; Ciardiello, F. Implications for KRAS status and EGFR-targeted therapies in metastatic CRC. Nat. Rev. Clin. Oncol. 2009, 6, 519–527. [Google Scholar]
- Chin, Y.T.; Hsieh, M.T.; Lin, C.Y.; Kuo, P.J.; Yang, Y.C.; Shih, Y.J.; Lai, H.Y.; Cheng, G.Y.; Tang, H.Y.; Lee, C.C.; et al. 2,3,5,4′-Tetrahydroxystilbene-2-O-beta-glucoside Isolated from Polygoni Multiflori Ameliorates the Development of Periodontitis. Mediators Inflamm. 2016, 2016, 6953459. [Google Scholar] [CrossRef] [Green Version]
- Tsai, P.W.; Lee, Y.H.; Chen, L.G.; Lee, C.J.; Wang, C.C. In Vitro and In Vivo Anti-Osteoarthritis Effects of 2,3,5,4′-Tetrahydroxystilbene-2-O-beta-d-Glucoside from Polygonum Multiflorum. Molecules 2018, 23, 571. [Google Scholar] [CrossRef] [Green Version]
- Fan, Y.F.; Guan, S.Y.; Luo, L.; Li, Y.J.; Yang, L.; Zhou, X.X.; Guo, G.D.; Zhao, M.G.; Yang, Q.; Liu, G. Tetrahydroxystilbene glucoside relieves the chronic inflammatory pain by inhibiting neuronal apoptosis, microglia activation, and GluN2B overexpression in anterior cingulate cortex. Mol. Pain. 2018, 14, 1744806918814367. [Google Scholar] [CrossRef]
- Ling, S.; Xu, J.W. Biological Activities of 2,3,5,4′-Tetrahydroxystilbene-2-O-beta-D-Glucoside in Antiaging and Antiaging-Related Disease Treatments. Oxid. Med. Cell. Longev. 2016, 2016, 4973239. [Google Scholar] [CrossRef] [Green Version]
- Liu, Q.L.; Xiao, J.H.; Ma, R.; Ban, Y.; Wang, J.L. Effect of 2,3,5,4′-tetrahydroxystilbene-2-O-beta-D-glucoside on lipoprotein oxidation and proliferation of coronary arterial smooth cells. J. Asian Nat. Prod. Res. 2007, 9, 689–697. [Google Scholar] [CrossRef]
- Long, T.; Wang, L.; Yang, Y.; Yuan, L.; Zhao, H.; Chang, C.C.; Yang, G.; Ho, C.T.; Li, S. Protective effects of trans-2,3,5,4′-tetrahydroxystilbene 2-O-beta-d-glucopyranoside on liver fibrosis and renal injury induced by CCl4via down-regulating p-ERK1/2 and p-Smad1/2. Food Funct. 2019, 10, 5115–5123. [Google Scholar] [CrossRef]
- Lv, L.; Shao, X.; Wang, L.; Huang, D.; Ho, C.T.; Sang, S. Stilbene glucoside from Polygonum multiflorum Thunb.: A novel natural inhibitor of advanced glycation end product formation by trapping of methylglyoxal. J. Agric. Food Chem. 2010, 58, 2239–2245. [Google Scholar] [CrossRef]
- Guan, S.; Su, W.; Wang, N.; Li, P.; Wang, Y. A potent tyrosinase activator from Radix Polygoni multiflori and its melanogenesis stimulatory effect in B16 melanoma cells. Phytother. Res. 2008, 22, 660–663. [Google Scholar] [CrossRef]
- Han, X.; Ling, S.; Gan, W.; Sun, L.; Duan, J.; Xu, J.W. 2,3,5,4′-tetrahydroxystilbene-2-O-beta-d-glucoside ameliorates vascular senescence and improves blood flow involving a mechanism of p53 deacetylation. Atherosclerosis 2012, 225, 76–82. [Google Scholar] [CrossRef]
- Jiang, C.Y.; Qin, X.Y.; Yuan, M.M.; Lu, G.J.; Cheng, Y. 2,3,5,4′-Tetrahydroxystilbene-2-O-beta-D-glucoside Reverses Stress-Induced Depression via Inflammatory and Oxidative Stress Pathways. Oxid. Med. Cell. Longev. 2018, 2018, 9501427. [Google Scholar] [CrossRef] [Green Version]
- Lee, S.Y.; Ahn, S.M.; Wang, Z.; Choi, Y.W.; Shin, H.K.; Choi, B.T. Neuroprotective effects of 2,3,5,4′-tetrahydoxystilbene-2-O-beta-D-glucoside from Polygonum multiflorum against glutamate-induced oxidative toxicity in HT22 cells. J. Ethnopharmacol. 2017, 195, 64–70. [Google Scholar] [CrossRef]
- Lin, C.Y.; Chin, Y.T.; Kuo, P.J.; Lee, H.W.; Huang, H.M.; Lin, H.Y.; Weng, I.T.; Hsiung, C.N.; Chan, Y.H.; Lee, S.Y. 2,3,5,4′-Tetrahydroxystilbene-2-O-beta-glucoside potentiates self-renewal of human dental pulp stem cells via the AMPK/ERK/SIRT1 axis. Int. Endod. J. 2018, 51, 1159–1170. [Google Scholar] [CrossRef] [PubMed]
- Lin, C.Y.; Kuo, P.J.; Chin, Y.T.; Weng, I.T.; Lee, H.W.; Huang, H.M.; Lin, H.Y.; Hsiung, C.N.; Chan, Y.H.; Lee, S.Y. Dental Pulp Stem Cell Transplantation with 2,3,5,4′-Tetrahydroxystilbene-2-O-beta-D-glucoside Accelerates Alveolar Bone Regeneration in Rats. J. Endod. 2019, 45, 435–441. [Google Scholar] [CrossRef] [PubMed]
- Song, F.; Zhao, J.; Hua, F.; Nian, L.; Zhou, X.X.; Yang, Q.; Xie, Y.H.; Tang, H.F.; Sun, J.Y.; Wang, S.W. Proliferation of rat cardiac stem cells is induced by 2, 3, 5, 4′-tetrahydroxystilbene-2-O-beta-D-glucoside in vitro. Life Sci. 2015, 132, 68–76. [Google Scholar] [CrossRef] [PubMed]
- Xiang, K.; Liu, G.; Zhou, Y.J.; Hao, H.Z.; Yin, Z.; He, A.D.; Da, X.W.; Xiang, J.Z.; Wang, J.L.; Ming, Z.Y. 2,3,5,4′-tetrahydroxystilbene-2-O-beta-D-glucoside (THSG) attenuates human platelet aggregation, secretion and spreading in vitro. Thromb. Res. 2014, 133, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.H.; Wang, W.Q.; Wang, J.L. Protective effect of tetrahydroxystilbene glucoside on cardiotoxicity induced by doxorubicin in vitro and in vivo. Acta Pharmacol. Sin. 2009, 30, 1479–1487. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; Yang, Q.; Song, F.; Bi, L.; Yuan, J.; Guan, S.; Yang, Q.; Wang, S. Tetrahydroxystilbene Glucoside Ameliorates Infrasound-Induced Central Nervous System (CNS) Injury by Improving Antioxidant and Anti-Inflammatory Capacity. Oxid. Med. Cell. Longev. 2020, 2020, 6576718. [Google Scholar] [CrossRef] [Green Version]
- Shen, J.; Zhang, Y.; Shen, H.; Pan, H.; Xu, L.; Yuan, L.; Ding, Z. The synergistic effect of 2,3,5,4′-Tetrahydroxystilbene-2-O-beta-d-glucoside combined with Adriamycin on MCF-7 breast cancer cells. Drug. Des. Devel Ther. 2018, 12, 4083–4094. [Google Scholar] [CrossRef] [Green Version]
- Xu, M.; Wang, C.; Zhu, M.; Wang, X.; Zhang, L.; Zhao, J. 2,3,5,4tetrahydroxy diphenylethylene2Oglucoside inhibits the adhesion and invasion of A549 human lung cancer cells. Mol. Med. Rep. 2017, 16, 8900–8906. [Google Scholar] [CrossRef] [Green Version]
- Lin, C.L.; Hsieh, S.L.; Leung, W.; Jeng, J.H.; Huang, G.C.; Lee, C.T.; Wu, C.C. 2,3,5,4′-tetrahydroxystilbene-2-O-beta-D-glucoside suppresses human colorectal cancer cell metastasis through inhibiting NF-kappaB activation. Int. J. Oncol. 2016, 49, 629–638. [Google Scholar] [CrossRef] [Green Version]
- Chang, Y.-Y.; Lin, H.-J.; Hsiao, L.-C.; Lin, Y.-F.; Chang, C.-S.; Liu, D.-Z. Reduction of breast tumor drug resistance by 2, 3, 5, 4′-tetrahydroxystilbene for exhibition synergic chemotherapeutic effect. PLoS ONE 2021, 16, e0260533. [Google Scholar]
- Singh, R.; Letai, A.; Sarosiek, K. Regulation of apoptosis in health and disease: The balancing act of BCL-2 family proteins. Nat. Rev. Mol. Cell Biol. 2019, 20, 175–193. [Google Scholar]
- Xie, Q.; Liu, Y.; Li, X. The interaction mechanism between autophagy and apoptosis in colon cancer. Transl. Oncol. 2020, 13, 100871. [Google Scholar] [CrossRef] [PubMed]
- Wu, Z.; Geng, Y.; Lu, X.; Shi, Y.; Wu, G.; Zhang, M.; Shan, B.; Pan, H.; Yuan, J. Chaperone-mediated autophagy is involved in the execution of ferroptosis. Proc. Natl. Acad. Sci. USA 2019, 116, 2996–3005. [Google Scholar]
- Ko, J.H.; Sethi, G.; Um, J.Y.; Shanmugam, M.K.; Arfuso, F.; Kumar, A.P.; Bishayee, A.; Ahn, K.S. The Role of Resveratrol in Cancer Therapy. Int. J. Mol. Sci. 2017, 18, 2589. [Google Scholar] [CrossRef] [Green Version]
- Rauf, A.; Imran, M.; Butt, M.S.; Nadeem, M.; Peters, D.G.; Mubarak, M.S. Resveratrol as an anti-cancer agent: A review. Crit. Rev. Food Sci. Nutr. 2018, 58, 1428–1447. [Google Scholar] [CrossRef]
- Lin, E.Y.; Bayarsengee, U.; Wang, C.C.; Chiang, Y.H.; Cheng, C.W. The natural compound 2,3,5,4′-tetrahydroxystilbene-2-O-beta-d glucoside protects against adriamycin-induced nephropathy through activating the Nrf2-Keap1 antioxidant pathway. Environ. Toxicol. 2018, 33, 72–82. [Google Scholar] [CrossRef] [Green Version]
- Patridge, E.; Gareiss, P.; Kinch, M.S.; Hoyer, D. An analysis of FDA-approved drugs: Natural products and their derivatives. Drug. Discov. Today 2016, 21, 204–207. [Google Scholar] [CrossRef]
- Cragg, G.M.; Newman, D.J. Natural products: A continuing source of novel drug leads. Biochim. Biophys. Acta 2013, 1830, 3670–3695. [Google Scholar] [CrossRef] [Green Version]
- Guo, Z. The modification of natural products for medical use. Acta Pharm. Sinica B 2017, 7, 119–136. [Google Scholar]
- Kung, C.-P.; Budina, A.; Balaburski, G.; Bergenstock, M.K.; Murphy, M. Autophagy in tumor suppression and cancer therapy. Crit. Rev. Eukaryot. Gene Expr. 2011, 21, 71–100. [Google Scholar] [CrossRef] [Green Version]
- Yang, W.S.; Stockwell, B.R. Synthetic lethal screening identifies compounds activating iron-dependent, nonapoptotic cell death in oncogenic-RAS-harboring cancer cells. Chem. Biol. 2008, 15, 234–245. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, M.; Monian, P.; Pan, Q.; Zhang, W.; Xiang, J.; Jiang, X. Ferroptosis is an autophagic cell death process. Cell Res. 2016, 26, 1021–1032. [Google Scholar]
- Field, J.K.; Spandidos, D.A. The role of ras and myc oncogenes in human solid tumours and their relevance in diagnosis and prognosis (review). Anticancer. Res. 1990, 10, 1–22. [Google Scholar] [PubMed]
- Tan, L.; Peng, D.; Cheng, Y. Significant position of C-myc in colorectal cancer: A promising therapeutic target. Clin. Transl. Oncol. 2022, 24, 2295–2304. [Google Scholar]
- Zhang, C.; Liu, X.; Jin, S.; Chen, Y.; Guo, R. Ferroptosis in cancer therapy: A novel approach to reversing drug resistance. Mol. Cancer 2022, 21, 47. [Google Scholar] [CrossRef]
- Zhang, X.; Ma, Y.; Ma, J.; Yang, L.; Song, Q.; Wang, H.; Lv, G. Glutathione peroxidase 4 as a therapeutic target for anti-colorectal cancer drug-tolerant persister cells. Front. Oncol. 2022, 12, 913669. [Google Scholar]
- Asada, Y.; Tsuruta, M.; Okabayashi, K.; Shigeta, K.; Ishida, T.; Shimada, T.; Suzumura, H.; Koishikawa, K.; Akimoto, S.; Hasegawa, H. Inhibition of heat-shock protein 27 reduces 5-fluorouracil-acquired resistance in human colon cancer cells. Anticancer. Res. 2021, 41, 1283–1290. [Google Scholar] [CrossRef]
- Zhang, K.; Wu, Y.; Chen, G.; Wang, H.; Liu, Y.; Zhou, Y. Heat shock protein 27 deficiency promotes ferrous ion absorption and enhances acyl-Coenzyme A synthetase long-chain family member 4 stability to promote glioblastoma cell ferroptosis. Cancer Cell. Int. 2023, 23, 5. [Google Scholar] [CrossRef]
- Hou, J.; Jiang, C.; Wen, X.; Li, C.; Xiong, S.; Yue, T.; Long, P.; Shi, J.; Zhang, Z. ACSL4 as a potential target and biomarker for anticancer: From molecular mechanisms to clinical therapeutics. Front. Pharmacol. 2022, 13, 949863. [Google Scholar]






Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, K.-Y.; Wei, P.-L.; Lee, C.-C.; Makondi, P.T.; Chen, H.-A.; Chang, Y.-Y.; Liu, D.-Z.; Huang, C.-Y.; Chang, Y.-J. 2,3,5,4′-Tetrahydroxystilbene (TG1), a Novel Compound Derived from 2,3,5,4′-Tetrahydroxystilbene-2-O-β-D-glucoside (THSG), Inhibits Colorectal Cancer Progression by Inducing Ferroptosis, Apoptosis, and Autophagy. Biomedicines 2023, 11, 1798. https://doi.org/10.3390/biomedicines11071798
Tsai K-Y, Wei P-L, Lee C-C, Makondi PT, Chen H-A, Chang Y-Y, Liu D-Z, Huang C-Y, Chang Y-J. 2,3,5,4′-Tetrahydroxystilbene (TG1), a Novel Compound Derived from 2,3,5,4′-Tetrahydroxystilbene-2-O-β-D-glucoside (THSG), Inhibits Colorectal Cancer Progression by Inducing Ferroptosis, Apoptosis, and Autophagy. Biomedicines. 2023; 11(7):1798. https://doi.org/10.3390/biomedicines11071798
Chicago/Turabian StyleTsai, Kuei-Yen, Po-Li Wei, Cheng-Chin Lee, Precious Takondwa Makondi, Hsin-An Chen, Yao-Yuan Chang, Der-Zen Liu, Chien-Yu Huang, and Yu-Jia Chang. 2023. "2,3,5,4′-Tetrahydroxystilbene (TG1), a Novel Compound Derived from 2,3,5,4′-Tetrahydroxystilbene-2-O-β-D-glucoside (THSG), Inhibits Colorectal Cancer Progression by Inducing Ferroptosis, Apoptosis, and Autophagy" Biomedicines 11, no. 7: 1798. https://doi.org/10.3390/biomedicines11071798





