The Expression Characteristics and Function of the RECQ Family in Pan-Cancer
Abstract
:1. Introduction
2. Methods
2.1. Source of Data and Its Processing
2.2. Analysis of RECQs for Mutations and CNVs
2.3. Evaluation of the Level of mRNA and Protein Expression
2.4. Building Networks of Protein Interactions
2.5. Perform Enrichment Analysis Using GO and KEGG
2.6. Analysis Related to the Immune System
2.7. Survival Analysis
2.8. Statistical Analysis
3. Results
3.1. Analysis of RECQs’ Expression, Interaction, and Functional Enrichment
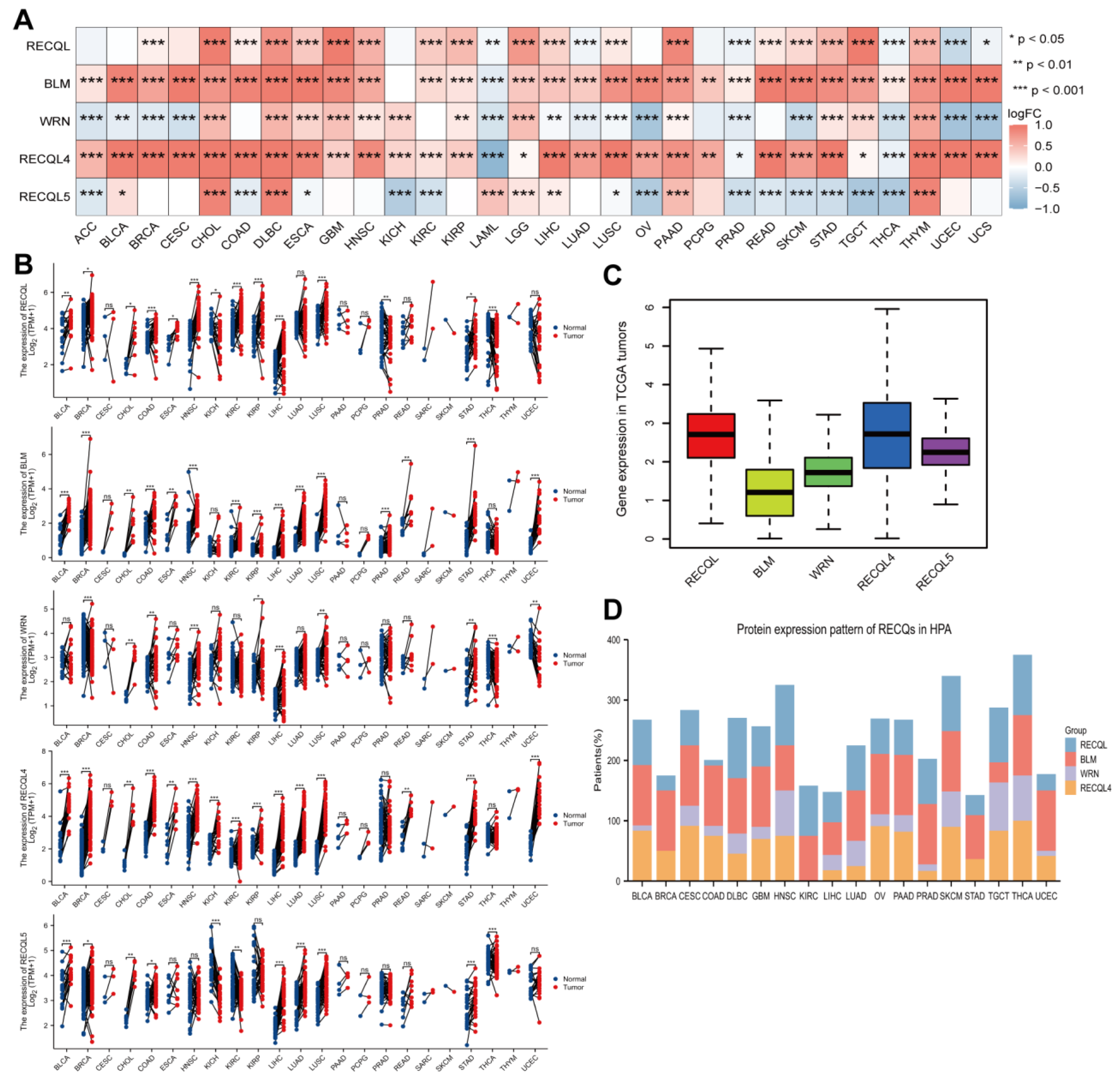
3.2. RECQs mRNA and Protein Expression in Various Cancer Types
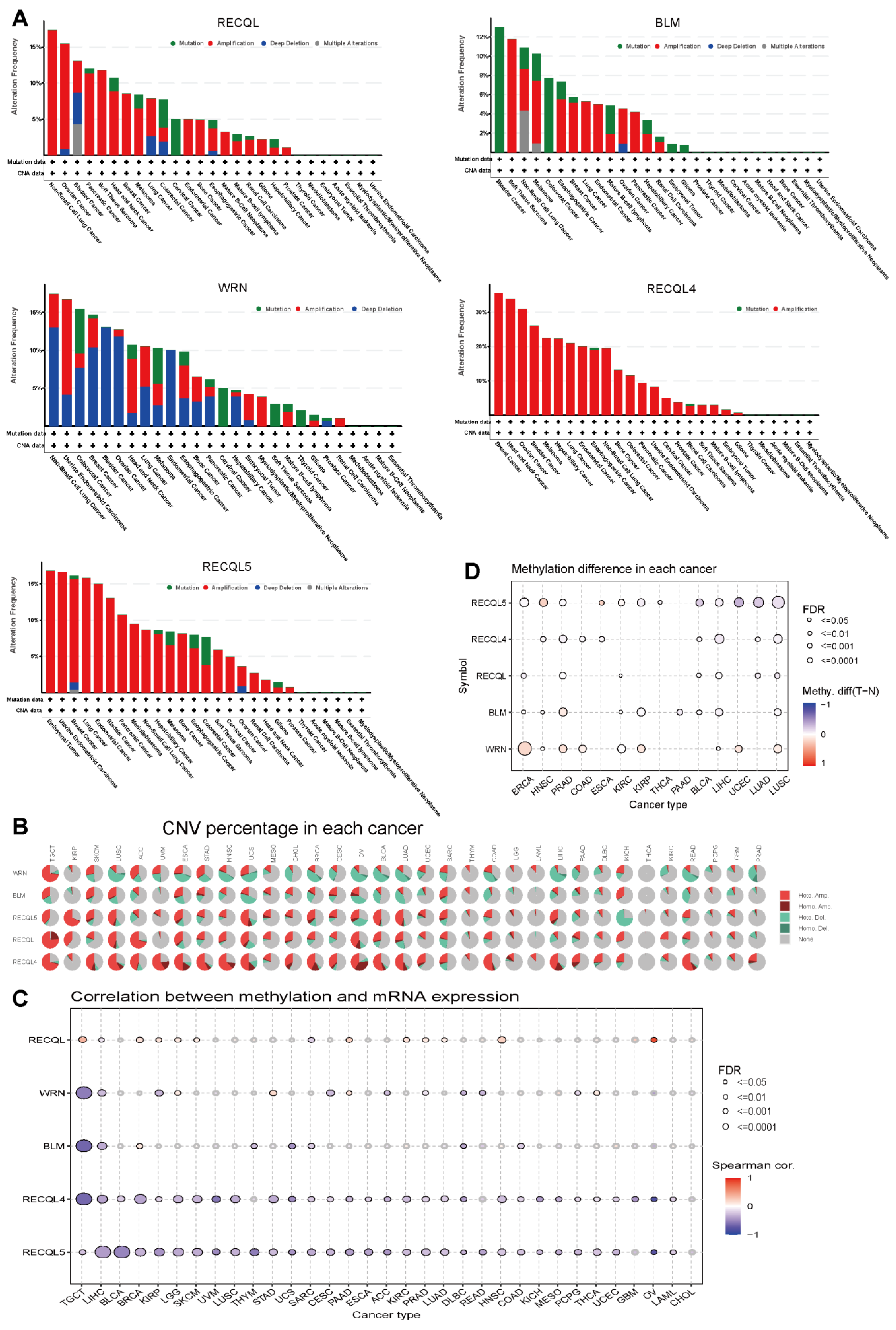
3.3. Variations in Genetic Alterations and Methylation of RECQs in Different Cancers
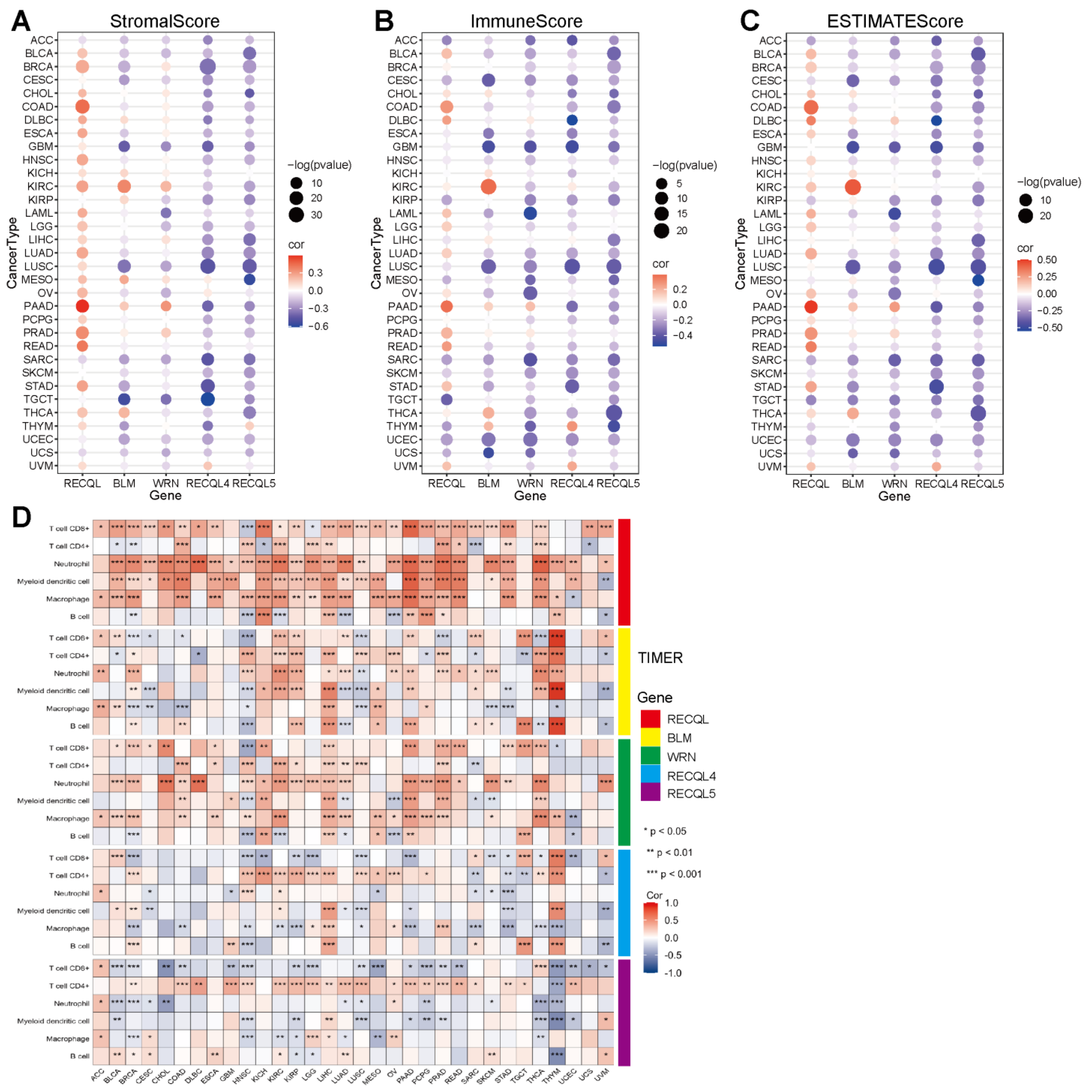
3.4. The Correlation between RECQs and Immune Infiltration in Pan-Cancer
3.5. The Relationship between mRNA Expression and the Predictive Significance of RECQs
3.6. The Relationship between the Expression of RECQs and the Clinical Characteristics in LIHC
3.7. Preliminary Verification of Characteristics of RECQL4 in LIHC
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem. Biol. Interact. 2006, 160, 1–40. [Google Scholar] [CrossRef]
- Basu, A.K.; Nohmi, T. Chemically-Induced DNA Damage, Mutagenesis, and Cancer. Int. J. Mol. Sci. 2018, 19, 1767. [Google Scholar] [CrossRef]
- Jackson, S.P.; Bartek, J. The DNA-damage response in human biology and disease. Nature 2009, 461, 1071–1078. [Google Scholar] [CrossRef] [PubMed]
- Najima, Y.; Tomizawa-Murasawa, M.; Saito, Y.; Watanabe, T.; Ono, R.; Ochi, T.; Suzuki, N.; Fujiwara, H.; Ohara, O.; Shultz, L.D.; et al. Induction of WT1-specific human CD8+ T cells from human HSCs in HLA class I Tg NOD/SCID/IL2rgKO mice. Blood 2016, 127, 722–734. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Riviere, I.; Gonen, M.; Wang, X.; Senechal, B.; Curran, K.J.; Sauter, C.; Wang, Y.; Santomasso, B.; Mead, E.; et al. Long-Term Follow-up of CD19 CAR Therapy in Acute Lymphoblastic Leukemia. N. Engl. J. Med. 2018, 378, 449–459. [Google Scholar] [CrossRef] [PubMed]
- Tanyi, J.L.; Bobisse, S.; Ophir, E.; Tuyaerts, S.; Roberti, A.; Genolet, R.; Baumgartner, P.; Stevenson, B.J.; Iseli, C.; Dangaj, D.; et al. Personalized cancer vaccine effectively mobilizes antitumor T cell immunity in ovarian cancer. Sci. Transl. Med. 2018, 10, eaao5931. [Google Scholar] [CrossRef]
- Benci, J.L.; Xu, B.; Qiu, Y.; Wu, T.J.; Dada, H.; Twyman-Saint Victor, C.; Cucolo, L.; Lee, D.S.M.; Pauken, K.E.; Huang, A.C.; et al. Tumor Interferon Signaling Regulates a Multigenic Resistance Program to Immune Checkpoint Blockade. Cell 2016, 167, 1540–1554.e12. [Google Scholar] [CrossRef]
- Zhang, J.; Liu, D.; Li, G.; Staveley-O’Carroll, K.F.; Graff, J.N.; Li, Z.; Wu, J.D. Antibody-mediated neutralization of soluble MIC significantly enhances CTLA4 blockade therapy. Sci. Adv. 2017, 3, e1602133. [Google Scholar] [CrossRef]
- Hong, L.K.; Chen, Y.; Smith, C.C.; Montgomery, S.A.; Vincent, B.G.; Dotti, G.; Savoldo, B. CD30-Redirected Chimeric Antigen Receptor T Cells Target CD30(+) and CD30(−) Embryonal Carcinoma via Antigen-Dependent and Fas/FasL Interactions. Cancer Immunol. Res. 2018, 6, 1274–1287. [Google Scholar] [CrossRef]
- Murciano-Goroff, Y.R.; Warner, A.B.; Wolchok, J.D. The future of cancer immunotherapy: Microenvironment-targeting combinations. Cell Res. 2020, 30, 507–519. [Google Scholar] [CrossRef]
- Larsen, N.B.; Hickson, I.D. RecQ Helicases: Conserved Guardians of Genomic Integrity. Adv. Exp. Med. Biol. 2013, 767, 161–184. [Google Scholar]
- Rezazadeh, S. RecQ helicases; at the crossroad of genome replication, repair, and recombination. Mol. Biol. Rep. 2012, 39, 4527–4543. [Google Scholar] [CrossRef]
- Du, X.; Zhang, C.; Yin, C.; Wang, W.; Yan, X.; Xie, D.; Zheng, X.; Zheng, Q.; Li, M.; Song, Z. High BLM Expression Predicts Poor Clinical Outcome and Contributes to Malignant Progression in Human Cholangiocarcinoma. Front. Oncol. 2021, 11, 633899. [Google Scholar] [CrossRef]
- Wang, G.; Cen, Y.; Wang, C.; Xiang, W.; Li, S.; Ming, Y.; Chen, L.; Zhou, J. Elevated RECQL1 expression predicts poor prognosis and associates with tumor immune infiltration in low-grade glioma. Transl. Cancer Res. 2022, 11, 1552–1564. [Google Scholar] [CrossRef]
- Cybulski, C.; Carrot-Zhang, J.; Kluzniak, W.; Rivera, B.; Kashyap, A.; Wokolorczyk, D.; Giroux, S.; Nadaf, J.; Hamel, N.; Zhang, S.; et al. Germline RECQL mutations are associated with breast cancer susceptibility. Nat. Genet. 2015, 47, 643–646. [Google Scholar] [CrossRef]
- Savva, C.; Sadiq, M.; Sheikh, O.; Karim, S.; Trivedi, S.; Green, A.R.; Rakha, E.A.; Madhusudan, S.; Arora, A. Werner Syndrome Protein Expression in Breast Cancer. Clin. Breast Cancer 2021, 21, 57–73.e7. [Google Scholar] [CrossRef] [PubMed]
- Marchena-Perea, E.M.; Salazar-Hidalgo, M.E.; Gomez-Sanz, A.; Arranz-Ledo, M.; Barroso, A.; Fernandez, V.; Tejera-Perez, H.; Pita, G.; Nunez-Torres, R.; Pombo, L.; et al. A Large Case-Control Study Performed in Spanish Population Suggests That RECQL5 Is the Only RECQ Helicase Involved in Breast Cancer Susceptibility. Cancers 2022, 14, 4738. [Google Scholar] [CrossRef]
- Fierheller, C.T.; Alenezi, W.M.; Tonin, P.N. The Genetic Analyses of French Canadians of Quebec Facilitate the Characterization of New Cancer Predisposing Genes Implicated in Hereditary Breast and/or Ovarian Cancer Syndrome Families. Cancers 2021, 13, 3406. [Google Scholar] [CrossRef] [PubMed]
- Tang, T.; Tan, X.; Wang, Z.; Wang, S.; Wang, Y.; Xu, J.; Wei, X.; Zhang, D.; Liu, Q.; Jiang, J. Germline Mutations in Patients with Early-Onset Prostate Cancer. Front. Oncol. 2022, 12, 826778. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Huang, T.T.; Nair, J.R.; An, D.; Zurcher, G.; Lampert, E.J.; McCoy, A.; Cimino-Mathews, A.; Swisher, E.M.; Radke, M.R.; et al. BLM overexpression as a predictive biomarker for CHK1 inhibitor response in PARP inhibitor-resistant BRCA-mutant ovarian cancer. Sci. Transl. Med. 2023, 15, eadd7872. [Google Scholar] [CrossRef]
- Goldman, M.J.; Craft, B.; Hastie, M.; Repecka, K.; McDade, F.; Kamath, A.; Banerjee, A.; Luo, Y.; Rogers, D.; Brooks, A.N.; et al. Visualizing and interpreting cancer genomics data via the Xena platform. Nat. Biotechnol. 2020, 38, 675–678. [Google Scholar] [CrossRef] [PubMed]
- Ritchie, M.E.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- Cerami, E.; Gao, J.; Dogrusoz, U.; Gross, B.E.; Sumer, S.O.; Aksoy, B.A.; Jacobsen, A.; Byrne, C.J.; Heuer, M.L.; Larsson, E.; et al. The cBio cancer genomics portal: An open platform for exploring multidimensional cancer genomics data. Cancer Discov. 2012, 2, 401–404. [Google Scholar] [CrossRef]
- Gao, J.; Aksoy, B.A.; Dogrusoz, U.; Dresdner, G.; Gross, B.; Sumer, S.O.; Sun, Y.; Jacobsen, A.; Sinha, R.; Larsson, E.; et al. Integrative analysis of complex cancer genomics and clinical profiles using the cBioPortal. Sci. Signal 2013, 6, pl1. [Google Scholar] [CrossRef]
- Liu, C.J.; Hu, F.F.; Xia, M.X.; Han, L.; Zhang, Q.; Guo, A.Y. GSCALite: A web server for gene set cancer analysis. Bioinformatics 2018, 34, 3771–3772. [Google Scholar] [CrossRef] [PubMed]
- Thul, P.J.; Akesson, L.; Wiking, M.; Mahdessian, D.; Geladaki, A.; Ait Blal, H.; Alm, T.; Asplund, A.; Bjork, L.; Breckels, L.M.; et al. A subcellular map of the human proteome. Science 2017, 356, eaal3321. [Google Scholar] [CrossRef]
- Uhlen, M.; Zhang, C.; Lee, S.; Sjostedt, E.; Fagerberg, L.; Bidkhori, G.; Benfeitas, R.; Arif, M.; Liu, Z.; Edfors, F.; et al. A pathology atlas of the human cancer transcriptome. Science 2017, 357, eaan2507. [Google Scholar] [CrossRef]
- Szklarczyk, D.; Gable, A.L.; Lyon, D.; Junge, A.; Wyder, S.; Huerta-Cepas, J.; Simonovic, M.; Doncheva, N.T.; Morris, J.H.; Bork, P.; et al. STRING v11: Protein-protein association networks with increased coverage, supporting functional discovery in genome-wide experimental datasets. Nucleic Acids Res. 2019, 47, D607–D613. [Google Scholar] [CrossRef]
- Mora, A.; Donaldson, I.M. iRefR: An R package to manipulate the iRefIndex consolidated protein interaction database. BMC Bioinform. 2011, 12, 455. [Google Scholar] [CrossRef]
- Kanehisa, M.; Goto, S. KEGG: Kyoto encyclopedia of genes and genomes. Nucleic Acids Res. 2000, 28, 27–30. [Google Scholar] [CrossRef]
- Yu, G.; Wang, L.G.; Han, Y.; He, Q.Y. clusterProfiler: An R package for comparing biological themes among gene clusters. OMICS 2012, 16, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Ru, B.; Wong, C.N.; Tong, Y.; Zhong, J.Y.; Zhong, S.S.W.; Wu, W.C.; Chu, K.C.; Wong, C.Y.; Lau, C.Y.; Chen, I.; et al. TISIDB: An integrated repository portal for tumor-immune system interactions. Bioinformatics 2019, 35, 4200–4202. [Google Scholar] [CrossRef]
- Sturm, G.; Finotello, F.; Petitprez, F.; Zhang, J.D.; Baumbach, J.; Fridman, W.H.; List, M.; Aneichyk, T. Comprehensive evaluation of transcriptome-based cell-type quantification methods for immuno-oncology. Bioinformatics 2019, 35, i436–i445. [Google Scholar] [CrossRef] [PubMed]
- Gyorffy, B. Survival analysis across the entire transcriptome identifies biomarkers with the highest prognostic power in breast cancer. Comput. Struct. Biotechnol. J. 2021, 19, 4101–4109. [Google Scholar] [CrossRef]
- Yarchoan, M.; Hopkins, A.; Jaffee, E.M. Tumor Mutational Burden and Response Rate to PD-1 Inhibition. N. Engl. J. Med. 2017, 377, 2500–2501. [Google Scholar] [CrossRef] [PubMed]
- Hellmann, M.D.; Ciuleanu, T.E.; Pluzanski, A.; Lee, J.S.; Otterson, G.A.; Audigier-Valette, C.; Minenza, E.; Linardou, H.; Burgers, S.; Salman, P.; et al. Nivolumab plus Ipilimumab in Lung Cancer with a High Tumor Mutational Burden. N. Engl. J. Med. 2018, 378, 2093–2104. [Google Scholar] [CrossRef]
- Zhao, P.; Li, L.; Jiang, X.; Li, Q. Mismatch repair deficiency/microsatellite instability-high as a predictor for anti-PD-1/PD-L1 immunotherapy efficacy. J. Hematol. Oncol. 2019, 12, 54. [Google Scholar] [CrossRef]
- Thakkar, M.K.; Lee, J.; Meyer, S.; Chang, V.Y. RecQ Helicase Somatic Alterations in Cancer. Front. Mol. Biosci. 2022, 9, 887758. [Google Scholar] [CrossRef]
- Shlien, A.; Tabori, U.; Marshall, C.R.; Pienkowska, M.; Feuk, L.; Novokmet, A.; Nanda, S.; Druker, H.; Scherer, S.W.; Malkin, D. Excessive genomic DNA copy number variation in the Li-Fraumeni cancer predisposition syndrome. Proc. Natl. Acad. Sci. USA 2008, 105, 11264–11269. [Google Scholar] [CrossRef]
- Anastasiadi, D.; Esteve-Codina, A.; Piferrer, F. Consistent inverse correlation between DNA methylation of the first intron and gene expression across tissues and species. Epigenetics Chromatin 2018, 11, 37. [Google Scholar] [CrossRef]
- Li, L.; Gao, M.; Song, B.; Zhang, H.; Wang, Y. Effects of RECQ1 helicase silencing on non-small cell lung cancer cells. Biomed. Pharmacother. 2016, 83, 1227–1232. [Google Scholar] [CrossRef]
- Kawabe, T.; Tsuyama, N.; Kitao, S.; Nishikawa, K.; Shimamoto, A.; Shiratori, M.; Matsumoto, T.; Anno, K.; Sato, T.; Mitsui, Y.; et al. Differential regulation of human RecQ family helicases in cell transformation and cell cycle. Oncogene 2000, 19, 4764–4772. [Google Scholar] [CrossRef] [PubMed]
- Opresko, P.L.; Calvo, J.P.; von Kobbe, C. Role for the Werner syndrome protein in the promotion of tumor cell growth. Mech. Ageing Dev. 2007, 128, 423–436. [Google Scholar] [CrossRef] [PubMed]
- Lu, L.; Jin, W.; Wang, L.L. RECQ DNA Helicases and Osteosarcoma. Adv. Exp. Med. Biol. 2020, 1258, 37–54. [Google Scholar]
- Wu, J.; Zhi, L.; Dai, X.; Cai, Q.; Ma, W. Decreased RECQL5 correlated with disease progression of osteosarcoma. Biochem. Biophys. Res. Commun. 2015, 467, 617–622. [Google Scholar] [CrossRef] [PubMed]
- Patterson, K.; Arya, L.; Bottomley, S.; Morgan, S.; Cox, A.; Catto, J.; Bryant, H.E. Altered RECQL5 expression in urothelial bladder carcinoma increases cellular proliferation and makes RECQL5 helicase activity a novel target for chemotherapy. Oncotarget 2016, 7, 76140–76150. [Google Scholar] [CrossRef] [PubMed]
- Bottcher, J.P.; Bonavita, E.; Chakravarty, P.; Blees, H.; Cabeza-Cabrerizo, M.; Sammicheli, S.; Rogers, N.C.; Sahai, E.; Zelenay, S.; Reis e Sousa, C. NK Cells Stimulate Recruitment of cDC1 into the Tumor Microenvironment Promoting Cancer Immune Control. Cell 2018, 172, 1022–1037.e14. [Google Scholar] [CrossRef]
- Cooper, J.; Giancotti, F.G. Integrin Signaling in Cancer: Mechanotransduction, Stemness, Epithelial Plasticity, and Therapeutic Resistance. Cancer Cell 2019, 35, 347–367. [Google Scholar] [CrossRef]
- Malta, T.M.; Sokolov, A.; Gentles, A.J.; Burzykowski, T.; Poisson, L.; Weinstein, J.N.; Kaminska, B.; Huelsken, J.; Omberg, L.; Gevaert, O.; et al. Machine Learning Identifies Stemness Features Associated with Oncogenic Dedifferentiation. Cell 2018, 173, 338–354.e15. [Google Scholar] [CrossRef]
- Krishnan, V.; Lim, D.X.E.; Hoang, P.M.; Srivastava, S.; Matsuo, J.; Huang, K.K.; Zhu, F.; Ho, K.Y.; So, J.B.Y.; Khor, C.; et al. DNA damage signalling as an anti-cancer barrier in gastric intestinal metaplasia. Gut 2020, 69, 1738–1749. [Google Scholar] [CrossRef]
- Wu, J.; Yu, X.; Zhu, H.; Chen, P.; Liu, T.; Yin, R.; Qiang, Y.; Xu, L. RelB is a potential molecular biomarker for immunotherapy in human pan-cancer. Front. Mol. Biosci. 2023, 10, 1178446. [Google Scholar] [CrossRef] [PubMed]
- Picco, G.; Cattaneo, C.M.; van Vliet, E.J.; Crisafulli, G.; Rospo, G.; Consonni, S.; Vieira, S.F.; Rodriguez, I.S.; Cancelliere, C.; Banerjee, R.; et al. Werner Helicase Is a Synthetic-Lethal Vulnerability in Mismatch Repair-Deficient Colorectal Cancer Refractory to Targeted Therapies, Chemotherapy, and Immunotherapy. Cancer Discov. 2021, 11, 1923–1937. [Google Scholar] [CrossRef]
- Calin, G.; Ranzani, G.N.; Amadori, D.; Herlea, V.; Matei, I.; Barbanti-Brodano, G.; Negrini, M. Somatic frameshift mutations in the Bloom syndrome BLM gene are frequent in sporadic gastric carcinomas with microsatellite mutator phenotype. BMC Genet. 2001, 2, 14. [Google Scholar] [CrossRef]
- Leslie, M. High TMB Predicts Immunotherapy Benefit. Cancer Discov. 2018, 8, 668. [Google Scholar]
- Samstein, R.M.; Lee, C.H.; Shoushtari, A.N.; Hellmann, M.D.; Shen, R.; Janjigian, Y.Y.; Barron, D.A.; Zehir, A.; Jordan, E.J.; Omuro, A.; et al. Tumor mutational load predicts survival after immunotherapy across multiple cancer types. Nat. Genet. 2019, 51, 202–206. [Google Scholar] [CrossRef]
- Overman, M.J.; McDermott, R.; Leach, J.L.; Lonardi, S.; Lenz, H.J.; Morse, M.A.; Desai, J.; Hill, A.; Axelson, M.; Moss, R.A.; et al. Nivolumab in patients with metastatic DNA mismatch repair-deficient or microsatellite instability-high colorectal cancer (CheckMate 142): An open-label, multicentre, phase 2 study. Lancet Oncol. 2017, 18, 1182–1191. [Google Scholar] [CrossRef] [PubMed]
- Luchini, C.; Bibeau, F.; Ligtenberg, M.J.L.; Singh, N.; Nottegar, A.; Bosse, T.; Miller, R.; Riaz, N.; Douillard, J.Y.; Andre, F.; et al. ESMO recommendations on microsatellite instability testing for immunotherapy in cancer, and its relationship with PD-1/PD-L1 expression and tumour mutational burden: A systematic review-based approach. Ann. Oncol. 2019, 30, 1232–1243. [Google Scholar] [CrossRef] [PubMed]
- McDonald, K.A.; Kawaguchi, T.; Qi, Q.; Peng, X.; Asaoka, M.; Young, J.; Opyrchal, M.; Yan, L.; Patnaik, S.; Otsuji, E.; et al. Tumor Heterogeneity Correlates with Less Immune Response and Worse Survival in Breast Cancer Patients. Ann. Surg. Oncol. 2019, 26, 2191–2199. [Google Scholar] [CrossRef] [PubMed]
- Kondratova, M.; Czerwinska, U.; Sompairac, N.; Amigorena, S.D.; Soumelis, V.; Barillot, E.; Zinovyev, A.; Kuperstein, I. A multiscale signalling network map of innate immune response in cancer reveals cell heterogeneity signatures. Nat. Commun. 2019, 10, 4808. [Google Scholar] [CrossRef]
- Mahmoodi, A.; Shoqafi, A.; Sun, P.; Giannakeas, V.; Cybulski, C.; Nofech-Mozes, S.; Masson, J.Y.; Sharma, S.; Samani, A.A.; Madhusudan, S.; et al. High Expression of RECQL Protein in ER-Positive Breast Tumours Is Associated With a Better Survival. Front. Oncol. 2022, 12, 877617. [Google Scholar] [CrossRef]
- Feng, S.; Qian, X.; Feng, D.; Zhang, X. Downregulation of BLM RecQ helicase inhibits proliferation, promotes the apoptosis and enhances the sensitivity of bladder cancer cells to cisplatin. Mol. Med. Rep. 2022, 26, 313. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, S.; Dutta, K.; Gupta, P.; Das, A.; Das, A.; Ghosh, S.K.; Patro, B.S. Targeting RECQL5 Functions, by a Small Molecule, Selectively Kills Breast Cancer In Vitro and In Vivo. J. Med. Chem. 2021, 64, 1524–1544. [Google Scholar] [CrossRef] [PubMed]
- Chun, S.G.; Yee, N.S. Werner syndrome as a hereditary risk factor for exocrine pancreatic cancer: Potential role of WRN in pancreatic tumorigenesis and patient-tailored therapy. Cancer Biol. Ther. 2010, 10, 430–437. [Google Scholar] [CrossRef]
- Lan, C.; Yamashita, Y.I.; Hayashi, H.; Nakagawa, S.; Imai, K.; Mima, K.; Kaida, T.; Matsumoto, T.; Maruno, M.; Liu, Z.; et al. High Expression of Bloom Syndrome Helicase is a Key Factor for Poor Prognosis and Advanced Malignancy in Patients with Pancreatic Cancer: A Retrospective Study. Ann. Surg. Oncol. 2022, 29, 3551–3564. [Google Scholar] [CrossRef]
- Lai, J.; Chen, W.; Zhao, A.; Huang, J. Determination of a DNA repair-related gene signature with potential implications for prognosis and therapeutic response in pancreatic adenocarcinoma. Front. Oncol. 2022, 12, 939891. [Google Scholar] [CrossRef] [PubMed]




| Characteristics | Total (N) | Univariate Analysis | Multivariate Analysis | ||
|---|---|---|---|---|---|
| Hazard Ratio (95% CI) | p Value | Hazard Ratio (95% CI) | p Value | ||
| Age | 373 | ||||
| ≤60 | 177 | Reference | |||
| >60 | 196 | 1.205 (0.850–1.708) | 0.295 | ||
| Gender | 373 | ||||
| Female | 121 | Reference | |||
| Male | 252 | 0.793 (0.557–1.130) | 0.2 | ||
| Race | 361 | ||||
| White | 185 | Reference | |||
| Asian and Black or African American | 176 | 0.791 (0.551–1.135) | 0.203 | ||
| BMI | 336 | ||||
| ≤25 | 177 | Reference | |||
| >25 | 159 | 0.798 (0.550–1.158) | 0.235 | ||
| Tumor status | 354 | ||||
| Tumor free | 202 | Reference | |||
| With tumor | 152 | 2.317 (1.590–3.376) | <0.001 | 1.794 (1.200–2.684) | 0.004 |
| Residual tumor | 344 | ||||
| R0 | 326 | Reference | |||
| R1 and R2 | 18 | 1.604 (0.812–3.169) | 0.174 | ||
| Pathologic stage | 349 | ||||
| Stage I and Stage II | 259 | Reference | |||
| Stage III and Stage IV | 90 | 2.504 (1.727–3.631) | <0.001 | 2.075 (1.393–3.091) | <0.001 |
| Adjacent hepatic tissue inflammation | 236 | ||||
| None | 118 | Reference | |||
| Mild and Severe | 118 | 1.194 (0.734–1.942) | 0.475 | ||
| AFP (ng/mL) | 279 | ||||
| ≤400 | 215 | Reference | |||
| >400 | 64 | 1.075 (0.658–1.759) | 0.772 | ||
| Albumin (g/dL) | 299 | ||||
| <3.5 | 69 | Reference | |||
| ≥3.5 | 230 | 0.897 (0.549–1.464) | 0.662 | ||
| Prothrombin time | 296 | ||||
| ≤4 | 207 | Reference | |||
| >4 | 89 | 1.335 (0.881–2.023) | 0.174 | ||
| Vascular invasion | 317 | ||||
| No | 208 | Reference | |||
| Yes | 109 | 1.344 (0.887–2.035) | 0.163 | ||
| RECQL (high vs. low) | 373 | 1.454 (1.026–2.060) | 0.035 | 1.047 (0.637–1.719) | 0.858 |
| BLM (high vs. low) | 373 | 1.270 (0.900–1.793) | 0.174 | ||
| WRN (high vs. low) | 373 | 1.473 (1.041–2.086) | 0.029 | 1.235 (0.752–2.027) | 0.405 |
| RECQL4 (high vs. low) | 373 | 1.672 (1.180–2.371) | 0.004 | 1.554 (1.042–2.318) | 0.031 |
| RECQL5 (high vs. low) | 373 | 1.554 (1.098–2.199) | 0.013 | 1.221 (0.825–1.809) | 0.318 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhou, Y.; Huang, X.; Wang, L.; Luo, Y. The Expression Characteristics and Function of the RECQ Family in Pan-Cancer. Biomedicines 2023, 11, 2318. https://doi.org/10.3390/biomedicines11082318
Zhou Y, Huang X, Wang L, Luo Y. The Expression Characteristics and Function of the RECQ Family in Pan-Cancer. Biomedicines. 2023; 11(8):2318. https://doi.org/10.3390/biomedicines11082318
Chicago/Turabian StyleZhou, Yuanyuan, Xucheng Huang, Liya Wang, and Yujia Luo. 2023. "The Expression Characteristics and Function of the RECQ Family in Pan-Cancer" Biomedicines 11, no. 8: 2318. https://doi.org/10.3390/biomedicines11082318
APA StyleZhou, Y., Huang, X., Wang, L., & Luo, Y. (2023). The Expression Characteristics and Function of the RECQ Family in Pan-Cancer. Biomedicines, 11(8), 2318. https://doi.org/10.3390/biomedicines11082318






