Cytocompatibility and Osteoinductive Properties of Collagen-Fibronectin Hydrogel Impregnated with siRNA Targeting Glycogen Synthase Kinase 3β: In Vitro Study
Abstract
:1. Introduction
2. Materials and Methods
2.1. siRNA Design
2.2. Polyplexes Containing siRNA and PEI Preparation
2.3. Fabrication of Collagen–Fibronectin Hydrogel Impregnated with Polyplexes, Containing siRNA GSK3β Molecules
2.4. siRNA GSK3β Molecules’ Release Monitoring
2.5. Cell Cultures
2.6. Immunophenotyping of hAD-MSCs
2.7. Assessment of Delivery Efficiency to the hAD-MSCs Cultures
2.8. MTT Assay
2.9. Staining of Cells Adhered to the Surface of Materials
2.10. GSK3β Gene Knockdown and Osteogenic Differentiation of hAD-MSCs
2.11. Evaluation of the Expression of the GSK3β Gene and Osteogenic Differentiation Marker Genes
2.12. Statistical Analysis
3. Results
3.1. Obtaining Collagen–Fibronectin Hydrogel Impregnated with siRNA GSK3β Molecules
3.2. Immunophenotypic Characteristics of hAD-MSCs
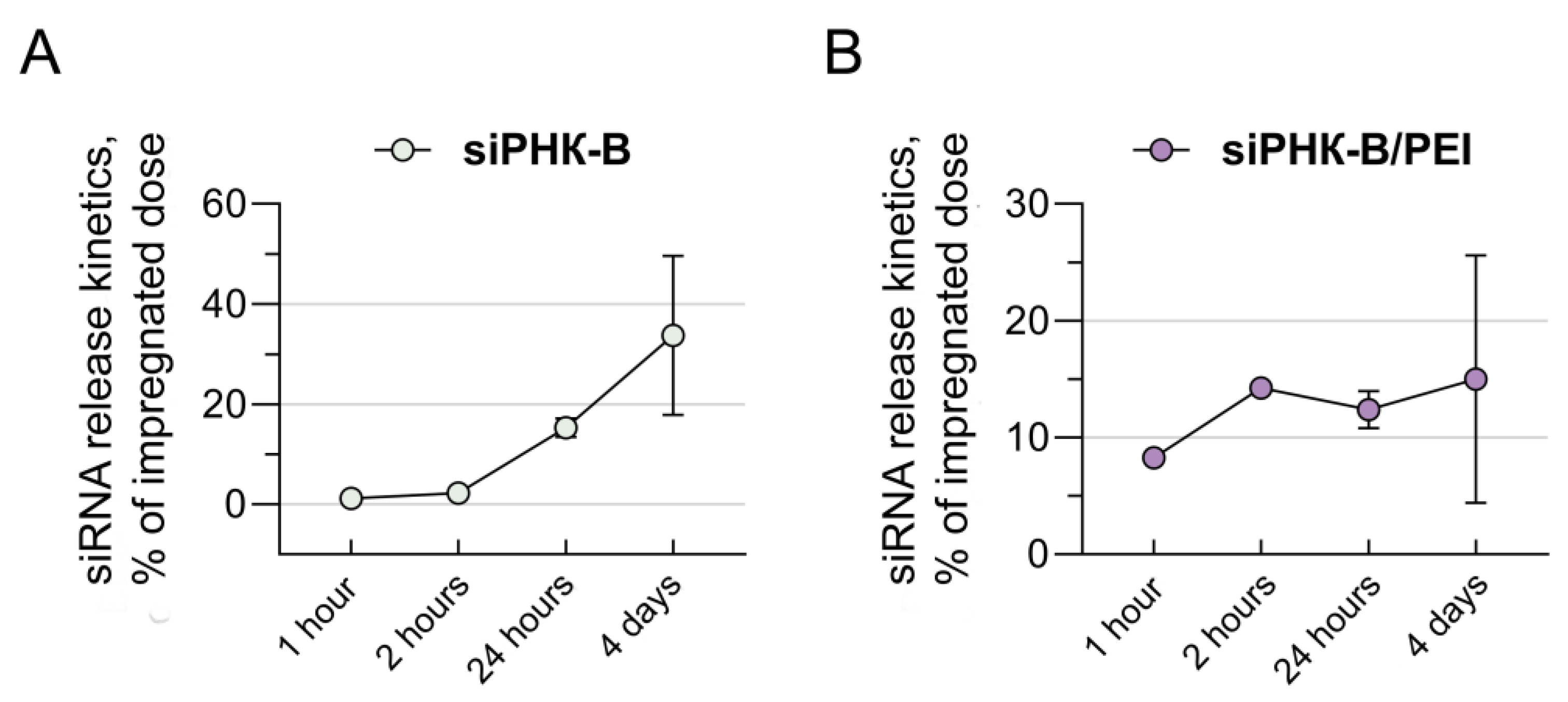
3.3. Investigation of the Kinetics of the Release of siRNA Molecules Impregnated in Collagen–Fibronectin Hydrogel
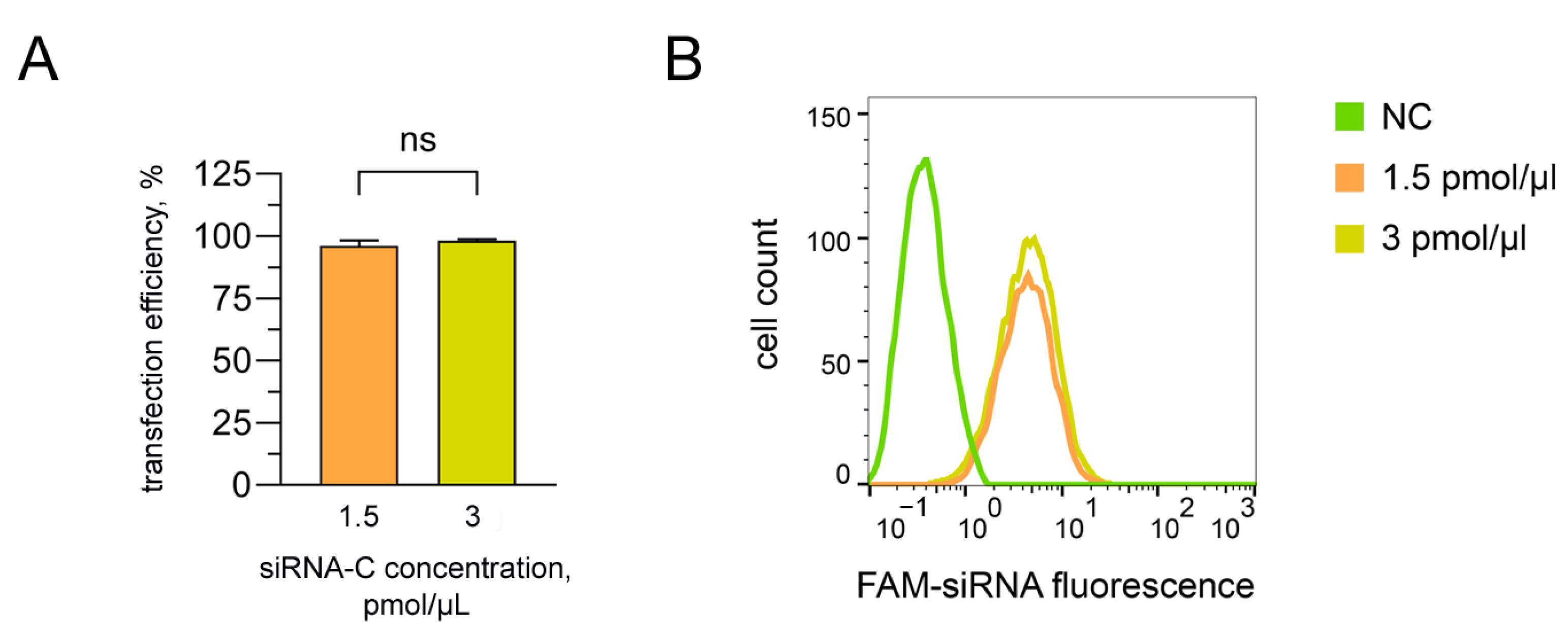
3.4. Assessment of the Transfection Efficiency of siRNA Molecules Impregnated in Collagen–Fibronectin Hydrogel
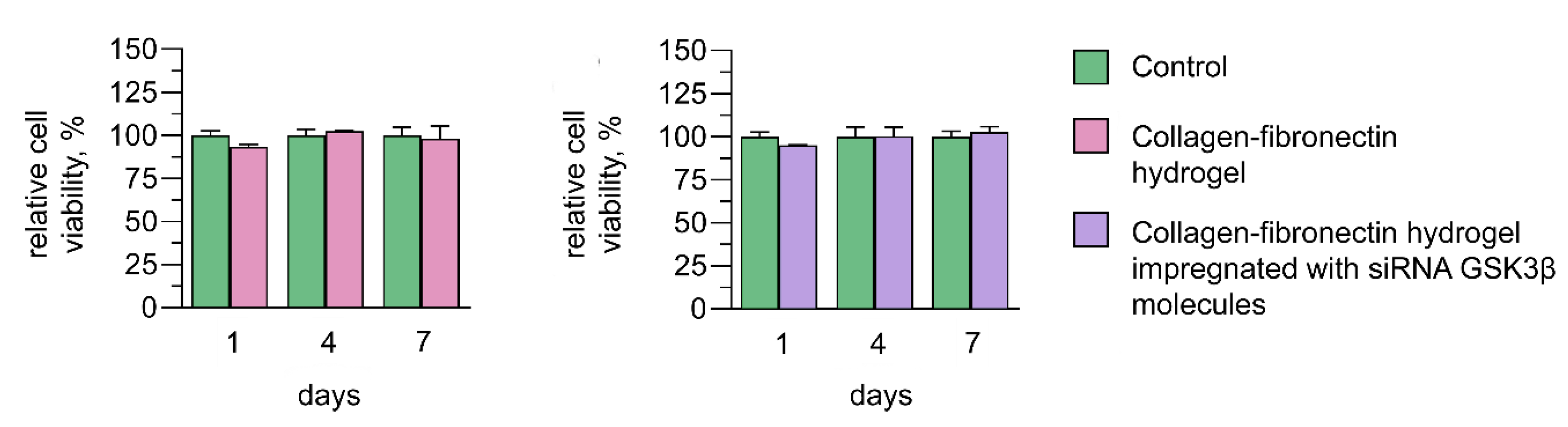
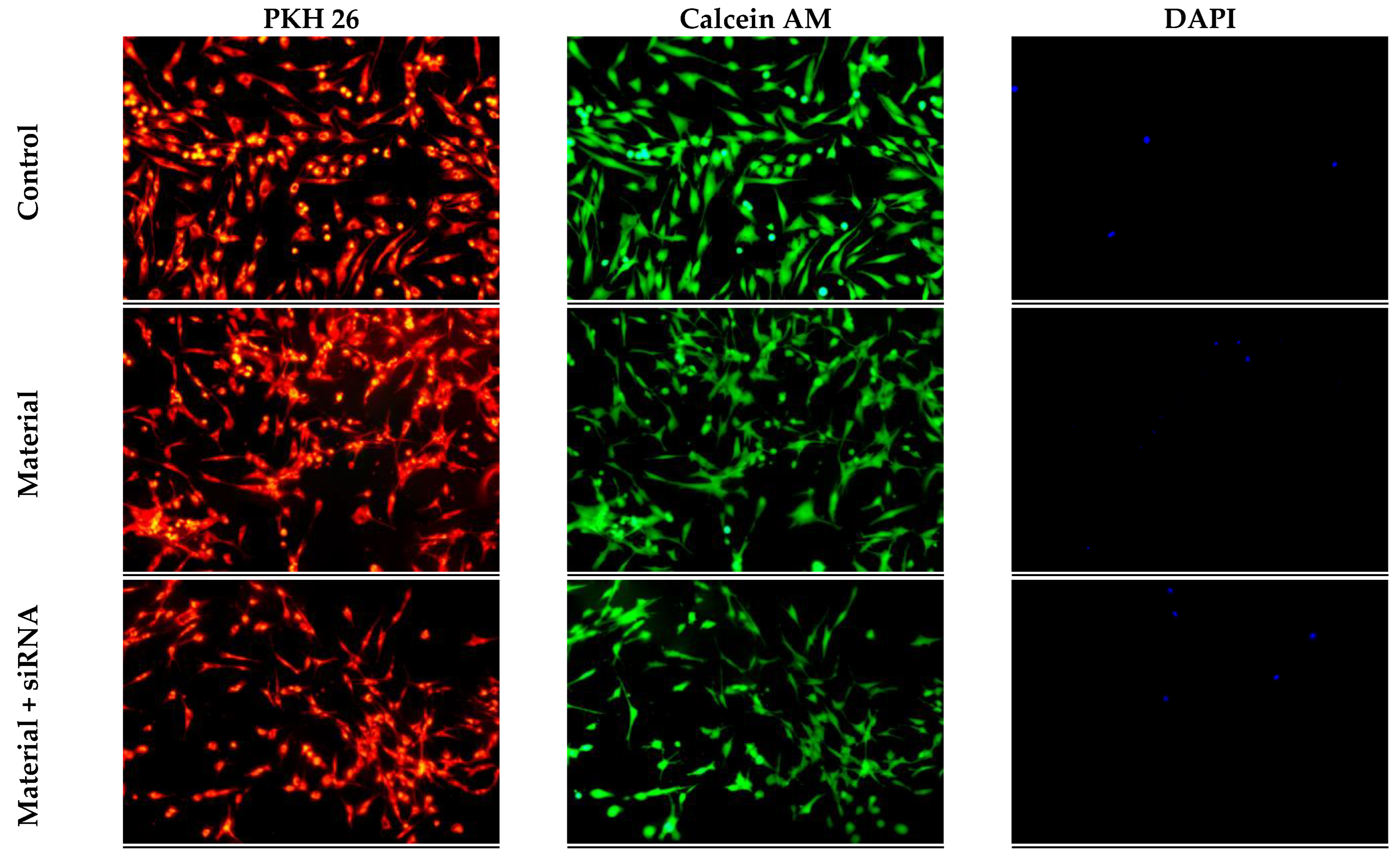
3.5. Cytotoxic Effect of Collagen–Fibronectin Hydrogel Impregnated with siRNA GSK3β Molecules
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
Appendix A
| Positive CD Markers of hAD-MSCs | Negative CD Markers of hAD-MSCs |
|---|---|
| CD 29+ (integrin β1), rabbit IgG, clone EP1041Y | CD 11b− PE, mouse IgG2B; clone 238446 |
| CD 44+, mouse IgG2a, clone F10-44-2 | CD 34− PE, mouse IgG1, clone QBEnd10 |
| CD 90+ (Thy1), mouse IgG1, clone AF-9 | CD 45− PE, mouse IgG1, clone 2D1 |
| CD73+ FAM, mouse IgG2B, clone 606112 | CD 79a− PE, mouse IgG1, clone 706931 |
| HLA-DR− PE, mouse IgG1, clone L203 |
| Name | Sequence in 5′ → 3′ Direction |
|---|---|
| ACTB forward | CCTGGAACCAGCACAATA |
| ACTB reverse | GGGCCGGACTAGTCATAC |
| ALPL forward | CTCGGAAGACACTCTGACCGT |
| ALPL reverse | TAGTCCACCATGGAGACATTCTCT |
| BMP-2 forward | ACTACCAGAAACGAGTGGGAA |
| BMP-2 reverse | GCATCTGTTCTCGGAAAACCT |
| GAPDH forward | GAAGGTGAAGGTCGGAGTACA |
| GAPDH reverse | TTCACACCCATGACGAGACAT |
| GSK3β forward | ACGGCACCCAAATATCAAACT |
| GSK3β reverse | AGCCAGAGGTGGATTACTTGA |
| OCN forward | CAGAGTCCAGCAAAGGTGCAG |
| OCN reverse | CTCCCAGCCATTGATACAGGT |
| OPN forward | GCCGAGGTGATAGTGTGGTT |
| OPN reverse | AACGGGGATGGCCTTGTATG |
| Runx2 forward | AGTGGACGACAGTCTGACTTT |
| Runx2 reverse | GGTGAATGCGGTAAGACTGGT |
References
- Fuentealba, L.C.; Eivers, E.; Ikeda, A.; Hurtado, C.; Kuroda, H.; Pera, E.M.; De Robertis, E.M. Integrating Patterning Signals: Wnt/GSK3 Regulates the Duration of the BMP/Smad1 Signal. Cell 2007, 131, 980–993. [Google Scholar] [CrossRef]
- Gao, C.; Xiao, G.; Hu, J. Regulation of Wnt/β-Catenin Signaling by Posttranslational Modifications. Cell Biosci. 2014, 4, 13. [Google Scholar] [CrossRef]
- Kugimiya, F.; Kawaguchi, H.; Ohba, S.; Kawamura, N.; Hirata, M.; Chikuda, H.; Azuma, Y.; Woodgett, J.R.; Nakamura, K.; Chung, U. GSK-3beta Controls Osteogenesis through Regulating Runx2 Activity. PLoS ONE 2007, 2, e837. [Google Scholar] [CrossRef]
- Kapinas, K.; Kessler, C.; Ricks, T.; Gronowicz, G.; Delany, A.M. MiR-29 Modulates Wnt Signaling in Human Osteoblasts through a Positive Feedback Loop. J. Biol. Chem. 2010, 285, 25221–25231. [Google Scholar] [CrossRef]
- Zhang, J.; Tu, Q.; Bonewald, L.F.; He, X.; Stein, G.; Lian, J.; Chen, J. Effects of MiR-335-5p in Modulating Osteogenic Differentiation by Specifically Downregulating Wnt Antagonist DKK1. J. Bone Miner. Res. 2011, 26, 1953–1963. [Google Scholar] [CrossRef]
- Hassan, M.Q.; Maeda, Y.; Taipaleenmaki, H.; Zhang, W.; Jafferji, M.; Gordon, J.A.R.; Li, Z.; Croce, C.M.; Van Wijnen, A.J.; Stein, J.L.; et al. MiR-218 Directs a Wnt Signaling Circuit to Promote Differentiation of Osteoblasts and Osteomimicry of Metastatic Cancer Cells. J. Biol. Chem. 2012, 287, 42084–42092. [Google Scholar] [CrossRef]
- Zhang, W.-B.; Zhong, W.-J.; Wang, L. A Signal-Amplification Circuit between MiR-218 and Wnt/β-Catenin Signal Promotes Human Adipose Tissue-Derived Stem Cells Osteogenic Differentiation. Bone 2014, 58, 59–66. [Google Scholar] [CrossRef]
- Vishal, M.; Vimalraj, S.; Ajeetha, R.; Gokulnath, M.; Keerthana, R.; He, Z.; Partridge, N.C.; Selvamurugan, N. MicroRNA-590-5p Stabilizes Runx2 by Targeting Smad7 During Osteoblast Differentiation: MICRORNA-590-5P STABILIZES RUNX2 BY TARGETING SMAD7. J. Cell. Physiol. 2017, 232, 371–380. [Google Scholar] [CrossRef]
- Tzeng, S.Y.; Hung, B.P.; Grayson, W.L.; Green, J.J. Cystamine-Terminated Poly(Beta-Amino Ester)s for SiRNA Delivery to Human Mesenchymal Stem Cells and Enhancement of Osteogenic Differentiation. Biomaterials 2012, 33, 8142–8151. [Google Scholar] [CrossRef]
- Wang, Z.; Xie, Q.; Yu, Z.; Zhou, H.; Huang, Y.; Bi, X.; Wang, Y.; Shi, W.; Sun, H.; Gu, P.; et al. A Regulatory Loop Containing MiR-26a, GSK3β and C/EBPα Regulates the Osteogenesis of Human Adipose-Derived Mesenchymal Stem Cells. Sci. Rep. 2015, 5, 15280. [Google Scholar] [CrossRef]
- Ghadakzadeh, S.; Mekhail, M.; Aoude, A.; Hamdy, R.; Tabrizian, M. Small Players Ruling the Hard Game: SiRNA in Bone Regeneration: SMALL PLAYERS RULING THE HARD GAME: SiRNA IN BONE REGENERATION. J. Bone Miner. Res. 2016, 31, 475–487. [Google Scholar] [CrossRef] [PubMed]
- Galitsyna, E.V.; Bukharova, T.B.; Vasilyev, A.V.; Goldshtein, D.V. MicroRNAs in the Regulation of Osteogenesis in Vitro and in Vivo: From Fundamental Mechanisms to Bone Diseases Pathogenesis. Genes Cells 2019, 14, 41–48. [Google Scholar] [CrossRef]
- Lam, J.K.W.; Chow, M.Y.T.; Zhang, Y.; Leung, S.W.S. SiRNA Versus MiRNA as Therapeutics for Gene Silencing. Mol. Ther.—Nucleic Acids 2015, 4, e252. [Google Scholar] [CrossRef]
- Yu, A.-M.; Jian, C.; Yu, A.H.; Tu, M.-J. RNA Therapy: Are We Using the Right Molecules? Pharmacol. Ther. 2019, 196, 91–104. [Google Scholar] [CrossRef]
- Saw, P.E.; Song, E.-W. SiRNA Therapeutics: A Clinical Reality. Sci. China Life Sci. 2020, 63, 485–500. [Google Scholar] [CrossRef]
- Robert, L.; Robert, B. Structural glycoproteins of connective tissue: Their role in morphogenesis and immunopathology. In Connective Tissues; Fricke, R., Hartmann, F., Eds.; Springer: Berlin/Heidelberg, Germany, 1974; pp. 240–256. [Google Scholar]
- Vyakhireva, J.V.; Filatova, A.Y.; Krivosheeva, I.A.; Skoblov, M.Y. siRNA-mediated gene silencing. Bull. RSMU 2017, 17–29. [Google Scholar] [CrossRef]
- Horova, V.; Hradilova, N.; Jelinkova, I.; Koc, M.; Svadlenka, J.; Brazina, J.; Klima, M.; Slavik, J.; Hyrslova Vaculova, A.; Andera, L. Inhibition of Vacuolar ATPase Attenuates the TRAIL-Induced Activation of Caspase-8 and Modulates the Trafficking of TRAIL Receptosomes. FEBS J. 2013, 280, 3436–3450. [Google Scholar] [CrossRef]
- Mosmann, T. Rapid Colorimetric Assay for Cellular Growth and Survival: Application to Proliferation and Cytotoxicity Assays. J. Immunol. Methods 1983, 65, 55–63. [Google Scholar] [CrossRef]
- Galitsyna, E.V.; Bukharova, T.B.; Buianova, A.A.; Davygora, K.S.; Goldshtein, D.V. Choice of an Effective System of Nonviral SiRNA Delivery to Multipotent Mesenchymal Stromal Cells. Appl. Biochem. Microbiol. 2021, 57, 823–827. [Google Scholar] [CrossRef]
- Weyand, B.; Dominici, M.; Hass, R.; Jacobs, R.; Kasper, C. (Eds.) Mesenchymal Stem Cells—Basics and Clinical Application I; Advances in Biochemical Engineering/Biotechnology; Springer: Berlin/Heidelberg, Germany, 2013; Volume 129. [Google Scholar] [CrossRef]
- Lv, H.; Zhang, S.; Wang, B.; Cui, S.; Yan, J. Toxicity of Cationic Lipids and Cationic Polymers in Gene Delivery. J. Control. Release 2006, 114, 100–109. [Google Scholar] [CrossRef]
- Shim, M.S.; Kwon, Y.J. Controlled Delivery of Plasmid DNA and SiRNA to Intracellular Targets Using Ketalized Polyethylenimine. Biomacromolecules 2008, 9, 444–455. [Google Scholar] [CrossRef] [PubMed]
- Han, X.; Fang, Q.; Yao, F.; Wang, X.; Wang, J.; Yang, S.; Shen, B.Q. The Heterogeneous Nature of Polyethylenimine-DNA Complex Formation Affects Transient Gene Expression. Cytotechnology 2009, 60, 63–75. [Google Scholar] [CrossRef]
- Kafil, V.; Omidi, Y. Cytotoxic Impacts of Linear and Branched Polyethylenimine Nanostructures in A431 Cells. BioImpacts 2011, 1, 23. [Google Scholar] [CrossRef]
- Lee, S.J.; Son, S.; Yhee, J.Y.; Choi, K.; Kwon, I.C.; Kim, S.H.; Kim, K. Structural Modification of SiRNA for Efficient Gene Silencing. Biotechnol. Adv. 2013, 31, 491–503. [Google Scholar] [CrossRef] [PubMed]
- Karsenty, G.; Wagner, E.F. Reaching a Genetic and Molecular Understanding of Skeletal Development. Dev. Cell 2002, 2, 389–406. [Google Scholar] [CrossRef] [PubMed]
- Guidotti, S.; Minguzzi, M.; Platano, D.; Santi, S.; Trisolino, G.; Filardo, G.; Mariani, E.; Borzì, R.M. Glycogen Synthase Kinase-3β Inhibition Links Mitochondrial Dysfunction, Extracellular Matrix Remodelling and Terminal Differentiation in Chondrocytes. Sci. Rep. 2017, 7, 12059. [Google Scholar] [CrossRef] [PubMed]
- Pilquil, C.; Alvandi, Z.; Opas, M. Calreticulin Regulates a Switch between Osteoblast and Chondrocyte Lineages Derived from Murine Embryonic Stem Cells. J. Biol. Chem. 2020, 295, 6861–6875. [Google Scholar] [CrossRef]
- Bali, S.K.; Bryce, D.; Prein, C.; Woodgett, J.R.; Beier, F. Glycogen Synthase Kinase 3 Alpha/Beta Deletion Induces Precocious Growth Plate Remodeling in Mice. J. Mol. Med. 2021, 99, 831–844. [Google Scholar] [CrossRef]
- Nelson, E.R.; Levi, B.; Sorkin, M.; James, A.W.; Liu, K.J.; Quarto, N.; Longaker, M.T. Role of GSK-3β in the Osteogenic Differentiation of Palatal Mesenchyme. PLoS ONE 2011, 6, e25847. [Google Scholar] [CrossRef]
- Clément-Lacroix, P.; Ai, M.; Morvan, F.; Roman-Roman, S.; Vayssière, B.; Belleville, C.; Estrera, K.; Warman, M.L.; Baron, R.; Rawadi, G. Lrp5-Independent Activation of Wnt Signaling by Lithium Chloride Increases Bone Formation and Bone Mass in Mice. Proc. Natl. Acad. Sci. USA 2005, 102, 17406–17411. [Google Scholar] [CrossRef]
- Gambardella, A.; Nagaraju, C.K.; O’Shea, P.J.; Mohanty, S.T.; Kottam, L.; Pilling, J.; Sullivan, M.; Djerbi, M.; Koopmann, W.; Croucher, P.I.; et al. Glycogen Synthase Kinase-3α/β Inhibition Promotes in Vivo Amplification of Endogenous Mesenchymal Progenitors with Osteogenic and Adipogenic Potential and Their Differentiation to the Osteogenic Lineage. J. Bone Miner. Res. 2011, 26, 811–821. [Google Scholar] [CrossRef]
- Marsell, R.; Sisask, G.; Nilsson, Y.; Sundgren-Andersson, A.K.; Andersson, U.; Larsson, S.; Nilsson, O.; Ljunggren, Ö.; Jonsson, K.B. GSK-3 Inhibition by an Orally Active Small Molecule Increases Bone Mass in Rats. Bone 2012, 50, 619–627. [Google Scholar] [CrossRef] [PubMed]
- Sisask, G.; Marsell, R.; Sundgren-Andersson, A.; Larsson, S.; Nilsson, O.; Ljunggren, Ö.; Jonsson, K.B. Rats Treated with AZD2858, a GSK3 Inhibitor, Heal Fractures Rapidly without Endochondral Bone Formation. Bone 2013, 54, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Gilmour, P.S.; O’Shea, P.J.; Fagura, M.; Pilling, J.E.; Sanganee, H.; Wada, H.; Courtney, P.F.; Kavanagh, S.; Hall, P.A.; Escott, K.J. Human Stem Cell Osteoblastogenesis Mediated by Novel Glycogen Synthase Kinase 3 Inhibitors Induces Bone Formation and a Unique Bone Turnover Biomarker Profile in Rats. Toxicol. Appl. Pharmacol. 2013, 272, 399–407. [Google Scholar] [CrossRef]
- Cuzzocrea, S.; Mazzon, E.; Di Paola, R.; Muià, C.; Crisafulli, C.; Dugo, L.; Collin, M.; Britti, D.; Caputi, A.P.; Thiemermann, C. Glycogen Synthase Kinase-3β Inhibition Attenuates the Degree of Arthritis Caused by Type II Collagen in the Mouse. Clin. Immunol. 2006, 120, 57–67. [Google Scholar] [CrossRef]
- Dawood, A.F.; Younes, S.; Alzamil, N.M.; Alradini, F.A.; Saja, M.F. Inhibition of Glycogen Synthase Kinase-3β Protects against Collagen Type II-Induced Arthritis Associated with the Inhibition of MiR155/24 and Inflammation and Upregulation of Apoptosis in Rats. Arch. Physiol. Biochem. 2022, 128, 679–687. [Google Scholar] [CrossRef]
- Somaiah, C.; Kumar, A.; Mawrie, D.; Sharma, A.; Patil, S.D.; Bhattacharyya, J.; Swaminathan, R.; Jaganathan, B.G. Collagen Promotes Higher Adhesion, Survival and Proliferation of Mesenchymal Stem Cells. PLoS ONE 2015, 10, e0145068. [Google Scholar] [CrossRef]
- Sakkas, A.; Wilde, F.; Heufelder, M.; Winter, K.; Schramm, A. Autogenous Bone Grafts in Oral Implantology-Is It Still a “Gold Standard”? A Consecutive Review of 279 Patients with 456 Clinical Procedures. Int. J. Implant. Dent. 2017, 3, 23. [Google Scholar] [CrossRef]
- Galindo-Moreno, P.; Avila, G.; Fernández-Barbero, J.E.; Mesa, F.; O’Valle-Ravassa, F.; Wang, H.-L. Clinical and Histologic Comparison of Two Different Composite Grafts for Sinus Augmentation: A Pilot Clinical Trial. Clin. Oral Implant. Res. 2008, 19, 755–759. [Google Scholar] [CrossRef]
- Parisi, L.; Toffoli, A.; Ghezzi, B.; Mozzoni, B.; Lumetti, S.; Macaluso, G.M. A Glance on the Role of Fibronectin in Controlling Cell Response at Biomaterial Interface. Jpn. Dent. Sci. Rev. 2020, 56, 50–55. [Google Scholar] [CrossRef]
- Sarinnaphakorn, L.; Di Silvio, L. Surface-Modified Titanium to Enhance Osseointegration in Dental Implants. In Cellular Response to Biomaterials; Elsevier: Amsterdam, The Netherlands, 2009; pp. 572–588. [Google Scholar] [CrossRef]
- Kazimierczak, P.; Przekora, A. Osteoconductive and Osteoinductive Surface Modifications of Biomaterials for Bone Regeneration: A Concise Review. Coatings 2020, 10, 971. [Google Scholar] [CrossRef]
- Biggs, M.J.P.; Dalby, M.J. Focal Adhesions in Osteoneogenesis. Proc. Inst. Mech. Eng. H 2010, 224, 1441–1453. [Google Scholar] [CrossRef] [PubMed]
- Kowalczewski, C.J.; Saul, J.M. Surface-Mediated Delivery of SiRNA from Fibrin Hydrogels for Knockdown of the BMP-2 Binding Antagonist Noggin. Acta Biomater. 2015, 25, 109–120. [Google Scholar] [CrossRef] [PubMed]
- Huynh, C.T.; Nguyen, M.K.; Naris, M.; Tonga, G.Y.; Rotello, V.M.; Alsberg, E. Light-Triggered RNA Release and Induction of HMSC Osteogenesis via Photodegradable, Dual-Crosslinked Hydrogels. Nanomedicine 2016, 11, 1535–1550. [Google Scholar] [CrossRef]
- Wang, Y.; Malcolm, D.W.; Benoit, D.S.W. Controlled and Sustained Delivery of SiRNA/NPs from Hydrogels Expedites Bone Fracture Healing. Biomaterials 2017, 139, 127–138. [Google Scholar] [CrossRef]


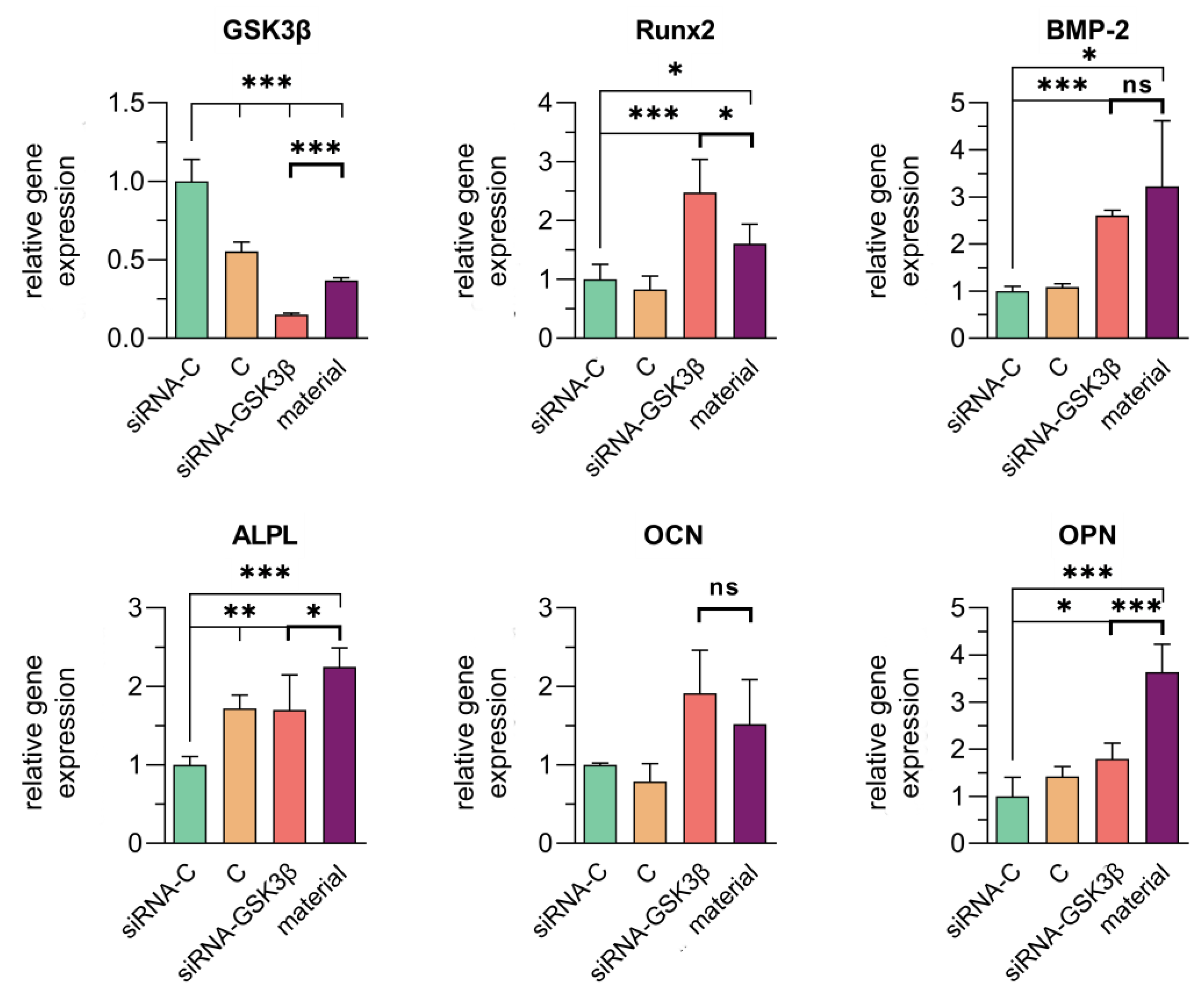
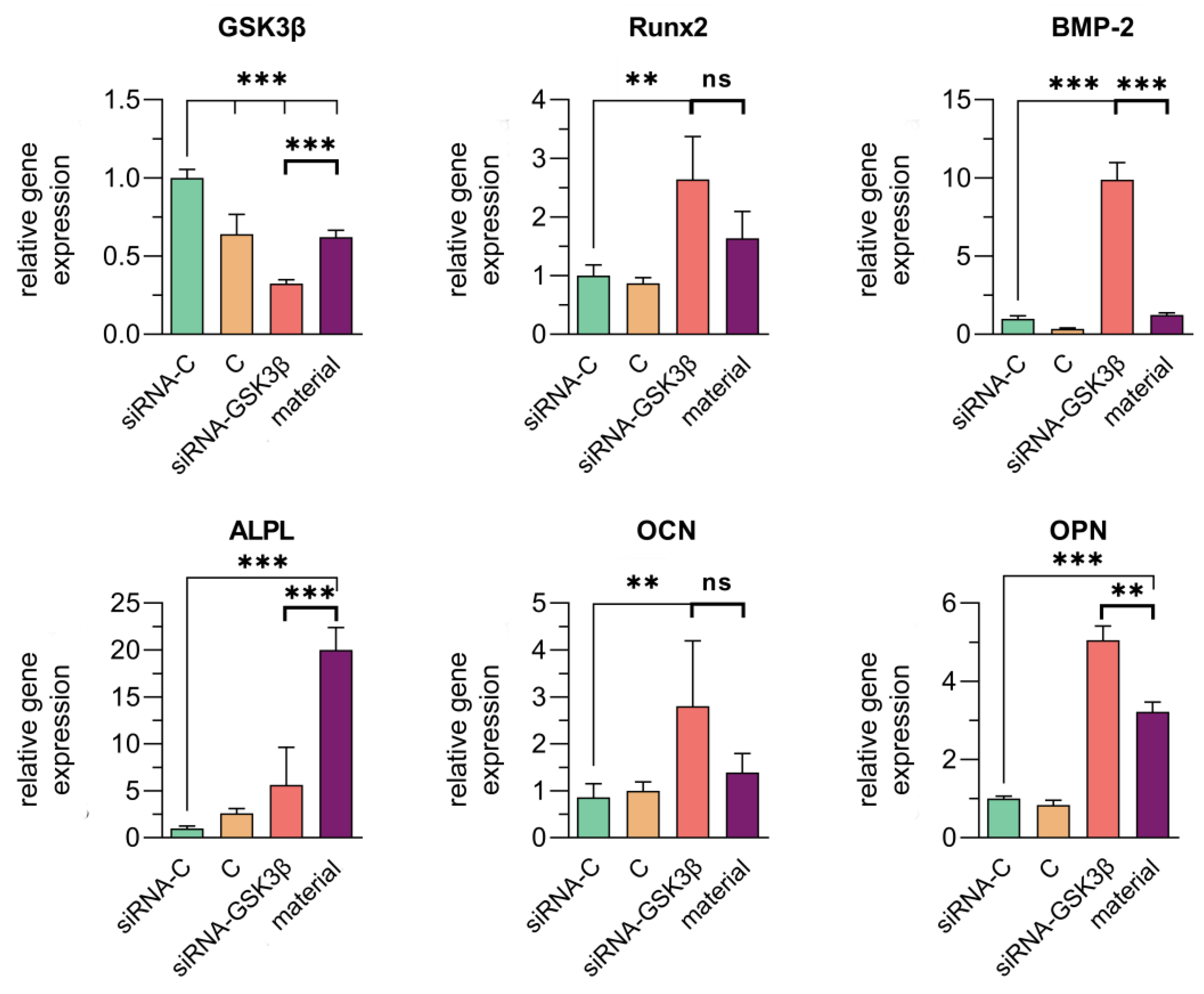
| Name | Strand | Sequence in 5′ → 3′ Direction |
|---|---|---|
| Control siRNA-FAM (siRNA-C) | Sense | FAM-AGGUCGAACUACGGGUCAAdTdC |
| Antisense | FAM-UUGACCCGUAGUUCGACCUdAdG | |
| Control siRNA-biotin (siRNA-B) | Sense | biotin-AGGUCGAACUACGGGUCAAdTdC |
| Antisense | biotin-UUGACCCGUAGUUCGACCUdAdG | |
| siRNA GSK3β | Sense | CCAACAAGGGAGCAAAUCAGAdTdT |
| Antisense | UCUGAUUUGCUCCCUUGUUGGdTdT |
| Reagent | Quantities | |
|---|---|---|
| Volume of siRNA, µg | 0.67 | 0.8 |
| Amount of siRNA, pmol | 50 | 60 |
| Amount of PEI at a ratio of 1:3, µg | 2 | 2.4 |
| Volume of DPBS for dilution of siRNA, μL | up to 25 | up to 25 |
| Volume of DPBS for dilution of PEI, μL | up to 25 | up to 25 |
| Positive and Negative CD Markers of hAD-MSCs | The Relative Number of Cells in the Culture |
|---|---|
| CD29+ (integrin β1) | 99.65% |
| CD44+ | 99.93% |
| CD73+ | 75.4% |
| CD90+ | 80.38% |
| CD11b−, CD34−, CD45−, CD79a−, HLA-DR− | 0.32% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Galitsyna, E.V.; Buianova, A.A.; Kozhukhov, V.I.; Domogatsky, S.P.; Bukharova, T.B.; Goldshtein, D.V. Cytocompatibility and Osteoinductive Properties of Collagen-Fibronectin Hydrogel Impregnated with siRNA Targeting Glycogen Synthase Kinase 3β: In Vitro Study. Biomedicines 2023, 11, 2363. https://doi.org/10.3390/biomedicines11092363
Galitsyna EV, Buianova AA, Kozhukhov VI, Domogatsky SP, Bukharova TB, Goldshtein DV. Cytocompatibility and Osteoinductive Properties of Collagen-Fibronectin Hydrogel Impregnated with siRNA Targeting Glycogen Synthase Kinase 3β: In Vitro Study. Biomedicines. 2023; 11(9):2363. https://doi.org/10.3390/biomedicines11092363
Chicago/Turabian StyleGalitsyna, Elena V., Anastasiia A. Buianova, Vadim I. Kozhukhov, Sergey P. Domogatsky, Tatiana B. Bukharova, and Dmitriy V. Goldshtein. 2023. "Cytocompatibility and Osteoinductive Properties of Collagen-Fibronectin Hydrogel Impregnated with siRNA Targeting Glycogen Synthase Kinase 3β: In Vitro Study" Biomedicines 11, no. 9: 2363. https://doi.org/10.3390/biomedicines11092363







