1. Introduction
Huntington’s disease (HD), an autosomal dominant monogenic neurodegenerative disease, was first described in 1872 by George Huntington, who reported an inherited choreiform disorder [
1] resulting from the expansion of cytosine, adenine, and guanine (CAG) tract in the
IT15 gene (Huntingtin gene,
HTT), located in the region 4p16.3. This protein encodes a polyglutamine, widely expressed in various tissues of the body and necessary for normal development [
2]. One of these processes occurs during the 13th week of pregnancy, known as “interkinetic nuclear migration”, and is essential for developing corticostriatal projections [
3].
Molecularly, normal polyglutamine differs from the mutated form in the length of the CAG repeats—those greater than 35 code for pathogenic polyglutamine.
At the functional level, under normal conditions, this protein forms aggregates that could exert a neuroprotective function. The mutated polyglutamine also produces these same aggregates, which lead to dysfunction that affects multiple cellular pathways, including DNA replication and repair, transcription, splicing, and mitochondrial function, with the striatum and the cortex being the storage location. Even though all of these mechanisms have been described, they remain poorly understood [
4].
HD manifests clinically with characteristics of motor dysfunction such as chorea, dystonia, bradykinesia, and rigidity, in addition to cognitive and psychiatric conditions [
5]. Phenotypically, the number of CAG repeats that the carrier has translated into the possible age of onset of the disease and the severity of symptoms in the function of their quantity. It affects both sexes equally, and the risk of passing the gene to the next generation is 50% [
6]. It generally manifests between 30 and 50 years of age, although an earlier form occurs before the age of 20 [
7] and has been associated with expansion in CAG repeats greater than 60 [
8].
The worldwide prevalence of HD is 2.71 per 100,000 inhabitants [
9]. In Colombia, orphan diseases are defined as those that are chronically debilitating, serious, life-threatening, and with a prevalence of less than 1 per 5000 individuals. HD is endemic in a municipality located 40 km from the capital of the Department of Atlántico on the Colombian Caribbean coast with a total population of 17,487 inhabitants, according to the municipality’s official website (
http://www.juandeacosta-atlantico.gov.co/, accessed on 12 January 2024). It stands out for being considered the second place with the highest number of patients affected by this disease worldwide. The estimated prevalence of HD in the Department of Atlántico is 0.2 per 10,000 inhabitants; however, in this municipality, it rises to 9.7 per 10,000 inhabitants [
10].
Although the diagnosis of this disease is based on the onset of several types of abnormal movements (such as chorea, dystonia, fine motor impairment, dysarthria, dysphagia, and gait disorders) [
8,
11,
12], the structural changes in the brain caused by volume loss, and defects in the corticstriatal network that cause dysfunction and degeneration of the striatum [
3] can occur much earlier than motor symptoms [
13]. They are a product of deposits of mutated
HTT, which appear from very early stages of embryonic development. Studies have described these alterations in human and murine brain tissue from week 13 of pregnancy [
3]. These tissues showed clear abnormalities in the developing cortex, including mislocalization of mutant
HTT and junctional complex proteins, defects in neuroprogenitor cell polarity and differentiation, abnormal ciliogenesis, and changes in mitosis and cell cycle progression. However, the path to degeneration is complex and involves pathogenic and compensatory mechanisms, so the estimated period between these changes in neurodevelopment (and their subsequent alterations and motor manifestations) cannot be established [
14].
The morphological changes brought about by this disease can be observed through neuroimaging techniques, such as structural magnetic resonance imaging (MRI) and brain volumetry, which allow for visualization and quantification of changes in the different regions of the brain [
15]. Structural MRI has been widely used in HD investigation to determine volumetric changes in both manifest and premanifest patients [
15]. Through this, different brain regions have been identified that have potential as biomarkers of progression in HD [
16].
Until this study, these brain structural volumetric analyses used tools such as parametric statistical mapping and geometric progression models [
17,
18]. Currently, analysis using artificial intelligence (AI) is considered a pioneering, novel, and future-oriented tool in this field.
The advent of AI and the application of deep learning algorithms in neuroimaging have made it possible to detect complex and subtle patterns by training with extensive datasets of brain images specific to the Colombian population. With this, a precise analysis of the brain volumes of this population was conducted. These datasets were carefully curated and validated to ensure representativeness and relevance in the local context. Through this training, AI can identify relevant patterns and characteristics, especially in the premanifest HD population. During the volumetric process, AI performs automatic segmentations of specific brain structures with high precision, facilitating the quantification of volumetric changes.
AI’s effectiveness in brain volumetry lies in its ability to rapidly process large datasets, enhance accuracy in identifying regions of interest, and ultimately serve as a valuable tool for early diagnosis and research in neurological diseases. This study proposes that the application of AI in neuroimaging could serve as an alternative method for identifying structural biomarkers long before patients present clinical symptoms, potentially up to 10 years earlier in premanifest patients. This capability is crucial for the early detection and intervention in HD, potentially altering the disease trajectory [
19,
20].
The integration of AI in neuroimaging is not only innovative but also supported by a growing body of literature that highlights its advantages in early disease detection and the analysis of complex datasets [
21,
22]. These studies underscore the transformative potential of AI in enhancing our understanding of HD and improving patient outcomes through earlier and more accurate diagnoses [
19].
3. Results
The sociodemographic characterization of the study participants is arranged by gender, age, and triplet expansion (CAG) (
Table 2). In terms of gender, 67.5% of participants are women, while 32.5% are men. Regarding age, the mean is represented according to the expansion number (34.53 [≤26], 27.5 [
25,
26,
27,
28,
29,
30,
31,
32,
33], and 39.25 [≥40], with the latter being more predominant). As for triplet expansion, 75% of participants had an expansion less than or equal to 26, indicating a normal state, while 20% had an expansion greater than 40, suggesting complete penetrance of the disease. Furthermore, intermediate cases (27–35) were identified in 5% of participants. One case of error (Fail) and two cases with data not available (NA) were recorded. After breaking down the data by gender, women double the population in our study.
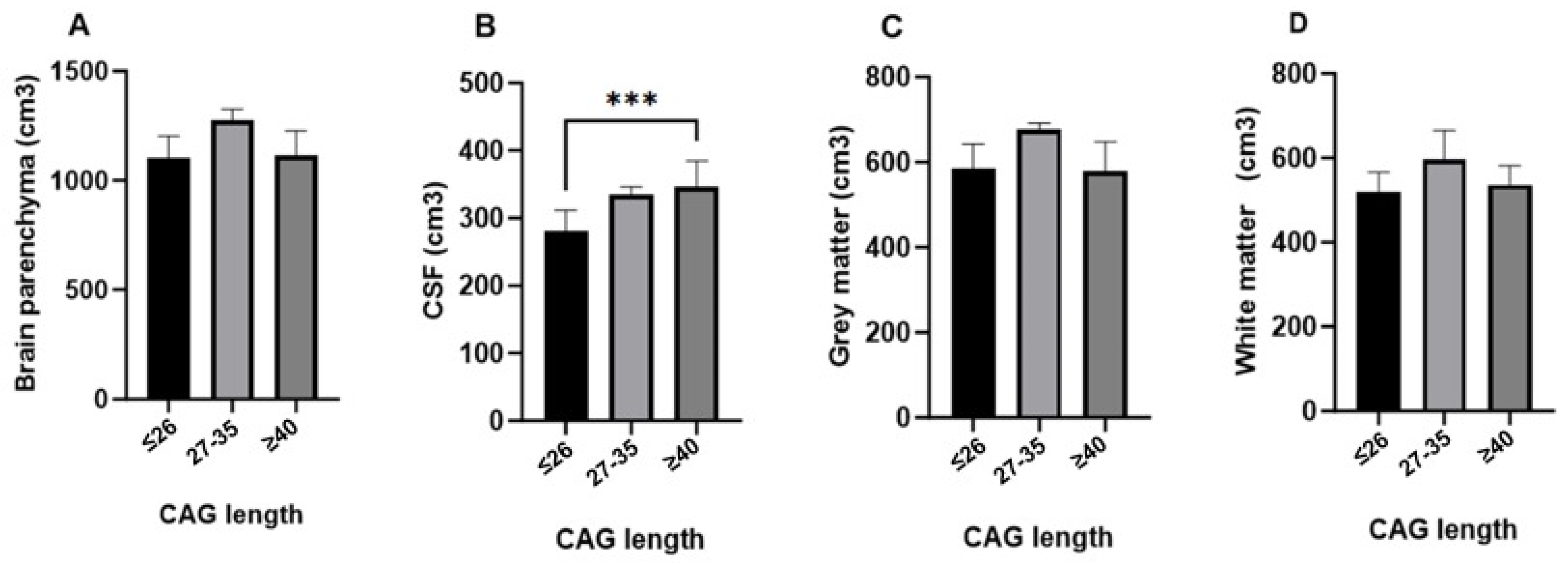
The analysis of brain parenchyma volume did not reveal any significant differences across the CAG groups (Kruskal–Wallis H = 4.526, p = 0.104). The mean parenchyma volume was 1115.14 cm3 in the CAG ≤ 26 group, with no significant differences observed in the CAG 27–35 and CAG ≥ 40 groups.
In contrast, a significant increase in cerebrospinal fluid (CSF) volume was observed in the CAG ≥ 40 group (Kruskal–Wallis H = 13.216, p = 0.001), suggesting possible brain atrophy in individuals with higher CAG repeat lengths. The mean CSF volume in the CAG ≤ 26 group was 294.92 cm3, with the CAG ≥ 40 group showing the highest mean rank. Regarding gray matter, the analysis showed no significant differences in volume between the groups (Kruskal–Wallis H = 4.366, p = 0.113). The CAG ≤ 26 group had a mean volume of 589.78 cm3, with similar trends observed across the other groups.
Finally, no significant differences were found in total white matter volume or in the volumes of the left and right hemispheres across the CAG groups (Kruskal–Wallis H values ranging from 2.885 to 4.209,
p > 0.05). The mean global white matter volume was 525.36 cm
3 in the CAG ≤ 26 group (
Table 3).
The analysis of brain volumes according to CAG triplet expansion reveals distinct patterns across different brain structures, as illustrated in
Figure 1. Brain parenchyma, gray matter, and white matter volumes did not show statistically significant differences between the groups (≤26, 27–35, and ≥40 CAG repeats), although there was a trend of slightly higher volumes in the intermediate group (27–35) compared to the other groups. In contrast, cerebrospinal fluid (CSF) volume showed a significant increase in the full penetrance group (≥40 CAG repeats) compared to the normal repeat group (≤26 CAG repeats), with a highly significant
p value (***
p < 0.00001). This finding suggests that greater CAG repeat lengths are associated with increased CSF volume, potentially reflecting brain atrophy. These results indicate a selective impact of CAG triplet expansion on CSF volume, while other brain structures remain relatively unaffected (
Figure 1).
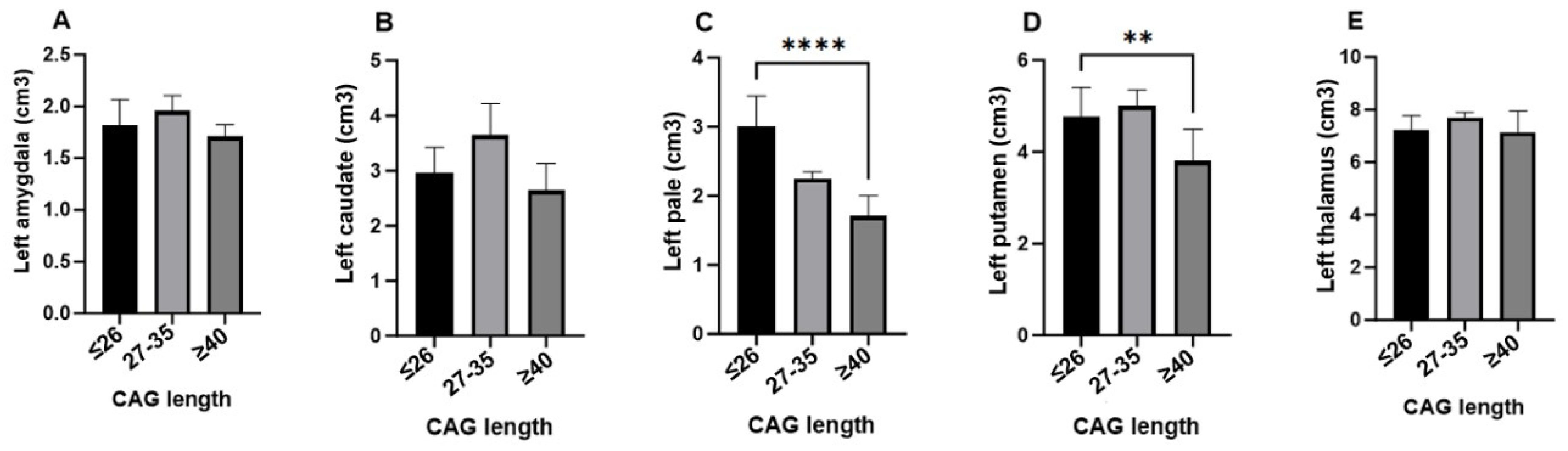
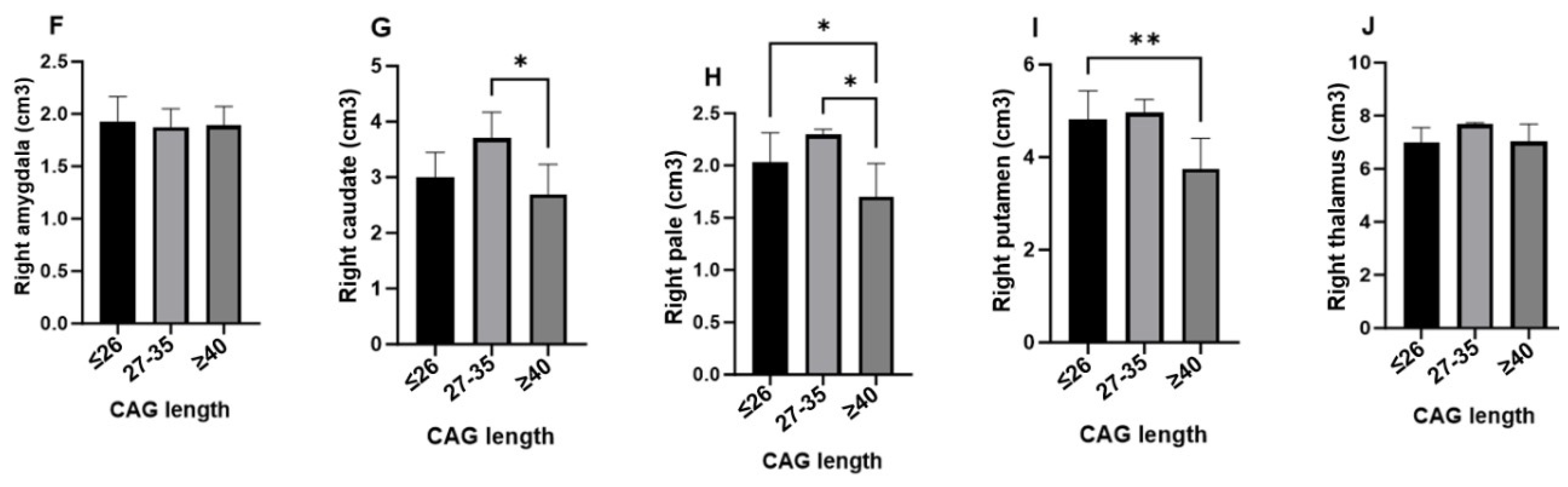
The analysis of subcortical brain structure volumes in relation to CAG triplet expansion reveals significant differences in specific brain regions in individuals at risk of developing Huntington’s disease. Notably, the volumes of the left pallidum (
p = 0.030) and right pallidum (
p = 0.018), as well as the left putamen (
p = 0.011) and right putamen (
p = 0.007), were significantly reduced in the full penetrance group (≥40 CAG repeats) compared to the other groups, suggesting that a higher number of CAG repeats significantly influences the atrophy of these structures. In contrast, no statistically significant differences were observed in the volumes of the left and right amygdala, left and right caudate, and left and right thalamus (
p > 0.05), indicating that these structures are not significantly affected by CAG triplet expansion in the premanifest stages of the disease. These findings highlight the selective impact of CAG triplet expansion on subcortical structures, with the pallidum and putamen showing greater susceptibility to early degeneration in Huntington’s disease (
Table 4).
Figure 2 illustrates the impact of CAG triplet expansion on the volumes of subcortical structures in individuals at risk of developing Huntington’s disease, categorized into three groups: ≤26 (normal), 27–35 (intermediate), and >40 (full penetrance). Significant reductions in the volumes of several subcortical structures were observed, particularly in the full penetrance group.
On the left side, subcortical structures, except for the thalamus, showed marked atrophy in the full penetrance group, suggesting a specific vulnerability to CAG triplet expansion in key regions such as the pallidum and putamen. On the right side, significant volume reductions were also observed in the caudate nucleus, globus pallidus, and putamen, consistent with the striatal atrophy characteristic of the disease. However, the right amygdala and thalamus-maintained volumes were comparable to those of the normal and intermediate groups, indicating possible preservation during the early stages of the disease.
These findings highlight the asymmetric and region-specific nature of brain atrophy in Huntington’s disease, with more pronounced effects on subcortical structures involved in motor control and cognitive processes, providing a deeper understanding of neurodegeneration in this disease.
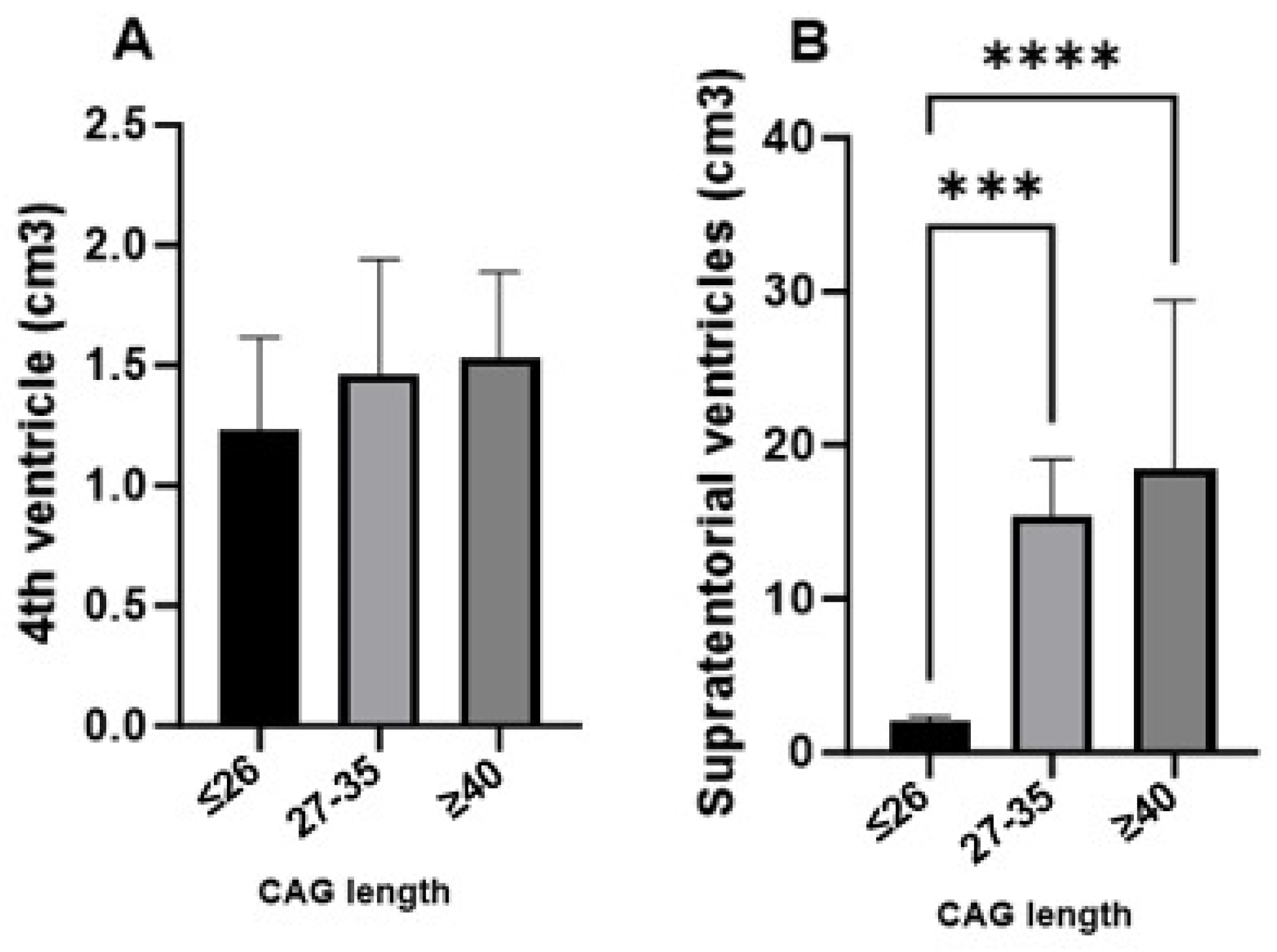
Table 5 highlights the impact of CAG triplet expansion on the volumes of the ventricular system, specifically focusing on the 4th ventricle and supratentorial ventricles in individuals at risk of developing Huntington’s disease. Participants were divided into three groups based on the number of CAG repeats: ≤26 (normal), 27–35 (intermediate), and ≥40 (full penetrance).
For the 4th ventricle, no statistically significant differences in volume were observed across the CAG groups (Kruskal–Wallis H = 3.245, p = 0.197), indicating that this structure remains relatively unaffected by CAG repeat expansion. The mean volume for the 4th ventricle was 1.2939 cm3 in the normal group, with higher average ranks observed in the intermediate and full penetrance groups.
In contrast, the analysis of the supratentorial ventricles revealed a significant increase in volume in the full penetrance group (Kruskal–Wallis H = 11.765, p = 0.003). The mean volume in the c group was 11.0939 cm3, with the highest volumes observed in the intermediate and full penetrance groups. This suggests that supratentorial ventricular enlargement is associated with increased CAG repeat lengths, likely reflecting the underlying brain atrophy characteristic of Huntington’s disease.
These findings indicate a differential impact of CAG expansion on the ventricular system, with significant enlargement of the supratentorial ventricles in individuals with higher CAG repeat numbers, while the 4th ventricle remains less affected. This pattern of ventricular dilation aligns with the progressive neurodegeneration seen in Huntington’s disease and may serve as an early marker of disease progression.
Figure 3 compares the volumes of the 4th ventricle (A) and supratentorial ventricles (B) across three groups based on CAG triplet expansion: ≤26 (normal), 27–35 (intermediate), and ≥40 (full penetrance). A significant finding from this analysis is the marked increase in ventricular system volume, both supratentorial and infratentorial, in the full penetrance group. This substantial enlargement, particularly evident in the supratentorial ventricles, is statistically significant, as indicated by the
p values (***
p < 0.00001, ****
p < 0.000001). These results suggest that individuals with higher CAG repeat lengths experience pronounced ventricular dilation, reflecting the extent of neurodegeneration associated with Huntington’s disease.
The results presented in
Table 6 indicate that CAG triplet expansion does not reveal a statistically significant impact on the volumes of infratentorial structures, including cerebellar white and gray matter and the brainstem, in individuals at risk for developing Huntington’s disease. Specifically, variations in mean volumes are observed among three groups (≤26, 27–35, and ≥40 repeats), with those possessing a higher number of repeats showing a trend toward reduced volumes, particularly in cerebellar gray matter. However, none of these differences reach statistical significance, with
p values exceeding 0.05 in all cases. These findings suggest that while there are fluctuations in the volumes of these brain structures, CAG triplet expansion does not have a significant effect on them within the analyzed cohort.
Figure 4 illustrates the comparison of infratentorial structure volumes across three groups of individuals categorized by CAG triplet expansion: ≤26 (normal), 27–35 (intermediate), and ≥40 (full penetrance). A significant finding from this analysis is the marked increase in left and right cerebellar gray matter in the intermediate group. The statistical significance of the differences observed between the groups is indicated by
p values, where *
p < 0.002 and **
p < 0.0002, highlighting significant differences in specific infratentorial structures associated with the extent of CAG triplet expansion.
The results presented in
Table 7 indicate that CAG triplet expansion does not have a statistically significant impact on the volumes of various cortical areas, including the frontal, insular, occipital, parietal, and temporal cortices, as well as the hippocampus, in individuals at risk of developing Huntington’s disease. Although variations in mean volumes were observed among groups with different numbers of CAG repeats (≤26, 27–35, and ≥40), these differences did not reach statistical significance in any of the regions analyzed. Specifically, in the frontal, parietal, and temporal cortices, as well as in the hippocampal regions, the
p values exceeded the threshold for significance in all cases, suggesting that, despite fluctuations in volumes, CAG triplet expansion does not have a significant effect on these cortical and hippocampal structures within the studied sample (
Table 7).
Figure 5 presents a comparative analysis of the volumes of various cortical areas and the hippocampus in individuals with different CAG triplet expansions, categorized into three groups: normal (≤26 repeats), intermediate (27–35 repeats), and full penetrance (≥40 repeats). The results reveal a significant increase in the volumes of the frontal, insular, occipital, parietal, and temporal cortices in the intermediate group compared to the normal and full penetrance groups. However, no statistically significant differences were observed in the volumes of the hippocampus, both left and right, among the three groups. These findings indicate that while CAG triplet expansion is associated with variations in cortical volumes, the hippocampus remains relatively stable regardless of the number of CAG triplet repeats.
Scheme 1 displays brain volumetry results obtained through nuclear magnetic resonance imaging in individuals with varying lengths of CAG repeats. Panel A shows the volumetric report of a patient with more than 45 CAG repeats, highlighting potential atrophic areas (indicated in blue) within the cerebral cortex, as well as regions requiring further examination (marked in orange). These results reveal a significant reduction in cortical volume and a more pronounced appearance of atrophic regions compared to Panel B.
Panel B illustrates the volumetric report of a patient with 16 CAG repeats, also identifying potentially atrophic areas (in blue) and zones for additional review (in orange) but to a lesser extent than in the patient with a higher CAG repeat expansion. The difference in the extent of atrophy between the two patients suggests a direct correlation between the number of CAG repeats and the severity of structural brain changes.
These findings support the hypothesis that a higher CAG repeat load is associated with increased cerebral atrophy and variations in brain volume, underscoring the importance of early and detailed evaluations in individuals at high risk for neurodegenerative diseases. Identifying atrophic areas and regions for further review may be crucial for implementing effective intervention and monitoring strategies in disease progression.
4. Discussion
This study explores the association between the number of triplet repeats and the volume of various brain structures in premanifest HD patients. This phase is characterized by the absence of notable motor symptoms, although underlying brain changes may already exist, as well as symptoms related to cognitive dysfunction and neuropsychiatric involvement [
31]. Early detection of changes in brain structures is essential for developing interventions since certain volumetric changes may precede the manifestation of clinical symptoms [
32].
When CSF was assessed, a progressive increase in volume was observed in the intermediate and full penetrance groups, consistent with data on brain atrophy (
Figure 3). These findings are more frequent in symptomatic patients and have even been detected up to 10 years before the onset of symptoms [
33,
34].
With regard to the white matter, the results are consistent with those of previous studies suggesting changes in brain conformation in the premanifest phase and even from childhood [
18,
32,
35,
36,
37], with a decrease in areas such as the corpus callosum and posterior white matter tracts [
18,
32,
35,
36,
37]. As the disease progresses to the symptomatic phase, white matter atrophy extends until it becomes global, which has been evidenced in volumetric analyses [
38] and in the TRACK-HD and PREDICT-HD reports, which describe the same progression. This is a finding repeated in our study where the full penetrance group shows a decrease in white matter (
Figure 4), thus supporting the usefulness of this biomarker to track the progression of the disease. The gray matter volume also shows a decrease in the full penetrance group, which is in line with studies indicating gradual changes in gray matter structures from the premanifest phase to the manifest phase of the disease [
39,
40].
As for the subcortical structures (
Figure 2), the striatum is considered the most affected structure in HD, and the underlying mechanisms explaining this regional preference are believed to be related to the instability of the mutation in different tissues. The TRACK-HD report has shown that the caudate volume exceeds that of the putamen at all phases of the disease [
11]. However, other studies based on structural resonances have indicated an alteration of 50% in the putamen and 20–30% in the caudate [
12]. Our study supports these findings by observing a more significant volume loss in the putamen compared to the caudate. These changes can predict conversion from the presymptomatic to the symptomatic phase in a period of up to 2 years [
28].
Regarding the globus pallidus, our results also indicate a decrease in its volume from the premanifest phase, with a notable change on the left side. As for the volume of the thalamus, although the literature emphasizes the significant atrophy in the late stages of HD, in our research, this volume remained relatively similar in the three groups.
Although not traditionally associated with HD, the amygdala showed differences in volume between groups, especially on the right side compared to the left, in addition to a slight increase in volume in the intermediate group and a slight decrease in the full penetrance group. This highlights the importance of further research in this area.
The ventricular system (
Figure 1 and
Figure 3), an indicator of total brain atrophy, revealed a progressive increase in its volume as the number of triplets grows, supporting the notion that ventricular dilation reflects the extension of the pathology in extrastriatal regions of the gray and white matter as the disease progresses [
41]. Ventricular dilation is seen approximately 5 years before the onset of motor symptoms and approximately a decade after the changes observed in the caudate owing to neuronal loss [
42]. Extrastriatal degeneration is also evident in the early stages of the clinical course of HD, which could be considered a global marker of progression, as well as total brain atrophy or ventricular enlargement [
43].
Our study found greater involvement of gray matter than cerebellar white matter (
Figure 4). Tissue loss of this structure has been reported in juvenile-onset HD and in some cases in adult-onset [
44,
45]. In most patients, these changes have been reported as moderate.
In studies of cerebellar atrophy, no correlation has been found with CAG repeat [
46,
47], and there is a nonsignificant trend toward an inverse correlation between cerebellar atrophy and disease duration, given that cerebellar neuronal loss occurs later in the clinical course of the disease [
48].
Finally, the brain stem nuclei were affected in HD. This structure shows a generalized neuronal loss with particular involvement of the substantia nigra, the precerebellar pontine nuclei, the inferior olive, the oculomotor reticulotegmental nucleus, the premotor oculomotor area, the interpositus raphe nucleus, the superior auditory olive, and the vestibular nuclei. Patients with HD present autonomic alterations and oculomotor dysfunction, some of which could be explained by brain stem pathology [
49]. To date, there is no evidence of brain stem involvement in the premanifest phase of HD. The findings of our study demonstrate that, rather than degeneration or loss of volume of this structure, there is an increase in volume in the intermediate group.
Regarding the brain volumes of cortical areas, both pathological and imaging studies have interpreted that the frontal lobes are altered in HD and that their degree of atrophy would be related to the severity of symptoms and general cognitive function. However, these relationships remained insignificant after considering the total brain volume [
41,
50]. In other research, individuals with pre-HD presented oculomotor and visuomotor deficits, possibly due to a loss of the visual and prefrontal cortex. In our study, no volumetric difference was observed between patients with normal AGC length and patients with full penetrance, with a paradoxical increase in volume in the intermediate group (
Figure 5).
As for the temporal, occipital, and parietal cortical volumes, it has been found that there may be signs of volume decrease in premanifest phases, but the atrophy itself occurs prior to the onset of symptoms or in the manifest clinical stage of HD, which is directly proportional to the expansion of triplets [
47,
51]. Despite this, as with the findings in the frontal lobe volume, there was also an increase in volume in the intermediate group, while the volume values between the normal and full penetrance groups were similar.
Structures such as the hippocampus and the insula show abnormal neurodegeneration in premanifest phases. However, the size of the effect is smaller than in the striatal structures, and these changes can occur up to 20 years before clinical diagnosis [
38]. In our study, the hippocampus volumes remain similar, except in the insular cortex, where volumes increase in the intermediate group.
Neurodegeneration in this disease follows a topographic and ordered distribution, starting from the tail of the caudate nucleus to the head and then the body. The most vulnerable neurons in HD are the medium-sized GABAergic spiny projection neurons, which constitute 90–95% of the neuronal population of the striatum, demonstrated by their specific characteristic of bilateral atrophy in this area [
52]. Other neuropathological changes occur in different areas of the cerebral cortex (layers III, IV, and V of the prefrontal cortex), which, due to their retrograde degenerative projections, explain the predominance of motor, cognitive, or emotional symptoms in each individual [
53].
Macroscopically, the thalamus is usually within normal limits but MRI studies have shown a variable loss in some of its nuclei due to astrocytosis and neuronal loss [
54]. The cerebellum, previously considered to be relatively unaffected, now shows a significant loss of Purkinje cells and neurons in the deep cerebellar nuclei [
55].
The blood–brain barrier is also affected, as aggregates of mutant HTT protein are present in all major components of the neurovascular unit, leading to structural, morphological, and functional changes in cerebral blood vessels [
56]. This is where neuroinflammation plays an important role, promoting the removal of cellular waste and acting on neurons, possibly contributing to neuronal death.
Mutant
HTT is expressed in the glial cells (astrocytes and microglia), thus reducing their neuroprotective function. It also mitigates the production and transport of brain-derived neurotrophic factor in the cortex through the corticostriatal pathway, with dysfunctional cortical signaling [
8]. An increase in multiple cytokines has also been observed both in plasma (interleukin [IL]-1b, IL-4, IL-6, IL-8, tumor necrosis factor [TNF]-α, and IL-10) and in the striatum (IL-6, IL-8, and TNF-α), especially IL-6, in carriers of presymptomatic mutations 16 years earlier, which would translate into early inflammatory changes.
Complement factors C1QC, C2, and C3, as well as other proteins associated with inflammatory pathways, such as peptidoglycan recognition protein 2 and apolipoprotein A4 have been found to increase in the CSF of HD patients compared to controls. The levels of these proteins in CSF followed a trend consistent with increased disease progression [
57].
Taken together, these findings enrich the understanding of volumetric variations in the premanifest phase of HD, thus contributing to the identification of potential biomarkers and highlighting the complexity of brain changes associated with triplet expansion. The divergence from the literature in some results underlines the need for additional research to consolidate and contextualize these findings in the context of the clinical heterogeneity of HD since, by the time the clinical stage begins, the disease has progressed markedly, and irreversible lesions are present. Therefore, it would be expected that interventions and treatments during the premanifest stages could be more effective [
33].
Limitations
The study presented several limitations, starting with the fact that the sample size was relatively small, 36 individuals from a specific population in the Colombian Caribbean, which could have limited the generalization of the findings to more diverse populations. In addition, selecting the sample among descendants of families affected by HD in a specific geographic location introduced a possible selection bias. The lack of genetic diversity within the sample could also have influenced the applicability of the results to populations with different genetic or ethnic backgrounds. Technical limitations in the acquisition of MRI and the interpretation of AI results should also have been considered. Despite the statistical analyses performed, the results should have been interpreted cautiously, recognizing the possibility of unmeasured variables that could have affected the findings.