Treatment of Refractory Mucosal Leishmaniasis Is Associated with Parasite Overexpression of HSP70 and ATPase and Reduced Host Hydrogen Peroxide Production (Brief Report)
Abstract
1. Introduction
2. Materials and Methods
2.1. Culture
2.2. Mass Spectrometry Analysis
2.2.1. Protein Extraction
2.2.2. Protein Digestion
2.2.3. Protein Identification
2.3. Hydrogen Peroxide Production
3. Results and Discussion
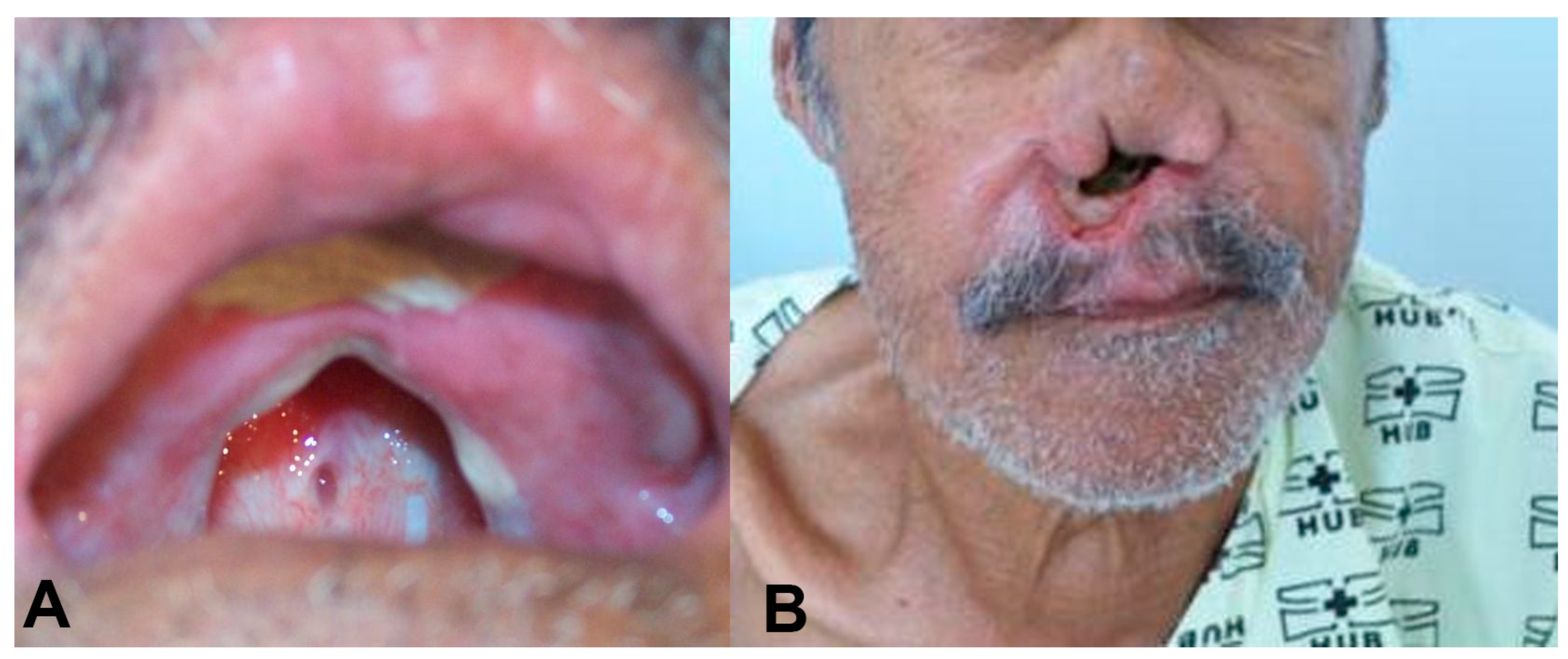
Clinical Case
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- World Health Organization. Leishmaniasis World Health Organization Site: World Health Organization. 2023. Available online: https://www.who.int/news-room/fact-sheets/detail/leishmaniasis (accessed on 13 June 2023).
- Hepburn, N. Cutaneous leishmaniasis. Clin. Exp. Dermatol. 2000, 25, 363–370. [Google Scholar] [CrossRef] [PubMed]
- Aronson, N.E.; Joya, C.A. Cutaneous leishmaniasis: Updates in diagnosis and management. Infect. Dis. Clin. 2019, 33, 101–117. [Google Scholar]
- Marsden, P.D. Mucosal leishmaniasis (“espundia” Escomel, 1911). Trans. R. Soc. Trop. Med. Hyg. 1986, 80, 859–876. [Google Scholar] [CrossRef]
- Ponte-Sucre, A.; Gamarro, F.; Dujardin, J.-C.; Barrett, M.P.; López-Vélez, R.; García-Hernández, R.; Pountain, A.W.; Mwenechanya, R.; Papadopoulou, B. Drug resistance and treatment failure in leishmaniasis: A 21st century challenge. PLoS Neglected Trop. Dis. 2017, 11, e0006052. [Google Scholar] [CrossRef] [PubMed]
- Koff, A.B.; Rosen, T. Treatment of cutaneous leishmaniasis. J. Am. Acad. Dermatol. 1994, 31, 693–708. [Google Scholar] [CrossRef]
- Borges, K.T.; Nogueira, L.S.C.; Sampaio, J.H.D.; Tauil, P.L.; Sampaio, R.N.R. Clinical, epidemiological and therapeuthic study of 402 patients with american cutaneous leishmaniasis attended at University Hospital of Brasilia, DF, Brazil. Bras. Dermatol. 2005, 80, 249–254. [Google Scholar]
- Biyani, N.; Singh, A.K.; Mandal, S.; Chawla, B.; Madhubala, R. Differential expression of proteins in antimony-susceptible and-resistant isolates of Leishmania donovani. Mol. Biochem. Parasitol. 2011, 179, 91–99. [Google Scholar] [CrossRef]
- Moreira, D.d.S.; Xavier, M.V.; Murta, S.M.F. Ascorbate peroxidase overexpression protects Leishmania braziliensis against trivalent antimony effects. Memórias Do Inst. Oswaldo Cruz 2018, 113, e180377. [Google Scholar] [CrossRef]
- Codonho, B.S.; Costa, S.d.S.; Peloso, E.d.F.; Joazeiro, P.P.; Gadelha, F.R.; Giorgio, S. HSP70 of Leishmania amazonensis alters resistance to different stresses and mitochondrial bioenergetics. Memórias Do Inst. Oswaldo Cruz 2016, 111, 460–468. [Google Scholar] [CrossRef] [PubMed]
- Rastrojo, A.; García-Hernández, R.; Vargas, P.; Camacho, E.; Corvo, L.; Imamura, H.; Dujardin, J.C.; Castanys, S.; Aguado, B.; Gamarro, F.; et al. Genomic and transcriptomic alterations in Leishmania donovani lines experimentally resistant to antileishmanial drugs. Int. J. Parasitol. Drugs Drug Resist. 2018, 8, 246–264. [Google Scholar] [CrossRef]
- Grimaldi, G.J.; Jaffe, C.L.; Mcmahon-Pratt, D.; Falqueto, A. A simple procedure for the isolation of leishmanial parasites and for the recovery of parasite virulence in avirulent stocks. Trans. R. Soc. Trop. Med. Hyg. 1984, 78, 560. [Google Scholar] [CrossRef] [PubMed]
- Brener, Z. Contribuição ao Estudo da Terapêutica Experimental da Doença de Chagas; Biblioteca Virtual em Saúde: Belo Horizonte, Brazil, 1961.
- Sussulini, A.; Garcia, J.S.; Mesko, M.F.; Moraes, D.P.; Flores, É.M.; Pérez, C.A.; Arruda, M.A. Evaluation of soybean seed protein extraction focusing on metalloprotein analysis. Microchim. Acta 2007, 158, 173–180. [Google Scholar] [CrossRef]
- Silva, J.C.; Denny, R.; Dorschel, C.A.; Gorenstein, M.; Kass, I.J.; Li, G.Z.; McKenna, T.; Nold, M.J.; Richardson, K.; Young, P.; et al. Quantitative proteomic analysis by accurate mass retention time pairs. Anal Chem. 2005, 77, 2187–2200. [Google Scholar] [CrossRef]
- Silva, J.C.; Gorenstein, M.V.; Li, G.-Z.; Vissers, J.P.; Geromanos, S.J. Absolute quantification of proteins by LCMSE: A virtue of parallel MS acquisition* S. Mol. Cell. Proteom. 2006, 5, 144–156. [Google Scholar] [CrossRef]
- Pick, E.; Keisari, Y. A simple colorimetric method for the measurement of hydrogen peroxide produced by cells in culture. J. Immunol. Methods 1980, 38, 161–170. [Google Scholar] [CrossRef] [PubMed]
- Gomes, C.M.; Paula, N.A.; Morais, O.O.; Soares, K.A.; Roselino, A.M.; Sampaio, R.N. Complementary exams in the diagnosis of American tegumentary leishmaniasis. Bras. Dermatol. 2014, 89, 701–709. [Google Scholar] [CrossRef] [PubMed]
- Cuba, C.C.; Llanos-Cuentas, E.A.; Barreto, A.C.; Magalhães, A.V.; Lago, E.L.; Reed, S.G.; Marsden, P.D. Human mucocutaneous leishmaniasis in Três Braços, Bahia-Brazil: An area of Leishmania braziliensis braziliensis transmission. I. Lab. Diagn. Rev. da Soc. Bras. de Med. Trop. 1984, 17, 161–167. [Google Scholar] [CrossRef]
- FIOCRUZ. Manual of Molecular Procedures: Instituto Oswaldo Cruz 2009; FIOCRUZ: Rio de Janeiro, Brazil, 2009. [Google Scholar]
- Gomes, C.M.; Cesetti, M.V.; de Paula, N.A.; Vernal, S.; Gupta, G.; Sampaio, R.N.; Roselino, A.M. Field Validation of SYBR Green- and TaqMan-Based Real-Time PCR Using Biopsy and Swab Samples to Diagnose American Tegumentary Leishmaniasis in an Area Where Leishmania (Viannia) braziliensis Is Endemic. J. Clin. Microbiol. 2017, 55, 526–534. [Google Scholar] [CrossRef]
- El-On, J. Current status and perspectives of the immunotherapy of leishmaniasis. Isr. Med. Assoc. J. IMAJ 2009, 11, 623–628. [Google Scholar]
- Pinart, M.; Rueda, J.-R.; Romero, G.A.; Pinzón-Flórez, C.E.; Osorio-Arango, K.; Maia-Elkhoury, A.N.S.; Reveiz, L.; Elias, V.M.; Tweed, J.A. Interventions for American cutaneous and mucocutaneous leishmaniasis. Cochrane Database Syst. Rev. 2020, 8, 1–329. [Google Scholar] [CrossRef]
- Mashayekhi Goyonlo, V.; Derakhshan, Z.; Darchini-Maragheh, E. Treatment of Cutaneous Leishmaniasis with Allopurinol Plus Itraconazole in Iran. Am. J. Trop. Med. Hyg. 2023, 108, 1164–1166. [Google Scholar] [CrossRef] [PubMed]
- Amato, V.S.; Padilha, A.R.; Nicodemo, A.C.; Duarte, M.I.; Valentini, M.; Uip, D.E.; Boulos, M.; Neto, V.A. Use of itraconazole in the treatment of mucocutaneous leishmaniasis: A pilot study. Int. J. Infect. Dis. 2000, 4, 153–157. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sampaio, R.N.R.; Paula CDRd Porto, C.; Motta JdOCd Pereira, L.I.d.A.; Martins, S.S.; Barroso, D.H.; Freire, G.S.M.; Gomes, C.M. A randomized, open-label clinical trial comparing the long-term effects of miltefosine and meglumine antimoniate for mucosal leishmaniasis. Rev. da Soc. Bras. de Med. Tropical. 2019, 52, e20180292. [Google Scholar] [CrossRef] [PubMed]
- Llanos-Cuentas, A.; Echevarria, J.; Cruz, M.; La Rosa, A.; Campos, P.; Campos, M.; Franke, E.; Berman, J.; Modabber, F.; Marr, J. Efficacy of sodium stibogluconate alone and in combination with allopurinol for treatment of mucocutaneous leishmaniasis. Clin. Infect. Dis. 1997, 25, 677–684. [Google Scholar] [CrossRef]
- Cunha, M.A.; Leão, A.C.; de Cassia Soler, R.; Lindoso, J.A. Efficacy and Safety of Liposomal Amphotericin B for the Treatment of Mucosal Leishmaniasis from the New World: A Retrospective Study. Am. J. Trop. Med. Hyg. 2015, 93, 1214–1218. [Google Scholar] [CrossRef] [PubMed]
- Santos, C.R.; Tuon, F.F.; Cieslinski, J.; de Souza, R.M.; Imamura, R.; Amato, V.S. Comparative study on liposomal amphotericin B and other therapies in the treatment of mucosal leishmaniasis: A 15-year retrospective cohort study. PLoS ONE 2019, 14, e0218786. [Google Scholar] [CrossRef] [PubMed]
- Lessa, H.A.; Machado, P.; Lima, F.; Cruz, A.A.; Bacellar, O.; Guerreiro, J.; Carvalho, E.M. Successful treatment of refractory mucosal leishmaniasis with pentoxifylline plus antimony. Am. J. Trop. Med. Hyg. 2001, 65, 87–89. [Google Scholar] [CrossRef]
- Goyonlo, V.M.; Vahabi-Amlashi, S.; Taghavi, F. Successful treatment by adding thalidomide to meglumine antimoniate in a case of refractory anthroponotic mucocutaneous leishmaniasis. Int. J. Parasitol. Drugs Drug Resistance. 2019, 11, 177–179. [Google Scholar] [CrossRef]
- Mayrink, W.; Botelho, A.C.d.C.; Magalhães, P.A.; Batista, S.M.; Lima, A.d.O.; Genaro, O.; Costa, C.A.D.; Melo, M.N.D.; Michalick, M.S.M.; Williams, P.; et al. Immunotherapy, immunochemotherapy and chemotherapy for American cutaneous leishmaniasis treatment. Rev. da Soc. Bras. de Med. Trop. 2006, 39, 14–21. [Google Scholar] [CrossRef]
- Sampaio, R.N.R. Pharmacotherapy in leishmaniasis: Old, new treatments, their impacts and expert opinion. Taylor Fr. 2023, 24, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Soto, J.; Toledo, J.; Valda, L.; Balderrama, M.; Rea, I.; Parra, R.; Ardiles, J.; Soto, P.; Gomez, A.; Molleda, F.; et al. Treatment of Bolivian mucosal leishmaniasis with miltefosine. Clin. Infect. Dis. 2007, 44, 350–356. [Google Scholar] [CrossRef] [PubMed]
- Soto, J.; Arana, B.A.; Toledo, J.; Rizzo, N.; Vega, J.C.; Diaz, A.; Luz, M.; Gutierrez, P.; Arboleda, M.; Berman, J.D.; et al. Miltefosine for new world cutaneous leishmaniasis. Clin. Infect. Dis. 2004, 38, 1266–1272. [Google Scholar] [CrossRef]
- Sampaio, R.; Marsden, P. Treatment of the mucosal form of leishmaniasis without response to glucantime, with liposomal amphotericin B. Rev. da Soc. Bras. de Med. Trop. 1997, 30, 125–128. [Google Scholar] [CrossRef] [PubMed]
- de Saldanha, R.R.; Martins-Papa, M.C.; Sampaio, R.N.R.; Muniz-Junqueira, M.I. Meglumine antimonate treatment enhances phagocytosis and TNF-α production by monocytes in human cutaneous leishmaniasis. Trans. R. Soc. Trop. Med. Hyg. 2012, 106, 596–603. [Google Scholar] [CrossRef]
- Tamarit, J.; Cabiscol, E.; Ros, J. Identification of the major oxidatively damaged proteins inEscherichia coli cells exposed to oxidative stress. J. Biol. Chem. 1998, 273, 3027–3032. [Google Scholar] [CrossRef]
- Muniz-Junqueira, M.I.; de Paula-Coelho, V.N. Meglumine antimonate directly increases phagocytosis, superoxide anion and TNF-α production, but only via TNF-α it indirectly increases nitric oxide production by phagocytes of healthy individuals, in vitro. Int. Immunopharmacol. 2008, 8, 1633–1638. [Google Scholar] [CrossRef]
- De Sarkar, S.; Chatterjee, M. Exploring Endoperoxides as Leishmanicidal Compounds. In Oxidative Stress in Microbial Diseases; Springer: Berlin/Heidelberg, Germany, 2019; p. 453. [Google Scholar]
- Barrett, M.P.; Kyle, D.E.; Sibley, L.D.; Radke, J.B.; Tarleton, R.L. Protozoan persister-like cells and drug treatment failure. Nat. Rev. Microbiol. 2019, 17, 607–620. [Google Scholar] [CrossRef]
- Mandell, M.A.; Beverley, S.M. Continual renewal and replication of persistent Leishmania major parasites in concomitantly immune hosts. Proc. Natl. Acad. Sci. USA 2017, 114, E801–E810. [Google Scholar] [CrossRef]
- Nirody, J.A.; Budin, I.; Rangamani, P. ATP synthase: Evolution, energetics, and membrane interactions. J. Gen. Physiol. 2020, 152, e201912475. [Google Scholar] [CrossRef]
- Lima, D.A.; Gonçalves, L.O.; Reis-Cunha, J.L.; Guimarães, P.A.S.; Ruiz, J.C.; Liarte, D.B.; Murta, S.M.F. Transcriptomic analysis of benznidazole-resistant and susceptible Trypanosoma cruzi populations. Parasit. Vectors 2023, 16, 167. [Google Scholar] [CrossRef] [PubMed]
- Mukherjee, A.; Padmanabhan, P.K.; Sahani, M.H.; Barrett, M.P.; Madhubala, R. Roles for mitochondria in pentamidine susceptibility and resistance in Leishmania donovani. Mol. Biochem. Parasitol. 2006, 145, 1–10. [Google Scholar] [CrossRef]
- Magalhães, R.D.M.; Duarte, M.C.; Mattos, E.C.; Martins, V.T.; Lage, P.S.; Chávez-Fumagalli, M.A.; Lage, D.P.; Menezes-Souza, D.; Regis, W.C.; Manso Alves, M.J.; et al. Identification of Differentially Expressed Proteins from Leishmania amazonensis Associated with the Loss of Virulence of the Parasites. PLoS Neglected Trop. Dis. 2014, 8, e2764. [Google Scholar] [CrossRef]
- Roy, A.; Ganguly, A.; BoseDasgupta, S.; Das, B.B.; Pal, C.; Jaisankar, P.; Majumder, H.K. Mitochondria-dependent reactive oxygen species-mediated programmed cell death induced by 3, 3′-diindolylmethane through inhibition of F0F1-ATP synthase in unicellular protozoan parasite Leishmania donovani. Mol. Pharmacol. 2008, 74, 1292–1307. [Google Scholar] [CrossRef] [PubMed]
- Bodley, A.L.; McGarry, M.W.; Shapiro, T.A. Drug cytotoxicity assay for African trypanosomes and Leishmania species. J. Infect. Dis. 1995, 172, 1157–1159. [Google Scholar] [CrossRef] [PubMed]
- Walker, J.; Gongora, R.; Vasquez, J.-J.; Drummelsmith, J.; Burchmore, R.; Roy, G.; Ouellette, M.; Gomez, M.A.; Saravia, N.G. Discovery of factors linked to antimony resistance in Leishmania panamensis through differential proteome analysis. Mol. Biochem. Parasitol. 2012, 183, 166–176. [Google Scholar] [CrossRef]
- Brandau, S.; Dresel, A.; Clos, J. High constitutive levels of heat-shock proteins in human-pathogenic parasites of the genus Leishmania. Biochem. J. 1995, 310, 225–232. [Google Scholar] [CrossRef]
- Cuervo, P.; De Jesus, J.B.; Saboia-Vahia, L.; Mendonça-Lima, L.; Domont, G.B.; Cupolillo, E. Proteomic characterization of the released/secreted proteins of Leishmania (Viannia) braziliensis promastigotes. J. Proteom. 2009, 73, 79–92. [Google Scholar] [CrossRef] [PubMed]
- Torres, D.C.; Ribeiro-Alves, M.; Romero, G.A.; Dávila, A.M.; Cupolillo, E. Assessment of drug resistance related genes as candidate markers for treatment outcome prediction of cutaneous leishmaniasis in Brazil. Acta Trop. 2013, 126, 132–141. [Google Scholar] [CrossRef]
- Inacio, J.D.; Gervazoni, L.; Canto-Cavalheiro, M.M.; Almeida-Amaral, E.E. The effect of (-)-epigallocatechin 3-O-gallate in vitro and in vivo in Leishmania braziliensis: Involvement of reactive oxygen species as a mechanism of action. PLoS Neglected Trop. Dis. 2014, 8, e3093. [Google Scholar] [CrossRef]
- Zininga, T.; Pooe, O.J.; Makhado, P.B.; Ramatsui, L.; Prinsloo, E.; Achilonu, I.; Dirr, H.; Shonhai, A. Polymyxin B inhibits the chaperone activity of Plasmodium falciparum Hsp70. Cell Stress Chaperones 2017, 22, 707–715. [Google Scholar] [CrossRef] [PubMed]
- Souza Ribeiro Costa, J.; Medeiros, M.; Yamashiro-Kanashiro, E.H.; Rocha, M.C.; Cotrim, P.C.; Stephano, M.A.; Lancellotti, M.; Tavares, G.D.; Oliveira-Nascimento, L. Biodegradable nanocarriers coated with polymyxin B: Evaluation of leishmanicidal and antibacterial potential. PLoS Neglected Trop. Dis. 2019, 13, e0007388. [Google Scholar] [CrossRef] [PubMed]
- Mendonça, M.G.; de Brito, M.E.; Rodrigues, E.H.; Bandeira, V.; Jardim, M.L.; Abath, F.G. Persistence of Leishmania parasites in scars after clinical cure of american cutaneous leishmaniasis: Is there a sterile cure? J. Infect. Dis. 2004, 189, 1018–1023. [Google Scholar] [CrossRef]
- Marsden, P.D.; Llanos-Cuentas, E.A.; Lago, E.L.; Cuba, C.C.; Barreto, A.C.; Costa, J.M.; Jones, T.C. Human mucocutaneous leishmaniasis in Três Braços, Bahia-Brazil. An area of Leishmania braziliensis braziliensis transmission. III-Mucosal disease presentation and initial evolution. Rev. Soc. Bras. Med. Trop. 1984, 17, 179–186. [Google Scholar] [CrossRef]
- Amato, V.; Tuon, F.; Imamura, R.; Abegao de Camargo, R.; Duarte, M.; Neto, V. Mucosal leishmaniasis: Description of case management approaches and analysis of risk factors for treatment failure in a cohort of 140 patients in Brazil. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 1026–1034. [Google Scholar] [CrossRef] [PubMed]
- Oliveira, F.S.; Valete-Rosalino, C.M.; Pacheco, S.J.; Costa, F.A.C.; Schubach, A.O.; Pacheco, R.S. American tegumentary leishmaniasis caused by Leishmania (Viannia) braziliensis: Assessment of parasite genetic variability at intra-and inter-patient levels. Parasites Vectors 2013, 6, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Pacheco, R.S.; Martinez, J.E.; Valderrama, L.; Momen, H.; Saravia, N.G. Genotypic polymorphisms in experimental metastatic dermal leishmaniasis. Mol. Biochem. Parasitol. 1995, 69, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Boité, M.C.; de Oliveira, T.S.; Ferreira, G.E.M.; Trannin, M.; dos Santos, B.N.; Porrozzi, R.; Cupolillo, E. Polymorphisms and ambiguous sites present in DNA sequences of Leishmania clones: Looking closer. Infect. Genet. Evol. 2014, 25, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Grimaldi, G.; Tesh, R. Leishmaniases of the New World: Current concepts and implications for future research. Clin. Microbiol. Rev. 1993, 6, 230–250. [Google Scholar] [CrossRef]
- Quaresma, P.F.; de Brito, C.F.A.; Rugani, J.M.N.; de Moura Freire, J.; de Paula Baptista, R.; Moreno, E.C.; Gontijo, R.C.; Rego, F.D.; Diniz, J.E.; Melo, M.N.; et al. Distinct genetic profiles of Leishmania (Viannia) braziliensis associate with clinical variations in cutaneous-leishmaniasis patients from an endemic area in Brazil. Parasitology 2018, 145, 1161–1169. [Google Scholar] [CrossRef]
- Sacks, D.L.; Melby, P.C. Animal models for the analysis of immune responses to leishmaniasis. Curr. Protoc. Immunol. 1998, 28, 19.2.1–19.2.20. [Google Scholar] [CrossRef]

| Drug | Year | Duration (Days) | Year–Dosage/Day–Duration (Days)–Evolution | Outcome |
|---|---|---|---|---|
| N-methyl glucamine | 1987 | 30 | 20 mg SbV/kg/day | Clinical cure |
| 1988 | 20 | 20 mg SbV/kg/day | Clinical cure | |
| 2002 | 30 | 20 mg SbV/kg/day | Clinical improvement | |
| N-methyl glucamine + allopurinol | 1996 | 30 | 20 mg SbV/kg/day + 15 mg/kg/day | Clinical cure |
| Aminosidine sulfate | 1993 | 25 | 16 mg/kg/day (2 series) | Clinical cure |
| 1994 | 25 | 16 mg/kg/day (1 series) | Clinical cure | |
| 1999 | 25 | 16 mg/kg/day (1 series) | Clinical improvement | |
| Itraconazol + allopurinol | 1996 | 15 | 200 mg + 15 mg/kg/day | Therapeutic failure |
| N-methyl glucamine + thalidomide | 2000 | 30 | 20 mg SbV/kg + 200 mg/day | Clinical cure |
| Liposomal amphotericin B | 1992 | 28 | 4050 mg cumulative dose | Clinical cure |
| 2002 | 90 | 2100 mg cumulative dose | Clinical improvement | |
| N-methyl-glucamine + pentoxifylline | 1998 | 30 | 20 mg SbV/kg + 1200 mg/day | Clinical improvement |
| Leishvacin + N-methyl glucamine | 2004 | 130 | From 0.1 mL in the first day to 1 mL in the 10th week, once per week; 15 mg SbV/kg every other day, series of 10 days with intervals of 10 days–13 weeks | Clinical improvement |
| Miltefosine | 2007 | 42 | 2 mg/kg/day | Clinical and parasitological cure |
| Pentamidine | 1995 | 48 | 1500 mg cumulative dose | Clinical cure |
| Liposomal amphotericin B + N-methyl glucamine | 2005 | 40 | 2000 mg cumulative dose + 20 mg SbV/kg/day | Clinical improvement |
| Liposomal amphotericin B + N-+ methyl glucamine + Leishvacin | 2006–2007 | 180 | Cumulative dose 3000 mg + 20 mg SbV/kg/day-(7 series) | Clinical improvement |
| Miltefosine + liposomal amphotericin B + pentoxyphylline | 2010 | 34 | 750 mg + 1200 mg cumulative doses. Treatment interruption due to creatinine increase | Clinical improvement |
| Liposomal amphotericin B + pentoxyphylline | 2011/2012 | 180 | 3000 mg cumulative dose + 1200 mg/day | Clinical improvement |
| Protein | L. (V.) braziliensis (Control) | L. (V.) braziliensis (Patient Isolate) |
|---|---|---|
| Putative heat shock protein 70-related protein 1 mitochondrial precursor Leishmania (V.) braziliensis | 32.38 | 65.33 |
| ATPase alpha subunit Leishmania (V.) braziliensis | 30.3553 | 166.27 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Urdapilleta, A.A.A.; Santos Alfani, A.d.O.; Barroso, D.H.; Vinecky, F.; Amaral Vaz Bandeira, S.d.G.; Andrade, A.C.; Taquita, J.A.; Bastos, I.M.D.; Sampaio, R.N.R. Treatment of Refractory Mucosal Leishmaniasis Is Associated with Parasite Overexpression of HSP70 and ATPase and Reduced Host Hydrogen Peroxide Production (Brief Report). Biomedicines 2024, 12, 2227. https://doi.org/10.3390/biomedicines12102227
Urdapilleta AAA, Santos Alfani AdO, Barroso DH, Vinecky F, Amaral Vaz Bandeira SdG, Andrade AC, Taquita JA, Bastos IMD, Sampaio RNR. Treatment of Refractory Mucosal Leishmaniasis Is Associated with Parasite Overexpression of HSP70 and ATPase and Reduced Host Hydrogen Peroxide Production (Brief Report). Biomedicines. 2024; 12(10):2227. https://doi.org/10.3390/biomedicines12102227
Chicago/Turabian StyleUrdapilleta, Ada Amália Ayala, Adriana de Oliveira Santos Alfani, Daniel Holanda Barroso, Felipe Vinecky, Suzana da Glória Amaral Vaz Bandeira, Alan Carvalho Andrade, Jorge Alex Taquita, Izabela Marques Dourado Bastos, and Raimunda Nonata Ribeiro Sampaio. 2024. "Treatment of Refractory Mucosal Leishmaniasis Is Associated with Parasite Overexpression of HSP70 and ATPase and Reduced Host Hydrogen Peroxide Production (Brief Report)" Biomedicines 12, no. 10: 2227. https://doi.org/10.3390/biomedicines12102227
APA StyleUrdapilleta, A. A. A., Santos Alfani, A. d. O., Barroso, D. H., Vinecky, F., Amaral Vaz Bandeira, S. d. G., Andrade, A. C., Taquita, J. A., Bastos, I. M. D., & Sampaio, R. N. R. (2024). Treatment of Refractory Mucosal Leishmaniasis Is Associated with Parasite Overexpression of HSP70 and ATPase and Reduced Host Hydrogen Peroxide Production (Brief Report). Biomedicines, 12(10), 2227. https://doi.org/10.3390/biomedicines12102227






