Integrative Multi-Omics Approach in Vascular Ehlers–Danlos Syndrome: Further Insights into the Disease Mechanisms by Proteomic Analysis of Patient Dermal Fibroblasts
Abstract
1. Introduction
2. Materials and Methods
2.1. Patients
2.2. Cell Culture Conditions
2.3. Label-Free Liquid Chromatography with Tandem Mass Spectrometry (LC-MS/MS)
2.4. MS Data Processing and Analysis
2.5. Bioinformatic Analyses
2.6. Cells Transfection
2.7. mRNA and miRNA Expression Analyses
2.8. Protein Quantification by Immunoblotting
2.9. Artificial Intelligence (AI)
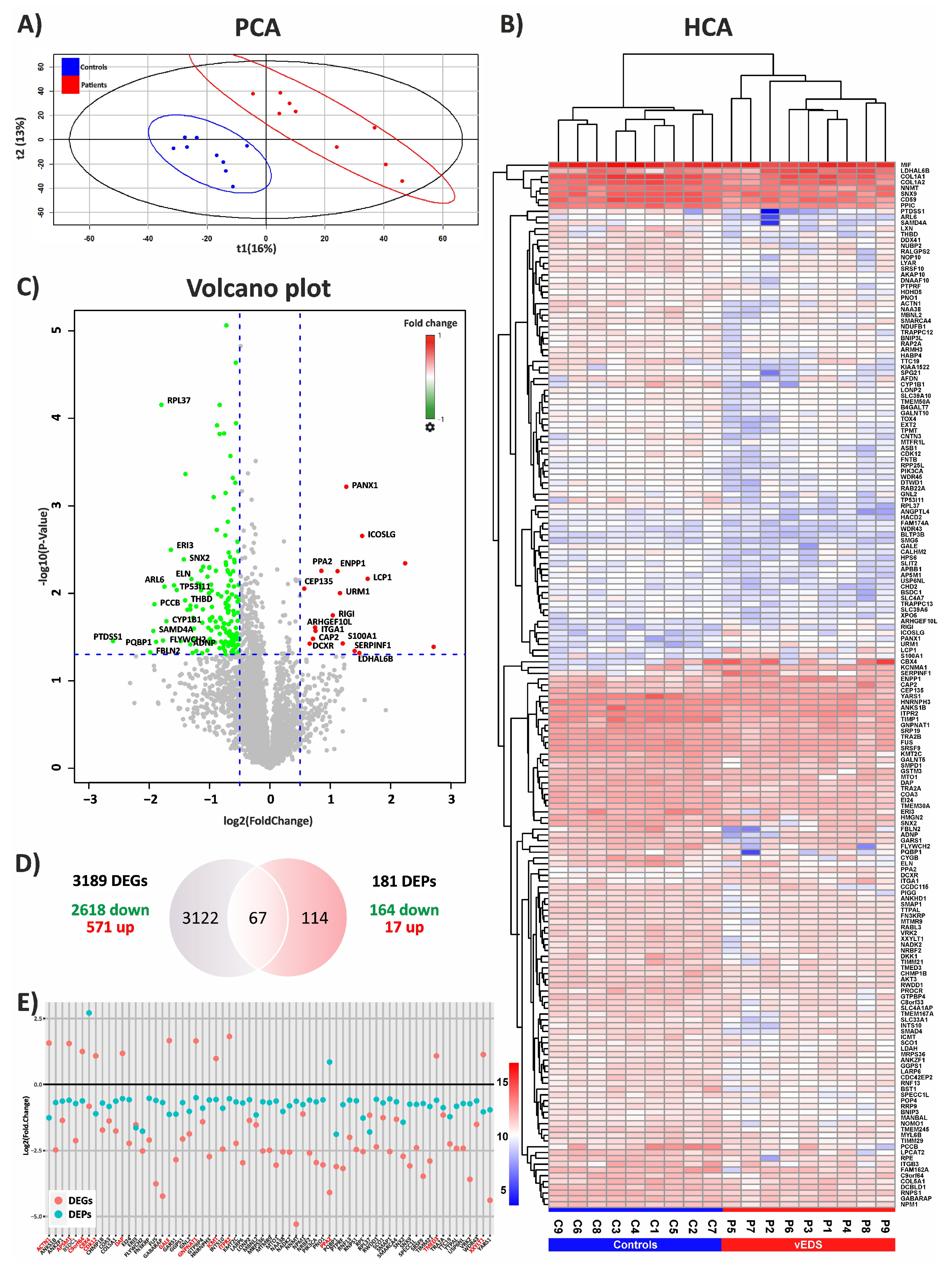
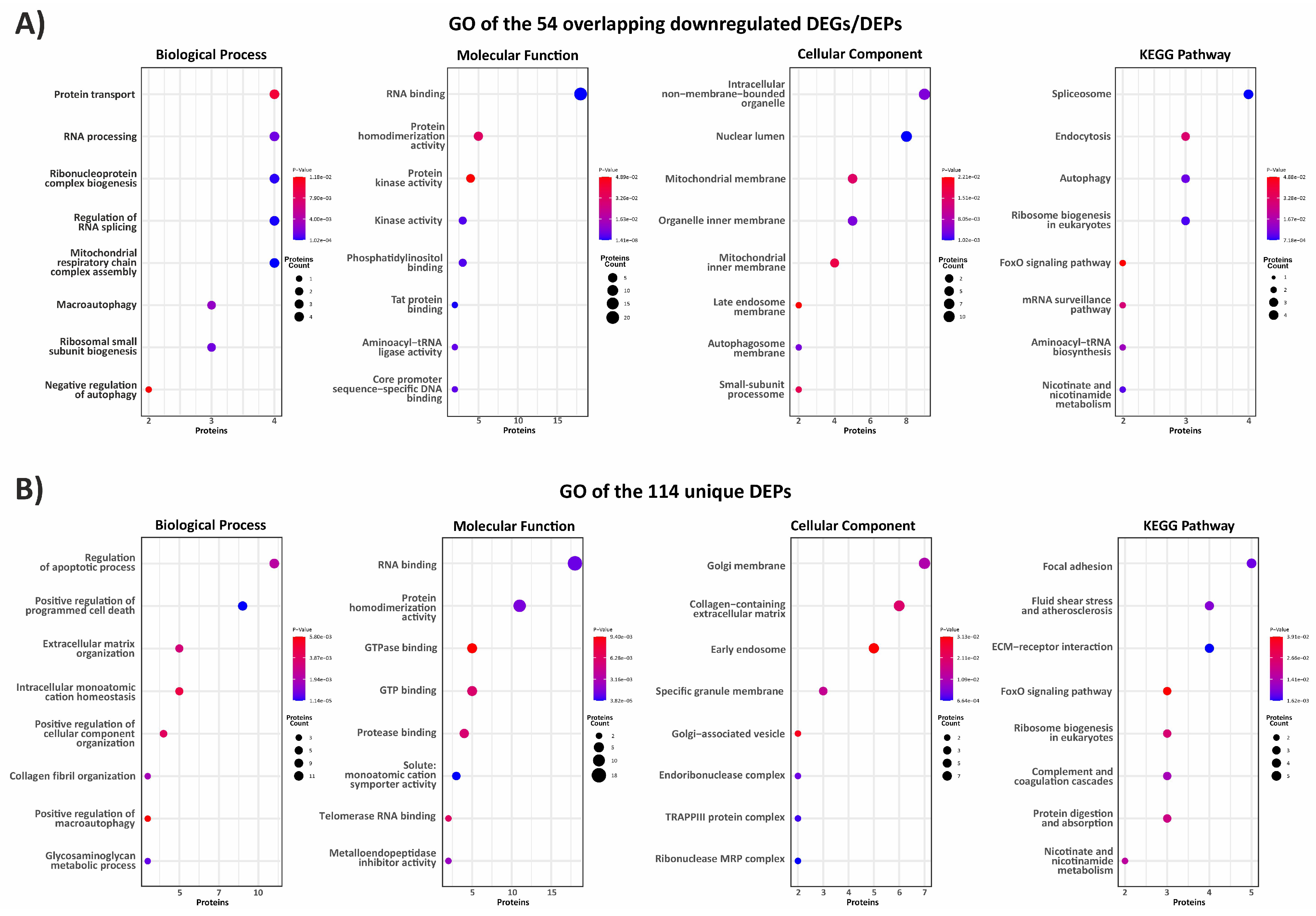
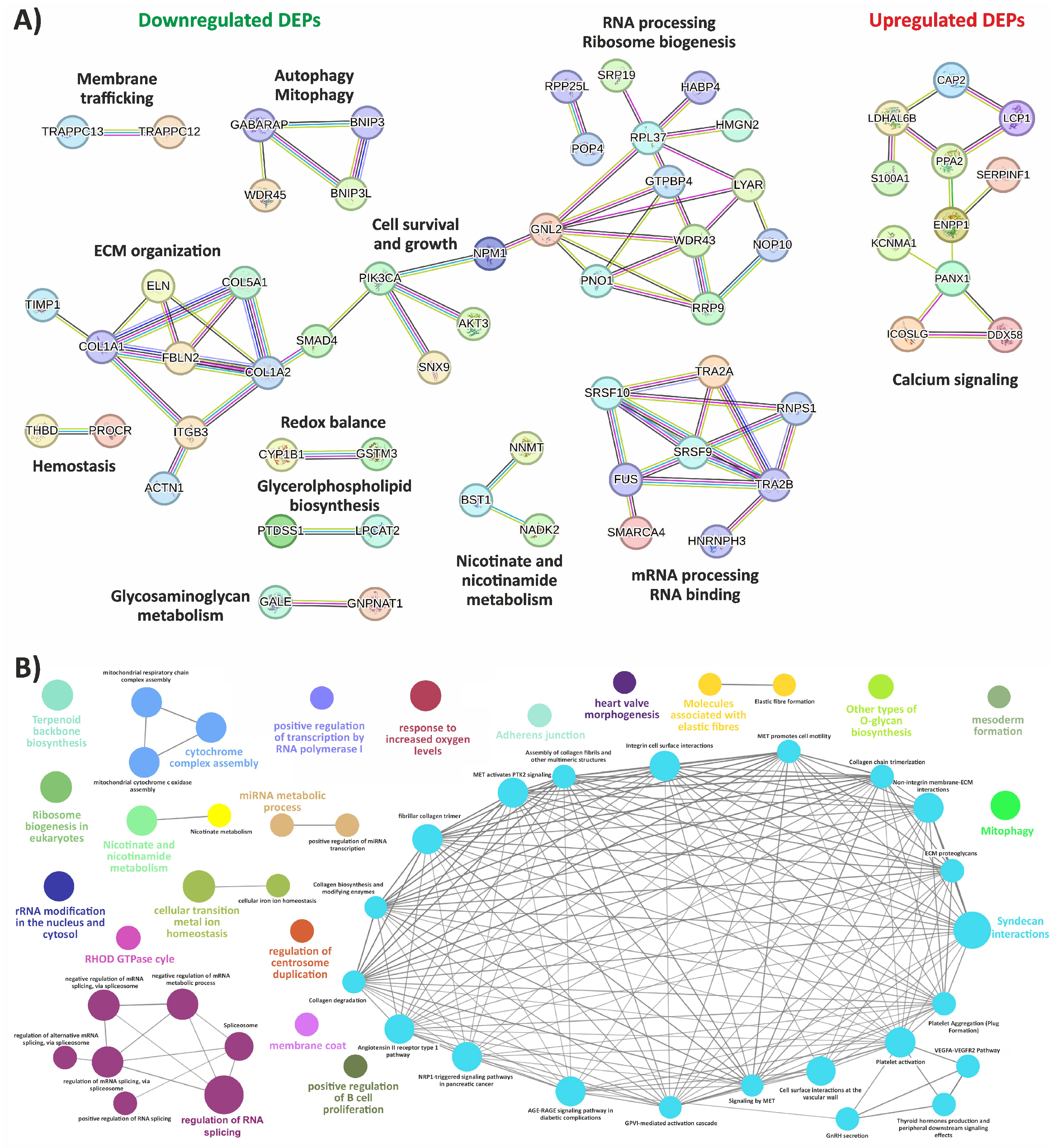
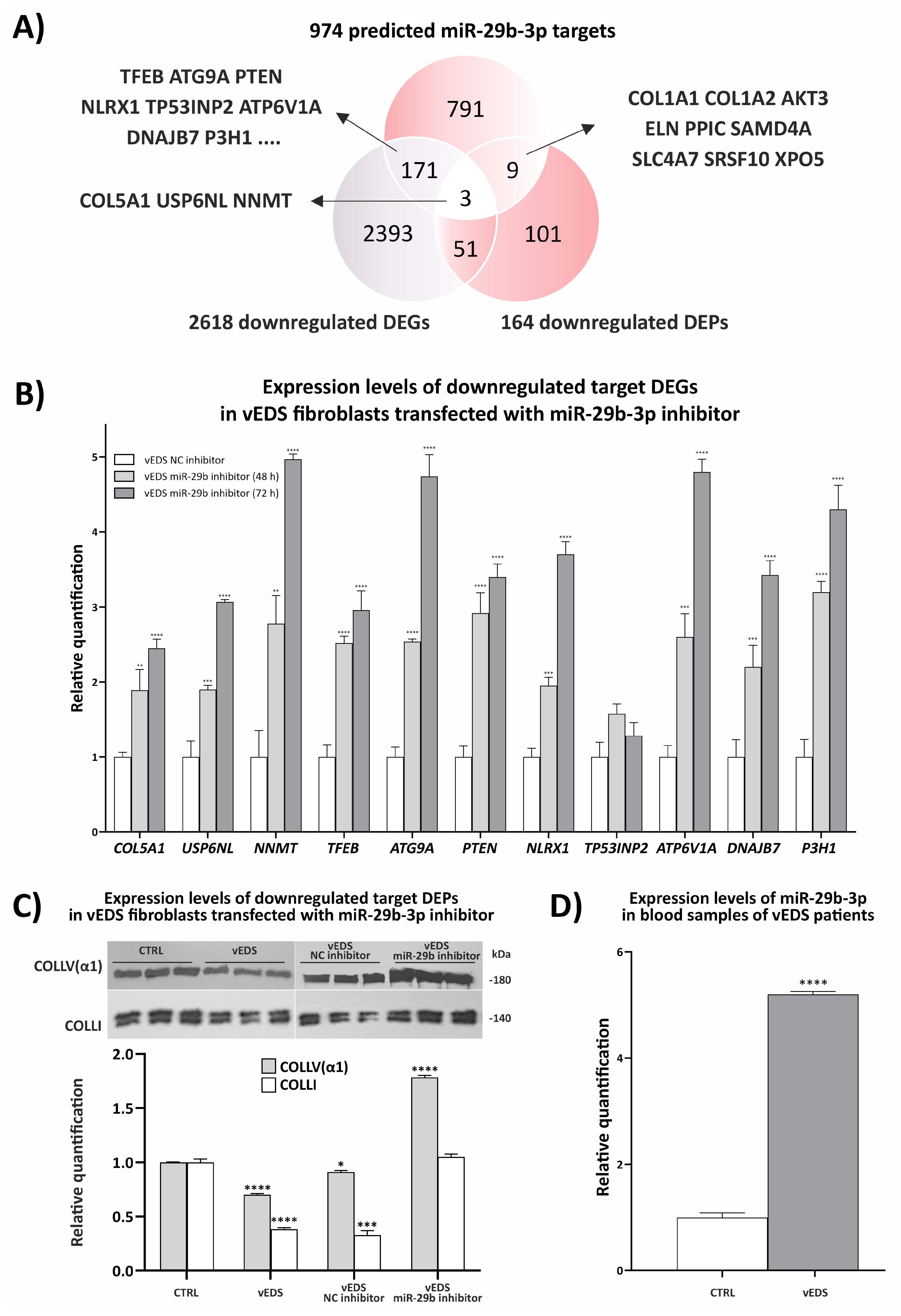
3. Results
3.1. Exploring the Proteomic Landscape of vEDS Patient Fibroblasts and Its Integration with Transcriptomic Insights
3.2. Exploring the Role of miR-29b-3p in Disease Mechanisms and Its Therapeutic Potential
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Malfait, F.; Castori, M.; Francomano, C.A.; Giunta, C.; Kosho, T.; Byers, P.H. The Ehlers-Danlos Syndromes. Nat. Rev. Dis. Prim. 2020, 6, 64. [Google Scholar] [CrossRef] [PubMed]
- Kuivaniemi, H.; Tromp, G. Type III Collagen (COL3A1): Gene and Protein Structure, Tissue Distribution, and Associated Diseases. Gene 2019, 707, 151–171. [Google Scholar] [CrossRef] [PubMed]
- Omar, R.; Malfait, F.; Van Agtmael, T. Four Decades in the Making: Collagen III and Mechanisms of Vascular Ehlers Danlos Syndrome. Matrix Biol. Plus 2021, 12, 100090. [Google Scholar] [CrossRef]
- Pepin, M.; Schwarze, U.; Superti-Furga, A.; Byers, P.H. Clinical and Genetic Features of Ehlers-Danlos Syndrome Type IV, the Vascular Type. N. Engl. J. Med. 2000, 342, 673–680. [Google Scholar] [CrossRef]
- Shalhub, S.; Byers, P.H.; Hicks, K.L.; Charlton-Ouw, K.; Zarkowsky, D.; Coleman, D.M.; Davis, F.M.; Regalado, E.S.; De Caridi, G.; Weaver, K.N.; et al. A Multi-Institutional Experience in the Aortic and Arterial Pathology in Individuals with Genetically Confirmed Vascular Ehlers-Danlos Syndrome. J. Vasc. Surg. 2019, 70, 1543–1554. [Google Scholar] [CrossRef] [PubMed]
- Shalhub, S.; Byers, P.H.; Hicks, K.L.; Coleman, D.M.; Davis, F.M.; De Caridi, G.; Weaver, K.N.; Miller, E.M.; Schermerhorn, M.L.; Shean, K.; et al. A Multi-Institutional Experience in Vascular Ehlers-Danlos Syndrome Diagnosis. J. Vasc. Surg. 2020, 71, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Shalhub, S.; Nkansah, R.; El-Ghazali, A.; Hillenbrand, C.J.; Vaidya, S.S.; Schwarze, U.; Byers, P.H. Splenic Artery Pathology Presentation, Operative Interventions, and Outcomes in 88 Patients with Vascular Ehlers-Danlos Syndrome. J. Vasc. Surg. 2023, 78, 394–404. [Google Scholar] [CrossRef]
- Frank, M.; Adham, S.; Seigle, S.; Legrand, A.; Mirault, T.; Henneton, P.; Albuisson, J.; Denarié, N.; Mazzella, J.M.; Mousseaux, E.; et al. Vascular Ehlers-Danlos Syndrome: Long-Term Observational Study. J. Am. Coll. Cardiol. 2019, 73, 1948–1957. [Google Scholar] [CrossRef]
- Demirdas, S.; van den Bersselaar, L.M.; Lechner, R.; Bos, J.; Alsters, S.I.M.; Baars, M.J.H.; Baas, A.F.; Baysal, Ö.; van der Crabben, S.N.; Dulfer, E.; et al. Vascular Ehlers-Danlos Syndrome: A Comprehensive Natural History Study in a Dutch National Cohort of 142 Patients. Circ. Genomic Precis. Med. 2024, 17, e003978. [Google Scholar] [CrossRef] [PubMed]
- Frank, M.; Adham, S.; Zinzindohoué, F.; Jeunemaitre, X. Natural History of Gastrointestinal Manifestations in Vascular Ehlers-Danlos Syndrome: A 17-Year Retrospective Review. J. Gastroenterol. Hepatol. 2019, 34, 857–863. [Google Scholar] [CrossRef]
- Murray, M.L.; Pepin, M.; Peterson, S.; Byers, P.H. Pregnancy-Related Deaths and Complications in Women with Vascular Ehlers-Danlos Syndrome. Genet. Med. 2014, 16, 874–880. [Google Scholar] [CrossRef] [PubMed]
- Haem, T.; Benson, B.; Dernoncourt, A.; Gondry, J.; Schmidt, J.; Foulon, A. Vascular Ehlers-Danlos Syndrome and Pregnancy: A Systematic Review. BJOG 2024, 131, 1620–1629. [Google Scholar] [CrossRef]
- Byers, P.H.; Belmont, J.; Black, J.; De Backer, J.; Frank, M.; Jeunemaitre, X.; Johnson, D.; Pepin, M.; Robert, L.; Sanders, L.; et al. Diagnosis, Natural History, and Management in Vascular Ehlers–Danlos Syndrome. Am. J. Med. Genet. Part C Semin. Med. Genet. 2017, 175, 40–47. [Google Scholar] [CrossRef] [PubMed]
- Ritelli, M.; Rovati, C.; Venturini, M.; Chiarelli, N.; Cinquina, V.; Castori, M.; Colombi, M. Application of the 2017 Criteria for Vascular Ehlers-Danlos Syndrome in 50 Patients Ascertained According to the Villefranche Nosology. Clin. Genet. 2020, 97, 287–295. [Google Scholar] [CrossRef]
- Bowen, J.M.; Hernandez, M.; Johnson, D.S.; Green, C.; Kammin, T.; Baker, D.; Keigwin, S.; Makino, S.; Taylor, N.; Watson, O.; et al. Diagnosis and Management of Vascular Ehlers-Danlos Syndrome: Experience of the UK National Diagnostic Service, Sheffield. Eur. J. Hum. Genet. 2023, 31, 749–760. [Google Scholar] [CrossRef]
- Eagleton, M.J. Arterial Complications of Vascular Ehlers-Danlos Syndrome. J. Vasc. Surg. 2016, 64, 1869–1880. [Google Scholar] [CrossRef]
- Ong, K.T.; Perdu, J.; De Backer, J.; Bozec, E.; Collignon, P.; Emmerich, J.; Fauret, A.L.; Fiessinger, J.N.; Germain, D.P.; Georgesco, G.; et al. Effect of Celiprolol on Prevention of Cardiovascular Events in Vascular Ehlers-Danlos Syndrome: A Prospective Randomised, Open, Blinded-Endpoints Trial. Lancet 2010, 376, 1476–1484. [Google Scholar] [CrossRef] [PubMed]
- Buso, G.; Paini, A.; Agabiti-Rosei, C.; De Ciuceis, C.; Bertacchini, F.; Stassaldi, D.; Salvetti, M.; Ritelli, M.; Venturini, M.; Colombi, M.; et al. Despite Celiprolol Therapy, Patients with Vascular Ehlers-Danlos Syndrome Remain at Risk of Vascular Events: A 12-Year Experience in an Italian Referral Center. Vasc. Med. 2023, 29. [Google Scholar] [CrossRef]
- Baderkhan, H.; Wanhainen, A.; Stenborg, A.; Stattin, E.L.; Björck, M. Celiprolol Treatment in Patients with Vascular Ehlers-Danlos Syndrome. Eur. J. Vasc. Endovasc. Surg. 2021, 61, 326–331. [Google Scholar] [CrossRef]
- Legrand Id, A.; Guery, C.; Faugeroux, J.; Fontaine Id, E.; Beugnon, C.; Lie Gianfermi, A.; Loisel-Ferreiraid, I.; Verpont, M.-C.; Adhamid, S.; Miraultid, T.; et al. Comparative Therapeutic Strategies for Preventing Aortic Rupture in a Mouse Model of Vascular Ehlers-Danlos Syndrome. PLoS Genet. 2022, 18, e1010059. [Google Scholar] [CrossRef] [PubMed]
- Bowen, C.J.; Giadrosic, J.F.C.; Burger, Z.; Rykiel, G.; Davis, E.C.; Helmers, M.R.; Benke, K.; MacFarlane, E.G.; Dietz, H.C. Targetable Cellular Signaling Events Mediate Vascular Pathology in Vascular Ehlers-Danlos Syndrome. J. Clin. Investig. 2020, 130, 686–698. [Google Scholar] [CrossRef] [PubMed]
- Buso, G.; Corvini, F.; Fusco, E.M.; Messina, M.; Cherubini, F.; Laera, N.; Paini, A.; Salvetti, M.; De Ciuceis, C.; Ritelli, M.; et al. Current Evidence and Future Perspectives in the Medical Management of Vascular Ehlers—Danlos Syndrome: Focus on Vascular Prevention. J. Clin. Med. 2024, 13, 4255. [Google Scholar] [CrossRef] [PubMed]
- Omar, R.; Lee, M.; Gonzalez-Trueba, L.; Lianos, S.; Hazarika, S.; Ammar, M.A.; Cassels, J.; Michie, A.M.; Bulleid, N.J.; Malfait, F.; et al. The Chemical Chaperone 4-Phenylbutyric Acid Rescues Molecular Cell Defects of COL3A1 Mutations That Cause Vascular Ehlers Danlos Syndrome. bioRxiv 2024. [Google Scholar] [CrossRef]
- Chiarelli, N.; Cinquina, V.; Martini, P.; Bertini, V.; Zoppi, N.; Venturini, M.; Ritelli, M.; Colombi, M. Deciphering Disease Signatures and Molecular Targets in Vascular Ehlers-Danlos Syndrome through Transcriptome and MiRNome Sequencing of Dermal Fibroblasts. Biochim. Biophys. Acta Mol. Basis Dis. 2024, 1870, 166915. [Google Scholar] [CrossRef]
- Terriaca, S.; Monastero, R.; Orlandi, A.; Balistreri, C.R. The Key Role of MiRNA in Syndromic and Sporadic Forms of Ascending Aortic Aneurysms as Biomarkers and Targets of Novel Therapeutic Strategies. Front. Genet. 2024, 15, 1365711. [Google Scholar] [CrossRef]
- Chiarelli, N.; Carini, G.; Zoppi, N.; Ritelli, M.; Colombi, M. Transcriptome Analysis of Skin Fibroblasts with Dominant Negative COL3A1 Mutations Provides Molecular Insights into the Etiopathology of Vascular Ehlers-Danlos Syndrome. PLoS ONE 2018, 13, e0191220. [Google Scholar] [CrossRef] [PubMed]
- Zoppi, N.; Chiarelli, N.; Ritelli, M.; Colombi, M. Multifaced Roles of the αvβ3 Integrin in Ehlers-Danlos and Arterial Tortuosity Syndromes’ Dermal Fibroblasts. Int. J. Mol. Sci. 2018, 19, 982. [Google Scholar] [CrossRef] [PubMed]
- Reggio, A.; Buonomo, V.; Berkane, R.; Bhaskara, R.M.; Tellechea, M.; Peluso, I.; Polishchuk, E.; Di Lorenzo, G.; Cirillo, C.; Esposito, M.; et al. Role of FAM134 Paralogues in Endoplasmic Reticulum Remodeling, ER-phagy, and Collagen Quality Control. EMBO Rep. 2021, 22, e52289. [Google Scholar] [CrossRef] [PubMed]
- Michalski, A.; Damoc, E.; Hauschild, J.P.; Lange, O.; Wieghaus, A.; Makarov, A.; Nagaraj, N.; Cox, J.; Mann, M.; Horning, S. Mass Spectrometry-Based Proteomics Using Q Exactive, a High-Performance Benchtop Quadrupole Orbitrap Mass Spectrometer. Mol. Cell. Proteom. 2011, 10, M111.011015. [Google Scholar] [CrossRef]
- Welten, S.M.J.; Goossens, E.A.C.; Quax, P.H.A.; Nossent, A.Y. The Multifactorial Nature of MicroRNAs in Vascular Remodelling. Cardiovasc. Res. 2016, 110, 6–22. [Google Scholar] [CrossRef] [PubMed]
- Rega, S.; Farina, F.; Bouhuis, S.; de Donato, S.; Chiesa, M.; Poggio, P.; Cavallotti, L.; Bonalumi, G.; Giambuzzi, I.; Pompilio, G.; et al. Multi-Omics in Thoracic Aortic Aneurysm: The Complex Road to the Simplification. Cell Biosci. 2023, 13, 131. [Google Scholar] [CrossRef] [PubMed]
- Verhagen, J.M.A.; Burger, J.; Bekkers, J.A.; Den Dekker, A.T.; von der Thüsen, J.H.; Zajec, M.; Brüggenwirth, H.T.; van der Sterre, M.L.T.; van den Born, M.; Luider, T.M.; et al. Multi-Omics Profiling in Marfan Syndrome: Further Insights into the Molecular Mechanisms Involved in Aortic Disease. Int. J. Mol. Sci. 2022, 23, 438. [Google Scholar] [CrossRef]
- Udugampolage, N.S.; Frolova, S.; Taurino, J.; Pini, A.; Martelli, F.; Voellenkle, C. Coding and Non-Coding Transcriptomic Landscape of Aortic Complications in Marfan Syndrome. Int. J. Mol. Sci. 2024, 25, 7367. [Google Scholar] [CrossRef] [PubMed]
- Lei, J.; Qiu, P.; Wu, Z.; Ding, A.; Hu, J.; Hou, J.; Jiang, Y.; Pu, H.; Huang, Q.; Zhang, X.; et al. Integrative Multi-Omics Analyses Reveal Vesicle Transport as a Potential Target for Thoracic Aortic Aneurysm. Comput. Biol. Med. 2024, 170, 108071. [Google Scholar] [CrossRef] [PubMed]
- Stratton, M.S.; Farina, F.M.; Elia, L. Epigenetics and Vascular Diseases. J. Mol. Cell. Cardiol. 2019, 133, 148. [Google Scholar] [CrossRef]
- Chistiakov, D.A.; Shkurat, T.P.; Melnichenko, A.A.; Grechko, A.V.; Orekhov, A.N. The Role of Mitochondrial Dysfunction in Cardiovascular Disease: A Brief Review. Ann. Med. 2018, 50, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Van Der Pluijm, I.; Burger, J.; Van Heijningen, P.M.; IJpma, A.; Van Vliet, N.; Milanese, C.; Schoonderwoerd, K.; Sluiter, W.; Ringuette, L.J.; Dekkers, D.H.W.; et al. Decreased Mitochondrial Respiration in Aneurysmal Aortas of Fibulin-4 Mutant Mice Is Linked to PGC1A Regulation. Cardiovasc. Res. 2018, 114, 1776–1793. [Google Scholar] [CrossRef] [PubMed]
- Németh, C.E.; Marcolongo, P.; Gamberucci, A.; Fulceri, R.; Benedetti, A.; Zoppi, N.; Ritelli, M.; Chiarelli, N.; Colombi, M.; Willaert, A.; et al. Glucose Transporter Type 10-Lacking in Arterial Tortuosity Syndrome-Facilitates Dehydroascorbic Acid Transport. FEBS Lett. 2016, 590, 1630–1640. [Google Scholar] [CrossRef] [PubMed]
- Zoppi, N.; Chiarelli, N.; Cinquina, V.; Ritelli, M.; Colombi, M. GLUT10 Deficiency Leads to Oxidative Stress and Non-Canonical Avβ3 Integrin-Mediated TGFβ Signalling Associated with Extracellular Matrix Disarray in Arterial Tortuosity Syndrome Skin Fibroblasts. Hum. Mol. Genet. 2015, 24, 6769–6787. [Google Scholar] [CrossRef]
- Oller, J.; Gabandé-Rodríguez, E.; Ruiz-Rodríguez, M.J.; Desdín-Micó, G.; Aranda, J.F.; Rodrigues-Diez, R.; Ballesteros-Martínez, C.; Blanco, E.M.; Roldan-Montero, R.; Acuña, P.; et al. Extracellular Tuning of Mitochondrial Respiration Leads to Aortic Aneurysm. Circulation 2021, 143, 2091–2109. [Google Scholar] [CrossRef] [PubMed]
- De Leonibus, C.; Maddaluno, M.; Ferriero, R.; Besio, R.; Cinque, L.; Lim, P.J.; Palma, A.; De Cegli, R.; Gagliotta, S.; Montefusco, S.; et al. Sestrin2 Drives ER-Phagy in Response to Protein Misfolding. Dev. Cell 2024, 59, 2035–2052.e10. [Google Scholar] [CrossRef] [PubMed]
- De Cavanagh, E.M.; Ferder, M.; Inserra, F.; Ferder, L. Angiotensin II, Mitochondria, Cytoskeletal, and Extracellular Matrix Connections: An Integrating Viewpoint. Am. J. Physiol. Heart Circ. Physiol. 2009, 296, H550–H558. [Google Scholar] [CrossRef] [PubMed]
- Chiarelli, N.; Ritelli, M.; Zoppi, N.; Colombi, M. Cellular and Molecular Mechanisms in the Pathogenesis of Classical, Vascular, and Hypermobile Ehlers—Danlos Syndromes. Genes 2019, 10, 609. [Google Scholar] [CrossRef]
- Nappi, F. Non-Coding RNA-Targeted Therapy: A State-of-the-Art Review. Int. J. Mol. Sci. 2024, 25, 3630. [Google Scholar] [CrossRef] [PubMed]
- Hibender, S.; Franken, R.; Van Roomen, C.; Ter Braake, A.; Van Der Made, I.; Schermer, E.E.; Gunst, Q.; Van Den Hoff, M.J.; Lutgens, E.; Pinto, Y.M.; et al. Resveratrol Inhibits Aortic Root Dilatation in the Fbn1C1039G/+ Marfan Mouse Model. Arterioscler. Thromb. Vasc. Biol. 2016, 36, 1618–1626. [Google Scholar] [CrossRef] [PubMed]
- Boon, R.A.; Dimmeler, S. MicroRNAs and Aneurysm Formation. Trends Cardiovasc. Med. 2011, 21, 172–177. [Google Scholar] [CrossRef] [PubMed]
- Maegdefessel, L.; Azuma, J.; Toh, R.; Merk, D.R.; Deng, A.; Chin, J.T.; Raaz, U.; Schoelmerich, A.M.; Raiesdana, A.; Leeper, N.J.; et al. Inhibition of MicroRNA-29b Reduces Murine Abdominal Aortic Aneurysm Development. J. Clin. Investig. 2012, 122, 497–506. [Google Scholar] [CrossRef]
- Merk, D.R.; Chin, J.T.; Dake, B.A.; Maegdefessel, L.; Miller, M.O.; Kimura, N.; Tsao, P.S.; Iosef, C.; Berry, G.J.; Mohr, F.W.; et al. MiR-29b Participates in Early Aneurysm Development in Marfan Syndrome. Circ. Res. 2012, 110, 312–324. [Google Scholar] [CrossRef] [PubMed]
- Chakraborty, C.; Sharma, A.R.; Sharma, G.; Lee, S.S. Therapeutic Advances of MiRNAs: A Preclinical and Clinical Update. J. Adv. Res. 2020, 28, 127–138. [Google Scholar] [CrossRef] [PubMed]
- Moraes, F.C.; Pichon, C.; Letourneur, D.; Chaubet, F. MiRNA Delivery by Nanosystems: State of the Art and Perspectives. Pharmaceutics 2021, 13, 1901. [Google Scholar] [CrossRef] [PubMed]
- Roberts, T.C.; Langer, R.; Wood, M.J.A. Advances in Oligonucleotide Drug Delivery. Nat. Rev. Drug Discov. 2020, 19, 673–694. [Google Scholar] [CrossRef]
- Hanna, J.; Hossain, G.S.; Kocerha, J. The Potential for MicroRNA Therapeutics and Clinical Research. Front. Genet. 2019, 10, 478. [Google Scholar] [CrossRef] [PubMed]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chiarelli, N.; Cinquina, V.; Zoppi, N.; Bertini, V.; Maddaluno, M.; De Leonibus, C.; Settembre, C.; Venturini, M.; Colombi, M.; Ritelli, M. Integrative Multi-Omics Approach in Vascular Ehlers–Danlos Syndrome: Further Insights into the Disease Mechanisms by Proteomic Analysis of Patient Dermal Fibroblasts. Biomedicines 2024, 12, 2749. https://doi.org/10.3390/biomedicines12122749
Chiarelli N, Cinquina V, Zoppi N, Bertini V, Maddaluno M, De Leonibus C, Settembre C, Venturini M, Colombi M, Ritelli M. Integrative Multi-Omics Approach in Vascular Ehlers–Danlos Syndrome: Further Insights into the Disease Mechanisms by Proteomic Analysis of Patient Dermal Fibroblasts. Biomedicines. 2024; 12(12):2749. https://doi.org/10.3390/biomedicines12122749
Chicago/Turabian StyleChiarelli, Nicola, Valeria Cinquina, Nicoletta Zoppi, Valeria Bertini, Marianna Maddaluno, Chiara De Leonibus, Carmine Settembre, Marina Venturini, Marina Colombi, and Marco Ritelli. 2024. "Integrative Multi-Omics Approach in Vascular Ehlers–Danlos Syndrome: Further Insights into the Disease Mechanisms by Proteomic Analysis of Patient Dermal Fibroblasts" Biomedicines 12, no. 12: 2749. https://doi.org/10.3390/biomedicines12122749
APA StyleChiarelli, N., Cinquina, V., Zoppi, N., Bertini, V., Maddaluno, M., De Leonibus, C., Settembre, C., Venturini, M., Colombi, M., & Ritelli, M. (2024). Integrative Multi-Omics Approach in Vascular Ehlers–Danlos Syndrome: Further Insights into the Disease Mechanisms by Proteomic Analysis of Patient Dermal Fibroblasts. Biomedicines, 12(12), 2749. https://doi.org/10.3390/biomedicines12122749











