DNA Methylation Demonstrates Bronchoalveolar Cell Senescence in People Living with HIV: An Observational Cohort Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Cohort
2.2. DNA Methylation Profiling
2.3. Alu and LINE-1 Imputation
2.4. Weighted Gene Correlation Network Analysis (WGCNA)
2.5. Differential Methylation Analysis
2.6. Pathway Enrichment Analysis
3. Results
3.1. Study Overview
3.2. Global Hypomethylation Is Observed in the BAL Cells of PLWH, but Not in COPD
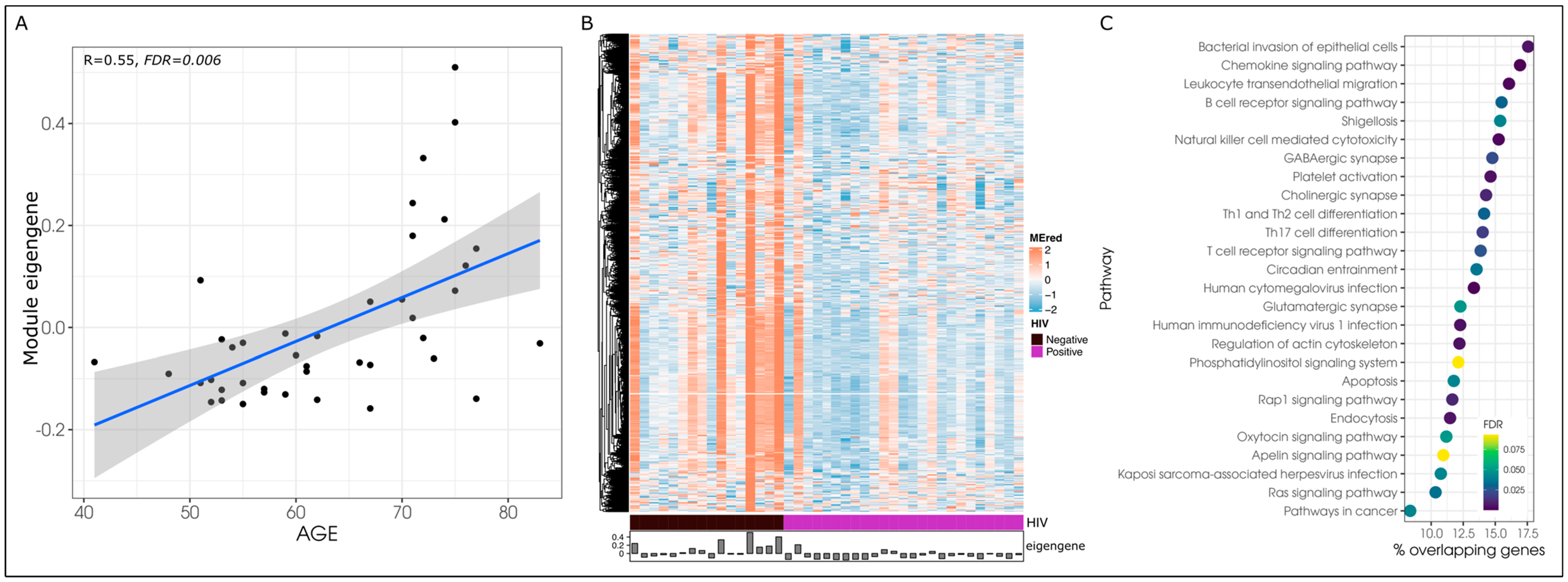
3.3. Age-Associated Co-Methylation Networks Are Dysregulated in HIV
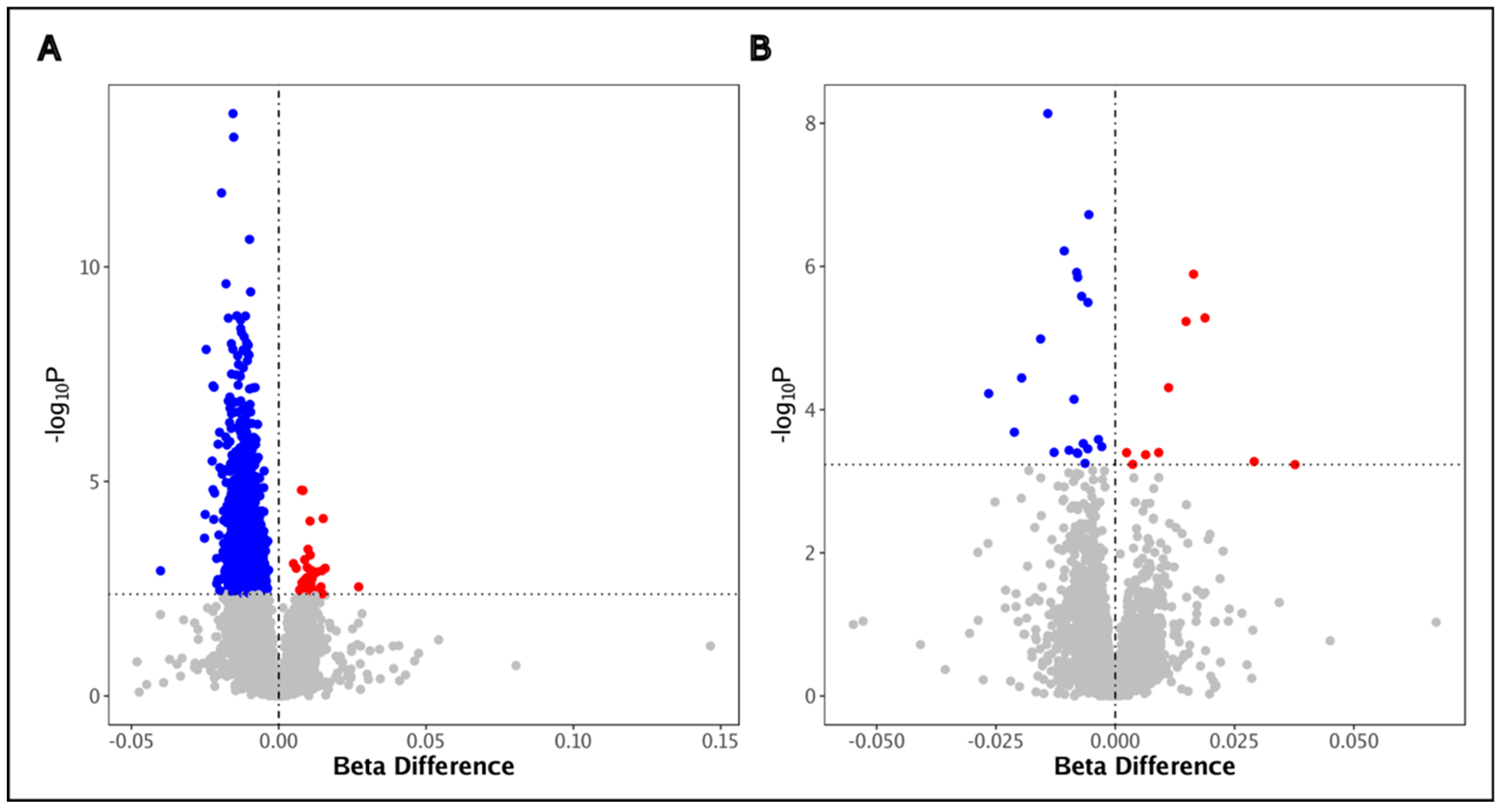
3.4. HIV Is Associated with DNA Methylation Alterations in BAL Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Currier, J.S.; Taylor, A.; Boyd, F.; Dezii, C.M.; Kawabata, H.; Burtcel, B.; Maa, J.-F.; Hodder, S. Coronary Heart Disease in HIV-Infected Individuals. JAIDS J. Acquir. Immune Defic. Syndr. 2003, 33, 506–512. [Google Scholar] [CrossRef] [PubMed]
- Obel, N.; Thomsen, H.F.; Kronborg, G.; Larsen, C.S.; Hildebrandt, P.R.; Sørensen, H.T.; Gerstoft, J. Ischemic Heart Disease in HIV-Infected and HIV-Uninfected Individuals: A Population-Based Cohort Study. Clin. Infect. Dis. 2007, 44, 1625–1631. [Google Scholar] [CrossRef] [PubMed]
- Lucas, G.M.; Lau, B.; Atta, M.G.; Fine, D.M.; Keruly, J.; Moore, R.D. Chronic Kidney Disease Incidence, and Progression to End-Stage Renal Disease, in HIV-Infected Individuals: A Tale of Two Races. J. Infect. Dis. 2008, 197, 1548–1557. [Google Scholar] [CrossRef] [PubMed]
- FFitzpatrick, M.E.; Kunisaki, K.M.; Morris, A. Pulmonary Disease in HIV-Infected Adults in the Era of Antiretroviral Therapy. AIDS Lond. Engl. 2018, 32, 277–292. [Google Scholar] [CrossRef] [PubMed]
- Wing, E.J. HIV and Aging. Int. J. Infect. Dis. 2016, 53, 61–68. [Google Scholar] [CrossRef] [PubMed]
- Brothers, T.D.; Rockwood, K. Biologic Aging, Frailty, and Age-Related Disease in Chronic HIV Infection. Curr. Opin. HIV AIDS 2014, 9, 412. [Google Scholar] [CrossRef]
- Konstantinidis, I.; Crothers, K.; Kunisaki, K.M.; Drummond, M.B.; Benfield, T.; Zar, H.J.; Huang, L.; Morris, A. HIV-Associated Lung Disease. Nat. Rev. Dis. Primer 2023, 9, 39. [Google Scholar] [CrossRef] [PubMed]
- Yang, C.X.; Schon, E.; Obeidat, M.; Kobor, M.S.; McEwen, L.; MacIsaac, J.; Lin, D.; Novak, R.M.; Hudson, F.; Klinker, H.; et al. Occurrence of Accelerated Epigenetic Aging and Methylation Disruptions in Human Immunodeficiency Virus Infection Before Antiretroviral Therapy. J. Infect. Dis. 2021, 223, 1681–1689. [Google Scholar] [CrossRef]
- Jones, P.A. Functions of DNA Methylation: Islands, Start Sites, Gene Bodies and Beyond. Nat. Rev. Genet. 2012, 13, 484–492. [Google Scholar] [CrossRef]
- Hernández Cordero, A.I.; Yang, C.X.; Yang, J.; Horvath, S.; Shaipanich, T.; MacIsaac, J.; Lin, D.T.S.; Kobor, M.S.; Guillemi, S.; Harris, M.; et al. Airway Aging and Methylation Disruptions in HIV-Associated Chronic Obstructive Pulmonary Disease. Am. J. Respir. Crit. Care Med. 2022, 206, 150–160. [Google Scholar] [CrossRef]
- Hernández Cordero, A.I.; Yang, C.X.; Yang, J.; Li, X.; Horvath, S.; Shaipanich, T.; MacIsaac, J.; Lin, D.; McEwen, L.; Kobor, M.S.; et al. The Relationship between the Epigenetic Aging Biomarker “Grimage” and Lung Function in Both the Airway and Blood of People Living with HIV: An Observational Cohort Study. EBioMedicine 2022, 83, 104206. [Google Scholar] [CrossRef]
- Hernandez Cordero, A.I.; Yang, C.X.; Obeidat, M.; Yang, J.; MacIsaac, J.; McEwen, L.; Lin, D.; Kobor, M.; Novak, R.; Hudson, F.; et al. DNA Methylation Is Associated with Airflow Obstruction in Patients Living with HIV. Thorax 2021, 76, 448–455. [Google Scholar] [CrossRef]
- Erichsen, L.; Beermann, A.; Arauzo-Bravo, M.J.; Hassan, M.; Dkhil, M.A.; Al-Quraishy, S.; Hafiz, T.A.; Fischer, J.C.; Santourlidis, S. Genome-Wide Hypomethylation of LINE-1 and Alu Retroelements in Cell-Free DNA of Blood Is an Epigenetic Biomarker of Human Aging. Saudi J. Biol. Sci. 2018, 25, 1220–1226. [Google Scholar] [CrossRef]
- Wilson, A.S.; Power, B.E.; Molloy, P.L. DNA Hypomethylation and Human Diseases. Biochim. Biophys. Acta 2007, 1775, 138–162. [Google Scholar] [CrossRef]
- Leitao Filho, F.S.; Takiguchi, H.; Akata, K.; Ra, S.W.; Moon, J.-Y.; Kim, H.K.; Cho, Y.; Yamasaki, K.; Milne, S.; Yang, J.; et al. Effects of Inhaled Corticosteroid/Long-Acting Β2-Agonist Combination on the Airway Microbiome of Patients with Chronic Obstructive Pulmonary Disease: A Randomized Controlled Clinical Trial (DISARM). Am. J. Respir. Crit. Care Med. 2021, 204, 1143–1152. [Google Scholar] [CrossRef]
- Ho, C.G.; Milne, S.; Li, X.; Yang, C.X.; Leitao Filho, F.S.; Cheung, C.Y.; Yang, J.S.W.; Hernández Cordero, A.I.; Yang, C.W.T.; Shaipanich, T.; et al. Airway Eosinophilia on Bronchoalveolar Lavage and the Risk of Exacerbations in COPD. Biomedicines 2022, 10, 1412. [Google Scholar] [CrossRef]
- Graham, B.L.; Steenbruggen, I.; Miller, M.R.; Barjaktarevic, I.Z.; Cooper, B.G.; Hall, G.L.; Hallstrand, T.S.; Kaminsky, D.A.; McCarthy, K.; McCormack, M.C.; et al. Standardization of Spirometry 2019 Update. An Official American Thoracic Society and European Respiratory Society Technical Statement. Am. J. Respir. Crit. Care Med. 2019, 200, e70–e88. [Google Scholar] [CrossRef]
- Triche, T.J.; Weisenberger, D.J.; Van Den Berg, D.; Laird, P.W.; Siegmund, K.D. Low-Level Processing of Illumina Infinium DNA Methylation BeadArrays. Nucleic Acids Res. 2013, 41, e90. [Google Scholar] [CrossRef]
- Teschendorff, A.E.; Marabita, F.; Lechner, M.; Bartlett, T.; Tegner, J.; Gomez-Cabrero, D.; Beck, S. A Beta-Mixture Quantile Normalization Method for Correcting Probe Design Bias in Illumina Infinium 450 k DNA Methylation Data. Bioinformatics 2013, 29, 189–196. [Google Scholar] [CrossRef]
- Hernandez Cordero, A.I.; Yang, C.X.; Li, X.; Yang, J.; Shaipanich, T.; MacIsaac, J.L.; Lin, D.T.S.; Kobor, M.S.; Horvath, S.; Man, S.F.P.; et al. The Blood DNA Methylation Clock GrimAge Is a Robust Surrogate for Airway Epithelia Aging. Biomedicines 2022, 10, 3094. [Google Scholar] [CrossRef]
- Hernandez Cordero, A.I.; Li, X.; Yang, C.X.; Yang, J.; MacIsaac, J.L.; Dever, K.; Kobor, M.S.; Milne, S.; van Eeden, S.F.; Shaipanich, T.; et al. Systemic and Airway Epigenetic Disruptions Are Associated with Health Status in COPD. Biomedicines 2023, 11, 134. [Google Scholar] [CrossRef] [PubMed]
- Rahmani, E.; Shenhav, L.; Schweiger, R.; Yousefi, P.; Huen, K.; Eskenazi, B.; Eng, C.; Huntsman, S.; Hu, D.; Galanter, J.; et al. Genome-Wide Methylation Data Mirror Ancestry Information. Epigenetics Chromatin 2017, 10, 1. [Google Scholar] [CrossRef] [PubMed]
- Houseman, E.A.; Accomando, W.P.; Koestler, D.C.; Christensen, B.C.; Marsit, C.J.; Nelson, H.H.; Wiencke, J.K.; Kelsey, K.T. DNA Methylation Arrays as Surrogate Measures of Cell Mixture Distribution. BMC Bioinform. 2012, 13, 86. [Google Scholar] [CrossRef] [PubMed]
- Yang, A.S.; Estécio, M.R.H.; Doshi, K.; Kondo, Y.; Tajara, E.H.; Issa, J.-P.J. A Simple Method for Estimating Global DNA Methylation Using Bisulfite PCR of Repetitive DNA Elements. Nucleic Acids Res. 2004, 32, e38. [Google Scholar] [CrossRef] [PubMed]
- Lange, N.E.; Sordillo, J.; Tarantini, L.; Bollati, V.; Sparrow, D.; Vokonas, P.; Zanobetti, A.; Schwartz, J.; Baccarelli, A.; Litonjua, A.A.; et al. Alu and LINE-1 Methylation and Lung Function in the Normative Ageing Study. BMJ Open 2012, 2, e001231. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Joyce, B.T.; Liu, L.; Zhang, Z.; Kibbe, W.A.; Zhang, W.; Hou, L. Prediction of Genome-Wide DNA Methylation in Repetitive Elements. Nucleic Acids Res. 2017, 45, 8697–8711. [Google Scholar] [CrossRef]
- Langfelder, P.; Horvath, S. WGCNA: An R Package for Weighted Correlation Network Analysis. BMC Bioinform. 2008, 9, 559. [Google Scholar] [CrossRef] [PubMed]
- Seale, K.; Horvath, S.; Teschendorff, A.; Eynon, N.; Voisin, S. Making Sense of the Ageing Methylome. Nat. Rev. Genet. 2022, 23, 585–605. [Google Scholar] [CrossRef] [PubMed]
- Venables, W.N.; Ripley, B.D. Modern Applied Statistics with S, 4th ed.; Statistics and Computing, Statistics, Computing Venables, W.N.: Statistics w.S-PLUS.; Springer: New York, NY, USA, 2002; ISBN 978-0-387-95457-8. [Google Scholar]
- Yu, G.; Wang, L.-G.; Han, Y.; He, Q.-Y. clusterProfiler: An R Package for Comparing Biological Themes Among Gene Clusters. OMICS J. Integr. Biol. 2012, 16, 284–287. [Google Scholar] [CrossRef]
- Liao, Y.; Wang, J.; Jaehnig, E.J.; Shi, Z.; Zhang, B. WebGestalt 2019: Gene Set Analysis Toolkit with Revamped UIs and APIs. Nucleic Acids Res. 2019, 47, W199–W205. [Google Scholar] [CrossRef]
- Schoepf, I.C.; Esteban-Cantos, A.; Thorball, C.W.; Rodés, B.; Reiss, P.; Rodríguez-Centeno, J.; Riebensahm, C.; Braun, D.L.; Marzolini, C.; Seneghini, M.; et al. Epigenetic Ageing Accelerates before Antiretroviral Therapy and Decelerates after Viral Suppression in People with HIV in Switzerland: A Longitudinal Study over 17 Years. Lancet Healthy Longev. 2023, 4, e211–e218. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, H.; Koga, H.; Katoh, Y.; Takahashi, S.; Nakayama, K.; Shin, H.-W. Functional Cross-Talk between Rab14 and Rab4 through a Dual Effector, RUFY1/Rabip4. Mol. Biol. Cell 2010, 21, 2746–2755. [Google Scholar] [CrossRef]
- Cormont, M.; Mari, M.; Galmiche, A.; Hofman, P.; Le Marchand-Brustel, Y. A FYVE-Finger-Containing Protein, Rabip4, Is a Rab4 Effector Involved in Early Endosomal Traffic. Proc. Natl. Acad. Sci. USA 2001, 98, 1637–1642. [Google Scholar] [CrossRef]
- Nag, S.; Rani, S.; Mahanty, S.; Bissig, C.; Arora, P.; Azevedo, C.; Saiardi, A.; van der Sluijs, P.; Delevoye, C.; van Niel, G.; et al. Rab4A Organizes Endosomal Domains for Sorting Cargo to Lysosome-Related Organelles. J. Cell Sci. 2018, 131, jcs216226. [Google Scholar] [CrossRef] [PubMed]
- Ohno, H.; Aguilar, R.C.; Fournier, M.C.; Hennecke, S.; Cosson, P.; Bonifacino, J.S. Interaction of Endocytic Signals from the HIV-1 Envelope Glycoprotein Complex with Members of the Adaptor Medium Chain Family. Virology 1997, 238, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Oriol-Tordera, B.; Esteve-Codina, A.; Berdasco, M.; Rosás-Umbert, M.; Gonçalves, E.; Duran-Castells, C.; Català-Moll, F.; Llano, A.; Cedeño, S.; Puertas, M.C.; et al. Epigenetic Landscape in the Kick-and-Kill Therapeutic Vaccine BCN02 Clinical Trial Is Associated with Antiretroviral Treatment Interruption (ATI) Outcome. EBioMedicine 2022, 78, 103956. [Google Scholar] [CrossRef]
- Dickson, S.J.; Batson, S.; Copas, A.J.; Edwards, S.G.; Singer, M.; Miller, R.F. Survival of HIV-Infected Patients in the Intensive Care Unit in the Era of Highly Active Antiretroviral Therapy. Thorax 2007, 62, 964–968. [Google Scholar] [CrossRef]
- Crothers, K.; Huang, L.; Goulet, J.L.; Goetz, M.B.; Brown, S.T.; Rodriguez-Barradas, M.C.; Oursler, K.K.; Rimland, D.; Gibert, C.L.; Butt, A.A.; et al. HIV Infection and Risk for Incident Pulmonary Diseases in the Combination Antiretroviral Therapy Era. Am. J. Respir. Crit. Care Med. 2011, 183, 388–395. [Google Scholar] [CrossRef]
- Twigg, H.L.; Soliman, D.M.; Day, R.B.; Knox, K.S.; Anderson, R.J.; Wilkes, D.S.; Schnizlein-Bick, C.T. Lymphocytic Alveolitis, Bronchoalveolar Lavage Viral Load, and Outcome in Human Immunodeficiency Virus Infection. Am. J. Respir. Crit. Care Med. 1999, 159, 1439–1444. [Google Scholar] [CrossRef]
- Costiniuk, C.T.; Salahuddin, S.; Farnos, O.; Olivenstein, R.; Pagliuzza, A.; Orlova, M.; Schurr, E.; De Castro, C.; Bourbeau, J.; Routy, J.-P.; et al. HIV Persistence in Mucosal CD4+ T Cells within the Lungs of Adults Receiving Long-Term Suppressive Antiretroviral Therapy. AIDS Lond. Engl. 2018, 32, 2279–2289. [Google Scholar] [CrossRef]
- Collini, P.J.; Bewley, M.A.; Mohasin, M.; Marriott, H.M.; Miller, R.F.; Geretti, A.-M.; Beloukas, A.; Papadimitropoulos, A.; Read, R.C.; Noursadeghi, M.; et al. HIV Gp120 in the Lungs of Antiretroviral Therapy-Treated Individuals Impairs Alveolar Macrophage Responses to Pneumococci. Am. J. Respir. Crit. Care Med. 2018, 197, 1604–1615. [Google Scholar] [CrossRef] [PubMed]

| PLWH | HIV-Uninfected | p | |
|---|---|---|---|
| n | 25 | 16 | - |
| Male Sex (%) | 92 | 56 | 0.016 |
| Age, years | 57.00 (53.00–62.00) | 72.00 (70.00–75.00) | 7.844 × 10−4 |
| BMI, kg/m2 | 24.54 (22.02–27.91) | 26.07 (24.66–29.41) | 0.279 |
| Pre-bronchodilator FEV1, L | 2.650 (2.06–3.06) | 2.490 (2.08–2.65) | 0.310 |
| Pre-bronchodilator FVC, L | 4.07 (3.51–4.71) | 3.24 (2.87–3.74) | 0.010 |
| Pre-bronchodilator FEV1/FVC (%) | 62.20 (54.93–74.94) | 74.13 (68.44–76.62) | 0.048 |
| Pre-bronchodilator FEV1% predicted | 77.00 (65.00–91.00) | 87.00 (74.25–96.75) | 0.195 |
| Pre-bronchodilator FEV1/FVC % predicted | 93.10 (81.00–105.00) | 92.00 (74.50–107.65) | 0.820 |
| Smoking status | 0.258 | ||
| Current smoker, (%) | 36 | 12 | |
| Former smoker, (%) | 56 | 69 | |
| Never smoker, (%) | 8 | 19 | |
| Smoking pack-years | 30 (11.50–45.00) | 30 (12.50–37.50) | 0.450 |
| Physician-diagnosed COPD, (%) | 76 | 25 | 0.003 |
| Bronchiectasis, (%) | 4 | 0 | ~1 |
| BAL neutrophils, (%) | 2.00 (1.00–5.50) | 3.63 (1.19–6.38) | 0.462 |
| BAL lymphocytes, (%) | 2.50 (1.50–5.50) | 2.75 (1.00–3.75) | 0.758 |
| BAL eosinophils, (%) | 0.25 (0.00–1.00) | 0.63 (0.23–4.06) | 0.041 |
| BAL macrophage, (%) | 92.50 (79.50–95.00) | 91.25 (73.75–94.25) | 0.679 |
| Inhaled corticosteroid use, (%) | 12 | 19 | 0.662 |
| CD4 T-cell count, cell/mm3 | 435.00 (355.00–590.00) | - | - |
| HIV viral load undetectable, (%) | 92 | - | - |
| ART, (%) | 96 | - | - |
| Hypertension, (%) | 20 | 44 | 0.161 |
| Diabetes, (%) | 8 | 19 | 0.362 |
| Module | #CpGs | #Genes | Effect | |||||
|---|---|---|---|---|---|---|---|---|
| Age | HIV | |||||||
| p-Value | FDR | R | p-Value | FDR | p-Value Age-Adj | |||
| Skyblue | 412 | 426 | 1.89 × 10−5 | 0.001 | 0.56 | 0.001 | 0.002 | 0.056 |
| Red | 2233 | 1150 | 1.44 × 10−4 | 0.006 | 0.55 | 4.50 × 10−4 | 0.002 | 0.044 |
| Yellow | 2394 | 1900 | 2.29 × 10−4 | 0.006 | −0.61 | 0.001 | 0.002 | 0.127 |
| Palevioletred3 | 142 | 150 | 2.54 × 10−3 | 0.043 | −0.46 | 0.003 | 0.006 | 0.103 |
| Pink | 1579 | 1346 | 3.38 × 10−3 | 0.043 | −0.45 | 0.007 | 0.007 | 0.151 |
| Floralwhite | 210 | 163 | 2.84 × 10−3 | 0.043 | 0.45 | 0.007 | 0.007 | 0.173 |
| Probe | Chr | p | FDR | Beta Difference (Reference Group: HIV-Uninfected) | Relation to CpG Island | Gene Symbol |
|---|---|---|---|---|---|---|
| Criteria: Lowest FDR | ||||||
| cg26126053 | 10 | 1.43 × 10−22 | 1.12 × 10−16 | −0.011 | Open Sea | DHX32 |
| cg00401660 | 3 | 8.05 × 10−18 | 3.17 × 10−12 | 0.018 | Open Sea | CPN2 |
| cg14118535 | 1 | 7.17 × 10−17 | 1.88 × 10−11 | −0.013 | Open Sea | FUBP1 |
| cg08589141 | 19 | 2.00 × 10−16 | 3.94 × 10−11 | 0.029 | North Shore | RGL3 |
| cg16636316 | 11 | 3.52 × 10−16 | 5.55 × 10−11 | 0.034 | Open Sea | OTOG |
| Criteria: Greatest Beta Difference | ||||||
| cg09733528 | 2 | 3.40 × 10−7 | 9.04 × 10−4 | −0.316 | Open Sea | ABCB11 |
| cg02630646 | 2 | 6.08 × 10−5 | 2.35 × 10−2 | −0.314 | Open Sea | -- |
| cg11424828 | 8 | 4.02 × 10−4 | 6.82 × 10−2 | 0.273 | Island | MYOM2 |
| cg21015022 | 7 | 4.07 × 10−5 | 1.87 × 10−2 | 0.254 | South Shore | PARP12 |
| cg17975832 | 12 | 7.01 × 10−4 | 9.21 × 10−2 | −0.250 | Open Sea | 7SK |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernandez Cordero, A.I.; Li, X.; Yang, J.; Yang, C.X.; Shaipanich, T.; MacIsaac, J.L.; Dever, K.; Kobor, M.S.; Montaner, J.; Harris, M.; et al. DNA Methylation Demonstrates Bronchoalveolar Cell Senescence in People Living with HIV: An Observational Cohort Study. Biomedicines 2024, 12, 1261. https://doi.org/10.3390/biomedicines12061261
Hernandez Cordero AI, Li X, Yang J, Yang CX, Shaipanich T, MacIsaac JL, Dever K, Kobor MS, Montaner J, Harris M, et al. DNA Methylation Demonstrates Bronchoalveolar Cell Senescence in People Living with HIV: An Observational Cohort Study. Biomedicines. 2024; 12(6):1261. https://doi.org/10.3390/biomedicines12061261
Chicago/Turabian StyleHernandez Cordero, Ana I., Xuan Li, Julia Yang, Chen Xi Yang, Tawimas Shaipanich, Julie L. MacIsaac, Kristy Dever, Michael S. Kobor, Julio Montaner, Marianne Harris, and et al. 2024. "DNA Methylation Demonstrates Bronchoalveolar Cell Senescence in People Living with HIV: An Observational Cohort Study" Biomedicines 12, no. 6: 1261. https://doi.org/10.3390/biomedicines12061261
APA StyleHernandez Cordero, A. I., Li, X., Yang, J., Yang, C. X., Shaipanich, T., MacIsaac, J. L., Dever, K., Kobor, M. S., Montaner, J., Harris, M., Guillemi, S., Man, S. F. P., Sin, D. D., & Leung, J. M. (2024). DNA Methylation Demonstrates Bronchoalveolar Cell Senescence in People Living with HIV: An Observational Cohort Study. Biomedicines, 12(6), 1261. https://doi.org/10.3390/biomedicines12061261





