CRISPR-Cas System, Antimicrobial Resistance, and Enterococcus Genus—A Complicated Relationship
Abstract
1. Introduction
2. Materials and Methods
2.1. Identification of Enterococcus Isolates
2.2. Genome Retrieval and Database Generation
- CRISPRCasdb Interface: The genomes of Enterococcus isolates are shown with CRISPR-Cas elements identified. The “Download Data” button allows users to download the genomic data.
- FASTA Format Sequence: Following data retrieval, the acquired information is depicted in the FASTA format, sourced from NCBI’s comprehensive nucleotide database. This format ensures accessibility and compatibility for subsequent analytical steps.
- Sequence Compilation in BioEdit: In BioEdit, genomes can be introduced by importing sequence data files in various formats. Users typically navigate to the “File” menu and select options such as “Open” or “Import” to load sequence files. Common file formats supported by BioEdit include FASTA, GenBank, and other commonly used sequence file formats. Once imported, the sequences are displayed in the BioEdit workspace, where users can perform various sequence manipulation and analysis tasks. The procured sequence data undergo compilation and comprehensive analysis within the BioEdit software environment. Here, the software showcases the aligned sequences, furnishing researchers with a structured platform for in-depth exploration and investigation.
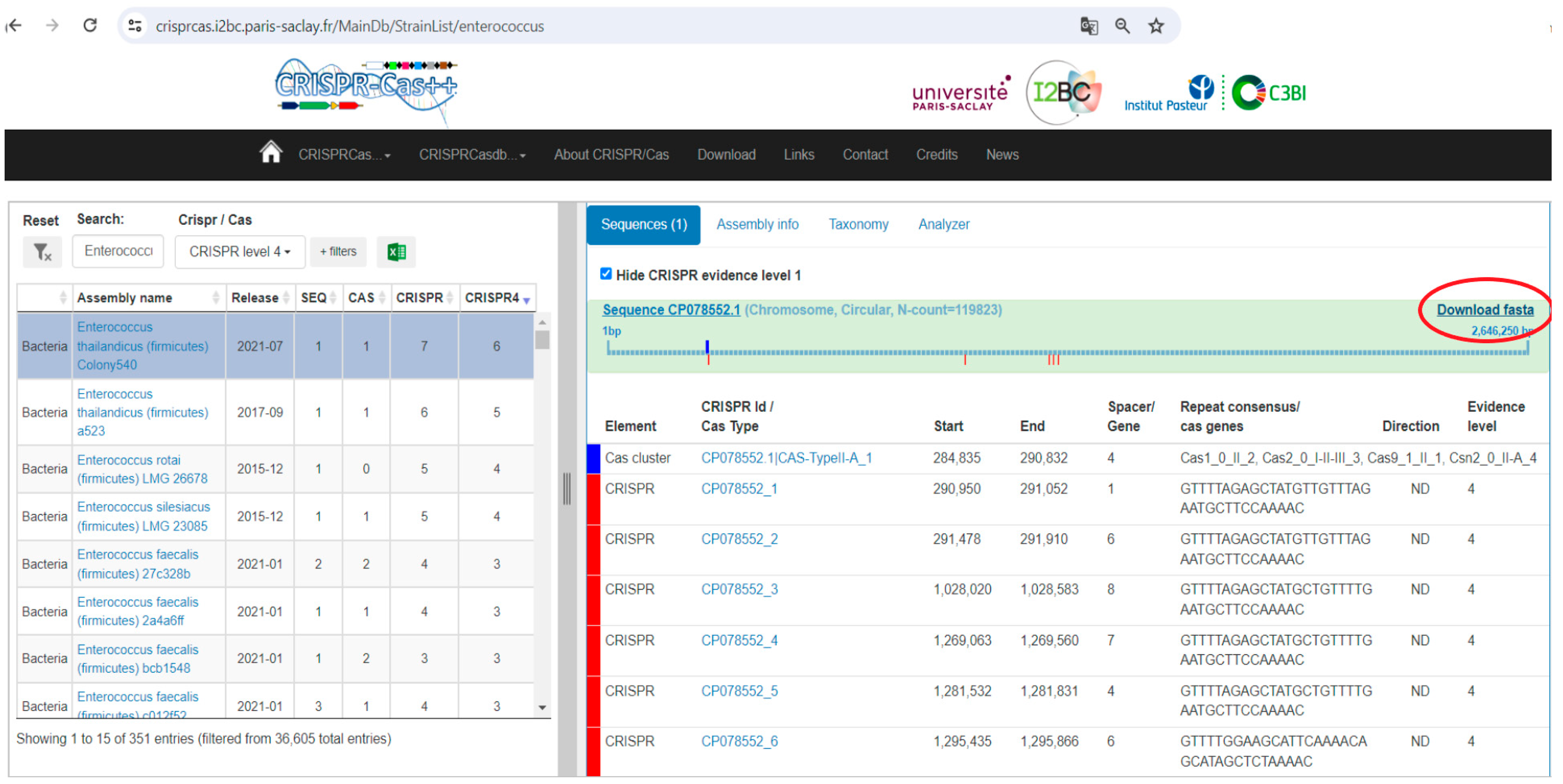
2.3. Identification of Antibiotic Resistance Genes and Statistical Analysis
3. Results and Discussion
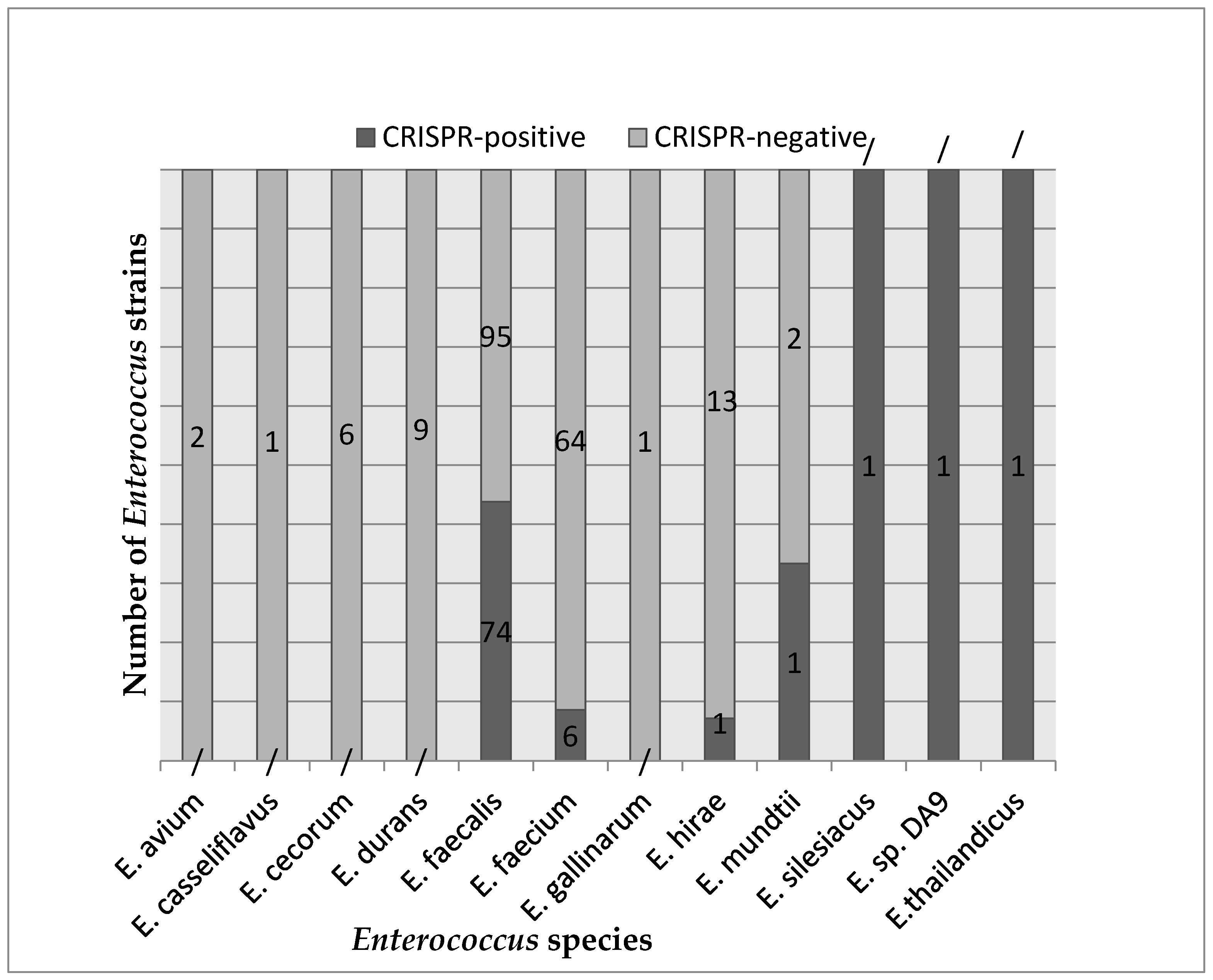
3.1. Comparative Analysis of CRISPR-Positive and CRISPR-Negative Enterococcus Genomes
3.2. Comparative Analysis of the Antibiotic Resistance Genes Found in the CRISPR-Positive and CRISPR-Negative Isolates
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- García-Solache, M.; Rice, L.B. The Enterococcus: A Model of Adaptability to Its Environment. Clin. Microbiol. Rev. 2019, 32, e00058-18. [Google Scholar] [CrossRef]
- Torres, C.; Alonso, C.A.; Ruiz-Ripa, L.; León-Sampedro, R.; Del Campo, R.; Coque, T.M. Antimicrobial Resistance in Enterococcus spp. of animal origin. Microbiol. Spectr. 2018, 6, 1–2. [Google Scholar] [CrossRef]
- Fiore, E.; Van Tyne, D.; Gilmore, M.S. Pathogenicity of Enterococci. Microbiol. Spectr. 2019, 7, 1–2. [Google Scholar] [CrossRef]
- Kim, M.-A.; Rosa, V.; Min, K.-S. Characterization of Enterococcus faecalis in different culture conditions. Sci. Rep. 2020, 10, 21867. [Google Scholar] [CrossRef]
- Toc, D.A.; Pandrea, S.L.; Botan, A.; Mihaila, R.M.; Costache, C.A.; Colosi, I.A.; Junie, L.M. Enterococcus raffinosus, Enterococcus durans and Enterococcus avium Isolated from a Tertiary Care Hospital in Romania—Retrospective Study and Brief Review. Biology 2022, 11, 598. [Google Scholar] [CrossRef]
- Monticelli, J.; Knezevich, A.; Luzzati, R.; Di Bella, S. Clinical management of non-faecium non-faecalis vancomycin-resistant enterococci infection. Focus on Enterococcus gallinarum and Enterococcus casseliflavus/flavescens. J. Infect. Chemother. 2018, 24, 237–246. [Google Scholar] [CrossRef]
- Butiuc-Keul, A.; Farkas, A.; Carpa, R.; Iordache, D. CRISPR-Cas System: The Powerful Modulator of Accessory Genomes in Prokaryotes. Microb. Physiol. 2022, 32, 2–17. [Google Scholar] [CrossRef]
- Ishino, Y.; Krupovic, M.; Forterre, P. History of CRISPR-Cas from Encounter with a Mysterious Repeated Sequence to Genome Editing Technology. J. Bacteriol. 2018, 200, e00580-17. [Google Scholar] [CrossRef]
- Iordache, D.; Baci, G.-M.; Căpriță, O.; Farkas, A.; Lup, A.; Butiuc-Keul, A. Correlation between CRISPR Loci Diversity in Three Enterobacterial Taxa. Int. J. Mol. Sci. 2022, 23, 12766. [Google Scholar] [CrossRef]
- Wang, H.; La Russa, M.; Qi, L.S. CRISPR/Cas9 in Genome Editing and Beyond. Annu. Rev. Biochem. 2016, 85, 227–264. [Google Scholar] [CrossRef]
- Barrangou, R.; Sontheimer, E.J.; Marraffini, L.A. Crispr: Biology and Applications; Wiley: Hoboken, NJ, USA, 2022; ISBN 9781683673798. [Google Scholar]
- Khambhati, K.; Bhattacharjee, G.; Gohil, N.; Dhanoa, G.K.; Sagona, A.P.; Mani, I.; Bui, N.L.; Chu, D.; Karapurkar, J.K.; Jang, S.H.; et al. Phage engineering and phage-assisted CRISPR-Cas delivery to combat multidrug-resistant pathogens. Bioeng. Transl. Med. 2023, 8, e10381. [Google Scholar] [CrossRef]
- Duan, C.; Cao, H.; Zhang, L.-H.; Xu, Z. Harnessing the CRISPR-Cas Systems to Combat Antimicrobial Resistance. Front. Microbiol. 2021, 12, 716064. [Google Scholar] [CrossRef]
- Tao, S.; Chen, H.; Li, N.; Liang, W. The Application of the CRISPR-Cas System in Antibiotic Resistance. Infect. Drug Resist. 2022, 15, 4155–4168. [Google Scholar] [CrossRef] [PubMed]
- Marraffini, L.A.; Sontheimer, E.J. CRISPR Interference Limits Horizontal Gene Transfer in Staphylococci by Targeting DNA. Science 2008, 322, 1843–1845. [Google Scholar] [CrossRef]
- Zhang, Q.; Ye, Y. Not all predicted CRISPR–Cas systems are equal: Isolated cas genes and classes of CRISPR like elements. BMC Bioinform. 2017, 18, 92. [Google Scholar] [CrossRef] [PubMed]
- Palmer, K.L.; Gilmore, M.S. Multidrug-Resistant Enterococci Lack CRISPR-cas. MBio 2010, 1, e00227-10. [Google Scholar] [CrossRef]
- Price, V.J.; Huo, W.; Sharifi, A.; Palmer, K.L. CRISPR-Cas and Restriction-Modification Act Additively against Conjugative Antibiotic Resistance Plasmid Transfer in Enterococcus faecalis. mSphere 2016, 1, e00064-16. [Google Scholar] [CrossRef] [PubMed]
- Sycheva, M.V.; Kartashova, O.L.; Shchepitova, N.E.; Safronov, A.A. Antibiotic Resistance of Enterococci Isolated from Healthy Humans and Patients with Various Pathologies. Antibiot. Khimioter. 2016, 61, 27–32. Available online: http://www.ncbi.nlm.nih.gov/pubmed/29533558 (accessed on 17 July 2024).
- Marin Garrido, A. Antimicrobial Resistance in Enterococci. J. Infect. Dis. Ther. 2014, 2, 150. [Google Scholar] [CrossRef]
- Sivertsen, A.; Pedersen, T.; Larssen, K.W.; Bergh, K.; Rønning, T.G.; Radtke, A.; Hegstad, K. A Silenced vanA Gene Cluster on a Transferable Plasmid Caused an Outbreak of Vancomycin-Variable Enterococci. Antimicrob. Agents Chemother. 2016, 60, 4119–4127. [Google Scholar] [CrossRef]
- Wardal, E.; Żabicka, D.; Hryniewicz, W.; Sadowy, E. VanA-Enterococcus faecalis in Poland: Hospital population clonal structure and vanA mobilome. Eur. J. Clin. Microbiol. Infect. Dis. 2022, 41, 1245–1261. [Google Scholar] [CrossRef]
- Sun, L.; Chen, Y.; Hua, X.; Chen, Y.; Hong, J.; Wu, X.; Jiang, Y.; van Schaik, W.; Qu, T.; Yu, Y. Tandem amplification of the vanM gene cluster drives vancomycin resistance in vancomycin-variable enterococci. J. Antimicrob. Chemother. 2020, 75, 283–291. [Google Scholar] [CrossRef]
- Nishioka, T.; Ogawa, W.; Kuroda, T.; Katsu, T.; Tsuchiya, T. Gene Cloning and Characterization of EfmA, a Multidrug Efflux Pump, from Enterococcus faecium. Biol. Pharm. Bull. 2009, 32, 483–488. [Google Scholar] [CrossRef]
- Reynolds, E.D.; Cove, J.H. Resistance to telithromycin is conferred by msr(A), msrC and msr(D) in Staphylococcus aureus. J. Antimicrob. Chemother. 2005, 56, 1179–1180. [Google Scholar] [CrossRef]
- Belloso Daza, M.V.; Milani, G.; Cortimiglia, C.; Pietta, E.; Bassi, D.; Cocconcelli, P.S. Genomic Insights of Enterococcus faecium UC7251, a Multi-Drug Resistant Strain From Ready-to-Eat Food, Highlight the Risk of Antimicrobial Resistance in the Food Chain. Front. Microbiol. 2022, 13, 894241. [Google Scholar] [CrossRef]
- Cordeiro-Moura, J.R.; Kraychete, G.B.; de Araújo Longo, L.G.; Corrêa, L.L.; da Silva, N.M.V.; Campana, E.H.; Oliveira, C.J.B.; Picão, R.C. Description and comparative genomic analysis of a mcr-1-carrying Escherichia coli ST683/CC155 recovered from touristic coastal water in Northeastern Brazil. Infect. Genet. Evol. 2022, 97, 105196. [Google Scholar] [CrossRef] [PubMed]
- Tao, S.; Chen, H.; Li, N.; Fang, Y.; Xu, Y.; Liang, W. Association of CRISPR-Cas System with the Antibiotic Resistance and Virulence Genes in Nosocomial Isolates of Enterococcus. Infect. Drug Resist. 2022, 15, 6939–6949. [Google Scholar] [CrossRef]
- Pourcel, C.; Touchon, M.; Villeriot, N.; Vernadet, J.-P.; Couvin, D.; Toffano-Nioche, C.; Vergnaud, G. CRISPRCasdb a successor of CRISPRdb containing CRISPR arrays and cas genes from complete genome sequences, and tools to download and query lists of repeats and spacers. Nucleic Acids Res. 2019, 48, D535–D544. [Google Scholar] [CrossRef]
- Alcock, B.P.; Huynh, W.; Chalil, R.; Smith, K.W.; Raphenya, A.R.; Wlodarski, M.A.; Edalatmand, A.; Petkau, A.; Syed, S.A.; Tsang, K.K.; et al. CARD 2023: Expanded curation, support for machine learning, and resistome prediction at the Comprehensive Antibiotic Resistance Database. Nucleic Acids Res. 2023, 51, D690–D699. [Google Scholar] [CrossRef]
- Lyons, C.; Raustad, N.; Bustos, M.A.; Shiaris, M. Incidence of Type II CRISPR1-Cas Systems in Enterococcus Is Species-Dependent. PLoS ONE 2015, 10, e0143544. [Google Scholar] [CrossRef]
- Tsegahun, A. Biofilm Formation by Enterococcus faecalis and Enterococcus faecium: Review. Int. J. Res. Stud. Biosci. 2019, 7, 5–10. [Google Scholar] [CrossRef]
- Tao, S.; Zhou, D.; Chen, H.; Li, N.; Zheng, L.; Fang, Y.; Xu, Y.; Jiang, Q.; Liang, W. Analysis of genetic structure and function of clustered regularly interspaced short palindromic repeats loci in 110 Enterococcus strains. Front. Microbiol. 2023, 14, 1177841. [Google Scholar] [CrossRef] [PubMed]
- Gholizadeh, P.; Aghazadeh, M.; Ghotaslou, R.; Rezaee, M.A.; Pirzadeh, T.; Cui, L.; Watanabe, S.; Feizi, H.; Kadkhoda, H.; Kafil, H.S. Role of CRISPR-Cas system on antibiotic resistance patterns of Enterococcus faecalis. Ann. Clin. Microbiol. Antimicrob. 2021, 20, 49. [Google Scholar] [CrossRef] [PubMed]
- Pursey, E.; Dimitriu, T.; Paganelli, F.L.; Westra, E.R.; van Houte, S. CRISPR-Cas is associated with fewer antibiotic resistance genes in bacterial pathogens. Philos. Trans. R. Soc. B Biol. Sci. 2022, 377, 20200464. [Google Scholar] [CrossRef]
- Price, V.J.; McBride, S.W.; Hullahalli, K.; Chatterjee, A.; Duerkop, B.A.; Palmer, K.L. Enterococcus faecalis CRISPR-Cas Is a Robust Barrier to Conjugative Antibiotic Resistance Dissemination in the Murine Intestine. mSphere 2019, 4, e00464-19. [Google Scholar] [CrossRef]
- dos Santos, B.A.; da Silva de Oliveira, J.; Parmanhani-da-Silva, B.M.; Ribeiro, R.L.; Teixeira, L.M.; Neves, F.P.G. CRISPR elements and their association with antimicrobial resistance and virulence genes among vancomycin-resistant and vancomycin-susceptible enterococci recovered from human and food sources. Infect. Genet. Evol. 2020, 80, 104183. [Google Scholar] [CrossRef]
- Pinilla-Redondo, R.; Russel, J.; Mayo-Muñoz, D.; Shah, S.A.; Garrett, R.A.; Nesme, J.; Madsen, J.S.; Fineran, P.C.; Sørensen, S.J. CRISPR-Cas systems are widespread accessory elements across bacterial and archaeal plasmids. Nucleic Acids Res. 2022, 50, 4315–4328. [Google Scholar] [CrossRef]
- Pinilla-Redondo, R.; Mayo-Muñoz, D.; Russel, J.; Garrett, R.A.; Randau, L.; Sørensen, S.J.; Shah, S.A. Type IV CRISPR–Cas systems are highly diverse and involved in competition between plasmids. Nucleic Acids Res. 2020, 48, 2000–2012. [Google Scholar] [CrossRef] [PubMed]
- Murugesan, A.C.; Varughese, H.S. Analysis of CRISPR–Cas system and antimicrobial resistance in Staphylococcus coagulans isolates. Lett. Appl. Microbiol. 2022, 75, 126–134. [Google Scholar] [CrossRef]
- Garneau, J.E.; Dupuis, M.-È.; Villion, M.; Romero, D.A.; Barrangou, R.; Boyaval, P.; Fremaux, C.; Horvath, P.; Magadán, A.H.; Moineau, S. The CRISPR/Cas bacterial immune system cleaves bacteriophage and plasmid DNA. Nature 2010, 468, 67–71. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, M.; McBride, S.W.; Hullahalli, K.; Palmer, K.L.; Duerkop, B.A. Conjugative delivery of CRISPR-Cas9 for the selective depletion of antibiotic-resistant enterococci. Antimicrob. Agents Chemother. 2019, 63, e01454-19. [Google Scholar] [CrossRef] [PubMed]
- Varahan, S.; Hancock, L.E. To Defend or Not To Defend: That’s the Question. mSphere 2016, 1, 16–18. [Google Scholar] [CrossRef] [PubMed]
- Huo, W.; Price, V.J.; Sharifi, A.; Zhang, M.Q.; Palmer, K.L. Enterococcus faecalis Strains with Compromised CRISPR-Cas Defense Emerge under Antibiotic Selection for a CRISPR-Targeted Plasmid. Appl. Environ. Microbiol. 2023, 89, e0012423. [Google Scholar] [CrossRef] [PubMed]

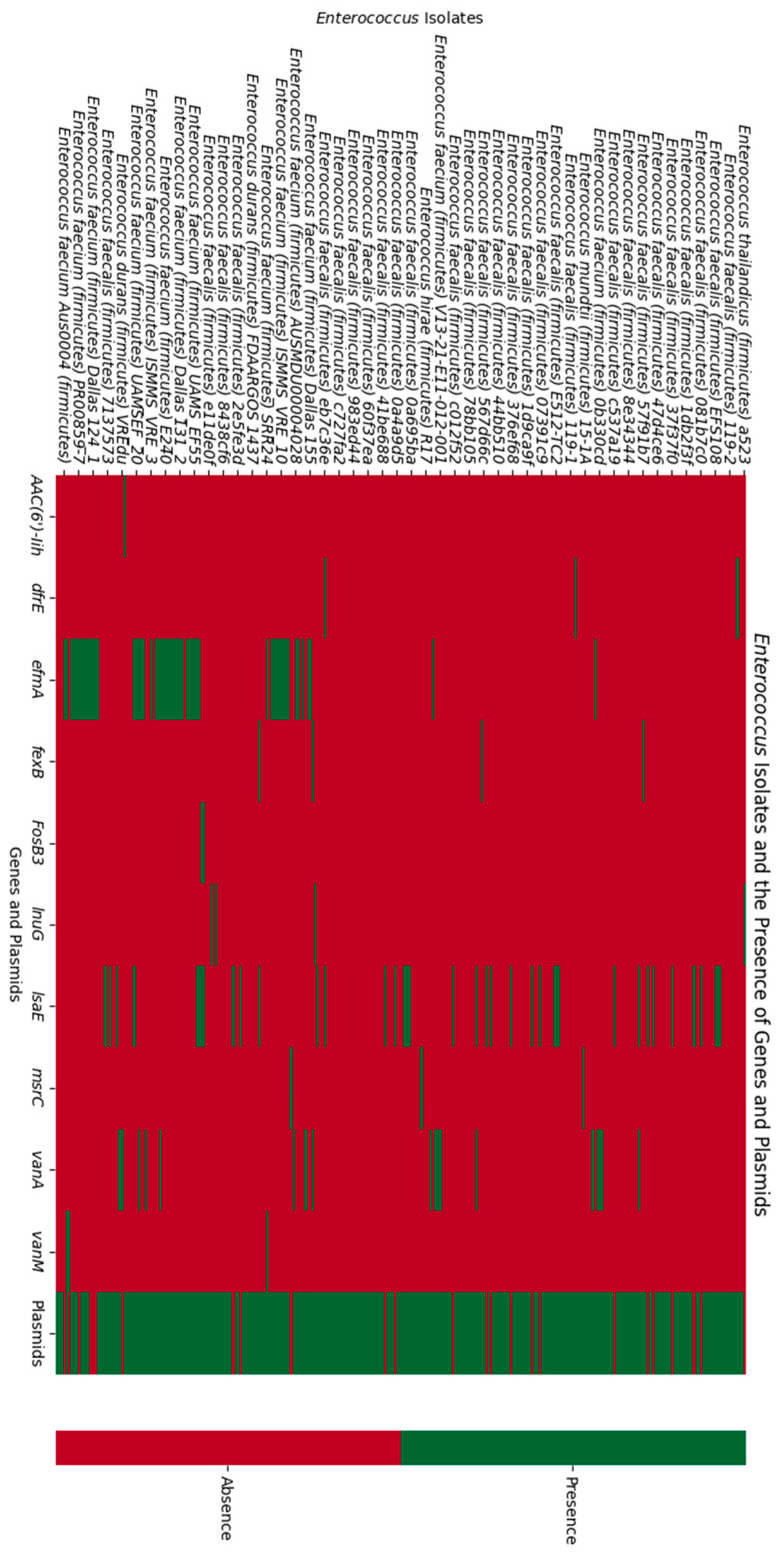

| Gene | CRISPR-Positive (n = 187 Sequences) | CRISPR-Negative (n = 529 Sequences) |
|---|---|---|
| AAC(6′)-Iih | 0 (0%) | 1 (0.19%) |
| dfrE | 1 (0.53%) | 1 (0.19%) |
| efmA | 1 (0.53%) | 52 (9.83%) |
| fexB | 1 (0.53%) | 2 (0.38%) |
| FosB3 | 0 (0%) | 2 (0.38%) |
| lnuG | 1 (0.53%) | 3 (0.57%) |
| lsaE | 12 (6.42%) | 17 (3.21%) |
| msrC | 1 (0.53%) | 1 (0.19%) |
| vanA | 5 (2.67%) | 9 (1.70%) |
| vanM | 0 (0%) | 3 (0.57%) |
| Gene | p (Fisher’s Exact Test) |
|---|---|
| efmA | p < 0.05 |
| fexB | 1 |
| FosB3 | 1 |
| lnuG | 1 |
| lsaE | 0.0812 |
| vanA | 0.3774 |
| vanM | 0.5713 |
| E. faecium | CRISPR-Positive | CRISPR-Negative | p (Fisher’s Exact Test) |
|---|---|---|---|
| efmA | 4.1% | 23.9% | 0.0034 |
| vanA | 20.8% | 4.1% | 0.007 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Costache, C.; Colosi, I.; Toc, D.-A.; Daian, K.; Damacus, D.; Botan, A.; Toc, A.; Pana, A.G.; Panaitescu, P.; Neculicioiu, V.; et al. CRISPR-Cas System, Antimicrobial Resistance, and Enterococcus Genus—A Complicated Relationship. Biomedicines 2024, 12, 1625. https://doi.org/10.3390/biomedicines12071625
Costache C, Colosi I, Toc D-A, Daian K, Damacus D, Botan A, Toc A, Pana AG, Panaitescu P, Neculicioiu V, et al. CRISPR-Cas System, Antimicrobial Resistance, and Enterococcus Genus—A Complicated Relationship. Biomedicines. 2024; 12(7):1625. https://doi.org/10.3390/biomedicines12071625
Chicago/Turabian StyleCostache, Carmen, Ioana Colosi, Dan-Alexandru Toc, Karla Daian, David Damacus, Alexandru Botan, Adelina Toc, Adrian Gabriel Pana, Paul Panaitescu, Vlad Neculicioiu, and et al. 2024. "CRISPR-Cas System, Antimicrobial Resistance, and Enterococcus Genus—A Complicated Relationship" Biomedicines 12, no. 7: 1625. https://doi.org/10.3390/biomedicines12071625
APA StyleCostache, C., Colosi, I., Toc, D.-A., Daian, K., Damacus, D., Botan, A., Toc, A., Pana, A. G., Panaitescu, P., Neculicioiu, V., Schiopu, P., Iordache, D., & Butiuc-Keul, A. (2024). CRISPR-Cas System, Antimicrobial Resistance, and Enterococcus Genus—A Complicated Relationship. Biomedicines, 12(7), 1625. https://doi.org/10.3390/biomedicines12071625








