Addressing Post-Acute COVID-19 Syndrome in Cancer Patients, from Visceral Obesity and Myosteatosis to Systemic Inflammation: Implications in Cardio-Onco-Metabolism
Abstract
:1. Introduction
2. Methods
3. Post-Acute COVID-19 Syndrome: A Clinical Scenario
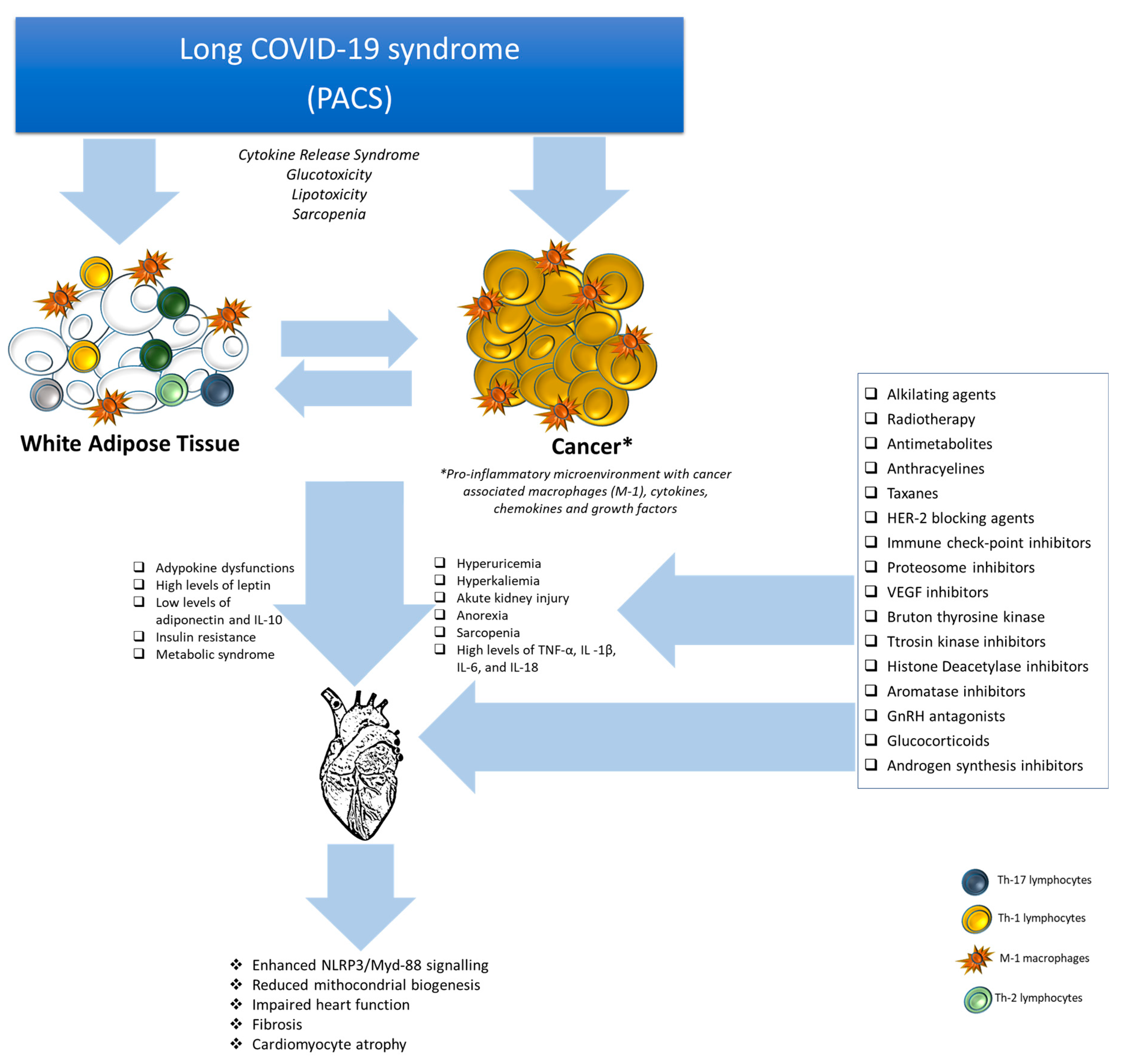
4. Post-Acute COVID-19 Syndrome and Cardiovascular Complications in Cancer Patients
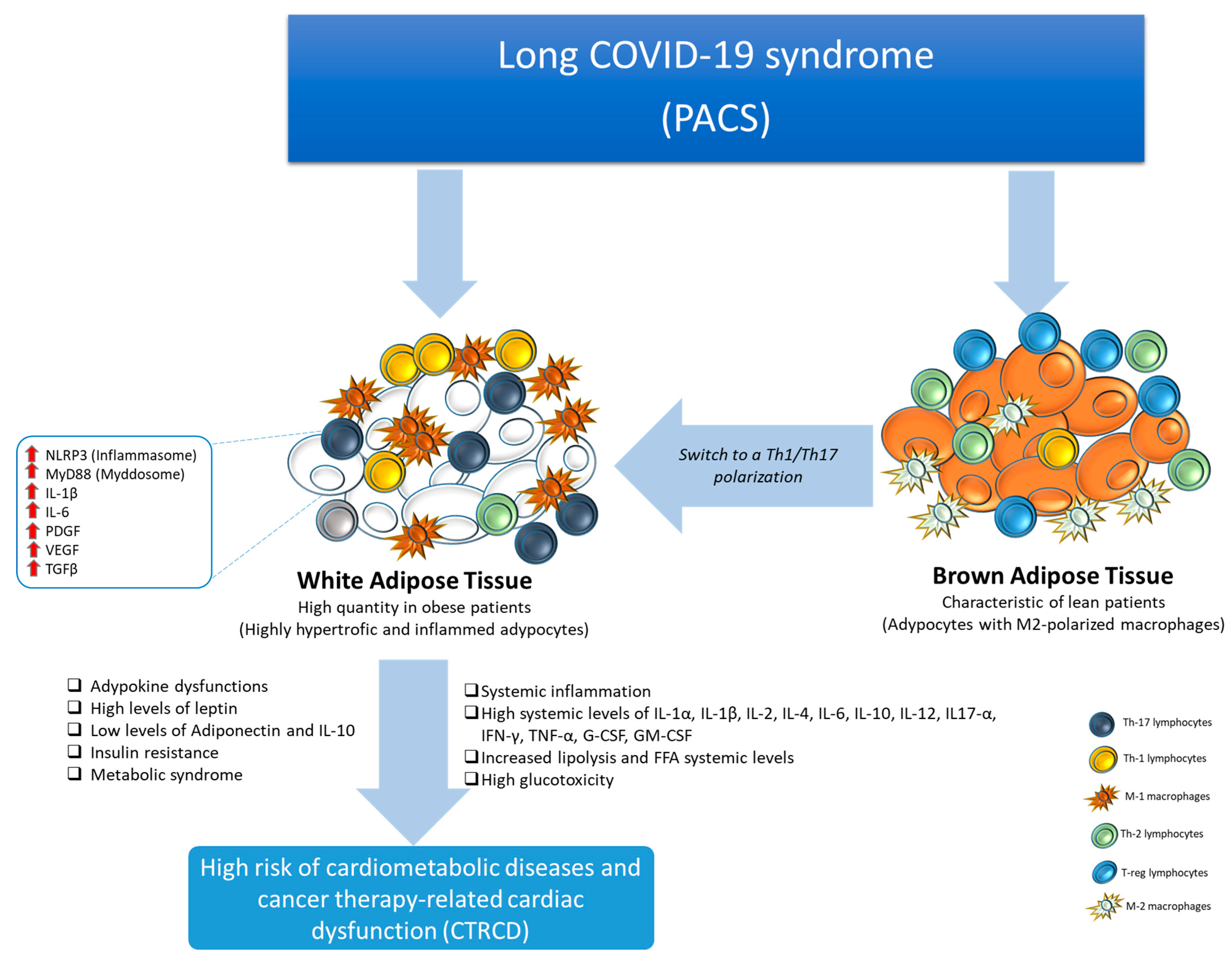
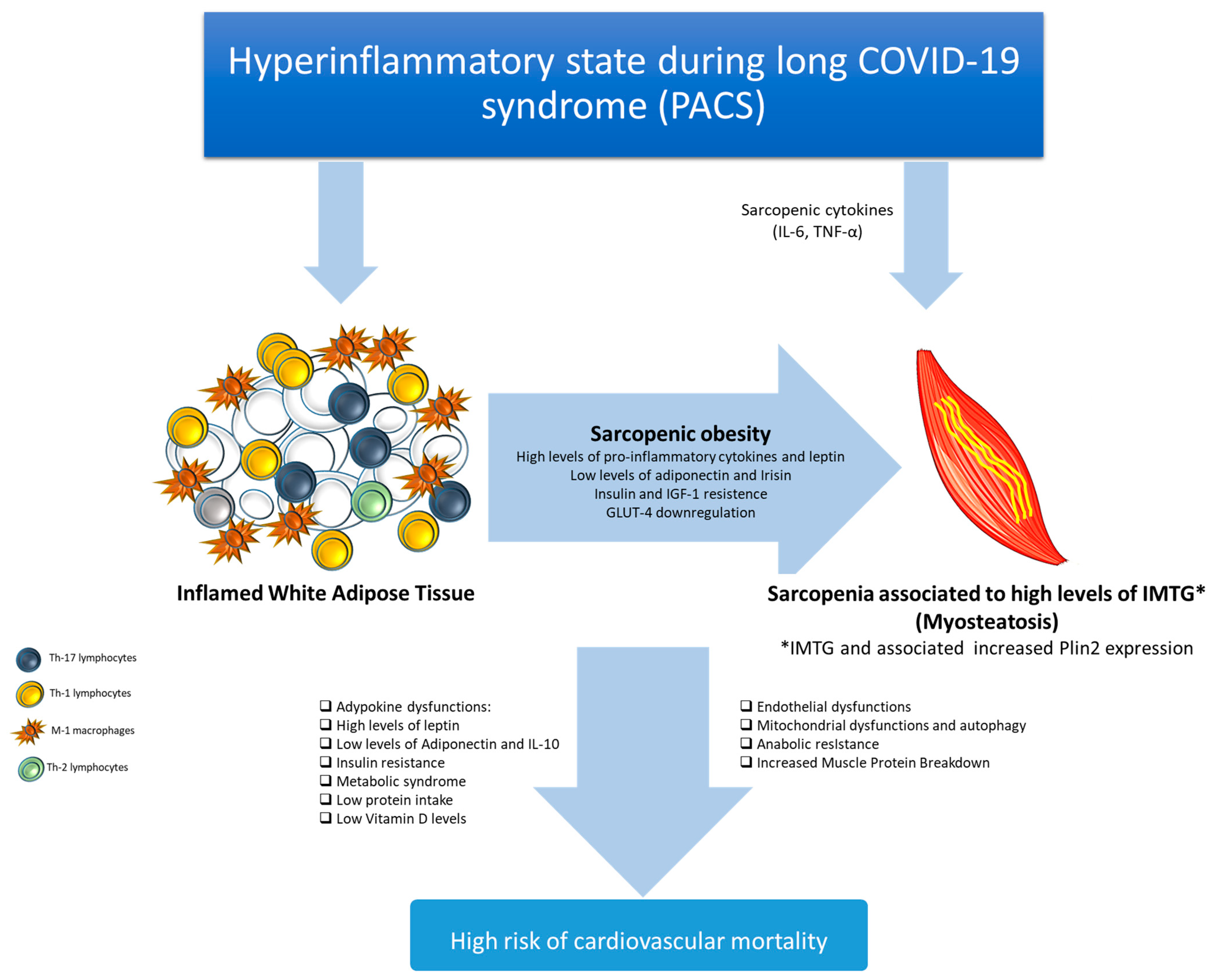
5. Post-Acute COVID-19 and Visceral Obesity
6. Post-Acute COVID-19 and Inflammation
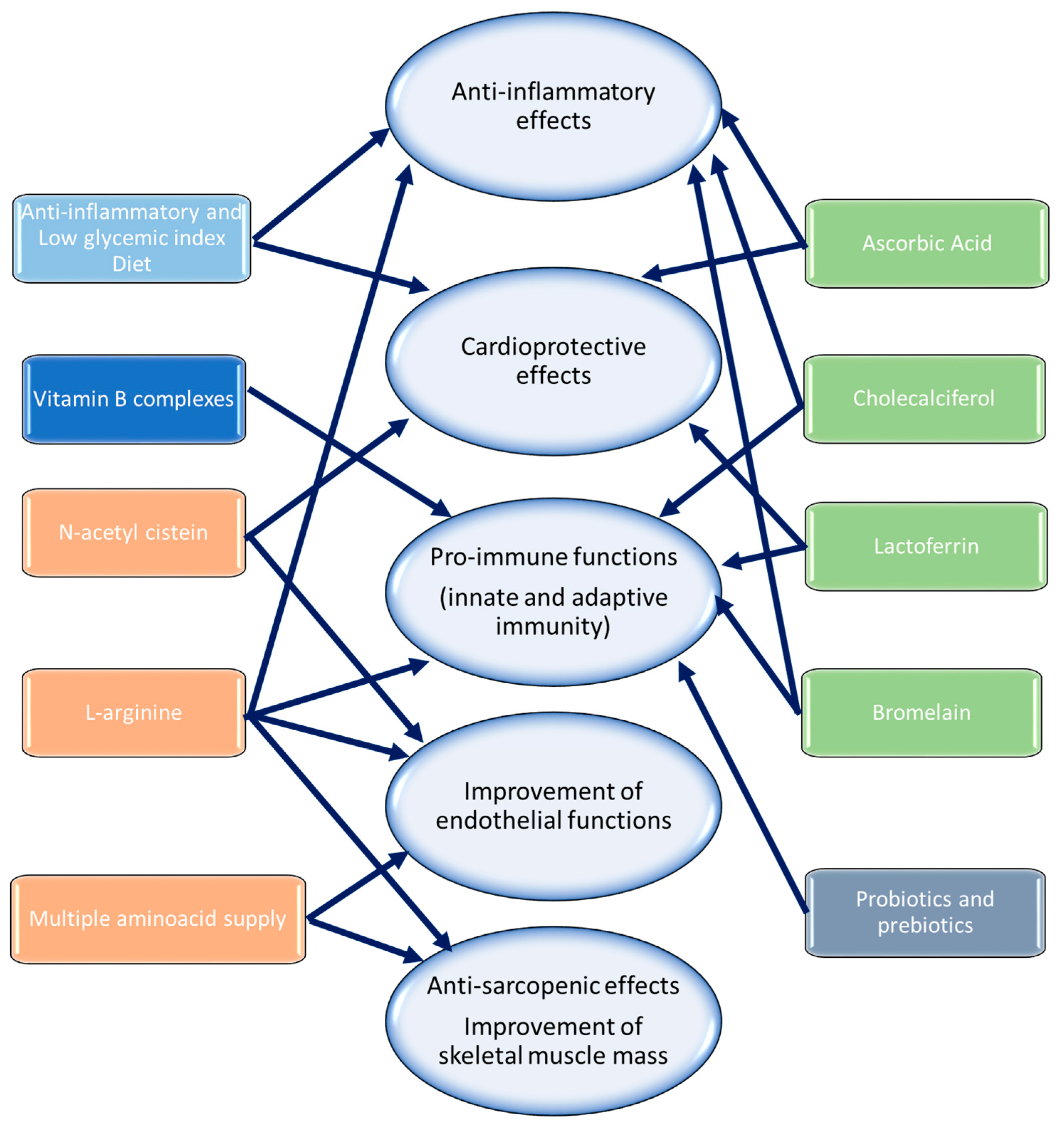
7. Suggestions to Reduce Cardiovascular Complications in Cancer Patients with Post-Acute COVID-19 Syndrome
| Therapeutic Option | Molecular Pathway | Clinical Outcomes | Ref. |
|---|---|---|---|
| Anti-inflammatory diet | ↓ COX-2, NLRP3, MyD88, pro-inflammatory cytokines | Anti-inflammatory systemic effects Cardioprotective effects | [171,172] |
| L-Arginine | ↑ T-lymphocyte survival, ↑ nitric oxide, cGMP, PKG ↓ IL-17, IL-1β | Pro-immune functions (innate and adaptive immunity) Anti-inflammatory systemic effects Improvement of endothelial functions | [173,174,175] |
| N-acetil cystein (NAC) | ↑ Cys, GSH, IL-10 ↓ HOCl, OH-, H2O2 ↓ NLRP3, MyD88 | Anti-inflammatory systemic effects Cardioprotective effects | [175,176] |
| Ascorbic acid, NAC, zinc, and iron | ↑ T-lymphocyte survival ↑ nitric oxide, cGMP, PKG ↓ IL-17, IL-1β, NLRP3, MyD88 | Pro-immune functions (innate and adaptive immunity) Anti-inflammatory systemic effects Improvement of endothelial functions | [177,178,179] |
| Cholecalciferol | ↓ NLRP3, MyD88, IL-6, and IL-17 ↑ CD8+ T-lymphocyte survival; Natural Killer cells ↑ IL-10 | Anti-inflammatory effects Pro-immune functions (innate and adaptive immunity) | [180,181,182,183,184] |
| Liposomal ascorbic acid | ↓ NLRP3, MyD88, IL-6, IL-17, IL-1β ↑ CD8+ T-lymphocyte survival; Natural Killer cells ↑ IL-10 | Anti-inflammatory effects Pro-immune functions (innate and adaptive immunity) | [185] |
| Multiple amino acid supplements (L-Leucine, L-Valine, L-Isoleucine, L-Lysine hydrochloride, L-Phenylalanine, L-Threonine, L-Methionine, L-Tryptophan) | ↓ ROS, IL-6, TNF-α, IL-1 ↑ Mitochondrial biogenesis, motor units, number of fibers ↑ Satellite cells function ↑ NO, PCG1-α | Anti-sarcopenic effects Improvement of skeletal muscle mass Improvement of endothelial functions | [187,188] |
| Lactoferrin | ↓ IL-1, IL-6, and IL-17 ↑ CD8+ T-lymphocyte survival; Natural Killer cells ↑ IL-10 ↓ ROS, MDA, 4-HNA | Antiviral effects Anti-inflammatory effects Pro-immune functions (innate and adaptive immunity) | [189,190,191,192,193] |
| Bromelain | ↓ IL-1, IL-6, IL-17, PGE2, COX-2 ↑ CD8+ T-lymphocyte survival; Natural Killer cells ↑ IL-10 ↓ ACE-2, TMPRSS2 | Antiviral effects Anti-inflammatory effects Pro-immune functions (innate and adaptive immunity) | [194,195,196,197,198,199,200,201,202,203,204,205,206] |
| Probiotics (Streptococcus thermophilus, Bifidobacterium breve, Bifidobacterium longum, Bifidobacterium infantis, Lactobacillus acidophilus, Lactobacillus plantarum, Lactobacillus paracasei, Lactobacillus delbrueckii, and lactobacillus reuteri) | ↑ IgA responses ↓ IL-17, IL-6, IL-1, TNF-α ↑ IL-15, IL-12, IL-21 ↑ CD8+ T-lymphocyte survival; Natural Killer cells | Pro-immune functions (innate and adaptive immunity) Enhancement of antibody production | [208,209,210,211] |
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Scholkmann, F.; May, C.A. COVID-19, post-acute COVID-19 syndrome (PACS, “long COVID”) and post-COVID-19 vaccination syndrome (PCVS, “post-COVIDvac-syndrome”): Similarities and differences. Pathol. Res. Pract. 2023, 246, 154497. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Albtoosh, A.S.; Toubasi, A.A.; Al Oweidat, K.; Hasuneh, M.M.; Alshurafa, A.H.; Alfaqheri, D.L.; Farah, R.I. New symptoms and prevalence of postacute COVID-19 syndrome among nonhospitalized COVID-19 survivors. Sci. Rep. 2022, 12, 16921. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Elseidy, S.A.; Awad, A.K.; Vorla, M.; Fatima, A.; Elbadawy, M.A.; Mandal, D.; Mohamad, T. Cardiovascular complications in the Post-Acute COVID-19 syndrome (PACS). Int. J. Cardiol. Heart Vasc. 2022, 40, 101012. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dixit, N.M.; Churchill, A.; Nsair, A.; Hsu, J.J. Post-Acute COVID-19 Syndrome and the cardiovascular system: What is known? Am. Heart J. Plus 2021, 5, 100025. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lim, S.H.; Lim, Y.C.; Zaki, R.A.; Johari, B.M.; Chang, C.Y.; Omar, S.F.S.; Azzeri, A.; Dahlui, M.; Kamarulzaman, A. Prevalence and predictors of post-acute COVID syndrome among infected healthcare workers at University Malaya Medical Centre. PLoS ONE 2024, 19, e0298376. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chiappelli, F.; Fotovat, L. Post acute COVID-19 syndrome (PACS)—Long COVID. Bioinformation 2022, 18, 908–911. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cavalcanti, I.D.L.; Soares, J.C.S. Impact of COVID-19 on cancer patients: A review. Asia Pac. J. Clin. Oncol. 2021, 17, 186–192. [Google Scholar] [CrossRef] [PubMed]
- Jee, J.; Foote, M.B.; Lumish, M.; Stonestrom, A.J.; Wills, B.; Narendra, V.; Avutu, V.; Murciano-Goroff, Y.R.; Chan, J.E.; Derkach, A.; et al. Chemotherapy and COVID-19 Outcomes in Patients With Cancer. J. Clin. Oncol. 2020, 38, 3538–3546. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Salvatore, M.; Hu, M.M.; Beesley, L.J.; Mondul, A.M.; Pearce, C.L.; Friese, C.R.; Fritsche, L.G.; Mukherjee, B. COVID-19 Outcomes by Cancer Status, Site, Treatment, and Vaccination. Cancer Epidemiol. Biomark. Prev. 2023, 32, 748–759. [Google Scholar] [CrossRef] [PubMed]
- Cottenet, J.; Tapia, S.; Arveux, P.; Bernard, A.; Dabakuyo-Yonli, T.S.; Quantin, C. Effect of Obesity among Hospitalized Cancer Patients with or without COVID-19 on a National Level. Cancers 2022, 14, 5660. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cho, H.; Park, Y.; Myung, S.K. Obesity and mortality in patients with COVID-19: A meta-analysis of prospective studies. Asia Pac. J. Clin. Nutr. 2024, 33, 56–65. [Google Scholar] [CrossRef] [PubMed]
- Corrêa, L.H.; Heyn, G.S.; Magalhaes, K.G. The Impact of the Adipose Organ Plasticity on Inflammation and Cancer Progression. Cells 2019, 8, 662. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Trayhurn, P. Hypoxia and adipose tissue function and dysfunction in obesity. Physiol. Rev. 2013, 93, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Sánchez, N. There and Back Again: Leptin Actions in White Adipose Tissue. Int. J. Mol. Sci. 2020, 21, 6039. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bełtowski, J. Adiponectin and resistin—New hormones of white adipose tissue. Med. Sci. Monit. 2003, 9, RA55–RA61. [Google Scholar] [PubMed]
- Beppu, L.Y.; Mooli, R.G.R.; Qu, X.; Marrero, G.J.; Finley, C.A.; Fooks, A.N.; Mullen, Z.P.; Frias, A.B., Jr.; Sipula, I.; Xie, B.; et al. Tregs facilitate obesity and insulin resistance via a Blimp-1/IL-10 axis. JCI Insight. 2021, 6, e140644. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Castellano-Castillo, D.; Ramos-Molina, B.; Cardona, F.; Queipo-Ortuño, M.I. Epigenetic regulation of white adipose tissue in the onset of obesity and metabolic diseases. Obes. Rev. 2020, 21, e13054. [Google Scholar] [CrossRef] [PubMed]
- Demir, L.; Oflazoğlu, U. The relationship between sarcopenia and serum irisin and TNF-α levels in newly diagnosed cancer patients. Support. Care Cancer 2023, 31, 586. [Google Scholar] [CrossRef] [PubMed]
- Lan, T.; Chen, L.; Wei, X. Inflammatory Cytokines in Cancer: Comprehensive Understanding and Clinical Progress in Gene Therapy. Cells 2021, 10, 100. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tsimberidou, A.M.; Keating, M.J. Hyperuricemic syndromes in cancer patients. Contrib. Nephrol. 2005, 147, 47–60. [Google Scholar] [CrossRef] [PubMed]
- Lacavalerie, M.R.; Pierre-Francois, S.; Agossou, M.; Inamo, J.; Cabie, A.; Barnay, J.L.; Neviere, R. Obese patients with long COVID-19 display abnormal hyperventilatory response and impaired gas exchange at peak exercise. Future Cardiol. 2022, 18, 577–584. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Quagliariello, V.; D’Aiuto, G.; Iaffaioli, R.V.; Berretta, M.; Buccolo, S.; Iovine, M.; Paccone, A.; Cerrone, F.; Bonanno, S.; Nunnari, G.; et al. Reasons why COVID-19 survivors should follow dietary World Cancer Research Fund/American Institute for Cancer Research (WCRF/AICR) recommendations: From hyper-inflammation to cardiac dysfunctions. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 3898–3907. [Google Scholar] [CrossRef] [PubMed]
- Zheng, C.; Chen, X.K.; Sit, C.H.; Liang, X.; Li, M.H.; Ma, A.C.; Wong, S.H. Effect of Physical Exercise-Based Rehabilitation on Long COVID: A Systematic Review and Meta-analysis. Med. Sci. Sports Exerc. 2024, 56, 143–154. [Google Scholar] [CrossRef] [PubMed]
- Davis, H.E.; McCorkell, L.; Vogel, J.M.; Topol, E.J. Long COVID: Major findings, mechanisms and recommendations. Nat. Rev. Microbiol. 2023, 21, 133–146, Erratum in Nat. Rev. Microbiol. 2023, 21, 408. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sandler, C.X.; Wyller, V.B.B.; Moss-Morris, R.; Buchwald, D.; Crawley, E.; Hautvast, J.; Katz, B.Z.; Knoop, H.; Little, P.; Taylor, R.; et al. Long COVID and Post-infective Fatigue Syndrome: A Review. Open Forum Infect. Dis. 2021, 8, ofab440. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Titze-de-Almeida, R.; Araújo Lacerda, P.H.; de Oliveira, E.P.; de Oliveira, M.E.F.; Vianna, Y.S.S.; Costa, A.M.; Pereira Dos Santos, E.; Guérard, L.M.C.; Ferreira, M.A.M.; Rodrigues Dos Santos, I.C.; et al. Sleep and memory complaints in long COVID: An insight into clustered psychological phenotypes. PeerJ 2024, 12, e16669. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Martone, A.M.; Tosato, M.; Ciciarello, F.; Galluzzo, V.; Zazzara, M.B.; Pais, C.; Savera, G.; Calvani, R.; Marzetti, E.; Robles, M.C.; et al. Sarcopenia as potential biological substrate of long COVID-19 syndrome: Prevalence, clinical features, and risk factors. J. Cachexia Sarcopenia Muscle 2022, 13, 1974–1982. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Piotrowicz, K.; Gąsowski, J.; Michel, J.P.; Veronese, N. Post-COVID-19 acute sarcopenia: Physiopathology and management. Aging Clin. Exp. Res. 2021, 33, 2887–2898. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zong, M.; Zhao, A.; Han, W.; Chen, Y.; Weng, T.; Li, S.; Tang, L.; Wu, J. Sarcopenia, sarcopenic obesity and the clinical outcome of the older inpatients with COVID-19 infection: A prospective observational study. BMC Geriatr. 2024, 24, 578. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Burges Watson, D.L.; Campbell, M.; Hopkins, C.; Smith, B.; Kelly, C.; Deary, V. Altered smell and taste: Anosmia, parosmia and the impact of long COVID-19. PLoS ONE 2021, 16, e0256998. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, J.; Liu, R.; Ma, H.; Zhang, W. The Pathogenesis of COVID-19-Related Taste Disorder and Treatments. J. Dent. Res. 2023, 102, 1191–1198. [Google Scholar] [CrossRef] [PubMed]
- Srivastava, A.; Nalroad Sundararaj, S.; Bhatia, J.; Singh Arya, D. Understanding long COVID myocarditis: A comprehensive review. Cytokine 2024, 178, 156584. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, A.; Tariq, R.; Jena, A.; Vesely, E.K.; Singh, S.; Khanna, S.; Sharma, V. Gastrointestinal manifestations of long COVID: A systematic review and meta-analysis. Therap. Adv. Gastroenterol. 2022, 15, 17562848221118403. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Mazza, M.G.; Palladini, M.; Poletti, S.; Benedetti, F. Post-COVID-19 Depressive Symptoms: Epidemiology, Pathophysiology, and Pharmacological Treatment. CNS Drugs 2022, 36, 681–702. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Álvarez-Santacruz, C.; Tyrkalska, S.D.; Candel, S. The Microbiota in Long COVID. Int. J. Mol. Sci. 2024, 25, 1330. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Riad, A.; Kassem, I.; Badrah, M.; Klugar, M. The manifestation of oral mucositis in COVID-19 patients: A case-series. Dermatol. Ther. 2020, 33, e14479. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sapkota, H.R.; Nune, A. Long COVID from rheumatology perspective—A narrative review. Clin. Rheumatol. 2022, 41, 337–348. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- de Jesus, M.; Chanda, A.; Grabauskas, T.; Kumar, M.; Kim, A.S. Cardiovascular disease and lung cancer. Front. Oncol. 2024, 14, 1258991. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lee, L.Y.; Cazier, J.B.; Angelis, V.; Arnold, R.; Bisht, V.; Campton, N.A.; Chackathayil, J.; Cheng, V.W.; Curley, H.M.; Fittall, M.W.; et al. COVID-19 mortality in patients with cancer on chemotherapy or other anticancer treatments: A prospective cohort study. Lancet 2020, 395, 1919–1926, Erratum in Lancet 2020, 396, 534. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wu, M.; Liu, S.; Wang, C.; Wu, Y.; Liu, J. Risk factors for mortality among lung cancer patients with COVID-19 infection: A systematic review and meta-analysis. PLoS ONE 2023, 18, e0291178. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Maruyama, S.; Wada, D.; Kanayama, S.; Shimazu, H.; Miyano, Y.; Inoue, A.; Kashihara, M.; Okuda, K.; Saito, F.; Nakamori, Y.; et al. The evaluation of risk factors for prolonged viral shedding during anti-SARS-CoV-2 monoclonal antibodies and long-term administration of antivirals in COVID-19 patients with B-cell lymphoma treated by anti-CD20 antibody. BMC Infect. Dis. 2024, 24, 715. [Google Scholar] [CrossRef] [PubMed]
- Potter, A.L.; Vaddaraju, V.; Venkateswaran, S.; Mansur, A.; Bajaj, S.S.; Kiang, M.V.; Jena, A.B.; Yang, C.J. Deaths Due to COVID-19 in Patients With Cancer During Different Waves of the Pandemic in the, U.S. JAMA Oncol. 2023, 9, 1417–1422. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Khoury, E.; Nevitt, S.; Madsen, W.R.; Turtle, L.; Davies, G.; Palmieri, C. Differences in Outcomes and Factors Associated With Mortality Among Patients With SARS-CoV-2 Infection and Cancer Compared With Those Without Cancer: A Systematic Review and Meta-analysis. JAMA Netw. Open 2022, 5, e2210880. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Salunke, A.A.; Nandy, K.; Pathak, S.K.; Shah, J.; Kamani, M.; Kottakota, V.; Thivari, P.; Pandey, A.; Patel, K.; Rathod, P.; et al. Impact of COVID-19 in cancer patients on severity of disease and fatal outcomes: A systematic review and meta-analysis. Diabetes Metab. Syndr. 2020, 14, 1431–1437. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tleyjeh, I.M.; Kashour, T.; Riaz, M.; Amer, S.A.; AlSwaidan, N.; Almutairi, L.; Halwani, R.; Assiri, A. Persistent COVID-19 symptoms at least one month after diagnosis: A national survey. J. Infect. Public Health 2022, 15, 578–585. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Caranci, N.; Di Girolamo, C.; Bartolini, L.; Fortuna, D.; Berti, E.; Sforza, S.; Giorgi Rossi, P.; Moro, M.L. General and COVID-19-Related Mortality by Pre-Existing Chronic Conditions and Care Setting during 2020 in Emilia-Romagna Region, Italy. Int. J. Environ. Res. Public Health 2021, 18, 13224. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Duong-Quy, S.; Huynh-Truong-Anh, D.; Nguyen-Thi-Kim, T.; Nguyen-Quang, T.; Tran-Ngoc-Anh, T.; Nguyen-Van-Hoai, N.; Do-Thi-Thu, M.; Nguyen-Chi, T.; Nguyen-Van, T.; Tang-Thi-Thao, T.; et al. Predictive Factors of Mortality in Patients with Severe COVID-19 Treated in the Intensive Care Unit: A Single-Center Study in Vietnam. Pulm Ther. 2023, 9, 377–394. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Meza-Torres, B.; Delanerolle, G.; Okusi, C.; Mayor, N.; Anand, S.; Macartney, J.; Gatenby, P.; Glampson, B.; Chapman, M.; Curcin, V.; et al. Differences in Clinical Presentation With Long COVID After Community and Hospital Infection and Associations With All-Cause Mortality: English Sentinel Network Database Study. JMIR Public Health Surveill. 2022, 8, e37668. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lipski, D.; Radziemski, A.; Wasiliew, S.; Wyrwa, M.; Szczepaniak-Chicheł, L.; Stryczyński, Ł.; Olasińska-Wiśniewska, A.; Urbanowicz, T.; Perek, B.; Tykarski, A.; et al. Assessment of COVID-19 risk factors of early and long-term mortality with prediction models of clinical and laboratory variables. BMC Infect. Dis. 2024, 24, 685. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Righi, E.; Mirandola, M.; Mazzaferri, F.; Razzaboni, E.; Zaffagnini, A.; Erbogasto, A.; Vecchia, I.D.; Auerbach, N.; Ivaldi, F.; Mongardi, M.; et al. Long-Term Patient-Centred Follow-up in a Prospective Cohort of Patients with COVID-19. Infect. Dis. Ther. 2021, 10, 1579–1590. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, J.; Chen, N.; Zhao, D.; Zhang, J.; Hu, Z.; Tao, Z. Clinical Characteristics of COVID-19 Patients Infected by the Omicron Variant of SARS-CoV-2. Front. Med. 2022, 9, 912367. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Moss, P. The T cell immune response against SARS-CoV-2. Nat. Immunol. 2022, 23, 186–193. [Google Scholar] [CrossRef] [PubMed]
- Shakiba, M.H.; Gemünd, I.; Beyer, M.; Bonaguro, L. Lung T cell response in COVID-19. Front. Immunol. 2023, 14, 1108716. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Queiroz, M.A.F.; das Neves, P.F.M.; Lima, S.S.; Lopes, J.d.C.; Torres, M.K.d.S.; Vallinoto, I.M.V.C.; de Brito, M.T.F.M.; da Silva, A.L.S.; Leite, M.d.M.; da Costa, F.P.; et al. Cytokine Profiles Associated With Acute COVID-19 and Long COVID-19 Syndrome. Front. Cell. Infect. Microbiol. 2022, 12, 922422. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gomes, S.M.R.; Brito, A.C.S.; Manfro, W.F.P.; Ribeiro-Alves, M.; Ribeiro, R.S.A.; da Cal, M.S.; Lisboa, V.D.C.; Abreu, D.P.B.; Castilho, L.D.R.; Porto, L.C.M.S.; et al. High levels of pro-inflammatory SARS-CoV-2-specific biomarkers revealed by in vitro whole blood cytokine release assay (CRA) in recovered and long-COVID-19 patients. PLoS ONE 2023, 18, e0283983. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- López-Hernández, Y.; Monárrez-Espino, J.; López, D.A.G.; Zheng, J.; Borrego, J.C.; Torres-Calzada, C.; Elizalde-Díaz, J.P.; Mandal, R.; Berjanskii, M.; Martínez-Martínez, E.; et al. The plasma metabolome of long COVID patients two years after infection. Sci. Rep. 2023, 13, 12420. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ageev, A.A.; Kozhevnikova, M.V.; Emelyanov, A.V.; Krivova, A.V.; Shumskaya, Y.F.; Musaeva, L.M.; Popova, L.V.; Naymann, Y.I.; Abdullaeva, G.B.; Privalova, E.V.; et al. The Effect of COVID-19 on Long-Term Cardiac Function in Patients With Chronic Heart Failure. Kardiologiia 2022, 62, 23–29, (In Russian, English). [Google Scholar] [CrossRef] [PubMed]
- Alfaro, C.; Sanmamed, M.F.; Rodríguez-Ruiz, M.E.; Teijeira, Á.; Oñate, C.; González, Á.; Ponz, M.; Schalper, K.A.; Pérez-Gracia, J.L.; Melero, I. Interleukin-8 in cancer pathogenesis, treatment and follow-up. Cancer Treat. Rev. 2017, 60, 24–31. [Google Scholar] [CrossRef] [PubMed]
- Hu, B.; Huang, S.; Yin, L. The cytokine storm and COVID-19. J. Med. Virol. 2021, 93, 250–256. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Paruchuri, S.S.H.; Farwa, U.E.; Jabeen, S.; Pamecha, S.; Shan, Z.; Parekh, R.; Lakkimsetti, M.; Alamin, E.; Sharma, V.; Haider, S.; et al. Myocarditis and Myocardial Injury in Long COVID Syndrome: A Comprehensive Review of the Literature. Cureus 2023, 15, e42444. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- McMaster, M.W.; Dey, S.; Fishkin, T.; Wang, A.; Frishman, W.H.; Aronow, W.S. The Impact of Long COVID-19 on the Cardiovascular System. Cardiol. Rev 2024. ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Lasagna, A.; Albi, G.; Figini, S.; Basile, S.; Sacchi, P.; Bruno, R.; Pedrazzoli, P. Long-COVID in Patients with Cancer Previously Treated with Early Anti-SARS-CoV-2 Therapies in an Out-of-Hospital Setting: A Single-Center Experience. Cancers 2023, 15, 1269. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yang, S.; Zhao, H.; Cui, R.; Ma, L.; Ge, X.; Fu, Q.; Yu, D.; Niu, X. Comparison of clinical outcomes and risk factors for COVID-19 infection in cancer patients without anticancer treatment and noncancer patients. Front. Public Health 2022, 10, 925519. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tanaka, T.; Nagasu, S.; Furuta, T.; Gobaru, M.; Suzuki, H.; Shimotsuura, Y.; Akiba, J.; Nomura, M.; Fujita, F.; Kawaguchi, T.; et al. Case report: A case of fulminant type 1 diabetes mellitus after COVID-19 vaccination during treatment of advanced gastric cancer: Pitfall in managing immune-related adverse events. Front. Oncol. 2023, 13, 1264281. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gazzaz, Z.J. Diabetes and COVID-19. Open Life Sci. 2021, 16, 297–302. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zaki, N.; Alashwal, H.; Ibrahim, S. Association of hypertension, diabetes, stroke, cancer, kidney disease, and high-cholesterol with COVID-19 disease severity and fatality: A systematic review. Diabetes Metab. Syndr. 2020, 14, 1133–1142. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bisceglia, I.; Canale, M.L.; Gallucci, G.; Turazza, F.M.; Lestuzzi, C.; Parrini, I.; Russo, G.; Maurea, N.; Quagliariello, V.; Oliva, S.; et al. Cardio-Oncology in the COVID Era (Co & Co): The Never Ending Story. Front. Cardiovasc. Med. 2022, 9, 821193, Erratum in Front. Cardiovasc. Med. 2022, 9, 903766; Erratum in Front. Cardiovasc. Med. 2023, 10, 1169176. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Krupka, S.; Hoffmann, A.; Jasaszwili, M.; Dietrich, A.; Guiu-Jurado, E.; Klöting, N.; Blüher, M. Consequences of COVID-19 on Adipose Tissue Signatures. Int. J. Mol. Sci. 2024, 25, 2908. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Martínez-Colón, G.J.; Ratnasiri, K.; Chen, H.; Jiang, S.; Zanley, E.; Rustagi, A.; Verma, R.; Chen, H.; Andrews, J.R.; Mertz, K.D.; et al. SARS-CoV-2 infection drives an inflammatory response in human adipose tissue through infection of adipocytes and macrophages. Sci. Transl. Med. 2022, 14, eabm9151. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Basolo, A.; Poma, A.M.; Bonuccelli, D.; Proietti, A.; Macerola, E.; Ugolini, C.; Torregrossa, L.; Giannini, R.; Vignali, P.; Basolo, F.; et al. Adipose tissue in COVID-19: Detection of SARS-CoV-2 in adipocytes and activation of the interferon-alpha response. J. Endocrinol. Investig. 2022, 45, 1021–1029. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Jing, X.; Wu, J.; Dong, C.; Gao, J.; Seki, T.; Kim, C.; Urgard, E.; Hosaka, K.; Yang, Y.; Long, S.; et al. COVID-19 instigates adipose browning and atrophy through VEGF in small mammals. Nat. Metab. 2022, 4, 1674–1683. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Muzyka, I.; Revenko, O.; Kovalchuk, I.; Savytska, M.; Bekesevych, A.; Kasko, R.; Zayachkivska, O. What is the role of brown adipose tissue in metabolic health: Lessons learned and future perspectives in the long COVID? Inflammopharmacology 2023, 31, 585–595. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Olivo, A.; Marlin, R.; Lazure, T.; Maisonnasse, P.; Bossevot, L.; Mouanga, C.; Lemaitre, J.; Pourcher, G.; Benoist, S.; Le Grand, R.; et al. Detection of SARS-CoV-2 in subcutaneous fat but not visceral fat, and the disruption of fat lymphocyte homeostasis in both fat tissues in the macaque. Commun. Biol. 2022, 5, 542. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Aghili, S.M.M.; Ebrahimpur, M.; Arjmand, B.; Shadman, Z.; Pejman Sani, M.; Qorbani, M.; Larijani, B.; Payab, M. Obesity in COVID-19 era, implications for mechanisms, comorbidities, and prognosis: A review and meta-analysis. Int. J. Obes. 2021, 45, 998–1016. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Haseeb, M.; Shafiq, A.; Sheikh, M.A.; Khan, M.F. Epipericardial Fat Necrosis and COVID-19. Eur. J. Case Rep. Intern. Med. 2024, 11, 004346. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Reiterer, M.; Rajan, M.; Gómez-Banoy, N.; Lau, J.D.; Gomez-Escobar, L.G.; Ma, L.; Gilani, A.; Alvarez-Mulett, S.; Sholle, E.T.; Chandar, V.; et al. Hyperglycemia in acute COVID-19 is characterized by insulin resistance and adipose tissue infectivity by SARS-CoV-2. Cell Metab. 2021, 33, 2174–2188.e5, Erratum in Cell Metab. 2021, 33, 2484. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Dugail, I.; Amri, E.Z.; Vitale, N. High prevalence for obesity in severe COVID-19: Possible links and perspectives towards patient stratification. Biochimie 2020, 179, 257–265. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Liu, X.; Zhang, Z.; Song, Y.; Xie, H.; Dong, M. An update on brown adipose tissue and obesity intervention: Function, regulation and therapeutic implications. Front. Endocrinol. 2023, 13, 1065263. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Malavazos, A.E.; Corsi Romanelli, M.M.; Bandera, F.; Iacobellis, G. Targeting the Adipose Tissue in COVID-19. Obesity 2020, 28, 1178–1179. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Quagliariello, V.; Bonelli, A.; Caronna, A.; Conforti, G.; Iovine, M.; Carbone, A.; Berretta, M.; Botti, G.; Maurea, N. SARS-CoV-2 Infection and Cardioncology: From Cardiometabolic Risk Factors to Outcomes in Cancer Patients. Cancers 2020, 12, 3316. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bakhtiari, M.; Asadipooya, K. Metainflammation in COVID-19. Endocr. Metab. Immune Disord Drug Targets 2022, 22, 1154–1166. [Google Scholar] [CrossRef] [PubMed]
- Huizinga, G.P.; Singer, B.H.; Singer, K. The Collision of Meta-Inflammation and SARS-CoV-2 Pandemic Infection. Endocrinology 2020, 161, bqaa154. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wool, G.D.; Miller, J.L. The Impact of COVID-19 Disease on Platelets and Coagulation. Pathobiology 2021, 88, 15–27. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, H.; Zhang, C.; Hua, W.; Chen, J. Saying no to SARS-CoV-2: The potential of nitric oxide in the treatment of COVID-19 pneumonia. Med. Gas Res. 2024, 14, 39–47. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tseng, Y.H. Adipose tissue in communication: Within and without. Nat. Rev. Endocrinol. 2023, 19, 70–71. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Steenblock, C.; Bechmann, N.; Beuschlein, F.; Wolfrum, C.; Bornstein, S.R. Do adipocytes serve as a reservoir for severe acute respiratory symptom coronavirus-2? J. Endocrinol. 2023, 258, e230027. [Google Scholar] [CrossRef] [PubMed]
- Saccon, T.D.; Mousovich-Neto, F.; Ludwig, R.G.; Carregari, V.C.; Dos Anjos Souza, A.B.; Dos Passos, A.S.C.; Martini, M.C.; Barbosa, P.P.; de Souza, G.F.; Muraro, S.P.; et al. SARS-CoV-2 infects adipose tissue in a fat depot- and viral lineage-dependent manner. Nat. Commun. 2022, 13, 5722. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhu, J.; Wilding, J.P.H.; Hu, J. Adipocytes in obesity: A perfect reservoir for SARS-CoV-2? Med. Hypotheses 2023, 171, 111020. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Quagliariello, V.; Paccone, A.; Iovine, M.; Cavalcanti, E.; Berretta, M.; Maurea, C.; Canale, M.L.; Maurea, N. Interleukin-1 blocking agents as promising strategy for prevention of anticancer drug-induced cardiotoxicities: Possible implications in cancer patients with COVID-19. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 6797–6812. [Google Scholar] [CrossRef] [PubMed]
- Petrelli, F.; Grappasonni, I.; Nguyen, C.T.T.; Tesauro, M.; Pantanetti, P.; Xhafa, S.; Cangelosi, G. Metformin and COVID-19: A systematic review of systematic reviews with meta-analysis. Acta Biomed. 2023, 94, e2023138. [Google Scholar] [CrossRef] [PubMed]
- Zareef, R.; Diab, M.; Al Saleh, T.; Makarem, A.; Younis, N.K.; Bitar, F.; Arabi, M. Aspirin in COVID-19: Pros and Cons. Front. Pharmacol. 2022, 13, 849628. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Su, T.; Zhong, B.; Tang, C.; Qiao, S.; Feng, Y.; Peng, H.; Gu, X. Correlation between epicardial adipose tissue and myocardial injury in patients with COVID-19. Front Physiol. 2024, 15, 1368542. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Favre, G.; Legueult, K.; Pradier, C.; Raffaelli, C.; Ichai, C.; Iannelli, A.; Redheuil, A.; Lucidarme, O.; Esnault, V. Visceral fat is associated to the severity of COVID-19. Metabolism 2021, 115, 154440. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tchernof, A.; Després, J.P. Pathophysiology of human visceral obesity: An update. Physiol. Rev. 2013, 93, 359–404. [Google Scholar] [CrossRef] [PubMed]
- Després, J.P. Health consequences of visceral obesity. Ann. Med. 2001, 33, 534–541. [Google Scholar] [CrossRef] [PubMed]
- Chaaban, N.; Høier, A.T.Z.B.; Andersen, B.V. A Detailed Characterisation of Appetite, Sensory Perceptional, and Eating-Behavioural Effects of COVID-19: Self-Reports from the Acute and Post-Acute Phase of Disease. Foods 2021, 10, 892. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Houben-Wilke, S.; Goërtz, Y.M.; Delbressine, J.M.; Vaes, A.W.; Meys, R.; Machado, F.V.; van Herck, M.; Burtin, C.; Posthuma, R.; Franssen, F.M.; et al. The Impact of Long COVID-19 on Mental Health: Observational 6-Month Follow-Up Study. JMIR Ment. Health 2022, 9, e33704. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lippi, G.; Mattiuzzi, C.; Sanchis-Gomar, F. Physical Activity, Long-COVID, and Inactivity: A Detrimental Endless Loop. J. Phys. Act. Health 2024, 21, 420–422. [Google Scholar] [CrossRef] [PubMed]
- Navas-Otero, A.; Calvache-Mateo, A.; Calles-Plata, I.; Valenza-Peña, G.; Hernández-Hernández, S.; Ortiz-Rubio, A.; Valenza, M.C. A lifestyle adjustments program in long COVID-19 improves symptomatic severity and quality of life. A randomized control trial. Patient Educ. Couns. 2024, 122, 108180. [Google Scholar] [CrossRef] [PubMed]
- Guntur, V.P.; Nemkov, T.; de Boer, E.; Mohning, M.P.; Baraghoshi, D.; Cendali, F.I.; San-Millán, I.; Petrache, I.; D’Alessandro, A. Signatures of Mitochondrial Dysfunction and Impaired Fatty Acid Metabolism in Plasma of Patients with Post-Acute Sequelae of COVID-19 (PASC). Metabolites 2022, 12, 1026. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Janochova, K.; Haluzik, M.; Buzga, M. Visceral fat and insulin resistance—What we know? Biomed. Pap. Med. Fac. Univ. Palacky Olomouc. Czech Repub. 2019, 163, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Al-Hakeim, H.K.; Al-Rubaye, H.T.; Jubran, A.S.; Almulla, A.F.; Moustafa, S.R.; Maes, M. Increased insulin resistance due to Long COVID is associated with depressive symptoms and partly predicted by the inflammatory response during acute infection. Braz. J. Psychiatry 2023, 45, 205–215. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bogdański, A.; Niziołek, P.; Kopeć, S.; Moszak, M. Epicardial Adipose Tissue: A Precise Biomarker for Cardiovascular Risk, Metabolic Diseases, and Target for Therapeutic Interventions. Cardiol. Rev. 2024. ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Mehta, R.; Bello-Chavolla, O.Y.; Mancillas-Adame, L.; Rodriguez-Flores, M.; Pedraza, N.R.; Encinas, B.R.; Carrión, C.I.P.; Ávila, M.I.J.; Valladares-García, J.C.; Vanegas-Cedillo, P.E.; et al. Epicardial adipose tissue thickness is associated with increased COVID-19 severity and mortality. Int. J. Obes. 2022, 46, 866–873. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Iacobellis, G. Epicardial adipose tissue in contemporary cardiology. Nat. Rev. Cardiol. 2022, 19, 593–606. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Boutari, C.; Mantzoros, C.S. A 2022 update on the epidemiology of obesity and a call to action: As its twin COVID-19 pandemic appears to be receding, the obesity and dysmetabolism pandemic continues to rage on. Metabolism 2022, 133, 155217. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cugno, M.; Gualtierotti, R.; Casazza, G.; Tafuri, F.; Ghigliazza, G.; Torri, A.; Costantino, G.; Montano, N.; Peyvandi, F. Mortality in Patients with COVID-19 on Renin Angiotensin System Inhibitor Long-Term Treatment: An Observational Study Showing that Things Are Not Always as They Seem. Adv. Ther. 2021, 38, 2709–2716. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rossi, A.P.; Donadello, K.; Schweiger, V.; Zamboni, G.A.; Dalla Valle, Z.; Zamboni, M.; Polati, E.; Gottin, L. Epicardial adipose tissue volume and CT-attenuation as prognostic factors for pulmonary embolism and mortality in critically ill patients affected by COVID-19. Eur. J. Clin. Nutr. 2023, 77, 105–111. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Meyer, H.J.; Aghayev, A.; Hinnrichs, M.; Borggrefe, J.; Surov, A. Epicardial Adipose Tissue as a Prognostic Marker in COVID-19. Vivo 2024, 38, 281–285. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Correa-de-Araujo, R.; Addison, O.; Miljkovic, I.; Goodpaster, B.H.; Bergman, B.C.; Clark, R.V.; Elena, J.W.; Esser, K.A.; Ferrucci, L.; Harris-Love, M.O.; et al. Myosteatosis in the Context of Skeletal Muscle Function Deficit: An Interdisciplinary Workshop at the National Institute on Aging. Front. Physiol. 2020, 11, 963. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Levy, D.; Giannini, M.; Oulehri, W.; Riou, M.; Marcot, C.; Pizzimenti, M.; Debrut, L.; Charloux, A.; Geny, B.; Meyer, A. Long Term Follow-Up of Sarcopenia and Malnutrition after Hospitalization for COVID-19 in Conventional or Intensive Care Units. Nutrients 2022, 14, 912. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Gambaro, S.E.; Zubiría, M.G.; Portales, A.E.; Rey, M.A.; Rumbo, M.; Giovambattista, A. M1 macrophage subtypes activation and adipocyte dysfunction worsen during prolonged consumption of a fructose-rich diet. J. Nutr. Biochem. 2018, 61, 173–182. [Google Scholar] [CrossRef] [PubMed]
- Li, C.W.; Yu, K.; Shyh-Chang, N.; Jiang, Z.; Liu, T.; Ma, S.; Luo, L.; Guang, L.; Liang, K.; Ma, W.; et al. Pathogenesis of sarcopenia and the relationship with fat mass: Descriptive review. J. Cachexia Sarcopenia Muscle 2022, 13, 781–794. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Murnane, L.C.; Forsyth, A.K.; Koukounaras, J.; Pilgrim, C.H.; Shaw, K.; Brown, W.A.; Mourtzakis, M.; Tierney, A.C.; Burton, P.R. Myosteatosis predicts higher complications and reduced overall survival following radical oesophageal and gastric cancer surgery. Eur. J. Surg. Oncol. 2021, 47, 2295–2303. [Google Scholar] [CrossRef] [PubMed]
- Zhao, W.; Zhang, Y.; Lin, S.; Li, Y.; Zhu, A.J.; Shi, H.; Liu, M. Identification of Ubr1 as an amino acid sensor of steatosis in liver and muscle. J. Cachexia Sarcopenia Muscle 2023, 14, 1454–1467. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Liu, F.P.; Guo, M.J.; Yang, Q.; Li, Y.Y.; Wang, Y.G.; Zhang, M. Myosteatosis is associated with coronary artery calcification in patients with type 2 diabetes. World J. Diabetes 2024, 15, 429–439. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lee, C.M.; Kang, J. Prognostic impact of myosteatosis in patients with colorectal cancer: A systematic review and meta-analysis. J. Cachexia Sarcopenia Muscle 2020, 11, 1270–1282. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Vedder, I.R.; Levolger, S.; Dierckx, R.A.J.O.; Zeebregts, C.J.; de Vries, J.P.M.; Viddeleer, A.R.; Bokkers, R.P.H. Effect of muscle depletion on survival in peripheral arterial occlusive disease: Quality over quantity. J. Vasc. Surg. 2020, 72, 2006–2016.e1. [Google Scholar] [CrossRef] [PubMed]
- Geladari, E.; Alexopoulos, T.; Kontogianni, M.D.; Vasilieva, L.; Mani, I.; Tenta, R.; Sevastianos, V.; Vlachogiannakos, I.; Alexopoulou, A. The Presence of Myosteatosis Is Associated with Age, Severity of Liver Disease and Poor Outcome and May Represent a Prodromal Phase of Sarcopenia in Patients with Liver Cirrhosis. J. Clin. Med. 2023, 12, 3332. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Aleixo, G.F.P.; Shachar, S.S.; Nyrop, K.A.; Muss, H.B.; Malpica, L.; Williams, G.R. Myosteatosis and prognosis in cancer: Systematic review and meta-analysis. Crit. Rev. Oncol. Hematol. 2020, 145, 102839. [Google Scholar] [CrossRef] [PubMed]
- Pozzuto, L.; Silveira, M.N.; Mendes, M.C.S.; Macedo, L.T.; Costa, F.O.; Martinez, C.A.R.; Coy, C.S.R.; da Cunha Júnior, A.D.; Carvalheira, J.B.C. Myosteatosis Differentially Affects the Prognosis of Non-Metastatic Colon and Rectal Cancer Patients: An Exploratory Study. Front. Oncol. 2021, 11, 762444. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Body, S.; Ligthart, M.A.P.; Rahman, S.; Ward, J.; May-Miller, P.; Pucher, P.H.; Curtis, N.J.; West, M.A.; Wessex Research Collaborative. Sarcopenia and Myosteatosis Predict Adverse Outcomes After Emergency Laparotomy: A Multi-center Observational Cohort Study. Ann. Surg. 2022, 275, 1103–1111. [Google Scholar] [CrossRef] [PubMed]
- Aleixo, G.F.P.; Williams, G.R.; Nyrop, K.A.; Muss, H.B.; Shachar, S.S. Muscle composition and outcomes in patients with breast cancer: Meta-analysis and systematic review. Breast Cancer Res. Treat. 2019, 177, 569–579. [Google Scholar] [CrossRef] [PubMed]
- Surov, A.; Meyer, H.J.; Ehrengut, C.; Zimmermann, S.; Schramm, D.; Hinnerichs, M.; Bär, C.; Borggrefe, J. Myosteatosis predicts short-term mortality in patients with COVID-19: A multicenter analysis. Nutrition 2024, 120, 112327. [Google Scholar] [CrossRef] [PubMed]
- De Lorenzo, R.; Palmisano, A.; Esposito, A.; Gnasso, C.; Nicoletti, V.; Leone, R.; Vignale, D.; Falbo, E.; Ferrante, M.; Cilla, M.; et al. Myosteatosis Significantly Predicts Persistent Dyspnea and Mobility Problems in COVID-19 Survivors. Front. Nutr. 2022, 9, 846901. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Fierro, P.; Martín, D.; Pariente-Rodrigo, E.; Pini, S.F.; Basterrechea, H.; Tobalina, M.; Petitta, B.; Bianconi, C.; Díaz-Salazar, S.; Bonome, M.; et al. Post-COVID-19 syndrome, inflammation and insulin resistance: A retrospective cohort study. Minerva Endocrinol. 2024. ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Kamba, A.; Daimon, M.; Murakami, H.; Otaka, H.; Matsuki, K.; Sato, E.; Tanabe, J.; Takayasu, S.; Matsuhashi, Y.; Yanagimachi, M.; et al. Association between Higher Serum Cortisol Levels and Decreased Insulin Secretion in a General Population. PLoS ONE 2016, 11, e0166077. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bastin, M.; Andreelli, F. Diabète et corticoïdes: Nouveautés et aspects pratiques [Corticosteroid-induced diabetes: Novelties in pathophysiology and management]. Rev. Med. Interne 2020, 41, 607–616. (In French) [Google Scholar] [CrossRef] [PubMed]
- Al-Hakeim, H.K.; Khairi Abed, A.; Rouf Moustafa, S.; Almulla, A.F.; Maes, M. Tryptophan catabolites, inflammation, and insulin resistance as determinants of chronic fatigue syndrome and affective symptoms in long COVID. Front Mol Neurosci. 2023, 16, 1194769. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kim, M.J.; Cho, Y.K.; Jung, H.N.; Kim, E.H.; Lee, M.J.; Jung, C.H.; Park, J.Y.; Kim, H.K.; Lee, W.J. Association Between Insulin Resistance and Myosteatosis Measured by Abdominal Computed Tomography. J. Clin. Endocrinol. Metab. 2023, 108, 3100–3110. [Google Scholar] [CrossRef] [PubMed]
- Wu, C.T.; Lidsky, P.V.; Xiao, Y.; Lee, I.T.; Cheng, R.; Nakayama, T.; Jiang, S.; Demeter, J.; Bevacqua, R.J.; Chang, C.A.; et al. SARS-CoV-2 infects human pancreatic β cells and elicits β cell impairment. Cell Metab. 2021, 33, 1565–1576.e5. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lyu, K.; Zhang, D.; Song, J.; Li, X.; Perry, R.J.; Samuel, V.T.; Shulman, G.I. Short-term overnutrition induces white adipose tissue insulin resistance through sn-1,2-diacylglycerol/PKCε/insulin receptor Thr1160 phosphorylation. JCI Insight 2021, 6, e139946. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lai, Y.J.; Liu, S.H.; Manachevakul, S.; Lee, T.A.; Kuo, C.T.; Bello, D. Biomarkers in long COVID-19: A systematic review. Front. Med. 2023, 10, 1085988. [Google Scholar] [CrossRef] [PubMed]
- Espín, E.; Yang, C.; Shannon, C.P.; Assadian, S.; He, D.; Tebbutt, S.J. Cellular and molecular biomarkers of long COVID: A scoping review. EBioMedicine 2023, 91, 104552. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Maamar, M.; Artime, A.; Pariente, E.; Fierro, P.; Ruiz, Y.; Gutiérrez, S.; Tobalina, M.; Díaz-Salazar, S.; Ramos, C.; Olmos, J.M.; et al. Post-COVID-19 syndrome, low-grade inflammation and inflammatory markers: A cross-sectional study. Curr. Med. Res. Opin. 2022, 38, 901–909. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yin, J.X.; Agbana, Y.L.; Sun, Z.S.; Fei, S.W.; Zhao, H.Q.; Zhou, X.N.; Chen, J.H.; Kassegne, K. Increased interleukin-6 is associated with long COVID-19: A systematic review and meta-analysis. Infect. Dis. Poverty 2023, 12, 43. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hirano, T. IL-6 in inflammation, autoimmunity and cancer. Int. Immunol. 2021, 33, 127–148. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Giannitrapani, L.; Mirarchi, L.; Amodeo, S.; Licata, A.; Soresi, M.; Cavaleri, F.; Casalicchio, S.; Ciulla, G.; Ciuppa, M.E.; Cervello, M.; et al. Can Baseline IL-6 Levels Predict Long COVID in Subjects Hospitalized for SARS-CoV-2 Disease? Int. J. Mol. Sci. 2023, 24, 1731. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bergantini, L.; Gangi, S.; d’Alessandro, M.; Cameli, P.; Perea, B.; Meocci, M.; Fabbri, G.; Bianchi, F.; Bargagli, E. Altered serum concentrations of IL-8, IL-32 and IL-10 in patients with lung impairment 6 months after COVID-19. Immunobiology 2024, 229, 152813. [Google Scholar] [CrossRef] [PubMed]
- Nigo, M.; Rasmy, L.; May, S.B.; Rao, A.; Karimaghaei, S.; Kannadath, B.S.; De la Hoz, A.; Arias, C.A.; Li, L.; Zhi, D. Real World Long-term Assessment of The Efficacy of Tocilizumab in Patients with COVID-19: Results From A Large De-identified Multicenter Electronic Health Record Dataset in the United States. Int. J. Infect. Dis. 2021, 113, 148–154. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bogard, G.; Barthelemy, J.; Hantute-Ghesquier, A.; Sencio, V.; Brito-Rodrigues, P.; Séron, K.; Robil, C.; Flourens, A.; Pinet, F.; Eberlé, D.; et al. SARS-CoV-2 infection induces persistent adipose tissue damage in aged golden Syrian hamsters. Cell Death Dis. 2023, 14, 75. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cox, A.R.; Chernis, N.; Masschelin, P.M.; Hartig, S.M. Immune Cells Gate White Adipose Tissue Expansion. Endocrinology 2019, 160, 1645–1658. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bauzá-Thorbrügge, M.; Vujičić, M.; Chanclón, B.; Palsdottir, V.; Pillon, N.J.; Benrick, A.; Wernstedt Asterholm, I. Adiponectin stimulates Sca1+CD34—Adipocyte precursor cells associated with hyperplastic expansion and beiging of brown and white adipose tissue. Metabolism 2024, 151, 155716. [Google Scholar] [CrossRef] [PubMed]
- Perrotta, F.; Scialò, F.; Mallardo, M.; Signoriello, G.; D’Agnano, V.; Bianco, A.; Daniele, A.; Nigro, E. Adiponectin, Leptin, and Resistin Are Dysregulated in Patients Infected by SARS-CoV-2. Int. J. Mol. Sci. 2023, 24, 1131. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, M.C.; Lee, C.J.; Yang, C.F.; Chen, Y.C.; Wang, J.H.; Hsu, B.G. Low serum adiponectin level is associated with metabolic syndrome and is an independent marker of peripheral arterial stiffness in hypertensive patients. Diabetol. Metab. Syndr. 2017, 9, 49. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Subramanian, S.; Liu, C.; Aviv, A.; Ho, J.E.; Courchesne, P.; Muntendam, P.; Larson, M.G.; Cheng, S.; Wang, T.J.; Mehta, N.N.; et al. Stromal cell-derived factor 1 as a biomarker of heart failure and mortality risk. Arter. Thromb. Vasc. Biol. 2014, 34, 2100–2105. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cambier, S.; Beretta, F.; Pörtner, N.; Metzemaekers, M.; de Carvalho, A.C.; Martens, E.; Kaes, J.; Aelbrecht, C.; Jacobs, C.; Van Mol, P.; et al. Proteolytic inactivation of CXCL12 in the lungs and circulation of COVID-19 patients. Cell. Mol. Life Sci. 2023, 80, 234. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Martínez-Fleta, P.; Vera-Tomé, P.; Jiménez-Fernández, M.; Requena, S.; Roy-Vallejo, E.; Sanz-García, A.; Lozano-Prieto, M.; López-Sanz, C.; Vara, A.; Lancho-Sánchez, Á.; et al. A Differential Signature of Circulating miRNAs and Cytokines Between COVID-19 and Community-Acquired Pneumonia Uncovers Novel Physiopathological Mechanisms of COVID-19. Front. Immunol. 2022, 12, 815651. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Noto, A.; Joo, V.; Mancarella, A.; Suffiotti, M.; Pellaton, C.; Fenwick, C.; Perreau, M.; Pantaleo, G. CXCL12 and CXCL13 Cytokine Serum Levels Are Associated with the Magnitude and the Quality of SARS-CoV-2 Humoral Responses. Viruses 2022, 14, 2665. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- D’Avila, H.; Lima, C.N.R.; Rampinelli, P.G.; Mateus, L.C.O.; Sousa Silva, R.V.; Correa, J.R.; Almeida, P.E. Lipid Metabolism Modulation during SARS-CoV-2 Infection: A Spotlight on Extracellular Vesicles and Therapeutic Prospects. Int. J. Mol. Sci. 2024, 25, 640. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yuen, T.T.; Chan, J.F.; Yan, B.; Shum, C.C.; Liu, Y.; Shuai, H.; Hou, Y.; Huang, X.; Hu, B.; Chai, Y.; et al. Targeting ACLY efficiently inhibits SARS-CoV-2 replication. Int. J. Biol. Sci. 2022, 18, 4714–4730. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kridel, S.J.; Lowther, W.T.; Pemble, C.W., 4th. Fatty acid synthase inhibitors: New directions for oncology. Expert Opin. Investig. Drugs 2007, 16, 1817–1829. [Google Scholar] [CrossRef] [PubMed]
- Wagner, N.; Wagner, K.D. Peroxisome Proliferator-Activated Receptors and the Hallmarks of Cancer. Cells 2022, 11, 2432. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wise, J. COVID-19: Metformin reduces the risk of developing long term symptoms by 40%, study finds. BMJ 2023, 381, 1306. [Google Scholar] [CrossRef] [PubMed]
- Kostev, K. Metformin, cancer, COVID-19, and longevity. Int. J. Clin. Pharmacol. Ther. 2023, 61, 93–95. [Google Scholar] [CrossRef] [PubMed]
- Davidsen, L.; Jensen, M.H.; Cook, M.E.; Vestergaard, P.; Knop, F.K.; Drewes, A.M.; Olesen, S.S. Metformin treatment is associated with reduced risk of hypoglycaemia, major adverse cardiovascular events, and all-cause mortality in patients with post-pancreatitis diabetes mellitus: A nationwide cohort study. Eur. J. Endocrinol. 2024, 190, 44–53. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.F.; Hong, C.T.; Chen, W.T.; Chan, L.; Chien, L.N. Metformin adherence and the risk of cardiovascular disease: A population-based cohort study. Ther. Adv. Chronic Dis. 2023, 14, 20406223231163115. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Al-Zadjali, J.; Al-Lawati, A.; Al Riyami, N.; Al Farsi, K.; Al Jarradi, N.; Boudaka, A.; Al Barhoumi, A.; Al Lawati, M.; Al Khaifi, A.; Musleh, A.; et al. Reduced HDL-cholesterol in long COVID-19: A key metabolic risk factor tied to disease severity. Clinics 2024, 79, 100344. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- MCCarthy, M.W. Metformin as a potential treatment for COVID-19. Expert Opin. Pharmacother. 2023, 24, 1199–1203. [Google Scholar] [CrossRef] [PubMed]
- Stromberg, S.; Baxter, B.A.; Dooley, G.; LaVergne, S.M.; Gallichotte, E.; Dutt, T.; Tipton, M.; Berry, K.; Haberman, J.; Natter, N.; et al. Relationships between plasma fatty acids in adults with mild, moderate, or severe COVID-19 and the development of post-acute sequelae. Front. Nutr. 2022, 9, 960409. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Joshi, C.; Jadeja, V.; Zhou, H. Molecular Mechanisms of Palmitic Acid Augmentation in COVID-19 Pathologies. Int. J. Mol. Sci. 2021, 22, 7127. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Yang, C.P.; Chang, C.M.; Yang, C.C.; Pariante, C.M.; Su, K.P. Long COVID and long chain fatty acids (LCFAs): Psychoneuroimmunity implication of omega-3 LCFAs in delayed consequences of COVID-19. Brain Behav. Immun. 2022, 103, 19–27. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Quaranta, P.; Scabia, G.; Storti, B.; Dattilo, A.; Quintino, L.; Perrera, P.; Di Primio, C.; Costa, M.; Pistello, M.; Bizzarri, R.; et al. SARS-CoV-2 Infection Alters the Phenotype and Gene Expression of Adipocytes. Int. J. Mol. Sci. 2024, 25, 2086. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Thomas, T.; Stefanoni, D.; Reisz, J.A.; Nemkov, T.; Bertolone, L.; Francis, R.O.; Hudson, K.E.; Zimring, J.C.; Hansen, K.C.; Hod, E.A.; et al. COVID-19 infection alters kynurenine and fatty acid metabolism, correlating with IL-6 levels and renal status. JCI Insight 2020, 5, e140327. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Petroni, A.; Paroni, R.; Aloisi, A.M.; Blasevich, M.; Haman, N.; Fessas, D. Thermogenic flux induced by lignoceric acid in peroxisomes isolated from HepG2 cells and from X-adrenoleukodystrophy and control fibroblasts. J. Cell. Physiol. 2019, 234, 18344–18348. [Google Scholar] [CrossRef] [PubMed]
- Das, U.N. Can essential fatty acids (EFAs) prevent and ameliorate post-COVID-19 long haul manifestations? Lipids Health Dis. 2024, 23, 112. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Liu, T.H.; Ho, C.H.; Chen, D.T.; Wu, J.Y.; Huang, P.Y.; Lai, C.C.; Hsieh, K.Y.; Su, K.P. Omega-3 polyunsaturated fatty acids and the psychiatric post-acute sequelae of COVID-19: A one-year retrospective cohort analysis of 33,908 patients. Brain Behav Immun. 2023, 114, 453–461. [Google Scholar] [CrossRef] [PubMed]
- Norton, A.; Olliaro, P.; Sigfrid, L.; Carson, G.; Paparella, G.; Hastie, C.; Kaushic, C.; Boily-Larouche, G.; Suett, J.C.; O’Hara, M.; et al. Long COVID: Tackling a multifaceted condition requires a multidisciplinary approach. Lancet Infect. Dis. 2021, 21, 601–602, Erratum in Lancet Infect. Dis. 2021, 21, e81. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Oldani, S.; Petrelli, F.; Dognini, G.; Borgonovo, K.; Parati, M.C.; Ghilardi, M.; Dottorini, L.; Cabiddu, M.; Luciani, A. COVID-19 and Lung Cancer Survival: An Updated Systematic Review and Meta-Analysis. Cancers 2022, 14, 5706. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Avancini, A.; Trestini, I.; Tregnago, D.; Wiskemann, J.; Lanza, M.; Milella, M.; Pilotto, S. Physical Activity for Oncological Patients in COVID-19 Era: No Time to Relax. JNCI Cancer Spectr. 2020, 4, pkaa071. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Lope, V.; Guerrero-Zotano, Á.; Fernández de Larrea-Baz, N.; Antolín, S.; Benavent Viñuales, M.; Bermejo, B.; Ruiz-Moreno, E.; Baena-Cañada, J.M.; París, L.; Antón, A.; et al. Cross-sectional and longitudinal associations of adherence to WCRF/AICR cancer prevention recommendations with health-related quality of life in breast cancer survivors. Health-EpiGEICAM study. J. Nutr. Health Aging 2024, 28, 100312. [Google Scholar] [CrossRef] [PubMed]
- Baldassarre, L.A.; Yang, E.H.; Cheng, R.K.; DeCara, J.M.; Dent, S.; Liu, J.E.; Rudski, L.G.; Strom, J.B.; Thavendiranathan, P.; Barac, A.; et al. Cardiovascular Care of the Oncology Patient During COVID-19: An Expert Consensus Document From the ACC Cardio-Oncology and Imaging Councils. J. Natl. Cancer Inst. 2021, 113, 513–522. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tosato, M.; Ciciarello, F.; Zazzara, M.B.; Pais, C.; Savera, G.; Picca, A.; Galluzzo, V.; Coelho-Júnior, H.J.; Calvani, R.; Marzetti, E.; et al. Nutraceuticals and Dietary Supplements for Older Adults with Long COVID-19. Clin. Geriatr. Med. 2022, 38, 565–591. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bonetti, G.; Medori, M.C.; Fioretti, F.; Farronato, M.; Nodari, S.; Lorusso, L.; Tartaglia, G.M.; Farronato, G.; Bellinato, F.; Gisondi, P.; et al. Dietary supplements for the management of COVID-19 symptoms. J. Prev. Med. Hyg. 2022, 63 (Suppl. 3), E221–E227. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Galluzzo, V.; Zazzara, M.B.; Ciciarello, F.; Savera, G.; Pais, C.; Calvani, R.; Picca, A.; Marzetti, E.; Landi, F.; Tosato, M.; et al. Fatigue in COVID-19 survivors: The potential impact of a nutritional supplement on muscle strength and function. Clin. Nutr. ESPEN 2022, 51, 215–221. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Rossato, M.S.; Brilli, E.; Ferri, N.; Giordano, G.; Tarantino, G. Observational study on the benefit of a nutritional supplement, supporting immune function and energy metabolism, on chronic fatigue associated with the SARS-CoV-2 post-infection progress. Clin. Nutr. ESPEN 2021, 46, 510–518. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Adebayo, A.; Varzideh, F.; Wilson, S.; Gambardella, J.; Eacobacci, M.; Jankauskas, S.S.; Donkor, K.; Kansakar, U.; Trimarco, V.; Mone, P.; et al. l-Arginine and COVID-19: An Update. Nutrients 2021, 13, 3951. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Tosato, M.; Calvani, R.; Picca, A.; Ciciarello, F.; Galluzzo, V.; Coelho-Júnior, H.J.; Di Giorgio, A.; Di Mario, C.; Gervasoni, J.; Gremese, E.; et al. Effects of l-Arginine Plus Vitamin C Supplementation on Physical Performance, Endothelial Function, and Persistent Fatigue in Adults with Long COVID: A Single-Blind Randomized Controlled Trial. Nutrients 2022, 14, 4984. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Trimarco, V.; Izzo, R.; Lombardi, A.; Coppola, A.; Fiorentino, G.; Santulli, G. Beneficial effects of L-Arginine in patients hospitalized for COVID-19: New insights from a randomized clinical trial. Pharmacol. Res. 2023, 191, 106702. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chiscano-Camón, L.; Ruiz-Rodriguez, J.C.; Plata-Menchaca, E.P.; Martin, L.; Bajaña, I.; Martin-Rodríguez, C.; Palmada, C.; Ferrer-Costa, R.; Camos, S.; Villena-Ortiz, Y.; et al. Vitamin C deficiency in critically ill COVID-19 patients admitted to intensive care unit. Front. Med. 2023, 10, 1301001. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kuźmicka, W.; Manda-Handzlik, A.; Cieloch, A.; Mroczek, A.; Demkow, U.; Wachowska, M.; Ciepiela, O. Zinc Supplementation Modulates NETs Release and Neutrophils’ Degranulation. Nutrients 2020, 13, 51. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hasan, R.; Rink, L.; Haase, H. Zinc signals in neutrophil granulocytes are required for the formation of neutrophil extracellular traps. Innate Immun. 2013, 19, 253–264. [Google Scholar] [CrossRef] [PubMed]
- di Filippo, L.; Frara, S.; Nannipieri, F.; Cotellessa, A.; Locatelli, M.; Rovere Querini, P.; Giustina, A. Low Vitamin D Levels Are Associated With Long COVID Syndrome in COVID-19 Survivors. J. Clin. Endocrinol. Metab. 2023, 108, e1106–e1116. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chen, K.Y.; Lin, C.K.; Chen, N.H. Effects of vitamin D and zinc deficiency in acute and long COVID syndrome. J. Trace Elem. Med. Biol. 2023, 80, 127278. [Google Scholar] [CrossRef] [PubMed]
- Izzo, R.; Trimarco, V.; Mone, P.; Aloè, T.; Capra Marzani, M.; Diana, A.; Fazio, G.; Mallardo, M.; Maniscalco, M.; Marazzi, G.; et al. Combining L-Arginine with vitamin C improves long-COVID symptoms: The LINCOLN Survey. Pharmacol. Res. 2022, 183, 106360. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sinopoli, A.; Sciurti, A.; Isonne, C.; Santoro, M.M.; Baccolini, V. The Efficacy of Multivitamin, Vitamin A, Vitamin B, Vitamin C, and Vitamin D Supplements in the Prevention and Management of COVID-19 and Long-COVID: An Updated Systematic Review and Meta-Analysis of Randomized Clinical Trials. Nutrients 2024, 16, 1345. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bian, A.L.; Hu, H.Y.; Rong, Y.D.; Wang, J.; Wang, J.X.; Zhou, X.Z. A study on relationship between elderly sarcopenia and inflammatory factors IL-6 and TNF-α. Eur. J. Med. Res. 2017, 22, 25. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Sakuma, K.; Hamada, K.; Yamaguchi, A.; Aoi, W. Current Nutritional and Pharmacological Approaches for Attenuating Sarcopenia. Cells 2023, 12, 2422. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Bolat, E.; Eker, F.; Kaplan, M.; Duman, H.; Arslan, A.; Saritaş, S.; Şahutoğlu, A.S.; Karav, S. Lactoferrin for COVID-19 prevention, treatment, and recovery. Front. Nutr. 2022, 9, 992733. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Matino, E.; Tavella, E.; Rizzi, M.; Avanzi, G.C.; Azzolina, D.; Battaglia, A.; Becco, P.; Bellan, M.; Bertinieri, G.; Bertoletti, M.; et al. Effect of Lactoferrin on Clinical Outcomes of Hospitalized Patients with COVID-19: The LAC Randomized Clinical Trial. Nutrients 2023, 15, 1285. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Berthon, B.S.; Williams, L.M.; Williams, E.J.; Wood, L.G. Effect of Lactoferrin Supplementation on Inflammation, Immune Function, and Prevention of Respiratory Tract Infections in Humans: A Systematic Review and Meta-analysis. Adv. Nutr. 2022, 13, 1799–1819. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Legrand, D. Overview of Lactoferrin as a Natural Immune Modulator. J. Pediatr. 2016, 173, S10–S15. [Google Scholar] [CrossRef] [PubMed]
- Salaris, C.; Scarpa, M.; Elli, M.; Bertolini, A.; Guglielmetti, S.; Pregliasco, F.; Blandizzi, C.; Brun, P.; Castagliuolo, I. Protective Effects of Lactoferrin against SARS-CoV-2 Infection In Vitro. Nutrients 2021, 13, 328. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hikisz, P.; Bernasinska-Slomczewska, J. Beneficial Properties of Bromelain. Nutrients 2021, 13, 4313. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Owoyele, B.V.; Bakare, A.O.; Ologe, M.O. Bromelain: A Review on its Potential as a Therapy for the Management of COVID-19. Niger. J. Physiol. Sci. 2020, 35, 10–19. [Google Scholar] [PubMed]
- Sagar, S.; Rathinavel, A.K.; Lutz, W.E.; Struble, L.R.; Khurana, S.; Schnaubelt, A.T.; Mishra, N.K.; Guda, C.; Palermo, N.Y.; Broadhurst, M.J.; et al. Bromelain inhibits SARS-CoV-2 infection via targeting ACE-2, TMPRSS2, and spike protein. Clin. Transl. Med. 2021, 11, e281. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Varilla, C.; Marcone, M.; Paiva, L.; Baptista, J. Bromelain, a Group of Pineapple Proteolytic Complex Enzymes (Ananas comosus) and Their Possible Therapeutic and Clinical Effects. A Summary. Foods 2021, 10, 2249. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Pavlidou, E.; Poulios, E.; Papadopoulou, S.K.; Fasoulas, A.; Dakanalis, A.; Giaginis, C. Clinical Evidence on the Potential Beneficial Effects of Diet and Dietary Supplements against COVID-19 Infection Risk and Symptoms’ Severity. Med. Sci. 2024, 12, 11. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Chakraborty, A.J.; Mitra, S.; Tallei, T.E.; Tareq, A.M.; Nainu, F.; Cicia, D.; Dhama, K.; Emran, T.B.; Simal-Gandara, J.; Capasso, R. Bromelain a Potential Bioactive Compound: A Comprehensive Overview from a Pharmacological Perspective. Life 2021, 11, 317, Erratum in Life 2024, 11, 317. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Engwerda, C.R.; Andrew, D.; Ladhams, A.; Mynott, T.L. Bromelain modulates T cell and B cell immune responses in vitro and in vivo. Cell. Immunol. 2001, 210, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Coelho Dos Reis, J.G.A.; Ferreira, G.M.; Lourenço, A.A.; Ribeiro, Á.L.; da Mata, C.P.D.S.M.; de Melo Oliveira, P.; Marques, D.P.A.; Ferreira, L.L.; Clarindo, F.A.; da Silva, M.F.; et al. Ex-vivo mucolytic and anti-inflammatory activity of BromAc in tracheal aspirates from COVID-19. Biomed. Pharmacother. 2022, 148, 112753. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Kritis, P.; Karampela, I.; Kokoris, S.; Dalamaga, M. The combination of bromelain and curcumin as an immune-boosting nutraceutical in the prevention of severe COVID-19. Metabol. Open 2020, 8, 100066. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhang, Y.; Sun, S.; Du, C.; Hu, K.; Zhang, C.; Liu, M.; Wu, Q.; Dong, N. Transmembrane serine protease TMPRSS2 implicated in SARS-CoV-2 infection is autoactivated intracellularly and requires N-glycosylation for regulation. J. Biol. Chem. 2022, 298, 102643. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hou, R.C.; Chen, Y.S.; Huang, J.R.; Jeng, K.C. Cross-linked bromelain inhibits lipopolysaccharide-induced cytokine production involving cellular signaling suppression in rats. J. Agric. Food Chem. 2006, 54, 2193–2198. [Google Scholar] [CrossRef] [PubMed]
- Soheilifar, S.; Bidgoli, M.; Hooshyarfard, A.; Shahbazi, A.; Vahdatinia, F.; Khoshkhooie, F. Effect of Oral Bromelain on Wound Healing, Pain, and Bleeding at Donor Site Following Free Gingival Grafting: A Clinical Trial. J. Dent. 2018, 15, 309–316. [Google Scholar] [PubMed] [PubMed Central]
- Leelakanok, N.; Petchsomrit, A.; Janurai, T.; Saechan, C.; Sunsandee, N. Efficacy and safety of bromelain: A systematic review and meta-analysis. Nutr. Health 2023, 29, 479–503. [Google Scholar] [CrossRef] [PubMed]
- Jurek, J.M.; Castro-Marrero, J. A Narrative Review on Gut Microbiome Disturbances and Microbial Preparations in Myalgic Encephalomyelitis/Chronic Fatigue Syndrome: Implications for Long COVID. Nutrients 2024, 16, 1545. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Wang, Y.H.; Limaye, A.; Liu, J.R.; Wu, T.N. Potential probiotics for regulation of the gut-lung axis to prevent or alleviate influenza in vulnerable populations. J. Tradit. Complement. Med. 2022, 13, 161–169. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Singh, K.; Rao, A. Probiotics: A potential immunomodulator in COVID-19 infection management. Nutr. Res. 2021, 87, 1–12. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Taufer, C.R.; da Silva, J.; Rampelotto, P.H. The Influence of Probiotic Lactobacilli on COVID-19 and the Microbiota. Nutrients 2024, 16, 1350. [Google Scholar] [CrossRef]
- Wiseman, M. The second World Cancer Research Fund/American Institute for Cancer Research expert report. Food, nutrition, physical activity, and the prevention of cancer: A global perspective. Proc. Nutr. Soc. 2008, 67, 253–256. [Google Scholar] [CrossRef] [PubMed]
- Linton, C.; Wright, H.H.; Wadsworth, D.P.; Schaumberg, M.A. Dietary Inflammatory Index and Associations with Sarcopenia Symptomology in Community-Dwelling Older Adults. Nutrients 2022, 14, 5319. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Cailleaux, P.E.; Déchelotte, P.; Coëffier, M. Novel dietary strategies to manage sarcopenia. Curr. Opin. Clin. Nutr. Metab. Care 2024, 27, 234–243. [Google Scholar] [CrossRef] [PubMed]
- Asher, A.; Tintle, N.L.; Myers, M.; Lockshon, L.; Bacareza, H.; Harris, W.S. Blood omega-3 fatty acids and death from COVID-19: A pilot study. Prostaglandins Leukot Essent Fat. Acids 2021, 166, 102250. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Ricker, M.A.; Haas, W.C. Anti-Inflammatory Diet in Clinical Practice: A Review. Nutr. Clin. Pract. 2017, 32, 318–325. [Google Scholar] [CrossRef] [PubMed]
- Barrea, L.; Grant, W.B.; Frias-Toral, E.; Vetrani, C.; Verde, L.; de Alteriis, G.; Docimo, A.; Savastano, S.; Colao, A.; Muscogiuri, G. Dietary Recommendations for Post-COVID-19 Syndrome. Nutrients 2022, 14, 1305. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zafar, M.I.; Mills, K.E.; Zheng, J.; Peng, M.M.; Ye, X.; Chen, L.L. Low glycaemic index diets as an intervention for obesity: A systematic review and meta-analysis. Obes. Rev. 2019, 20, 290–315. [Google Scholar] [CrossRef] [PubMed]
- Ludwig, D.S.; Majzoub, J.A.; Al-Zahrani, A.; Dallal, G.E.; Blanco, I.; Roberts, S.B. High glycemic index foods, overeating, and obesity. Pediatrics 1999, 103, E26. [Google Scholar] [CrossRef] [PubMed]
- Hieronimus, B.; Medici, V.; Lee, V.; Nunez, M.V.; Sigala, D.M.; Bremer, A.A.; Cox, C.L.; Keim, N.L.; Schwarz, J.M.; Pacini, G.; et al. Effects of Consuming Beverages Sweetened with Fructose, Glucose, High-Fructose Corn Syrup, Sucrose, or Aspartame on OGTT-Derived Indices of Insulin Sensitivity in Young Adults. Nutrients 2024, 16, 151. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Turesky, R.J. Mechanistic Evidence for Red Meat and Processed Meat Intake and Cancer Risk: A Follow-up on the International Agency for Research on Cancer Evaluation of 2015. Chimia 2018, 72, 718–724. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quagliariello, V.; Canale, M.L.; Bisceglia, I.; Maurea, C.; Gabrielli, D.; Tarantini, L.; Paccone, A.; Inno, A.; Oliva, S.; Cadeddu Dessalvi, C.; et al. Addressing Post-Acute COVID-19 Syndrome in Cancer Patients, from Visceral Obesity and Myosteatosis to Systemic Inflammation: Implications in Cardio-Onco-Metabolism. Biomedicines 2024, 12, 1650. https://doi.org/10.3390/biomedicines12081650
Quagliariello V, Canale ML, Bisceglia I, Maurea C, Gabrielli D, Tarantini L, Paccone A, Inno A, Oliva S, Cadeddu Dessalvi C, et al. Addressing Post-Acute COVID-19 Syndrome in Cancer Patients, from Visceral Obesity and Myosteatosis to Systemic Inflammation: Implications in Cardio-Onco-Metabolism. Biomedicines. 2024; 12(8):1650. https://doi.org/10.3390/biomedicines12081650
Chicago/Turabian StyleQuagliariello, Vincenzo, Maria Laura Canale, Irma Bisceglia, Carlo Maurea, Domenico Gabrielli, Luigi Tarantini, Andrea Paccone, Alessandro Inno, Stefano Oliva, Christian Cadeddu Dessalvi, and et al. 2024. "Addressing Post-Acute COVID-19 Syndrome in Cancer Patients, from Visceral Obesity and Myosteatosis to Systemic Inflammation: Implications in Cardio-Onco-Metabolism" Biomedicines 12, no. 8: 1650. https://doi.org/10.3390/biomedicines12081650







