Aristolochia trilobata: Identification of the Anti-Inflammatory and Antinociceptive Effects
Abstract
1. Introduction
2. Experimental Section
2.1. Animals
2.2. Plant Material
2.3. Drugs and Treatments
2.4. Formalin-Induced Licking Behavior
2.5. Thermal Nociception Model (Hot-Plate)
2.6. Evaluation of the Possible Mechanisms of Antinociception of A. Trilobata EO and Sulcatyl Acetate
2.7. Carrageenan-Induced Leukocyte Migration into the Subcutaneous Air Pouch (SAP)
2.8. Cell Culture
2.9. Cell Viability Assay
2.10. Quantification of TNF-α and IL-1β
2.11. Nitrate and Nitrite Measurement
2.12. Detection of Enzymes Expression
2.13. Statistical Analysis
3. Results
3.1. Antinociceptive Effect
3.1.1. Formalin-Induced Licking Behavior
3.1.2. Hot-Plate Model
3.1.3. Mechanism of Antinociceptive Action
3.2. Anti-Inflammatory Effect
3.2.1. Leukocyte Migration
3.2.2. Protein Extravasation
3.2.3. Nitric Oxide Production
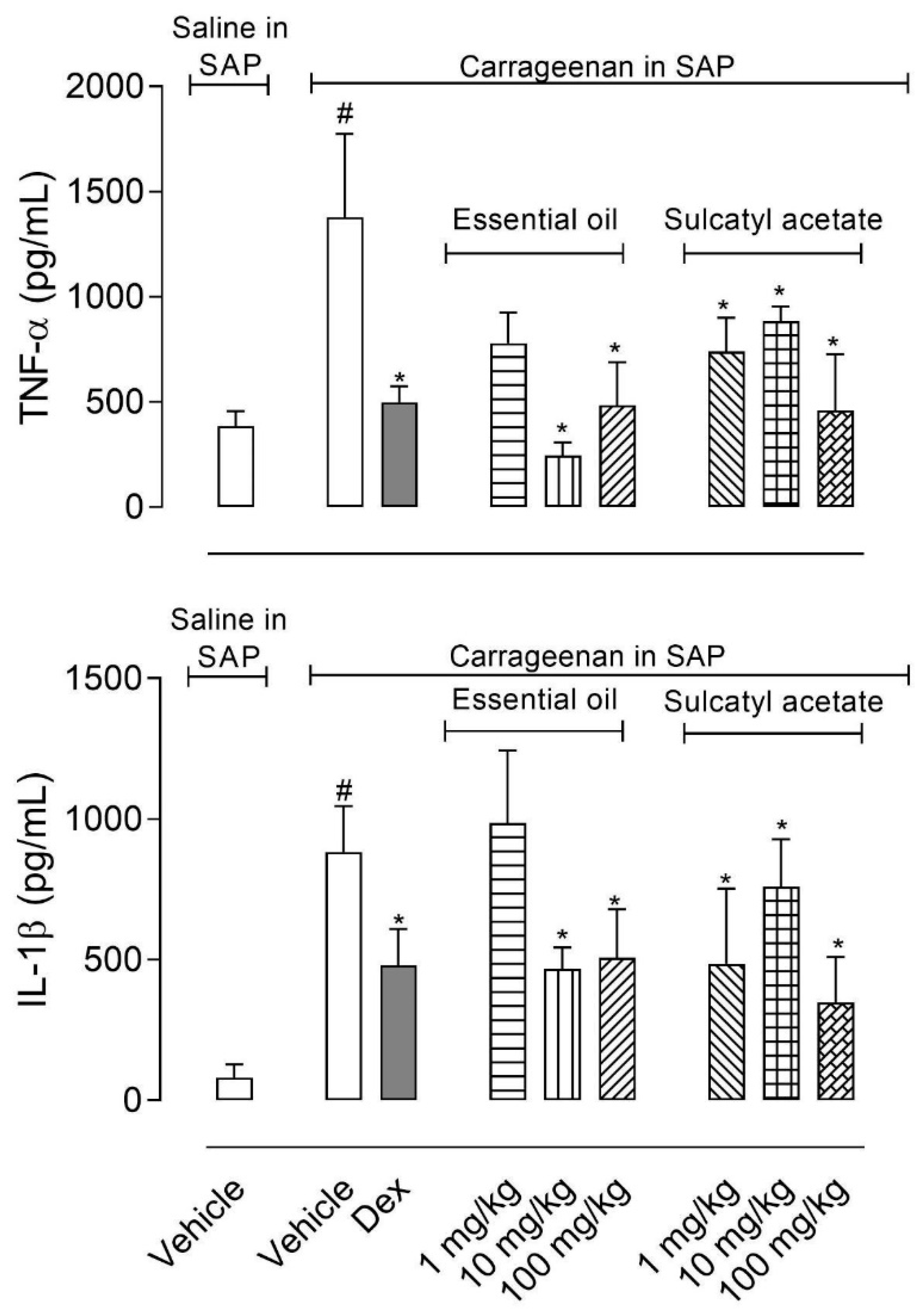
3.2.4. Cytokine Production
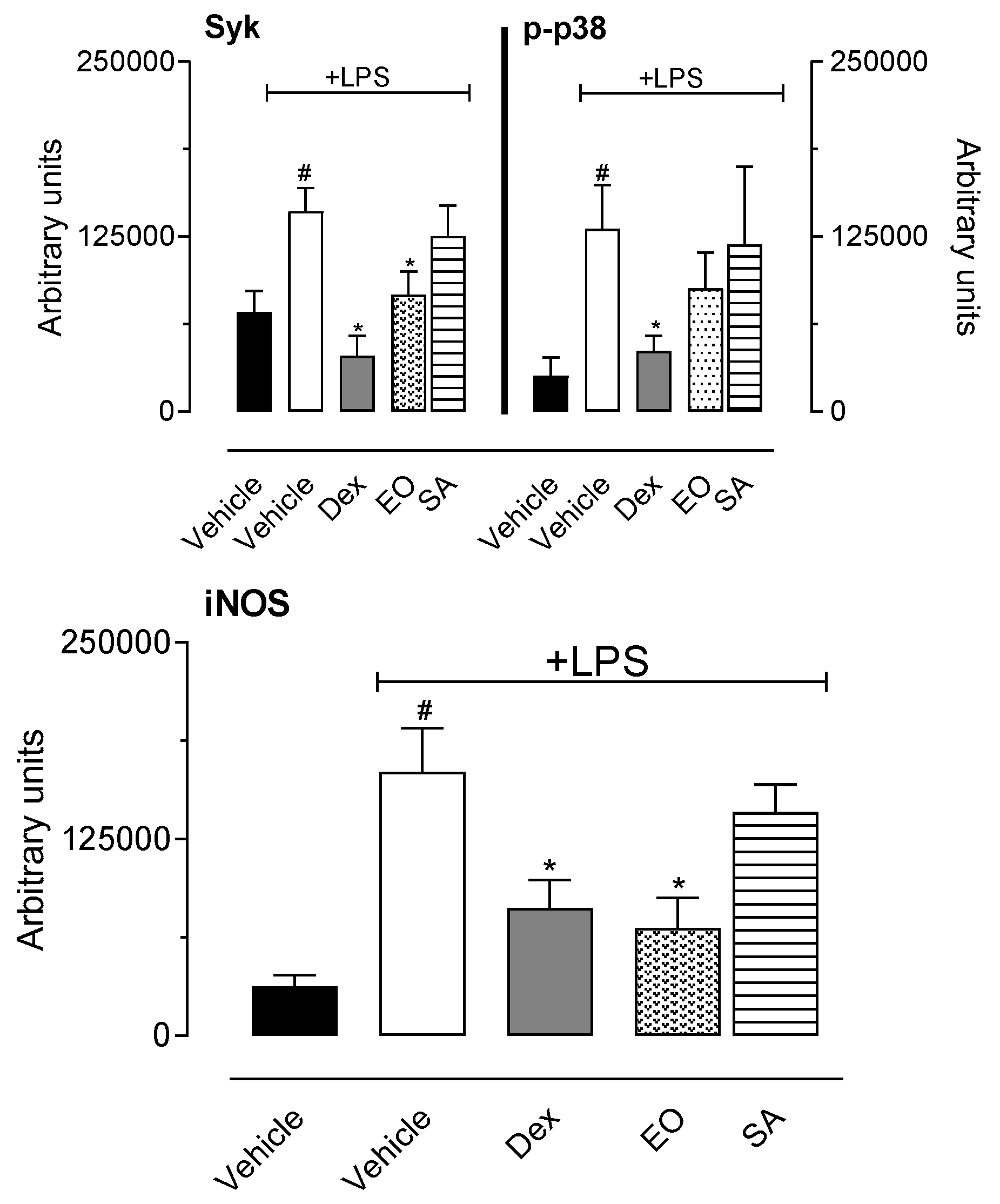
3.2.5. In Vitro Cell Viability and Nitric Oxide Production
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Levine, J.D.; Reichling, D.B. Peripheral mechanisms of inflammatory pain. In Textbook of Pain, 4th ed.; Will, P.A., Melzack, R., Eds.; Churchil Livingstone: London, UK, 1999; pp. 59–84. [Google Scholar]
- Harirforoosh, S.; Asghar, W.; Jamali, F. Adverse effects of nonsteroidal anti-inflammatory drugs: An update of gastrointestinal, cardiovascular and renal complications. J. Pharm. Pharm. Sci. 2013, 16, 821–847. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, M.; Chan, J.; Wanke, S.; Neinhuis, C.; Simmonds, M. Local uses of Aristolochia species and content of nephrotoxic aristolochic acid 1 and 2 a global assessment based on bibliographic sources. J. Ethnopharmacol. 2008, 17, 108–144. [Google Scholar] [CrossRef] [PubMed]
- de Oliveira, B.M.S.; Melo, C.R.; Alves, P.B.; Abraão, A.S.; Santos, A.C.C.; Santana, A.S.; Araújo, A.P.A.; Nascimento, P.E.S.; Blank, A.F.; Bacci, L. Essential oil of Aristolochia trilobata: Synthesis, routes of exposure, acute toxicity, binary mixtures and behavioral effects on leaf-cutting ants. Molecules 2017, 22, 335. [Google Scholar] [CrossRef] [PubMed]
- Lans, C. Comparison of plants used for skin and stomach problems in Trinidad and Tobago with Asian ethnomedicine. J. Ethnobiol. Ethnomed. 2007, 3, 3. [Google Scholar] [CrossRef] [PubMed]
- Sosa, S.; Balick, M.J.; Arvigo, R.; Esposito, R.G.; Pizza, C.; Altinier, G. Screening of the topical anti-inflammatory activity of some Central American plants. J. Ethnopharmacol. 2002, 81, 211–215. [Google Scholar] [CrossRef]
- Quintans, J.S.S.; Alves, R.S.; Santos, D.A.; Serafini, M.R.; Alves, P.B.; Costa, E.V.; Zengin, G.; Quintans-Júnior, L.J.; Guimarães, A.G. Antinociceptive effect of Aristolochia trilobata stem essential oil and 6-methyl-5-hepten-2yl acetate, its main compound, in rodents. Zeitschrift Für Naturforschung C 2017, 72, 93–97. [Google Scholar] [CrossRef]
- Santos, D.A. Volatile constituents of Aristolochia trilobata L. (Aristolochiaceae): A rich source of sulcatyl acetate. Quim. Nova 2014, 37, 977–981. [Google Scholar]
- Sahley, T.L.; Berntson, G.G. Antinociceptive effects of central and systemic administration of nicotine in the rat. Psychopharmacology 1979, 65, 279–283. [Google Scholar] [CrossRef]
- Matheus, M.E.; Berrondo, L.F.; Vieitas, E.C.; Menezes, S.F.; Fernandes, P.D. Evaluation of the antinociceptive properties from Brillantaisia palisotii Lindau stems extracts. J. Ethnopharmacol. 2005, 102, 377–381. [Google Scholar] [CrossRef]
- Fernandes, P.D.; Zardo, P.D.; Figueiredo, G.S.M.; Silva, B.A.; Pinto, A.C. Anti-inflammatory properties of convolutamydine A and two structural analogues. Life Sci. 2014, 116, 16–24. [Google Scholar] [CrossRef]
- Sedgwick, A.D.; Lees, P. Studies of eicosanoid production in the air pouch model of synovial inflammation. Agents Actions 1986, 18, 439–446. [Google Scholar] [CrossRef] [PubMed]
- Raymundo, L.J.R.P.; Guilhon, C.C.; Alviano, D.S.; Matheus, M.E.; Antoniolli, A.R.; Cavalcanti, S.C.H.; Alves, P.B.; Alviano, C.S.; Fernandes, P.D. Characterization of the anti-inflammatory and antinociceptive activities of the Hyptis pectinata (L.) Poit essential oil. J. Ethnopharmacol. 2011, 134, 725–732. [Google Scholar] [CrossRef] [PubMed]
- Denizot, F.; Lang, R. Rapid colorimetric assay for cell growth and survival. Modifications to the tetrazolium dye procedure giving improved sensitivity and reliability. J. Immunol. Meth. 1986, 89, 271–277. [Google Scholar] [CrossRef]
- Bartholomew, B. A rapid method for the assay of nitrate in urine using the nitrate reductase enzyme of Escherichia coli. Food Chem. Toxicol. 1984, 22, 541–543. [Google Scholar] [CrossRef]
- Green, L.C.; Wagner, D.A.; Glogowski, J.; Skipper, P.L.; Wisnok, J.S.; Tannenbaum, S.R. Analysis of nitrate, nitrite, and [5N] nitrate in biological fluids. Anal. Biochem. 1982, 126, 131–138. [Google Scholar] [CrossRef]
- Fernandes, P.D.; Araujo, H.M.; Riveros-Moreno, V.; Assreuy, J. Depolymerization of macrophage microfilaments prevents induction and inhibits activity of nitric oxide synthase. Eur. J. Cell Biol. 1996, 71, 356–362. [Google Scholar]
- do Amaral, J.F.; Silva, M.I.; Neto, M.R.; Neto, P.F.; Moura, B.A.; de Melo, C.T.; de Araújo, F.L.; de Sousa, D.P.; de Vasconcelos, P.F.; de Vasconcelos, S.M.; et al. Antinociceptive effect of the monoterpene R-(+)-limonene in mice. Biol. Pharm. Bull. 2007, 30, 1217–1220. [Google Scholar] [CrossRef]
- Kaiamoto, T.; Hatakeyama, Y.; Takahashi, K.; Imagawa, T.; Tominaga, M.; Ohta, T. Involvement of transient receptor potential A1 channel in algesic and analgesic actions of the organic compound limonene. Eur. J. Pain 2016, 20, 1155–1165. [Google Scholar] [CrossRef]
- Egea, J.; Rosa, A.O.; Lorrio, S.; Barrio, L.; Cuadrado, A.; López, M.G. Haeme oxygenase-1 overexpression via nAChRs and the transcription factor Nrf2 has antinociceptive effects in the formalin test. Pain 2009, 146, 75–83. [Google Scholar] [CrossRef]
- Rosa, O.A.; Egea, J.; Lorrio, S.; Rojo, A.I.; Cuadrado, A.; Lopez, M.G. Nrf2-mediated haeme oxygenase-1 up-regulation induced by cobalt protoporphyrin has antinociceptive effects against inflammatory pain in the formalin test in mice. Pain 2008, 132, 332–339. [Google Scholar] [CrossRef]
- Xie, Y.F.; Huo, F.Q.; Tang, J.S. Cerebral cortex modulation of pain. Acta Pharmacol. Sin. 2009, 30, 31–41. [Google Scholar] [CrossRef] [PubMed]
- Malmberg, A.B.; Yaksh, T.L. Cyclooxygenase inhibition and the spinal release of prostaglandin E2 nd amino acids evoked by paw formalin injection: A microdialysis study in unanesthetized rats. J. Neurosci. 1995, 15, 2768–2776. [Google Scholar] [CrossRef] [PubMed]
- Shibata, M.; Ohkubo, T.; Takahashi, H.; Inoki, R. Modified formalin test: Characteristic biphasic pain response. Pain 1989, 38, 347–352. [Google Scholar] [CrossRef]
- Teng, C.J.; Abbott, F.V. The formalin test: A dose-response analysis at three developmental stages. Pain 1998, 76, 337–347. [Google Scholar] [CrossRef]
- Ward, L.; Wright, E.; McMahon, S.B. A comparison of the effects of noxious and innocuous counter stimuli on experimentally induced itch and pain. Pain 1996, 64, 129–138. [Google Scholar] [CrossRef]
- Wheeler-Aceto, H.; Porreca, F.; Cowan, A. The rat paw formalin test: Comparison of noxious agents. Pain 1990, 40, 229–238. [Google Scholar] [CrossRef]
- Hunskaar, S.; Hole, K. The formalin test in mice: Dissociation between inflammatory and non-inflammatory pain. Pain 1987, 30, 103–114. [Google Scholar] [CrossRef]
- Jaffery, G.; Coleman, J.W.; Huntley, J.; Bell, E.B. Mast cell recovery following chronic treatment with compound 48/80. Int. Arch. Allergy Immunol. 1994, 105, 274–280. [Google Scholar] [CrossRef]
- Parada, C.A.; Tambeli, C.H.; Cunha, F.Q.; Ferreira, S.H. The major role of peripheral release of histamine and 5-hydroxytryptamine in formalin-induced nociception. Neuroscience 2001, 102, 937–944. [Google Scholar] [CrossRef]
- Di Rosa, M.; Sorrentino, L.; Parente, L. Non-steroidal anti-inflammatory drugs and leucocyte emigration. J. Pharm. Pharmacol. 1972, 24, 575–577. [Google Scholar] [CrossRef]
- Ferrandiz, M.L.; Gil, B.; Sanz, M.L.; Ubeda, A.; Gonzalez, E.; Negrete, R.; Pacheco, S.; Paya, M.; Alcarz, M.L. Effect of bakuchiol on leukocytes function and some inflammatory responses in mice. J. Pharm. Pharmacol. 1996, 48, 975–980. [Google Scholar] [CrossRef] [PubMed]
- Saleh, T.S.; Calixto, J.B.; Medeiros, Y.S. Anti-inflammatory effects of theophylline, cromolyn and salbutamol in a murine model of pleurisy. Br. J. Pharmacol. 1996, 118, 811–819. [Google Scholar] [CrossRef] [PubMed]
- Fröde, T.S.; Medeiros, Y.S. Myeloperoxidase and adenosine-deaminase levels in the pleural fluid leakage induced by carrageenan in the mouse model of pleurisy. Mediat. Inflamm. 2001, 10, 223–227. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, M.B.; Farges, R.C.; Fröde, T.S. Involvement of steroids in anti-inflammatory effects of PK11195 in a murine model of pleurisy. Mediat. Inflamm. 2004, 13, 93–103. [Google Scholar] [CrossRef]
- Koo, H.J.; Lim, K.H.; Jung, H.J.; Park, E.H. Anti-inflammatory evaluation of gardenioa extract, geniposide and genipin. J. Ethnopharmacol. 2005, 103, 496–500. [Google Scholar] [CrossRef]
- Rau, B.M.; Kruger, C.M.; Hasel, C.; Oliveira, V.; Rubie, C.; Beger, H.G.; Schilling, M.K. Effects of immunosuppressive and immunostimulative treatment on pancreatic injury and mortality in severe acute experimental pancreatitis. Pancreas 2006, 33, 174–183. [Google Scholar] [CrossRef]
- Gurik, T.J.; Korbut, R.; Adamek-Gurik, T. Nitric oxide and superoxide in inflammation and immune regulation. J. Physiol. Pharmacol. 2003, 54, 469–487. [Google Scholar]
- Paya, M.; Pastor, P.G.; Coloma, J.; Alcaraz, M.J. Nitric oxide synthase and cyclo-oxygenase pathways in the inflammatory response induced by zymosan in the rat air pouch. Br. J. Pharmacol. 1997, 120, 1445–1452. [Google Scholar] [CrossRef]
- Yi, Y.S.; Son, Y.J.; Ryou, C.; Sung, G.H.; Kim, J.H.; Chol, J.Y. Functional roles of Syk in macrophage-mediated inflammatory responses. Mediat. Inflamm. 2014, 270302. [Google Scholar] [CrossRef]
- Lee, J.C.; Laydon, J.T.; McDonnell, P.C.; Gallagher, T.F.; Kumar, S.; Green, D.; McNulty, D.; Blumenthal, M.J.; Heys, J.R.; Landvatter, S.W. A protein kinase involved in the regulation of inflammatory cytokine biosynthesis. Nature 1994, 372, 739–746. [Google Scholar] [CrossRef]
- Hwang, D.; Jang, B.C.; Yu, G.; Boudreau, M. Expression of mitogen inducible cyclooxygenase induced by lipopolysaccharide: Mediation through both mitogen-activated protein kinase and NF-kappaB signaling pathways in macrophages. Biochem. Pharmacol. 1997, 54, 87–96. [Google Scholar] [CrossRef]
- Paul, A.; Cuenda, A.; Bryant, C.E.; Murray, J.; Chilvers, E.R.; Cohen, P.; Gould, G.W.; Plevin, R. Involvement of mitogen-activated protein kinase homologues in the regulation of lipopolysaccharide-mediated induction of cyclo-oxygenase-2 but not nitric oxide synthase in RAW 264.7 macrophages. Cell. Signal. 1999, 11, 491–497. [Google Scholar] [CrossRef]
- Ridley, S.H.; Dean, J.L.; Sarsfield, S.J.; Brook, M.; Clark, A.R.; Saklatvala, J. A p38 MAP kinase inhibitor regulates stability of interleukin-1-induced cyclooxygenase-2 Mrna. FEBS Lett. 1998, 439, 75–80. [Google Scholar] [CrossRef]







© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salomé, D.d.C.; Cordeiro, N.d.M.; Valério, T.S.; Santos, D.d.A.; Alves, P.B.; Alviano, C.S.; Moreno, D.S.A.; Fernandes, P.D. Aristolochia trilobata: Identification of the Anti-Inflammatory and Antinociceptive Effects. Biomedicines 2020, 8, 111. https://doi.org/10.3390/biomedicines8050111
Salomé DdC, Cordeiro NdM, Valério TS, Santos DdA, Alves PB, Alviano CS, Moreno DSA, Fernandes PD. Aristolochia trilobata: Identification of the Anti-Inflammatory and Antinociceptive Effects. Biomedicines. 2020; 8(5):111. https://doi.org/10.3390/biomedicines8050111
Chicago/Turabian StyleSalomé, Dayana da Costa, Natália de Morais Cordeiro, Tayná Sequeira Valério, Darlisson de Alexandria Santos, Péricles Barreto Alves, Celuta Sales Alviano, Daniela Sales Alviano Moreno, and Patricia Dias Fernandes. 2020. "Aristolochia trilobata: Identification of the Anti-Inflammatory and Antinociceptive Effects" Biomedicines 8, no. 5: 111. https://doi.org/10.3390/biomedicines8050111
APA StyleSalomé, D. d. C., Cordeiro, N. d. M., Valério, T. S., Santos, D. d. A., Alves, P. B., Alviano, C. S., Moreno, D. S. A., & Fernandes, P. D. (2020). Aristolochia trilobata: Identification of the Anti-Inflammatory and Antinociceptive Effects. Biomedicines, 8(5), 111. https://doi.org/10.3390/biomedicines8050111