Abstract
Synthetic and naturally occurring nano-sized particles present versatile vehicles for the delivery of therapy in a range of clinical settings. Their small size and modifiable physicochemical properties support refinement of targeting capabilities, immune response, and therapeutic cargo, but rapid clearance from the body and limited efficacy remain a major challenge. This highlights the need for a local sustained delivery system for nanoparticles (NPs) and extracellular vesicles (EVs) at the target site that will ensure prolonged exposure, maximum efficacy and dose, and minimal toxicity. Biocompatible hydrogels loaded with therapeutic NPs/EVs hold immense promise as cell-free sustained and targeted delivery systems in a range of disease settings. These bioscaffolds ensure retention of the nano-sized particles at the target site and can also act as controlled release systems for therapeutics over a prolonged period of time. The encapsulation of stimuli sensitive components into hydrogels supports the release of the content on-demand. In this review, we highlight the prospect of the sustained and prolonged delivery of these nano-sized therapeutic entities from hydrogels for broad applications spanning tissue regeneration and cancer treatment. Further understanding of the parameters controlling the release rate of these particles and efficient transfer of cargo to target cells will be fundamental to success.
1. Introduction
Nanoparticles (NPs) are nanoscale entities consisting primarily of lipid and polymeric structures. The ability of NPs to encapsulate components including proteins, peptides, nucleic acids and small molecules has resulted in widespread interest in their potential as targeted delivery agents for therapeutic drugs [1]. Over the last few years, the significance of nano-sized particles has escalated due to their potential use in the clinical setting for the treatment of various diseases as they can elicit site specific effects as well as be tuned for time-controlled release [2,3,4]. NPs can also be modified or engineered to elicit an enhanced therapeutic effect at the target site due to improved pharmacokinetics and pharmacodynamics, and active intracellular delivery [5]. Although NPs are known to be readily phagocytized by macrophages as an immediate host immune response to a foreign body, these limitations are now being overcome through the modulation of physicochemical properties such as surface charge, size, steric effects, and surface modifications.
For example, many nanoparticles are now being coated with different types of polymers in order to bypass the immune system as they form a hydrophilic coating [5,6]. Nanotherapeutics have the potential to ensure enhanced bioavailability, prolonged effects of the therapeutic gene or drug at the target site, as well as abated chemical/enzymatic degradation resulting in enhanced stability [7,8]. The commonly employed nanoparticles include biodegradable NPs, lipid-based NPs, polymeric NPs, and micelles that were found to be potential drug delivery systems. Among these, phospholipid-based vesicles known as liposomes have been prominent nanocarriers for the delivery of therapeutic cargo. This is due to the phospholipid bilayers that contain an internal hydrophilic compartment, supporting the ability to encapsulate hydrophilic as well as hydrophobic constituents effectively based on their synthesis [9,10]. The first nanomedicines to be approved for clinical use by the FDA were liposomes. Liposome formulations carrying anti-cancer drugs, such as doxorubicin and amphotericin B, were approved in the 1990s. Liposomes usually have shorter half-lives due to rapid clearance of these particles by circulating macrophages. PEGylation of NPs was found to minimize clearance and was adopted for the treatment of various malignancies, fungal infections, and macular degeneration [11,12,13,14]. A liposomal Irinotecan is the most recently approved drug carrier that works by passively targeting pancreatic cancer as a topoisomerase I inhibitor [11]. Over the years more than 25 inorganic NPs have also been approved for clinical use by the FDA, with the first one being Cosmo Fer for treating iron deficiency in 1974 [15].
Similar to the synthetic NPs, nature has its own nanoparticles known as extracellular vesicles (EVs), which are lipid bound vesicles secreted by almost all cell types into the extracellular space. Recent findings have highlighted the role of EVs in intercellular communication resulting in the transfer of a wide range of nucleic acids, peptides and proteins that are now being developed for the treatment of multiple diseases [16]. The composition of these vesicles is mainly governed by the cell of origin, the local niche, the pathways involved in biogenesis and the cargo sorting routes. “EVs” is an umbrella term that has been advocated for nano-sized vesicles including microvesicles, exosomes and apoptotic bodies, which are defined by their respective size and biogenesis pathway [17,18]. These vesicles resemble synthetic liposomes as they are bound by lipid membranes that provide protection against proteases and nucleases [19,20]. EVs are therefore an intense topic of research at the moment as they have emerged as potent carriers for the delivery of therapeutic cargo mainly because they are believed to be the fingerprint of the secreting cell, and ideally could bypass the immune system if isolated from immune-compatible cell sources [21]. The use of EVs also bears the potential to replace cell therapy and its associated complications such as undesirable immunological reactions, the transfer of mutated or damaged genetic material and transformation potential [20]. The small size of EVs enables easy transit across biological barriers, the potential to bypass the immune system and to home to the target site based on its surface modifications [20].
Currently, the majority (>30) of EV-based clinical trials focus on employing them as a tool for diagnosis of various diseases by isolating EVs from body fluids and detection of EV cargo indicative of disease. Based on recent reports there are >20 ongoing clinical trials that employ EV therapeutics for different diseases [22,23]. A recently completed randomized controlled study highlighted the efficacy of platelet rich plasma (PRP) containing EVs for the treatment of temporal bone cavities [24]. Another ongoing clinical trial is also focused on the use of PRP rich in EVs for the treatment of ear infections [25]. As there are many ongoing clinical trials with Mesenchymal Stromal Cells (MSCs) mainly attributing to their regenerative capacity, MSC-EVs have also been studied to analyze their potential for therapeutic benefit. A trial for the treatment of advanced stages of colorectal cancer involved subcutaneous administration of 2 × 1012 ascites-derived EVs weekly for a month and was found to be safe and effective by eliciting antigen-specific T lymphocyte response [26]. Another ongoing Phase I clinical trial for the treatment of metastatic pancreatic ductal carcinoma caused by a mutation in KrasG12D, involves treatment with MSC-EVs carrying siRNA to this KrasG12D mutation. Patients will be systemically injected with three doses of EVs biweekly for three courses and will be monitored for a year [27]. Several clinical trials employing MSC-EV treatment are now focused on the treatment of the coronavirus (SARS-COVID-2). Aerosol administration of 2 × 108 vesicles/3 mL for a week is also being carried out on COVID-19 patients in China [28]. Although clinical trials results have not yet been released, multiple studies are ongoing, such as the phase-2 clinical trial for moderate to severe COVID-19, thereby highlighting the immense and broad potential of therapeutic EVs [29].
2. Limitations in the Current Route of Administration of NPs and EVs
Although NPs and EVs offer exciting potential as drug carrier systems, they come with certain limitations. When introduced in the body, they are subjected to several physical and biological obstacles that alter the dosage of the NPs/EVs reaching the target site. These blocks mainly include phagocytic sequestration, aggregation, harsh flow and shear forces, varying pH, diffusion, renal clearance, and much more [30]. The nano size and suitable structure on the one hand enables these particles to traverse multiple physiological barriers, but on the other hand results in rapid clearance from the body. Studies by Takahashi et al., revealed that B16-BL6 murine melanoma cell EVs are rapidly cleared from the circulation when administered via direct intravenous, intraperitoneal or subcutaneous injections and are found to be accumulated at non-specific sites such as the liver, spleen, gastrointestinal tracts, kidneys and lungs [31,32]. Wiklander et al. [33] also reported that the majority of injected exosomes were rapidly cleared by macrophages in the reticuloendothelial system. The clearance of exosomes by the immune system was found to vary considerably based on the route of administration and the different cell sources. Another study using exosomes for the treatment of pancreatic cancer indicated the ability of CD47 positive exosomes to evade phagocytosis by circulating monocytes, increasing their half-life in circulation [34]. Along with rapid clearance, another major limitation is the struggle of mass production of pure and consistent EVs [20,35]. Dynamic distribution also results in decreased retention of the drug at the target site as well as increased risk of exposing healthy tissues to undesired toxicity. These impediments associated with the systemic delivery of NPs/EVs limit the wide use of nanomedicines in the clinic [36]. This highlights the need for a local sustained delivery system at the target site that will ensure prolonged exposure, maximum efficacy and minimal toxicity [36]. In order to enhance the therapeutic index, many studies now focus on the establishment of a controlled drug delivery system that incorporates NPs/EVs into biomaterials [35,36,37,38,39,40,41,42].
3. Hydrogels as Reservoirs for Sustained Delivery of NPs/EVs
This review highlights the use of sustained delivery systems for the targeted delivery of NPs/EVs using highly porous biomaterials known as hydrogels. These have been extensively used as efficient drug delivery systems that ensure sustained release of the therapeutics encapsulated within. These 3D constructs of insoluble matrices are formed by the crosslinking of hydrophilic copolymers, macromers or homopolymers [43]. Hydrogel efficiency is attributed to the cross-linked networks of water-soluble polymers that provide a 3D structure that is highly porous, resulting in enhanced encapsulation of therapeutic particles as well as ensuring sustained release [43].
These hydrophilic polymers bear the potential to absorb water from minimal amounts up to a thousand times their dry weight giving them the ability to either be chemically stable or subsequently disintegrate. The commonly employed polymers for biomedical applications include synthetic hydrogels such as PEG (poly-ethylene glycol), PVA (Poly-vinyl alcohol) and PHEMA (poly(2-hydroxyethyl methacrylate)) or natural polymers such as collagen, hyaluronic acid, chitosan, alginate and agarose [44]. Most of the hydrogels commonly employed are biocompatible as they are produced from naturally occurring components of the human body such as collagen, hyaluronic acid, fibrin, dextran, and so forth [45]. Many studies have focused on the use of hydrogels for encapsulation of cells [46,47,48,49], however due to the limitations associated with cell therapies many approaches are now based on sustained drug delivery systems containing therapeutic NPs.
These have been incorporated into hydrogels through a variety of approaches, including the mixing of NPs with monomers, incorporating them into the polymeric solution. NPs can also be added to pre-made gels by the process of breathing, where hydrogels are soaked in a solution of NPs to enable their uptake by swelling, or by in-situ conversion where NPs are formed inside the hydrogels by the use of a precursor [50]. Previous studies have achieved sustained release of NPs by regulating the ratio between the mean mesh size of the hydrogels and the diameter of the NPs [51]. The retention and release of therapeutic proteins are mainly regulated by the mesh size of the hydrogels [52]; however, as hydrogels are not homogenous physical enmeshments, the crosslinked network is not sufficient for encasing the NPs. Therefore, many studies are now focused on electrostatic interactions that can be established in polymers for tuning the release kinetics [51]. Therapeutic drugs/NPs are retained in the hydrogel matrix by hydrophobic or charge interactions, hydrogen bonding, stereocomplexation, covalent crosslinking or small molecule cross linking [43]. The stability of NPs in the hydrogel system is also an important consideration. This has been addressed through alteration of the amount of crosslinkers used, and also through the use of surfactants or microgels [53]. Recent studies have reported the capacity of the side chains of synthetic hydrogels to enhance stability when their chemical functionality is altered [54]. The monomers or semi-polymer chains of hydrogels have also been reported to be key regulators of maintaining the size and shape of silver NPs [55]. The highly porous nature enables enhanced drug loading capacity along with the subsequent release from hydrogel complexes based on the diffusion coefficient of the small molecule [43]. The encapsulation of stimuli sensitive components into these “intelligent hydrogels” enables systemic release of the content on-demand. A popular example of such well controlled systems is the glucose-sensitive hydrogels that modulate insulin levels in patients based on the glucose concentration at that moment in order to maintain stable blood sugar levels at all times [37,56].
Hydrogels can also be formulated into different shapes and thickness based on requirements. As a result, these are now being extensively used in clinical practice as well as experimental medicine. Currently, 30 injectable hydrogels have been approved by the FDA with applications mainly focusing on skin regeneration, osteoarthritis and spinal cord regeneration [57]. Many synthetic hydrogels such as polyacrylamide gels as well as natural gels such as hyaluronan and hydroxyethyl cellulose are being investigated for the treatment of osteoarthritis [58]. Hydrogels have also been extensively investigated for ocular therapeutics; the only candidate for age-related macular degeneration (AMD) in clinical trials is the Ocular Therapeutix’s OTX-TKI (tyrosine kinase inhibitor microcrystals in PEG hydrogel) [57]. Compared to other groups of synthetic biomaterials, hydrogels are very similar to the living tissues of the human body due to their water absorption capacity. Due to this they possess the unique ability to undergo various alterations in the gel structure such as swelling, dissolution or degradation that results in sustained release of the therapeutic particles upon external stimuli, making them responsive to factors in the microenvironment such as temperature and pH, thereby making them potential candidates for sustained drug delivery systems. Advances in hydrogel technologies have spiked over the past few decades for several biomedical applications especially those involving sustained drug delivery. Therefore, many novel hydrogel based delivery systems have been established with multiple small molecule drugs, NPs and EVs to fulfil the escalating demand of the pharmaceutical and medical industry (Table 1).

Table 1.
Nanoparticles (NPs) delivered via biomaterial scaffolds for tissue repair in representative examples in various injury models.
4. Sustained Delivery of NPs/EVs for Tissue Regeneration
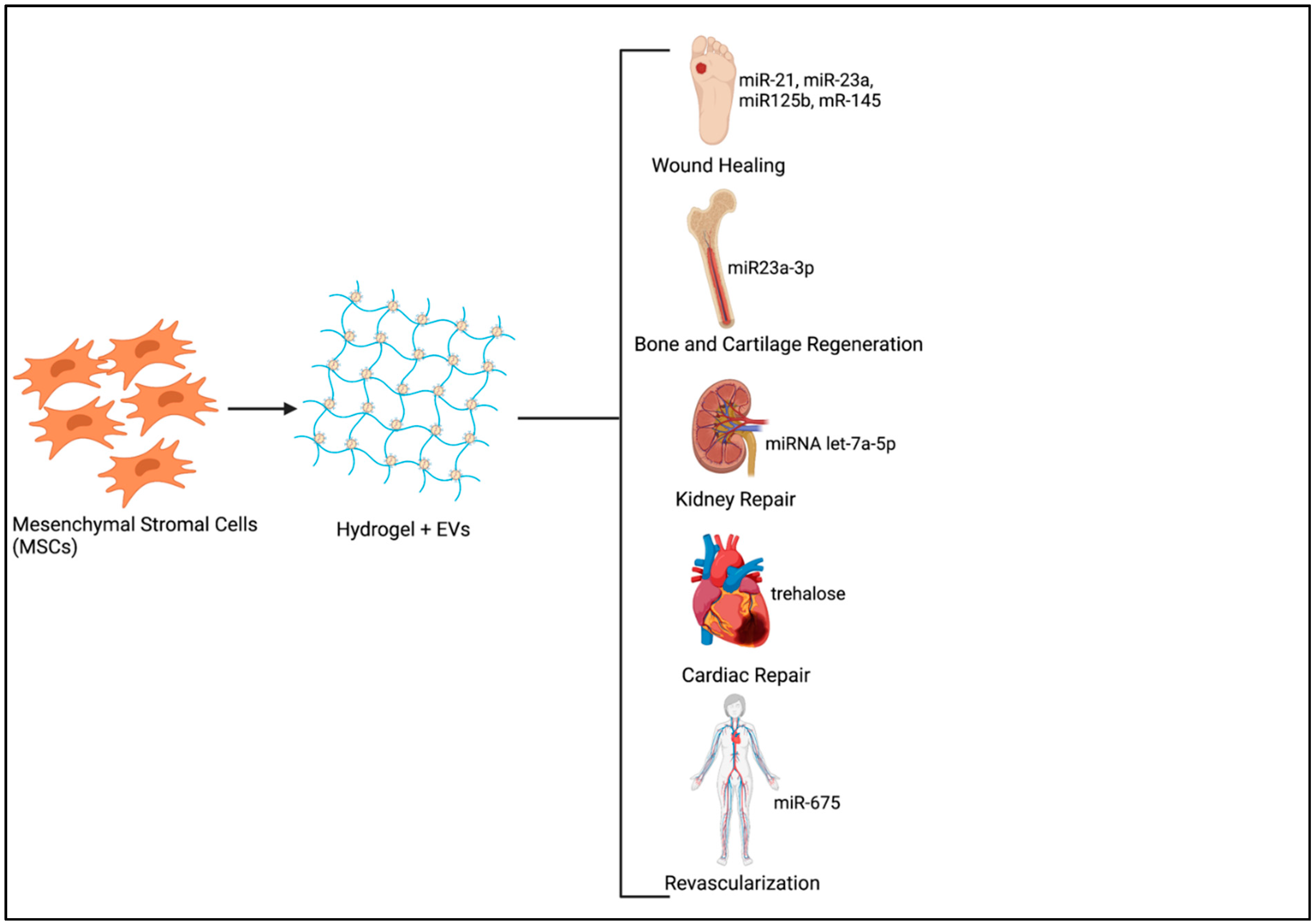
Harnessing the body’s own regenerative capacity has been extensively researched over several decades. Many studies have focused on the use of different therapeutics for triggering endogenous regeneration, such as instructive biomaterial scaffolds, NPs, small molecules, and so forth [72]. Therapeutic NPs have been exploited for potential regenerative applications mainly due to their versatility in terms of altering surface chemistry, size and potential to be used as delivery vehicles for the transfer of drugs, growth factors, small molecules, or genetic material [73,74]. Tissue repair and regeneration require extended periods of exposure to therapeutic molecules with increased uptake of these substances at the injured site by endocytosis being crucial for exerting the expected biological effects. The encapsulation of EVs in a hydrogel introduced at the tissue defect is therefore the best way forward as it not only enhances EV half-life by minimizing non-specific binding at other sites, but also ensures optimal therapeutic effects at minimal doses [44,75] (Figure 1).
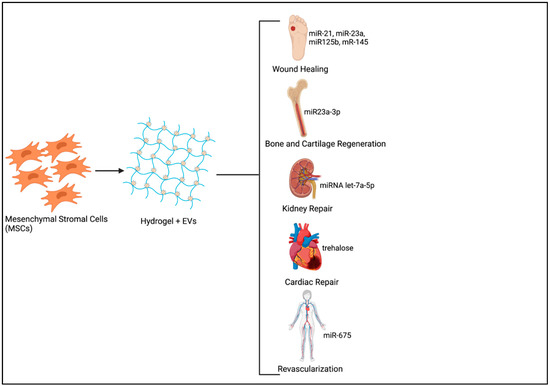
Figure 1.
Sustained Delivery system of MSC derived Extracellular vesicles (EVs) for Tissue Regeneration. (Image created using www.Biorender.com-paid subscription, accessed on 29 June 2021, Biorender 2021).
4.1. Wound Healing
Limited systemic treatments are available for chronic injuries such as diabetic wounds mainly due to dysregulation of the cellular response as well as high incidence of infections, therefore novel therapeutic approaches are constantly being developed. The delayed healing not only results in infections such as gangrene and eventual amputation of the infected organ but can also be lethal in severe cases. The conventional treatment with limited success includes antibacterial dressings with plasters and biocompatible hydrogels [40,76,77,78]. Multifold studies over the years have highlighted the regenerative capacity of metal NPs especially gold and silver NPs mainly attributing efficacy to the tunable size and surface characteristics.
For example, sustained release of silver NPs through polymeric hydrogels has been found to be promising for wound healing due to the antimicrobial and healing properties [64,74]. Hydrogels responsive to varying pH have also been found to be effective drug delivery systems for wound healing. A recent study highlighted the potential of silver NPs loaded in pH responsive hydrogels as a potential candidate for wound healing. This was due to the unique property of switching to a hydrated state in the alkaline wound environment, resulting in the sustained release of silver NPs with antibacterial properties [65]. Another study assessing the efficacy of gold nanorods released from different hydrogels namely polyethylene glycol (PEG) and cationic poly allyl amine hydrochloride (PAH) demonstrated prominent wound healing ability with complete closure of the wound within 14 days of the treatment along with improved skin re-epithelialization and collagen formation as well as noticeable impact on the inflammatory gene expression [79]. Combination hydrogel composed of chitosan and poly-vinyl alcohol dispensing heparinized zinc NPs has been considered as an effective wound dressing material attributing to its enhanced antibacterial and rapid wound healing properties with complete wound closure within 14 days [66]. Another study highlighted the immunomodulatory effects of MSC exosomes when delivered using polymeric fibrous scaffolds in a murine model. The scaffolding material and exosomes were found to attract immune cells by modulating macrophage, T helper cell and T regulatory cell responses and in turn promoting tissue repair [80].
Stable EV delivery systems comprising of biocompatible hydrogels are also being extensively studied for their therapeutic regenerative capacity. EVs derived from Human-Gingival MSCs (hGMSC-EVs) when incorporated into chitosan/silk hydrogel sponge were found to significantly promote wound healing by augmenting re-epithelialization along with extracellular matrix (ECM) deposition and remodeling, and angiogenesis in a diabetic rat model [40]. Another study using commercially available hydro-matrix hydrogel highlighted the wound healing ability of sustained exposure to Human Umbilical cord derived MSCs (hUCMSC-EVs) enriched with microRNAs (miR-21, miR-23a, miR-125b and miR-145). Prolonged exposure to these EVs resulted in a significant decline in scarring as well as myofibroblast formation in the in-vivo model [81]. Adipose stromal cell derived EVs (ADSC-EVs) when encased in alginate hydrogels were also found to elicit similar wound healing properties by not only improving the wound closure and promoting angiogenesis, but also enhancing collagen deposition at the injured site [82].
4.2. Bone and Cartilage Regeneration
The demand for tissue engineering solutions for effective bone and cartilage regeneration has escalated over the years due to an aging population with associated rapid decline in bone healing ability. Bio-printed 3D scaffolds have been observed to provide the required mechanical support that accelerates bone regeneration [83,84]. Biocompatible hydrogels used as reservoirs of therapeutic NPs and growth factors have also been assessed for their efficacy in bone regeneration. A significant increase in bone regeneration was also observed in a nanofiber scaffold of poly(ε-caprolactone) (PCL) and poly(glycerol sebacate) (PGS) packed with hydroxyapatite NPs (HANPs) and simvastatin (SIM) with a significant increase in mineralization and bone formation [85]. Thermosensitive chitosan based hydrogels were found to bear notable potential for continuous local delivery of stromal cell-derived factor-1α (SDF-1α) loaded NPs for the treatment of calvarial defects [67].
Studies have previously confirmed the clinical efficacy of chitosan hydrogels for the delivery of growth factors and soluble factors to promote chondrogenic regeneration [86]. Synthetic hydrogels have also been investigated as potential therapy for articular cartilage damage. To simulate the lubrication of cartilage, which is critical to function, Lin et al. [87] employed multilamellar vesicles (MLVs) to incorporate phosphatidylcholine (PC) dimyristoylphosphatidylcholine (DMPC) or hydrogenated soy (HSPC) lipids into synthetic hydrogels. The lipids were distributed through microreservoirs to support the continuous replenishment of lubrication as they wear. This lipid incorporation was found to result in a 95%–99% reduction in friction [87]. This promising data support the potential of self-lubricating hydrogels for reduction of the impact of friction and wear [87]. Improved cartilage repair was observed with a composite hydrogel system of silk fibroin loaded with BMP-2 as well as chitosan NPs containing TGF-β1. The in vitro and in-vivo models not only displayed enhanced biocompatibility but also improved chondrogenesis at the cartilage defect when compared to the hydrogel alone [68].
Stem cell derived EVs were found to improve osteogenic differentiation by enhancing osteogenic gene expression through paracrine signaling factors. MSC derived EVs from dental tissues have also been observed to promote skeletal regeneration when administered to the damaged bone site for prolonged time frames using different scaffolding biomaterials [41]. Studies using synthetic Poly-Lactide Scaffold (PLA) or natural collagen scaffold for assessing the prolonged effects of therapeutic EVs over a period of 6 weeks also demonstrated pro-osteogenic alterations at the site of damage. The sustained exposure to EVs not only supported formation of new bone nodules and increased mineralization by having an effect on the osteogenic genes, but also promoted the vascular network that resulted in rapid bone regeneration [41,88]. Complex combination hydrogels comprising of coralline hydroxyapatite, silk fibroin, glycol chitosan along with synthetic polyethylene glycol (PEG) for the delivery of hUCMSC-EVs were found to accelerate bone ossification by escalating BMP-2 and collagen deposition and promoting angiogenesis. The factors contributing to the sustained delivery of EVs were the uniform pore size and connectivity in the hydrogel along with its hydrophilicity, making it an attractive biomaterial as the therapeutic effects were observed over a period of 90 days at the site of injury [89].
With limited success through innate healing, the clinical demand for therapeutic options for articular cartilage repair is ever increasing. Articular cartilage lesions were significantly restored when treated with induced pluripotent stem cell cells (iPSC)-derived exosomes integrated with a Photoinduced Imine Crosslinking (PIC) hydrogel [35]. The exosome-hydrogel tissue patch not only maintained a consistent supply of EVs but also integrated with the native cartilage cells over a period of 14 days making this acellular hydrogel glue scaffold a promising candidate for cartilage regeneration [35]. MSC-EVs enriched with miR-23a-3p delivered using a Gelma/nanoclay hydrogel were also found to significantly promote cartilage regeneration by activating PTEN/AKT signaling pathway [90].
4.3. Vascularization and Cardiac Repair
Consistent supply of oxygen and nutrients is essential for the regeneration and maintenance of cells and tissues. Scaffolds promoting vascularization have thereby been extensively developed in order to provide the required support that would mimic the native extracellular matrix (ECM). However, these lack biological constituents that would enhance revascularization [42]. Employing synthetic NP delivery based hydrogel systems have shown significant effects in promoting revascularization by sustained local delivery of angiogenic factors such as VEGF. The delivery of free VEGF using a hydrogel resulted in rapid release over a period of 24hrs, however upon integration of PGA-NPs loaded with VEGF in the hydrogel, sustained discharge in vitro for up to 35 days was observed thereby accentuating its potential use for numerous cardiac applications [59].
Immobilization of placenta derived MSC-EVs onto synthetic scaffolds was also found to promote revascularization [42]. The EVs were immobilized on to the scaffolds using integrin ligand LLP2A, which enhanced attachment of EVs to the scaffold surface. The ECM-EV complexes were found to inhibit endothelial cell apoptosis as well as promote angiogenesis at the ischemic site [42]. Another study, where natural silk-fibroin hydrogels were loaded with stem cell-derived EVs enriched with miR-675 displayed a significant increase in blood perfusion over a period of 28 days when compared to the miR-675 EVs alone in an aging-induced vascular dysfunction murine model [91].
Being a major contributor of deaths worldwide, novel therapeutic options for cardiovascular defects have been extensively researched as these result in excessive heart muscle damage with minimal regenerative potential. The current therapies are mostly mechanical devices that assist the heart functioning however they do not aid in restoring and repairing the damaged heart tissues [92]. Polygycerol sebacate acrylate-based polymers combined with EVs were found to support active release of EVs over a period of 14 days in an in-vivo model of MI. This highlighted the potential of the hydrogel to protect the EVs from being lost during circulation. It was also observed that the polymer remained at the implanted site of the epicardium for over a month without eliciting a significant immune response thereby indicating its potential use for targeted local delivery of therapeutic molecules [93]. Another study compared the efficacy of small EVs administered through a sodium alginate hydrogel to EVs that were systemically injected into an MI murine model [94]. The sEVs incorporated in the hydrogel were found to have sustained release over a period of 14 days that resulted in enhanced angiogenesis at the infarcted site. When compared to the systemically administered sEVs the hydrogel group also drastically abated cardiac apoptosis and fibrosis having an eventual impact on restoring cardiac function [94].
4.4. Neuronal Regeneration
Nerve injuries are clinically challenging due to their limited regenerative potential. The conventional treatments mainly included autologous nerve grafts that are now being replaced by artificial nerve guidance conduits (NGCs) due to reduced availability of nerve graft, multiple surgeries for isolating donor graft and risk of developing neuromas. A recent study for the treatment of peripheral nerve injury focused on targeted therapeutics by using alginate hydrogels consisting of laminin- coated Poly(l-lactide-co-glycolide) (PLGA) conduit containing a combination of gold NPs, brain derived growth factor (BDNF) and adipose derived stem cells [69]. The complex system of the hydrogel, NPs, growth-factors and cells were found to significantly promote axonal regeneration and remyelination. They elicited a synergistic effect in a rat sciatic transection model [69]. Another study focused on the delivery of cell adhesive tetrapeptide modified conductive poly(3,4-ethylenedioxythiophene) nanoparticles (PEDOT NPs) from a biocompatible chitin scaffold. The conductive scaffold was found to be highly porous and biocompatible. When introduced in an in-vivo model improved nerve regeneration was observed based on thickness of the regenerated myelin and the area of the muscle fibers. Increased adhesive potential of the schwann cells as well as improved angiogenesis was also observed at the site of injury. Therefore, this electrically active chitin scaffold was suggested to not only be a potential substitute for a nerve guidance conduit but also a beneficial scaffold material for delivery of therapeutics for bone and muscle regeneration [70].
Limited regenerative capacity of spinal cord injuries demands novel therapeutic options. Hydrogels, with their increased water retaining capacity, not only provide the desired mechanical properties and support but are also promising drug delivery vehicles eliciting prolonged therapeutic effects. Mahya et al. [71] demonstrated the consistent delivery of berberine loaded chitosan NPs using a hybrid hydrogel of alginate and chitosan. Combining the gels with endometrial stem cells was also found to elicit a profound effect on the spinal cord regenerative capacity. These complex hydrogel and drug loaded NP systems not only abated the volume of the cavity but also resulted in reduced recruitment of inflammatory cells along with a decline in cell apoptosis and necrosis rates. These results highlighted the sustained release of the berberine loaded NPs resulted in increased neuroprotective effects [71].
4.5. Liver and Kidney Regeneration
As stem cell derived EVs are well known to promote healing and regeneration in multiple chronic diseases over time, the sustained release of EVs from hydrogels has been employed in various disease models to analyze the enhanced therapeutic effects of EVs due to increased bioavailability. Mardpour et al. [95], demonstrated the therapeutic effects of embryonic stem cell derived EV (ES-MSC-EVs) laden polyethylene glycol (PEG) macromeres when administered in a chronic liver injury (CLI) murine model. When systemically administered, these EVs were cleared from the system within 24 h; however, the EV loaded PEG macromeres ensured stable release of EVs for 4 weeks in vitro. The prolonged exposure of the fibrotic liver tissue to the hydrogel encapsulated EVs resulted in a drastic decline in fibrosis and apoptosis rates when compared to the systemically administered EVs [95]. MSC-EVs have also been found to alleviate renal injuries, although their application for kidney repair has been challenging due to rapid clearance from the body. However, sustained release of EVs enriched with miRNAs from RGD-Biotin hydrogel in an in-vivo model was found to not only enhance EV retention in the kidneys but also improved the antifibrotic and antiapoptotic effects at functional as well as molecular levels over a period of 7 days [60]. EVs encased in collagen hydrogels were also found to elicit similar effects in an acute kidney injury murine model by not only promoting proliferation and anti-apoptotic effects but also augmenting revascularization at the fibrotic site [96] (Table 2).

Table 2.
Extra cellular vehicles (EVs) delivered via biomaterial scaffolds for tissue repair in representative examples in various injury models.
5. Sustained Delivery of EVs/NPs for Cancer Therapy
Given the potential to load NPs with a variety of anti-cancer agents, and to modify the surface for tumor-targeted delivery, there is immense potential to employ NPs as therapeutics in the cancer setting. Many conventional systemic cancer therapies offer poor targeting and extensive side effects, which could be overcome with the use of NPs [99]. Polymeric conjugates and liposomes are FDA approved nanocarriers for targeted therapy. Although these NPs have huge potential in the cancer setting only few have made it to the market as there are considerable challenges in the release and uptake of these NPs, primarily the issue of accumulation in the liver and spleen when systemically administered in patients. Another limitation is the reduced retention of these particles in the tumor resulting in reduction in its efficacy [99].
Nature’s own nanocarriers, EVs, have also been highlighted as potent carriers of anti-cancer drugs or genetic material based on the unique characteristics of improving drug bioavailability, tumor-targeted delivery, reduced toxicity and protection of therapeutic genetic material/drugs by the lipid bilayer [100,101]. Although EV based therapeutics bear immense potential in the cancer setting, their clinical potential remains limited as a sustained delivery system of EVs is yet to be established [101]. While the number of studies employing hydrogels in the cancer setting is currently limited, this holds immense promise for tumor-targeted delivery of drugs/cargo, potentially enhancing drug availability and minimizing side effects of chemotherapeutic agents [60].
Thermosensitive hydrogels composed of hydroxypropyl cellulose, silk fibroin and glycerol loaded with gambogic acid carrying NPs were found to elicit enhanced therapeutic effects in a murine model of gastric cancer when compared to systemic delivery of the drugs [60]. The constant sustained release of the drug-loaded NPs at the site of the tumor resulted in increased ability to penetrate the gastric tumors and retain therapeutic drug dosages at the tumor site [60]. The increasing demand for controlled drug delivery systems has been mainly due to the limited bioavailability of only a few hours of drug loaded NPs when administered intravenously. The administration of a common chemotherapeutic drug, doxorubicin, when loaded on to micelles through a polymeric hydrogel was found to elicit enhanced anti-cancer effects for a period of 1 week when administered to breast cancer cells in vitro [102]. When treated with low concentrations of doxorubicin NPs a significant decline in the viability of the cancer cells was still noticed thereby highlighting the prolonged bioactivity of the hydrogel-released drug loaded micelles even at low concentrations [61]. Another study assessing the efficacy of siRNA loaded oligopeptide-modified poly(β-aminoester) NPs from polyamidoamine (PAMAM) dendrimer scaffold was found to elicit promising therapeutic effects in a breast cancer murine model [103]. Doxorubicin loaded polydopamine NPs were also shown to elicit tumor ameliorating effects when administered in a murine model over a period of 15days using a self-healing hydrogel [104]. The nanocomposite hydrogel being thermo-responsive was found to be very effective and easy to administer as the gel could be injected in a liquid sate via an intratumoral injection and, when exposed to the NIR laser, the hydrogel heated up, resulting in the release of the drug loaded NPs [104]. Hydrogels and NP/EV complexes could therefore be the way forward for enabling the targeted and sustained delivery of anti-cancer therapeutics.
6. Conclusions
Regardless of the disease setting, targeted therapeutics with increased bioavailability and persistence, and reduced toxicity to healthy tissue are a primary requirement. Natural and synthetic EVs and NPs have been explored as a therapeutic option for tissue regeneration and cancer, but clinical application has remained limited due to the lack of sustained delivery systems to ensure localized delivery of efficacious drug doses. Herein, we highlight the prospect of the sustained and prolonged delivery of these nano-sized therapeutic entities from hydrogels for broad applications spanning tissue regeneration and cancer treatment [10,20]. While synthetic NPs can be reproducibly manufactured, the clinical use of EVs was previously limited due to issues associated with the isolation and storage of large quantities of pure and consistent EVs. However, advances in technologies and guidelines that support reproducible EV production, isolation and characterization on a large scale have begun to ameliorate that challenge [17]. The use of bioreactors supports mass EV production through enhanced sheer stress or compression, or exposure to hypoxic conditions to increase EV release [105]. Encapsulation of EVs/NPs in biocompatible hydrogels will support more targeted, tunable delivery and reduce systemic loss of the therapeutic payload, with examples including the controlled release of EVs from scaffolds in response to alterations in pH [89,106].
The release kinetics of nano-sized particles is not only based on the characteristics of the NPs but also the mechanical properties, breakdown rate and stress distribution of the biomaterial. Therefore, optimization of these biomaterials in parallel with the NPs is critical [106]. An in vitro study assessing the controlled release of NPs from hydrogels highlighted the role of electric charge interactions in enabling sustained release of the particles. The superficial charges of NPs and hydrogel mean mesh size were exploited to control NP release [51]. As most of the studies involving the delivery of EVs that use hydrogels are still in the early stages, prolonged release rates are yet to be achieved. This will be a critical milestone in the clinical translation of these hydrogel-EV composites [20]. Although limited studies have been performed to date in the cancer setting, the hydrogel system could potentially support continual NP release for prolonged periods to target metastatic sites and reduce disease progression and/or recurrence. The use of biocompatible hydrogels combined with EVs derived from a compatible cell source (e.g., MSCs) will also limit any immunogenic response. In summary, hydrogels loaded with therapeutic NPs/EVs hold immense promise as cell-free sustained and targeted delivery systems in a range of disease settings. Further understanding of the parameters controlling the release rate of these particles and the efficient transfer of cargo to target cells will be fundamental to success.
Author Contributions
This work was conceived by Y.C. and R.M.D. The original draft preparation and writing: Y.C. and R.M.D. Review and editing: R.M.D., A.J.L. and G.P.D. All authors have read and agreed to the published version of the manuscript.
Funding
Y.C. is supported by Science Foundation Ireland and the Engineering and Physical Sciences Research Council Centre for Doctoral Training in Engineered Tissues for Discovery, Industry and Medicine (Grant numbers 18/EPSRC-CDT/3583 and EP/S02347X/1). This publication has emanated from research supported in part by a grant from Science Foundation Ireland (SFI) and the European Regional Development Fund (ERDF) under grant number 13/RC/2073_2.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Davis, M.E.; Chen, Z.; Shin, D.M. Nanoparticle therapeutics: An emerging treatment modality for cancer. Nat. Rev. Drug Discov. 2008, 7, 771–782. [Google Scholar] [CrossRef] [PubMed]
- Hamidi, M.; Azadi, A.; Rafiei, P. Hydrogel nanoparticles in drug delivery. Adv. Drug Deliv. Rev. 2008, 60, 1638–1649. [Google Scholar] [CrossRef] [PubMed]
- Lim, Y.H.; Tiemann, K.M.; Hunstad, D.A.; Elsabahy, M.; Wooley, K.L. Polymeric nanoparticles in development for treatment of pulmonary infectious diseases: Nanoparticles in treatment of pulmonary infectious diseases. Wiley Interdiscip. Rev. Nanomed. Nanobiotechnol. 2016, 8, 842–871. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jin, G.-Z.; Chakraborty, A.; Lee, J.-H.; Knowles, J.C.; Kim, H.-W. Targeting with nanoparticles for the therapeutic treatment of brain diseases. J. Tissue Eng. 2020, 11, 204173141989746. [Google Scholar] [CrossRef] [Green Version]
- Zolnik, B.S.; González-Fernández, Á.; Sadrieh, N.; Dobrovolskaia, M.A. Minireview: Nanoparticles and the immune system. Endocrinology 2010, 151, 458–465. [Google Scholar] [CrossRef]
- Dobrovolskaia, M.A.; Shurin, M.; Shvedova, A.A. Current understanding of interactions between nanoparticles and the immune system. Toxicol. Appl. Pharmacol. 2016, 299, 78–89. [Google Scholar] [CrossRef] [Green Version]
- Zhou, X.; He, X.; Shi, K.; Yuan, L.; Yang, Y.; Liu, Q.; Ming, Y.; Yi, C.; Qian, Z. Injectable thermosensitive hydrogel containing erlotinib-loaded hollow mesoporous silica nanoparticles as a localized drug delivery system for NSCLC therapy. Adv. Sci. 2020, 7, 2001442. [Google Scholar] [CrossRef]
- Peng, Q.; Sun, X.; Gong, T.; Wu, C.-Y.; Zhang, T.; Tan, J.; Zhang, Z.-R. Injectable and biodegradable thermosensitive hydrogels loaded with PHBHHx nanoparticles for the sustained and controlled release of insulin. Acta Biomater. 2013, 9, 5063–5069. [Google Scholar] [CrossRef]
- Allen, T.M.; Cullis, P.R. Liposomal drug delivery systems: From concept to clinical applications. Adv. Drug Deliv. Rev. 2013, 65, 36–48. [Google Scholar] [CrossRef]
- Alavi, M. Application of various types of liposomes in drug delivery systems. Adv. Pharm. Bull. 2017, 7, 3–9. [Google Scholar] [CrossRef]
- Wang-Gillam, A.; Li, C.-P.; Bodoky, G.; Dean, A.; Shan, Y.-S.; Jameson, G.; Macarulla, T.; Lee, K.-H.; Cunningham, D.; Blanc, J.F.; et al. Nanoliposomal irinotecan with fluorouracil and folinic acid in metastatic pancreatic cancer after previous gemcitabine-based therapy (NAPOLI-1): A global, randomised, open-label, phase 3 trial. Lancet Lond. Engl. 2016, 387, 545–557. [Google Scholar] [CrossRef]
- Bobo, D.; Robinson, K.J.; Islam, J.; Thurecht, K.J.; Corrie, S.R. Nanoparticle-based medicines: A review of FDA-approved materials and clinical trials to date. Pharm. Res. 2016, 33, 2373–2387. [Google Scholar] [CrossRef] [PubMed]
- Gabizon, A.; Catane, R.; Uziely, B.; Kaufman, B.; Safra, T.; Cohen, R.; Martin, F.; Huang, A.; Barenholz, Y. Prolonged circulation time and enhanced accumulation in malignant exudates of doxorubicin encapsulated in polyethylene-glycol coated liposomes. Cancer Res. 1994, 54, 987–992. [Google Scholar] [PubMed]
- Hann, I.M.; Prentice, H.G. Lipid-based amphotericin B: A review of the last 10 years of use. Int. J. Antimicrob. Agents 2001, 17, 161–169. [Google Scholar] [CrossRef]
- Huang, H.; Feng, W.; Chen, Y.; Shi, J. Inorganic nanoparticles in clinical trials and translations. Nano Today 2020, 35, 100972. [Google Scholar] [CrossRef]
- Vissers, C.; Ming, G.; Song, H. Nanoparticle technology and stem cell therapy team up against neurodegenerative disorders. Adv. Drug Deliv. Rev. 2019, 148, 239–251. [Google Scholar] [CrossRef]
- Théry, C.; Witwer, K.W.; Aikawa, E.; Alcaraz, M.J.; Anderson, J.D.; Andriantsitohaina, R.; Antoniou, A.; Arab, T.; Archer, F.; Atkin-Smith, G.K.; et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): A position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 2018, 7, 1535750. [Google Scholar] [CrossRef] [Green Version]
- O’Neill, C.P.; Gilligan, K.E.; Dwyer, R.M. Role of extracellular vesicles (EVs) in cell stress response and resistance to cancer therapy. Cancers 2019, 14, 136. [Google Scholar] [CrossRef] [Green Version]
- Konoshenko MYu Lekchnov, E.A.; Vlassov, A.V.; Laktionov, P.P. Isolation of extracellular vesicles: General methodologies and latest trends. BioMed Res. Int. 2018, 2018, 8545347. [Google Scholar] [CrossRef]
- Riau, A.K.; Ong, H.S.; Yam, G.H.F.; Mehta, J.S. Sustained delivery system for stem cell-derived exosomes. Front. Pharmacol. 2019, 10, 1368. [Google Scholar] [CrossRef]
- Bunggulawa, E.J.; Wang, W.; Yin, T.; Wang, N.; Durkan, C.; Wang, Y.; Durkan, C.; Wang, Y.; Yang, G. Recent advancements in the use of exosomes as drug delivery systems. J. Nanobiotechnol. 2018, 16, 81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mendt, M.; Rezvani, K.; Shpall, E. Mesenchymal stem cell-derived exosomes for clinical use. Bone Marrow Transpl. 2019, 54, 789–792. [Google Scholar] [CrossRef] [PubMed]
- Klyachko, N.L.; Arzt, C.J.; Li, S.M.; Gololobova, O.A.; Batrakova, E.V. Extracellular vesicle-based therapeutics: Preclinical and clinical investigations. Pharmaceutics 2020, 12, 1171. [Google Scholar] [CrossRef]
- Vozel, D. Efficacy of Platelet- and Extracellular Vesicle-Rich Plasma for the Treatment of Chronically Inflamed Post-Surgical Temporal Bone Cavities: A Randomised Controlled Clinical Study, Report No.: Study/NCT04281901. November 2020. Available online: https://clinicaltrials.gov/ct2/show/study/NCT04281901 (accessed on 1 July 2020).
- Steiner, N. Use of Autologous Plasma Rich in Platelets and Extracellular Vesicles in the Surgical Treatment of Chronic Otitis Media. Report No.: NCT04761562. February 2021. Available online: https://clinicaltrials.gov/ct2/show/NCT0476156 (accessed on 1 July 2021).
- Dai, S.; Wei, D.; Wu, Z.; Zhou, X.; Wei, X.; Huang, H.; Li, G. Phase I clinical trial of autologous ascites-derived exosomes combined with GM-CSF for colorectal cancer. Mol. Ther. 2008, 16, 782–790. [Google Scholar] [CrossRef] [PubMed]
- Anderson Cancer Center. Phase I Study of Mesenchymal Stromal Cells-Derived Exosomes with KrasG12D siRNA for Metastatic Pancreas Cancer Patients Harboring KrasG12D Mutation, Report No.: NCT03608631. April 2021. Available online: https://clinicaltrials.gov/ct2/show/NCT03608631 (accessed on 19 May 2021).
- Ruijin Hospital. A Pilot Clinical Study on Aerosol Inhalation of the Exosomes Derived from Allogenic Adipose Mesenchymal Stem Cells in the Treatment of Severe Patients with Novel Coronavirus Pneumonia, Report No.: Results/NCT04276987. September 2020. Available online: https://clinicaltrials.gov/ct2/show/results/NCT04276987 (accessed on 19 May 2021).
- Athens Medical Society. A Phase II Randomized, Single-Blind Dose Study to Evaluate the Safety and Efficacy of Exosomes Overexpressing CD24 in 10^9 Dose versus 10^10 Dose, for the Prevention of Clinical Deterioration in Patients with Moderate or Severe COVID-19. Report No.: NCT04902183. June 2021. Available online: https://clinicaltrials.gov/ct2/show/NCT04902183 (accessed on 6 July 2021).
- Wilhelm, S.; Tavares, A.J.; Dai, Q.; Ohta, S.; Audet, J.; Dvorak, H.F.; Chan, W.C.W. Analysis of nanoparticle delivery to tumours. Nat. Rev. Mater. 2016, 1, 16014. [Google Scholar] [CrossRef]
- Takahashi, Y.; Nishikawa, M.; Shinotsuka, H.; Matsui, Y.; Ohara, S.; Imai, T.; Takakura, Y. Visualization and in vivo tracking of the exosomes of murine melanoma B16-BL6 cells in mice after intravenous injection. J. Biotechnol. 2013, 165, 77–84. [Google Scholar] [CrossRef]
- Smyth, T.; Kullberg, M.; Malik, N.; Smith-Jones, P.; Graner, M.W.; Anchordoquy, T.J. Biodistribution and delivery efficiency of unmodified tumor-derived exosomes. J. Control. Release 2015, 199, 145–551. [Google Scholar] [CrossRef] [Green Version]
- Wiklander, O.P.B.; Nordin, J.Z.; O’Loughlin, A.; Gustafsson, Y.; Corso, G.; Mäger, I.; Vader, P.; Lee, Y.; Sork, H.; Seow, Y.; et al. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J. Extracell. Vesicles 2015, 4, 26316. [Google Scholar] [CrossRef] [Green Version]
- Kamerkar, S.; LeBleu, V.S.; Sugimoto, H.; Yang, S.; Ruivo, C.F.; Melo, S.A.; Lee, J.J.; Kalluri, R. Exosomes facilitate therapeutic targeting of oncogenic kras in pancreatic cancer. Nature 2017, 546, 498–503. [Google Scholar] [CrossRef]
- Liu, X.; Yang, Y.; Li, Y.; Niu, X.; Zhao, B.; Wang, Y.; Bao, C.; Xie, Z.; Lin, Q.; Zhu, L. Integration of stem cell-derived exosomes with in situ hydrogel glue as a promising tissue patch for articular cartilage regeneration. Nanoscale 2017, 9, 4430–4438. [Google Scholar] [CrossRef]
- Oliva, N.; Conde, J.; Wang, K.; Artzi, N. Designing hydrogels for on-demand therapy. ACC Chem. Res. 2017, 50, 669–679. [Google Scholar] [CrossRef] [PubMed]
- Merino, S.; Martín, C.; Kostarelos, K.; Prato, M.; Vázquez, E. Nanocomposite hydrogels: 3D polymer–nanoparticle synergies for on-demand drug delivery. ACS Nano 2015, 9, 4686–4697. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gao, W.; Zhang, Y.; Zhang, Q.; Zhang, L. Nanoparticle-hydrogel: A hybrid biomaterial system for localized drug delivery. Ann. Biomed. Eng. 2016, 44, 2049–2061. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Y.; Zhang, J.; Chen, M.; Gong, H.; Thamphiwatana, S.; Eckmann, L.; Gao, W.; Zhang, L. A bioadhesive nanoparticle–hydrogel hybrid system for localized antimicrobial drug delivery. ACS Appl. Mater. Interfaces 2016, 8, 18367–18374. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shi, Q.; Qian, Z.; Liu, D.; Sun, J.; Wang, X.; Liu, H.; Xu, J.; Guo, X. GMSC-derived exosomes combined with a chitosan/silk hydrogel sponge accelerates wound healing in a diabetic rat skin defect model. Front. Physiol. 2017, 8, 904. [Google Scholar] [CrossRef] [PubMed]
- Diomede, F. Three-dimensional printed PLA scaffold and human gingival stem cell-derived extracellular vesicles: A new tool for bone defect repair. Stem Cell Res. Ther. 2018, 21, 104. [Google Scholar] [CrossRef] [Green Version]
- Hao, D.; Swindell, H.S.; Ramasubramanian, L.; Liu, R.; Lam, K.S.; Farmer, D.L.; Wang, A. Extracellular matrix mimicking nanofibrous scaffolds modified with mesenchymal stem cell-derived extracellular vesicles for improved vascularization. Front. Bioeng. Biotechnol. 2020, 8, 633. [Google Scholar] [CrossRef] [PubMed]
- Hoare, T.R.; Kohane, D.S. Hydrogels in drug delivery: Progress and challenges. Polymer 2008, 49, 1993–2007. [Google Scholar] [CrossRef] [Green Version]
- Slaughter, B.V.; Khurshid, S.S.; Fisher, O.Z.; Khademhosseini, A.; Peppas, N.A. Hydrogels in regenerative medicine. Adv. Mater. 2009, 21, 3307–3329. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, C.-C.; Metters, A.T. Hydrogels in controlled release formulations: Network design and mathematical modeling. Adv. Drug Deliv. Rev. 2006, 58, 1379–1408. [Google Scholar] [CrossRef]
- Hoffman, A.S. Hydrogels for biomedical applications. Adv. Drug Deliv. Rev. 2012, 6, 18–23. [Google Scholar] [CrossRef]
- Dong, Y.A.S.; Rodrigues, M.; Li, X.; Kwon, S.H.; Kosaric, N.; Khong, S.; Gao, Y.; Wang, W.; Gurtner, G.C. Injectable and tunable gelatin hydrogels enhance stem cell retention and improve cutaneous wound healing. Adv. Funct. Mater. 2017, 27, 1606619. [Google Scholar] [CrossRef]
- Hsu, F.Y.; Tsai, S.W.; Wang, F.F.; Wang, Y.J. The collagen-containing alginate/poly(l-lysine)/alginate microcapsules. Artif. Cells Blood Substit. Biotechnol. 2000, 28, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Liu, V.A.; Bhatia, S.N. Three-dimensional photopatterning of hydrogels containing living cells. Biomed. Microdev. 2002, 10, 257–266. [Google Scholar] [CrossRef]
- Dannert, C.; Stokke, B.T.; Dias, R.S. Nanoparticle-hydrogel composites: From molecular interactions to macroscopic behavior. Polymers 2019, 11, 275. [Google Scholar] [CrossRef] [Green Version]
- Mauri, E.; Negri, A.; Rebellato, E.; Masi, M.; Perale, G.; Rossi, F. Hydrogel-nanoparticles composite system for controlled drug delivery. Gels 2018, 4, 74. [Google Scholar] [CrossRef] [Green Version]
- Rehmann, M.S.; Skeens, K.M.; Kharkar, P.M.; Ford, E.M.; Maverakis, E.; Lee, K.H.; Kloxin, A.M. Tuning and predicting mesh size and protein release from step growth hydrogels. Biomacromolecules 2017, 18, 3131–3142. [Google Scholar] [CrossRef]
- Wang, C.; Flynn, N.T.; Langer, R. Controlled structure and properties of thermoresponsive nanoparticle–hydrogel composites. Adv. Mater. 2004, 16, 1074–1079. [Google Scholar] [CrossRef]
- Bronstein, L.M.; Platonova, O.A.; Yakunin, A.N.; Yanovskaya, I.M.; Valetsky, P.M.; Dembo, A.T.; Makhaeva, E.E.; Mironov, A.V.; Khokhlov, A.R. Complexes of polyelectrolyte gels with oppositely charged surfactants: Interaction with metal ions and metal nanoparticle formation. Langmuir 1998, 14, 252–259. [Google Scholar] [CrossRef]
- Murali Mohan, Y.; Vimala, K.; Thomas, V.; Varaprasad, K.; Sreedhar, B.; Bajpai, S.K.; Raju, K.M. Controlling of silver nanoparticles structure by hydrogel networks. J. Colloid. Interface Sci. 2010, 342, 73–82. [Google Scholar] [CrossRef]
- Siegel, R.A.; Gu, Y.; Lei, M.; Baldi, A.; Nuxoll, E.E.; Ziaie, B. Hard and soft micro- and nanofabrication: An integrated approach to hydrogel-based biosensing and drug delivery. J. Control. Release 2010, 141, 303–313. [Google Scholar] [CrossRef] [Green Version]
- Mandal, A.; Clegg, J.R.; Anselmo, A.C.; Mitragotri, S. Hydrogels in the clinic. Bioeng. Transl. Med. 2020, 5, e10158. [Google Scholar] [CrossRef] [Green Version]
- Tonbul, M.; Adas, M.; Bekmezci, T.; Kara, A.D. Intra-articular polyacrylamide hydrogel injections are not innocent. Case Rep. Orthop. 2014, 2014, e150709. [Google Scholar] [CrossRef] [PubMed]
- O’Dwyer, J. Development of a nanomedicine-loaded hydrogel for sustained delivery of an angiogenic growth factor to the ischaemic myocardium. Drug Deliv. Transl. Res. 2020, 10, 440–454. [Google Scholar] [CrossRef] [PubMed]
- Zhang, D.; Chu, Y.; Qian, H.; Qian, L.; Shao, J.; Xu, Q.; Li, R.; Zhang, Q.; Wu, F.; Liu, B.; et al. Antitumor activity of thermosensitive hydrogels packaging gambogic acid nanoparticles and tumor-penetrating peptide iRGD against gastric cancer. Int. J. Nanomed. 2020, 15, 735–747. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ono, R.J.; Lee, A.L.Z.; Voo, Z.X.; Venkataraman, S.; Koh, B.W.; Yang, Y.Y.; Hedrick, J.L. Biodegradable strain-promoted click hydrogels for encapsulation of drug-loaded nanoparticles and sustained release of therapeutics. Biomacromolecules 2017, 18, 2277–2285. [Google Scholar] [CrossRef]
- Zhou, L.; Chen, F.; Hou, Z.; Chen, Y.; Luo, X. Injectable self-healing CuS nanoparticle complex hydrogels with antibacterial, anti-cancer, and wound healing properties. Chem. Eng. J. 2021, 409, 128224. [Google Scholar] [CrossRef]
- Wang, C.; Zhao, N.; Yuan, W. NIR/thermoresponsive injectable self-healing hydrogels containing polydopamine nanoparticles for efficient synergistic cancer thermochemotherapy. ACS Appl. Mater. Interfaces 2020, 12, 9118–9131. [Google Scholar] [CrossRef]
- Diniz, F.R.; Maia, R.C.A.P.; Rannier Andrade, L.; Andrade, L.N.; Vinicius Chaud, M.; da Silva, C.F.; Correa, C.B.; de Albuquerque, R.L.C.; da Costa, L.P.; Shin, S.R.; et al. Silver nanoparticles-composing alginate/gelatine hydrogel improves wound healing in vivo. Nanomaterials 2020, 10, 390. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haidari, H.; Kopecki, Z.; Sutton, A.T.; Garg, S.; Cowin, A.J.; Vasilev, K. pH-responsive “Smart” hydrogel for controlled delivery of silver nanoparticles to infected wounds. Antibiotics 2021, 10, 49. [Google Scholar] [CrossRef]
- Khorasani, M.T.; Joorabloo, A.; Adeli, H.; Milan, P.B.; Amoupour, M. Enhanced antimicrobial and full-thickness wound healing efficiency of hydrogels loaded with heparinized ZnO nanoparticles: In vitro and in vivo evaluation. Int. J. Biol. Macromol. 2021, 166, 200–212. [Google Scholar] [CrossRef]
- Mi, L.; Liu, H.; Gao, Y.; Miao, H.; Ruan, J. Injectable nanoparticles/hydrogels composite as sustained release system with stromal cell-derived factor-1α for calvarial bone regeneration. Int. J. Biol. Macromol. 2017, 101, 341–347. [Google Scholar] [CrossRef]
- Li, Y.; Liu, Y.; Guo, Q. Silk fibroin hydrogel scaffolds incorporated with chitosan nanoparticles repair articular cartilage defects by regulating TGF-β1 and BMP-2. Arthritis Res. Ther. 2021, 23, 50. [Google Scholar] [CrossRef]
- Jahromi, M.; Razavi, S.; Seyedebrahimi, R.; Reisi, P.; Kazemi, M. Regeneration of rat sciatic nerve using PLGA conduit containing rat ADSCs with controlled release of BDNF and gold nanoparticles. J. Mol. Neurosci. 2021, 71, 746–760. [Google Scholar] [CrossRef]
- Huang, L.; Yang, X.; Deng, L.; Ying, D.; Lu, A.; Zhang, L.; Yu, A.; Duan, B. Biocompatible chitin hydrogel incorporated with PEDOT nanoparticles for peripheral nerve repair. ACS Appl. Mater. Interfaces 2021, 13, 16106–16117. [Google Scholar] [CrossRef] [PubMed]
- Mahya, S.; Ai, J.; Shojae, S.; Khonakdar, H.A.; Darbemamieh, G.; Shirian, S. Berberine loaded chitosan nanoparticles encapsulated in polysaccharide-based hydrogel for the repair of spinal cord. Int. J. Biol. Macromol. 2021, 182, 82–90. [Google Scholar] [CrossRef] [PubMed]
- van Rijt, S.; Habibovic, P. Enhancing regenerative approaches with nanoparticles. J. R. Soc. Interface 2017, 14, 20170093. [Google Scholar] [CrossRef] [PubMed]
- Saleh, B.; Dhaliwal, H.K.; Portillo-Lara, R.; Sani, E.S.; Abdi, R.; Amiji, M.M.; Annabi, N. Local immunomodulation using an adhesive hydrogel loaded with miRNA-laden nanoparticles promotes wound healing. Small 2019, 15, 1902232. [Google Scholar] [CrossRef]
- Tan, H.-L.; Teow, S.-Y.; Pushpamalar, J. Application of metal nanoparticle–hydrogel composites in tissue regeneration. Bioengineering 2019, 6, 17. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xie, M.; Wu, D.; Li, G.; Yang, J.; Zhang, Y.S. Exosomes targeted towards applications in regenerative medicine. Nano Sel. 2021, 2, 880–908. [Google Scholar] [CrossRef]
- Ramírez, O.J.; Alvarez, S.; Contreras-Kallens, P.; Aguayo, S. Type I collagen hydrogels as a delivery matrix for royal jelly derived extracellular vesicles. Drug Deliv. 2020, 27, 12. [Google Scholar] [CrossRef]
- Miguel, S.P. Thermoresponsive chitosan–agarose hydrogel for skin regeneration. Carbohydr. Polym. 2014, 8, 366–373. [Google Scholar] [CrossRef]
- Chouhan, D.; Lohe, T.; Samudrala, P.K.; Mandal, B.B. In situ forming injectable silk fibroin hydrogel promotes skin regeneration in full thickness burn wounds. Adv. Healthc. Mater. 2018, 7, 1801092. [Google Scholar] [CrossRef]
- Mahmoud, N.N.; Hikmat, S.; Abu Ghith, D.; Hajeer, M.; Hamadneh, L.; Qattan, D.; Khalil, E.A. Gold nanoparticles loaded into polymeric hydrogel for wound healing in rats: Effect of nanoparticles’ shape and surface modification. Int. J. Pharm. 2019, 565, 174–186. [Google Scholar] [CrossRef] [PubMed]
- Su, N.; Hao, Y.; Wang, F.; Hou, W.; Chen, H.; Luo, Y. Mesenchymal stromal exosome–functionalized scaffolds induce innate and adaptive immunomodulatory responses toward tissue repair. Sci. Adv. 2021, 7, eabf7207. [Google Scholar] [CrossRef]
- Fang, S.; Xu, C.; Zhang, Y.; Xue, C.; Yang, C.; Bi, H.; Qian, X.; Wu, M.; Ji, K.; Zhao, Y.; et al. Umbilical cord-derived mesenchymal stem cell-derived exosomal MicroRNAs suppress myofibroblast differentiation by inhibiting the transforming growth factor-β/SMAD2 pathway during wound healing: uMSC exosomes suppress scar formation. Stem Cells Transl. Med. 2016, 5, 1425–1439. [Google Scholar] [CrossRef] [PubMed]
- Shafei, S.; Khanmohammadi, M.; Heidari, R.; Ghanbari, H.; Taghdiri Nooshabadi, V.; Farzamfar, S.; Akbariqomi, M.; Sanikhani, N.S.; Absalam, M.; Tavoosidana, G. Exosome loaded alginate hydrogel promotes tissue regeneration in full-thickness skin wounds: An in vivo study. J. Biomed. Mater. Res. A 2020, 108, 545–556. [Google Scholar] [CrossRef]
- Zhao, Y.; Cui, Z.; Liu, B.; Xiang, J.; Qiu, D.; Tian, Y.; Qu, X.; Yang, Z. An injectable strong hydrogel for bone reconstruction. Adv. Healthc. Mater. 2019, 8, 1900709. [Google Scholar] [CrossRef] [PubMed]
- Raucci, M.G.; Demitri, C.; Soriente, A.; Fasolino, I.; Sannino, A.; Ambrosio, L. Gelatin/nano-hydroxyapatite hydrogel scaffold prepared by sol-gel technology as filler to repair bone defects. J. Biomed. Mater. Res. A 2018, 14, 2007–2019. [Google Scholar] [CrossRef]
- Rezk, A.I.; Kim, K.-S.; Kim, C.S. Poly(ε-caprolactone)/poly(glycerol sebacate) composite nanofibers incorporating hydroxyapatite nanoparticles and simvastatin for bone tissue regeneration and drug delivery applications. Polymers 2020, 12, 2667. [Google Scholar] [CrossRef]
- Choi, B.; Kim, S.; Fan, J.; Kowalski, T.; Petrigliano, F.; Evseenko, D.; Lee, M. Covalently conjugated transforming growth factor-β1 in modular chitosan hydrogels for the effective treatment of articular cartilage defects. Biomater. Sci. 2015, 3, 742–752. [Google Scholar] [CrossRef] [PubMed]
- Lin, W.; Kluzek, M.; Iuster, N.; Shimoni, E.; Kampf, N.; Goldberg, R.; Klein, J. Cartilage-inspired, lipid-based boundary-lubricated hydrogels. Science 2020, 370, 335–338. [Google Scholar] [CrossRef] [PubMed]
- Diomede, F.; D’Aurora, M.; Gugliandolo, A.; Merciaro, I.; Ettorre, V.; Bramanti, A.; Piatelli, A.; Gatta, V.; Mazzon, E.; Fontana, A.; et al. A novel role in skeletal segment regeneration of extracellular vesicles released from periodontal-ligament stem cells. Int. J. Nanomed. 2018, 13, 3805–3825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, L.; Wang, J.; Zhou, X.; Sun, J.; Zhu, B.; Duan, C.; Chen, P.; Guo, X.; Zhang, T.; Guo, H. A new self-healing hydrogel containing hucMSC-derived exosomes promotes bone regeneration. Front. Bioeng. Biotechnol. 2020, 8, 564731. [Google Scholar] [CrossRef] [PubMed]
- Hu, H. miR-23a-3p-abundant small extracellular vesicles released from Gelma/nanoclay hydrogel for cartilage. J. Extracell. Vesicles 2020, 9, 19. [Google Scholar] [CrossRef]
- Han, C. Delivery of miR-675 by stem cell-derived exosomes encapsulated in silk fibroin hydrogel prevents aging-induced vascular dysfunction in mouse hindlimb. Mater. Sci. 2019, 11, 322–332. [Google Scholar] [CrossRef] [PubMed]
- Riaud, M.; Martinez, M.C.; Montero-Menei, C.N. Scaffolds and extracellular vesicles as a promising approach for cardiac regeneration after myocardial infarction. Pharmaceutics 2020, 12, 1195. [Google Scholar] [CrossRef]
- Hamada, T.; Dubois, J.L.N.; Bellamy, V.; Pidial, L.; Hagège, A.; Pereira, M.N.; Menasche, P. In vitro controlled release of extracellular vesicles for cardiac repair from poly(glycerol sebacate) acrylate-based polymers. Acta Biomater. 2020, 115, 92–103. [Google Scholar] [CrossRef]
- Lv, K.; Li, Q.; Zhang, L.; Wang, Y.; Zhong, Z.; Zhao, J.; Lin, X.; Wang, J.; Zhu, K.; Xiao, C.; et al. Incorporation of small extracellular vesicles in sodium alginate hydrogel as a novel therapeutic strategy for myocardial infarction. Theranostics 2019, 9, 7403–7416. [Google Scholar] [CrossRef]
- Mardpour, S.; Ghanian, M.H.; Sadeghi-Abandansari, H.; Mardpour, S.; Nazari, A.; Shekari, F.; Baharvand, H. Hydrogel-mediated sustained systemic delivery of mesenchymal stem cell-derived extracellular vesicles improves hepatic regeneration in chronic liver failure. ACS Appl. Mater. Interfaces 2019, 11, 37421–37433. [Google Scholar] [CrossRef]
- Liu, Y.; Cui, J.; Wang, H.; Hezam, K.; Zhao, X.; Huang, H.; Chen, S.; Han, Z.; Han, Z.C.; Guo, Z.; et al. Enhanced therapeutic effects of MSC-derived extracellular vesicles with an injectable collagen matrix for experimental acute kidney injury treatment. Stem Cell Res. Ther. 2020, 11, 161. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, C.; Shang, Y.; Chen, X.; Midgley, A.C.; Wang, Z.; Zhu, D.; Wu, J.; Chen, P.; Wu, L.; Wang, X.; et al. Supramolecular nanofibers containing arginine-glycine-aspartate (RGD) peptides boost therapeutic efficacy of extracellular vesicles in kidney repair. ACS Nano 2020, 14, 12133–12147. [Google Scholar] [CrossRef] [PubMed]
- Xin, L.; Lin, X.; Zhou, F.; Li, C.; Wang, X.; Yu, H.; Pan, Y.; Fei, H.; Ma, L.; Zhang, S. A scaffold laden with mesenchymal stem cell-derived exosomes for promoting endometrium regeneration and fertility restoration through macrophage immunomodulation. Acta Biomater. 2020, 113, 252–266. [Google Scholar] [CrossRef]
- Pérez-Herrero, E.; Fernández-Medarde, A. Advanced targeted therapies in cancer: Drug nanocarriers, the future of chemotherapy. Eur. J. Pharm. Biopharm. 2015, 93, 52–79. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Zhang, Y.; Cai, G.; Li, Q. Exosomes as actively targeted nanocarriers for cancer therapy. Int. J. Nanomed. 2020, 15, 4257–4273. [Google Scholar] [CrossRef]
- Xu, Z.; Zeng, S.; Gong, Z.; Yan, Y. Exosome-based immunotherapy: A promising approach for cancer treatment. Mol. Cancer 2020, 19, 160. [Google Scholar] [CrossRef] [PubMed]
- Senapati, S.; Mahanta, A.K.; Kumar, S.; Maiti, P. Controlled drug delivery vehicles for cancer treatment and their performance. Signal. Transduct. Target Ther. 2018, 3, 7. [Google Scholar] [CrossRef] [Green Version]
- Segovia, N.; Pont, M.; Oliva, N.; Ramos, V.; Borrós, S.; Artzi, N. Hydrogel doped with nanoparticles for local sustained release of siRNA in breast cancer. Adv. Healthc. Mater. 2015, 4, 271–280. [Google Scholar] [CrossRef]
- Tang, W.; Liu, B.; Wang, S.; Liu, T.; Fu, C.; Ren, X.; Tan, L.; Duan, W.; Meng, X. Doxorubicin-loaded ionic liquid–polydopamine nanoparticles for combined chemotherapy and microwave thermal therapy of cancer. RSC Adv. 2016, 6, 32434–32440. [Google Scholar] [CrossRef]
- Maumus, M.; Rozier, P.; Boulestreau, J.; Jorgensen, C.; Noël, D. Mesenchymal stem cell-derived extracellular vesicles: Opportunities and challenges for clinical translation. Front. Bioeng. Biotechnol. 2020, 8, 997. [Google Scholar] [CrossRef]
- Yan, H.C.; Yu, T.T.; Li, J.; Qiao, Y.Q.; Wang, L.C.; Zhang, T.; Li, Q.; Zhou, Y.H.; Liu, D.W. The delivery of extracellular vesicles loaded in biomaterial scaffolds for bone regeneration. Front. Bioeng. Biotechnol. 2020, 8, 1015. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).