Repurposing Antipsychotics for Cancer Treatment
Abstract
1. Introduction
2. Typical Antipsychotics
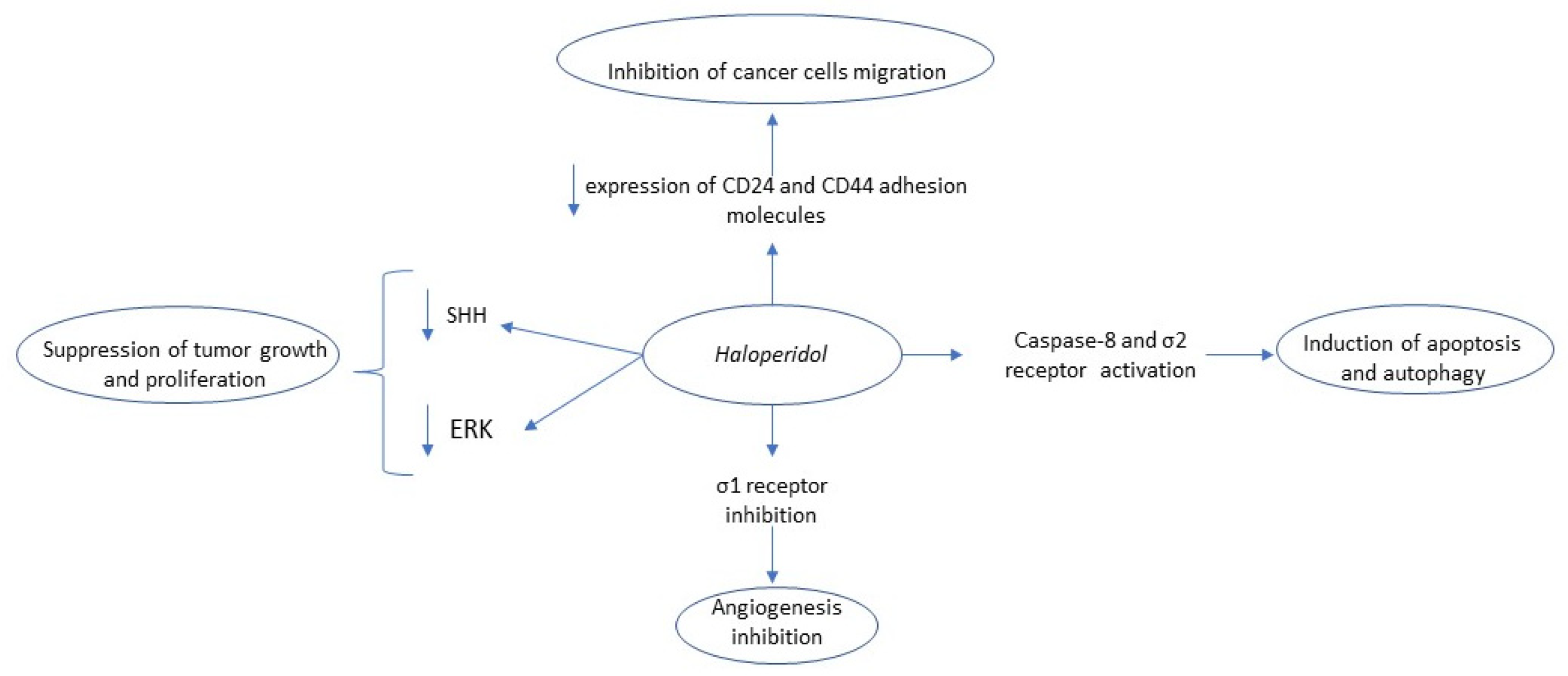
2.1. Haloperidol
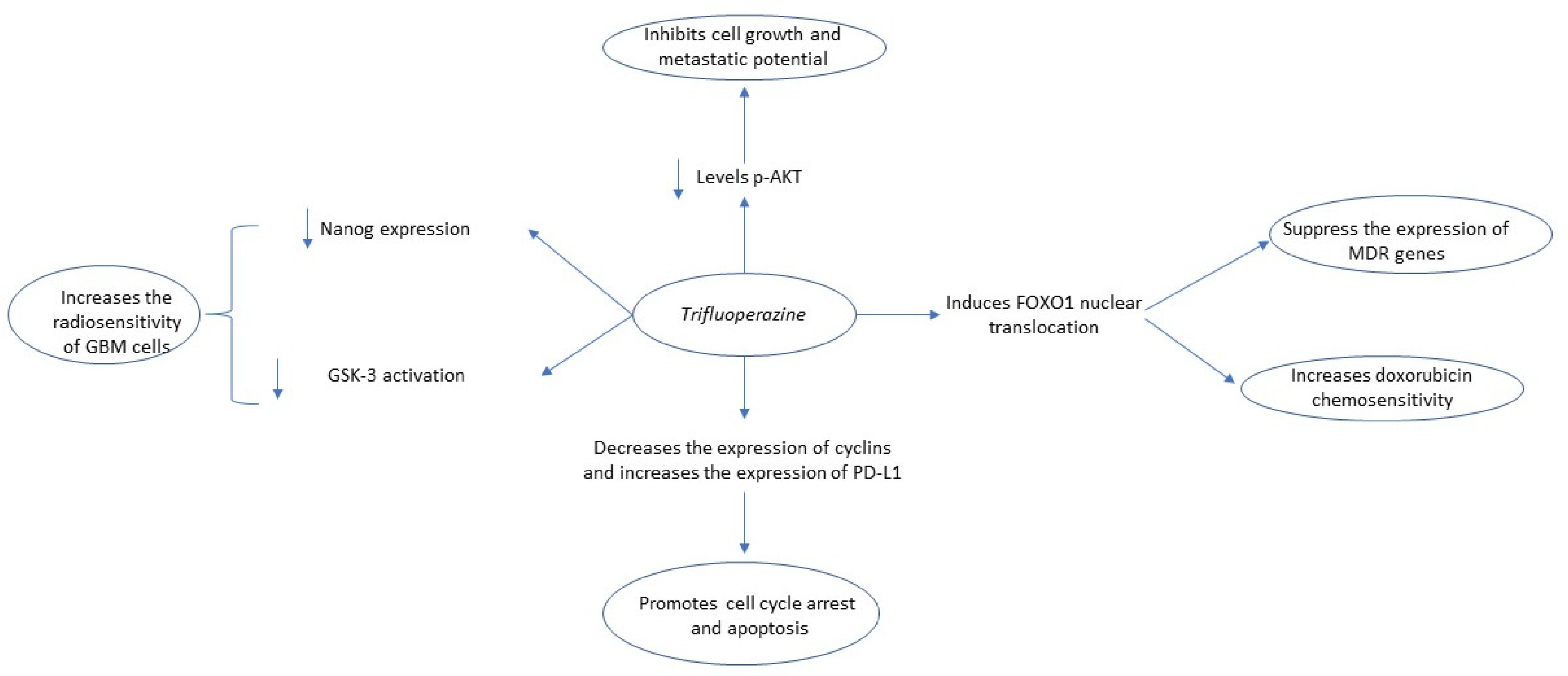
2.2. Trifluoperazine
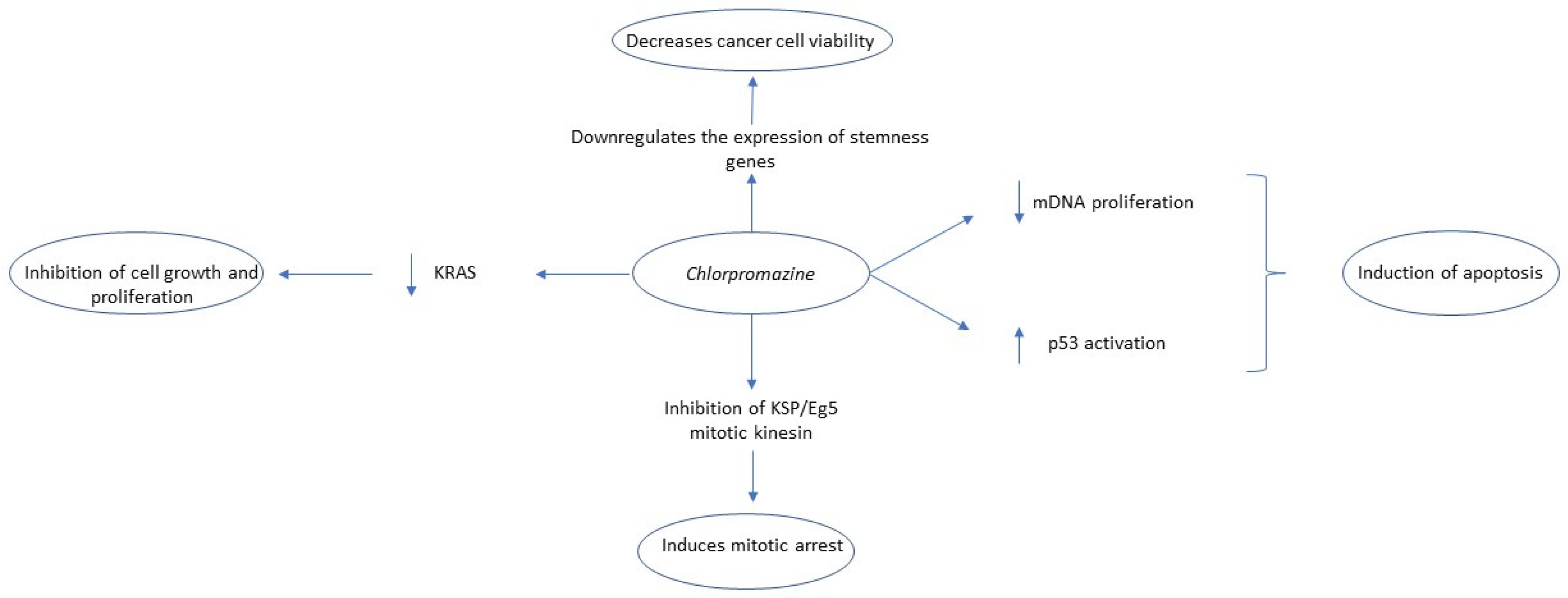
2.3. Chlorpromazine
2.4. Pimozide
2.5. Fluspirilene
2.6. Penfluridol
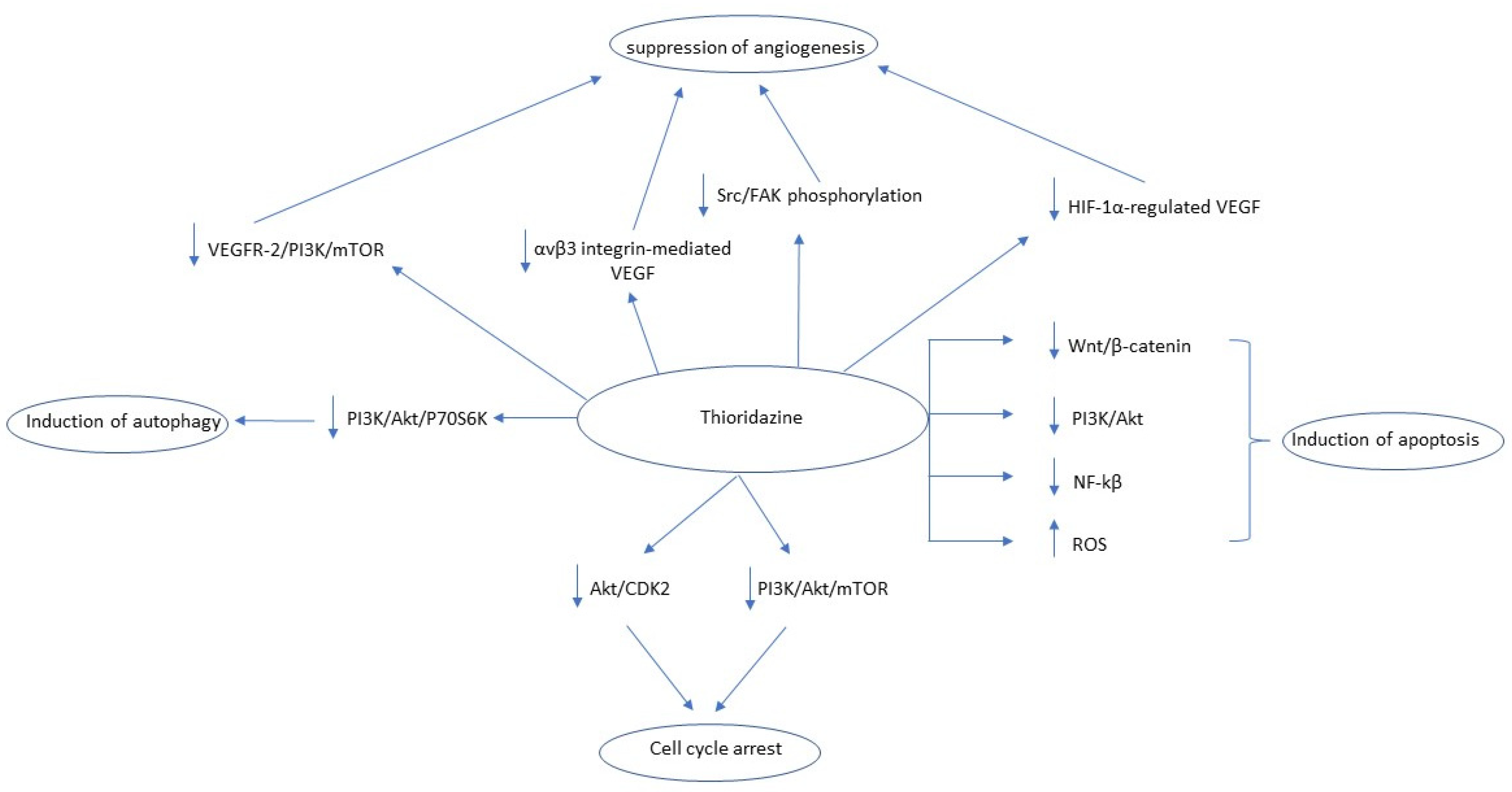
2.7. Thioridazine (THD)
3. Atypical Antipsychotics
3.1. Quetiapine
3.2. Olanzapine
3.3. Risperidone
3.4. Aripiprazole (ARP)
3.5. Clozapine (CLZ)
4. D3 Receptors’ Modulation and Cancer Treatment
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef] [PubMed]
- Housman, G.; Byler, S.; Heerboth, S.; Lapinska, K.; Longacre, M.; Snyder, N.; Sarkar, S.; Housman, G.; Byler, S.; Heerboth, S.; et al. Drug Resistance in Cancer: An Overview. Cancers 2014, 6, 1769–1792. [Google Scholar] [CrossRef]
- Ciociola, A.A.; Cohen, L.B.; Kulkarni, P.; Kefalas, C.; Buchman, A.; Burke, C.; Cain, T.; Connor, J.; Ehrenpreis, E.D.; Fang, J.; et al. How Drugs are Developed and Approved by the FDA: Current Process and Future Directions. Am. J. Gastroenterol. 2014, 109, 620–623. [Google Scholar] [CrossRef]
- Pushpakom, S.; Iorio, F.; Eyers, P.A.; Escott, K.J.; Hopper, S.; Wells, A.; Doig, A.; Guilliams, T.; Latimer, J.; McNamee, C.; et al. Drug repurposing: Progress, challenges and recommendations. Nat. Rev. Drug Discov. 2019, 18, 41–58. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.W. A history of antipsychotic drug development. Compr. Psychiatry 1999, 40, 407–414. [Google Scholar] [CrossRef]
- Miyamoto, S.; Duncan, E.G.; E Marx, C.; Lieberman, A.J. Treatments for schizophrenia: A critical review of pharmacology and mechanisms of action of antipsychotic drugs. Mol. Psychiatry 2004, 10, 79–104. [Google Scholar] [CrossRef]
- Fond, G.; Macgregor, A.; Attal, J.; Larue, A.; Brittner, M.; Ducasse, D.; Capdevielle, D. Antipsychotic drugs: Pro-cancer or anti-cancer? A systematic review. Med. Hypotheses 2012, 79, 38–42. [Google Scholar] [CrossRef]
- Dalton, O.S.; Johansen, C.; Poulsen, A.H.; Nørgaard, M.; Sørensen, H.T.; McLaughlin, J.K.; Mortensen, P.B.; Friis, S. Cancer risk among users of neuroleptic medication: A population-based cohort study. Br. J. Cancer 2006, 95, 934–939. [Google Scholar] [CrossRef]
- López-Muñoz, F.; Alamo, C. The consolidation of neuroleptic therapy: Janssen, the discovery of haloperidol and its introduction into clinical practice. Brain Res. Bull. 2009, 79, 130–141. [Google Scholar] [CrossRef]
- Beach, S.R.; Gross, A.F.; Hartney, K.E.; Taylor, J.B.; Rundell, J.R. Intravenous haloperidol: A systematic review of side effects and recommendations for clinical use. Gen. Hosp. Psychiatry 2020, 67, 42–50. [Google Scholar] [CrossRef] [PubMed]
- Saeedi, H.; Remington, G.; Christensen, B.K. Impact of haloperidol, a dopamine D2 antagonist, on cognition and mood. Schizophr. Res. 2006, 85, 222–231. [Google Scholar] [CrossRef]
- Wahba, M.; Messiha, B.A.S.; Abo-Saif, A.A. Ramipril and haloperidol as promising approaches in managing rheumatoid arthritis in rats. Eur. J. Pharmacol. 2015, 765, 307–315. [Google Scholar] [CrossRef] [PubMed]
- Hoertel, N.; Sánchez-Rico, M.; Vernet, R.; Jannot, A.-S.; Neuraz, A.; Blanco, C.; Lemogne, C.; Airagnes, G.; Paris, N.; Daniel, C.; et al. Observational study of haloperidol in hospitalized patients with COVID-19. PLoS ONE 2021, 16, e0247122. [Google Scholar] [CrossRef] [PubMed]
- Ji, C.; Liu, N.; Tu, J.; Li, Z.; Han, G.; Li, J.; Sheng, C. Drug Repurposing of Haloperidol: Discovery of New Benzocyclane Derivatives as Potent Antifungal Agents against Cryptococcosis and Candidiasis. ACS Infect. Dis. 2019, 6, 768–786. [Google Scholar] [CrossRef] [PubMed]
- Papadopoulos, F.; Isihou, R.; Alexiou, G.A.; Tsalios, T.; Vartholomatos, E.; Markopoulos, G.S.; Sioka, C.; Tsekeris, P.; Kyritsis, A.P.; Galani, V. Haloperidol Induced Cell Cycle Arrest and Apoptosis in Glioblastoma Cells. Biomedicines 2020, 8, 595. [Google Scholar] [CrossRef]
- Hendouei, N.; Saghafi, F.; Shadfar, F.; Hosseinimehr, S.J. Molecular mechanisms of anti-psychotic drugs for improvement of cancer treatment. Eur. J. Pharmacol. 2019, 856, 172402. [Google Scholar] [CrossRef]
- Liu, Z.; Jiang, X.; Gao, L.; Liu, X.; Li, J.; Huang, X.; Zeng, T. Synergistic Suppression of Glioblastoma Cell Growth by Combined Application of Temozolomide and Dopamine D2 Receptor Antagonists. World Neurosurg. 2019, 128, e468–e477. [Google Scholar] [CrossRef]
- He, Y.; Li, J.; Koga, T.; Ma, J.; Dhawan, S.; Suzuki, Y.; Furnari, F.; Prabhu, V.V.; E Allen, J.; Chen, C.C. Epidermal growth factor receptor as a molecular determinant of glioblastoma response to dopamine receptor D2 inhibitors. Neuro-Oncology 2020, 23, 400–411. [Google Scholar] [CrossRef]
- Lauth, M.; Rohnalter, V.; Bergström, Å.; Kooshesh, M.; Svenningsson, P.; Toftgård, R. Antipsychotic Drugs Regulate Hedgehog Signaling by Modulation of 7-Dehydrocholesterol Reductase Levels. Mol. Pharmacol. 2010, 78, 486–496. [Google Scholar] [CrossRef]
- Kim, S.H.; Lee, H.Y.; Yi, H.; Ahn, Y.M.; Kim, Y.S. Haloperidol induces demethylation and expression of the dual specificity phosphatase 6 gene in MIA PaCa-2 human pancreatic cancer cells. Life Sci. 2012, 91, 1317–1322. [Google Scholar] [CrossRef]
- Jandaghi, P.; Najafabadi, H.S.; Bauer, A.S.; Papadakis, A.I.; Fassan, M.; Hall, A.; Monast, A.; von Knebel Doeberitz, M.; Neoptolemos, J.P.; Costello, E.; et al. Expression of DRD2 Is Increased in Human Pancreatic Ductal Adenocarcinoma and Inhibitors Slow Tumor Growth in Mice. Gastroenterology 2016, 151, 1218–1231. [Google Scholar] [CrossRef]
- Sozio, P.; Fiorito, J.; Di Giacomo, V.; Di Stefano, A.; Marinelli, L.; Cacciatore, I.; Cataldi, A.; Pacella, S.; Turkez, H.; Parenti, C.; et al. Haloperidol metabolite II prodrug: Asymmetric synthesis and biological evaluation on rat C6 glioma cells. Eur. J. Med. Chem. 2015, 90, 1–9. [Google Scholar] [CrossRef]
- Marrazzo, A.; Fiorito, J.; Zappalà, L.; Prezzavento, O.; Ronsisvalle, S.; Pasquinucci, L.; Scoto, G.M.; Bernardini, R.; Ronsisvalle, G. Antiproliferative activity of phenylbutyrate ester of haloperidol metabolite II [(±)-MRJF4] in prostate cancer cells. Eur. J. Med. Chem. 2011, 46, 433–438. [Google Scholar] [CrossRef]
- Di Giacomo, V.; Di Valerio, V.; Rapino, M.; Bosco, D.; Cacciatore, I.; Ciulla, M.; Marrazzo, A.; Fiorito, J.; DI Stefano, A.; Cataldi, A. MRJF4, a novel histone deacetylase inhibitor, induces p21 mediated autophagy in PC3 prostate cancer cells. Cell. Mol. Boil. 2015, 61, 17–23. [Google Scholar]
- Barbaraci, C.; Giurdanella, G.; Leotta, C.G.; Longo, A.; Amata, E.; Dichiara, M.; Pasquinucci, L.; Turnaturi, R.; Prezzavento, O.; Cacciatore, I.; et al. Haloperidol Metabolite II Valproate Ester (S)-(−)-MRJF22: Preliminary Studies as a Potential Multifunctional Agent against Uveal Melanoma. J. Med. Chem. 2021, 64, 13622–13632. [Google Scholar] [CrossRef]
- Feng, Z.; Xia, Y.; Gao, T.; Xu, F.; Lei, Q.; Peng, C.; Yang, Y.; Xue, Q.; Hu, X.; Wang, Q.; et al. The antipsychotic agent trifluoperazine hydrochloride suppresses triple-negative breast cancer tumor growth and brain metastasis by inducing G0/G1 arrest and apoptosis. Cell Death Dis. 2018, 9, 1–15. [Google Scholar] [CrossRef]
- Bhat, K.; Saki, M.; Vlashi, E.; Cheng, F.; Duhachek-Muggy, S.; Alli, C.; Yu, G.; Medina, P.; He, L.; Damoiseaux, R.; et al. The dopamine receptor antagonist trifluoperazine prevents phenotype conversion and improves survival in mouse models of glioblastoma. Proc. Natl. Acad. Sci. USA 2020, 117, 11085–11096. [Google Scholar] [CrossRef] [PubMed]
- Caragher, S.; Shireman, J.; Huang, M.; Miska, J.; Atashi, F.; Baisiwala, S.; Park, C.H.; Saathoff, M.; Warnke, L.; Xiao, T.; et al. Activation of Dopamine Receptor 2 Prompts Transcriptomic and Metabolic Plasticity in Glioblastoma. J. Neurosci. 2019, 39, 1982–1993. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.; Xu, R.; Zhang, C.; Xu, Y.; Han, M.; Huang, B.; Chen, A.; Qiu, C.; Thorsen, F.; Prestegarden, L.; et al. Trifluoperazine, a novel autophagy inhibitor, increases radiosensitivity in glioblastoma by impairing homologous recombination. J. Exp. Clin. Cancer Res. 2017, 36, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Luo, X.; Cheng, Y. Trifluoperazine prevents FOXO1 nuclear excretion and reverses doxorubicin-resistance in the SHG44/DOX drug-resistant glioma cell line. Int. J. Mol. Med. 2018, 42, 3300–3308. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.-T.; Wu, A.; Chang, P.M.-H.; Chen, K.-Y.; Yang, C.-N.; Yang, S.-C.; Ho, C.-C.; Chen, C.-C.; Kuo, Y.-L.; Lee, P.-Y.; et al. Trifluoperazine, an Antipsychotic Agent, Inhibits Cancer Stem Cell Growth and Overcomes Drug Resistance of Lung Cancer. Am. J. Respir. Crit. Care Med. 2012, 186, 1180–1188. [Google Scholar] [CrossRef]
- Kang, S.; Hong, J.; Lee, J.M.; Moon, H.E.; Jeon, B.; Choi, J.; Yoon, N.A.; Paek, S.H.; Roh, E.J.; Lee, C.J.; et al. Trifluoperazine, a Well-Known Antipsychotic, Inhibits Glioblastoma Invasion by Binding to Calmodulin and Disinhibiting Calcium Release Channel IP3R. Mol. Cancer Ther. 2016, 16, 217–227. [Google Scholar] [CrossRef]
- Pulkoski-Gross, A.; Li, J.; Zheng, C.; Li, Y.; Ouyang, N.; Rigas, B.; Zucker, S.; Cao, J. Repurposing the Antipsychotic Trifluoperazine as an Antimetastasis Agent. Mol. Pharmacol. 2014, 87, 501–512. [Google Scholar] [CrossRef]
- Xia, Y.; Jia, C.; Xue, Q.; Jiang, J.; Xie, Y.; Wang, R.; Ran, Z.; Xu, F.; Zhang, Y.; Ye, T. Antipsychotic Drug Trifluoperazine Suppresses Colorectal Cancer by Inducing G0/G1 Arrest and Apoptosis. Front. Pharmacol. 2019, 10, 1029. [Google Scholar] [CrossRef] [PubMed]
- Jiang, J.; Huang, Z.; Chen, X.; Luo, R.; Cai, H.; Wang, H.; Zhang, H.; Sun, T.; Zhang, Y. Trifluoperazine Activates FOXO1-Related Signals to Inhibit Tumor Growth in Hepatocellular Carcinoma. DNA Cell Biol. 2017, 36, 813–821. [Google Scholar] [CrossRef]
- Ding, L.; Cao, J.; Lin, W.; Chen, H.; Xiong, X.; Ao, H.; Yu, M.; Lin, J.; Cui, Q. The Roles of Cyclin-Dependent Kinases in Cell-Cycle Progression and Therapeutic Strategies in Human Breast Cancer. Int. J. Mol. Sci. 2020, 21. [Google Scholar] [CrossRef]
- Kamgar-Dayhoff, P.; Brelidze, T.I. Multifaceted effect of chlorpromazine in cancer: Implications for cancer treatment. Oncotarget 2021, 12, 1406–1426. [Google Scholar] [CrossRef]
- Abbruzzese, C.; Matteoni, S.; Persico, M.; Villani, V.; Paggi, M.G. Repurposing chlorpromazine in the treatment of glioblastoma multiforme: Analysis of literature and forthcoming steps. J. Exp. Clin. Cancer Res. 2020, 39, 1–3. [Google Scholar] [CrossRef] [PubMed]
- Matteoni, S.; Matarrese, P.; Ascione, B.; Buccarelli, M.; Ricci-Vitiani, L.; Pallini, R.; Villani, V.; Pace, A.; Paggi, M.G.; Abbruzzese, C. Anticancer Properties of the Antipsychotic Drug Chlorpromazine and Its Synergism with Temozolomide in Restraining Human Glioblastoma Proliferation In Vitro. Front. Oncol. 2021, 11, 635472. [Google Scholar] [CrossRef] [PubMed]
- Aas, A.T.; Brun, A.; Pero, R.W.; Salford, L.G. Chlorpromazine in combination with nitrosourea inhibits experimental glioma growth. Br. J. Neurosurg. 1994, 8, 187–192. [Google Scholar] [CrossRef]
- Oliva, C.R.; Zhang, W.; Langford, C.; Suto, M.J.; Griguer, C.E. Repositioning chlorpromazine for treating chemoresistant glioma through the inhibition of cytochrome c oxidase bearing the COX4-1 regulatory subunit. Oncotarget 2017, 8, 37568–37583. [Google Scholar] [CrossRef] [PubMed]
- Shin, S.Y.; Kim, C.G.; Kim, S.H.; Kim, Y.S.; Lim, Y.; Lee, Y.H. Chlorpromazine activates p21Waf1/Cip1gene transcription via early growth response-1 (Egr-1) in C6 glioma cells. Exp. Mol. Med. 2010, 42, 395–405. [Google Scholar] [CrossRef]
- Shin, S.Y.; Lee, K.S.; Choi, Y.-K.; Lim, H.J.; Lee, H.G.; Lim, Y.; Lee, Y.H. The antipsychotic agent chlorpromazine induces autophagic cell death by inhibiting the Akt/mTOR pathway in human U-87MG glioma cells. Carcinogenesis 2013, 34, 2080–2089. [Google Scholar] [CrossRef]
- Jhou, A.-J.; Chang, H.-C.; Hung, C.-C.; Lin, H.-C.; Lee, Y.-C.; Liu, W.-T.; Han, K.-F.; Lai, Y.-W.; Lin, M.-Y.; Lee, C.-H. Chlorpromazine, an antipsychotic agent, induces G2/M phase arrest and apoptosis via regulation of the PI3K/AKT/mTOR-mediated autophagy pathways in human oral cancer. Biochem. Pharmacol. 2020, 184, 114403. [Google Scholar] [CrossRef]
- Kurita, J.-I.; Hirao, Y.; Nakano, H.; Fukunishi, Y.; Nishimura, Y. Sertraline, chlorprothixene, and chlorpromazine characteristically interact with the REST-binding site of the corepressor mSin3, showing medulloblastoma cell growth inhibitory activities. Sci. Rep. 2018, 8, 13763. [Google Scholar] [CrossRef] [PubMed]
- Zhelev, Z.; Ohba, H.; Bakalova, R.; Hadjimitova, V.; Ishikawa, M.; Shinohara, Y.; Baba, Y. Phenothiazines suppress proliferation and induce apoptosis in cultured leukemic cells without any influence on the viability of normal lymphocytes. Cancer Chemother. Pharmacol. 2004, 53, 267–275. [Google Scholar] [CrossRef]
- Rai, S.; Tanaka, H.; Suzuki, M.; Espinoza, J.L.; Kumode, T.; Tanimura, A.; Yokota, T.; Oritani, K.; Watanabe, T.; Kanakura, Y.; et al. Chlorpromazine eliminates acute myeloid leukemia cells by perturbing subcellular localization of FLT3-ITD and KIT-D816V. Nat. Commun. 2020, 11, 1–14. [Google Scholar] [CrossRef]
- Lee, M.S.; Johansen, L.; Zhang, Y.; Wilson, A.; Keegan, M.; Avery, W.; Elliott, P.; Borisy, A.A.; Keith, C.T. The Novel Combination of Chlorpromazine and Pentamidine Exerts Synergistic Antiproliferative Effects through Dual Mitotic Action. Cancer Res. 2007, 67, 11359–11367. [Google Scholar] [CrossRef]
- Martins, A.S.; Ordóñez, J.L.; Amaral, A.T.; Prins, F.; Floris, G.; Debiec-Rychter, M.; Hogendoorn, P.C.W.; de Álava, E. IGF1R Signaling in Ewing Sarcoma Is Shaped by Clathrin-/Caveolin-Dependent Endocytosis. PLoS ONE 2011, 6, e19846. [Google Scholar] [CrossRef]
- Liang, Z.; Zang, Y.Q.; Lu, Y.; Dong, Q.P.; Dong, K.T.; Zhou, H.F. Chlorpromazine hydrochloride plays a tumor suppressive role in diffuse large B lymphoma by promoting the expression of S1PR2. Chin. J. Ind. Hyg. Occup. Dis. 2021, 39, 418–423. [Google Scholar] [CrossRef]
- Lee, W.-Y.; Lee, W.-T.; Cheng, C.-H.; Chen, K.-C.; Chou, C.-M.; Chung, C.-H.; Sun, M.-S.; Cheng, H.-W.; Ho, M.-N.; Lin, C.-W. Repositioning antipsychotic chlorpromazine for treating colorectal cancer by inhibiting sirtuin 1. Oncotarget 2015, 6, 27580–27595. [Google Scholar] [CrossRef] [PubMed]
- Buscail, L.; Bournet, B.; Cordelier, P. Role of oncogenic KRAS in the diagnosis, prognosis and treatment of pancreatic cancer. Nat. Rev. Gastroenterol. Hepatol. 2020, 17, 153–168. [Google Scholar] [CrossRef]
- Eisenberg, S.; Giehl, K.; Henis, Y.I.; Ehrlich, M. Differential Interference of Chlorpromazine with the Membrane Interactions of Oncogenic K-Ras and Its Effects on Cell Growth. J. Biol. Chem. 2008, 283, 27279–27288. [Google Scholar] [CrossRef] [PubMed]
- Yde, C.W.; Clausen, M.P.; Bennetzen, M.V.; Lykkesfeldt, A.E.; Mouritsen, O.G.; Guerra, B. The antipsychotic drug chlorpromazine enhances the cytotoxic effect of tamoxifen in tamoxifen-sensitive and tamoxifen-resistant human breast cancer cells. Anti-Cancer Drugs 2009, 20, 723–735. [Google Scholar] [CrossRef]
- Yang, C.-E.; Lee, W.-Y.; Cheng, H.-W.; Chung, C.-H.; Mi, F.-L.; Lin, C.-W. The antipsychotic chlorpromazine suppresses YAP signaling, stemness properties, and drug resistance in breast cancer cells. Chem.-Biol. Interact. 2019, 302, 28–35. [Google Scholar] [CrossRef]
- Shaw, V.; Srivastava, S.; Srivastava, S.K. Repurposing antipsychotics of the diphenylbutylpiperidine class for cancer therapy. Semin. Cancer Biol. 2021, 68, 75–83. [Google Scholar] [CrossRef]
- Hongo, H.; Kosaka, T.; Suzuki, Y.; Oya, M. Discovery of a New Candidate Drug to Overcome Cabazitaxel-Resistant Gene Signature in Castration-Resistant Prostate Cancer by in Silico Screening. Prostate Cancer Prostatic Dis. 2021. [Google Scholar] [CrossRef]
- Kim, U.; Kim, C.-Y.; Lee, J.M.; Ryu, B.; Kim, J.; Shin, C.; Park, J.-H. Pimozide Inhibits the Human Prostate Cancer Cells through the Generation of Reactive Oxygen Species. Front. Pharmacol. 2020, 10, 1517. [Google Scholar] [CrossRef] [PubMed]
- Cai, N.; Zhou, W.; Ye, L.-L.; Chen, J.; Liang, Q.-N.; Chang, G.; Chen, J.-J. The STAT3 inhibitor pimozide impedes cell proliferation and induces ROS generation in human osteosarcoma by suppressing catalase expression. Am. J. Transl. Res. 2017, 9, 3853–3866. [Google Scholar] [PubMed]
- Dees, S.; Pontiggia, L.; Jasmin, J.-F.; Mercier, I. Phosphorylated STAT3 (Tyr705) as a biomarker of response to pimozide treatment in triple-negative breast cancer. Cancer Biol. Ther. 2020, 21, 506–521. [Google Scholar] [CrossRef]
- Ranjan, A.; Kaushik, I.; Srivastava, S.K. Pimozide Suppresses the Growth of Brain Tumors by Targeting STAT3-Mediated Autophagy. Cells 2020, 9, 2141. [Google Scholar] [CrossRef] [PubMed]
- Dakir, E.-H.; Pickard, A.; Srivastava, K.; McCrudden, C.M.; Gross, S.; Lloyd, S.; Zhang, S.-D.; Margariti, A.; Morgan, R.; Rudland, P.S.; et al. The anti-psychotic drug pimozide is a novel chemotherapeutic for breast cancer. Oncotarget 2018, 9, 34889–34910. [Google Scholar] [CrossRef]
- Strobl, J.S.; Kirkwood, K.; Lantz, T.K.; Lewine, A.M.; A Peterson, V.; Worley, J.F. Inhibition of human breast cancer cell proliferation in tissue culture by the neuroleptic agents pimozide and thioridazine. Cancer Res. 1990, 50, 5399–5405. [Google Scholar] [PubMed]
- Rondanin, R.; Simoni, D.; Maccesi, M.; Romagnoli, R.; Grimaudo, S.; Pipitone, R.M.; Meli, M.; Cascio, A.; Tolomeo, M. Effects of Pimozide Derivatives on pSTAT5 in K562 Cells. ChemMedChem 2017, 12, 1183–1190. [Google Scholar] [CrossRef]
- Nelson, E.A.; Walker, S.R.; Weisberg, E.; Bar-Natan, M.; Barrett, R.; Gashin, L.B.; Terrell, S.; Klitgaard, J.L.; Santo, L.; Addorio, M.R.; et al. The STAT5 inhibitor pimozide decreases survival of chronic myelogenous leukemia cells resistant to kinase inhibitors. Blood 2011, 117, 3421–3429. [Google Scholar] [CrossRef] [PubMed]
- Nelson, E.A.; Walker, S.R.; Xiang, M.; Weisberg, E.; Bar-Natan, M.; Barrett, R.; Liu, S.; Kharbanda, S.; Christie, A.L.; Nicolais, M.; et al. The STAT5 Inhibitor Pimozide Displays Efficacy in Models of Acute Myelogenous Leukemia Driven by FLT3 Mutations. Genes Cancer 2012, 3, 503–511. [Google Scholar] [CrossRef]
- Xiao, Z.; Liang, J.; Deng, Q.; Song, C.; Yang, X.; Liu, Z.; Shao, Z.; Zhang, K.; Wang, X.; Li, Z. Pimozide augments bromocriptine lethality in prolactinoma cells and in a xenograft model via the STAT5/cyclin D1 and STAT5/Bcl-xL signaling pathways. Int. J. Mol. Med. 2020, 47, 113–124. [Google Scholar] [CrossRef]
- Subramaniam, D.; Angulo, P.; Ponnurangam, S.; Dandawate, P.; Ramamoorthy, P.; Srinivasan, P.; Iwakuma, T.; Weir, S.J.; Chastain, K.; Anant, S. Suppressing STAT5 signaling affects osteosarcoma growth and stemness. Cell Death Dis. 2020, 11, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Fako, V.; Yu, Z.; Henrich, C.J.; Ransom, T.; Budhu, A.S.; Wang, X.W. Inhibition of wnt/β-catenin Signaling in Hepatocellular Carcinoma by an Antipsychotic Drug Pimozide. Int. J. Biol. Sci. 2016, 12, 768–775. [Google Scholar] [CrossRef]
- Gould, R.J.; Murphy, K.M.; Reynolds, I.J.; Snyder, S.H. Antischizophrenic drugs of the diphenylbutylpiperidine type act as calcium channel antagonists. Proc. Natl. Acad. Sci. USA 1983, 80, 5122–5125. [Google Scholar] [CrossRef]
- Shi, X.-N.; Li, H.; Yao, H.; Liu, X.; Li, L.; Leung, K.-S.; Kung, H.-F.; Lu, D.; Wong, M.-H.; Lin, M.C.-M. In Silico Identification and In Vitro and In Vivo Validation of Anti-Psychotic Drug Fluspirilene as a Potential CDK2 Inhibitor and a Candidate Anti-Cancer Drug. PLoS ONE 2015, 10, e0132072. [Google Scholar] [CrossRef] [PubMed]
- Dong, Y.; Furuta, T.; Sabit, H.; Kitabayashi, T.; Jiapaer, S.; Kobayashi, M.; Ino, Y.; Todo, T.; Teng, L.; Hirao, A.; et al. Identification of antipsychotic drug fluspirilene as a potential anti-glioma stem cell drug. Oncotarget 2017, 8, 111728–111741. [Google Scholar] [CrossRef]
- Patil, S.P.; Pacitti, M.F.; Gilroy, K.S.; Ruggiero, J.C.; Griffin, J.D.; Butera, J.J.; Notarfrancesco, J.M.; Tran, S.; Stoddart, J.W. Identification of antipsychotic drug fluspirilene as a potential p53-MDM2 inhibitor: A combined computational and experimental study. J. Comput. Mol. Des. 2014, 29, 155–163. [Google Scholar] [CrossRef]
- Janssen, P.A.; Niemegeers, C.J.; Schellekens, K.H.; Lenaerts, F.M.; Verbruggen, F.J.; Van Nueten, J.M.; Schaper, W.K. The pharmacology of penfluridol (R 16341) a new potent and orally long-acting neuroleptic drug. Eur. J. Pharmacol. 1970, 11, 139–154. [Google Scholar] [CrossRef]
- Santi, C.M.; Cayabyab, F.S.; Sutton, K.G.; McRory, J.E.; Mezeyova, J.; Hamming, K.S.; Parker, D.; Stea, A.; Snutch, T.P. Differential Inhibition of T-Type Calcium Channels by Neuroleptics. J. Neurosci. 2002, 22, 396–403. [Google Scholar] [CrossRef]
- Zaman, A.U.; Sajib, S.; Cucullo, L.; Mikelis, C.M.; German, N.A. Analogs of penfluridol as chemotherapeutic agents with reduced central nervous system activity. Bioorganic Med. Chem. Lett. 2018, 28, 3652–3657. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, A.; Gupta, P.; Srivastava, S.K. Penfluridol: An Antipsychotic Agent Suppresses Metastatic Tumor Growth in Triple-Negative Breast Cancer by Inhibiting Integrin Signaling Axis. Cancer Res. 2015, 76, 877–890. [Google Scholar] [CrossRef]
- Hamidi, H.; Ivaska, J. Every step of the way: Integrins in cancer progression and metastasis. Nat. Rev. Cancer 2018, 18, 533–548. [Google Scholar] [CrossRef]
- Hedrick, E.; Li, X.; Safe, S. Penfluridol Represses Integrin Expression in Breast Cancer through Induction of Reactive Oxygen Species and Downregulation of Sp Transcription Factors. Mol. Cancer Ther. 2016, 16, 205–216. [Google Scholar] [CrossRef] [PubMed]
- Hedrick, E.; Cheng, Y.; Jin, U.-H.; Kim, K.; Safe, S. Specificity protein (Sp) transcription factors Sp1, Sp3 and Sp4 are non-oncogene addiction genes in cancer cells. Oncotarget 2016, 7, 22245–22256. [Google Scholar] [CrossRef] [PubMed]
- Hedrick, E.; Lee, S.-O.; Doddapaneni, R.; Singh, M.; Safe, S. NR4A1 Antagonists Inhibit β1-Integrin-Dependent Breast Cancer Cell Migration. Mol. Cell. Biol. 2016, 36, 1383–1394. [Google Scholar] [CrossRef]
- Chien, W.; Sun, Q.-Y.; Lee, K.L.; Ding, L.-W.; Wuensche, P.; Torres-Fernandez, L.A.; Tan, S.Z.; Tokatly, I.; Zaiden, N.; Poellinger, L.; et al. Activation of protein phosphatase 2A tumor suppressor as potential treatment of pancreatic cancer. Mol. Oncol. 2015, 9, 889–905. [Google Scholar] [CrossRef]
- Wu, S.-Y.; Wen, Y.-C.; Ku, C.-C.; Yang, Y.-C.; Chow, J.-M.; Yang, S.-F.; Lee, W.-J.; Chien, M.-H. Penfluridol triggers cytoprotective autophagy and cellular apoptosis through ROS induction and activation of the PP2A-modulated MAPK pathway in acute myeloid leukemia with different FLT3 statuses. J. Biomed. Sci. 2019, 26, 1–13. [Google Scholar] [CrossRef]
- Ranjan, A.; Srivastava, S.K. Penfluridol suppresses glioblastoma tumor growth by Akt-mediated inhibition of GLI1. Oncotarget 2017, 8, 32960–32976. [Google Scholar] [CrossRef] [PubMed]
- Kim, H.; Chong, K.; Ryu, B.K.; Park, K.J.; Yu, M.O.; Lee, J.; Chung, S.; Choi, S.; Park, M.J.; Chung, Y.G.; et al. Repurposing Penfluridol in Combination with Temozolomide for the Treatment of Glioblastoma. Cancers 2019, 11, 1310. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, A.; Srivastava, S.K. Penfluridol suppresses pancreatic tumor growth by autophagy-mediated apoptosis. Sci. Rep. 2016, 6, 26165. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, A.; German, N.; Mikelis, C.; Srivenugopal, K.; Srivastava, S.K. Penfluridol induces endoplasmic reticulum stress leading to autophagy in pancreatic cancer. Tumor Biol. 2017, 39, 1010428317705517. [Google Scholar] [CrossRef] [PubMed]
- Hung, W.-Y.; Chang, J.-H.; Cheng, Y.; Cheng, G.-Z.; Huang, H.-C.; Hsiao, M.; Chung, C.-L.; Lee, W.-J.; Chien, M.-H. Autophagosome accumulation-mediated ATP energy deprivation induced by penfluridol triggers nonapoptotic cell death of lung cancer via activating unfolded protein response. Cell Death Dis. 2019, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Guo, J.Y.; White, E. Autophagy, Metabolism, and Cancer. Cold Spring Harb. Symp. Quant. Biol. 2016, 81, 73–78. [Google Scholar] [CrossRef]
- Ranjan, A.; Wright, S.; Srivastava, S.K. Immune consequences of penfluridol treatment associated with inhibition of glioblastoma tumor growth. Oncotarget 2017, 8, 47632–47641. [Google Scholar] [CrossRef] [PubMed]
- Gupta, N.; Gupta, P.; Srivastava, S.K. Penfluridol overcomes paclitaxel resistance in metastatic breast cancer. Sci. Rep. 2019, 9, 5066. [Google Scholar] [CrossRef] [PubMed]
- Du, J.; Shang, J.; Chen, F.; Zhang, Y.; Yin, N.; Xie, T.; Zhang, H.; Yushuo, Z.; Liu, F. A CRISPR/Cas9–Based Screening for Non-Homologous End Joining Inhibitors Reveals Ouabain and Penfluridol as Radiosensitizers. Mol. Cancer Ther. 2017, 17, 419–431. [Google Scholar] [CrossRef] [PubMed]
- Wu, L.; Liu, Y.-Y.; Li, Z.-X.; Zhao, Q.; Wang, X.; Yu, Y.; Wang, Y.-Y.; Wang, Y.-Q.; Luo, F. Anti-tumor effects of penfluridol through dysregulation of cholesterol homeostasis. Asian Pac. J. Cancer Prev. 2014, 15, 489–494. [Google Scholar] [CrossRef]
- Thanacoody, R.H. Thioridazine: The good and the bad. Recent Pat. Anti-Infect. Drug Discov. 2011, 6, 92–98. [Google Scholar] [CrossRef]
- Cowap, J.; Hardy, J. Thioridazine in the management of cancer-related sweating. J. Pain Symptom Manag. 1998, 15, 266. [Google Scholar]
- Zhukovsky, D.S. Fever and sweats in the patient with advanced cancer. Hematol. Clin. N. Am. 2002, 16, 579–588. [Google Scholar] [CrossRef]
- Ly, K.L.; Chidgey, J.; Addington-Hall, J.; Hotopf, M. Depression in palliative care: A systematic review. Part 2. Treatment. Palliat. Med. 2002, 16, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Chu, C.-W.; Ko, H.-J.; Chou, C.-H.; Cheng, T.-S.; Cheng, H.-W.; Liang, Y.-H.; Lai, Y.-L.; Lin, C.-Y.; Wang, C.; Loh, J.-K.; et al. Thioridazine Enhances P62-Mediated Autophagy and Apoptosis through Wnt/β-Catenin Signaling Pathway in Glioma Cells. Int. J. Mol. Sci. 2019, 20, 473. [Google Scholar] [CrossRef]
- Johannessen, T.; Hasan-Olive, M.; Zhu, H.; Denisova, O.; Grudic, A.; Latif, A.; Saed, H.; Varughese, J.K.; Røsland, G.V.; Yang, N.; et al. Thioridazine inhibits autophagy and sensitizes glioblastoma cells to temozolomide. Int. J. Cancer 2018, 144, 1735–1745. [Google Scholar] [CrossRef] [PubMed]
- Cheng, H.-W.; Liang, Y.-H.; Kuo, Y.-L.; Chuu, C.-P.; Lin, C.-Y.; Lee, M.-H.; Wu, A.T.H.; Yeh, C.-T.; Chen, E.I.-T.; Whangpeng, J.; et al. Identification of thioridazine, an antipsychotic drug, as an antiglioblastoma and anticancer stem cell agent using public gene expression data. Cell Death Dis. 2015, 6, e1753. [Google Scholar] [CrossRef]
- Gil-Ad, I.; Shtaif, B.; Levkovitz, Y.; Dayag, M.; Zeldich, E.; Weizman, A. Characterization of Phenothiazine-Induced Apoptosis in Neuroblastoma and Glioma Cell Lines: Clinical Relevance and Possible Application for Brain-Derived Tumors. J. Mol. Neurosci. 2004, 22, 189–198. [Google Scholar] [CrossRef]
- Seo, S.U.; Cho, H.K.; Min, K.-J.; Woo, S.M.; Kim, S.; Park, J.-W.; Kim, S.H.; Choi, Y.H.; Keum, Y.S.; Hyun, J.W.; et al. Thioridazine enhances sensitivity to carboplatin in human head and neck cancer cells through downregulation of c-FLIP and Mcl-1 expression. Cell Death Dis. 2017, 8, e2599. [Google Scholar] [CrossRef]
- Park, M.S.; Dong, S.M.; Kim, B.-R.; Seo, S.H.; Kang, S.; Lee, E.-J.; Lee, S.-H.; Rho, S.B. Thioridazine inhibits angiogenesis and tumor growth by targeting the VEGFR-2/PI3K/mTOR pathway in ovarian cancer xenografts. Oncotarget 2014, 5, 4929–4934. [Google Scholar] [CrossRef]
- Qian, G.; Dai, L.; Yu, T. Thioridazine Sensitizes Cisplatin against Chemoresistant Human Lung and Ovary Cancer Cells. DNA Cell Biol. 2019, 38, 718–724. [Google Scholar] [CrossRef]
- Rho, S.B.; Kim, B.-R.; Kang, S. A gene signature-based approach identifies thioridazine as an inhibitor of phosphatidylinositol-3′-kinase (PI3K)/AKT pathway in ovarian cancer cells. Gynecol. Oncol. 2011, 120, 121–127. [Google Scholar] [CrossRef] [PubMed]
- Byun, H.-J.; Lee, J.H.; Kim, B.-R.; Kang, S.; Dong, S.M.; Park, M.S.; Lee, S.-H.; Park, S.H.; Rho, S.B. Anti-angiogenic effects of thioridazine involving the FAK-mTOR pathway. Microvasc. Res. 2012, 84, 227–234. [Google Scholar] [CrossRef] [PubMed]
- Yong, M.; Yu, T.; Tian, S.; Liu, S.; Xu, J.; Hu, J.; Hu, L. DR2 blocker thioridazine: A promising drug for ovarian cancer therapy. Oncol. Lett. 2017, 14, 8171–8177. [Google Scholar] [CrossRef] [PubMed]
- Song, Y.; Li, L.; Chen, J.; Chen, H.; Cui, B.; Feng, Y.; Zhang, P.; Zhang, Q.; Xia, Y.; Luo, M. Thioridazine hydrochloride: An antipsychotic agent that inhibits tumor growth and lung metastasis in triple-negative breast cancer via inducing G0/G1 arrest and apoptosis. Cell Cycle 2020, 19, 3521–3533. [Google Scholar] [CrossRef]
- Tegowski, M.; Fan, C.; Baldwin, A.S. Thioridazine inhibits self-renewal in breast cancer cells via DRD2-dependent STAT3 inhibition, but induces a G1 arrest independent of DRD2. J. Biol. Chem. 2018, 293, 15977–15990. [Google Scholar] [CrossRef]
- Yin, T.; He, S.; Shen, G.; Ye, T.; Guo, F.; Wang, Y. Dopamine receptor antagonist thioridazine inhibits tumor growth in a murine breast cancer model. Mol. Med. Rep. 2015, 12, 4103–4108. [Google Scholar] [CrossRef]
- Gong, L.; Wang, Y.; Tong, S.; Liu, L.; Niu, L.; Yuan, Y.; Bao, Y. Mechanism of Killing Effect of Thioridazine on Human Lung Cancer PC9 Cells. Chin. J. Lung Cancer 2015, 18, 727–733. [Google Scholar] [CrossRef]
- Kang, S.; Dong, S.M.; Kim, B.-R.; Park, M.S.; Trink, B.; Byun, H.-J.; Rho, S.B. Thioridazine induces apoptosis by targeting the PI3K/Akt/mTOR pathway in cervical and endometrial cancer cells. Apoptosis 2012, 17, 989–997. [Google Scholar] [CrossRef]
- Mao, M.; Yu, T.; Hu, J.; Hu, L. Dopamine D2 receptor blocker thioridazine induces cell death in human uterine cervical carcinoma cell line SiHa. J. Obstet. Gynaecol. Res. 2015, 41, 1240–1245. [Google Scholar] [CrossRef]
- Meng, Q.; Sun, X.; Wang, J.; Wang, Y.; Wang, L. The important application of thioridazine in the endometrial cancer. Am. J. Transl. Res. 2016, 8, 2767–2775. [Google Scholar] [PubMed]
- Gil-Ad, I.; Shtaif, B.; Levkovitz, Y.; Nordenberg, J.; Taler, M.; Korov, I.; Weizman, A. Phenothiazines induce apoptosis in a B16 mouse melanoma cell line and attenuate in vivo melanoma tumor growth. Oncol. Rep. 2006, 15, 107–112. [Google Scholar] [CrossRef] [PubMed]
- Jiang, X.; Chen, Z.; Shen, G.; Jiang, Y.; Wu, L.; Li, X.; Wang, G.; Yin, T. Psychotropic agent thioridazine elicits potent in vitro and in vivo anti-melanoma effects. Biomed. Pharmacother. 2018, 97, 833–837. [Google Scholar] [CrossRef] [PubMed]
- Spengler, G.; Molnar, J.; Viveiros, M.; Amaral, L. Thioridazine induces apoptosis of multidrug-resistant mouse lymphoma cells transfected with the human ABCB1 and inhibits the expression of P-glycoprotein. Anticancer. Res. 2011, 31, 4201–4205. [Google Scholar]
- Nagel, D.; Spranger, S.; Vincendeau, M.; Grau, M.; Raffegerst, S.; Kloo, B.; Hlahla, D.; Neuenschwander, M.; von Kries, J.P.; Hadian, K.; et al. Pharmacologic Inhibition of MALT1 Protease by Phenothiazines as a Therapeutic Approach for the Treatment of Aggressive ABC-DLBCL. Cancer Cell 2012, 22, 825–837. [Google Scholar] [CrossRef]
- Aslostovar, L.; Boyd, A.L.; Almakadi, M.; Collins, T.J.; Leong, D.P.; Tirona, R.G.; Kim, R.B.; Julian, J.A.; Xenocostas, A.; Leber, B.; et al. A Phase 1 Trial Evaluating Thioridazine in Combination with Cytarabine in Patients with Acute Myeloid Leukemia. Blood Adv. 2018, 2, 1935–1945. [Google Scholar] [CrossRef]
- Mu, J.; Xu, H.; Yang, Y.; Huang, W.; Xiao, J.; Li, M.; Tan, Z.; Ding, Q.; Zhang, L.; Lü, J.; et al. Thioridazine, an antipsychotic drug, elicits potent antitumor effects in gastric cancer. Oncol. Rep. 2014, 31, 2107–2114. [Google Scholar] [CrossRef]
- Loehr, A.; Pierpont, T.; Gelsleichter, E.; Galang, A.; Fernandez, I.; Moore, E.; Guo, M.; Miller, A.; Weiss, R. Targeting Cancer Stem Cells with Differentiation Agents as an Alternative to Genotoxic Chemotherapy for the Treatment of Malignant Testicular Germ Cell Tumors. Cancers 2021, 13, 2045. [Google Scholar] [CrossRef]
- Lu, M.; Li, J.; Luo, Z.; Zhang, S.; Xue, S.; Wang, K.; Shi, Y.; Zhang, C.; Chen, H.; Li, Z. Roles of dopamine receptors and their antagonist thioridazine in hepatoma metastasis. OncoTargets Ther. 2015, 8, 1543–1552. [Google Scholar] [CrossRef]
- Min, K.-J.; Seo, B.R.; Bae, Y.C.; Yoo, Y.H.; Kwon, T.K. Antipsychotic agent thioridazine sensitizes renal carcinoma Caki cells to TRAIL-induced apoptosis through reactive oxygen species-mediated inhibition of Akt signaling and downregulation of Mcl-1 and c-FLIP(L). Cell Death Dis. 2014, 5, e1063. [Google Scholar] [CrossRef]
- Singh, V.; Jaiswal, P.K.; Ghosh, I.; Koul, H.K.; Yu, X.; De Benedetti, A. Targeting the TLK1/NEK1 DDR axis with Thioridazine suppresses outgrowth of androgen independent prostate tumors. Int. J. Cancer 2019, 145, 1055–1067. [Google Scholar] [CrossRef]
- Shen, J.; Ma, B.; Zhang, X.; Sun, X.; Han, J.; Wang, Y.; Chu, L.; Xu, H.; Yang, Y. Thioridazine has potent antitumor effects on lung cancer stem-like cells. Oncol. Lett. 2017, 13, 1563–1568. [Google Scholar] [CrossRef] [PubMed]
- Yue, H.; Huang, D.; Qin, L.; Zheng, Z.; Hua, L.; Wang, G.; Huang, J.; Huang, H. Targeting Lung Cancer Stem Cells with Antipsychological Drug Thioridazine. BioMed Res. Int. 2016, 2016, 1–7. [Google Scholar] [CrossRef]
- Zhang, C.; Gong, P.; Liu, P.; Zhou, N.; Zhou, Y.; Wang, Y. Thioridazine elicits potent antitumor effects in colorectal cancer stem cells. Oncol. Rep. 2016, 37, 1168–1174. [Google Scholar] [CrossRef]
- Sachlos, E.; Risueño, R.M.; Laronde, S.; Shapovalova, Z.; Lee, J.-H.; Russell, J.; Malig, M.; McNicol, J.D.; Fiebig-Comyn, A.; Graham, M.; et al. Identification of Drugs Including a Dopamine Receptor Antagonist that Selectively Target Cancer Stem Cells. Cell 2012, 149, 1284–1297. [Google Scholar] [CrossRef]
- Wang, Y.; Huang, N.; Li, H.; Liu, S.; Chen, X.; Yu, S.; Wu, N.; Bian, X.-W.; Shen, H.-Y.; Li, C.; et al. Promoting oligodendroglial-oriented differentiation of glioma stem cell: A repurposing of quetiapine for the treatment of malignant glioma. Oncotarget 2017, 8, 37511–37524. [Google Scholar] [CrossRef] [PubMed]
- Bhat, K.; Saki, M.; Cheng, F.; He, L.; Zhang, L.; Ioannidis, A.; Nathanson, D.; Tsang, J.; Bensinger, S.J.; Nghiemphu, P.L.; et al. Dopamine Receptor Antagonists, Radiation, and Cholesterol Biosynthesis in Mouse Models of Glioblastoma. J. Natl. Cancer Inst. 2021, 113, 1094–1104. [Google Scholar] [CrossRef]
- Chen, V.C.-H.; Chan, H.-L.; Hsu, T.-C.; Lu, M.-L.; Lee, Y.-C.; Lee, Y.; Siow, J.Y.; McIntyre, R.S.; Zhou, A.J.; Tzang, B.-S.; et al. New use for old drugs: The protective effect of atypical antipsychotics on hepatocellular carcinoma. Int. J. Cancer 2018, 144, 2428–2439. [Google Scholar] [CrossRef]
- Lee, Y.-J.; Chung, J.-G.; Tan, Z.-L.; Hsu, F.-T.; Liu, Y.-C.; Lin, S.-S. ERK/AKT Inactivation and Apoptosis Induction Associate With Quetiapine-inhibited Cell Survival and Invasion in Hepatocellular Carcinoma Cells. Vivo 2020, 34, 2407–2417. [Google Scholar] [CrossRef] [PubMed]
- Navari, R.M.; Qin, R.; Ruddy, K.J.; Liu, H.; Powell, S.F.; Bajaj, M.; Dietrich, L.; Biggs, D.; Lafky, J.M.; Loprinzi, C.L. Olanzapine for the Prevention of Chemotherapy-Induced Nausea and Vomiting. N. Engl. J. Med. 2016, 375, 134–142. [Google Scholar] [CrossRef] [PubMed]
- Karpel-Massler, G.; Kast, R.E.; Westhoff, M.-A.; Dwucet, A.; Welscher, N.; Nonnenmacher, L.; Hlavac, M.; Siegelin, M.D.; Wirtz, C.R.; Debatin, K.-M.; et al. Olanzapine inhibits proliferation, migration and anchorage-independent growth in human glioblastoma cell lines and enhances temozolomide’s antiproliferative effect. J. Neuro-Oncol. 2014, 122, 21–33. [Google Scholar] [CrossRef] [PubMed]
- Sanomachi, T.; Suzuki, S.; Kuramoto, K.; Takeda, H.; Sakaki, H.; Togashi, K.; Seino, S.; Yoshioka, T.; Okada, M.; Kitanaka, C. Olanzapine, an Atypical Antipsychotic, Inhibits Survivin Expression and Sensitizes Cancer Cells to Chemotherapeutic Agents. Anticancer Res. 2017, 37, 6177–6188. [Google Scholar] [CrossRef] [PubMed]
- Chopko, T.C.; Lindsley, C.W. Classics in Chemical Neuroscience: Risperidone. ACS Chem. Neurosci. 2018, 9, 1520–1529. [Google Scholar] [CrossRef]
- Möller, H.-J. Risperidone: A review. Expert Opin. Pharmacother. 2005, 6, 803–818. [Google Scholar] [CrossRef]
- Dilly, S.J.; Clark, A.J.; Marsh, A.; Mitchell, D.A.; Cain, R.; Fishwick, C.W.; Taylor, P.C. A chemical genomics approach to drug reprofiling in oncology: Antipsychotic drug risperidone as a potential adenocarcinoma treatment. Cancer Lett. 2017, 393, 16–21. [Google Scholar] [CrossRef]
- Yang, S.-Y.; He, X.-Y.; Schulz, H. Multiple functions of type 10 17β-hydroxysteroid dehydrogenase. Trends Endocrinol. Metab. 2005, 16, 167–175. [Google Scholar] [CrossRef]
- He, X.; Yang, X.Y.H.A.S.Y. Roles of Type 10 17beta-Hydroxysteroid Dehydrogenase in Intracrinology and Metabolism of Isoleucine and Fatty Acids. Endocr. Metab. Immune Disord.-Drug Targets 2006, 6, 95–102. [Google Scholar] [CrossRef]
- Wang, J.-S.; Zhu, H.-J.; Markowitz, J.S.; Donovan, J.L.; Yuan, H.-J.; DeVane, C.L. Antipsychotic Drugs Inhibit the Function of Breast Cancer Resistance Protein. Basic Clin. Pharmacol. Toxicol. 2008, 103, 336–341. [Google Scholar] [CrossRef] [PubMed]
- Gündoğdu, U.D.; Çoban, F.; Liman, R.; Şelli, M.E. Investigation of Risperidone’s anti-tumor activity on MCF-7 breast cancer cells. Ann. Clin. Anal. Med. 2021, 12, 1267–1271. [Google Scholar] [CrossRef]
- Persad, R.; Liu, C.; Wu, T.-T.; Houlihan, P.S.; Hamilton, S.R.; Diehl, A.M.; Rashid, A. Overexpression of Caspase-3 in Hepatocellular Carcinomas. Mod. Pathol. 2004, 17, 861–867. [Google Scholar] [CrossRef] [PubMed]
- Prommer, E. Aripiprazole. Am. J. Hosp. Palliat. Med. 2015, 34, 180–185. [Google Scholar] [CrossRef]
- Badran, A.; Tul-Wahab, A.; Zafar, H.; Mohammad, N.; Imad, R.; Khan, M.A.; Baydoun, E.; Choudhary, M.I. Antipsychotics drug aripiprazole as a lead against breast cancer cell line (MCF-7) in vitro. PLoS ONE 2020, 15, e0235676. [Google Scholar] [CrossRef]
- Lee, H.; Kang, S.; Sonn, J.K.; Lim, Y. Dopamine receptor D2 activation suppresses the radiosensitizing effect of aripiprazole via activation of AMPK. FEBS Open Bio 2019, 9, 1580–1588. [Google Scholar] [CrossRef]
- Suzuki, S.; Okada, M.; Kuramoto, K.; Takeda, H.; Sakaki, H.; Watarai, H.; Sanomachi, T.; Seino, S.; Yoshioka, T.; Kitanaka, C. Aripiprazole, an Antipsychotic and Partial Dopamine Agonist, Inhibits Cancer Stem Cells and Reverses Chemoresistance. Anticancer Res. 2016, 36, 5153–5162. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.Y.; Tae, I.H.; Lee, B.-M.; Kim, H.S.; Yoon, S. Low Doses of the Anti-psychotic Drug Aripiprazole Have Strong P-gp-inhibitory Activity and Sensitize Anti-mitotic Drug-resistant Cancer Cells. Anticancer Res. 2018, 38, 5101–5108. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Lee, S.H.; Park, J.H.; Lee, J.-S.; Park, J.W.; Kim, J.R.; Lee, S.H.; Kim, H.S.; Yoon, S. A Low Dose of Aripiprazole Has the Strongest Sensitization Effect among 19 Repositioned Bipolar Drugs in P-gp-overexpressing Drug-resistant Cancer Cells. Anticancer Res. 2021, 41, 687–697. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.S.; Yoo, B.C.; Yang, W.S.; Han, S.Y.; Jeong, D.; Song, J.M.; Kim, K.-H.; Aravinthan, A.; Kim, J.H.; Kim, J.-H.; et al. Src is the primary target of aripiprazole, an atypical antipsychotic drug, in its anti-tumor action. Oncotarget 2017, 9, 5979–5992. [Google Scholar] [CrossRef] [PubMed]
- Schmid, C.L.; Streicher, J.M.; Meltzer, H.Y.; Bohn, L.M. Clozapine Acts as an Agonist at Serotonin 2A Receptors to Counter MK-801-Induced Behaviors through a βArrestin2-Independent Activation of Akt. Neuropsychopharmacology 2014, 39, 1902–1913. [Google Scholar] [CrossRef]
- Yin, Y.-C.; Lin, C.-C.; Chen, T.-T.; Chen, J.-Y.; Tsai, H.-J.; Wang, C.-Y.; Chen, S.-Y. Clozapine Induces Autophagic Cell Death in Non-Small Cell Lung Cancer Cells. Cell. Physiol. Biochem. 2015, 35, 945–956. [Google Scholar] [CrossRef]
- Massari, N.A.; Medina, V.A.; Cricco, G.P.; Lamas, D.J.M.; Sambuco, L.; Pagotto, R.; Ventura, C.; Ciraolo, P.J.; Pignataro, O.; Bergoc, R.M.; et al. Antitumor activity of histamine and clozapine in a mouse experimental model of human melanoma. J. Dermatol. Sci. 2013, 72, 252–262. [Google Scholar] [CrossRef]
- Shin, S.Y.; Choi, B.H.; Ko, J.; Kim, S.H.; Kim, Y.S.; Lee, Y.H. Clozapine, a neuroleptic agent, inhibits Akt by counteracting Ca2+/calmodulin in PTEN-negative U-87MG human glioblastoma cells. Cell. Signal. 2006, 18, 1876–1886. [Google Scholar] [CrossRef]
- Geyer, M.A. Gerhard Gross. Handbook of Experimental Pharmacology. Vol. 213, Novel Antischizophrenia Treatments; Springer: Heidelberg, Germany, 2012. [Google Scholar]
- Torrisi, S.A.; Salomone, S.; Geraci, F.; Caraci, F.; Bucolo, C.; Drago, F.; Leggio, G.M. Buspirone Counteracts MK-801-Induced Schizophrenia-Like Phenotypes through Dopamine D3 Receptor Blockade. Front. Pharmacol. 2017, 8, 710. [Google Scholar] [CrossRef]
- Wang, Y.; Yang, X.; Song, X.; Zhao, L.; Wei, J.; Wang, J.; Tian, H.; Zheng, C.; Wei, M.; Wang, Q.; et al. Co-treatment of buspirone with atypical antipsychotic drugs (AAPDs) improved neurocognitive function in chronic schizophrenia. Schizophr. Res. 2019, 209, 135–140. [Google Scholar] [CrossRef]
- Pendergrass, J.C.; Targum, S.D.; Harrison, J.E. Cognitive Impairment Associated with Cancer: A Brief Review. Innov. Clin. Neurosci. 2018, 15, 36–44. [Google Scholar] [PubMed]
- Hussein, N.; Amawi, H.; Karthikeyan, C.; Hall, F.S.; Mittal, R.; Trivedi, P.; Ashby, C.R.; Tiwari, A.K. The dopamine D 3 receptor antagonists PG01037, NGB2904, SB277011A, and U99194 reverse ABCG2 transporter-mediated drug resistance in cancer cell lines. Cancer Lett. 2017, 396, 167–180. [Google Scholar] [CrossRef]
- Edinoff, A.; Ruoff, M.T.; Ghaffar, Y.T.; Rezayev, A.; Jani, D.; Kaye, A.M.; Cornett, E.M.; Kaye, A.D.; Viswanath, O.; Urits, I. Cariprazine to Treat Schizophrenia and Bipolar Disorder in Adults. Psychopharmacol. Bull. 2020, 50, 83–117. [Google Scholar]
- Hussein, N.; Ashby, J.C.R.; Amawi, H.; Nyinawabera, A.; Vij, A.; Khare, V.M.; Karthikeyan, C.; Tiwari, A.K. Cariprazine, A. Dopamine D2/D3 Receptor Partial Agonist, Modulates ABCG2-Mediated Multidrug Resistance in Cancer. Cancers 2018, 10, 308. [Google Scholar] [CrossRef] [PubMed]
- Neifeld, J.P.; Tormey, D.C.; A Baker, M.; Meyskens, F.L.; Taub, R.N. Phase II trial of the dopaminergic inhibitor pimozide in previously treated melanoma patients. Cancer Treat. Rep. 1983, 67, 155–157. [Google Scholar] [PubMed]

| Cancer | Drug | Start Date | Title | NCT | Phase | Results | Estimated Completion Year/Date |
|---|---|---|---|---|---|---|---|
| Glioblastoma | Chlorpromazine | 15 December 2019 | Repurposing chlorpromazine for the Treatment of Glioblastoma (RACTAC) | NCT04224441 | II | Not posted | 15 December 2022 |
| Metastatic Melanoma [162] | Pimozide | n/s | Phase II trial of pimozide in previously treated melanoma patients | n/a | II | 17% of the evaluated patients had complete or partial response for at least 8 months | Completed (1983) |
| Acute myeloid leukemia | Thioridazine (THD) | July 2014 | Safety study of Thioridazine in combination with Cytarabine to treat relapsed or refractory Acute Myeloid Leukemia (THORIDAL) | NCT02096289 | I | A reduction up to 55% in blast levels was noted in 85% of the patients | Completed (September 2016) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vlachos, N.; Lampros, M.; Voulgaris, S.; Alexiou, G.A. Repurposing Antipsychotics for Cancer Treatment. Biomedicines 2021, 9, 1785. https://doi.org/10.3390/biomedicines9121785
Vlachos N, Lampros M, Voulgaris S, Alexiou GA. Repurposing Antipsychotics for Cancer Treatment. Biomedicines. 2021; 9(12):1785. https://doi.org/10.3390/biomedicines9121785
Chicago/Turabian StyleVlachos, Nikolaos, Marios Lampros, Spyridon Voulgaris, and George A. Alexiou. 2021. "Repurposing Antipsychotics for Cancer Treatment" Biomedicines 9, no. 12: 1785. https://doi.org/10.3390/biomedicines9121785
APA StyleVlachos, N., Lampros, M., Voulgaris, S., & Alexiou, G. A. (2021). Repurposing Antipsychotics for Cancer Treatment. Biomedicines, 9(12), 1785. https://doi.org/10.3390/biomedicines9121785






