Less Can Be More: The Hormesis Theory of Stress Adaptation in the Global Biosphere and Its Implications
Abstract
:1. Introduction
2. Evolutionary Origin
2.1. The Beginnings
2.2. Nrf2 and Its Role in Anti-Oxidative and Anti-Inflammatory Cellular Responses
3. Low-Dose Radiation (LDR) Mediated Hormesis Effect in the Immune System
3.1. LDR and Innate Immunity
3.2. LDR and Adaptive Immunity
4. Other Hormetic Effects in the Immune System
4.1. Protection by Immunological Memory
4.2. Increase of Longevity and Tissue Protection by Macrophages as Hormesis Effects against Biological Threats
4.3. Hormetic Effects on the Immune System by Sportive Exercise
5. Clinical Implications
5.1. Low Stimulatory Effects of Toxic Compounds
5.2. Psychiatry
5.3. Neurodegenerative Diseases
5.4. Cardiovascular Diseases (CVD)
5.5. Metabolic Syndrom
5.6. Autoimmune Diseases
5.7. Acute Respiratory Distress Syndrome (ARDS)
5.8. Multidrug Interaction
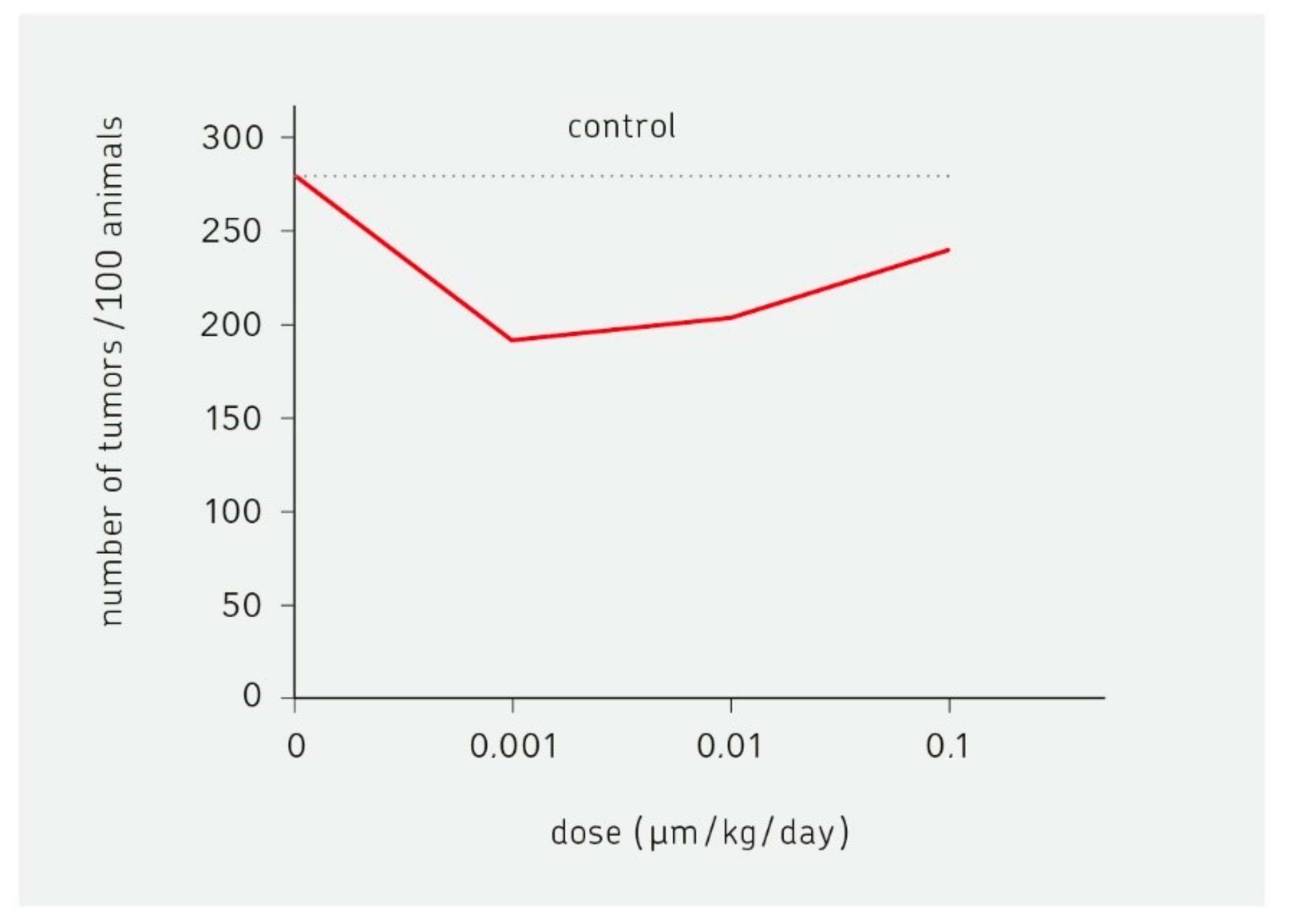
6. Is Less More in Cancer Therapy?
6.1. Historic Aspects
6.2. Hormetic Aspects of Targeted Therapies, Oncolytic Viruses and Cancer Vaccines
6.2.1. Hormetic Aspects of Small Molecule Inhibitors (SMIs)
6.2.2. Hormetic Aspects of Antigen Recognition by the Immune System
6.2.3. Hormetic Aspects of Oncolytic Viruses and Cancer Vaccines.
6.3. Low-Dose T Cell Triggering and Cytotoxic Effector Function
6.3.1. mRNA-Based Vaccines
6.3.2. Peptide-Based Vaccines
6.4. Mitohormesis, Macrophages and Case Reports
7. Hormesis Effects in Plants
8. Archaic Environmental Stress Response as an Example of Hormesis
9. Global Aspects
10. Discussion
11. Summary and Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Calabbrese, E.J.; Baldwin, L.A. Hormesis: U-shaped dose responses and their centrality to toxicology. Trends Pharmacol. Sci. 2001, 22, 285–291. [Google Scholar] [CrossRef] [PubMed]
- Agathokleous, E.; Kitao, M.; Calabrese, E.J. Hormesis: Highly generalizable and beyond laboratory. Trends Plant Sci. 2020, 25, 1076–1086. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J.; Blain, R. The occurence of hormetic dose responses in the toxicological literature, the hormesis database: An overview. Toxicol. Appl. Pharmacol. 2005, 202, 285–301. [Google Scholar] [CrossRef] [PubMed]
- Agathokleous, E.; Barcelo, D.; Tsatsakis, A.; Calabrese, E.J. Hydrocarbon-induced hormesis: 101 Years of evidence at the margin? Environ. Pollut. 2020, 265 Pt B, 11846. [Google Scholar] [CrossRef]
- Fernàndez, J.P.S. The downfall of the linear non-threshold model. Rev. Esp. Med. Nucl. Imagen. Mol. 2020, 39, 303–315. [Google Scholar] [CrossRef]
- Calabrese, E.J. Hormesis: Principles and applications. Homeopathy 2015, 104, 69–82. [Google Scholar] [CrossRef]
- Schirrmacher, V. Mitochondria at work: New insights into regulation and dysregulation of cellular energy supply and metabolism. Biomedicines 2020, 8, 526. [Google Scholar] [CrossRef]
- Sthijns, M.M.J.P.E.; Weseler, A.R.; Bast, A.; Haenen, G.R.M.M. Time in Redox Adaptation Processes: From Evolution to Hormesis. Int. J. Mol. Sci. 2016, 17, 1649. [Google Scholar] [CrossRef] [Green Version]
- Rico, A. Chemo-defence system. C. R. Acad. Sci. III 2001, 324, 97–106. [Google Scholar] [CrossRef]
- Xu, Y.; Liu, S.; Chen, F.; Wang, Z. pH affects the hormesis profiles of personal care product components on luminescence of the bacteria Vibrio qinghaiensis sp.-Q67. Sci. Total Environ. 2020, 713, 136656. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Chen, Y.; Yang, W.; Qiao, Z.; Zhang, X. Complete genome sequence of fish-pathogenic Aeromonas hydrophila HX-3 and a comparative analysis: Insights into virulence factors and quorum sensing. Sci. Rep. 2020, 10, 15479. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.; Wang, D.; Wu, X.; Lin, Z.; Long, X.; Liu, Y. Hormetic mechanism of sulfonamides on Aliivibrio fischeri luminescence based on a bacterial cell-cell communication. Chemospere 2019, 215, 793–799. [Google Scholar] [CrossRef] [PubMed]
- Sun, H.; Zhang, X.; Wang, D.; Lin, Z. Insights into the role of energy source in hormesis through diauxic growth of bacteria in mixed cultivation systems. Chemosphere 2020, 261, 127669. [Google Scholar] [CrossRef] [PubMed]
- Nicolic, B.; Vasilijevic, B.; Culafic, D.; Vukovic-Gacic, B.; Knezevic-Vukcevic, J. Comparative study of genotoxic, antigenotoxic and cytotoxic activities of monoterpenes camphor, eucalyptol and thujone in bacteria and mammalian cells. Chem. Biol. Interact. 2015, 242, 263–271. [Google Scholar] [CrossRef]
- Kudryasheva, N.S.; Rozhko, T.V. Effect of low-dose ionizing radiation on luminous marine bacteria: Radiation hormesis and toxicity. J. Environ. Radioact. 2015, 142, 68–77. [Google Scholar] [CrossRef]
- Zemva, J.; Fink, C.A.; Fleming, T.H.; Schmidt, L.; Loft, A.; Herzig, S.; Knieß, R.A.; Mayer, M.; Bukau, B.; Nawroth, P.P.; et al. Hormesis enables cells to handle accumulating toxic metabolites during increased energy flux. Redox Biol. 2017, 13, 674–686. [Google Scholar] [CrossRef] [PubMed]
- Kubohara, Y.; Kikuchi, H. Dictyostelium: An important source of structural and functional diversity in drug discovery. Cells 2019, 8, 6. [Google Scholar] [CrossRef] [Green Version]
- Moore, M.N.; Shaw, J.P.; Adams, D.R.F.; Viarengo, A. Anti-oxidative cellular protection effect of fasting-induced autophagy as a mechanism of hormesis. Mar. Environ. Res. 2015, 107, 35–44. [Google Scholar] [CrossRef] [Green Version]
- Matai, L.; Sarker, G.C.; Chamoli, M.; Malik, Y.; Kumar, S.S.; Rautela, U.; Jana, N.R.; Chakraborty, K.; Mukhopadhyay, A. Dietary restriction improves proteasis and increases life span through endoplasmic reticulum hormesis. Proc. Natl. Acad. Sci. USA 2019, 116, 17383–17392. [Google Scholar] [CrossRef] [Green Version]
- Kishimoto, S.; Uno, M.; Okabe, E.; Nono, M.; Nishida, E. Environmental stresses induce transgenerationally inheritable survival advantages via germline-to-soma communication in Caenorhabditis elegans. Nat. Commun. 2017, 8, 14031. [Google Scholar] [CrossRef] [Green Version]
- Le Bourg, E. Characterization of the positive effects of mild stress on ageing and resistance to stress. Biogerontology 2020, 21, 485–493. [Google Scholar] [CrossRef] [PubMed]
- Le Bourg, E. Combining three mild stresses in Drosophila melanogaster flies does not have a more positive effect on resistance to a severe cold stress than combining two mild stresses. Biogerontology 2017, 18, 275–284. [Google Scholar] [CrossRef]
- Le Bourg, E. Life-time protection against severe heat stress by exposing young Drosophila melanogaster flies to a mild cold stress. Biogerontology 2016, 17, 409–415. [Google Scholar] [CrossRef]
- Berry, R., 3rd; Lopez-Martinez, G. A dose of experimental hormesis: When mild stress protects and improves animal performance. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 2020, 242, 110658. [Google Scholar] [CrossRef] [PubMed]
- Shushimita, S.; Grefhorst, A.; Steenbergen, J.; de Bruin, R.W.F.; Ijzermans, J.N.M.; Themmen, A.P.N.; Dor, F.J.M.F. Protection against renal ischemia-reperfusion injury through hormesis? Dietary intervention versus cold exposure. Life Sci. 2016, 144, 69–79. [Google Scholar] [CrossRef]
- Teets, N.M.; Gantz, J.D.; Kawarasaki, Y. Rapid cold hardening: Ecological relevance, physiological mechanisms and new perspectives. J. Exp. Biol. 2020, 223 Pt 3, jeb203448. [Google Scholar] [CrossRef]
- Calabrese, E.J. Hormesis: Path and progression to significance. Int. J. Mol. Sci. 2018, 19, 2871. [Google Scholar] [CrossRef] [Green Version]
- Saha, S.; Buttari, B.; Panieri, E.; Profumo, E.; Saso, L. An overview of Nrf2 signaling pathway and ist role in inflammation. Molecules 2020, 25, 5474. [Google Scholar] [CrossRef] [PubMed]
- Cui, J.; Yang, G.; Pan, Z.; Zhao, Y.; Liang, X.; Li, W.; Cai, L. Hormetic response to low-dose radiation: Focus on the immune system and its clinical implications. Int. J. Mol. Sci. 2017, 18, 280. [Google Scholar] [CrossRef] [PubMed]
- Yang, G.; Kong, Q.; Wang, G.; Jin, H.; Zhou, L.; Yu, D.; Niu, C.; Han, W.; Li, W.; Cui, J. Low-dose ionizing radiation induces direct activation of natural killer cells and provides a novel approach for adoptive cellular immunotherapy. Cancer Biother. Radiopharm. 2014, 29, 428–434. [Google Scholar] [CrossRef] [Green Version]
- Klug, F.; Prakash, H.; Huber, P.E.; Seibel, T.; Bender, N.; Halama, N.; Pfirschke, C.; Voss, R.H.; Timke, C.; Umansky, L.; et al. Low-dose irradiation programs macrophage differentiation to an iNOS+/M1 phenotype that orchestrates effective T cell immunotherapy. Cancer Cell 2013, 34, 589–602. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schaue, D.; Marples, B.; Trott, K.R. The effects of low-dose X-irradiation on the oxidative burst in stimulated macrophages. Int. J. Radiat. Biol. 2002, 78, 567–576. [Google Scholar] [CrossRef]
- Jahns, J.; Anderegg, U.; Saalbach, A.; Rosin, B.; Patties, I.; Glasow, A.; Kamprad, M.; Scholz, M.; Hildebrandt, G. Influence of low dose irradiation on differentiation, maturation and T-cell activation of human dendritic cells. Mutat. Res. 2011, 710, 32–39. [Google Scholar] [CrossRef] [PubMed]
- Shigematsu, A.; Adachi, Y.; Koike-Kiriyama, N.; Suzuki, Y.; Iwasaki, M.; Koike, Y.; Nakano, K.; Mukaide, H.; Imamura, M.; Ikehara, S. Effects of low-dose irradiation on enhancement of immunity by dendritic cells. J. Radiat. Res. 2007, 48, 51–55. [Google Scholar] [CrossRef] [Green Version]
- Song, K.H.; Kim, M.H.; Kang, S.M.; Jung, S.Y.; Ahn, J.; Woo, H.J.; Nam, S.Y.; Hwang, S.G.; Ryu, S.Y.; Song, J.Y. Analysis of the immune cell populations and cytokine profiles in murine splenocytes exposed to whole-body low-dose irradiation. Int. J. Radiat. Biol. 2015, 91, 795–803. [Google Scholar] [CrossRef]
- Shankar, B.; Pandey, R.; Sainis, K. Radiation-induced bystander effects and adaptive response in murine lymphocytes. Int. J. Radiat. Biol. 2006, 82, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Rizvi, A.; Pecaut, M.J.; Slater, J.M.; Subramaniam, S.; Gridley, D.S. Low-dose γ-rays modify CD4+ T cell signalling response to simulated solar particle event protons in a mouse model. Int. J. Radiat. Biol. 2011, 87, 24–35. [Google Scholar] [CrossRef]
- Liu, S.Z.; Zhang, Y.C.; Su, X. Effect of low dose radiation on the expression of TCR/CD3 and CD25 on mouse thymocyte plasma membrane. Clin. J. Pathophysiol. 1995, 11, 2–5. [Google Scholar]
- Liu, R.; Xiong, S.; Zhang, L.; Chu, Y. Enhancement of antitumor immunity by low-dose total body irradiation is associated with selectively decreasing the proportion and number of T regulatory cells. Cell. Mol. Immunol. 2010, 7, 157–162. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, X.D.; Liu, S.Z.; Ma, S.M.; Liu, Y. Expression of IL-10 in mouse spleen at mRNA and protein level after whole-body X-irradiation. Chin. J. Radiol. Med. Prot. 2001, 22, 10–12. [Google Scholar]
- Rho, H.S.; Park, S.S.; Lee, C.E. Gamma irradiation up-regulates expression of B cell differentiation molecule CD23 by NF-κB activation. J. Biochem. Mol. Biol. 2004, 37, 507–514. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ye, S.; Yuan, D.; Xie, Y.; Pan, Y.; Shao, C. Role of DNA methylation in long-term low-dose γ-rays induced adaptive response in human B lymphoblast cells. Int. J. Radiat. Biol. 2013, 89, 898–906. [Google Scholar] [CrossRef] [PubMed]
- Lall, R.; Ganapathy, S.; Yang, M.; Xiao, S.; Xu, T.; Su, H.; Shadfan, M.; Asara, J.M.; Ha, C.S.; Ben-Sahra, I.; et al. Low-dose radiation exposure induces a HIF-1-mediated adaptive and protective metabolic response. Cell Death Differ. 2014, 21, 836–844. [Google Scholar] [CrossRef] [PubMed]
- Csaba, G. Hormesis and immunity: A review. Acta Microbiol. Immunol. Hung. 2019, 66, 155–168. [Google Scholar] [CrossRef] [Green Version]
- Muri, J.; Kopf, M. Redox regulation of immunometabolism. Nat. Rev. Immunol. 2020. [Google Scholar] [CrossRef]
- Kaiser, J. Sipping from a poisoned chalice. Science 2003, 302, 376–379. [Google Scholar] [CrossRef] [PubMed]
- Collins, N.; Han, S.; Enamorado, M.; Link, V.M.; Huang, B.; Moseman, E.A.; Kishton, R.J.; Shannon, J.P.; Dixit, D.; Schwab, S.R.; et al. The bone marrow protects and optimizes immunological memory during dietary restriction. Cell 2019, 178, 1088–1101. [Google Scholar] [CrossRef] [PubMed]
- McClure, C.D.; Zhong, W.; Hunt, V.L.; Chapman, F.M.; Hill, F.V.; Priest, K. Hormesis results in trade-offs with immunity. Evolution 2014, 68, 2225–2233. [Google Scholar] [CrossRef] [Green Version]
- Calabrese, E.J.; Giordano, J.J.; Kozumbo, W.J.; Leak, R.K.; Bhatia, T.N. Hormesis mediates dose-sensitive shifts in macrophage activation patterns. Pharmacol. Res. 2018, 137, 236–249. [Google Scholar] [CrossRef]
- Clanton, R.; Saucier, D.; Ford, J.; Akabani, G. Microbial influences on hormesis, oncogenesis, and therapy: A review of the literature. Environ. Res. 2015, 142, 239–256. [Google Scholar] [CrossRef]
- Ortega, E. The “bioregulatory effect of exercise” on the innate/inflammatory responses. J. Physiol. Biochem. 2016, 72, 361–369. [Google Scholar] [CrossRef]
- An, J.; Li, F.; Qin, Y.; Zhang, H.; Ding, S. Low concentrations of FA exhibits the hormesis effect by affecting cell division and the Warburg effect. Ecotoxicol. Environ. Saf. 2019, 183, 109576. [Google Scholar] [CrossRef]
- Sies, H.; Feinendegen, L.E. Radiation hormesis: The link to nanomolar hydrogen peroxide. Antioxid. Redox Signal. 2017, 27, 596–598. [Google Scholar] [CrossRef]
- Castillo-Quan, J.I.; Li, L.; Kinghorn, K.J.; Ivanov, D.K.; Tain, L.S.; Slack, C.; Kerr, F.; Nespital, T.; Thornton, J.; Hardy, J.; et al. Lithium promotes longevity through GSK3/NRF2-dependent hormesis. Cell Rep. 2016, 15, 638–650. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pennisi, M.; Crupi, R.; Di Paola, R.; Ontario, M.L.; Bella, R.; Calabrese, E.J.; Crea, R.; Cuzzocrea, S.; Calabrese, V. Inflammasomes, hormesis, and antioxidants in neuroinflammation: Role of NRLP3 in Alzheimer disease. J. Neurosci. Res. 2017, 95, 1360–1372. [Google Scholar] [CrossRef]
- Huang, X.; Li, J.; Song, S.; Wang, L.; Lin, Z.; Ouyang, Z.; Yu, R. Hormesis effect of hydrogen peroxide on the promoter activity of neuropeptide receptor PAC1-R. J. Food Biochem. 2019, 43, e12877. [Google Scholar] [CrossRef]
- Scuto, M.C.; Mancuso, C.; Tomasello, B.; Ontario, M.L.; Cavallaro, A.; Frasca, F.; Maiolino, L.; Salinaro, A.T.; Calabrese, E.J.; Calabrese, V. Curcumin, hormesis and the nervous system. Nutrients 2019, 11, 2417. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rainey, N.E.; Moustapha, A.; Petit, P.X. Curcumin, a multifaceted hormetic agent, mediates an intricate crosstalk between mitochondrial turnover, autophagy, and apoptosis. Oxid. Med. Cell. Longev. 2020, 2020, 3656419. [Google Scholar] [CrossRef] [PubMed]
- Calabrese, E.J. Hormesis and Ginseng: Ginseng mixtures and individual constituents commonly display hormesis dose responses, especially for neuroprotective effects. Molecules 2020, 25, 2719. [Google Scholar] [CrossRef]
- Gòmez-Sierra, T.; Medina-Campos, O.N.; Solano, J.D.; Ibarro-Rubio, M.E.; Pedraza-Chaverri, J. Isoliquiritigenin pretreatment induces endoplasmatic reticulum stress-mediated hormesis and attenuates cisplatin-induced oxidative stress and damage in LLC-PK1 cells. Molecules 2020, 25, 4442. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Hanekamp, J.C.; Hanekamp, Y.N.; Kapoor, R.; Dhawan, G.; Agathokleous, E. Chloroquine commonly induces hormetic dose responses. Sci. Total Environ. 2020, 755 Pt 1, 142436. [Google Scholar] [CrossRef]
- Calabrese, E.J.; Tsatsakis, A.; Agathokleous, E.; Giordano, J.; Calabrese, V. Does green tea induce hormesis? Dose-Response 2020, 18. [Google Scholar] [CrossRef]
- Antonnuci, S.; Mulvey, J.F.; Burger, N.; Di Sante, M.; Hall, A.R.; Hinchy, E.C.; Caldwell, S.T.; Gruszczyk, A.V.; Deshwal, S.; Hartley, R.C.; et al. Selective mitochondrial superoxide generation in vivo is cardioprotective through hormesis. Free Radic. Biol. Med. 2019, 134, 678–687. [Google Scholar] [CrossRef]
- Fernandez-Ortiz, M.; Sayed, R.K.A.; Fernandez-Martinez, J.; Cionfrini, A.; Aranda-Martinez, P.; Escames, G.; de Haro, T.; Acuna-Castroviejo, D. Melatonin/Nrf2/NLRP3 connection in mouse heart mitochondria during aging. Antioxidants 2020, 9, 1187. [Google Scholar] [CrossRef] [PubMed]
- Keller, A.; Hull, S.E.; Elajaili, H.; Johnston, A.; Knaub, L.A.; Chun, J.H.; Walker, L.; Nozik-Grayck, E.; Reusch, J.E.B. (-)-Epicatechin modulates mitochondrial redox in vascular cell models of oxidative stress. Oxid. Med. Cell. Longev. 2020, 2020, 6392629. [Google Scholar] [CrossRef] [PubMed]
- Mattson, M.P.; Moehl, K.; Ghena, N.; Schmaedick, M.; Cheng, A. Intermittent metabolic switching, neuroplasticity and brain health. Nat. Rev. Neurosci. 2018, 19, 63–80. [Google Scholar] [CrossRef]
- Zhu, X.; Wei, Y.; Yang, B.; Yin, X.; Guo, X. The mitohormetic response a spart of the cytoprotection mechanism of berberine. Mol. Med. 2020, 26, 10. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, H.M.; White, D.M.; Kraig, R.P. Strategies for study of neuroprotection from cold-preconditioning. J. Vis. Exp. 2010, 43, e2192. [Google Scholar] [CrossRef] [Green Version]
- Rojas, J.C.; Gonzales-Lima, F. Neurological and psychological applications of transcranial lasers and LEDs. Biochem. Pharmacol. 2013, 86, 447–457. [Google Scholar] [CrossRef] [PubMed]
- Abdel Meguid, M.H.; Hamad, Y.H.; Swilam, R.S.; Barakat, M.S. Relation of interleukin-6 in rheumatoid arthritis patients to systemic bone loss and structural bone damage. Rheumatol. Int. 2013, 33, 697–703. [Google Scholar] [CrossRef]
- Tago, F.; Tsukimoto, M.; Nakatsukasa, H.; Kojima, S. Repeated 0.5-Gy gamma irradiation attenuates autoimmune disease in MRL-lpr/lpr mice with suppression of CD3+CD4−CD8−B220+ t-cell proliferation and with up-regulation of CD4+CD25+Foxp3+ regulatory T cells. Radiat. Res. 2008, 169, 59–66. [Google Scholar] [CrossRef]
- Artukovic, M.; Ikic, M.; Kustelega, J.; Artukovic, I.N.; Kaliterna, D.M. Influence of UV radiation on immunological system and occurence of autoimmune diseases. Coll. Antropol. 2010, 34, 175–178. [Google Scholar]
- Nakatsukasa, H.; Tsukimoto, M.; Tokunaga, A.; Kojima, S. Repeated gamma irradiation attenuates collagen-induced arthritis via up-regulation of regulatory T cells but not by damaging lymphocytes directly. Radiat. Res. 2010, 174, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Dhawan, G.; Kapoor, R.; Dhawan, R.; Singh, R.; Monga, B.; Giordano, J.; Calabrese, E.J. Low dose radiation therapy as a potential life saving treatment for COVID-19-induced acute respiratory distress syndrome (ARDS). Radiother. Oncol. 2020, 147, 212–216. [Google Scholar] [CrossRef]
- Li, G.; Fan, Y.; Lai, Y.; Han, T.; Li, Z.; Zhou, P.; Pan, P.; Wang, W.; Hu, D.; Liu, X.; et al. Coronavirus infections and immune responses. J. Med. Virol. 2020, 92, 424. [Google Scholar] [CrossRef]
- Prompetchara, E.; Ketloy, C.; Palaga, T. Immune responses in COVID-19 and potential vaccines: Lessons learned from SARS and MERS epidemic. Asian Pac. J. Allergy Immunol. 2020, 38, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Fung, S.; Yuen, K.; Ye, Z.; Chan, C.; Jin, D. A tug-of-war between severe acute respiratory syndrome coronavirus 2 and host antiviral defence: Lessons from other pathogenic viruses. Emerg. Microbes Infect. 2020, 9, 558–570. [Google Scholar] [CrossRef] [PubMed]
- Kory, P.; Meduri, G.U.; Iglesias, J.; Varon, J.; Marik, P.E. Clinical and scientific rationale for the “MATH+” hospital treatment protocol for COVID-19. J. Intensiv. Care Med. 2020, 36, 135–156. [Google Scholar] [CrossRef]
- Kory, P.; Meduri, G.U.; Iglesias, J.; Varon, J.; Berkowitz, K.; Kornfeld, H.; Vinjevoll, E.; Mitchell, S.; Wagshul, F.; Marik, P.E. Review of the Emerging Evidence Demonstrating the Efficacy of Ivermectin in the Prophylaxis and Treatment of COVID-19. Front Line COVID-19 CRITICAL CARE ALLIANCE. Prophylaxis & Treatment Protocols for COVID-19. 2020. Available online: https://covid19criticalcare.com/wp-content/uploads/2020/11/FLCCC-Ivermectin-in-the-prophylaxis-and-treatment-of-COVID-19.pdf (accessed on 18 December 2020).
- Jacob, R.S.; de Souza Santos, L.V.; d’Auriol, M.; Lebron, Y.A.R.; Moreira, V.R.; Lange, L.C. Diazepam, metformin, omeprazole and simvastatin: A full discussion of individual and mixture acute toxicity. Ecotoxicology 2020, 29, 1062–1071. [Google Scholar] [CrossRef] [PubMed]
- Fisher, B. Biological research in the evolution of cancer surgery: A personal perspective. Cancer Res. 2008, 68, 10007–10020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lowenbraun, S.; DeVita, V.T.; Serpick, A.A. Combination chemotherapy with nitrogen mustard, vincristine, procarbazine and prednisone in lymphosarcoma and reticulum cell sarcoma. Cancer 1970, 25, 1018–1025. [Google Scholar] [CrossRef]
- Schirrmacher, V. Quo Vadis Cancer Therapy? Lambert Academic Publishing: Beau-Bassin, Mauritius, 2017; pp. 1–353. [Google Scholar]
- Cardoso, F.; van’t Veer, L.J.; Bogaerts, J.; Slaets, L.; Viale, G.; Delaloge, S.; Pierga, J.-Y.; Brain, E.; Causeret, S.; DeLorenzi, M.; et al. 70-Gene signature as an aid to treatment decisions in early-stage breast cancer. N. Engl. J. Med. 2016, 375, 717–729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gaya, A.; Akle, C.A.; Mudan, S.; Grange, J. The concept of hormesis in cancer therapy—Is less more? Cureus 2015, 7, e261. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fan, H.; Wu, Y.; Yu, S.; Li, X.; Wang, A.; Wang, S.; Chen, W.; Lu, Y. Critical role of mTOR in regulating aerobic glycolysis in carcinogenesis (Review). Int. J. Oncol. 2020, 58, 9–19. [Google Scholar] [CrossRef] [PubMed]
- Feuerer, M.; Beckhove, P.; Garbi, N.; Mahnke, Y.; Limmer, A.; Hommel, M.; Hämmerling, G.J.; Kyewsky, B.; Hamann, A.; Umansky, V.; et al. Bone marrow as a priming site for T-cell responses tp blood-borne antigen. Nat. Med. 2003, 9, 1151–1157. [Google Scholar] [CrossRef] [PubMed]
- Schirrmacher, V. New insights into mechanisms of long-term protective anti-tumor immunity induced by cancer vaccines modified by virus infection. Biomedicines 2020, 8, 55. [Google Scholar] [CrossRef] [Green Version]
- Bai, L.; Beckhove, P.; Feuerer, M.; Umansky, V.; Choi, C.; Solomayer, F.S.; Diel, I.J.; Schirrmacher, V. Cognate interactions between memory T cells and tumor antigen-presenting dendritic cells from bone marrow of breast cancer patients: Bidirectional cell stimulation, survival and antitumor activity in vivo. Int. J. Cancer 2003, 103, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Cassel, W.A.; Garrett, R.E. Newcastle disease virus as an antineoplastic agent. Cancer 1965, 18, 863–868. [Google Scholar] [CrossRef]
- Heicappell, R.; Schirrmacher, V.; von Hoegen, P.; Ahlert, T.; Appelhans, B. Prevention of metastatic spread by postoperative immunotherapy with virally modified autologous tumor cells. I. Parameters for optimal therapeutic effects. Int. J. Cancer 1986, 37, 569–577. [Google Scholar] [CrossRef] [PubMed]
- Von Hoegen, P.; Zawatzky, R.; Schirrmacher, V. Modification of tumor cells by a low dose of Newcastle disease virus. III. Potentiation of tumor-specific cytolytic T cell activity via induction of interferon-alpha/beta. Cell. Immunol. 1990, 126, 80–90. [Google Scholar] [CrossRef] [PubMed]
- Schild, H.; von Hoegen, P.; Schirrmacher, V. Modification of tumor cells by a low dose of Newcastle disease virus. II. Augmented tumor-specific T cell response as a result of CD4+ and CD8+ immune T cell cooperation. Cancer Immunol. Immunother. 1989, 28, 22–28. [Google Scholar] [CrossRef] [PubMed]
- Von Hoegen, P.; Weber, E.; Schirrmacher, V. Modification of tumor cells by a low dose of Newcastle disease virus. I. Augmentation of the tumor-specific T cell response in the absence of an anti-viral response. Eur. J. Immunol. 1988, 18, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Schirrmacher, V.; Haas, C.; Bonifer, R.; Ahlert, T.; Gerhards, R.; Ertel, C. Human tumor cell modification by virus infection: An efficient and safe way to produce cancer vaccine with pleiotropic immune stimulatory properties when using Newcastle disease virus. Gene Ther. 1999, 6, 63–73. [Google Scholar] [CrossRef] [PubMed]
- Ahlert, T.; Sauerbrei, W.; Bastert, G.; Ruhland, S.; Bartik, B.; Simiantonaki, N.; Schumacher, J.; Häcker, B.; Schumacher, M.; Schirrmacher, V. Tumor cell number and viability as quality and efficacy parameters of autologous virus-modified cancer vaccines in patients with breast or ovarian cancer. J. Clin. Oncol. 1997, 15, 1354–1366. [Google Scholar] [CrossRef] [PubMed]
- Schirrmacher, V.; Lorenzen, D.; Van Gool, S.W.; Stuecker, W. A new strategy of cancer immunotherapy combining hyperthermia/oncolytic virus pretreatment with specific autologous anti-tumor vaccination—A review. Austin Oncol. Case Rep. 2017, 2, 1006. [Google Scholar]
- Van Gool, S.W.; Makalowski, J.; Feyen, O.; Prix, L.; Schirrmacher, V.; Stuecker, W. The induction of immunogenic cell death (ICD) during maintenance chemotherapy and susequent multimodal immunotherapy for glioblastoma (GBM). Austin Oncol. Case Rep. 2018, 3, 1010. [Google Scholar]
- Van Gool, S.W.; Makalowski, J.; Fiore, S.; Sprenger, T.; Prix, L.; Schirrmacher, V.; Stuecker, W. Randomized controlled immunotherapy clinical trials for GBM challenged. Cancers 2021, 13, 32. [Google Scholar] [CrossRef] [PubMed]
- Puhlmann, J.; Puehler, F.; Mumberg, D.; Boukamp, P.; Beier, R. Rac1 is required for oncolytic NDV replication in human cancer cells and establishes a link between tumorigenesis and sensitivity to oncolytic virus. Oncogene 2010, 29, 2205–2215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abdulla, J.M.; Mustafa, Z.; Ideris, A. Newcastle disease virus interaction in targeted therapy against proliferation and invasion pathways of glioblastoma multiforme. Biomed. Res. Int. 2014, 2014, 386470. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ren, S.; Rehman, Z.U.; Shi, M.; Yang, B.; Qu, Y.; Yang, X.F.; Shao, Q.; Meng, C.; Yang, Z.; Gao, X.; et al. Syncytia generated by hemagglutinin-neuraminidase and fusion protein of virulent Newcastle disease virus induce complete autophagy by activating AMPK-mTORC1-ULK1 signaling. Vet. Microbiol. 2019, 230, 283–290. [Google Scholar] [CrossRef]
- Ch’ng, W.C.; Stanbridge, E.J.; Yusoff, K.; Shafee, N. The oncolytic activity of Newcastle disease virus in clear cell renal carcinoma cells in normoxic and hypoxic conditions: The interplay between von Hippel-Lindau and interferon-β signaling. J. Interferon Cytokine Res. 2013, 33, 346–354. [Google Scholar] [CrossRef] [Green Version]
- Schirrmacher, V.; van Gool, S.; Stuecker, W. Breaking therapy resistance: An update on oncolytic Newcasle disease virus for improvements of cancer therapy. Biomedicines 2019, 7, 66. [Google Scholar] [CrossRef] [Green Version]
- Schirrmacher, V. Cancer vaccines and oncolytic viruses exert profoudly lower side effects in cancer patients than other systemic therapies: A comparative analysis. Biomedicines 2020, 8, 61. [Google Scholar] [CrossRef] [Green Version]
- Bai, L.; Koopmann, J.; Fiola, C.; Fournier, P.; Schirrmacher, V. Dendritic cells pulsed with viral oncolysates potently stimalate autologous T cells from cancer patients. Int. J. Oncol. 2002, 21, 685–694. [Google Scholar] [PubMed]
- Shevtsov, M.; Balogi, Z.; Khachatryan, W.; Gao, H.; Vigh, L.; Multhoff, G. Membrane- associated heat shock proteins in oncolgy: From basic research to new theranostic targets. Cells 2020, 9, 1263. [Google Scholar] [CrossRef]
- Camandona, V.L.; Rios-Anjos, R.M.; Alegria, T.G.P.; Pereira, F.; Bicev, R.N.; da Cunha, F.M.; Digiampietri, L.A.; de Barros, M.H.; Netto, L.E.S.; Ferreira-Junior, J.R. Expression of human HSP27 in yeast extends replicative lifespan and uncovers a hormetic response. Biogerontology 2020, 21, 559–575. [Google Scholar] [CrossRef]
- Kurd, N.; Robey, E.A. T-cell selection in the thymus. A spatial and temporal perspective. Immunol. Rev. 2016, 271, 114–126. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Melssen, M.; Slingluff, C.L. Vaccines targeting helper T cells for cancer immunotherapy. Curr. Opin. Immunol. 2017, 47, 85–92. [Google Scholar] [CrossRef] [PubMed]
- Hoerr, I.; Obst, R.; Rammensee, H.G.; Jung, G. In vivo application of RNA leads to induction of specific cytotoxic T lymphocytes and antibodies. Eur. J. Immunol. 2000, 30, 1–7. [Google Scholar] [CrossRef]
- Fournier, P.; Wilden, H.; Schirrmacher, V. Importance of retinoic acid-inducible gene I and of receptor for type I interferon for cellular resistance to infection by Newcastle disease virus. Int. J. Oncol. 2012, 40, 287–298. [Google Scholar] [CrossRef] [Green Version]
- Zaslawsky, E.; Hershberg, U.; Seto, J.; Pham, A.M.; Marques, S.; Duke, J.L.; Wetmur, J.G.; Tenoever, B.R.; Sealfon, S.C.; Kleinstein, S.H. Antiviral response dictated by choreographed cascade of transcription factors. J. Immunol. 2010, 184, 2908–2917. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schirrmacher, V.; Förg, P.; Dalemans, W.; Chlichlia, K.; Zeng, Y.; Fournier, P.; von Hoegen, P. Intra-pinna anti-tumor vaccination with self-replicating infectious RNA or with DNA encoding a model tumor antigen and a cytokine. Gene Ther. 2000, 7, 1137–1147. [Google Scholar] [CrossRef] [Green Version]
- Förg, P.; von Hoegen, P.; Dalemans, W.; Schirrmacher, V. Superiority of the ear pinna over muscle tissue as site for DNA vaccination. Gene Ther. 1998, 5, 789–797. [Google Scholar] [CrossRef] [PubMed]
- Toes, R.E.M.; Offringa, R.; Blom, R.J.J.; Melief, C.J.M.; Kast, W.M. Peptide vaccination can lead to enhanced tumor growth through specific T-cell tolerance induction. Proc. Natl. Acad. Sci. USA 1996, 93, 7855–7860. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rini, B.I.; Stenzl, A.; Zdrojowy, R.; Kogan, M.; Shkolnik, M.; Oudard, S.; Weikert, S.; Bracarda, S.J.; Crabb, S.; Bedke, J.; et al. IMA901, a multipeptide cancer vaccine, plus sunitinib versus sunitinib alone, as first-line therapy for advanced or metastatic renal cell carcinoma (IMPRINT): A multicentre, open-label, randomized, controlled, phase 3 trial. Lancet Oncol. 2016, 17, 1599–1611. [Google Scholar] [CrossRef]
- Nelde, A.; Rammensee, H.G.; Walz, J.S. The peptide vaccine of the future. Mol. Cell. Proteom. 2021, 20, 100022. [Google Scholar] [CrossRef]
- Zhao, T.; Du, J.; Zeng, H. Interplay between endoplasmic reticulum stress and non-coding RNAs in cancer. J. Hematol. Oncol. 2020, 13, 163. [Google Scholar] [CrossRef] [PubMed]
- Kenny, T.C.; Gomez, M.; Germain, D. Mitohormesis, UPRmt, and the complexity of mitochondrial DNA landscapes in cancer. Cancer Res. 2019, 79, 6057–6066. [Google Scholar] [CrossRef] [Green Version]
- Nokin, M.; Dutieux, F.; Bellier, J.; Peulen, O.; Uchida, K.; Spiegel, D.A.; Cochrane, J.R.; Hutton, C.A.; Castronovo, V.; Bellahcene, A. Hormetic potential of methylglyoxal, a side-product of glycolysis, in switching tumours from growth to death. Sci. Rep. 2017, 7, 11722. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calvani, M.; Subbiani, A.; Vignoli, M.; Favre, C. Spotlight on ROS and ß3-adrenoreceptors fighting in cancer cells. Oxid. Med. Cell. Longev. 2019, 2019, 6346529. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Werner, L.; Dreyer, J.H.; Hartmann, D.; Barros, M.H.M.; Büttner-Herold, M.; Grittner, U.; Niedobitek, G. Tumor-associated macrophages in classical Hodgkin lymphoma: Hormetic relationship to outcome. Sci. Rep. 2020, 10, 9410. [Google Scholar] [CrossRef]
- Oshi, M.; Angarita, F.A.; Tokumaru, Y.; Yan, L.; Matsuyama, R.; Endo, I.; Takabe, K. High expression of Nrf2 is associated with increased tumor-infiltrating lymphocytes and cancer immunity in ER-positive/HER2-negative breast cancer. Cancer 2020, 12, 3856. [Google Scholar] [CrossRef] [PubMed]
- Rocha, C.R.R.; Rocha, A.R.; Silva, M.M.; Gomes, L.R.; Latancia, M.T.; Tomaz, M.A.; de Souza, I.; Monteiro, L.K.S.; Menck, C.F.M. Revealing temozolomide resistance mechanisms via genome-wide CRISPR libraries. Cells 2020, 9, 2573. [Google Scholar] [CrossRef]
- Angeli, J.P.F.; Meierjohann, S. NRF2 dependent stress defence in tumor antioxidant control and immune evasion. Pigment Cell Melanoma Res. 2020, 34, 268–279. [Google Scholar] [CrossRef]
- Kojima, S.; Tsukimoto, M.; Shimura, N.; Koga, H.; Murata, A.; Takara, T. Treatment of cancer and inflammation with low-dose ionizing radiation: Three case reports. Dose-Response 2017, 15, 1–7. [Google Scholar] [CrossRef]
- Kojima, S.; Cuttler, J.M.; Inoguchi, K.; Yorozu, K.; Horii, T.; Shimura, N.; Koga, H.; Murata, A. Radon therapy is very promising as a primary or an adjuvant treatment for different types of cancers: 4 case reports. Dose-Response 2019, 17, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Malkowski, E.; Sitka, K.; Szopinski, M.; Gieron, Z.; Pogrzeba, M.; Kalaji, H.M.; Zieleznik-Rusinowska, P. Hormesis in plants: The role of oxidative stress, auxins and photosynthesis in corn treated with Cd or Pb. Int. J. Mol. Sci. 2020, 21, 2099. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sillen, W.M.A.; Thijs, S.; Abbamondi, G.R.; De La Torre Roche, R.; Weyens, N.; White, J.C.; Vangronsveld, J. Nanoparticle treatment of maize analyzed through the metatranscriptome: Compromised nitrogen cycling, possible phytopathogen selection, and plant hormesis. Microbiome 2020, 8, 127. [Google Scholar] [CrossRef] [PubMed]
- Jalai, A.; de Oliveira, J.C., Jr.; Ribeiro, J.S.; Fernandez, G.C.; Mariano, G.G.; Trindade, V.D.R.; Dos Reis, A.R. Hormesis in plants. Physiological and biochemical responses. Ecotoxicol. Environ. Saf. 2021, 207, 111225. [Google Scholar] [CrossRef]
- Checa, J.; Aran, J.M. Reactive oxygen species: Drivers of physiological and pathological processes. J. Inflamm. Res. 2020, 13, 1057–1073. [Google Scholar] [CrossRef]
- Trejo-Tellez, L.I.; Garcia-Jimenez, A.; Escobar-Sepulveda, H.F.; Ramirez-Olvera, S.M.; Bello-Bello, J.J.; Gomez-Merino, F.C. Silicon induces hormetic dose-response effects on growth and concentrations of chlorophylls, amino acids and sugars in pepper plants during early developmental stage. Peer J. 2020, 8, e9224. [Google Scholar] [CrossRef] [PubMed]
- Hackley, R.K.; Schmid, A.K. Global transcriptional programs in archaea share features with eukaryotic environmental stress response. J. Mol. Med. 2019, 431, 4147–4166. [Google Scholar] [CrossRef]
- Sprang, A.; Saw, J.H.; Jorgensen, S.L.; Zaremba-Niedz-wiedzka, K.; Matijn, J.; Lind, A.E.; van Eijk, R.; Schleper, C.; Guy, L.; Ettema, T.J.G. Complex archaea that bridge the gap between prpkaryocytes and eukaryocytes. Nature 2015, 521, 173–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agathokleous, E.; Kitao, M.; Calabrese, E.J. New insights into the role of melatonin in plants and animals. Chem. Biol. Interact. 2019, 299, 163–167. [Google Scholar] [CrossRef] [PubMed]
- Constantini, D. Hormesis promotes evolutionary change. Dose-Response 2019, 17. [Google Scholar] [CrossRef] [Green Version]
- Chirumbolo, S.; Bjorklund, G. PERM hypothesis: The fundamental machinery able to elucidate the role of xenobiotics and hormesis in cell survival and homeostasis. Int. J. Mol. Sci. 2017, 18, 165. [Google Scholar] [CrossRef]
- Rossnerova, A.; Izzotti, A.; Pulliero, A.; Bast, A.; Rattan, S.I.S.; Rossner, P. The molecular mechanisms of adaptive response related to environmental stress. Int. J. Mol. Sci. 2020, 21, 7053. [Google Scholar] [CrossRef] [PubMed]
- Agathokleous, E.; Kitao, M.; Calabrese, E.J. Environmental hormesis and ist fundamental biological basis: Rewriting the history of toxicology. Environ. Res. 2018, 165, 274–278. [Google Scholar] [CrossRef] [PubMed]
- Perego, M.; Tyurin, V.A.; Tyurina, Y.Y.; Yellets, J.; Nacarelli, T.; Lin, C.; Nefedova, Y.; Kossenkov, A.; Liu, Q.; Sreedhar, S.; et al. Reactivation of dormant tumor cells by modified lipids derived from stress-actvated neutrophiles. Sci. Transl. Med. 2020, 12, eabb5817. [Google Scholar] [CrossRef]
- Rodriguez-Cano, A.M.; Caldaza-Mendoza, C.C.; Estrada-Gutierrez, G.; Mendoza-Ortega, J.A.; Perichart-Perera, O. Nutrients, mitochondrial function, and perinatal health. Nutrients 2020, 72, 2166. [Google Scholar] [CrossRef]
- Wu, Y.; Song, J.; Wang, Y.; Wang, X.; Culmsee, C.; Zhu, C. The potential role of ferroptosis in neonatal brain injury. Front. Neurosci. 2019, 13, 115. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Babich, R.; Hamlin, H.; Thayer, L.; Dorr, M.; Wie, Z.; Neilson, A.; Jayasundara, N. Mitochondrial response and resilience to anthropogenic chemicals during embryonic development. Comp. Biochem. Physiol. C Toxicol. Pharmacol. 2020, 233, 108759. [Google Scholar] [CrossRef]
- Dietert, R.R.; Piepenbrink, M.S. The managed immune system: Protecting the womb to delay the tomb. Hum. Exp. Toxicol. 2008, 27, 129–134. [Google Scholar] [CrossRef]
- Alloo, F.; Arizpe, L.; Belford, T.S.; Clark, J.; Csanadi, J.; Davies, P.J.; George, S.; Hyden, G.; Jonsson, U. The Bellagio Declaration: Overcoming hunger in the 1990’s. Dev. Dialogue 1989, 2, 177–184. [Google Scholar] [PubMed]
- Elvevoll, E.O.; James, D. The emerging importance of dietary lipids, quantity and quality, in the global disease burden: The potential of aquatic resources. Nutr. Health 2001, 15, 155–167. [Google Scholar] [CrossRef]
- Kerac, M.; Blencowe, H.; Grijalva-Eternod, C.; McGrath, M.; Shoham, J.; Cole, T.J.; Seal, A. Prevalence of wasting among under 6-month-old infants in developing countries and implications of new case definitions using WHO growth standards: A secondary data analysis. Arch. Dis. Child. 2011, 96, 1008–1013. [Google Scholar] [CrossRef] [Green Version]
- Hill, Y.; Kiefer, A.W.; Silva, P.L.; Van Yperen, N.W.; Meijer, R.R.; Fischer, N.; Den Hartigh, R.J.R. Antifragility in climbing: Determining optimal stress loads for athletic performance training. Front. Psychol. 2020, 11, 272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Agathokleous, E.; Calabrese, E.J. Environmental toxicology and ecotoxicology: How clean is clean? Rethinking dose-response analysis. Sci. Total Environ. 2020, 746, 138769. [Google Scholar] [CrossRef] [PubMed]
- Belz, R.G.; Duke, S.O. Herbicides and plant hormesis. Pest Manag. Sci. 2014, 70, 698–707. [Google Scholar] [CrossRef]
- Zhang, Y.; Calabrese, E.J.; Zhang, J.; Gao, D.; Qin, M.; Lin, Z. A trigger mechanism of herbicides to phytoplankton blooms: From the standpoint of hormesis involving cytochrome b559, reactive oxygen species and nitric oxide. Water Res. 2020, 173, 115584. [Google Scholar] [CrossRef]
- Kino, K. The prospective mathematical idea satisfying both radiation hormesis under low radiation doses and linear non-threshold theory under high radiation doses. Genes Environ. 2020, 42, 4. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Devic, C.; Ferlazzo, M.L.; Berthel, E.; Foray, N. Influence of individual radiosensitivity on the hormesis phenomenon: Toward a mechanistic explanation based on the nucleoshutting of ATM protein. Dose-Response 2020, 18. [Google Scholar] [CrossRef] [PubMed]
| Stress Feature | Inducer/Modulator/Target I/M/T | Mol Mechanism | Effect |
|---|---|---|---|
| Oxidative stress | Glutathion system (M) | TF Nrf2 | Homeostasis |
| Chemodefence | Metals, genotoxics (I) | inducibility | Protection |
| pH | Sulfonamides (I) QS luxR (M) | Adenyl cyclase | Energy |
| UV light | Monoterpens (M) | 4NQO | UV protection |
| Radiation | Luminous marine bacteria (T) | 3 response levels | Adaptive response |
| RCS and ROS | PQS of yeast (M) | Mithormesis, glycohormesis | Protection |
| Fasting | Unicellular to multicellular transformation by Dictyostelium (M) | Polyketide differentiation Inducing factor 1 | Reproduction cycle |
| Fasting | Marine snails (T) Caenorhabditis (worm) (T) | Autophagy SNK-1/Nrf | Reduction of lipofuscin Epigenetic memory |
| Stressor | Response (Part A) | Response (Part B) | Effect |
|---|---|---|---|
| ROS, ER | 1. Nrf2 phosphorylation and release from Keap complex 2. Nrf2-P translocation to nucleus | 3. Heterodimerization with cMaf, 4. Binding to ARE 5. Transcription of HO-1, NQO1, GCLM | Anti-oxidation |
| TLR | 1. NFκB phosphorylation and release from IKK complex 2. Translocation of NFκB to nucleus 3. Induction of proinflammatory cytokines | 4. Induction of HO-1 expression via Nrf2 5. Inhibition of NFκB activation via Nrf2 6. Blocking degradation of IkB-a 7. Degradation of NFκB via Nrf2 8. Inhibition of nuclear translocation via Nrf2 | Anti-inflammation |
| Stressor | Sensor/Modulator/Target S/M/T | Mol Mechanism | Effect |
|---|---|---|---|
| LDR | NK cells (S) | p38/MAPK | cytotoxicity |
| LDR | Macrophages, M1 (S) | iNOS, oxidative burst | Orchestration of T cell immunotherapy |
| LDR | CD4 and CD8 T cells (S) T regulatory cells (S) | p38/MAPK, NFκB, JNK IL-10 down | Cytokine secretion, CTL activity downregulation |
| LDR | B cells (S) | NFκB, CD23 | OXPHOS shift to aerobic glycosylation |
| Fungus spore toxin | Drosophila (T) | Increased longevity and fecundity; decreased immune function | |
| Biological threats, infection by microbes | Macrophages (S) | M1/M2 shift | Tissue protection |
| Transient dietary restriction (DR) | Memory T cells (M), conservation in bone marrow | CXCR4/CXCL12 adipogenesis | Enhanced protective function |
| Stressor | Syndrome/Modulator/ Target S/M/T | Mol Mechanism | Effect |
|---|---|---|---|
| Toxic compound Li | Psychiatry (S) | GSK-3, Nrf-2 | Stress resistance Longevity |
| Ag-Nanoparticles (Ag-NPs) | Astroglioma cells (T) | MuD and p38/ERK | Beneficial |
| Formaldehyde | Bronchial epithelial cells (T) | CyclinD-cdk4, E2F1 | Warburg effect |
| LDR | H2O2 signaling (M) | Nrf2/Keap1, NFkB | Redox signaling |
| ROS | Neurodegenerative disorders (S), Curcumin (M) Inflammasomes (T) | Mitochondria, autophagy, apoptosis | Protection |
| LDR | Autoimmune diseases (S) | Upregulation of Treg Inhibition of cytokines | Regulation of negative effects |
| H2O2 | NLRP3 inflammasome (T) | PAC1-R | Neuroprotection, neurotrophic and neurogenesis effects |
| ROS | Cardiovascular diseases (S), MitoPQ (M) | Ca2+ homeostasis, mitochondrial homeostasis | Cardioprotection |
| ROS | Vascular cells (T), EPICAT (M) | Vasodilation | Mitochondrial redox regulation |
| Feature | Inducer/Modulator/Target I/M/T | Mol Mechanism | Effect |
|---|---|---|---|
| Small molecule inhibitor (SMI) | mTOR (T): Aerobic glycolysis, Truncated TCA cycle, MG production (M) | Metabolism of glucose, amino acids, fatty acids, lipids, nucleotides | Targeted inhibition by SMIs of carcinoma growth, MG as hormetin |
| Oncolytic virus | NDV (I): low-dose optimum for oncolysis, CTL induction and DTH reactivity | HSP27 phosphorylation, proteasomal protein degradation | Oncolysis, Immunogenic cell death (ICD), immune stimulation |
| SR59230A | ß3-adrenoreceptor (M) | Increase of ROS and cancer cell death | Hormetic low-dose anti-cancer effect |
| Tumor infiltrating macrophage | Hodgkin lymphoma (T) | CD68+, CD163 | Intermediate numbers associated with better prognosis |
| LDR | Cancer and ulceratice colitis (T) | Radiation hormesis | Three case reports of positve effects |
| Radon | Cancer (T), primary or adjuvant treatment | Radiation hormesis | Four case reports of positive effects |
| Herbicid | Modulator/Target M/T | Mol Mechanism | Effect |
|---|---|---|---|
| Metal: Cd or Pb | ROS (M) | Increase in auxin andflavonol | Hormetic stimulation of shoot growth |
| Metal: Ag-NP | Maize (T) | Positive effect on plants roots | Negative effect on rhizome |
| Glyphosate, 2,4-D, Paraquat | ROS (M) | H2O2 as signaling molecule | Increased water transport causing cell expansion |
| Silicon (Si) | Si accumulators: rize, wheat, barley, sugarcane, soybean, sugarbeet (T) | Si binding to hydroxyl groups of proteins involved in signaling | Hormetic effect on growth, chlorophyll, amino acids and sugars |
| Environmental Stressor | Species/Genes/Transcription | Response Criterium |
|---|---|---|
| Heat shock 25 °C to 37 °C | Halobacterium salinarum iESR: 724 genes rESR: 276 genes | 1. Global, stereotypical transcripttional reprogramming |
| Heat shock 25 °C to 37 °C | H. salinarum Repression of genes involved in ribosome biosynthesis and translation | 2. Induced and repressed genes enriched for distinct functions |
| Paraquat Redox cycling agent | H. salinarum 4 mM caused higher response than 0.25 mM | 3. Duration and magnitude of the transcriptional response dendent on intensity of stress |
| Reciprocal environmental shift 37 °C to 25 °C | H. salinarum Rapid recovery from stress without ESR-like transcriptional characteristics | 4. Induction of the transcriptional response specific to stress exposure |
| Stress Type | Example | Mol Mechanism | Effect |
|---|---|---|---|
| Physical | Temperature (heat, frost), radiation, exercise | Nrf2, glutathion DNA methylation, microRNA | Cell protection Apoptosis, autophagy, cell cycle regulation, DNA repair and turnover |
| Chemical | ROS, Li, Si, Ag, Cd, Pb Monoterpenes Methylglyoxal | Proteasome, endoplasmic reticulum, PQS, mitochondria | Cell survival Epigenetic memory Evolutionary flexibility |
| Biochemical | Dietary restriction, Pharmacological drugs | Glucose-ketone switch PAC1-R | Metabolic switching Neuroprotection |
| Biologic | Hormone: melatonin Oncolytic virus: NDV | Calcium HSP27, Type I IFN | Adaptation to circardian rhythm Oncolysis, Immunogenic cell death |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schirrmacher, V. Less Can Be More: The Hormesis Theory of Stress Adaptation in the Global Biosphere and Its Implications. Biomedicines 2021, 9, 293. https://doi.org/10.3390/biomedicines9030293
Schirrmacher V. Less Can Be More: The Hormesis Theory of Stress Adaptation in the Global Biosphere and Its Implications. Biomedicines. 2021; 9(3):293. https://doi.org/10.3390/biomedicines9030293
Chicago/Turabian StyleSchirrmacher, Volker. 2021. "Less Can Be More: The Hormesis Theory of Stress Adaptation in the Global Biosphere and Its Implications" Biomedicines 9, no. 3: 293. https://doi.org/10.3390/biomedicines9030293
APA StyleSchirrmacher, V. (2021). Less Can Be More: The Hormesis Theory of Stress Adaptation in the Global Biosphere and Its Implications. Biomedicines, 9(3), 293. https://doi.org/10.3390/biomedicines9030293






