NF-κB in Gastric Cancer Development and Therapy
Abstract
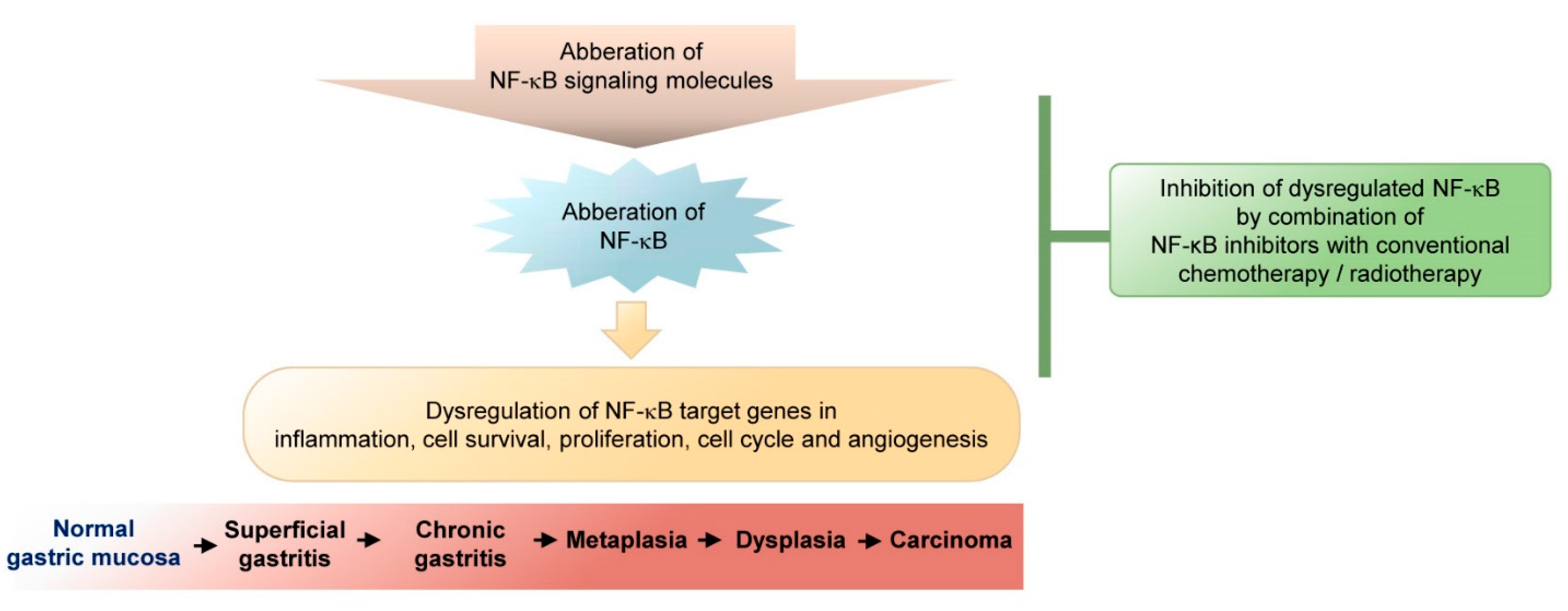
:1. Introduction
2. Dysregulation of NF-κB in Gastric Cancer
2.1. NF-κB Signaling
2.2. NF-κB Gene Polymorphisms
2.3. Gene Polymorphisms in NF-κB Signaling Molecules
2.4. Modulation of NF-κB Regulation in Gastric Cancer
3. NF-κB-Regulated Genes and Their Relevance for Gastric Cancer Development
3.1. Immune Response Mediators
3.2. iNOS and COX2
3.3. Effectors in Proliferation, Cell Cycle, Apoptosis, and Invasion
4. Therapeutic Targeting of NF-κB in Gastric Cancer
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Activator protein 1 | AP1 |
| 3,3’-diindolylmethane | DIM |
| ADP-glycero-β-D-manno-heptose | ADP-hep |
| Ataxia-telangiectasia mutated | ATM |
| B cell activation factor | BAF |
| B-cell lymphoma 2 | Bcl-2 |
| Bcl-2-associated X protein | Bax |
| Beta-transducin repeat containing E3 ubiquitin protein ligase | β-TrCP |
| Cadherin-1 | CDH1 |
| Cag pathogenicity island | CagPAI |
| Cannabinoid receptor 1 | CNR1 |
| Carcinoembryonic antigen-related cell adhesion molecule 19 | CEACAM19 |
| Caspase-associated recruitment domains | CARDs |
| Chemokine (C-X-C motif) ligand 1 | CXCL1 |
| Chemokine (C-X-C motif) ligand 2 | CXCL2 |
| Connective tissue growth factor | CTGF |
| CXC chemokine receptor 4 | CXCR4 |
| CXC chemokine receptor 4 | CXCR4 |
| CXC motif chemokine ligand 11 | CXCL11 |
| Cyclin-dependent kinase 2 | CDK-2 |
| Cyclooxygenase-2 | COX-2 |
| Deacetylase sirtuin 1 | SIRT1 |
| DNA methyltransferase 3 | DNMT3 |
| DNA repair protein | Ku |
| Dopamine and cAMP-regulated phosphoprotein 32,000 Da | DARPP-32 |
| Epidermal growth factor receptor | EGRF |
| Epstein–Barr virus (EBV) latent membrane protein 2 | LMP2A |
| Fibroblast growth factor-inducible 14 | FN14 |
| Gastric adenocarcinomas | non-cardia GCs |
| Gastric cancer | GC |
| Gastric mucosa-associated lymphoid tissue | MALT |
| Gastroesophageal junction adenocarcinomas | cardia GC |
| Gastrokine 1 | GKN1 |
| Granulocyte-macrophage colony-stimulating factor | GM-CSF |
| Growth-regulated oncogene | GRO-α |
| Growth-regulated protein beta | GRO-β |
| Growth-regulated protein gamma | GRO-γ |
| HOX transcript antisense RNA | HOTAIR |
| Human epidermal growth factor receptor 2 | HER2 |
| Human telomerase reverse transcriptase | hTERT |
| Hypoxia inducible factor 1 alpha | HIF-1α |
| Immunohistochemistry | IHC |
| Immunohistochemistry analysis | ICH |
| Inducible nitric oxide synthase | iNOS, NOS2 |
| Inhibitor of growth 4 | ING4 |
| Interleukin 1b | IL-1b |
| IκB kinase | IKK |
| Lipopolysaccharide | LPS |
| Lymphotoxin β | LTβ |
| Lymphotoxin β | LTβ |
| Matrix metallopeptidase 9 | MMP9 |
| Mesenchymal stromal cells | MSCs |
| Mucosa-associated lymphoid tissue | MALT |
| Myeloid differentiation primary response 88 | MyD88 |
| NADPH oxidase 1 | NOX1 |
| NADPH oxidase organizer 1 | Noxo1 |
| NF-κB essential modulator | NEMO |
| NF-κB inducing kinase | NIK |
| NF-κB1 | p50 |
| NF-κB2 | p52 |
| Nitric oxide | NO |
| Non-cardia gastric adenocarcinoma | NCGC |
| Nonsteroidal anti-inflammatory drugs | NSAIDs |
| O6-methylguanin-DNA-methyltransferase | MGMT |
| Oncogenes latent membrane protein 1 | LMP1 |
| Oncoprotein metadherin | MTDH |
| Open reading frames | ORFs |
| p21-activated kinases | PAKs |
| Phosphatase and tensin homolog | PTEN |
| Phosphatase of regenerating liver-3 | PRL-3 |
| Poly r(C)-binding protein | PCBP |
| Programmed death 1 | PD-1 |
| Programmed-death ligand 1 | PD-L1 |
| Prostaglandin E2 | PGE2 |
| Protection of telomeres 1 | POT1 |
| Protein alpha-kinase 1 | ALPK1 |
| Reactive oxygen species | ROS |
| Receptor activator of NF-κB | RANK |
| Receptor interacting serine/threonine kinase 2 | RIPK2 |
| Rel homology domain | RHD |
| Repressor/activator protein 1 | RAP1 |
| Sex determining region Y (SRY)-box 2 | SOX2 |
| Signal transducers and activators of transcription 3 | STAT3 |
| Stress protein metallothionein 2A | MT2A |
| T regulatory | Treg |
| TNF-induced protein 3-interacting protein 1 | TNIP1 |
| Toll-like receptors | TLRs |
| TRAF-interacting protein with forkhead-associated domain | TIFA |
| Transcription factor nuclear factor kappa B | NF-κB |
| Transforming growth factor b kinase 1 | TAK1 |
| Trefoil factor 1 | TFF1 |
| Tumor necrosis factor receptor-associated factor | TRAF |
| Tumor necrosis factor | TNF |
| Tumor-associated macrophages | TAMs |
| Type IV secretion system | T4SS |
| Vascular endothelial growth factor | VEGF |
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef] [Green Version]
- Torre, L.A.; Siegel, R.L.; Ward, E.M.; Jemal, A. Global Cancer Incidence and Mortality Rates and Trends--An Update. Cancer Epidemiol. Biomark. Prev. 2015, 25, 16–27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Balakrishnan, M.; George, R.; Sharma, A.; Graham, D.Y. Changing Trends in Stomach Cancer Throughout the World. Curr. Gastroenterol. Rep. 2017, 19, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Waldum, H.L.; Fossmark, R. Types of Gastric Carcinomas. Int. J. Mol. Sci. 2018, 19, 4109. [Google Scholar] [CrossRef] [Green Version]
- Rawla, P.; Barsouk, A. Epidemiology of gastric cancer: Global trends, risk factors and prevention. Gastroenterol. Rev. 2019, 14, 26–38. [Google Scholar] [CrossRef]
- Poorolajal, J.; Moradi, L.; Mohammadi, Y.; Cheraghi, Z.; Gohari-Ensaf, F. Risk factors for stomach cancer: A systematic review and meta-analysis. Epidemiol. Health 2020, 42, e2020004. [Google Scholar] [CrossRef] [PubMed]
- Biological agents. A review of human carcinogens. IARC Monogr. Eval. Carcinog. Risks Hum. 2012, 100, 1–441. [Google Scholar]
- De Martel, C.; Georges, D.; Bray, F.; Ferlay, J.; Clifford, G.M. Global burden of cancer attributable to infections in 2018: A worldwide incidence analysis. Lancet Glob. Health 2020, 8, e180–e190. [Google Scholar] [CrossRef] [Green Version]
- Hooi, J.K.Y.; Lai, W.Y.; Ng, W.K.; Suen, M.M.; Underwood, F.E.; Tanyingoh, D.; Malfertheiner, P.; Graham, D.Y.; Wong, V.W.; Wu, J.C.; et al. Global Prevalence of Helicobacter pylori Infection: Systematic Review and Meta-Analysis. Gastroenterology 2017, 153, 420–429. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kotilea, K.; Bontems, P.; Touati, E. Epidemiology, Diagnosis and Risk Factors of Helicobacter pylori Infection. Adv. Exp. Med. Biol. 2019, 1149, 17–33. [Google Scholar] [CrossRef]
- Lordick, F.; Kang, Y.-K.; Chung, H.; Salman, P.; Oh, S.C.; Bodoky, G.; Kurteva, G.; Volovat, C.; Moiseyenko, V.; Gorbunova, V.; et al. Capecitabine and cisplatin with or without cetuximab for patients with previously untreated advanced gastric cancer (EXPAND): A randomised, open-label phase 3 trial. Lancet Oncol. 2013, 14, 490–499. [Google Scholar] [CrossRef]
- Charalampakis, N.; Economopoulou, P.; Kotsantis, I.; Tolia, M.; Schizas, D.; Liakakos, T.; Elimova, E.; Ajani, J.A.; Psyrri, A. Medical management of gastric cancer: A 2017 update. Cancer Med. 2017, 7, 123–133. [Google Scholar] [CrossRef] [Green Version]
- Choi, S.I.; Park, B.; Joo, J.; Kim, Y.-I.; Lee, J.Y.; Kim, C.G.; Choi, I.J.; Kook, M.-C.; Cho, S.-J. Three-year interval for endoscopic screening may reduce the mortality in patients with gastric cancer. Surg. Endosc. 2019, 33, 861–869. [Google Scholar] [CrossRef]
- Joharatnam-Hogan, N.; Shiu, K.K.; Khan, K. Challenges in the treatment of gastric cancer in the older patient. Cancer Treat. Rev. 2020, 85, 101980. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.-H.; Choi, Y.W.; Kang, S.Y.; Jeong, G.S.; Lee, H.W.; Jeong, S.H.; Park, J.S.; Ahn, M.S.; Sheen, S.S. Combination versus single-agent as palliative chemotherapy for gastric cancer. BMC Cancer 2020, 20, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taniguchi, K.; Karin, M. NF-κB, inflammation, immunity and cancer: Coming of age. Nat. Rev. Immunol. 2018, 18, 309–324. [Google Scholar] [CrossRef] [PubMed]
- Neumann, M.; Naumann, M. Beyond IκBs: Alternative regulation of NF-KB activity. FASEB J. 2007, 21, 2642–2654. [Google Scholar] [CrossRef] [PubMed]
- Kang, M.-J.; Ryu, B.; Lee, M.; Han, J.; Lee, J.; Ha, T.; Byun, D.; Chae, K.; Lee, B.; Chun, H.S.; et al. NF-κB Activates Transcription of the RNA-Binding Factor HuR, via PI3K-AKT Signaling, to Promote Gastric Tumorigenesis. Gastroenterology 2008, 135, 2030–2042.e3. [Google Scholar] [CrossRef]
- Strickertsson, J.A.B.; Desler, C.; Martin-Bertelsen, T.; Machado, A.M.D.; Wadstrøm, T.; Winther, O.; Rasmussen, L.J.; Friis-Hansen, L. Enterococcus faecalis Infection Causes Inflammation, Intracellular Oxphos-Independent ROS Production, and DNA Damage in Human Gastric Cancer Cells. PLoS ONE 2013, 8, e63147. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Choi, H.; Lee, S.K. TAX1BP1 downregulation by EBV-miR-BART15-3p enhances chemosensitivity of gastric cancer cells to 5-FU. Arch. Virol. 2017, 162, 369–377. [Google Scholar] [CrossRef] [PubMed]
- Dolcet, X.; Llobet, D.; Pallares, J.; Matias-Guiu, X. NF-kB in development and progression of human cancer. Virchows. Archiv. 2005, 446, 475–482. [Google Scholar] [CrossRef]
- Ooi, C.-H.; Ivanova, T.; Wu, J.; Lee, M.; Tan, I.; Tao, J.; Ward, L.; Koo, J.H.; Gopalakrishnan, V.; Zhu, Y.; et al. Oncogenic Pathway Combinations Predict Clinical Prognosis in Gastric Cancer. PLoS Genet. 2009, 5, e1000676. [Google Scholar] [CrossRef] [Green Version]
- Xu, D.; Li, J.; Li, R.-Y.; Lan, T.; Xiao, C.; Gong, P. PD-L1 Expression Is Regulated By NF-κB During EMT Signaling In Gastric Carcinoma. OncoTargets Ther. 2019, 12, 10099–10105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xiang, S.; Zhao, Z.; Zhang, T.; Zhang, B.; Meng, M.; Cao, Z.; Zhou, Q. Triptonide effectively suppresses gastric tumor growth and metastasis through inhibition of the oncogenic Notch1 and NF-κB signaling pathways. Toxicol. Appl. Pharmacol. 2020, 388, 114870. [Google Scholar] [CrossRef] [PubMed]
- Napetschnig, J.; Wu, H. Molecular Basis of NF-κB Signaling. Annu. Rev. Biophys. 2013, 42, 443–468. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pfannkuch, L.; Hurwitz, R.; Trauisen, J.; Sigulla, J.; Poeschke, M.; Matzner, L.; Kosma, P.; Schmid, M.; Meyer, T.F. ADP heptose, a novel pathogen-associated molecular pattern identified in Helicobacter pylori. FASEB J. 2019, 33, 9087–9099. [Google Scholar] [CrossRef] [Green Version]
- Zhou, P.; She, Y.; Dong, N.; Li, P.; He, H.; Borio, A.; Wu, Q.; Lu, S.; Ding, X.; Cao, Y.; et al. Alpha-kinase 1 is a cytosolic innate immune receptor for bacterial ADP-heptose. Nat. Cell Biol. 2018, 561, 122–126. [Google Scholar] [CrossRef]
- Sokolova, O.; Maubach, G.; Naumann, M. MEKK3 and TAK1 synergize to activate IKK complex in Helicobacter pylori infection. Biochim. Biophys. Acta BBA Bioenerg. 2014, 1843, 715–724. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cildir, G.; Low, K.C.; Tergaonkar, V. Noncanonical NF-κB Signaling in Health and Disease. Trends Mol. Med. 2016, 22, 414–429. [Google Scholar] [CrossRef] [PubMed]
- Maubach, G.; Feige, M.H.; Lim, M.C.; Naumann, M. NF-kappaB-inducing kinase in cancer. Biochim. Biophys. Acta BBA Bioenerg. 2019, 1871, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Feige, M.H.; Vieth, M.; Sokolova, O.; Täger, C.; Naumann, M. Helicobacter pylori induces direct activation of the lymphotoxin beta receptor and non-canonical nuclear factor-kappa B signaling. Biochim. Biophys. Acta BBA Bioenerg. 2018, 1865, 545–550. [Google Scholar] [CrossRef]
- Hua, T.; Qinsheng, W.; Xuxia, W.; Shuguang, Z.; Ming, Q.; Zhenxiong, L.; Jingjie, W. Nuclear Factor-Kappa B1 is Associated With Gastric Cancer in a Chinese Population. Medicine 2014, 93, e279. [Google Scholar] [CrossRef]
- Tuncay, S.S.; Okyay, P.; Bardakci, F. Identification of NF-κB1 and NF-κBIA Polymorphisms Using PCR–RFLP Assay in a Turkish Population. Biochem. Genet. 2009, 48, 104–112. [Google Scholar] [CrossRef]
- Arisawa, T.; Tahara, T.; Shiroeda, H.; Yamada, H.; Nomura, T.; Hayashi, R.; Saito, T.; Fukuyama, T.; Otsuka, T.; Nakamura, M.; et al. NFKB1 polymorphism is associated with age-related gene methylation in Helicobacter pylori-infected subjects. Int. J. Mol. Med. 2012, 30, 255–262. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lo, S.-S.; Chen, J.-H.; Wu, C.-W.; Lui, W.-Y. Functional polymorphism of NFKB1 promoter may correlate to the susceptibility of gastric cancer in aged patients. Surgery 2009, 145, 280–285. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.; Gao, X.; Chen, Y.; Ni, J.; Yu, Y.; Li, S.; Guo, L. Association of an NFKB1 intron SNP (rs4648068) with gastric cancer patients in the Han Chinese population. BMC Gastroenterol. 2012, 12, 87. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Lu, R.; Zheng, H.; Xiao, R.; Feng, J.; Wang, H.; Gao, X.; Guo, L. The NFKB1 polymorphism (rs4648068) is associated with the cell proliferation and motility in gastric cancer. BMC Gastroenterol. 2015, 15, 21. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- O’Reilly, L.A.; Putoczki, T.L.; Mielke, L.; Low, J.T.; Lin, A.; Preaudet, A.; Herold, M.; Yaprianto, K.; Tai, L.; Kueh, A.; et al. Loss of NF-κB1 Causes Gastric Cancer with Aberrant Inflammation and Expression of Immune Checkpoint Regulators in a STAT-1-Dependent Manner. Immunity 2018, 48, 570–583.e8. [Google Scholar] [CrossRef] [Green Version]
- Burkitt, M.D.; Williams, J.; Duckworth, C.; O’Hara, A.; Hanedi, A.; Varro, A.; Caamano, J.; Pritchard, D.M. Signaling mediated by the NF-κB sub-units NF-κB1, NF-κB2 and c-Rel differentially regulate Helicobacter felis-induced gastric carcinogenesis in C57BL/6 mice. Oncogene 2013, 32, 5563–5573. [Google Scholar] [CrossRef]
- Ishikawa, H.; Carrasco, D.; Claudio, E.; Ryseck, R.-P.; Bravo, R. Gastric Hyperplasia and Increased Proliferative Responses of Lymphocytes in Mice Lacking the COOH-terminal Ankyrin Domain of NF-κB2. J. Exp. Med. 1997, 186, 999–1014. [Google Scholar] [CrossRef] [Green Version]
- Wang, S.; Tian, L.; Zeng, Z.; Zhang, M.; Wu, K.; Chen, M.; Fan, D.; Hu, P.; Sung, J.J.; Yu, J. IκBα polymorphism at promoter region (rs2233408) influences the susceptibility of gastric cancer in Chinese. BMC Gastroenterol. 2010, 10, 15. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhang, M.; Zeng, Z.; Tian, L.; Wu, K.; Chu, J.; Fan, D.; Hu, P.; Sung, J.J.; Yu, J. IκBα polymorphisms were associated with increased risk of gastric cancer in a southern Chinese population: A case–control study. Life Sci. 2011, 88, 792–797. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Wu, C.; Cai, Y.; Liu, B. Association of NFKB1 and NFKBIA gene polymorphisms with susceptibility of gastric cancer. Tumor Biol. 2017, 39, 1010428317717107. [Google Scholar] [CrossRef] [Green Version]
- Gong, Y.; Zhao, W.; Jia, Q.; Dai, J.; Chen, N.; Chen, Y.; Gu, D.; Huo, X.; Chen, J. IKBKB rs2272736 is Associated with Gastric Cancer Survival. Pharm. Pers. Med. 2020, 13, 345–352. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Shi, Y.; Na, Y.; Zhang, Q.; Cao, S.; Duan, X.; Zhang, X.; Yang, H.; Jin, T.; Li, Y. Genetic polymorphisms in TNIP1 increase the risk of gastric carcinoma. Oncotarget 2016, 7, 40500–40507. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Luo, J.; Song, J.; Zhang, H.; Zhang, F.; Liu, H.; Li, L.; Zhang, Z.; Chen, L.; Zhang, M.; Lin, D.; et al. Melatonin mediated Foxp3-downregulation decreases cytokines production via the TLR2 and TLR4 pathways in H. pylori infected mice. Int. Immunopharmacol. 2018, 64, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Ngo, V.N.; Young, R.M.; Schmitz, R.; Jhavar, S.; Xiao, W.; Lim, K.-H.; Kohlhammer, H.; Xu, W.; Yang, Y.; Zhao, H.; et al. Oncogenically active MYD88 mutations in human lymphoma. Nature 2011, 470, 115–119. [Google Scholar] [CrossRef] [Green Version]
- Ota, M.; Tahara, T.; Otsuka, T.; Jing, W.; Nomura, T.; Hayashi, R.; Shimasaki, T.; Nakamura, M.; Shibata, T.; Arisawa, T. Association between receptor interacting serine/threonine kinase 2 polymorphisms and gastric cancer susceptibility. Oncol. Lett. 2018, 15, 3772–3778. [Google Scholar] [CrossRef]
- Ng, M.T.H.; Hof, R.V.T.; Crockett, J.C.; Hope, M.E.; Berry, S.; Thomson, J.; McLean, M.; McColl, K.E.L.; El-Omar, E.; Hold, G.L. Increase in NF-κB Binding Affinity of the Variant C Allele of the Toll-Like Receptor 9 −1237T/C Polymorphism Is Associated with Helicobacter pylori -Induced Gastric Disease. Infect. Immun. 2010, 78, 1345–1352. [Google Scholar] [CrossRef] [Green Version]
- Marta, Ż.-N.; Agnieszka, W.; Jacek, P.; Jeleń, A.; Adrian, K.; Dagmara, S.-K.; Sałagacka-Kubiak, A.; Balcerczak, E. NFKB2 gene expression in patients with peptic ulcer diseases and gastric cancer. Mol. Biol. Rep. 2020, 47, 2015–2021. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wan, H.-Y.; Guo, L.-M.; Liu, T.; Liu, M.; Li, X.; Tang, H. Regulation of the transcription factor NF-kappaB1 by microRNA-9 in human gastric adenocarcinoma. Mol. Cancer 2010, 9, 10–16. [Google Scholar] [CrossRef] [Green Version]
- Yue, Y.; Zhou, T.; Gao, Y.; Zhang, Z.; Li, L.; Liu, L.; Shi, W.; Su, L.; Cheng, B. High mobility group box 1/toll-like receptor 4/myeloid differentiation factor 88 signaling promotes progression of gastric cancer. Tumor Biol. 2017, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, Q.; Tian, S.; Liu, Z.; Dong, W. Knockdown of RIPK2 Inhibits Proliferation and Migration, and Induces Apoptosis via the NF-κB Signaling Pathway in Gastric Cancer. Front. Genet. 2021, 12, 627464. [Google Scholar] [CrossRef] [PubMed]
- Pimentel-Nunes, P.; Gonçalves, N.; Boal-Carvalho, I.; Afonso, L.; Lopes, P.; Jr, R.R.A.; Henrique, R.; Moreira-Dias, L.; Leite-Moreira, A.; Dinis-Ribeiro, M. Helicobacter pyloriInduces Increased Expression of Toll-Like Receptors and Decreased Toll-Interacting Protein in Gastric Mucosa that Persists Throughout Gastric Carcinogenesis. Helicobacter 2013, 18, 22–32. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.R.; Peng, J.C.; Qiao, Y.Q.; Zhu, M.M.; Zhao, D.; Shen, J.; Ran, Z.H. Helicobacter pylori regulates TLR4 and TLR9 during gastric carcinogenesis. Int. J. Clin. Exp. Pathol. 2014, 7, 6950–6955. [Google Scholar] [PubMed]
- Gong, Y.; Tao, L.; Jing, L.; Liu, D.; Hu, S.; Liu, W.; Zhou, N.; Xie, Y. Association of TLR4 and Treg in Helicobacter pylori Colonization and Inflammation in Mice. PLoS ONE 2016, 11, e0149629. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ye, H.-H.; Hua, R.; Yu, L.; Wu, K.-J.; Fei, S.-J.; Qin, X.; Song, Y.; Cao, J.-L.; Zhang, Y.-M. Abnormal Expression of Toll-Like Receptor 4 Is Associated with Susceptibility to Ethanol-Induced Gastric Mucosal Injury in Mice. Dig. Dis. Sci. 2013, 58, 2826–2839. [Google Scholar] [CrossRef]
- Sasaki, N.; Morisaki, T.; Hashizume, K.; Yao, T.; Tsuneyoshi, M.; Noshiro, H.; Nakamura, K.; Yamanaka, T.; Uchiyama, A.; Tanaka, M.; et al. Nuclear factor-kappaB p65 (RelA) transcription factor is constitutively activated in human gastric carcinoma tissue. Clin. Cancer Res. 2001, 7, 4136–4142. [Google Scholar]
- Huang, T.; Kang, W.; Zhang, B.; Wu, F.; Dong, Y.; Tong, J.H.M.; Yang, W.; Zhou, Y.; Zhang, L.; Cheng, A.S.L.; et al. miR-508-3p concordantly silences NFKB1 and RELA to inactivate canonical NF-κB signaling in gastric carcinogenesis. Mol. Cancer 2016, 15, 9. [Google Scholar] [CrossRef] [Green Version]
- Li, G.; Wang, Z.; Ye, J.; Zhang, X.; Wu, H.; Peng, J.; Song, W.; Chen, C.; Cai, S.; He, Y.; et al. Uncontrolled Inflammation Induced by AEG-1 Promotes Gastric Cancer and Poor Prognosis. Cancer Res. 2014, 74, 5541–5552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jiao, Y.; Yang, H.; Qian, J.; Gong, Y.; Liu, H.; Wu, S.; Cao, L.; Tang, L. miR-3664-5P suppresses the proliferation and metastasis of gastric cancer by attenuating the NF-κB signaling pathway through targeting MTDH. Int. J. Oncol. 2019, 54, 845–858. [Google Scholar] [CrossRef] [Green Version]
- Kim, S.S.; Ahn, C.H.; Kang, M.R.; Kim, Y.R.; Kim, H.S.; Yoo, N.J.; Lee, S.H. Expression of CARD6, an NF-κB activator, in gastric, colorectal and oesophageal cancers. Pathology 2010, 42, 50–53. [Google Scholar] [CrossRef]
- Zhang, C.; Tian, W.; Meng, L.; Qu, L.; Shou, C. PRL-3 promotes gastric cancer migration and invasion through a NF-κB-HIF-1α-miR-210 axis. J. Mol. Med. 2015, 94, 401–415. [Google Scholar] [CrossRef]
- Gong, Y.; Xiang, X.-J.; Feng, M.; Chen, J.; Fang, Z.-L.; Xiong, J.-P. CUL4A promotes cell invasion in gastric cancer by activating the NF-κB signaling pathway. Biol. Targets Ther. 2017, 11, 45–53. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Canedo, P.; Durães, C.; Pereira, F.P.; Regalo, G.; Lunet, N.; Barros, H.; Carneiro, F.; Seruca, R.; Rocha, J.; Machado, J.C. Tumor Necrosis Factor Alpha Extended Haplotypes and Risk of Gastric Carcinoma. Cancer Epidemiol. Biomark. Prev. 2008, 17, 2416–2420. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xing, R.; Cui, J.T.; Xia, N.; Lu, Y.Y. GKN1 inhibits cell invasion in gastric cancer by inactivating the NF-kappaB pathway. Discov. Med. 2015, 19, 65–71. [Google Scholar] [PubMed]
- Wang, Y.; Wu, H.; Wu, X.; Bian, Z.; Gao, Q. Interleukin 17A Promotes Gastric Cancer Invasiveness via NF-κB Mediated Matrix Metalloproteinases 2 and 9 Expression. PLoS ONE 2014, 9, e96678. [Google Scholar] [CrossRef]
- Lee, K.-A.; Park, J.H.; Sohn, T.-S.; Kim, S.; Rhee, J.-C.; Kim, J.-W. Interaction of polymorphisms in the Interleukin 1B-31 and general transcription factor 2A1 genes on the susceptibility to gastric cancer. Cytokine 2007, 38, 96–100. [Google Scholar] [CrossRef]
- Zhu, Y.-W.; Yan, J.-K.; Li, J.-J.; Ou, Y.-M.; Yang, Q. Knockdown of Radixin Suppresses Gastric Cancer Metastasis In Vitro by Up-Regulation of E-Cadherin via NF-κB/Snail Pathway. Cell. Physiol. Biochem. 2016, 39, 2509–2521. [Google Scholar] [CrossRef]
- Kwon, O.-H.; Park, S.-J.; Kang, T.-W.; Kim, M.; Kim, J.-H.; Noh, S.-M.; Song, K.-S.; Yoo, H.-S.; Wang, Y.; Pocalyko, D.; et al. Elevated fibroblast growth factor-inducible 14 expression promotes gastric cancer growth via nuclear factor-κB and is associated with poor patient outcome. Cancer Lett. 2012, 314, 73–81. [Google Scholar] [CrossRef] [PubMed]
- Li, S.; Fan, T.; Liu, H.; Chen, J.; Qin, C.; Ren, X. Tumor suppressor ING4 overexpression contributes to proliferation and invasion inhibition in gastric carcinoma by suppressing the NF-κB signaling pathway. Mol. Biol. Rep. 2013, 40, 5723–5732. [Google Scholar] [CrossRef] [PubMed]
- Soutto, M.; Belkhiri, A.; Piazuelo, M.B.; Schneider, B.; Peng, D.; Jiang, A.; Washington, M.K.; Kokoye, Y.; Crowe, S.E.; Zaika, A.; et al. Loss of TFF1 is associated with activation of NF-κB–mediated inflammation and gastric neoplasia in mice and humans. J. Clin. Investig. 2011, 121, 1753–1767. [Google Scholar] [CrossRef]
- Mao, Z.; Ma, X.; Rong, Y.; Cui, L.; Wang, X.; Wu, W.; Zhang, J.; Jin, D. Connective tissue growth factor enhances the migration of gastric cancer through downregulation of E-cadherin via the NF-κB pathway. Cancer Sci. 2010, 102, 104–110. [Google Scholar] [CrossRef]
- Zhao, H.; Xu, J.; Wang, Y.; Jiang, R.; Li, X.; Zhang, L.; Che, Y. Knockdown of CEACAM19 suppresses human gastric cancer through inhibition of PI3K/Akt and NF-κB. Surg. Oncol. 2018, 27, 495–502. [Google Scholar] [CrossRef] [PubMed]
- Lim, J.W.; Kim, H.; Kim, K.H. Expression of Ku70 and Ku80 Mediated by NF-κB and Cyclooxygenase-2 Is Related to Proliferation of Human Gastric Cancer Cells. J. Biol. Chem. 2002, 277, 46093–46100. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pan, Y.; Huang, J.; Xing, R.; Yin, X.; Cui, J.; Li, W.; Yu, J.; Lu, Y. Metallothionein 2A inhibits NF-κB pathway activation and predicts clinical outcome segregated with TNM stage in gastric cancer patients following radical resection. J. Transl. Med. 2013, 11, 173. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lu, J.; Zhang, L.; Chen, X.; Lu, Q.; Yang, Y.; Liu, J.; Ma, X. SIRT1 counteracted the activation of STAT3 and NF-κB to repress the gastric cancer growth. Int. J. Clin. Exp. Med. 2014, 7, 5050–5058. [Google Scholar] [PubMed]
- Zhang, Y.; Liu, W.; Zhang, W.; Wang, W.; Song, Y.; Xiao, H.; Luo, B. Constitutive activation of the canonical NF-κB signaling pathway in EBV-associated gastric carcinoma. Virology 2019, 532, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Xia, J.-T.; Chen, L.-Z.; Jian, W.-H.; Wang, K.-B.; Yang, Y.-Z.; He, W.-L.; He, Y.-L.; Chen, D.; Li, W. MicroRNA-362 induces cell proliferation and apoptosis resistance in gastric cancer by activation of NF-κB signaling. J. Transl. Med. 2014, 12, 33. [Google Scholar] [CrossRef] [Green Version]
- Bai, T.-L.; Liu, Y.-B.; Li, B.-H. MiR-411 inhibits gastric cancer proliferation and migration through targeting SETD6. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 3344–3350. [Google Scholar]
- Chen, P.; Guo, H.; Wu, X.; Li, J.; Duan, X.; Ba, Q.; Wang, H. Epigenetic silencing of microRNA-204 by Helicobacter pylori augments the NF-κB signaling pathway in gastric cancer development and progression. Carcinogenesis 2020, 41, 430–441. [Google Scholar] [CrossRef] [PubMed]
- Tsai, C.-C.; Chen, T.-Y.; Tsai, K.-J.; Lin, M.-W.; Hsu, C.-Y.; Wu, D.-C.; Tsai, E.-M.; Hsieh, T.-H. NF-κB/miR-18a-3p and miR-4286/BZRAP1 axis may mediate carcinogenesis in Helicobacter pylori―Associated gastric cancer. Biomed. Pharmacother. 2020, 132, 110869. [Google Scholar] [CrossRef] [PubMed]
- Chaturvedi, R.; Asim, M.; Romero–Gallo, J.; Barry, D.P.; Hoge, S.; De Sablet, T.; Delgado, A.G.; Wroblewski, L.E.; Piazuelo, M.B.; Yan, F.; et al. Spermine Oxidase Mediates the Gastric Cancer Risk Associated with Helicobacter pylori CagA. Gastroenterology 2011, 141, 1696–1708.E2. [Google Scholar] [CrossRef] [Green Version]
- Wang, M.; Busuttil, R.A.; Pattison, S.; Neeson, P.; Boussioutas, A. Immunological battlefield in gastric cancer and role of immunotherapies. World J. Gastroenterol. 2016, 22, 6373–6384. [Google Scholar] [CrossRef]
- Umakoshi, M.; Takahashi, S.; Itoh, G.; Kuriyama, S.; Sasaki, Y.; Yanagihara, K.; Yashiro, M.; Maeda, D.; Goto, A.; Tanaka, M. Macrophage-mediated transfer of cancer-derived components to stromal cells contributes to establishment of a pro-tumor microenvironment. Oncogene 2019, 38, 2162–2176. [Google Scholar] [CrossRef]
- Wang, Y.; Zhai, J.; Zhang, T.; Han, S.; Zhang, Y.; Yao, X.; Shen, L. Tumor-Associated Neutrophils Can Predict Lymph Node Metastasis in Early Gastric Cancer. Front. Oncol. 2020, 10, 570113. [Google Scholar] [CrossRef]
- Lee, K.H.; Bae, S.H.; Lee, J.L.; Hyun, M.S.; Kim, S.H.; Song, S.K.; Kim, H.S. Relationship between Urokinase-Type Plasminogen Receptor, Interleukin-8 Gene Expression and Clinicopathological Features in Gastric Cancer. Oncology 2004, 66, 210–217. [Google Scholar] [CrossRef]
- Kido, S.; Kitadai, Y.; Hattori, N.; Haruma, K.; Kido, T.; Ohta, M.; Tanaka, S.; Yoshihara, M.; Sumii, K.; Ohmoto, Y.; et al. Interleukin 8 and vascular endothelial growth factor—prognostic factors in human gastric carcinomas? Eur. J. Cancer 2001, 37, 1482–1487. [Google Scholar] [CrossRef]
- Kitadai, Y.; Haruma, K.; Mukaida, N.; Ohmoto, Y.; Matsutani, N.; Yasui, W.; Yamamoto, S.; Sumii, K.; Kajiyama, G.; Fidler, I.J.; et al. Regulation of disease-progression genes in human gastric carcinoma cells by interleukin 8. Clin. Cancer Res. 2000, 6, 2735–2740. [Google Scholar] [PubMed]
- Tu, S.; Bhagat, G.; Cui, G.; Takaishi, S.; Kurt-Jones, E.A.; Rickman, B.; Betz, K.S.; Penz-Oesterreicher, M.; Bjorkdahl, O.; Fox, J.G.; et al. Overexpression of Interleukin-1β Induces Gastric Inflammation and Cancer and Mobilizes Myeloid-Derived Suppressor Cells in Mice. Cancer Cell 2008, 14, 408–419. [Google Scholar] [CrossRef] [Green Version]
- Wu, H.; Xu, J.-B.; He, Y.-L.; Peng, J.-J.; Zhang, X.-H.; Chen, C.-Q.; Li, W.; Cai, S.-R. Tumor-associated macrophages promote angiogenesis and lymphangiogenesis of gastric cancer. J. Surg. Oncol. 2012, 106, 462–468. [Google Scholar] [CrossRef]
- Li, W.; Zhang, X.; Wu, F.; Zhou, Y.; Bao, Z.; Li, H.; Zheng, P.; Zhao, S. Gastric cancer-derived mesenchymal stromal cells trigger M2 macrophage polarization that promotes metastasis and EMT in gastric cancer. Cell Death Dis. 2019, 10, 1–16. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Xu, J.; Yu, Y.; He, X.; Niu, N.; Li, X.; Zhang, R.; Hu, J.; Ma, J.; Yu, X.; Sun, Y.; et al. Tumor-associated macrophages induce invasion and poor prognosis in human gastric cancer in a cyclooxygenase-2/MMP9-dependent manner. Am. J. Transl. Res. 2019, 11, 6040–6054. [Google Scholar] [PubMed]
- Kim, J.W.; Nam, K.H.; Ahn, S.-H.; Park, D.J.; Kim, H.-H.; Kim, S.H.; Chang, H.; Lee, J.-O.; Kim, Y.J.; Lee, H.S.; et al. Prognostic implications of immunosuppressive protein expression in tumors as well as immune cell infiltration within the tumor microenvironment in gastric cancer. Gastric Cancer 2016, 19, 42–52. [Google Scholar] [CrossRef] [Green Version]
- Junttila, A.; Helminen, O.; Väyrynen, J.P.; Ahtiainen, M.; Kenessey, I.; Jalkanen, S.; Mecklin, J.-P.; Kellokumpu, I.; Kuopio, T.; Böhm, J.; et al. Immunophenotype based on inflammatory cells, PD-1/PD-L1 signalling pathway and M2 macrophages predicts survival in gastric cancer. Br. J. Cancer 2020, 123, 1625–1632. [Google Scholar] [CrossRef] [PubMed]
- Shao, Y.; Sun, K.; Xu, W.; Li, X.-L.; Shen, H.; Sun, W.-H. Helicobacter pyloriinfection, gastrin and cyclooxygenase-2 in gastric carcinogenesis. World J. Gastroenterol. 2014, 20, 12860–12873. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Liu, M.; Liu, X.; Huang, S.; Li, L.; Song, B.; Li, H.; Ren, Q.; Hu, Z.; Zhou, Y.; et al. COX-2 regulates E-cadherin expression through the NF-κB/Snail signaling pathway in gastric cancer. Int. J. Mol. Med. 2013, 32, 93–100. [Google Scholar] [CrossRef] [Green Version]
- Gu, H.; Huang, T.; Shen, Y.; Liu, Y.; Zhou, F.; Jin, Y.; Sattar, H.; Wei, Y. Reactive Oxygen Species-Mediated Tumor Microenvironment Transformation: The Mechanism of Radioresistant Gastric Cancer. Oxidative Med. Cell. Longev. 2018, 2018, 1–8. [Google Scholar] [CrossRef] [Green Version]
- Kim, J.M.; Kim, J.S.; Jung, H.C.; Oh, Y.-K.; Chung, H.-Y.; Lee, C.-H.; Song, I.S. Helicobacter pylori infection activates NF-κB signaling pathway to induce iNOS and protect human gastric epithelial cells from apoptosis. Am. J. Physiol. Liver Physiol. 2003, 285, G1171–G1180. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chang, C.-S.; Chen, W.-N.; Lin, H.-H.; Wu, C.-C.; Wang, C.-J. Increased oxidative DNA damage, inducible nitric oxide synthase, nuclear factor κ B expression and enhanced antiapoptosis-related proteins inHelicobacter pylori-infected non-cardiac gastric adenocarcinoma. World J. Gastroenterol. 2004, 10, 2232–2240. [Google Scholar] [CrossRef]
- Huang, F.-Y.; Chan, A.O.-O.; Rashid, A.; Wong, D.K.-H.; Cho, C.-H.; Yuen, M.-F. Helicobacter pyloriinduces promoter methylation of E-cadherin via interleukin-1β activation of nitric oxide production in gastric cancer cells. Cancer 2012, 118, 4969–4980. [Google Scholar] [CrossRef]
- Wong, C.C.; Kang, W.; Xu, J.; Qian, Y.; Luk, S.T.Y.; Chen, H.; Li, W.; Zhao, L.; Zhang, X.; Chiu, P.W.; et al. Prostaglandin E2 induces DNA hypermethylation in gastric cancer in vitro and in vivo. Theranostics 2019, 9, 6256–6268. [Google Scholar] [CrossRef]
- Takeshima, H.; Niwa, T.; Yamashita, S.; Takamura-Enya, T.; Iida, N.; Wakabayashi, M.; Nanjo, S.; Abe, M.; Sugiyama, T.; Kim, Y.-J.; et al. TET repression and increased DNMT activity synergistically induce aberrant DNA methylation. J. Clin. Investig. 2020, 130, 5370–5379. [Google Scholar] [CrossRef]
- Konturek, P.C.; Kania, J.; Konturek, J.W.; Nikiforuk, A.; Konturek, S.J.; Hahn, E.G.H. pylori infection, atrophic gastritis, cytokines, gastrin, COX-2, PPAR gamma and impaired apoptosis in gastric carcinogenesis. Med. Sci. Monit. 2003, 9, SR53–SR66. [Google Scholar]
- Tiwari, S.K.; Shaik, A.S.; Shaik, A.P.; Alyousef, A.A.; Bardia, A.; Habeeb, A.; Khan, A.A.; Sultana, S.A.; Pasha, S.A. Gene expression patterns of COX-1, COX-2 and iNOS in H. Pylori infected histopathological conditions. Microb. Pathog. 2019, 135, 103634. [Google Scholar] [CrossRef]
- Liao, W.; Ye, T.; Liu, H. Prognostic Value of Inducible Nitric Oxide Synthase (iNOS) in Human Cancer: A Systematic Review and Meta-Analysis. BioMed. Res. Int. 2019, 2019, 6304851. [Google Scholar] [CrossRef] [Green Version]
- Koc, D.O.; Kimiloglu, E. Relation of cyclooxygenase-2 expression with premalignant gastric lesions. Acta Gastroenterol. Belg. 2020, 83, 249–254. [Google Scholar]
- Grivennikov, S.I.; Karin, M. Dangerous liaisons: STAT3 and NF-κB collaboration and crosstalk in cancer. Cytokine Growth Factor Rev. 2010, 21, 11–19. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, G.-F.; Deng, C.-S.; Xiong, Y.-Y.; Gong, L.-L.; Wang, B.-C.; Luo, J. Expression of nuclear factor-kappa B and target genes in gastric precancerous lesions and adenocarcinoma: Association withHelicobactor pyloricagA (+) infection. World J. Gastroenterol. 2004, 10, 491–496. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Zeng, J.; Guo, Q.; Liang, X.; Shen, L.; Li, S.; Sun, Y.; Li, W.; Liu, S.; Yu, H.; et al. Mutual amplification of HNF4α and IL-1R1 composes an inflammatory circuit in Helicobacter pylori associated gastric carcinogenesis. Oncotarget 2016, 7, 11349–11363. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, F.; Xu, Y.; Liu, C.; Ma, C.; Zou, S.; Xu, X.; Jia, J.; Liu, Z. NF-κB/miR-223-3p/ARID1A axis is involved in Helicobacter pylori CagA-induced gastric carcinogenesis and progression. Cell Death Dis. 2018, 9, 1–13. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ma, J.; Liu, J.; Wang, Z.; Gu, X.; Fan, Y.; Zhang, W.; Xu, L.; Zhang, J.; Cai, D. NF-kappaB-dependent MicroRNA-425 upregulation promotes gastric cancer cell growth by targeting PTEN upon IL-1β induction. Mol. Cancer 2014, 13, 40. [Google Scholar] [CrossRef] [Green Version]
- Echizen, K.; Horiuchi, K.; Aoki, Y.; Yamada, Y.; Minamoto, T.; Oshima, H.; Oshima, M. NF-κB-induced NOX1 activation promotes gastric tumorigenesis through the expansion of SOX2-positive epithelial cells. Oncogene 2019, 38, 4250–4263. [Google Scholar] [CrossRef]
- Hu, Z.; Liu, X.; Tang, Z.; Zhou, Y.; Qiao, L. Possible regulatory role of Snail in NF-?B-mediated changes in E-cadherin in gastric cancer. Oncol. Rep. 2012, 29, 993–1000. [Google Scholar] [CrossRef] [Green Version]
- Wang, W.; Luo, H.-S.; Yu, B.-P. Expression of NF-κB and human telomerase reverse transcriptase in gastric cancer and precancerous lesions. World J. Gastroenterol. 2004, 10, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.-J.; Zeng, S.; Xie, R.; Hu, C.-J.; Wang, S.-M.; Wu, Y.-Y.; Xiao, Y.-F.; Yang, S.-M. hTERT promotes gastric intestinal metaplasia by upregulating CDX2 via NF-κB signaling pathway. Oncotarget 2017, 8, 26969–26978. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, Y.; Ukaji, T.; Koide, N.; Umezawa, K. Inhibition of Late and Early Phases of Cancer Metastasis by the NF-κB Inhibitor DHMEQ Derived from Microbial Bioactive Metabolite Epoxyquinomicin: A Review. Int. J. Mol. Sci. 2018, 19, 729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lambrou, G.I.; Hatziagapiou, K.; Vlahopoulos, S. Inflammation and tissue homeostasis: The NF-κB system in physiology and malignant progression. Mol. Biol. Rep. 2020, 47, 4047–4063. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.; Nie, S.; Lv, Z.; Wen, J.; Yuan, Y. Infiltration of Immunoinflammatory Cells and Related Chemokine/Interleukin Expression in Different Gastric Immune Microenvironments. J. Immunol. Res. 2020, 2020, 1–13. [Google Scholar] [CrossRef]
- Algood, H.M.S.; Gallo-Romero, J.; Wilson, K.T.; Peek, J.R.M.; Cover, T.L. Host response toHelicobacter pyloriinfection before initiation of the adaptive immune response. FEMS Immunol. Med. Microbiol. 2007, 51, 577–586. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dixon, B.R.E.A.; Hossain, R.; Patel, R.V.; Algood, H.M.S. Th17 Cells in Helicobacter pylori Infection: A Dichotomy of Help and Harm. Infect. Immun. 2019, 87, e00363-19. [Google Scholar] [CrossRef] [PubMed]
- Yuzhalin, A. The role of interleukin DNA polymorphisms in gastric cancer. Hum. Immunol. 2011, 72, 1128–1136. [Google Scholar] [CrossRef] [PubMed]
- El-Omar, E.M.; Rabkin, C.S.; Gammon, M.D.; Vaughan, T.L.; Risch, H.A.; Schoenberg, J.B.; Stanford, J.L.; Mayne, S.T.; Goedert, J.; Blot, W.J.; et al. Increased risk of noncardia gastric cancer associated with proinflammatory cytokine gene polymorphisms. Gastroenterology 2003, 124, 1193–1201. [Google Scholar] [CrossRef]
- Ramis, I.B.; Vianna, J.S.; Gonçalves, C.V.; von Groll, A.; Dellagostin, O.A.; da Silva, P.E.A. Polymorphisms of the IL-6, IL-8 and IL-10 genes and the risk of gastric pathology in patients infected with Helicobacter pylori. J. Microbiol. Immunol. Infect. 2017, 50, 153–159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gray, B.M.; Fontaine, C.A.; Poe, S.A.; Eaton, K.A. Complex T Cell Interactions Contribute to Helicobacter pylori Gastritis in Mice. Infect. Immun. 2013, 81, 740–752. [Google Scholar] [CrossRef] [Green Version]
- Pantano, F.; Berti, P.; Guida, F.M.; Perrone, G.; Vincenzi, B.; Amato, M.M.C.; Righi, D.; Dell’Aquila, E.; Graziano, F.; Catalano, V.; et al. The role of macrophages polarization in predicting prognosis of radically resected gastric cancer patients. J. Cell. Mol. Med. 2013, 17, 1415–1421. [Google Scholar] [CrossRef] [Green Version]
- Ju, X.; Zhang, H.; Zhou, Z.; Chen, M.; Wang, Q. Tumor-associated macrophages induce PD-L1 expression in gastric cancer cells through IL-6 and TNF-ɑ signaling. Exp. Cell Res. 2020, 396, 112315. [Google Scholar] [CrossRef]
- Betzler, A.C.; Theodoraki, M.-N.; Schuler, P.J.; Döscher, J.; Laban, S.; Hoffmann, T.K.; Brunner, C. NF-κB and Its Role in Checkpoint Control. Int. J. Mol. Sci. 2020, 21, 3949. [Google Scholar] [CrossRef]
- Mendis, S.; Gill, S. Cautious Optimism—The Current Role of Immunotherapy in Gastrointestinal Cancers. Curr. Oncol. 2020, 27, 59–68. [Google Scholar] [CrossRef]
- Gobert, A.P.; Wilson, K.T. The Immune Battle against Helicobacter pylori Infection: NO Offense. Trends Microbiol. 2016, 24, 366–376. [Google Scholar] [CrossRef] [Green Version]
- Rodrigues, R.P.; Ardisson, J.S.; Gonçalves, R.D.C.R.; Oliveira, T.B.; Da Silva, V.B.; Kawano, D.F.; Kitagawa, R.R. Search for Potential Inducible Nitric Oxide Synthase Inhibitors with Favorable ADMET Profiles for the Therapy of Helicobacter pylori Infections. Curr. Top. Med. Chem. 2020, 19, 2795–2804. [Google Scholar] [CrossRef]
- Andrade, F.D.C.P.D.; Mendes, A.N. Computational analysis of eugenol inhibitory activity in lipoxygenase and cyclooxygenase pathways. Sci. Rep. 2020, 10, 1–14. [Google Scholar] [CrossRef]
- Konturek, P.C.; Rembiasz, K.; Burnat, G.; Konturek, S.J.; Tusinela, M.; Bielanski, W.; Rehfeld, J.; Karcz, D.; Hahn, E. Effects of Cyclooxygenase-2 Inhibition on Serum and Tumor Gastrins and Expression of Apoptosis-Related Proteins in Colorectal Cancer. Dig. Dis. Sci. 2006, 51, 779–787. [Google Scholar] [CrossRef]
- Guo, Q.; Li, Q.; Wang, J.; Liu, M.; Wang, Y.; Chen, Z.; Ye, Y.; Guan, Q.; Zhou, Y. A comprehensive evaluation of clinical efficacy and safety of celecoxib in combination with chemotherapy in metastatic or postoperative recurrent gastric cancer patients. Medicine 2019, 98, e16234. [Google Scholar] [CrossRef] [PubMed]
- Nam, S.Y.; Ko, Y.S.; Jung, J.; Yoon, J.; Kim, Y.H.; Choi, Y.J.; Park, J.W.; Chang, M.S.; Kim, W.H.; Lee, B.L. A hypoxia-dependent upregulation of hypoxia-inducible factor-1 by nuclear factor-κB promotes gastric tumour growth and angiogenesis. Br. J. Cancer 2010, 104, 166–174. [Google Scholar] [CrossRef] [Green Version]
- Yoon, J.; Cho, S.J.; Ko, Y.S.; Park, J.; Shin, D.H.; Hwang, I.C.; Han, S.Y.; Nam, S.Y.; Kim, M.A.; Chang, M.S.; et al. A synergistic interaction between transcription factors nuclear factor-κB and signal transducers and activators of transcription 3 promotes gastric cancer cell migration and invasion. BMC Gastroenterol. 2013, 13, 29. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yao, X.; Smolka, A.J. Gastric Parietal Cell Physiology and Helicobacter pylori–Induced Disease. Gastroenterology 2019, 156, 2158–2173. [Google Scholar] [CrossRef] [Green Version]
- Waldum, H.; Mjønes, P. Towards Understanding of Gastric Cancer Based upon Physiological Role of Gastrin and ECL Cells. Cancers 2020, 12, 3477. [Google Scholar] [CrossRef] [PubMed]
- Ding, L.; Sontz, E.A.; Saqui-Salces, M.; Merchant, J.L. Interleukin-1β Suppresses Gastrin via Primary Cilia and Induces Antral Hyperplasia. Cell. Mol. Gastroenterol. Hepatol. 2021, 11, 1251–1266. [Google Scholar] [CrossRef]
- Zhu, S.; Soutto, M.; Chen, Z.; Peng, D.; Romero-Gallo, J.; Krishna, U.S.; Belkhiri, A.; Washington, M.K.; Peek, R.; El-Rifai, W. Helicobacter pylori-induced cell death is counteracted by NF-κB-mediated transcription of DARPP-32. Gut 2017, 66, 761.1–762. [Google Scholar] [CrossRef] [Green Version]
- Ren, H.-Y.; Huang, G.-L.; Liu, W.-M.; Zhang, W.; Liu, Y.; Su, G.-Q.; Shen, D.-Y. IL-1β induced RXRα overexpression through activation of NF-κB signaling in gastric carcinoma. Biomed. Pharmacother. 2016, 78, 329–334. [Google Scholar] [CrossRef]
- Lv, D.-D.; Zhou, L.-Y.; Tang, H. Hepatocyte nuclear factor 4α and cancer-related cell signaling pathways: A promising insight into cancer treatment. Exp. Mol. Med. 2021, 53, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, N.; Morisaki, T.; Nakashima, H.; Tasaki, A.; Kubo, M.; Kuga, H.; Nakahara, C.; Nakamura, K.; Noshiro, H.; Yao, T.; et al. Interleukin 1β Enhances Invasive Ability of Gastric Carcinoma through Nuclear Factor-κB Activation. Clin. Cancer Res. 2004, 10, 1853–1859. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Acharyya, S.; Oskarsson, T.; Vanharanta, S.; Malladi, S.; Kim, J.; Morris, P.G.; Manova-Todorova, K.; Leversha, M.; Hogg, N.; Seshan, V.E.; et al. A CXCL1 Paracrine Network Links Cancer Chemoresistance and Metastasis. Cell 2012, 150, 165–178. [Google Scholar] [CrossRef] [Green Version]
- Kwon, C.H.; Moon, H.J.; Park, H.J.; Choi, J.H.; Park, D.Y. S100A8 and S100A9 promotes invasion and migration through p38 mitogen-activated protein kinase-dependent NF-κB activation in gastric cancer cells. Mol. Cells 2013, 35, 226–234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhang, Z.; Fan, B.; Liu, F.; Song, N.; Peng, Y.; Ma, W.; Ma, R.; Dong, T.; Liu, S. HOX transcript antisense RNA is elevated in gastric carcinogenesis and regulated by the NF-κB pathway. J. Cell. Biochem. 2019, 120, 10548–10555. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Che, X.; Zhang, S.; Guo, T.; He, X.; Liu, Y.; Qu, X. Positive Cross-Talk Between CXC Chemokine Receptor 4 (CXCR4) and Epidermal Growth Factor Receptor (EGFR) Promotes Gastric Cancer Metastasis via the Nuclear Factor kappa B (NF-kB)-Dependent Pathway. Med. Sci. Monit. 2020, 26. [Google Scholar] [CrossRef] [PubMed]
- Yamanaka, N.; Sasaki, N.; Tasaki, A.; Nakashima, H.; Kubo, M.; Morisaki, T.; Noshiro, H.; Yao, T.; Tsuneyoshi, M.; Tanaka, M.; et al. Nuclear factor-kappaB p65 is a prognostic indicator in gastric carcinoma. Anticancer Res. 2004, 24, 1071–1075. [Google Scholar]
- Ye, S.; Long, Y.-M.; Rong, J.; Xie, W.-R. Nuclear factor kappa B: A marker of chemotherapy for human stage IV gastric carcinoma. World J. Gastroenterol. 2008, 14, 4739–4744. [Google Scholar] [CrossRef]
- Lee, B.L.; Lee, H.S.; Jung, J.; Cho, S.J.; Chung, H.Y.; Kim, W.H.; Jin, Y.-W.; Kim, C.S.; Nam, S.Y. Nuclear Factor-κB Activation Correlates with Better Prognosis and Akt Activation in Human Gastric Cancer. Clin. Cancer Res. 2005, 11, 2518–2525. [Google Scholar] [CrossRef] [Green Version]
- Kinoshita, J.; Fushida, S.; Harada, S.; Makino, I.; Nakamura, K.; Oyama, K.; Fujita, H.; Ninomiya, I.; Fujimura, T.; Kayahara, M.; et al. PSK enhances the efficacy of docetaxel in human gastric cancer cells through inhibition of nuclear factor-κB activation and survivin expression. Int. J. Oncol. 2010, 36, 593–600. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kwon, O.-H.; Kim, J.H.; Kim, S.-Y.; Kim, Y.S. TWEAK/Fn14 signaling mediates gastric cancer cell resistance to 5-fluorouracil via NF-κB activation. Int. J. Oncol. 2013, 44, 583–590. [Google Scholar] [CrossRef] [Green Version]
- Zhi, X.; Tao, J.; Xiang, G.; Cao, H.; Liu, Z.; Yang, K.; Lv, C.; Ni, S. APRIL Induces Cisplatin Resistance in Gastric Cancer Cells via Activation of the NF-κB Pathway. Cell. Physiol. Biochem. 2015, 35, 571–585. [Google Scholar] [CrossRef]
- Rohwer, N.; Dame, C.; Haugstetter, A.; Wiedenmann, B.; Detjen, K.; Schmitt, C.A.; Cramer, T. Hypoxia-Inducible Factor 1α Determines Gastric Cancer Chemosensitivity via Modulation of p53 and NF-κB. PLoS ONE 2010, 5, e12038. [Google Scholar] [CrossRef] [Green Version]
- Manu, K.A.; Shanmugam, M.K.; Li, F.; Chen, L.; Siveen, K.S.; Ahn, K.S.; Kumar, A.P.; Sethi, G. Simvastatin sensitizes human gastric cancer xenograft in nude mice to capecitabine by suppressing nuclear factor-kappa B-regulated gene products. J. Mol. Med. 2014, 92, 267–276. [Google Scholar] [CrossRef] [Green Version]
- Shi, H.; Sun, Y.; Ruan, H.; Ji, C.; Zhang, J.; Wu, P.; Li, L.; Huang, C.; Jia, Y.; Zhang, X.; et al. 3,3′-Diindolylmethane Promotes Gastric Cancer Progression via β-TrCP-Mediated NF-κB Activation in Gastric Cancer-Derived MSCs. Front. Oncol. 2021, 11, 603533. [Google Scholar] [CrossRef] [PubMed]
- Inoue, M.; Matsumoto, S.; Saito, H.; Tsujitani, S.; Ikeguchi, M. Intraperitoneal administration of a small interfering RNA targeting nuclear factor-kappa B with paclitaxel successfully prolongs the survival of xenograft model mice with peritoneal metastasis of gastric cancer. Int. J. Cancer 2008, 123, 2696–2701. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Haruki, K.; Shiba, H.; Fujiwara, Y.; Furukawa, K.; Iwase, R.; Uwagawa, T.; Misawa, T.; Ohashi, T.; Yanaga, K. Inhibition of Nuclear Factor-κB Enhances the Antitumor Effect of Paclitaxel Against Gastric Cancer with Peritoneal Dissemination in Mice. Dig. Dis. Sci. 2012, 58, 123–131. [Google Scholar] [CrossRef]
- Sohma, I.; Fujiwara, Y.; Sugita, Y.; Yoshioka, A.; Shirakawa, M.; Moon, J.-H.; Takiguchi, S.; Miyata, H.; Yamasaki, M.; Mori, M.; et al. Parthenolide, an NF-?B inhibitor, suppresses tumor growth and enhances response to chemotherapy in gastric cancer. Cancer Genom. Proteom. 2011, 8, 39–47. [Google Scholar]
- Freitas, R.H. NF-κB-IKKβ Pathway as a Target for Drug Development: Realities, Challenges and Perspectives. Curr. Drug Targets 2018, 19, 1933–1942. [Google Scholar] [CrossRef]
- Ramadass, V.; Vaiyapuri, T.; Tergaonkar, V. Small Molecule NF-kB Pathway Inhibitors in Clinic. Int. J. Mol. Sci. 2020, 21, 5164. [Google Scholar] [CrossRef] [PubMed]
- Nakata, W.; Hayakawa, Y.; Nakagawa, H.; Sakamoto, K.; Kinoshita, H.; Takahashi, R.; Hirata, Y.; Maeda, S.; Koike, K. Anti-tumor activity of the proteasome inhibitor bortezomib in gastric cancer. Int. J. Oncol. 2011, 39, 1529–1536. [Google Scholar] [CrossRef]
- Zhang, J.; Pu, K.; Bai, S.; Peng, Y.; Li, F.; Ji, R.; Guo, Q.; Sun, W.; Wang, Y. The anti-alcohol dependency drug disulfiram inhibits the viability and progression of gastric cancer cells by regulating the Wnt and NF-κB pathways. J. Int. Med Res. 2020, 48, 300060520925996. [Google Scholar] [CrossRef]
- Gayed, D.D.T.; Wodeyar, J.; Wang, Z.-X.; Wei, X.; Yao, Y.-Y.; Chen, X.-X.; Du, Z.; Chen, J.-C. Prognostic values of inhibitory κB kinases mRNA expression in human gastric cancer. Biosci. Rep. 2019, 39, 20180617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sakamoto, K.; Hikiba, Y.; Nakagawa, H.; Hayakawa, Y.; Yanai, A.; Akanuma, M.; Ogura, K.; Hirata, Y.; Kaestner, K.H.; Omata, M.; et al. Inhibitor of κB Kinase Beta Regulates Gastric Carcinogenesis via Interleukin-1α Expression. Gastroenterology 2010, 139, 226–238.e6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, L.; Ruan, Y.; Wang, X.; Min, L.; Shen, Z.; Sun, Y.; Qin, X. BAY 11-7082, a nuclear factor-κB inhibitor, induces apoptosis and S phase arrest in gastric cancer cells. J. Gastroenterol. 2013, 49, 864–874. [Google Scholar] [CrossRef]
- Yan, Y.; Qian, H.; Cao, Y.; Zhu, T. Nuclear factor-κB inhibitor Bay11-7082 inhibits gastric cancer cell proliferation by inhibiting Gli1 expression. Oncol. Lett. 2021, 21, 1–7. [Google Scholar] [CrossRef]
- Al-Ishaq, R.K.; Overy, A.J.; Büsselberg, D. Phytochemicals and Gastrointestinal Cancer: Cellular Mechanisms and Effects to Change Cancer Progression. Biomolecules 2020, 10, 105. [Google Scholar] [CrossRef] [Green Version]
- Han, H.; Lim, J.W.; Kim, H. Lycopene Inhibits Activation of Epidermal Growth Factor Receptor and Expression of Cyclooxygenase-2 in Gastric Cancer Cells. Nutrients 2019, 11, 2113. [Google Scholar] [CrossRef] [Green Version]
- Yu, L.-L.; Wu, J.-G.; Dai, N.; Si, J.-Μ. Curcumin reverses chemoresistance of human gastric cancer cells by downregulating the NF-?B transcription factor. Oncol. Rep. 2011, 26, 1197–1203. [Google Scholar] [CrossRef] [Green Version]
- Qiu, P.; Zhang, S.; Zhou, Y.; Zhu, M.; Kang, Y.; Chen, D.; Wang, J.; Zhou, P.; Li, W.; Xu, Q.; et al. Synthesis and evaluation of asymmetric curcuminoid analogs as potential anticancer agents that downregulate NF-κB activation and enhance the sensitivity of gastric cancer cell lines to irinotecan chemotherapy. Eur. J. Med. Chem. 2017, 139, 917–925. [Google Scholar] [CrossRef] [PubMed]
- Sokolova, O.; Naumann, M. NF-κB Signaling in Gastric Cancer. Toxins 2017, 9, 119. [Google Scholar] [CrossRef]
- Liu, D.; Shi, K.; Fu, M.; Chen, F. Melatonin indirectly decreases gastric cancer cell proliferation and invasion via effects on cancer-associated fibroblasts. Life Sci. 2021, 277, 119497. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.H.; Huang, J.Q.; Zheng, G.F.; Lam, S.K.; Karlberg, J.; Wong, B.C.-Y. Non-steroidal anti-inflammatory drug use and the risk of gastric cancer: A systematic review and meta-analysis. J. Natl. Cancer Inst. 2003, 95, 1784–1791. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rodríguez, L.A.G.; Soriano-Gabarró, M.; Vora, P.; Soriano, L.C. Low-dose aspirin and risk of gastric and oesophageal cancer: A population-based study in the United Kingdom using The Health Improvement Network. Int. J. Cancer 2020, 147, 2394–2404. [Google Scholar] [CrossRef]
- Bindu, S.; Mazumder, S.; Bandyopadhyay, U. Non-steroidal anti-inflammatory drugs (NSAIDs) and organ damage: A current perspective. Biochem. Pharmacol. 2020, 180, 114147. [Google Scholar] [CrossRef]
- Bindu, S.; Mazumder, S.; Dey, S.; Pal, C.; Goyal, M.; Alam, A.; Iqbal, M.; Sarkar, S.; Siddiqui, A.A.; Banerjee, C.; et al. Nonsteroidal anti-inflammatory drug induces proinflammatory damage in gastric mucosa through NF-κB activation and neutrophil infiltration: Anti-inflammatory role of heme oxygenase-1 against nonsteroidal anti-inflammatory drug. Free Radic. Biol. Med. 2013, 65, 456–467. [Google Scholar] [CrossRef]
- Ko, I.-G.; Jin, J.-J.; Hwang, L.; Kim, S.-H.; Kim, C.-J.; Han, J.H.; Kwak, M.S.; Yoon, J.Y.; Jeon, J.W. Evaluating the mucoprotective effect of polydeoxyribonucleotide against indomethacin-induced gastropathy via the MAPK/NF-κB signaling pathway in rats. Eur. J. Pharmacol. 2020, 874, 172952. [Google Scholar] [CrossRef]
- Sexton, R.E.; Al Hallak, M.N.; Uddin, H.; Diab, M.; Azmi, A.S. Gastric Cancer Heterogeneity and Clinical Outcomes. Technol. Cancer Res. Treat. 2020, 19, 1533033820935477. [Google Scholar] [CrossRef]
- Ricci, A.; Rizzo, A.; Llimpe, F.R.; Di Fabio, F.; De Biase, D.; Rihawi, K. Novel HER2-Directed Treatments in Advanced Gastric Carcinoma: AnotHER Paradigm Shift? Cancers 2021, 13, 1664. [Google Scholar] [CrossRef]
- Muro, K.; Chung, H.; Shankaran, V.; Geva, R.; Catenacci, D.; Gupta, S.; Eder, J.P.; Golan, T.; Le, D.T.; Burtness, B.; et al. Pembrolizumab for patients with PD-L1-positive advanced gastric cancer (KEYNOTE-012): A multicentre, open-label, phase 1b trial. Lancet Oncol. 2016, 17, 717–726. [Google Scholar] [CrossRef]
- Ghidini, M.; Petrillo, A.; Botticelli, A.; Trapani, D.; Parisi, A.; La Salvia, A.; Sajjadi, E.; Piciotti, R.; Fusco, N.; Khakoo, S. How to Best Exploit Immunotherapeutics in Advanced Gastric Cancer: Between Biomarkers and Novel Cell-Based Approaches. J. Clin. Med. 2021, 10, 1412. [Google Scholar] [CrossRef] [PubMed]
- Wainberg, Z.A.; Fuchs, C.S.; Tabernero, J.; Shitara, K.; Muro, K.; Van Cutsem, E.; Bang, Y.-J.; Chung, H.C.; Yamaguchi, K.; Varga, E.; et al. Efficacy of Pembrolizumab Monotherapy for Advanced Gastric/Gastroesophageal Junction Cancer with Programmed Death Ligand 1 Combined Positive Score ≥ 10. Clin. Cancer Res. 2021, 27, 1923–1931. [Google Scholar] [CrossRef] [PubMed]
| Gene Name | Genetic Aberration | Comments | References |
|---|---|---|---|
| NFKB1 | SNP_rs230521 | observed in GC patients | [32] |
| SNP_rs28362491 (−94 ins/del ATTG) | associated with diffuse GC, accelerate severe gastric inflammation | [33,34] | |
| SNP_rs4648068 | increased risk of GC | [36,37] | |
| homozygous deletion | invasive GC, gastric atrophy in mice | [38,39] | |
| NFKB2 | homozygous deletion | gastric hyperplasia, early postnatal death | [40] |
| suppressed in gastric mucosal lesions | [39] | ||
| NFKBIA | SNP_rs2233408 T/C homozygote | GC susceptibility | [41] |
| SNP_rs2233408 T heterozygote | reduced GC risk in intestinal-type non-cardiac GC | ||
| SNP_rs17103265 | risk factor for gastric carcinogenesis | [42] | |
| SNP_rs696 | cardia GC susceptibility | [43] | |
| SNP_rs2233406 | non-cardia GC susceptibility | ||
| IΚBKB | SNP_rs2272736 A homozygote | prolonged overall survival time | [44] |
| TNIP1 | SNP_rs7708392 | associated with GC risk | [45] |
| SNP_rs10036748 | |||
| MYD88 | deletion, mutation | gastric mucosal damage, carcinogenesis | [46] |
| L265P mutant | observed in gastric mucosa-associated lymphoid tissue (MALT) lymphomas | [47] | |
| RIPK2 | SNP_rs16900627 | increased risk of intestinal GC | [48] |
| TLR9 | SNP_rs5743836 (−1237 T/C) | associated with H. pylori-induced GC | [49] |
| NF-κB Regulated Genes | Comments | References |
|---|---|---|
| IL-8 | correlates with diffuse-type GC | [87] |
| correlates with depth of invasion, venous and lymphatic invasion, low survival rate, enhances cell migration and invasion | [88] | |
| IL-17 | positively associates with GC, enhances cell migration and invasion | [67,89] |
| IL-1β | promotes gastric dysplasia to GC | [90] |
| COX2, MMP9, VEGF | TAMs induce COX2, MMP9, VEGF expression, promote invasion/migration in GC | [91,92,93] |
| PD-L1 | relates to a less advanced stage, intestinal type GC | [94] |
| associates with poor prognosis for GC patients | [95] | |
| NO, PGE2 | potentiates the infiltration of macrophages in stomach tissue, promotes an inflammatory environment | [96,97,98] |
| promotes tissue healing via eliminating infectious agents, increasing tissue microcirculation and cell restitution | [99] | |
| accelerates turnover of epithelial cells, increasing the mutagenesis rate in inflamed tissue | [100,101,102,103] | |
| iNOS, COX-2 | contributes to a gradual progress of gastric carcinogenesis | [104,105,106,107] |
| STAT3 | contributes to GC development and progression | [108] |
| c-myc, cyclinD1 | high expression in intestinal-type GC | [109] |
| HNF4α | HNF4α overexpression correlates with sustained inflammation and GC | [110] |
| miR-223-3p, miR-18a-3p, miR-4286 | expression in gastric cancer cells and tissues, links to proliferation and gastric carcinogenesis | [80,111] |
| miR-425 | promotes proliferation of GC | [112] |
| Noxo1 | associates with gastritis and GC | [113] |
| Snail1 | downregulation of E-cadherin in GC tissue | [114] |
| hTERT | promotes intestinal metaplasia | [115,116] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chaithongyot, S.; Jantaree, P.; Sokolova, O.; Naumann, M. NF-κB in Gastric Cancer Development and Therapy. Biomedicines 2021, 9, 870. https://doi.org/10.3390/biomedicines9080870
Chaithongyot S, Jantaree P, Sokolova O, Naumann M. NF-κB in Gastric Cancer Development and Therapy. Biomedicines. 2021; 9(8):870. https://doi.org/10.3390/biomedicines9080870
Chicago/Turabian StyleChaithongyot, Supattra, Phatcharida Jantaree, Olga Sokolova, and Michael Naumann. 2021. "NF-κB in Gastric Cancer Development and Therapy" Biomedicines 9, no. 8: 870. https://doi.org/10.3390/biomedicines9080870






