Effect of a Larger Flush Volume on Bioavailability and Efficacy of Umbilical Venous Epinephrine during Neonatal Resuscitation in Ovine Asphyxial Arrest
Abstract
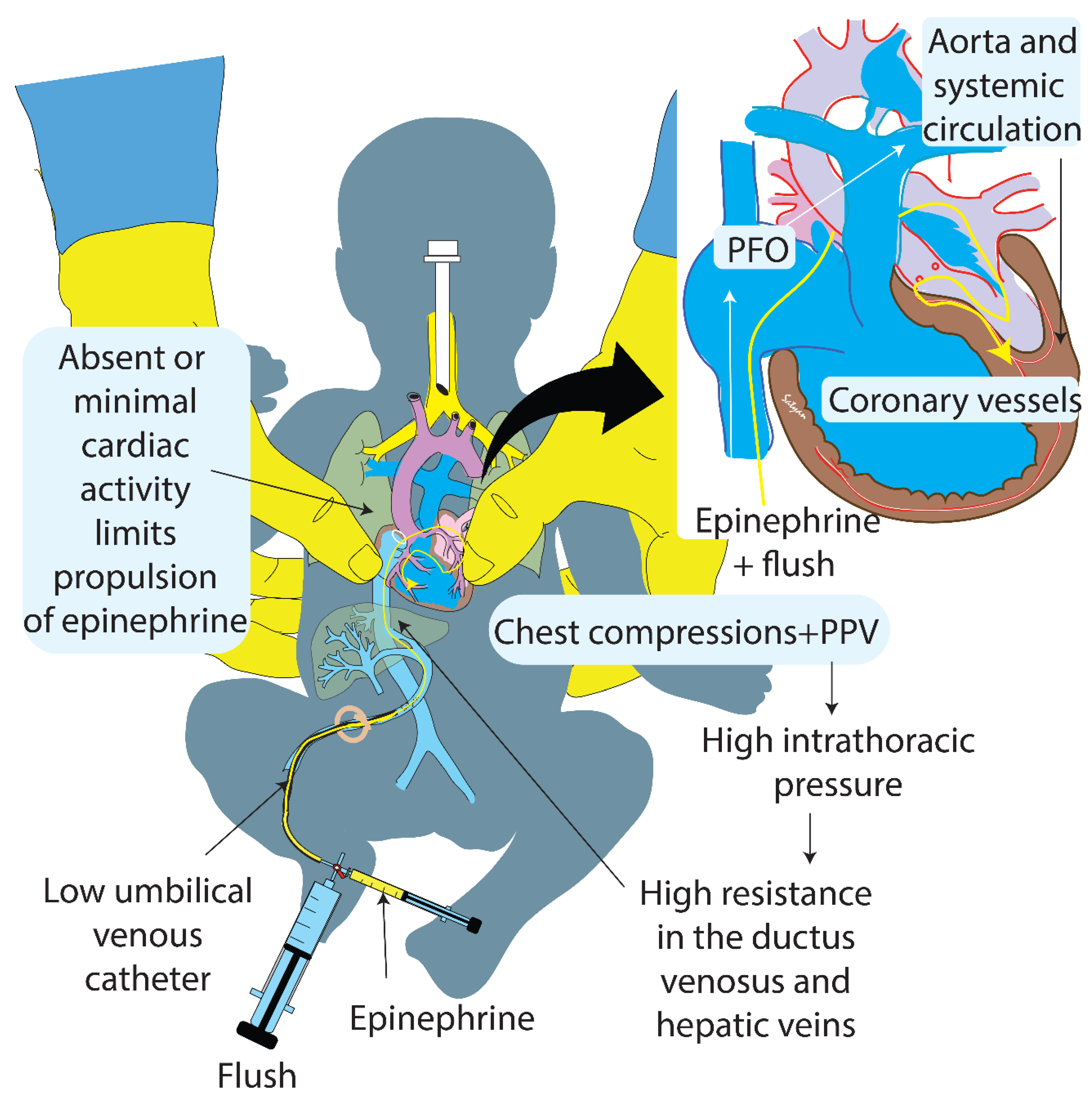
:1. Introduction
2. Materials and Methods
2.1. Fetal Instrumentation
2.2. Asphyxial Arrest and Resuscitation
2.3. Primary and Secondary Outcomes
2.4. Data Collection and Statistical Analysis
3. Results
3.1. Incidence of ROSC and Time to Achieve ROSC
3.2. Cumulative Dose of Epinephrine and Epinephrine Concentrations in Plasma
3.3. Post-ROSC Hemodynamics
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wyckoff, M.H.; Wyllie, J.; Aziz, K.; De Almeida, M.F.; Fabres, J.; Fawke, J.; Guinsburg, R.; Hosono, S.; Isayama, T.; Kapadia, V.S.; et al. Neonatal life support: 2020 international consensus on cardiopulmonary resuscitation and emergency cardiovascular care science with treatment recommendations. Circulation 2020, 142, S185–S221. [Google Scholar] [CrossRef] [PubMed]
- Isayama, T.; Mildenhall, L.; Schmölzer, G.M.; Kim, H.-S.; Rabi, Y.; Ziegler, C.; Liley, H.G. International Liaison Committee on Resuscitation Newborn Life Support Task Force The route, dose, and interval of epinephrine for neonatal resuscitation: A systematic review. Pediatrics 2020, 146, e20200586. [Google Scholar] [CrossRef] [PubMed]
- Vali, P.; Chandrasekharan, P.; Rawat, M.; Gugino, S.; Koenigsknecht, C.; Helman, J.; Jusko, W.J.; Mathew, B.; Berkelhamer, S.; Nair, J.; et al. Evaluation of timing and route of epinephrine in a neonatal model of asphyxial arrest. J. Am. Heart. Assoc. 2017, 6, e004402. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nair, J.; Vali, P.; Gugino, S.F.; Koenigsknecht, C.; Helman, J.; Nielsen, L.C.; Chandrasekharan, P.; Rawat, M.; Berkelhamer, S.; Mathew, B.; et al. Bioavailability of endotracheal epinephrine in an ovine model of neonatal resuscitation. Early Hum. Dev. 2019, 130, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Aziz, K.; Lee, H.C.; Escobedo, M.B.; Hoover, A.V.; Kamath-Rayne, B.D.; Kapadia, V.S.; Magid, D.J.; Niermeyer, S.; Schmölzer, G.M.; Szyld, E.; et al. Part 5: Neonatal resuscitation: 2020 american heart association guidelines for cardiopulmonary resuscitation and emergency cardiovascular care. Circulation 2020, 142, S524–S550. [Google Scholar] [CrossRef] [PubMed]
- American Academy of Pediatrics; American Heart Association. Textbook of Neonatal Resuscitation, 7th ed.; American Academy of Pediatrics: Healdsburg, CA, USA, 2016. [Google Scholar]
- Vali, P.; Sankaran, D.; Rawat, M.; Berkelhamer, S.; Lakshminrusimha, S. Epinephrine in neonatal resuscitation. Children 2019, 6, 51. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foglia, E.E.; Weiner, G.; De Almeida, M.F.B.; Wyllie, J.; Wyckoff, M.H.; Rabi, Y.; Guinsburg, R. International liaison committee on resuscitation neonatal life support task force duration of resuscitation at birth, mortality, and neurodevelopment: A systematic review. Pediatrics 2020, 146, e20201449. [Google Scholar] [CrossRef] [PubMed]
- Vali, P.; Weiner, G.M.; Sankaran, D.; Lakshminrusimha, S. What is the optimal initial dose of epinephrine during neonatal resuscitation in the delivery room? J. Perinatol. 2021, 1–5. [Google Scholar] [CrossRef]
- NRP 8th Edition Busy People Update. 2021. Available online: https://downloads.aap.org/AAP/PDF/NRP%208th%20Edition%20Busy%20People%20Update%20(1).pdf (accessed on 1 March 2021).
- Kilkenny, C.; Browne, W.J.; Cuthill, I.C.; Emerson, M.; Altman, D.G. Improving bioscience research reporting: The arrive guidelines for reporting animal research. PLoS Biol. 2010, 8, e1000412. [Google Scholar] [CrossRef] [PubMed]
- Sankaran, D.; Chandrasekharan, P.K.; Gugino, S.F.; Koenigsknecht, C.; Helman, J.; Nair, J.; Mathew, B.; Rawat, M.; Vali, P.; Nielsen, L.; et al. Randomised trial of epinephrine dose and flush volume in term newborn lambs. Arch. Dis. Child. Fetal Neonatal Ed. 2021. [Google Scholar] [CrossRef] [PubMed]
- Vali, P.; Gugino, S.; Koenigsknecht, C.; Helman, J.; Chandrasekharan, P.; Rawat, M.; Lakshminrusimha, S.; Nair, J. The perinatal asphyxiated lamb model: A model for newborn resuscitation. J. Vis. Exp. 2018, 2018, e57553. [Google Scholar] [CrossRef] [PubMed]
- Sankaran, D.; Chen, P.; Alhassen, Z.; Lesneski, A.; Hardie, M.; Lakshminrusimha, S.; Vali, P. Optimal inspired oxygen weaning strategy following return of spontaneous circulation after perinatal asphyxial arrest. Sect. Neonatal Perinat. Med. Program 2021, 147, 757–759. [Google Scholar] [CrossRef]
- Hirvonen, L.; Peltonen, T.; Ruokola, M. Angiocardiography of the newborn with contrast injected into the umbilical vein. Ann. Paediatr. Fenn. 1961, 7, 124–130. [Google Scholar] [PubMed]
- Bhasin, H.; Kohli, C. Myocardial dysfunction as a predictor of the severity and mortality of hypoxic ischaemic encephalopathy in severe perinatal asphyxia: A case-control study. Paediatr. Int. Child Health 2019, 39, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Rajakumar, P.; Bhat, B.V.; Sridhar, M.; Balachander, J.; Konar, B.; Narayanan, P.; Chetan, G. Cardiac enzyme levels in myocardial dysfunction in newborns with perinatal asphyxia. Indian J. Pediatr. 2008, 75, 1223–1225. [Google Scholar] [CrossRef] [PubMed]
- Popescu, M.R.; Panaitescu, A.M.; Pavel, B.; Zagrean, L.; Peltecu, G.; Zagrean, A.-M. Getting an early start in understanding perinatal asphyxia impact on the cardiovascular system. Front. Pediatr. 2020, 8. [Google Scholar] [CrossRef] [PubMed]
- Wyckoff, M.H.; Perlman, J.M.; Laptook, A.R. Use of volume expansion during delivery room resuscitation in near-term and term infants. Pediatric 2005, 115, 950–955. [Google Scholar] [CrossRef] [PubMed]
- Kasdorf, E.; Perlman, J.M. Strategies to prevent reperfusion injury to the brain following intrapartum hypoxia-ischemia. Semin. Fetal Neonatal Med. 2013, 18, 379–384. [Google Scholar] [CrossRef] [PubMed]
- Berg, R.A.; Otto, C.W.; Kern, K.B.; Hilwig, R.W.; Sanders, A.B.; Henry, C.P.; Ewy, G.A. A randomized, blinded trial of high-dose epinephrine versus standard-dose epinephrine in a swine model of pediatric asphyxial cardiac arrest. Crit. Care Med. 1996, 24, 1695–1700. [Google Scholar] [CrossRef] [PubMed]

| Flush Volume | 1-mL Flush n = 7 | 2.5-mL Flush n = 15 | p-Value |
|---|---|---|---|
| Gestational age (days) | 142 (2) | 140 (1) | 0.97 |
| Weight (kg) | 4.45 (1.3) | 3.6 (0.8) | 0.07 |
| Sex distribution n (%) | 4 females (57%) | 6 females (40%) | 0.45 |
| Time to cardiac arrest (min) | 14.7 (3.6) | 15.6 (4.6) | 0.89 |
| ROSC incidence with the 1st dose of epinephrine n (%) | 3 (42.8%) | 12 (80%) | 0.08 |
| ROSC incidence n (%) | 5 (71.4%) | 13 (86.6%) | 0.38 |
| Median time to ROSC from time of epinephrine and flush (s) | 95 (60–120) | 72 (56–111) | 0.71 |
| Cumulative dose of epinephrine (mg/kg) median (interquartile range) | 0.06 (0.03–0.075) | 0.03 (0.03–0.03) | 0.26 |
| Mean blood pressure at 10 min after ROSC (mmHg) | 64 (25) | 65 (15) | 0.26 0.96 |
| Heart rate at 10 min after ROSC (beats per minute) | 195 (14) | 194 (13) | 0.88 |
| Left Carotid artery blood flow at 10 min after ROSC (ml/kg/min) | 26 (6) | 31 (13) | 0.47 |
| Parameter | 1-mL Flush | 2.5-mL Flush | p-Value |
|---|---|---|---|
| Plasma epinephrine concentration at 1 min after epinephrine dose among all the lambs studied (ng/mL). | 494 (171) | 519 (140) | 0.92 |
| Plasma epinephrine concentration at 1 min after epinephrine and flush among lambs that achieved ROSC with 1st dose (ng/mL) | 572 (50) | 545 (165) | 0.94 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sankaran, D.; Vali, P.; Chandrasekharan, P.; Chen, P.; Gugino, S.F.; Koenigsknecht, C.; Helman, J.; Nair, J.; Mathew, B.; Rawat, M.; et al. Effect of a Larger Flush Volume on Bioavailability and Efficacy of Umbilical Venous Epinephrine during Neonatal Resuscitation in Ovine Asphyxial Arrest. Children 2021, 8, 464. https://doi.org/10.3390/children8060464
Sankaran D, Vali P, Chandrasekharan P, Chen P, Gugino SF, Koenigsknecht C, Helman J, Nair J, Mathew B, Rawat M, et al. Effect of a Larger Flush Volume on Bioavailability and Efficacy of Umbilical Venous Epinephrine during Neonatal Resuscitation in Ovine Asphyxial Arrest. Children. 2021; 8(6):464. https://doi.org/10.3390/children8060464
Chicago/Turabian StyleSankaran, Deepika, Payam Vali, Praveen Chandrasekharan, Peggy Chen, Sylvia F. Gugino, Carmon Koenigsknecht, Justin Helman, Jayasree Nair, Bobby Mathew, Munmun Rawat, and et al. 2021. "Effect of a Larger Flush Volume on Bioavailability and Efficacy of Umbilical Venous Epinephrine during Neonatal Resuscitation in Ovine Asphyxial Arrest" Children 8, no. 6: 464. https://doi.org/10.3390/children8060464






