Monkeypox Infection 2022: An Updated Narrative Review Focusing on the Neonatal and Pediatric Population
Abstract
:1. Introduction
2. Materials and Methods
3. Results
3.1. Monkeypox Virus: Generalities
3.2. Clinical Presentation, Transmission and Prevention
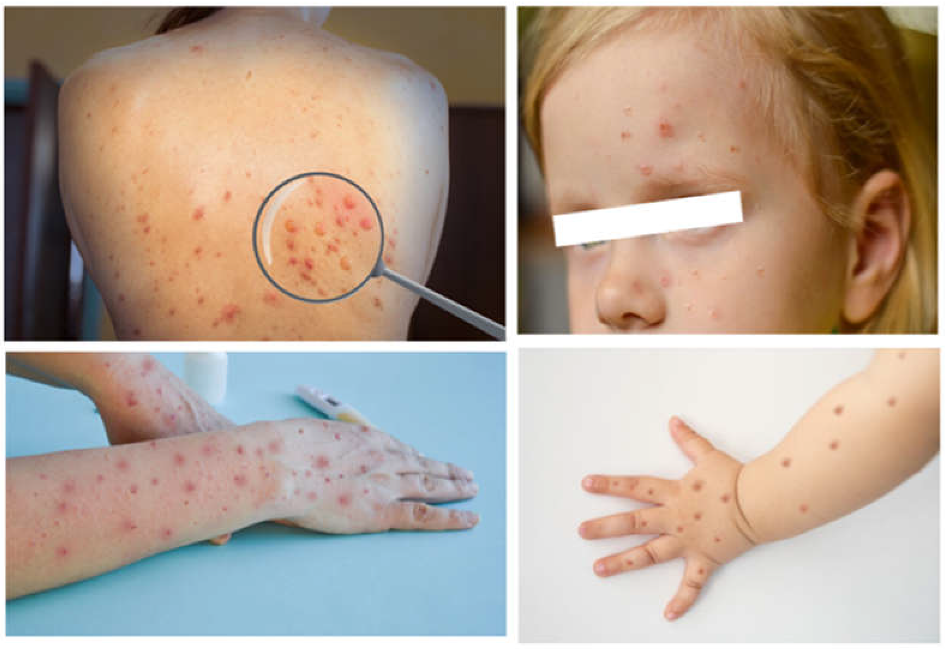
3.2.1. Monkeypox in the Pediatric Population
3.2.2. Monkeypox in Pregnancy and in Newborns
3.3. Diagnosis
3.4. Treatment
3.4.1. Antiviral Medicines
- (a)
- Cidofovir (Vistide) is primarily used as a treatment for retinitis, encephalitis and oesophagitis caused by cytomegalovirus, especially in people with HIV. It is the phosphorylated active metabolite of brincidovir. In-vitro and preclinical studies showed that is effective against poxviruses [4];
- (b)
- Brincidofovir (Tembexa) is available as oral suspension/tablets, approved by FDA for smallpox disease [58]. Both drugs are inhibitors of DNA replication with a broad spectrum of activity against multiple families of double-stranded DNA viruses.
- (c)
- Tecovirimat (ST-246): is an antiviral medication which impairs the function of the VP37 envelope protein necessary for the formation of the extracellular enveloped virus required for cell-to-cell transmission; it has more specific activity against orthopoxviruses [58]. It has been approved by Food and Drug Administration (FDA), used to treat human smallpox disease but can be used against MPV. Tecovirimat is given orally (TPOXX®: 200 mg capsule) or as an injectable formulation [52]. Capsules should be taken within 30 min after a full meal with moderate to high fat. Per CDC guidelines, for those who cannot swallow they can be opened and mixed with liquids/soft food [59] Because it is an inducer of cytochrome P450 (CYP) 3A and CYP2B6, co-administration with this drug may lead to reduced plasma exposures of sensitive substrates of CYP3A4 or CYP2B6, reducing the effects. Because of the presence in its IV formulation of a potentially nephrotoxic substance (hydroxypropyl-β-cyclodextrin), it is advisable to dose creatinine clearance (CrCl) and liver function before starting treatment. Intravenous therapy is safe in mild/moderate renal impairment but is contraindicated in severe nephropathies (CrCl < 30 mL/min), both in adults and children [39]. Dose adjustments for oral therapy instead are not required in the case of mild, moderate, severe nephropathy or even in patients requiring hemodialysis in end-stage renal disease [60]. Although reduced fertility due to testicular toxicity was found in mouse models, no human data are available [60].
3.4.2. Children’s Treatment
3.4.3. Treatment during Pregnancy and Breast-Feeding
3.5. Vaccines: Pre- and Post-Exposure Prophylaxis
3.5.1. Vaccine in Adults
3.5.2. Vaccines in the Pediatric Population
3.5.3. Vaccines during Pregnancy
3.6. Post-Exposure Measures
3.7. Population, Ethics and Risk of Discrimination
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Moore, M.J.; Rathish, B.; Zahra, F. Monkeypox; Stat Pearls Publishing LLC: Treasure Island, FL, USA, 2022. Available online: www.ncbi.nlm.nih.gov/books/NBK574519/ (accessed on 18 November 2022).
- Ladnyj, I.D.; Ziegler, P.; Kima, E. A human infection caused by monkeypox virus in Basankusu Territory, Democratic Republic of the Congo. Bull. World Health Organ. 1972, 46, 593–597. [Google Scholar] [PubMed]
- Reynolds, M.G.; Yorita, K.L.; Kuehnert, M.J.; Davidson, W.B.; Huhn, G.D.; Holman, R.C.; Damon, I.K. Clinical Manifestations of Human Monkeypox Influenced by Route of Infection. J. Infect. Dis. 2006, 194, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Acharya, A.; Gendelman, H.E.; Byrareddy, S.N. The 2022 outbreak and the pathobiology of the monkeypox virus. J. Autoimmun. 2022, 131, 102855. [Google Scholar] [CrossRef] [PubMed]
- CDC. 2022 Monkeypox Outbreak Global Map. Available online: https://www.cdc.gov/poxvirus/monkeypox/response/2022/world-map.html (accessed on 18 November 2022).
- ECDC. 2022 Monkeypox Situation Update. Available online: https://www.ecdc.europa.eu/en/news-events/monkeypox-situation-update (accessed on 18 November 2022).
- WHO. Monkeypox Outbreak 2022. Available online: https://www.who.int/emergencies/situation-reports (accessed on 18 November 2022).
- Hennessee, I.; Shelus, V.; McArdle, C.E.; Wolf, M.; Schatzman, S.; Carpenter, A.; Minhaj, F.S.; Petras, J.K.; Cash-Goldwasser, S.; Maloney, M.; et al. Epidemiologic and Clinical Features of Children and Adolescents Aged < 18 Years with Monkeypox—United States, 17 May–24 September 2022. MMWR Morb. Mortal. Wkly. Rep. 2022, 71, 1407–1411. [Google Scholar] [CrossRef] [PubMed]
- WHO Director-General Declares the Ongoing Monkeypox Outbreak a Public Health Emergency of International Concern. Available online: https://www.who.int/europe/news/item/23-07-2022-who-director-general-declares-the-ongoing-monkeypox-outbreak-a-public-health-event-of-international-concern (accessed on 18 November 2022).
- Alakunle, E.; Moens, U.; Nchinda, G.; Okeke, M. Monkeypox Virus in Nigeria: Infection Biology, Epidemiology, and Evolution. Viruses 2020, 12, 1257. [Google Scholar] [CrossRef] [PubMed]
- Likos, A.M.; Sammons, S.A.; Olson, V.A.; Frace, A.M.; Li, Y.; Olsen-Rasmussen, M.; Davidson, W.; Galloway, R.; Khristova, M.L.; Reynolds, M.G.; et al. A tale of two clades: Monkeypox viruses. J. Gen. Virol. 2005, 86 Pt 10, 2661–2672. [Google Scholar] [CrossRef] [PubMed]
- WHO. Variants Names. Available online: https://www.who.int/news/item/12-08-2022-monkeypox--experts-give-virus-variants-new-names (accessed on 18 November 2022).
- Agrati, C.; Cossarizza, A.; Mazzotta, V.; Grassi, G.; Casetti, R.; De Biasi, S.; Pinnetti, C.; Gili, S.; Mondi, A.; Cristofanelli, F.; et al. Immunological Signature in Human Cases of Monkeypox Infection in 2022 Outbreak. Lancet Infect. Dis. 2022. [Google Scholar] [CrossRef]
- Johnston, S.C.; Johnson, J.C.; Stonier, S.W.; Lin, K.L.; Kisalu, N.K.; Hensley, L.E.; Rimoin, A.W. Cytokine modulation correlates with severity of monkeypox disease in humans. J. Clin. Virol. 2015, 63, 42–45. [Google Scholar] [CrossRef]
- Sanjuán, R.; Domingo-Calap, P. Mechanisms of viral mutation. Cell. Mol. Life Sci. 2016, 73, 4433–4448. [Google Scholar] [CrossRef] [Green Version]
- Gigante, C.M.; Plumb, M.; Ruprecht, A.; Zhao, H.; Wicker, V.; Wilkins, K.; Matheny, A. Genomic deletions and rearrangements in monkeypox virus from the 2022 outbreak, USA. bioRxiv 2022. [Google Scholar] [CrossRef]
- Wassenaar, T.M.; Wanchai, V.; Ussery, D.W. Comparison of Monkeypox virus genomes from the 2017 Nigeria outbreak and the 2022 outbreak. J. Appl. Microbiol. 2022, 133, 3690–3698. [Google Scholar] [CrossRef] [PubMed]
- Kozlov, M. The monkeypox virus is mutating. Are scientists worried? Nature 2022. epub ahead of print. [Google Scholar] [CrossRef] [PubMed]
- Angelo, K.M.; Petersen, B.W.; Hamer, D.H.; Schwartz, E.; Brunette, G. Monkeypox transmission among international travellers—Serious monkey business? J. Travel Med. 2019, 26, taz002. [Google Scholar] [CrossRef] [PubMed]
- York, A. The bodily distribution of monkeypox virus. Nat. Rev. Genet. 2022, 20, 703. [Google Scholar] [CrossRef] [PubMed]
- Palich, R.; Burrel, S.; Monsel, G.; Nouchi, A.; Bleibtreu, A.; Seang, S.; Bérot, V.; Brin, C.; Gavaud, C.; Wakim, Y.; et al. Viral loads in clinical samples of men with monkeypox virus infection: A French case series. Lancet Infect. Dis. 2022. [Google Scholar] [CrossRef]
- Lum, F.-M.; Torres-Ruesta, A.; Tay, M.Z.; Lin, R.T.P.; Lye, D.C.; Rénia, L.; Ng, L.F.P. Monkeypox: Disease epidemiology, host immunity and clinical interventions. Nat. Rev. Immunol. 2022, 22, 597–613. [Google Scholar] [CrossRef]
- Di Gennaro, F.; Veronese, N.; Marotta, C.; Shin, J.I.; Koyanagi, A.; Silenzi, A.; Antunes, M.; Saracino, A.; Bavaro, D.F.; Soysal, P.; et al. Human Monkeypox: A Comprehensive Narrative Review and Analysis of the Public Health Implications. Microorganisms 2022, 10, 1633. [Google Scholar] [CrossRef]
- Vouga, M.; Nielsen-Saines, K.; Dashraath, P.; Baud, D. The monkeypox outbreak: Risks to children and pregnant women. Lancet Child Adolesc. Health 2022, 6, 751–753. [Google Scholar] [CrossRef]
- D’Antonio, F.; Pagani, G.; Buca, D.; Khalil, A. Monkeypox infection in pregnancy: A systematic review and metaanalysis. Am. J. Obstet. Gynecol. MFM 2022, 5, 100747. [Google Scholar] [CrossRef]
- CDC. Monkeypox in Animals. Available online: https://www.cdc.gov/poxvirus/monkeypox/veterinarian/monkeypox-in-animals.html (accessed on 18 November 2022).
- Grant, R.; Nguyen, L.-B.L.; Breban, R. Modelling human-to-human transmission of monkeypox. Bull. World Health Organ. 2020, 98, 638–640. [Google Scholar] [CrossRef]
- WHO. Disease Outbreak News. Available online: https://www.who.int/emergencies/disease-outbreak-news/item/2022-DON396 (accessed on 18 November 2022).
- Pastula, D.M.; Tyler, K.L. An Overview of Monkeypox Virus and Its Neuroinvasive Potential. Ann. Neurol. 2022, 92, 527–531. [Google Scholar] [CrossRef]
- CDC. Clinical Recognition. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/clinical-recognition.html (accessed on 18 November 2022).
- Sejvar, J.J.; Chowdary, Y.; Schomogyi, M.; Stevens, J.; Patel, J.; Karem, K.; Fischer, M.; Kuehnert, M.J.; Zaki, S.R.; Paddock, C.D.; et al. Human Monkeypox Infection: A Family Cluster in the Midwestern United States. J. Infect. Dis. 2004, 190, 1833–1840. [Google Scholar] [CrossRef]
- Kaler, J.; Hussain, A.; Flores, G.; Kheiri, S.; Desrosiers, D. Monkeypox: A Comprehensive Review of Transmission, Pathogenesis, and Manifestation. Cureus 2022, 14, e26531. [Google Scholar] [CrossRef]
- Adalja, A.; Inglesby, T. A Novel International Monkeypox Outbreak. Ann. Intern. Med. 2022, 175, 1175–1176. [Google Scholar] [CrossRef]
- Nörz, D.; Pfefferle, S.; Brehm, T.T.; Franke, G.; Grewe, I.; Knobling, B.; Aepfelbacher, M.; Huber, S.; Klupp, E.M.; Jordan, S.; et al. Evidence of surface contamination in hospital rooms occupied by patients infected with monkeypox, Germany, June 2022. Eurosurveillance 2022, 27, 2200477. [Google Scholar] [CrossRef]
- CDC. Isolation and Infection Control at Home. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/infection-control-home.html (accessed on 18 November 2022).
- Dashraath, P.; Nielsen-Saines, K.; Mattar, C.; Musso, D.; Tambyah, P.; Baud, D. Guidelines for pregnant individuals with monkeypox virus exposure. Lancet 2022, 400, 21–22. [Google Scholar] [CrossRef]
- Bunge, E.M.; Hoet, B.; Chen, L.; Lienert, F.; Weidenthaler, H.; Baer, L.R.; Steffen, R. The changing epidemiology of human monkeypox—A potential threat? A systematic review. PLOS Negl. Trop. Dis. 2022, 16, e0010141. [Google Scholar] [CrossRef]
- Clinical Considerations for Monkeypox in Children and Adolescents. Available online: https://www.cdc.gov/poxvirus/monkeypox/clinicians/pediatric.html (accessed on 18 November 2022).
- Monkeypox—AAP Book American Academy of Pediatrics. Available online: https://www.aap.org/en/patient-care/monkeypox/ (accessed on 18 November 2022).
- Van Furth, A.M.T.; van der Kuip, M.; van Els, A.L.; Fievez, L.C.; van Rijckevorsel, G.G.; van den Ouden, A.; Jonges, M.; Welkers, M.R. Paediatric monkeypox patient with unknown source of infection, the Netherlands, June 2022. Eurosurveillance 2022, 27, 2200552. [Google Scholar] [CrossRef]
- CDC Schools, Early Care and Education Programs, and Other Settings Serving Children or Adolescents. Available online: www.cdc.gov/poxvirus/monkeypox/schools/faq.html (accessed on 18 November 2022).
- Ramnarayan, P.; Mitting, R.; Whittaker, E.; Marcolin, M.; O’Regan, C.; Sinha, R.; Bennett, A.; Moustafa, M.; Tickner, N.; Gilchrist, M.; et al. Neonatal Monkeypox Virus Infection. N. Engl. J. Med. 2022, 387, 1618–1620. [Google Scholar] [CrossRef]
- Obstetric Care Considerations for Monkeypox. Available online: https://www.acog.org/clinical-information/physician-faqs/obstetric-care-considerations-monkeypox (accessed on 18 November 2022).
- Wisner, K. Monkeypox: A Brief for Perinatal Nurses. MCN Am. J. Matern. Child Nurs. 2022. [Google Scholar] [CrossRef]
- Khalil, A.; Samara, A.; O’Brien, P.; Morris, E.; Draycott, T.; Lees, C.; Ladhani, S. Monkeypox and pregnancy: What do obstetricians need to know? Ultrasound Obstet. Gynecol. 2022, 60, 22–27. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.; Samara, A.; O’Brien, P.; Ladhani, S. Call for a unified approach to Monkeypox infection in pregnancy: Lessons from the COVID-19 pandemic. Nat. Commun. 2022, 13, 5038. [Google Scholar] [CrossRef] [PubMed]
- Khalil, A.; Samara, A.; O’Brien, P.; Coutinho, C.M.; Duarte, G.; Quintana, S.M.; Ladhani, S.N. Monkeypox in pregnancy: Update on current outbreak. Lancet Infect. Dis. 2022, 22, 1534–1535. [Google Scholar] [CrossRef] [PubMed]
- Rash Illness Testing Protocol. Acute, Generalized Vesicular or Pustular Rash Illness Testing Protocol in the United States. Available online: https://www.cdc.gov/smallpox/lab-personnel/laboratory-procedures/rash-testing.html (accessed on 15 October 2022).
- Mccollum, A.M.; Damon, I.K. Human Monkeypox. Clin. Infect. Dis. 2014, 58, 260–267. [Google Scholar] [CrossRef] [Green Version]
- Thornhill, J.P.; Barkati, S.; Walmsley, S.; Rockstroh, J.; Antinori, A.; Harrison, L.B.; Palich, R.; Nori, A.; Reeves, I.; Habibi, M.S.; et al. Monkeypox Virus Infection in Humans across 16 Countries—April–June 2022. N. Engl. J. Med. 2022, 387, 679–691. [Google Scholar] [CrossRef] [PubMed]
- WHO. Laboratory Testing for the Monkeypox Virus: Interim Guidance 23 May 2022. Available online: https://www.who.int/publications/i/item/WHO-MPX-laboratory-2022.1 (accessed on 18 November 2022).
- Singhal, T.; Kabra, S.K.; Lodha, R. Monkeypox: A Review. Indian J. Pediatr. 2022, 89, 955–960. [Google Scholar] [CrossRef] [PubMed]
- MacNeil, A.; Reynolds, M.; Braden, Z.; Carroll, D.S.; Bostik, V.; Karem, K.; Smith, S.K.; Davidson, W.; Li, Y.; Moundeli, A.; et al. Transmission of Atypical Varicella-Zoster Virus Infections Involving Palm and Sole Manifestations in an Area with Monkeypox Endemicity. Clin. Infect. Dis. 2009, 48, e6–e8. [Google Scholar] [CrossRef] [Green Version]
- Baud, D.; Nielsen-Saines, K.; Dashraath, P. Approach to monkeypox in pregnancy: Conjecture is best guided by evidence. Am. J. Obstet. Gynecol. 2022. epub ahead of print. [Google Scholar] [CrossRef]
- Webb, E.; Rigby, I.; Michelen, M.; Dagens, A.; Cheng, V.; Rojek, A.M.; Dahmash, D.; Khader, S.; Gedela, K.; Norton, A.; et al. Availability, scope and quality of monkeypox clinical management guidelines globally: A systematic review. BMJ Glob. Health 2022, 7, e009838. [Google Scholar] [CrossRef]
- CDC. Monkeypox. Available online: https://www.cdc.gov/poxvirus/monkeypox/if-sick/treatment.html#:~:text=There%20are%20no%20treatments%20specifically,and%20treat%20monkeypox%20virus%20infections (accessed on 18 November 2022).
- Adler, H.; Gould, S.; Hine, P.; Snell, L.B.; Wong, W.; Houlihan, C.F.; Osborne, J.C.; Rampling, T.; Beadsworth, M.B.; Duncan, C.J.; et al. NHS England High Consequence Infectious Diseases (Airborne) Network. Clinical features and management of human monkeypox: A retrospective observational study in the UK. Lancet Infect. Dis. 2022, 22, 1153–1162, Erratum in Lancet Infect. Dis. 2022, 22, e177. [Google Scholar] [CrossRef]
- Siegrist, A.E.; Sassine, J. Antivirals with Activity Against Monkeypox: A Clinically Oriented Review. Clin. Infect. Dis. 2022. epub ahead of print. [Google Scholar] [CrossRef]
- CDC. Guidance for Tecovirimat Use. Available online: https://www.cdc.gov/poxvirus/monkeypox/pdf/tecovirimat-ind-protocol-cdc-irb.pdf (accessed on 18 November 2022).
- Expanded Access IND Protocol: Use of Tecovirimat (TPOXX®) for Treatment of Human Non-Variola Orthopoxvirus Infections in Adults and Children IND No. 116,039 CDC IRB No. 6402 Version 6.1 10 August 2022. Available online: https://solanocounty.com/documents/TecovirimatINDProtocol_CDCIRB6402_v5.105.20.22_versionfor2022MPXOutbreak.pdf (accessed on 18 November 2022).
- O’Laughlin, K.; Tobolowsky, F.A.; Elmor, R.; Overton, R.; O’Connor, S.M.; Damon, I.K.; Petersen, B.W.; Rao, A.K.; Chatham-Stephens, K.; Yu, P.; et al. Clinical Use of Tecovirimat (Tpoxx) for Treatment of Monkeypox Under an Investigational New Drug Protocol—United States, May–August 2022. MMWR Morb. Mortal. Wkly. Rep. 2022, 71, 1190–1195. [Google Scholar] [CrossRef]
- Rabaan, A.A.; Abas, A.H.; Tallei, T.E.; Al-Zaher, M.A.; Al-Sheef, N.M.; Fatimawali; Al-Nass, E.Z.; Al-Ebrahim, E.A.; Effendi, Y.; Idroes, R.; et al. Monkeypox Outbreak 2022: What We Know So Far and Its Potential Drug Targets and Management Strategies. J. Med. Virol. 2022. epub ahead of print. [Google Scholar] [CrossRef]
- Red Book Online Outbreak: Monkeypox Virus Outbreak. Available online: https://publications.aap.org/redbook/resources/20705?autologincheck=redirected?nfToken=00000000-0000-0000-0000-000000000000 (accessed on 18 November 2022).
- Medscape Drug and Disease. Tecovirimat. Available online: https://reference.medscape.com/drug/tpoxx-tecovirimat-1000237 (accessed on 18 November 2022).
- EMA European Medicine Agency—Tecovirimat. Available online: www.ema.europa.eu/en/medicines/human/EPAR/tecovirimat-siga (accessed on 18 November 2022).
- Grosenbach, D.W.; Jordan, R.; Hruby, D.E. Development of the small-molecule antiviral ST-246® as a smallpox therapeutic. Futur. Virol. 2011, 6, 653–671. [Google Scholar] [CrossRef] [Green Version]
- Rizk, J.G.; Lippi, G.; Henry, B.M.; Forthal, D.N.; Rizk, Y. Prevention and Treatment of Monkeypox. Drugs 2022, 82, 957–963. [Google Scholar] [CrossRef]
- Priyamvada, L.; Carson, W.C.; Ortega, E.; Navarra, T.; Tran, S.; Smith, T.G.; Pukuta, E.; Muyamuna, E.; Kabamba, J.; Nguete, B.U.; et al. Serological responses to the MVA-based JYNNEOS monkeypox vaccine in a cohort of participants from the Democratic Republic of Congo. Vaccine 2022, 40, 7321–7327. [Google Scholar] [CrossRef]
- Islam, M.R.; Hossain, M.J.; Roy, A.; Hasan, A.H.M.N.; Rahman, M.A.; Shahriar, M.; Bhuiyan, M.A. Repositioning potentials of smallpox vaccines and antiviral agents in monkeypox outbreak: A rapid review on comparative benefits and risks. Health Sci. Rep. 2022, 5, e798. [Google Scholar] [CrossRef]
- Kozlov, M. Monkeypox vaccination begins—Can the global outbreaks be contained? Nature 2022, 606, 444–445. [Google Scholar] [CrossRef]
- US Centers for Disease Control and Prevention (CDC). Vaccination. Timing of Post-exposure Prophylaxis. Available online: https://www.cdc.gov/poxvirus/monkeypox/interim-considerations/overview.html (accessed on 18 November 2022).
- Overton, E.T.; Lawrence, S.J.; Stapleton, J.T.; Weidenthaler, H.; Schmidt, D.; Koenen, B.; Silbernagl, G.; Nopora, K.; Chaplin, P. A randomized phase II trial to compare safety and immunogenicity of the MVA-BN smallpox vaccine at various doses in adults with a history of AIDS. Vaccine 2020, 38, 2600–2607. [Google Scholar] [CrossRef]
- CDC—Interim Clinical Considerations for Use of JYNNEOS and ACAM2000 Vaccines during the 2022 U.S. Monkeypox Outbreak. Available online: https://www.cdc.gov/poxvirus/monkeypox/health-departments/vaccine-considerations.html (accessed on 18 November 2022).
- Afolabi, M.O.; Ishola, D.; Manno, D.; Keshinro, B.; Bockstal, V.; Rogers, B.; Owusu-Kyei, K.; Serry-Bangura, A.; Swaray, I.; Lowe, B.; et al. Safety and immunogenicity of the two-dose heterologous Ad26.ZEBOV and MVA-BN-Filo Ebola vaccine regimen in children in Sierra Leone: A randomised, double-blind, controlled trial. Lancet Infect. Dis. 2022, 22, 110–122. [Google Scholar] [CrossRef]
- O’Shea, J.; Filardo, T.D.; Morris, S.B.; Weiser, J.; Petersen, B.; Brooks, J.T. Interim Guidance for Prevention and Treatment of Monkeypox in Persons with HIV Infection—United States, August 2022. MMWR Morb. Mortal. Wkly. Rep. 2022, 71, 1023–1028. [Google Scholar] [CrossRef] [PubMed]
- Cohen, J.M.; Bamford, A.; Eisen, S.; Emonts, M.; Ho, D.; Kadambari, S.; Kenny, J.; Lyall, H.; Owens, S.; Porter, D.; et al. Royal College of Paediatrics and Child Health Monkeypox Working Group. Comment Title: Care of children exposed to monkeypox. Lancet Reg. Health Eur. 2022, 21, 100514. [Google Scholar] [CrossRef] [PubMed]
- FDA Highlights of Prescribing Information. CNJ-016, Vaccinia Immune Globulin Intravenous (Human). Available online: www.fda.gov/media/78174/download (accessed on 18 November 2022).
- Shrewsbury, D. Blame and shame are harming our response to monkeypox. BMJ 2022, 378, o2134. [Google Scholar] [CrossRef]
- Isaacs, D. Monkeypox. J. Paediatr. Child Health 2022, 58, 1290–1292. [Google Scholar] [CrossRef]
- JYNNEOS® (MVA-BN) Smallpox (Monkeypox) Vaccine Description. Available online: https://www.precisionvaccinations.com/vaccines/jynneos-smallpox-monkeypox-vaccine (accessed on 18 November 2022).
- Forni, D.; Cagliani, R.; Molteni, C.; Clerici, M.; Sironi, M. Monkeypox virus: The changing facets of a zoonotic pathogen. Infect. Genet. Evol. 2022, 105, 105372. [Google Scholar] [CrossRef]
- Mukherjee, A.G.; Wanjari, U.R.; Kannampuzha, S.; Das, S.; Murali, R.; Namachivayam, A.; Renu, K.; Ramanathan, G.; Doss, C.; Vellingiri, B.; et al. The pathophysiological and immunological background of the monkeypox virus infection: An update. J. Med. Virol. 2022. epub ahead of print. [Google Scholar] [CrossRef]
- Technical Report 3. Available online: https://www.cdc.gov/poxvirus/monkeypox/cases-data/technical-report/report-3.html (accessed on 18 November 2022).
- Yao, K. The diversity of clinical manifestations of human monkeypox should be emphasized in practice. Pediatr. Investig. 2022, 6, 224–225. [Google Scholar] [CrossRef]
| Oral ** | Intravenous |
|---|---|
The latest update published by the CDC (version 6.2 24 October 2022) on the use of Tecovirimat in children [60] provides additional indications of its use in children from birth:
| <35 kg: 6 mg/kg by IV infusion in <6 h. Volume of infusion depends on the weight. In children < 2 years of age,
For children <3 kg the drug is allowed according to the CDC report on treatment of 24 October 2022, but dose adjustments may be necessary in infants based on the general condition and weight [60]. |
| ACAM2000 | MVA-BN ** | LC16m8 |
|---|---|---|
| (Live vaccinia virus) Second-generation vaccine | (Attenuated, non-replicating vaccinia virus) | (Modified vaccinia virus) |
| Administration by multiple percutaneous puncture device of the skin surface. Single dose with lesion at the site of inoculation. Not recommended in A. Individuals with
Possible adverse cardiac reactions. | Administration in two subcutaneous doses, 4 weeks apart, no lesions at the inoculum site. Safe for immunocompromised patients because the virus does not replicate. If not large available, a single dose may be administered. | Single dose administration. Licensed in Japan Safer then ACAM2000, given the lower replication capacity. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gaeta, F.; De Caro, F.; Franci, G.; Pagliano, P.; Vajro, P.; Mandato, C. Monkeypox Infection 2022: An Updated Narrative Review Focusing on the Neonatal and Pediatric Population. Children 2022, 9, 1832. https://doi.org/10.3390/children9121832
Gaeta F, De Caro F, Franci G, Pagliano P, Vajro P, Mandato C. Monkeypox Infection 2022: An Updated Narrative Review Focusing on the Neonatal and Pediatric Population. Children. 2022; 9(12):1832. https://doi.org/10.3390/children9121832
Chicago/Turabian StyleGaeta, Francesca, Francesco De Caro, Gianluigi Franci, Pasquale Pagliano, Pietro Vajro, and Claudia Mandato. 2022. "Monkeypox Infection 2022: An Updated Narrative Review Focusing on the Neonatal and Pediatric Population" Children 9, no. 12: 1832. https://doi.org/10.3390/children9121832






