A Value-Added Utilization Method of Sugar Production By-Products from Rice Straw: Extraction of Lignin and Evaluation of Its Antioxidant Activity
Abstract
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Process for Producing Glucose from Rice Straw
2.3. Lignin Extraction
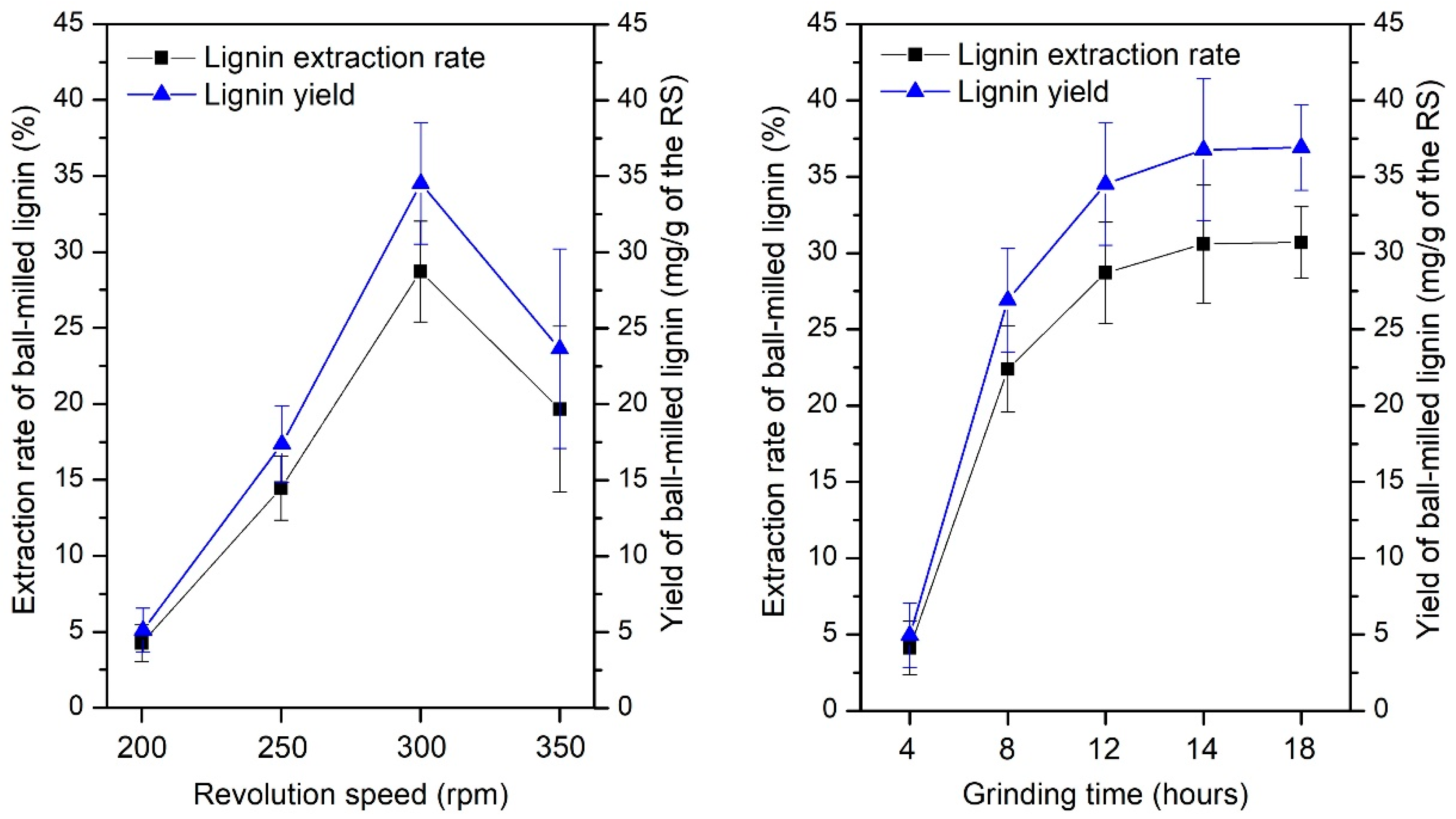
2.3.1. Extraction of Lignin from PTA Pretreatment Concentrated Filtrate
2.3.2. Extraction of Lignin from EH Residue
2.4. Analysis and Characterization
2.4.1. Chemical Composition Analysis
2.4.2. Determination of Phenolic Hydroxyl Groups
2.4.3. Determination of Molecular Weight
2.4.4. FT-IR Analysis
2.4.5. Calculation Formulas and Data Analysis
2.5. Evaluation of Antioxidant Activity
3. Results and Discussion
3.1. Effects of Treatment Process on Chemical Composition of Rice Straw
3.2. Subsection
3.3. Characterization of Different Lignin Samples
3.4. Antioxidative Activities of Different Lignin Samples
3.5. Mass Balance
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tao, J.M.; Li, S.; Ye, F.Y.; Zhou, Y.; Lei, L.; Zhao, G.H. Lignin—An underutilized, renewable and valuable material for food industry. Crit. Rev. Food Sci. Nutr. 2020, 60, 2011–2033. [Google Scholar] [CrossRef] [PubMed]
- Sun, R.C. Lignin Source and Structural Characterization. ChemSusChem 2020, 13, 4385–4393. [Google Scholar] [CrossRef] [PubMed]
- Rinaldi, R.; Jastrzebski, R.; Clough, M.T.; Ralph, J.; Kennema, M.; Bruijnincx, P.C.A.; Weckhuysen, B.M. Paving the Way for Lignin Valorisation: Recent Advances in Bioengineering, Biorefining and Catalysis. Angew. Chem. Int. Ed. 2016, 55, 8164–8215. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kumar, A.; Anushree; Kumar, J.; Bhaskar, T. Utilization of lignin: A sustainable and eco-friendly approach. J. Energy Inst. 2020, 93, 235–271. [Google Scholar] [CrossRef]
- Houfani, A.A.; Anders, N.; Spiess, A.C.; Baldrian, P.; Benallaoua, S. Insights from enzymatic degradation of cellulose and hemicellulose to fermentable sugars–A review. Biomass Bioenergy 2020, 134, 105481. [Google Scholar] [CrossRef]
- Zhang, C.F.; Wang, F. Catalytic Lignin Depolymerization to Aromatic Chemicals. Acc. Chem. Res. 2020, 53, 470–484. [Google Scholar] [CrossRef] [PubMed]
- Shu, R.Y.; Li, R.X.; Lin, B.Q.; Wang, C.; Cheng, Z.D.; Chen, Y. A review on the catalytic hydrodeoxygenation of lignin-derived phenolic compounds and the conversion of raw lignin to hydrocarbon liquid fuels. Biomass Bioenergy 2020, 132, 105432. [Google Scholar] [CrossRef]
- Morgana, M.; Viola, E.; Zimbardi, F.; Cerone, N.; Romanelli, A.; Valerio, V. Depolymerization and Hydrogenation of Organosolv Eucalyptus Lignin by Using Nickel Raney Catalyst. Processes 2021, 9, 1093. [Google Scholar] [CrossRef]
- Chio, C.L.; Sain, M.; Qin, W.S. Lignin utilization: A review of lignin depolymerization from various aspects. Renew. Sustain. Energy Rev. 2019, 107, 232–249. [Google Scholar] [CrossRef]
- Torres, L.A.Z.; Woiciechowski, A.L.; Tanobe, V.O.D.; Karp, S.G.; Lorenci, L.C.G.; Faulds, C.; Soccol, C.R. Lignin as a potential source of high-added value compounds: A review. J. Clean. Prod. 2020, 263, 121499. [Google Scholar] [CrossRef]
- Yoo, C.G.; Meng, X.Z.; Pu, Y.Q.; Ragauskas, A.J. The critical role of lignin in lignocellulosic biomass conversion and recent pretreatment strategies: A comprehensive review. Bioresour. Technol. 2020, 301, 122784. [Google Scholar] [CrossRef] [PubMed]
- Shen, X.J.; Sun, R.C. Recent advances in lignocellulose prior-fractionation for biomaterials, biochemicals, and bioenergy. Carbohydr. Polym. 2021, 261, 117884. [Google Scholar] [CrossRef] [PubMed]
- Chiorcea-Paquim, A.M.; Enache, T.A.; Gil, E.D.; Oliveira-Brett, A.M. Natural phenolic antioxidants electrochemistry: Towards a new food science methodology. Compr. Rev. Food Sci. Food Saf. 2020, 19, 1680–1726. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.M.; Yuan, T.Q.; Song, G.Y.; Sun, R.C. Advanced and versatile lignin-derived biodegradable composite film materials toward a sustainable world. Green Chem. 2021, 23, 3790–3817. [Google Scholar] [CrossRef]
- Piccinino, D.; Capecchi, E.; Tomaino, E.; Gabellone, S.; Gigli, V.; Avitabile, D.; Saladino, R. Nano-Structured Lignin as Green Antioxidant and UV Shielding Ingredient for Sunscreen Applications. Antioxidants 2021, 10, 274. [Google Scholar] [CrossRef] [PubMed]
- Lauberte, L.; Telysheva, G.; Cravotto, G.; Andersone, A.; Janceva, S.; Dizhbite, T.; Arshanitsa, A.; Jurkjane, V.; Vevere, L.; Grillo, G.; et al. Lignin-Derived antioxidants as value-added products obtained under cavitation treatments of the wheat straw processing for sugar production. J. Clean. Prod. 2021, 303, 126369. [Google Scholar] [CrossRef]
- Tha, E.L.; Matos, M.; Avelino, F.; Lomonaco, D.; Rodrigues-Souza, I.; Gagosian, V.S.C.; Cestari, M.M.; Magalhaes, W.L.E.; Leme, D.M. Safety aspects of kraft lignin fractions: Discussions on the in chemico antioxidant activity and the induction of oxidative stress on a cell-based in vitro model. Int. J. Biol. Macromol. 2021, 182, 977–986. [Google Scholar] [CrossRef]
- Wei, X.X.; Liu, Y.; Luo, Y.D.; Shen, Z.; Wang, S.F.; Li, M.F.; Zhang, L.M. Effect of organosolv extraction on the structure and antioxidant activity of eucalyptus kraft lignin. Int. J. Biol. Macromol. 2021, 187, 462–470. [Google Scholar] [CrossRef]
- Su, C.; Gan, T.; Liu, Z.L.; Chen, Y.; Zhou, Q.; Xia, J.Y.; Cao, Y.F. Enhancement of the antioxidant abilities of lignin and lignin-carbohydrate complex from wheat straw by moderate depolymerization via LiCl/DMSO solvent catalysis. Int. J. Biol. Macromol. 2021, 184, 369–379. [Google Scholar] [CrossRef]
- Sapouna, I.; Lawoko, M. Deciphering lignin heterogeneity in ball milled softwood: Unravelling the synergy between the supramolecular cell wall structure and molecular events. Green Chem. 2021, 23, 3348–3364. [Google Scholar] [CrossRef]
- Qin, Z.; Yang, Q.L.; Cheng, X.C.; Liu, H.M.; Wang, X.D. Structural features, chemical composition, antioxidant activities of organosolv lignins extracted from black and white sesame capsules and stalks. Ind. Crops Prod. 2021, 169, 113677. [Google Scholar] [CrossRef]
- Hasan, G.; Musajan, D.; He, M.Y.; Hou, G.B.; Li, Y.; Yimit, M. Study on extraction of cotton stalk lignin by different methods and its antioxidant property in polypropylene. Sep. Sci. Technol. 2022, 57, 263–273. [Google Scholar] [CrossRef]
- Zhang, S.M.; Zhang, Y.; Liu, L.; Fang, G.Z. Antioxidant Activity of Organosolv Lignin Degraded Using SO42−/ZrO2 as Catalyst. BioResources 2015, 10, 6819–6829. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.M.; Liu, L.; Fang, G.Z.; Yan, N.; Ren, S.X.; Ma, Y.L. Hydrogenolysis and Activation of Soda Lignin Using [BMIM]Cl as a Catalyst and Solvent. Polymers 2017, 9, 279. [Google Scholar] [CrossRef] [Green Version]
- Zhang, S.M.; Su, L.; Liu, L.; Fang, G.Z. Degradation on hydrogenolysis of soda lignin using CuO/SO42−/ZrO2 as catalyst. Ind. Crops Prod. 2015, 77, 451–457. [Google Scholar] [CrossRef]
- Zhang, S.M.; Fang, G.Z.; Chen, H.T.; Lang, Q. The Effect of Degradation of Soda Lignin Using Pd/SO42−/ZrO2 as a Catalyst: Improved Reactivity and Antioxidant Activity. Polymers 2019, 11, 1218. [Google Scholar] [CrossRef] [Green Version]
- Yang, S.; Yuan, T.Q.; Sun, R.C. Structural Elucidation of Whole Lignin in Cell Walls of Triploid of Populus tomentosa Carr. ACS Sustainable Chem. Eng. 2016, 4, 1006–1015. [Google Scholar] [CrossRef]
- Zinovyev, G.; Sumerskii, I.; Rosenau, T.; Balakshin, M.; Potthast, A. Ball Milling’s Effect on Pine Milled Wood Lignin’s Structure and Molar Mass. Molecules 2018, 23, 2223. [Google Scholar] [CrossRef] [Green Version]
- Sitotaw, Y.W.; Habtu, N.G.; Gebreyohannes, A.Y.; Nunes, S.P.; Van Gerven, T. Ball milling as an important pretreatment technique in lignocellulose biorefineries: A review. Biomass Convers. Biorefin. 2021. [Google Scholar] [CrossRef]
- Dou, J.Z.; Kim, H.; Li, Y.D.; Padmakshan, D.; Yue, F.X.; Ralph, J.; Vuorinen, T. Structural Characterization of Lignins from Willow Bark and Wood. J. Agric. Food Chem. 2018, 66, 7294–7300. [Google Scholar] [CrossRef]
- Samuel, R.; Pu, Y.Q.; Raman, B.; Ragauskas, A.J. Structural Characterization and Comparison of Switchgrass Ball-milled Lignin Before and After Dilute Acid Pretreatment. Appl. Biochem. Biotechnol. 2010, 162, 62–74. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.M.; Yuan, T.Q.; Peng, F.; Xu, F.; Sun, R.C. Separation and Structural Characterization of Lignin from Hybrid Poplar Based on Complete Dissolution in DMSO/LiCl. Sep. Sci. Technol. 2010, 45, 2497–2506. [Google Scholar] [CrossRef]
- Chen, W.J.; Zhao, B.C.; Cao, X.F.; Yuan, T.Q.; Shi, Q.; Wang, S.F.; Sun, R.C. Structural Features of Alkaline Dioxane Lignin and Residual Lignin from Eucalyptus grandis × E. urophylla. J. Agric. Food Chem. 2019, 67, 968–974. [Google Scholar] [CrossRef] [PubMed]
- Zhang, S.M.; Mei, T.H.; Zhu, C.H.; Shang, H.M.; Gao, S.S.; Qin, L.Y.; Chen, H.T. A Combination Method of Liquid Hot Water and Phosphotungstic Acid Pretreatment for Improving the Enzymatic Saccharification Efficiency of Rice Straw. Energies 2022, 15, 3636. [Google Scholar] [CrossRef]
- Sluiter, A.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D. Determination of Extractives in Biomass. Technical Report NREL/TP-510-42619. Available online: https://www.nrel.gov/docs/gen/fy08/42619.pdf (accessed on 17 July 2005).
- Sluiter, A.; Hames, B.; Ruiz, R.; Scarlata, C.; Sluiter, J.; Templeton, D.; Crocker, D. Determination of Structural Carbohydrates and Lignin in Biomass. Technical Report NREL/TP-510-42618. Available online: https://www.nrel.gov/docs/gen/fy13/42618.pdf (accessed on 3 August 2012).
- Gärtner, A.; Gellerstedt, G.; Tamminen, T. Determination of phenolic hydroxyl groups in residual lignin using a modified UV-method. Nord. Pulp Pap. Res. J. 1999, 14, 163–170. [Google Scholar] [CrossRef]
- Guerra, A.; Filpponen, I.; Lucia, L.; Argyropoulos, D.S. Comparative evaluation of three lignin isolation protocols for various wood species. J. Agric. Food Chem. 2006, 54, 9696–9705. [Google Scholar] [CrossRef]
- Yang, G.R.; An, X.Y.; Yang, S.L. The Effect of Ball Milling Time on the Isolation of Lignin in the Cell Wall of Different Biomass. Front. Bioeng. Biotech. 2021, 9, 807625. [Google Scholar] [CrossRef]
- Wen, J.L.; Sun, S.L.; Yuan, T.Q.; Sun, R.C. Structural elucidation of whole lignin from Eucalyptus based on preswelling and enzymatic hydrolysis. Green Chem. 2015, 17, 1589–1596. [Google Scholar] [CrossRef]
- Zhang, A.P.; Liu, C.F.; Sun, R.C.; Xie, J. Extraction, Purification, and Characterization of Lignin Fractions from Sugarcane Bagasse. BioResources 2013, 8, 1604–1614. [Google Scholar] [CrossRef] [Green Version]
- Wang, Z.W.; Zhu, X.T.; Deuss, P.J. The effect of ball milling on birch, pine, reed, walnut shell enzymatic hydrolysis recalcitrance and the structure of the isolated residual enzyme lignin. Ind. Crops Prod. 2021, 167, 113493. [Google Scholar] [CrossRef]
- Wang, H.Y.; Zhao, L.H.; Ren, J.L.; He, B.H. Structural Changes of Alkali Lignin under Ozone Treatment and Effect of Ozone-Oxidized Alkali Lignin on Cellulose Digestibility. Processes 2022, 10, 559. [Google Scholar] [CrossRef]
- Faix, O. Classification of Lignins from Different Botanical Origins by FT-IR Spectroscopy. Holzforschung 1991, 45, 21–27. [Google Scholar] [CrossRef]



| Run | Sample | Solid Recovery | Chemical Composition (%) | ||||
|---|---|---|---|---|---|---|---|
| (%) | Cellulose | Hemicellulose | Lignin | Extract | Moisture | ||
| — 1 | RS | 100 | 37.80 ± 0.08 | 18.39 ± 0.26 | 14.60 ± 0.16 | 11.34 ± 0.23 | 3.70 ± 0.10 |
| Soxhlet extraction | Extract-free RS | 85.02 ± 0.28 | 44.47 ± 0.13 | 21.64 ± 0.31 | 17.18 ± 0.20 | N.D. 2 | N.D. |
| Pretreatment | LHW-PTA RS | 60.38 ± 0.42 | 55.12 ± 0.21 | 4.38 ± 0.35 | 19.67 ± 0.31 | N.D. | N.D. |
| Enzymatic hydrolysis | EH residue | 33.83 ± 0.72 | 24.43 ± 0.86 | 3.66 ± 0.18 | 35.54 ± 0.51 | N.D. | N.D. |
| Sample | Purity (%) | Phenolic Hydroxyl Groups (mmol·g−1) | Mw (g·mol−1) | Mn (g·mol−1) | Mw/Mn |
|---|---|---|---|---|---|
| Ether-extracted lignin | 98.7 ± 0.3 | 2.40 ± 0.02 | 3200 | 1400 | 2.29 |
| Ball-milled lignin | 99.6 ± 0.1 | 2.21 ± 0.01 | 10,200 | 6800 | 1.50 |
| Soda lignin | 92.2 ± 0.2 | 1.70 ± 0.03 | 7700 | 4900 | 1.57 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, S.; Ma, L.; Gao, S.; Zhu, C.; Yan, Y.; Liu, X.; Li, L.; Chen, H. A Value-Added Utilization Method of Sugar Production By-Products from Rice Straw: Extraction of Lignin and Evaluation of Its Antioxidant Activity. Processes 2022, 10, 1210. https://doi.org/10.3390/pr10061210
Zhang S, Ma L, Gao S, Zhu C, Yan Y, Liu X, Li L, Chen H. A Value-Added Utilization Method of Sugar Production By-Products from Rice Straw: Extraction of Lignin and Evaluation of Its Antioxidant Activity. Processes. 2022; 10(6):1210. https://doi.org/10.3390/pr10061210
Chicago/Turabian StyleZhang, Shengming, Liangfei Ma, Shushan Gao, Chonghao Zhu, Yehong Yan, Xiaohan Liu, Longhai Li, and Haitao Chen. 2022. "A Value-Added Utilization Method of Sugar Production By-Products from Rice Straw: Extraction of Lignin and Evaluation of Its Antioxidant Activity" Processes 10, no. 6: 1210. https://doi.org/10.3390/pr10061210







