Effect of Interaction between Carbon Dioxide and Fluid Phase/Rock Interface on Carbon Dioxide Storage
Abstract
:1. Introduction
2. Experimental Part
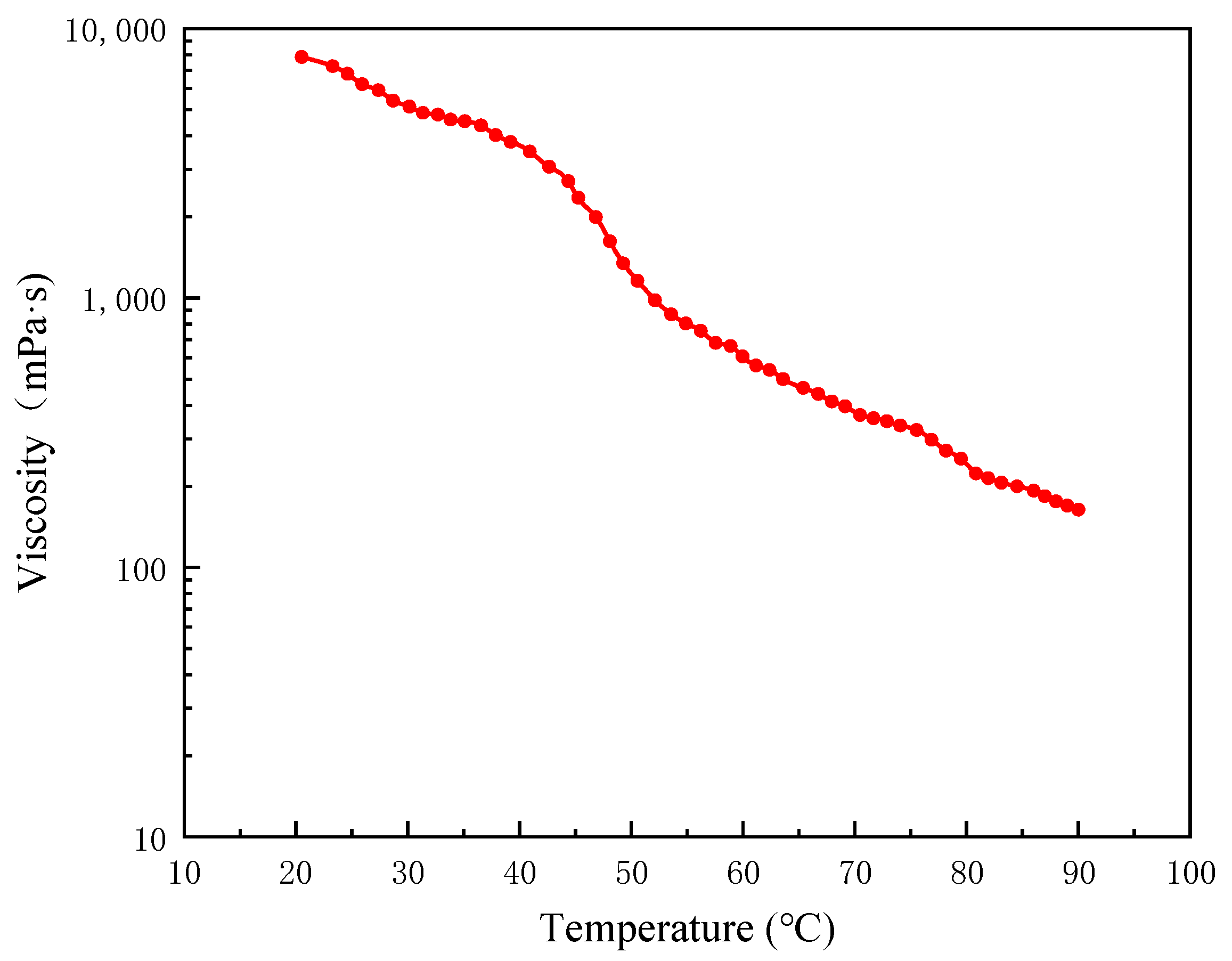
2.1. Experimental Materials
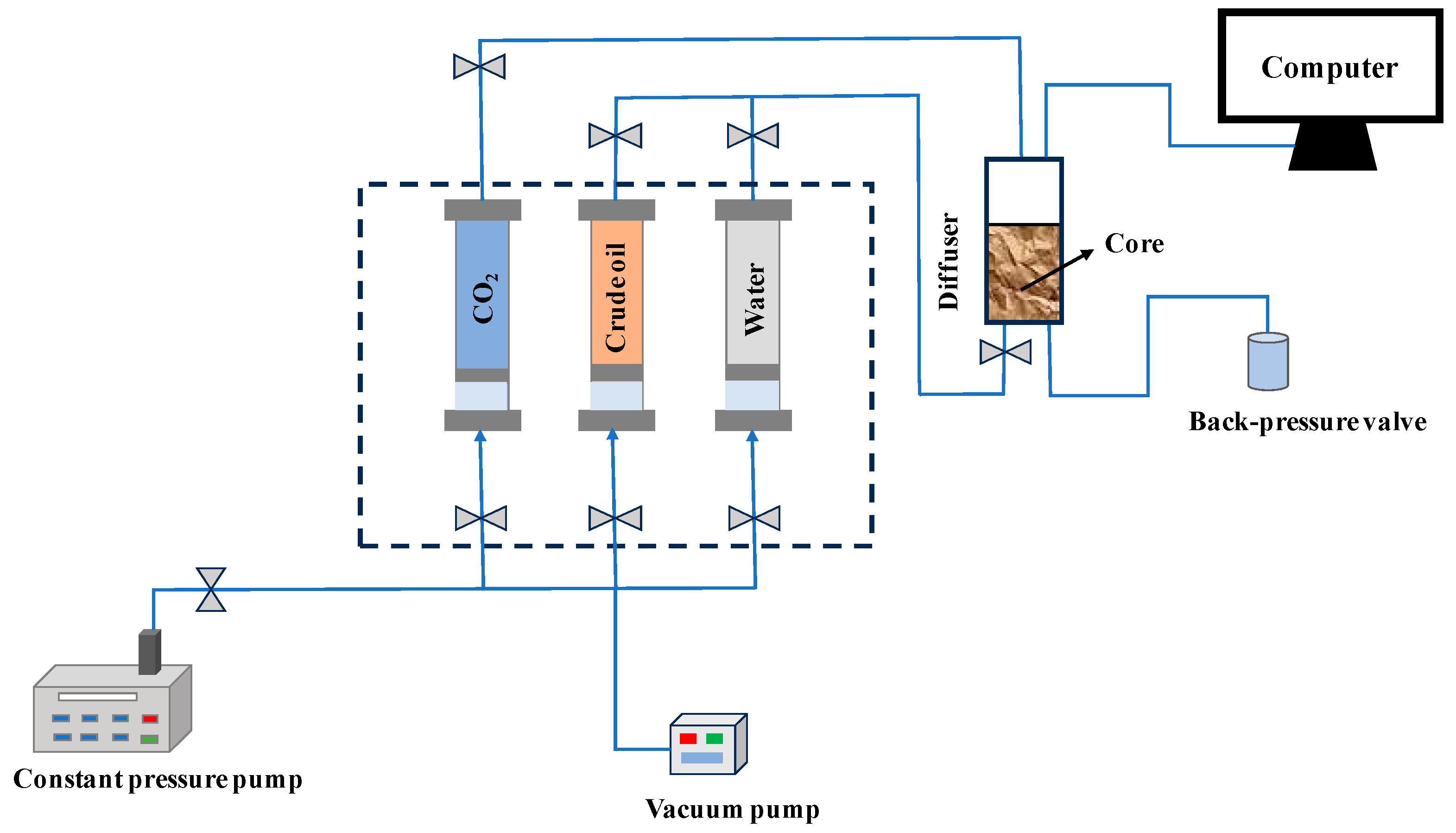
2.2. Experimental Devices
2.3. Experimental Methods
2.3.1. Solubility Measurement of CO2 in Pure Liquid
2.3.2. Diffusion Experiment with CO2 in Pure Liquid
2.3.3. Diffusion Measurement of CO2 in Porous Medium
2.3.4. Static Erosion Experiment
2.3.5. Dynamic Erosion Experiment
2.3.6. Dynamic Flooding Experiment
3. Results and Discussion
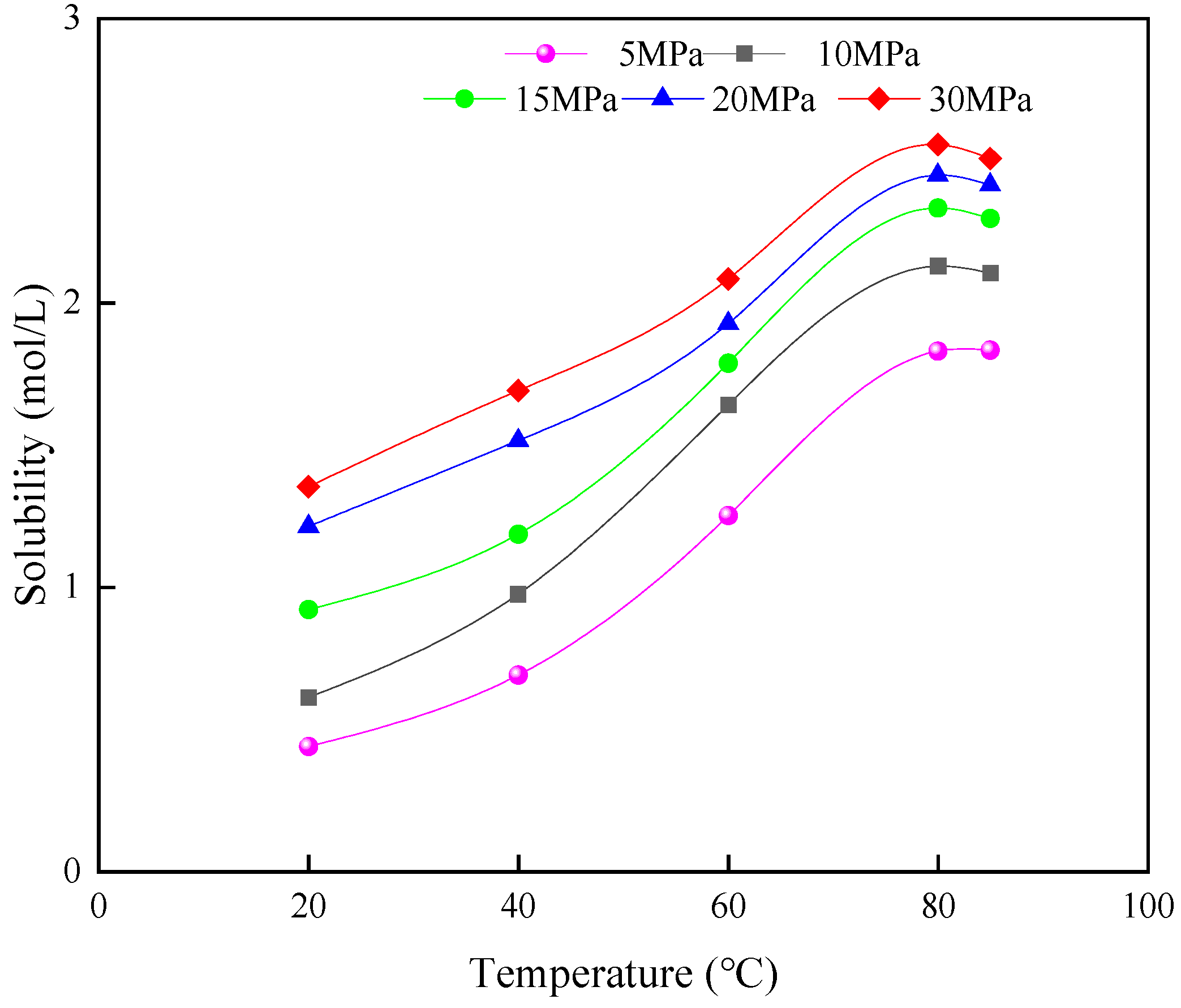
3.1. Solubility of CO2 in the Formation Fluid
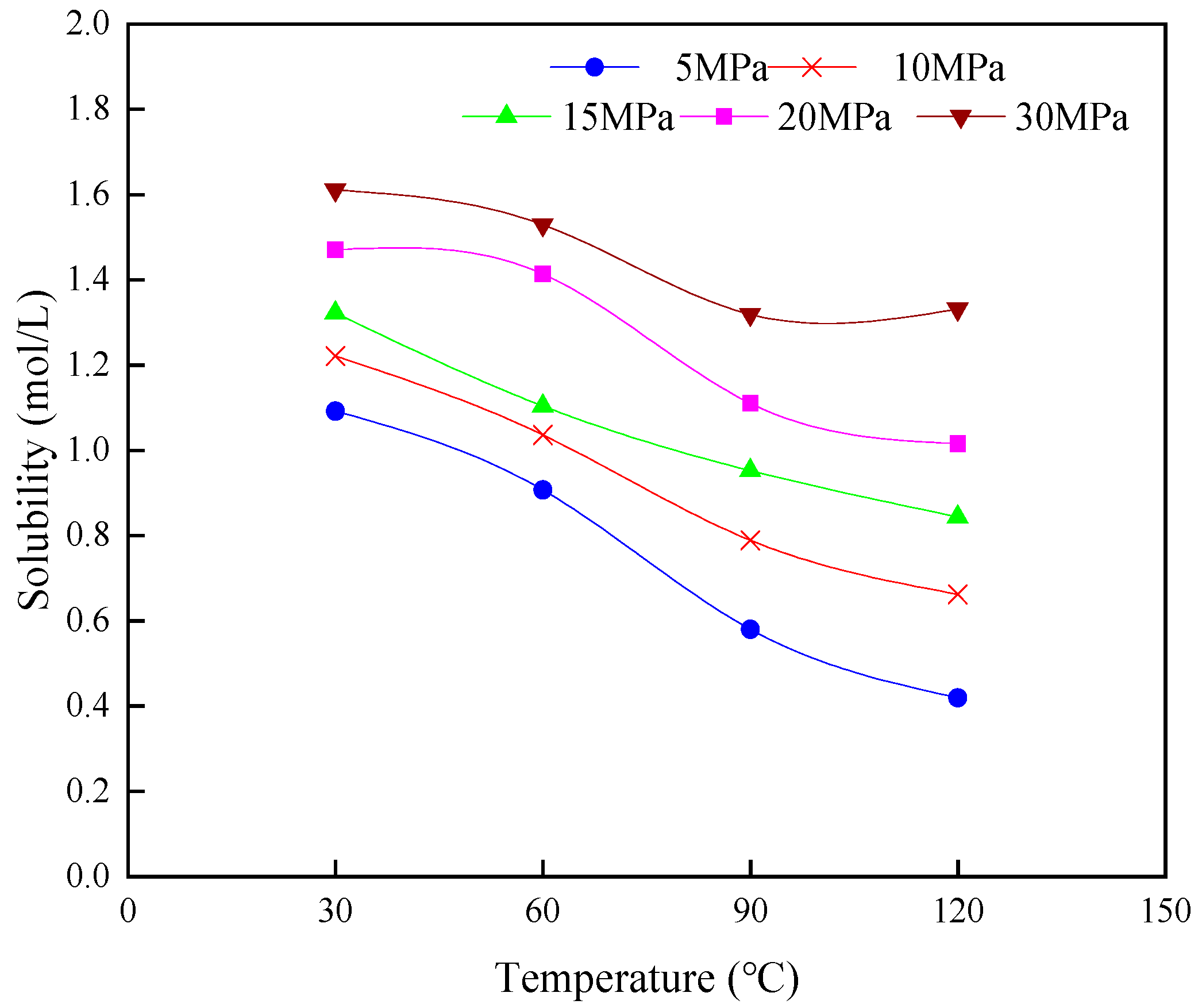
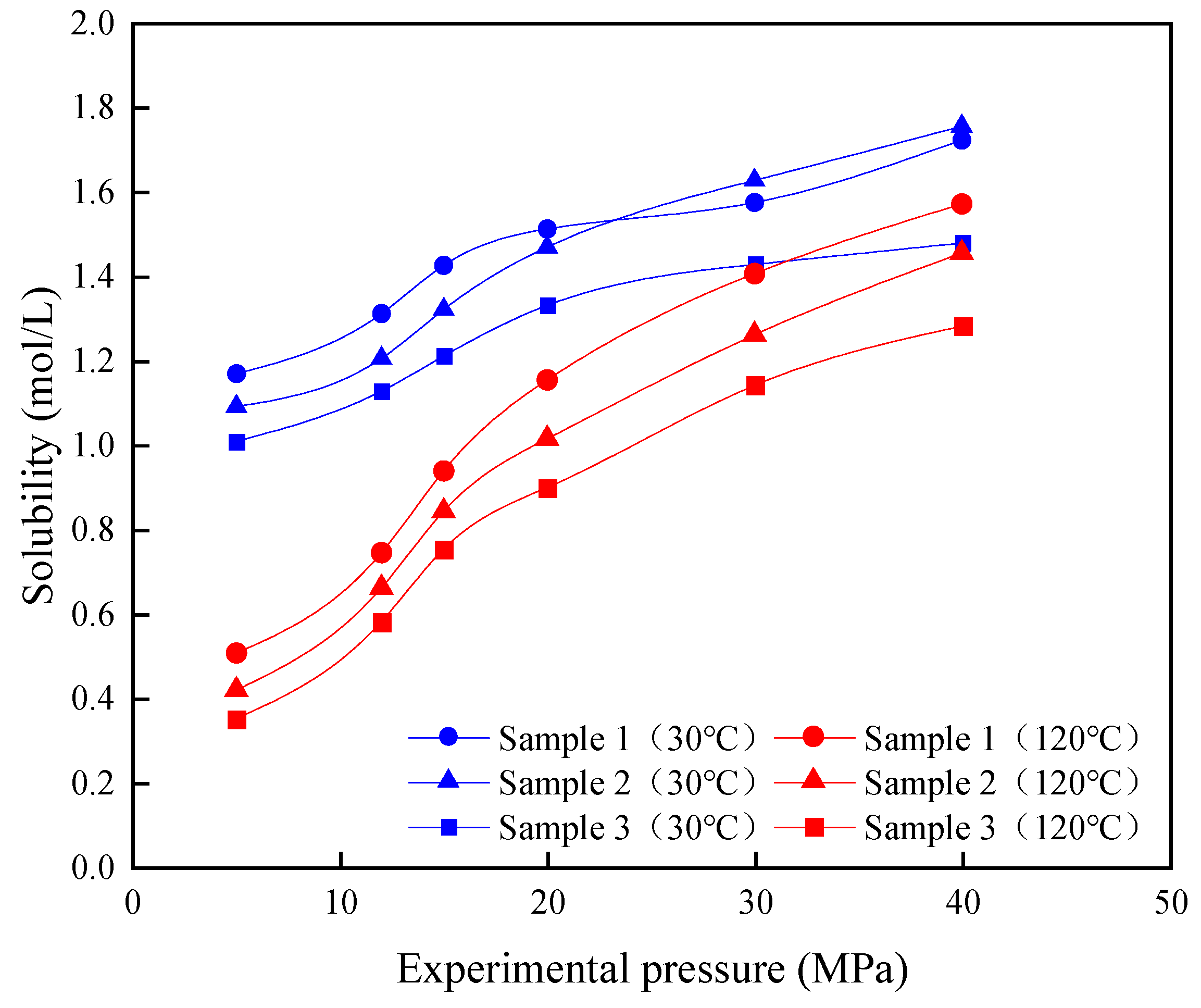
3.1.1. Solubility of CO2 in the Formation Water
3.1.2. Solubility of CO2 in Crude Oil
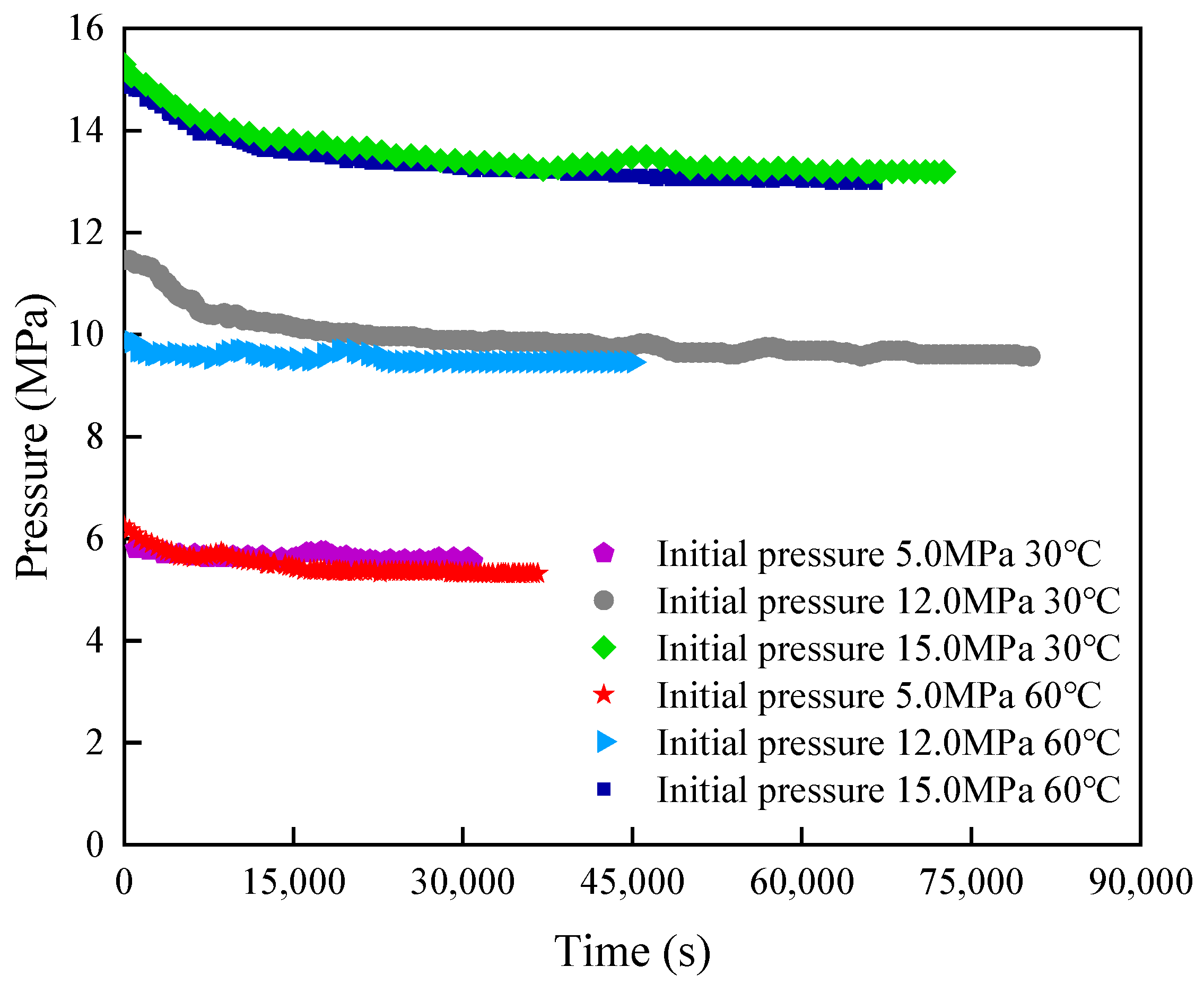
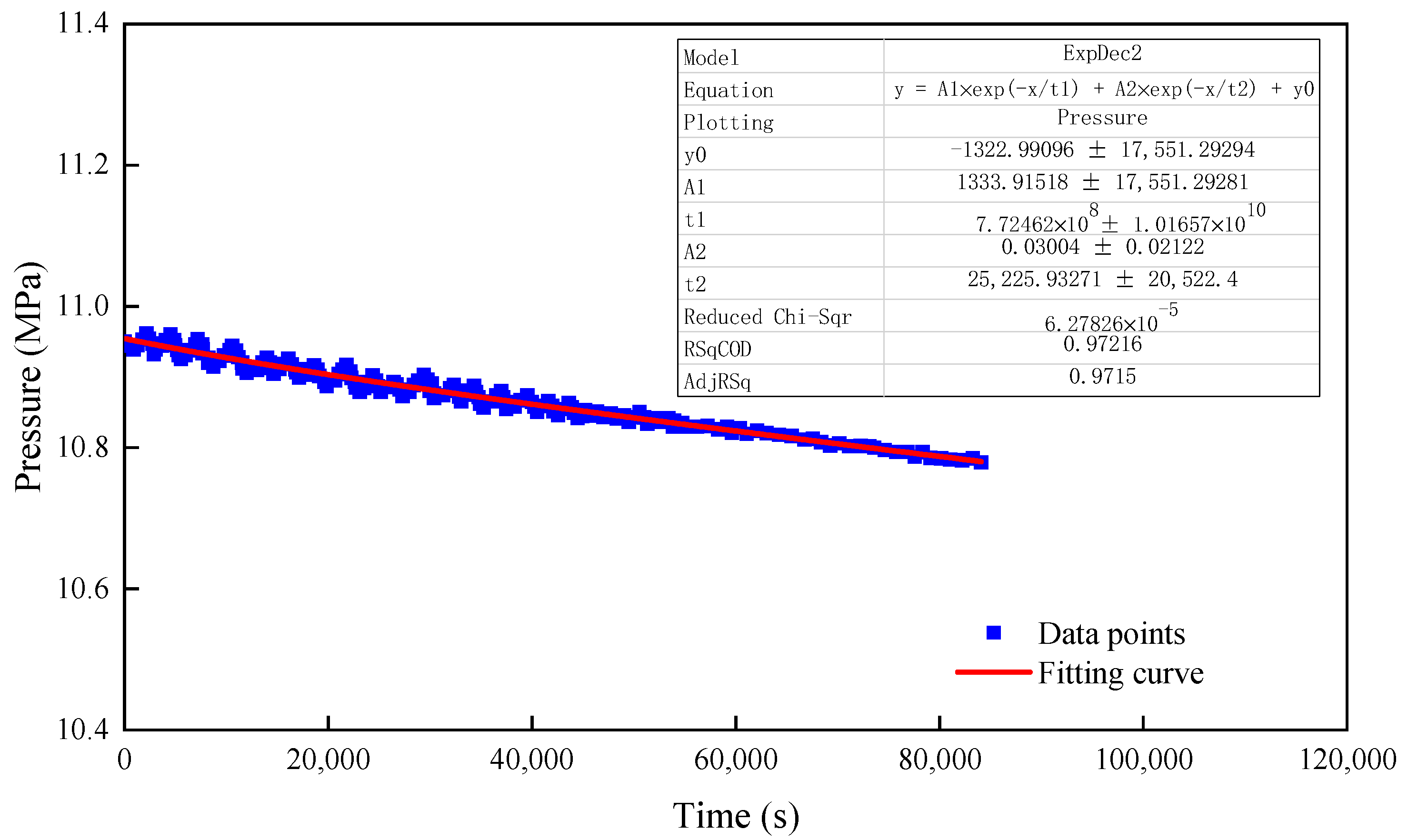
3.2. Diffusion Law of CO2 in the Formation Fluid
3.2.1. Diffusion Experiment Involving CO2 in Pure Water
3.2.2. Diffusion Experiment Involving CO2 in Crude Oil
3.2.3. Diffusion Experiment Involving CO2 in Porous Medium
3.3. Interaction of CO2–Water–Rock System
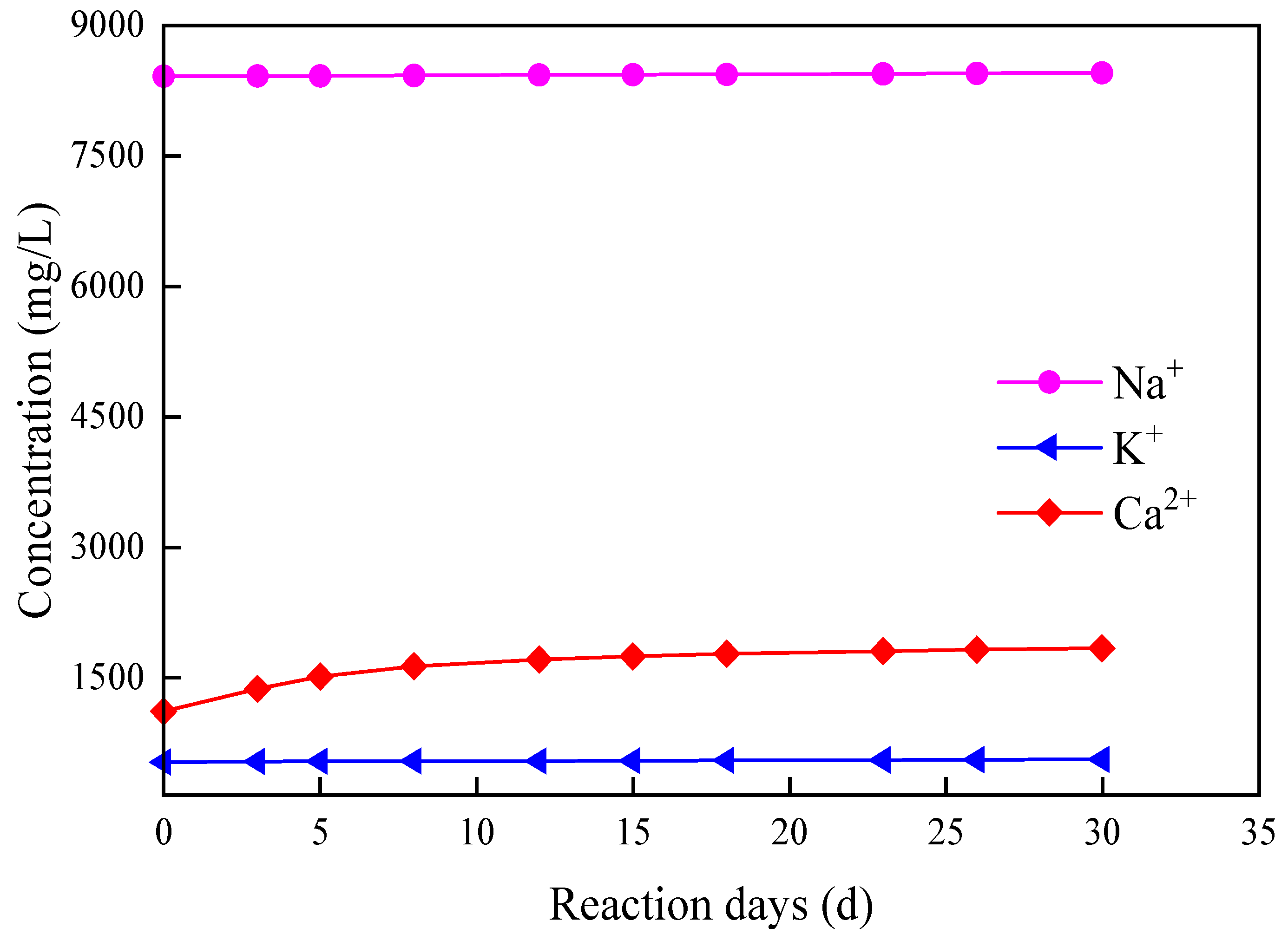
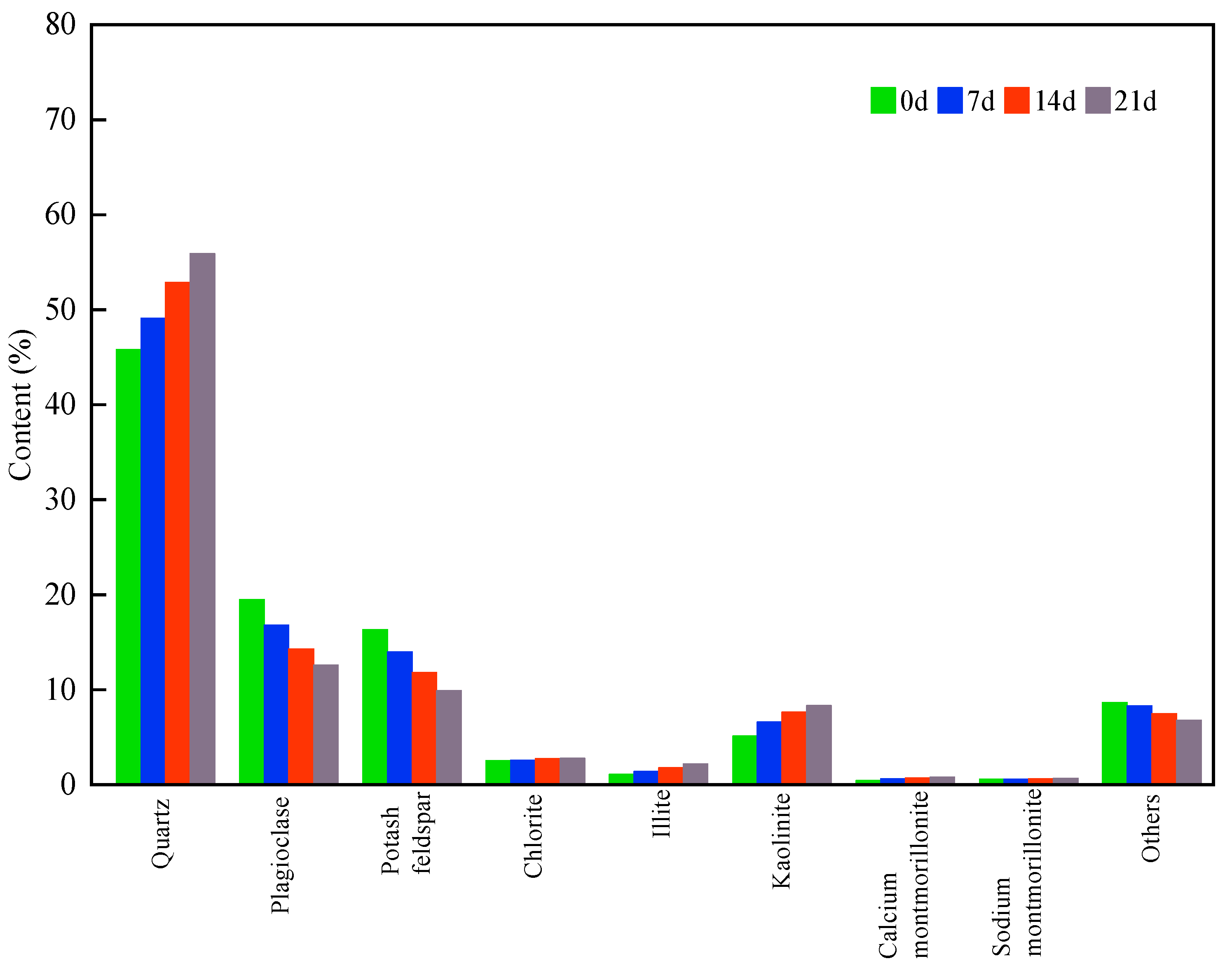
3.3.1. Static Erosion Experiment
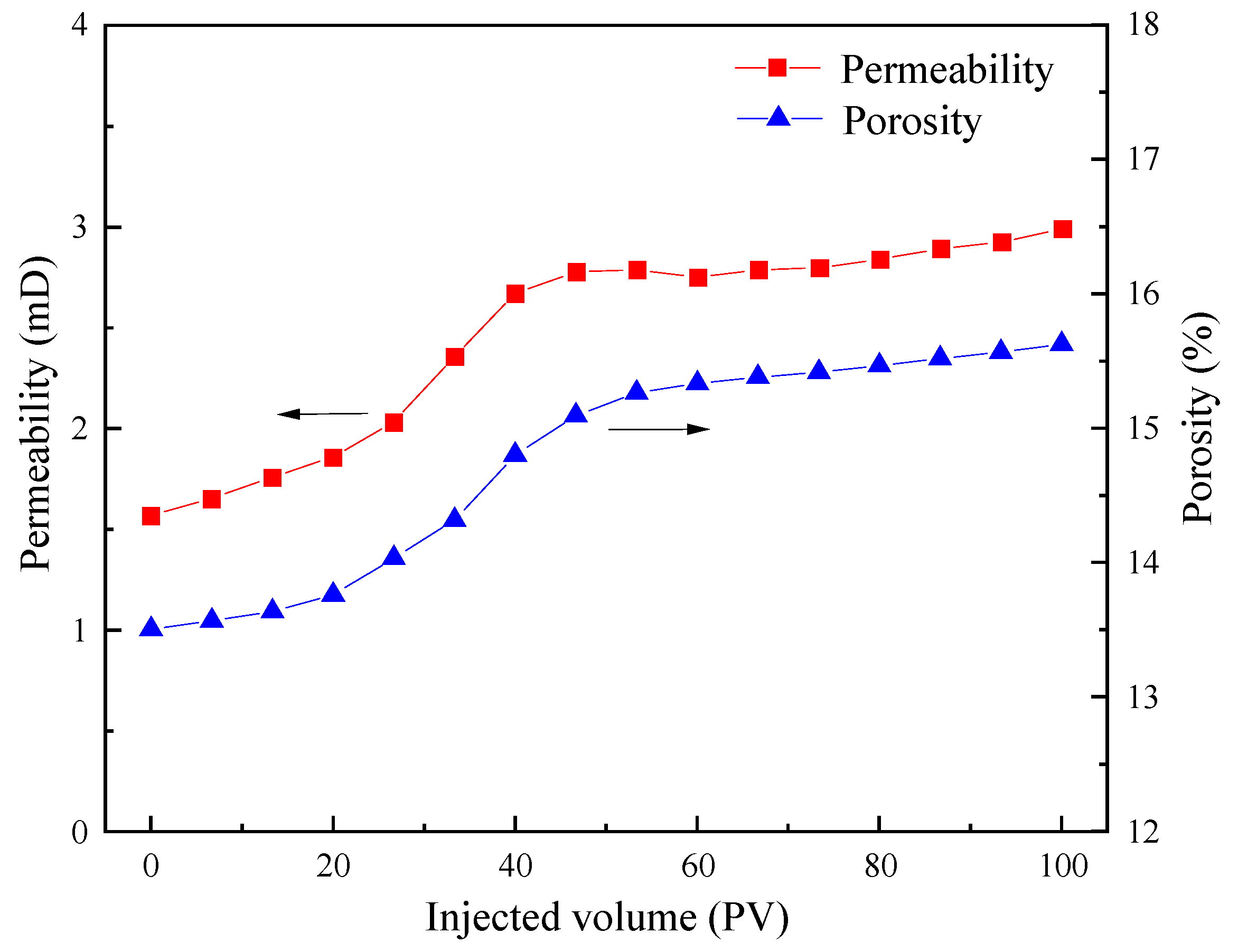
3.3.2. Dynamic Erosion Experiments
3.4. Storage following the Process of CO2 Flooding
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Lokhorst, A.; Wildenborg, T. Introduction on CO2 Geological storage-classification of storage options. Oil Gas Sci. Technol. 2005, 60, 513–515. [Google Scholar] [CrossRef]
- Streit, J.E.; Hillis, R.R. Estimating fault stability and sustainable fluid pressures for underground storage of CO2 in porous rock. Energy 2004, 29, 1445–1456. [Google Scholar] [CrossRef]
- Zendehboudi, S.; Khan, A.; Carlisle, S.; Leonenko, Y. Ex situ dissolution of CO2: A new engineering methodology based on mass-transfer perspective for enhancement of CO2 sequestration. Energy Fuels 2011, 25, 3323–3333. [Google Scholar] [CrossRef]
- Tayeb, F.; Rouabhi, A.; AbuAisha, M.; Valtz, A. Kinetics of CO2 dissolution for underground applications. Geoenergy Sci. Eng. 2023, 230, 212061. [Google Scholar] [CrossRef]
- Damen, K.; Faaij, A.; Turkenburg, W. Health, safety and environmental risks of underground CO2 storage-overview of mechanisms and current knowledge. Climatic Change 2006, 74, 289–318. [Google Scholar] [CrossRef]
- Zhang, Z.; Wang, T.; Blunt, M.J.; Anthony, E.J.; Park, A.-H.A.; Hughes, R.W.; Webley, P.A.; Yan, J. Advances in Carbon Capture, Utilization and Storage; Elsevier: Amsterdam, The Netherlands, 2020; Volume 278, p. 115627. [Google Scholar]
- Nocito, F.; Dibenedetto, A. Atmospheric CO2 mitigation technologies: Carbon capture utilization and storage. Curr. Opin. Green Sustain. Chem. 2020, 21, 34–43. [Google Scholar] [CrossRef]
- Orr, F.M., Jr. Carbon capture, utilization, and storage: An update. Spe J. 2018, 23, 2444–2455. [Google Scholar] [CrossRef]
- Gilfillan, S.M.; Lollar, B.S.; Holland, G.; Blagburn, D.; Stevens, S.; Schoell, M.; Cassidy, M.; Ding, Z.; Zhou, Z.; Lacrampe-Couloume, G. Solubility trapping in formation water as dominant CO2 sink in natural gas fields. Nature 2009, 458, 614–618. [Google Scholar] [CrossRef]
- Ahmadi, P.; Chapoy, A. CO2 solubility in formation water under sequestration conditions. Fluid Phase Equilibria 2018, 463, 80–90. [Google Scholar] [CrossRef]
- Zhang, W.; Wu, S.; Ren, S.; Zhang, L.; Li, J. The modeling and experimental studies on the diffusion coefficient of CO2 in saline water. J. CO2 Util. 2015, 11, 49–53. [Google Scholar] [CrossRef]
- Li, S.; Li, Z.; Dong, Q. Diffusion coefficients of supercritical CO2 in oil-saturated cores under low permeability reservoir conditions. J. CO2 Util. 2016, 14, 47–60. [Google Scholar] [CrossRef]
- Askari, G.; Rostami, B.; Ghasemi, M. Impact of water saturation on diffusion coefficients determined by constant volume diffusion method. J. Pet. Sci. Eng. 2022, 212, 110290. [Google Scholar] [CrossRef]
- Miller, J.S.; Jones, R.A. A Laboratory Study to Determine Physical Characteristics of Heavy Oil after CO2 Saturation. In SPE/DOE Enhanced Oil Recovery Symposium; OnePetro: Richardson, TX, USA, 1981. [Google Scholar]
- Riazi, M.R. A new method for experimental measurement of diffusion coefficients in reservoir fluids. J. Pet. Sci. Eng. 1996, 14, 235–250. [Google Scholar] [CrossRef]
- Geerlings, H.; Zevenhoven, R. CO2 mineralization-bridge between storage and utilization of CO2. Annu. Rev. Chem. Biomol. Eng. 2013, 4, 103–117. [Google Scholar] [CrossRef]
- Kelemen, P.; Benson, S.M.; Pilorgé, H.; Psarras, P.; Wilcox, J. An overview of the status and challenges of CO2 storage in minerals and geological formations. Front. Clim. 2019, 1, 9. [Google Scholar] [CrossRef]
- Assen, A.H.; Belmabkhout, Y.; Adil, K.; Lachehab, A.; Hassoune, H.; Aggarwal, H. Advances on CO2 storage. Synthetic porous solids, mineralization and alternative solutions. Chem. Eng. J. 2021, 419, 129569. [Google Scholar] [CrossRef]
- Ryzhenko, B. Genesis of dawsonite mineralization: Thermodynamic analysis and alternatives. Geochem. Int. 2006, 44, 835–840. [Google Scholar] [CrossRef]
- Izgec, O.; Demiral, B.; Bertin, H.; Akin, S. CO2 injection into saline carbonate aquifer formations I. Transp. Porous Media 2008, 72, 1–24. [Google Scholar] [CrossRef]
- Tiong, M.; Gholami, R.; Abid, K.; Rahman, M.E. Nanomodification: An efficient method to improve cement integrity in CO2 storage sites. J. Nat. Gas Sci. Eng. 2020, 84, 103612. [Google Scholar] [CrossRef]
- Zhao, X.; Liao, X.; He, L. The evaluation methods for CO2 storage in coal beds, in China. J. Energy Inst. 2016, 89, 389–399. [Google Scholar] [CrossRef]
- Ajoma, E.; Sungkachart, T.; Saira; Yin, H.; Le-Hussain, F. A laboratory study of coinjection of water and CO2 to improve oil recovery and CO2 storage: Effect of fraction of CO2 injected. SPE J. 2021, 26, 2139–2147. [Google Scholar] [CrossRef]
- Li, Z.; Su, Y.; Shen, F.; Huang, L.; Ren, S.; Hao, Y.; Wang, W.; Meng, Y.; Fan, Y. Investigation of CO2 storage and EOR of alternating N2 and CO2 injection using experiments and numerical simulation. Fuel 2023, 340, 127517. [Google Scholar] [CrossRef]
- Yao, Y.; Li, X. A study on CO2 flooding effectiveness and its geologic storage. Xinjiang Pet. Geol. 2009, 30, 493. [Google Scholar]
- Yang, Z.; Wu, W.; Dong, Z.; Lin, M.; Zhang, S.; Zhang, J. Reducing the minimum miscibility pressure of CO2 and crude oil using alcohols. Colloids Surf. A: Physicochem. Eng. Asp. 2019, 568, 105–112. [Google Scholar] [CrossRef]
- Kavousi, A.; Torabi, F.; Chan, C.W.; Shirif, E. Experimental measurement and parametric study of CO2 solubility and molecular diffusivity in heavy crude oil systems. Fluid Phase Equilibria 2014, 371, 57–66. [Google Scholar] [CrossRef]
- Li, W.; Nan, Y.; You, Q.; Jin, Z. CO2 solubility in brine in silica nanopores in relation to geological CO2 sequestration in tight formations: Effect of salinity and pH. Chem. Eng. J. 2021, 411, 127626. [Google Scholar] [CrossRef]
- Rezk, M.G.; Foroozesh, J.; Abdulrahman, A.; Gholinezhad, J. CO2 diffusion and dispersion in porous media: Review of advances in experimental measurements and mathematical models. Energy Fuels 2021, 36, 133–155. [Google Scholar] [CrossRef]
- Farquhar, S.; Pearce, J.; Dawson, G.; Golab, A.; Sommacal, S.; Kirste, D.; Biddle, D.; Golding, S. A fresh approach to investigating CO2 storage: Experimental CO2-water-rock interactions in a low-salinity reservoir system. Chem. Geol. 2015, 399, 98–122. [Google Scholar] [CrossRef]
- Li, Z.; Su, Y.; Li, L.; Hao, Y.; Wang, W.; Meng, Y.; Zhao, A. Evaluation of CO2 storage of water alternating gas flooding using experimental and numerical simulation methods. Fuel 2022, 311, 122489. [Google Scholar] [CrossRef]









| Sample Number | Ionic Species | Na+ + K+ | Ca2+ | Mg2+ | Cl− | SO42− | CO32− | HCO3− | Total Mineralization |
|---|---|---|---|---|---|---|---|---|---|
| 1 | Concentration (mg/L) | 2281 | 420 | 215 | 3250 | 491 | 0 | 0 | 13,500 |
| 2 | 8922 | 1105 | 2427 | 16,280 | 227 | 0 | 288 | 29,249 | |
| 3 | 2738 | 7206 | 5052 | 27,740 | 0 | 0 | 6110 | 62,428 |
| Density/ (g·cm−3) | Ground Viscosity/ (mPa·s) | Saturates/wt% | Aromatics /wt% | Resin /wt% | Asphaltene /wt% |
|---|---|---|---|---|---|
| 0.912 | 126 | 46.1 | 25.8 | 27.06 | 1.04 |
| Core Number | Length/cm | Diameter/cm | Porosity/% | Irreducible Water Saturation/% | Fluid Measuring Permeability/mD |
|---|---|---|---|---|---|
| Sample 1 | 6.635 | 2.501 | 13.34 | 27.84 | 1.65 |
| Sample 2 | 6.816 | 2.505 | 13.46 | 27.56 | 1.77 |
| Sample 3 | 6.130 | 2.496 | 14.41 | 27.44 | 1.68 |
| Core Number | Length/cm | Diameter/cm | Porosity/% | Fluid Measuring Permeability/mD |
|---|---|---|---|---|
| Sample 1 | 6.731 | 2.504 | 13.57 | 1.55 |
| Sample 2 | 7.815 | 2.506 | 13.89 | 1.67 |
| Sample 3 | 8.134 | 2.496 | 14.16 | 1.71 |
| Diameter/cm | Length/cm | Porosity/% | Fluid Measuring Permeability/mD |
|---|---|---|---|
| 2.58 | 30.14 | 13.502 | 1.57 |
| Parameter | Level | |||||
|---|---|---|---|---|---|---|
| Temperature/°C | 30.0 | 60.0 | ||||
| Initial pressure/MPa | 5.0 | 12.0 | 15.0 | 5.0 | 12.0 | 15.0 |
| Coefficient of diffusion/m2·s−1 (×10−8) | 3.15 | 3.51 | 3.65 | 4.01 | 4.62 | 4.96 |
| Parameter | Level | |||||
|---|---|---|---|---|---|---|
| Temperature/°C | 30.0 | 60.0 | ||||
| Initial pressure/MPa | 5.0 | 12.0 | 15.0 | 5.0 | 12.0 | 15.0 |
| Coefficient of diffusion/m2·s−1 (×10−8) | 4.97 | 5.32 | 5.52 | 5.97 | 6.35 | 6.62 |
| Parameter | Level | ||
|---|---|---|---|
| Temperature/°C | 60.0 | ||
| Initial pressure/MPa | 5.0 | 12.0 | 15.0 |
| Diffusion coefficient/m2·s−1 (×10−10) | 3.20 | 3.67 | 4.16 |
| Experiment Number | Volume of Injected Water, mL | Conversion Factor | Average Value of Conversion Factors |
|---|---|---|---|
| 1 | 496.55 | 144.8 | 142.1 |
| 2 | 496.42 | 139.6 | |
| 3 | 496.48 | 142.0 |
| Mineral Type | Capacity of Solidified CO2 per Unit Mass of Mineral (g/g) | Mineral Consumption (g) | Amount of Solidified CO2 (g) |
|---|---|---|---|
| NaAlSi3O8 | 0.167 | 0.24 | 0.040 |
| Ca(AlSi3O8)2 | 0.170 | 0.62 | 0.105 |
| KAlSi3O8 | 0.721 | 0.55 | 0.397 |
| Al2Si2O5(OH)4 | 0.341 | 0.12 | 0.041 |
| Form of Storage | Free and Irreducible Storage | Mineralized Storage | Dissolved Storage |
|---|---|---|---|
| Storage volume, mL | 640.871 | 294.912 | 121.559 |
| Storage proportion, % | 60.6 | 27.9 | 11.5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cao, X.; Feng, Q.; Ji, Y. Effect of Interaction between Carbon Dioxide and Fluid Phase/Rock Interface on Carbon Dioxide Storage. Processes 2023, 11, 3331. https://doi.org/10.3390/pr11123331
Cao X, Feng Q, Ji Y. Effect of Interaction between Carbon Dioxide and Fluid Phase/Rock Interface on Carbon Dioxide Storage. Processes. 2023; 11(12):3331. https://doi.org/10.3390/pr11123331
Chicago/Turabian StyleCao, Xiaopeng, Qihong Feng, and Yanfeng Ji. 2023. "Effect of Interaction between Carbon Dioxide and Fluid Phase/Rock Interface on Carbon Dioxide Storage" Processes 11, no. 12: 3331. https://doi.org/10.3390/pr11123331
APA StyleCao, X., Feng, Q., & Ji, Y. (2023). Effect of Interaction between Carbon Dioxide and Fluid Phase/Rock Interface on Carbon Dioxide Storage. Processes, 11(12), 3331. https://doi.org/10.3390/pr11123331





