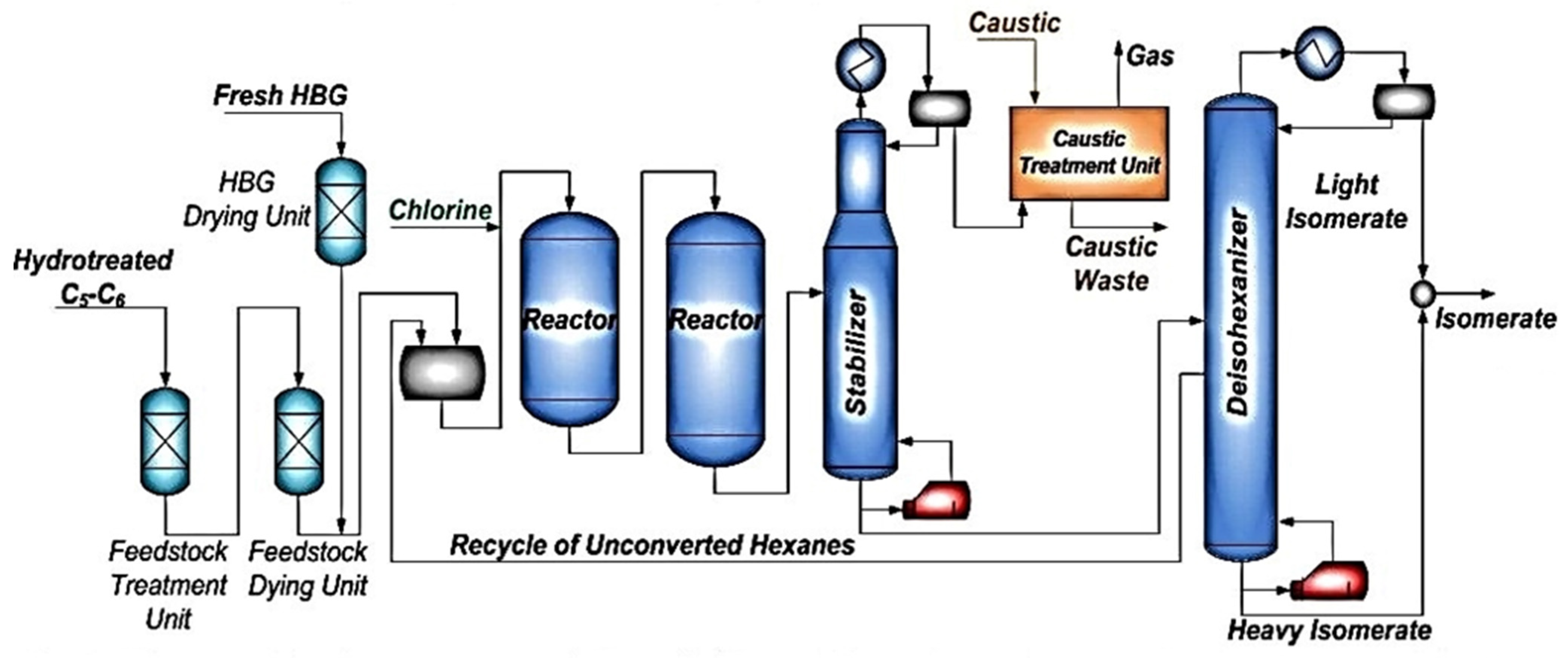
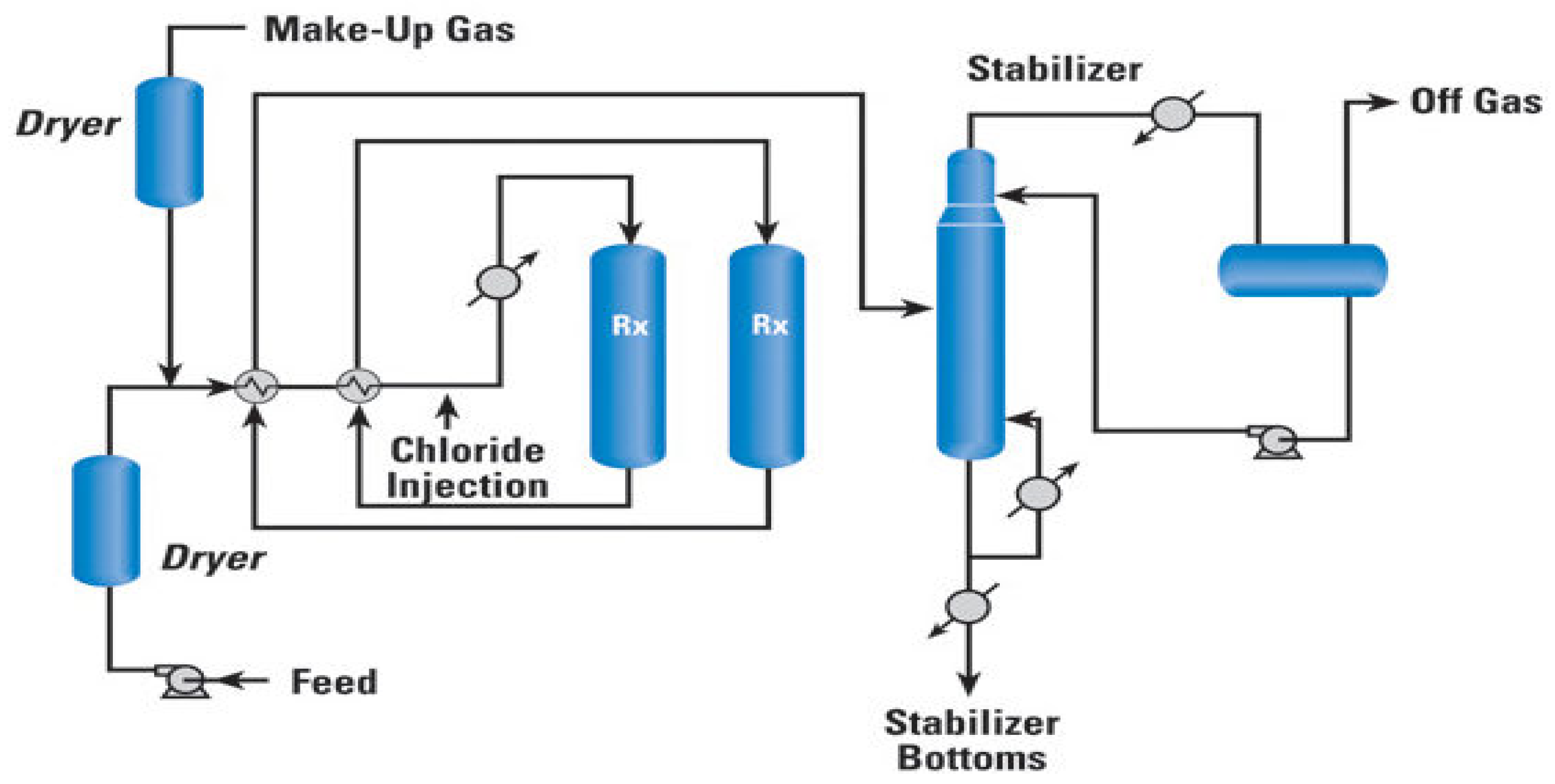
1. Introduction
Isomerization is a process that has become one of the most promising techniques for upgrading gasoline quality; this is due to the process’s ability to produce high-octane gasoline by converting straight-chain paraffins to the branched forms of iso-paraffins, while simultaneously reducing the number of pollutants released into the environment. There are different types of isomerization; one of the most common types is geometric isomerization. Geometric isomerization results in the same molecular formula, but the atoms are arranged differently in space due to the presence of double bonds. Another type of isomerization is positional isomerization; positional isomers have the same molecular formula but differ in the location of the functional group in the carbon chain [
1].
Catalytic reforming and isomerization are the two main processes used to improve octane number by hydrocarbon molecule rearrangement [
2,
3]. However, catalytic reforming also involves breaking down large hydrocarbons into smaller more valuable molecules [
4]. The two processes differ in feedstock, operating conditions, and quality of the product; catalytic reforming has a limited ability to process naphtha with a high content of normal paraffin. The reformate, produced from the reforming process, has a much higher content of benzene than permissible by the current environmental regulations in many countries.
Isomerization is considered as a more refined and economical way of increasing the octane rating. The quality of isomerate depends on several factors such as temperature, naphthene content, and liquid hourly space velocity (LHSV); increasing temperature positively affects the reaction rates. In addition, as the naphthene content of the feed increases, the amount of hydrogen required to open the naphthene ring increases. However, a severe reduction in LHSV will result in channeling [
5].
Many studies have been done to improve the performance of the isomerization process [
6,
7]. Naqvi et al. reviewed the isomerization catalyst used, main reactions, reaction mechanisms, and classification of the isomerization processes [
8]. Nikitav Checantsev and Gyngazova introduced a mathematical model for light naphtha isomerization units with different compositions of raw materials, which gives isomerate composition agreement with experimental data obtained from the industrial isomerization units of Russian refineries. Their proposed isomerization mathematical model enables the user to compare the efficiency of different isomerization units and select the more suitable variant of process optimization for a given raw material. The calculations are carried out on an isomerization process scheme with recycling n-pentane [
9].
Hamadi and Kadhim introduced a material balance and kinetic model for penex isomerization, in which material balance calculations have been performed for the prediction of kinetics, which is the rate constant for conversion of n-paraffin to olefin “K
1”, rate constant for conversion of olefin to i-paraffins “K
2”, and activation energy. Their study showed that increasing temperature results in an increase in K
1 and a decrease in K
2 [
10]. The optimization of process variables was introduced by Shahata et al. in 2018; in their work, the variables affecting isomerization product octane numbers such as feed composition, temperature, hydrogen-to-feed ratio, and LHSV have been analyzed and optimized using response surface methodology (RSM) [
11]. Chuzlov et al. developed a mathematical model for a light naphtha catalytic isomerization unit, where the plant operation with catalytic isomerization and separation columns has been optimized. They aimed to select the optimal modes of separation columns to achieve the desired separation between the units [
12]. Jarullah et al. introduced a new naphtha isomerization process, called AJAM, where the isomerization reactor model was validated using data from Baiji North Refinery (BNR). In their study, it is found that adding a de-isopentanizer “DIP” has a positive effect on the research octane number (RON), isomerate properties, and operation cost. Their study concluded that the proposed AJAM isomerization process gives the maximum RON, isomerate yield, and minimum cost compared to molecular sieve technologies [
13]. Nagabhatla Viswanadham et al. converted naphtha feedstock into high-octane gasoline blending stock, which is rich in iso-paraffin and suitable for fuel applications. They used three catalyst systems, which exhibit different acidities. Nano crystalline ZSM-5, containing inter-crystalline voids, is also studied in their research to investigate the effect of micro porosity on the product selectivity; then, the quality of isomerate in terms of total iso-paraffins is analyzed. Studies were also conducted on two single-component feeds, n-heptane, and n-octane, to understand the effect of hydrocarbon chain length on the reactivity and product selectivity in the process [
14]. Yu. N. Lebedevincrease et al. showed that fitting isomerization units with a de-isohexanizer “DIH” tower increases the RON of isomerate by a minimum of 4–5 points; the product from the stabilizer is fed to this tower to separate low-octane n-hexane and methyl pentanes, which are taken back with the side stream into the reactor for repeated conversion [
15].
The availability of low-value naphtha and other such feedstocks calls for the development of efficient methods for adding value to the feedstocks through octane number enhancement. The feedstock has a low octane number most times (<60), which is not reliable for fuel applications, in which high-octane gasoline is required. Therefore, the isomerization of n-paraffins attracted much attention for the refinery processes. In addition, Euro-4 and Euro-5 standards did not only restrict the content of benzene to less than 1 vol.%, but also put restrictions on the total aromatics content; it must be less than 35 vol.%. To meet these environmental restrictions, reformate is usually diluted with isomerate in the ratio of 1:1. So, isomerization became the largest tonnage process after reforming. Saad Zafer showed that UOP offers several schemes, in which low-octane components are separated and recycled back to the reactors. These recycling modes of operation can lead to the production of a product with a higher octane number. He also confirmed that the addition of a de-isopentanizer (DIP) and a super de-isohexanizer (DIH) would achieve the highest octane from a fractionation hydrocarbon recycle flow scheme. Moreover, the scheme with de-isopentanizer (DIP) before the reactor section allows the production of isomerate with high octane number, increases the conversion level of n-pentanes, and at the same time reduces the reactor duty [
16].
In this research, a new modification is developed by adding two fractionators before and after the reactor, where more economic savings to the refinery could be achieved. The improvement in process economics is not only related to obtaining high-quality product, but also comes from operating cost savings due to optimizing process conditions. This improvement was illustrated by applying the proposed modifications to the investigated case study.
The study shows how octane number is affected by separating i-pentane from feed before entering the reactor and recycling n-hexane to the reactor. The optimum conditions to produce the highest octane with minimum cost are also found in this study, using Aspen HYSYS V.12.1. Soave Redlich Kwong (SRK) fluid package is used to provide an estimation for the isomerate composition and the properties of all process streams. The Peng–Robinson Equation of State (EOS) is generally the recommended property package, as it predicts properties of mixtures ranging from well-defined light hydrocarbon systems to complex oil mixtures and provides optimized state equations for the rigorous handling of hydrocarbon systems. However, it is approved that the SRK fluid package is more suitable for isomerization reaction calculations [
17].
3. Conclusions
The aim of the current research work is directed to studying, improving, and optimizing an isomerization plant located in Alexandria, Egypt. Three types of changes in the main structure of the isomerization process are introduced. The changes consider the addition of one or two fractionation columns before or after the reactor. The purpose of these fractionators is separating the unreacted normal (unbranched) paraffins to be recycled to the reactor for increasing the octane number of the isomerate product. The simulation and optimization tool used in this paper is Aspen HYSYS version 12.1.
The simulation results showed that the octane number and ROI of the isomerization process using two fractionators (a de-hexanizer and a de-isopentanizer) before and after the reactor increased to 86.5 and 29.9%, respectively. For the process of adding a de-isopentanizer before the reactor, the octane number and ROI reached 78.15 and 20%, respectively. Regarding the isomerization process with adding a de-hexanizer after the reactor, the octane number of 81 and ROI of 17% are obtained. Therefore, adding two fractionators to the original isomerization plant is more profitable with the highest product octane number compared with the other investigated configurations.
This work also studies the optimization of this selected best process to be more profitable. The optimization of this process seeks to find the optimum conditions for the reactor feed. The optimization results showed that the optimum temperature and pressure of the reactor feed are 155 °C and 45.4 bar_g, respectively. It is noticed that the obtained optimum conditions of the reactor feed stream led to the maximum product’s octane number and minimum heat flow of the de-hexanizer and stabilizer, which resulted in a significant decrease in utility costs. The optimization results reveal an increase in octane numbers by 7% and a decrease in the total cost by 13%. These results prove the economic effectiveness of the proposed modification and optimization on the existent isomerization process, which can be applied to similar processes to increase their profitability.