1. Introduction
The application scope of vanadium has expanded beyond iron-/steelmaking, military, and medical industries [
1,
2,
3,
4] to include functional materials (e.g., nanocomposites), high-performance alloys, and all-vanadium liquid-flow batteries [
5,
6,
7,
8,
9,
10,
11,
12,
13]. In particular, the abovementioned batteries represent a new high-efficiency energy storage technology and energy-development direction, which highlights the strategic importance of vanadium resources [
14,
15,
16]. The steel industry is a major area of interest for vanadium products. Vanadium has greatly helped to improve the hardness, toughness, wear resistance, and high temperature-resistance of steel alloys products. With the continuous development and expansion of new fields, such as vanadium-based materials, vanadium nitrogen alloys, vanadium electrode products, high-purity vanadium-containing materials, and all vanadium liquid flow batteries, the market demand for high-quality vanadium-containing products will continue to increase. This rapid expansion places high requirements on the quality and quantity of vanadium compounds, of which V
2O
5 is the most stable and widely used [
17,
18]. Therefore, much attention has been drawn to the facile and efficient production of high-purity V
2O
5.
V
2O
5 is mainly produced through hydrometallurgical and chlorination methods, among which the most common method of separating and purifying metallurgical-grade vanadium products is through hydrometallurgical methods to achieve the purification and enrichment of vanadium [
19,
20]. However, all hydrometallurgical processes widely used for vanadium recovery, e.g., ion exchange, solvent extraction, adsorption, and precipitation [
21], exhibit certain drawbacks. For example, emulsification and flocculation during solvent extraction, rapid extractant loss, organic phase loss through entrainment, re-extraction difficulty, and impurity transfer compromise product quality and complicate organic-phase purification. The ion exchange method has a long cycle time and high salt consumption and generates excessive regeneration waste streams, and the presence of organic substances can contaminate ion exchange resins and discharge a large amount of salty wastewater [
22,
23,
24], which can easily cause the corrosion of pipelines. In addition, when there are multiple ions in the solution, different resins need to be selected for different ions, resulting in poor universality. The use of chemical precipitation requires the introduction of large amounts of chemical agents, resulting in the secondary pollution of precipitated waste residues.
Unlike their hydrometallurgical counterparts, chlorination-based processes offered the benefits of simplicity, low pollution, low cost, and high selectivity [
25,
26,
27,
28,
29,
30,
31,
32], and the preparation of V
2O
5 from vanadium-containing raw materials with chlorinating agents is therefore drawing much attention [
33]. Chlorination is typically performed using Cl
2, and the resulting VOCl
3 (purity ≥ 99.9%) reacts with NH
3·H
2O in an aqueous medium to afford precipitates that are subsequently converted into V
2O
5 [
34,
35,
36]. Although this method is time-efficient, it requires the handling of the highly corrosive and toxic Cl
2 and VOCl
3, thus necessitating the use of highly corrosion-resistant industrial equipment and strict safety protocols. In addition, the use of high roasting temperatures increases energy consumption and, hence, production costs. Zheng et al. [
37] used FeCl
X as a chlorination agent to extract vanadium from vanadium-bearing titanium magnetite. Under a roasting temperature of 900~1300 K and oxygen atmosphere, the extraction rate of vanadium increases with the increase in temperature and then decreases with the increase in temperature. Using FeCl
3 as a chlorinating agent and holding at 1100 K for 2 h, the extraction rate of vanadium can reach 32%. Du et al. [
38] extracted 96.36% V and 4.23% Ti from tailings containing 10% petroleum coke via chlorination roasting for 1 h at 800 °C with a chlorine pressure fraction of [P(Cl
2)/P(Cl
2 + N
2)] = 0.5. Further purification of the collected chlorinated products resulted in the production of VOCl
3 with a purity higher than 99.99%. Wu et al. [
27] proposed a method of recovering vanadium from carbonaceous gold ores based on the use of NaCl as a chlorinating agent to separate vanadium, gold, zinc, and iron. However, the temperature of the suggested chlorination reaction is as high as 800 °C. Further, we found that VOCl
3 can be prepared below 200 °C when AlCl
3 is used as a chlorinating agent. Jiang et al. [
1] used anhydrous aluminum chloride and sodium chloride to purify industrial-grade V
2O
5 with a purity of 96% at low temperatures of 170 °C, and the high-purity V
2O
5 with a purity of at least 99.97% was obtained. The novel method of V
2O
5 preparation is highly selective for vanadium. Therefore, using a chlorination method to prepare high-purity V
2O
5 has received great attention.
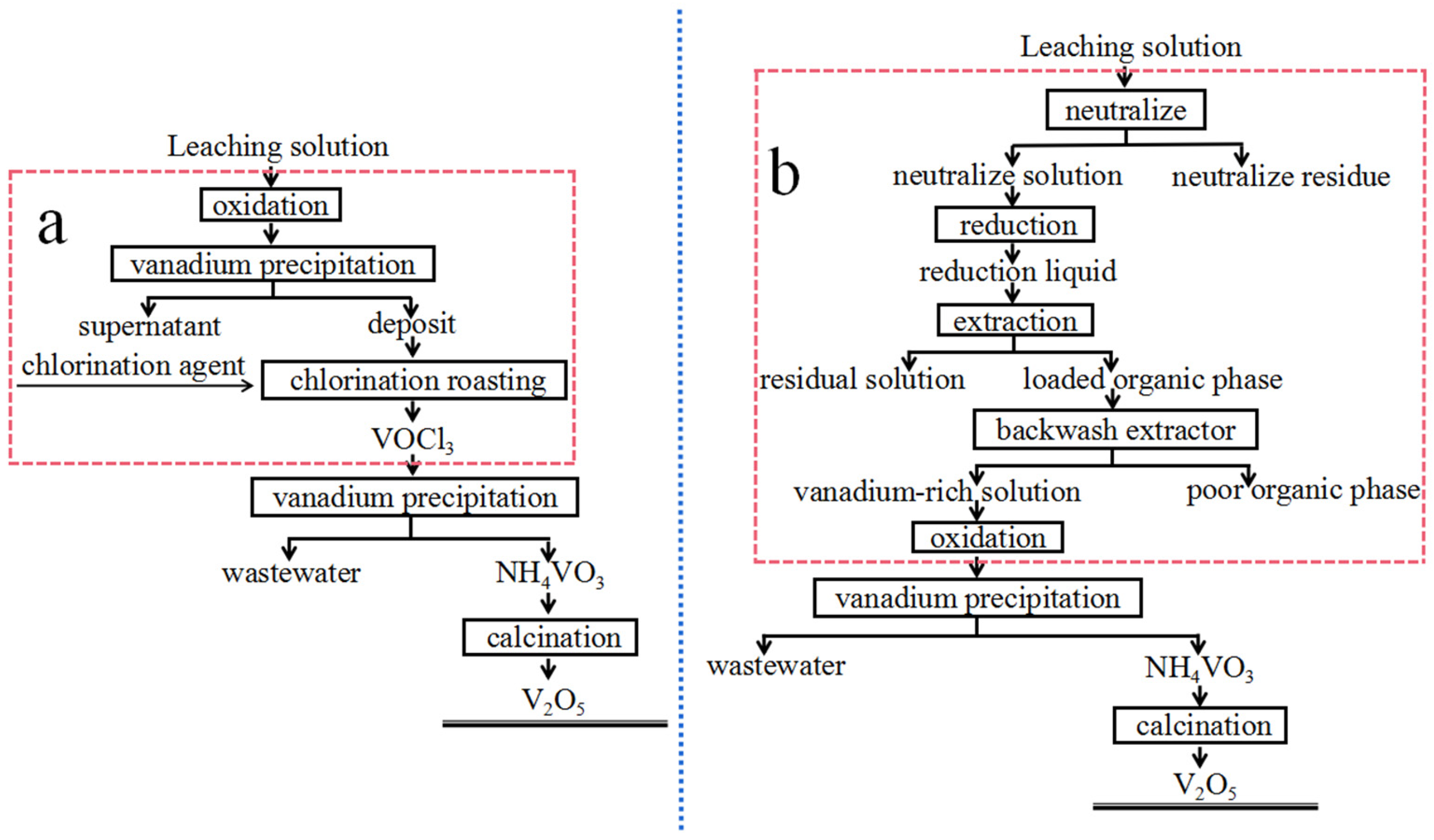
Herein, based on the abovementioned previous works, we developed a short-process method of preparing V2O5 from shale acid leaching solution. Based on the difference in boiling points of VOCl3 and FeCl3, MgCl2, NaCl, KCl, CrCl3, MoCl6 and NiCl2, through the use of chlorinated metallurgical methods, a vanadium precipitate was obtained by precipitating vanadium from acid leaching solution; the vanadium precipitate was roasted with AlCl3 at a low temperature, and then, a high-purity VOCl3 product was obtained, which was then hydrolyzed through treatment with NH3·H2O, and the precipitate was roasted to obtain high-purity V2O5. Unlike conventional chlorination techniques, our method avoided the ion exchange, chemical precipitation, solvent extraction, and other undesired processes, and the overall preparation process of V2O5 has been shortened. The short preparation process of V2O5 in this study also avoids the use of the toxic and corrosive Cl2 and does not require excessively high temperatures, thus holding great promise for cost-effective and facile vanadium recovery.
4. Conclusions
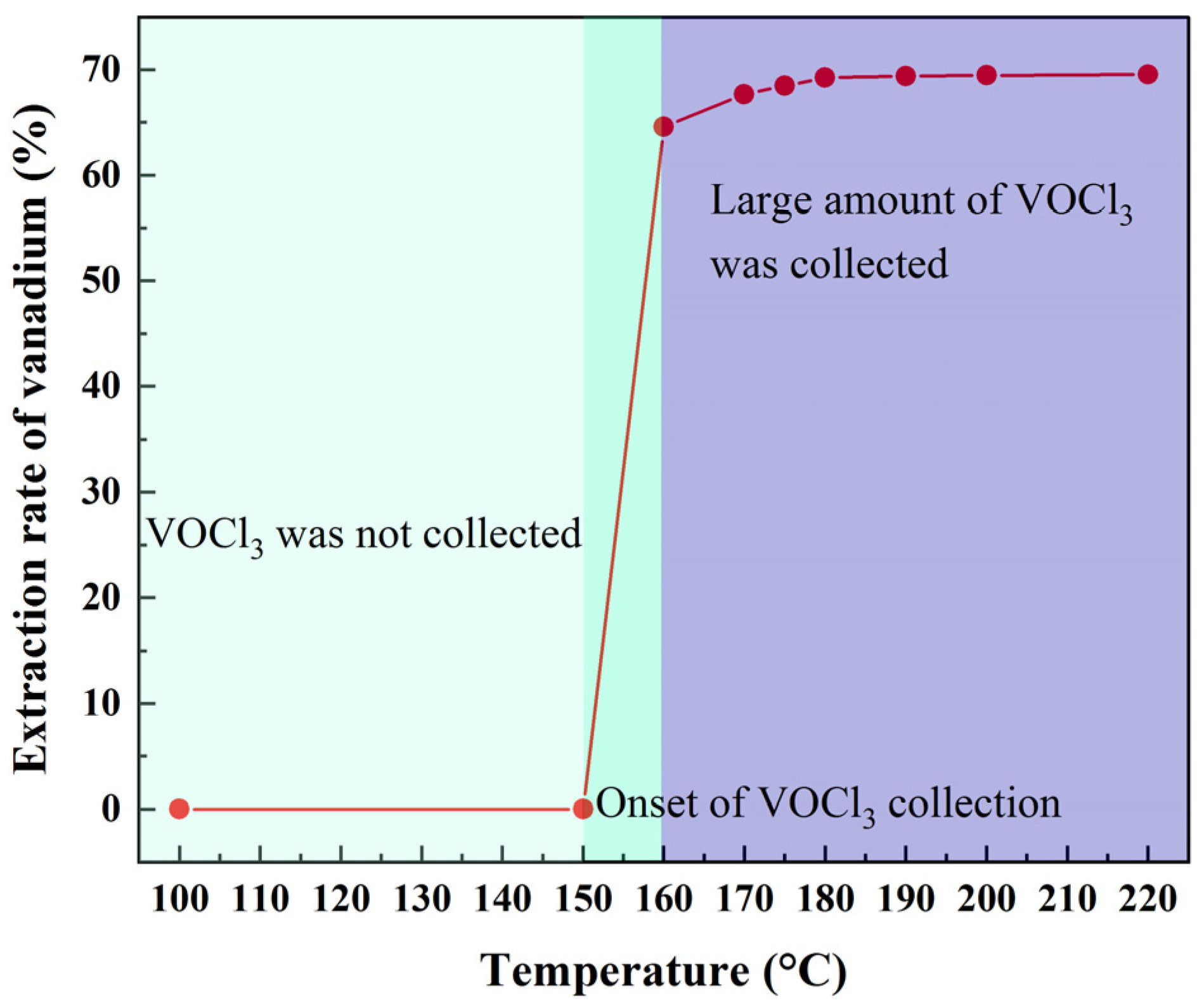
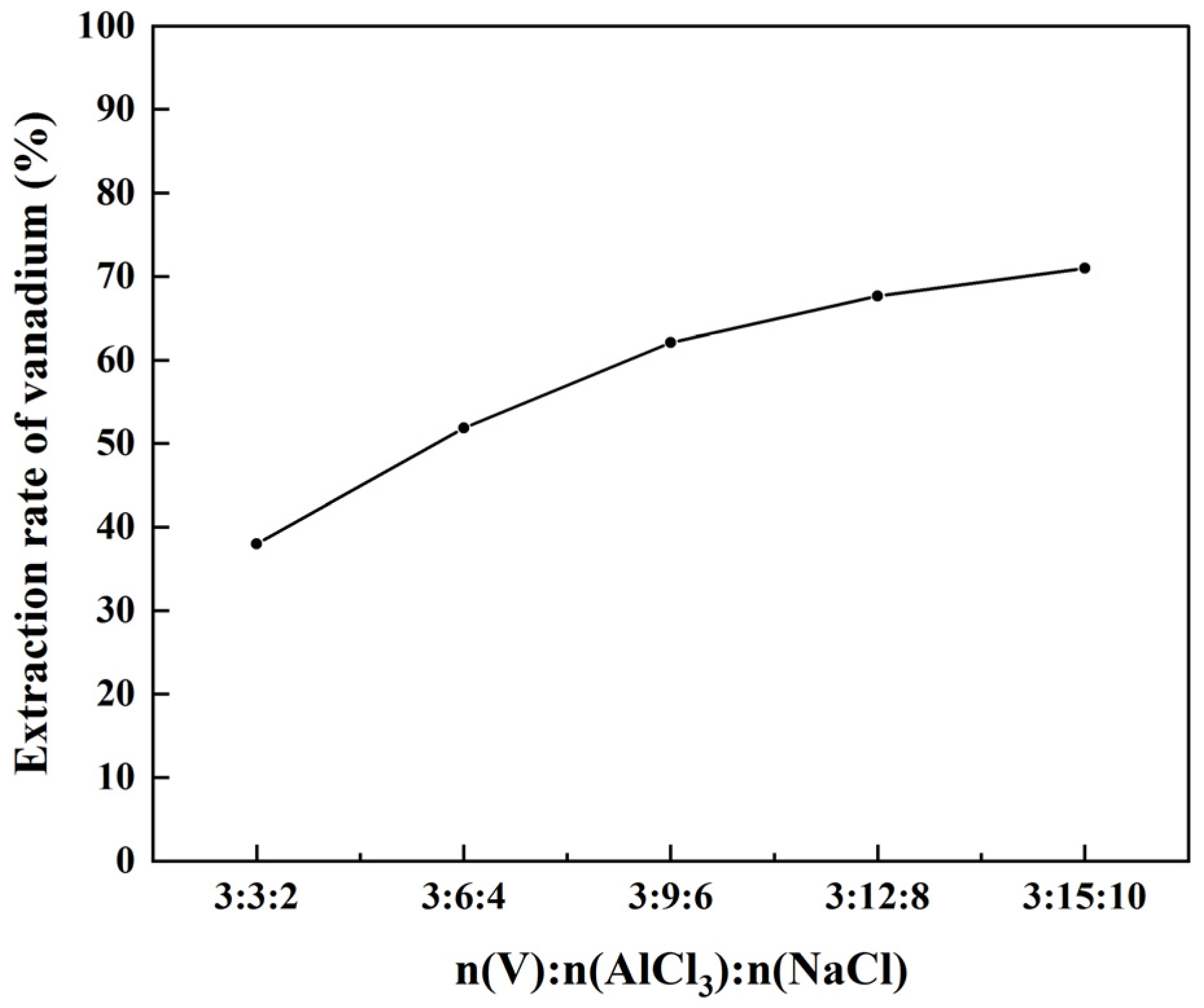
This study describes a short process of preparing V2O5 from shale acid leaching solution at a low temperature via precipitation followed by chlorination with AlCl3 at a low temperature (170 °C). The addition of NaCl can improve the vanadium extraction rate and purity of VOCl3. The obtained VOCl3 (purity ≥ 99.9%) is dissolved in ultrapure water, the solution is treated with aqueous ammonia, and the precipitate is roasted to afford V2O5. The optimal process parameters are as follows: chlorination temperature = 170 °C, chlorination time = 120 min, V:AlCl3:NaCl = 3:12:8 (mol/mol/mol) [chlorination], precipitation temperature = 50 °C, precipitation time = 120 min, NH3:V = 1.34 (mol/mol), and vanadium concentration = 32 g/L [precipitation]. The V2O5 obtained under the optimal conditions had a purity of 99.86%. Compared with the traditional hydrometallurgical process of V2O5 preparation, our method is characterized by simple operation and a short preparation process.
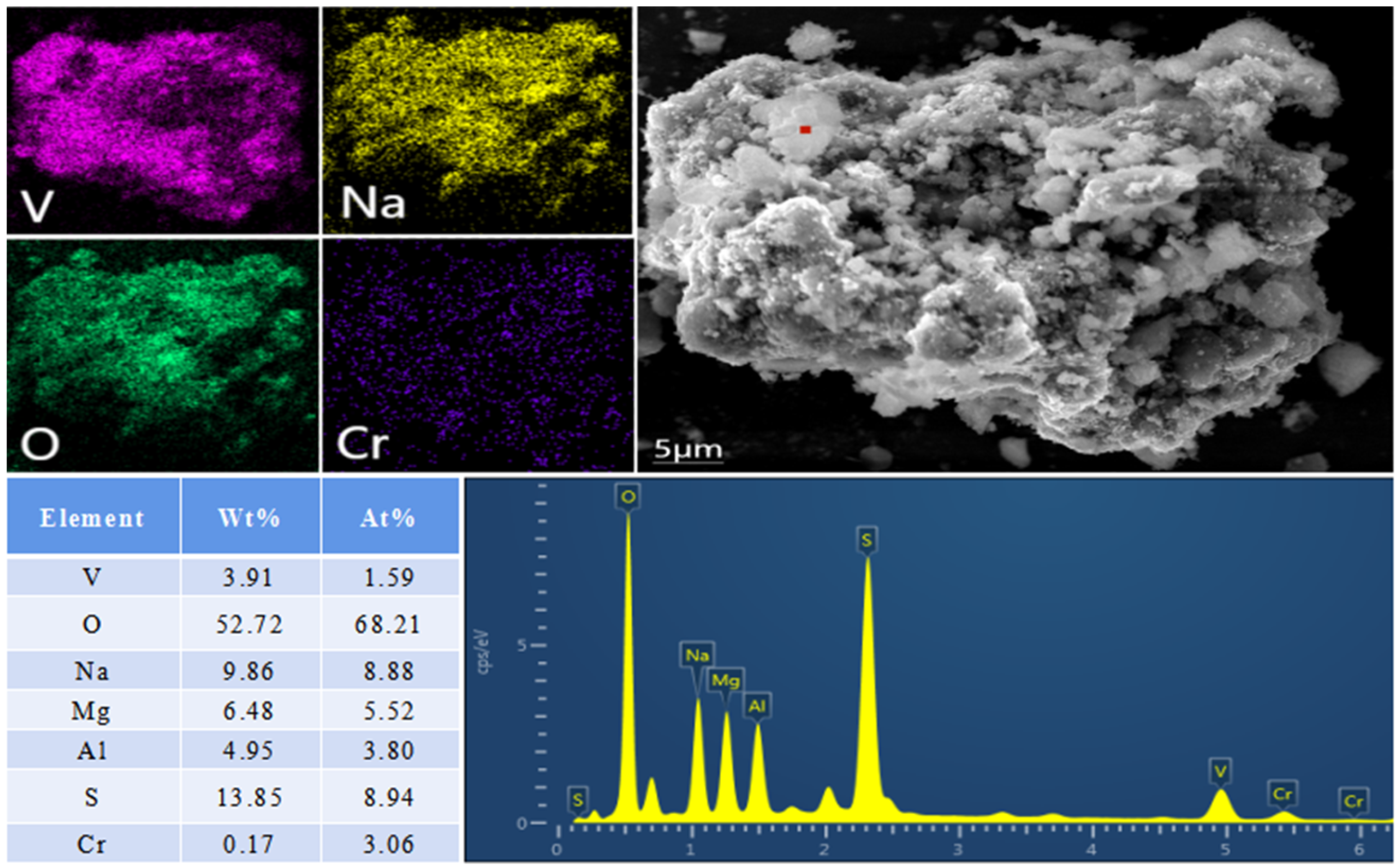
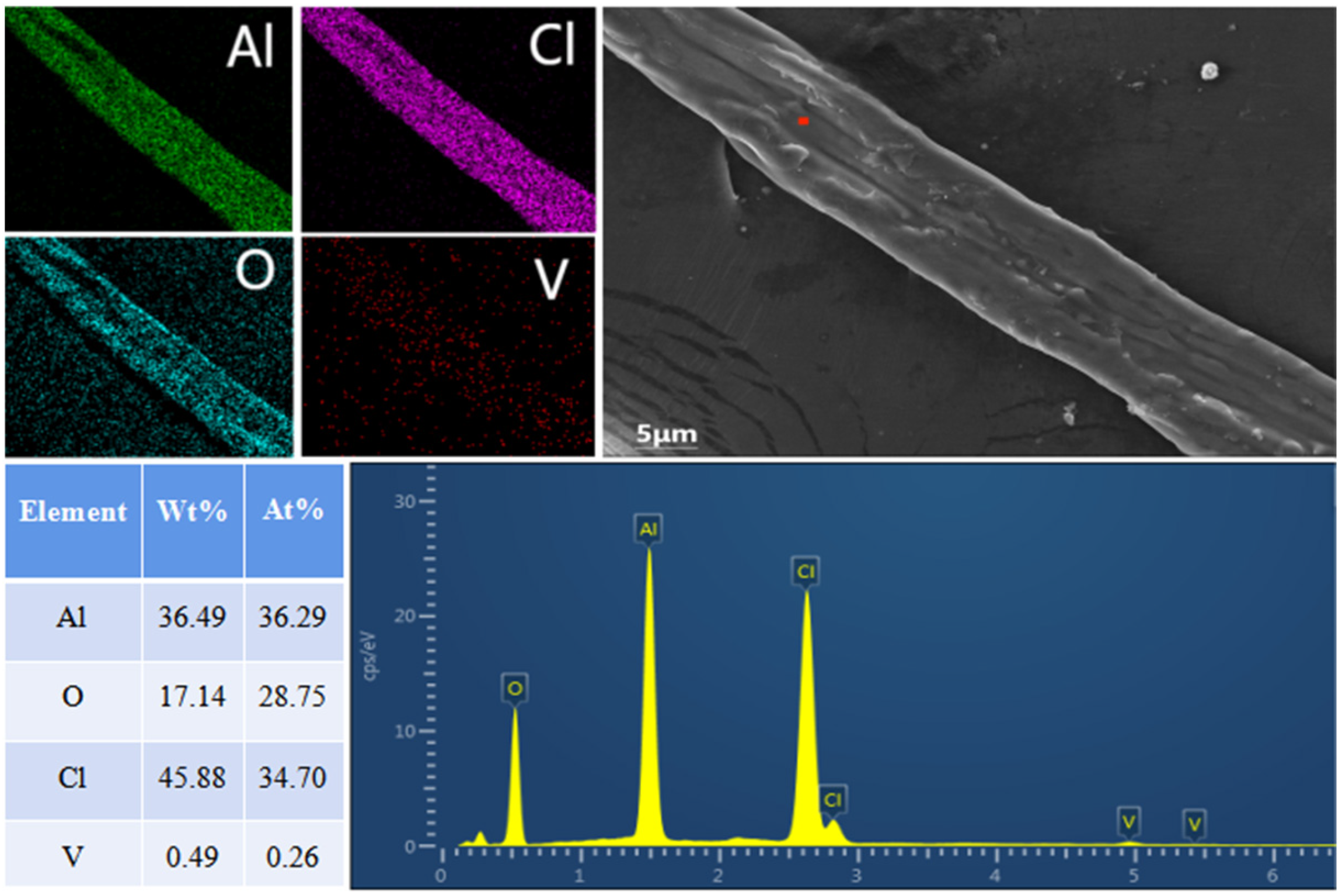
In addition, we found fibrous substances in the residue of the chlorination reaction. By means of XRD and SEM–EDS analysis of fibrous substances, this was determined to be AlOCl. Based on the formation of AlOCl during the chlorination reaction, the chlorination reaction mechanism was defined as the oxygen atoms in Na3VO4 and NaVO3 first replacing the two chlorine atoms in AlCl3 and the replaced chlorine atoms chlorinating Na3VO4 and NaVO3 to generate VOCl3. The chlorine atom in AlCl3 is gradually replaced.