1. Introduction
Lipophilicity affects the bioavailability, degree of biodegradation, and chemical activity of chemical compounds, including drugs. The action of the drug can be divided into three main stages, which include: pharmaceutical, pharmacokinetic, and pharmacodynamic phases. Lipophilicity plays a role in each of these steps [
1,
2,
3,
4]. Drug design is an important field of science that is experiencing a renaissance. Knowledge of intermolecular interactions, which are the basis for the development of diseases, has enabled the development of new drug structures and the planning of innovative treatments. Designing drugs is a long-term process and requires extensive knowledge in the field of chemistry, pharmacology, and related fields. The improvement of the therapeutic index of a drug is achieved by increasing the solubility and ability to penetrate biological membranes. When designing new drugs, these parameters are modified by differentiating the hydrophilic–lipophilic balance [
1,
4]. In practice, optimal lipophilicity values are sought, as a significant increase in fat solubility leads to excessive interaction of subsequent analogs with lipids, which results in their irreversible retention in the lipid layer of the cell membrane [
1,
2,
4]. For these reasons, the study of lipophilicity is of great practical importance. The measure of the lipophilicity of a chemical compound is the partition coefficient P, expressed as logP, and defined as the ratio of the equilibrium concentrations of the dissolved substance in the system of two phases:
n-octanol (C
o) and water (C
w) [
3,
5]:
The logP value allows us to understand the hydrophilic or hydrophobic (lipophilic) nature of a molecule. Indeed, if logP is positive, it means that the molecule under consideration is more soluble in n-octanol than in water, reflecting its lipophilic nature. Conversely, if the logP is negative, the molecule is hydrophilic. A logP of zero means that the molecule is as soluble in one solvent as it is in the other. Compounds with 0 < logP < 1 are characterized by poor lipophilicity. On the other hand, compounds for which −1 < logP < 0 are characterized by weak hydrophilicity.
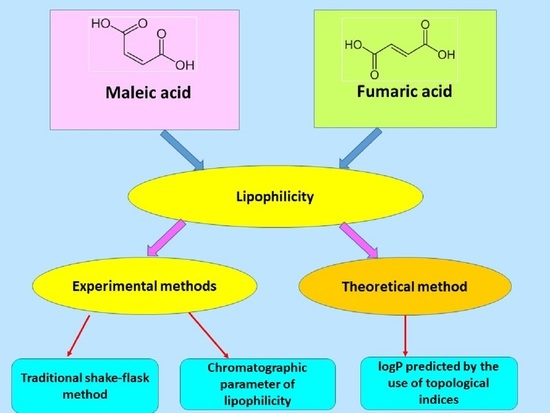
Lipophilicity can be determined experimentally: using the classic extraction method in the
n-octanol–water system (shake-flask), reversed-phase thin-layer chromatography (RP-TLC), and reversed-phase high-performance liquid chromatography (RP-HPLC) [
3,
6]. The measurements of the partition coefficients by equilibration methods are frequently difficult to take, or even impossible due to the impurity or instability of the compounds, a strong preference of compounds for one of the two phases of the system, or the formation of stable emulsions after shaking. However, due to these limitations, the procedure is very often replaced by chromatographic techniques.
Fumaric acid (FA) and maleic acid (MA) belong to the α-hydroxy acids. FA is
trans-butenedioic acid, whereas MA is
cis-isomer of fumaric acid (
Figure 1).
Fumaric acid (FA) appears in many plants, including mosses and mushrooms [
7]. In recent years it has been proven that FA is useful in cancer research, cardiology, neurology, and immunology. It has also been used as an element in the design of certain drug carriers and scaffolds that are based on biomaterials [
8,
9,
10,
11,
12]. It has a sour taste similar to grapes [
13]. It serves a role as a gustatory, oxyntic, and antioxidant substance. MA is mostly used in cosmetology and stomatology. It has an exfoliating function in scrubs. In recent years it has been implied that maleic acid could be used in stomatology as a smear layer, removing disinfectant for the rinsing of root canals. The use of a MA solution for irrigation changes the wettability of the root canal’s dentin, which has an impact on bacterial adhesion, as well as the interaction between the root canal’s dentin and the filling material [
14,
15,
16]. MA is also being used in stomatology, in combination with triclosan and PVM/MA copolymer [
17,
18,
19].
A different configuration (cis/trans) within the double bond causes differences in the physicochemical properties of FA and MA that have been defined experimentally [
7,
13,
20,
21,
22,
23,
24,
25].
Table 1 presents basic experimental [
7,
13,
20,
21,
22,
23,
24,
25] and theoretical [
26,
27] physicochemical properties of fumaric and maleic acids.
The different values of logP
exp in the
n-octanol–water system were obtained for MA and FA (
Table 1). The logP
exp values were equal from −1.30 to −0.48 [
20,
22,
24] and from 0.33 to 0.46 [
21,
23,
25] for MA and FA, respectively. Whereas, the theoretical logP, predicted by using Virtual Computational Chemistry Laboratory (xlogP3, AlogPs, AlogP, AClogP, mlogP, xlogP2) and Molinspiration Cheminformatics (milogP), have identical values for MA and FA. For example, the theoretical AlogPs is 0.21 for both fumaric acid and maleic acid. To our knowledge, the lipophilicity of maleic and fumaric acids has not yet been investigated by the TLC technique. Therefore, in this work, the lipophilicity of fumaric and maleic acids was determined by use of the classical shake-flask method in the
n-octanol–water system, the RP-TLC method, and new methods of calculation of the partition coefficient on the basis of the topological indices derived from the distance matrix.
2. Materials and Methods
2.1. Chemicals and Reference Standards
Fumaric acid (Sigma-Aldrich, St. Louis, MO, USA) and maleic acid (Fluka Chemie GmbH, Buchs, Switzerland) both had a degree of purity ≥ 99.0%. Methanol, dioxane, ammonia (25%), ethanol (POCh, Gliwice, Poland), and n-octanol (Sigma-Aldrich, St. Louis, MO, USA) were analytical grade. Distilled water at pH = 7.00 (Department of Analytical Chemistry, Sosnowiec, Poland) was also used. Chromatographic plates (E. Merck, Darmstadt, Germany): silica gel 60F254 (#1.05554), TLC RP8F254s (#1.15424), HPTLC RP18WF254 (#1.13124), and HPTLC CNF254s (#1.12571) were used in the thin-layer chromatography analysis.
2.2. Determining the Experimental Partition Coefficient (LogPexp) Using the Shake-Flask Method
2.2.1. Preparation of Samples
Water (pH = 7.00) saturated with n-octanol, and n-octanol saturated with water (pH = 7.00) were used in the study. A volume of 25 mL of n-octanol saturated with water, and 25 mL of water saturated with n-octanol were poured into the separatory funnel, and 100 mg of FA or MA was added. The extraction was carried out for 60 min at 20 °C. The extracted mixtures were left for 24 h to establish thermodynamic equilibrium. After this time, the aqueous phases were taken for quantitative analysis of FA and MA.
2.2.2. Normal-Phase Thin-Layer Chromatography (NP-TLC)
NP-TLC combined with densitometry on silica gel 60F
254 plates (#1.05554, E. Merck, Darmstadt, Germany) was used to determinate FA and MA. The water fraction, after extraction of FA and MA, was spotted on the chromatographic plate. A mixture of ethanol, ammonia 25%, and water in a volume composition of 6:1:1 was used as the mobile phase [
28]. Methyl red (100 mg in 100 mL of methanol) [
28] was used as the visualizing reagent of FA and MA. A Camag densitometer was used for the quantitative analysis of FA and MA. Scanning parameters were: speed 20 mm/s, resolution 100 µm/step, and slit size 12 × 0.4 mm. Spectrodensitometric analysis was carried out in the wavelength range of 200–800 nm (
Figure S1), and densitometry scanning was performed at λ
max = 215 nm. Each track was scanned five times. The content of FA and MA in the aqueous phase was calculated from the appropriate calibration curves. Standard solutions of FA and MA in water saturated with
n-octanol were used to construct the calibration equations. FA and MA were applied to the plates at 4, 8, 12, 16, 20, 24, 28, 32, 36, and 40 µg. Next, logP
exp for FA and MA was calculated using Equation (1). The analyzes were repeated six times.
2.2.3. Validation of the Normal-Phase Thin-Layer Chromatography Method for Determination of Experimental Partition Coefficient (LogPexp)
The NP-TLC method was validated according to the ICH guidelines [
29].
Linearity of Detector Response and Range
The linearity of the TLC method was evaluated by the analysis series of the standard solutions (in water saturated with
n-octanol) of FA and MA at the following concentrations 0.80, 1.60, 2.40, 3.20, 4.00, 4.80, 5.60, 6.40, 7.20, and 8.00 mg mL
−1. The solutions (5 μL) were applied to the same plate. The calibration plots were developed by plotting peak area versus concentration of FA and MA (
Table S1, Figures S2 and S3, respectively).
The experiments were performed in five different analyses.
Accuracy
The accuracy of the TLC method was determined by investigating the recovery percentage of the studied FA and MA at three concentration levels covering the low, medium, and high regions of the calibration plot by spotting on a chromatographic plate (n = 5 for each concentration level, where n represent the number of analyses). The resulting spots were analyzed by the use of the TLC procedure described above.
Precision
The precision was evaluated in terms of the repeatability (intraday) and interday precision. The repeatability of the proposed method was determined by the analysis of three replicates of the sample solutions at three concentration levels under the same operating conditions over a short interval of time (the same day). The intermediate (interday) precision was determined for three sample solutions at three concentration levels by an analyst who performed the analysis over a period of one week. To determine the precision of the procedure, the concentrations were prepared independently, and experiments were performed in three different analyses. The precision was evaluated as the coefficient of variation, CV [%]. The obtained peak areas were used to calculate the mean value and the CV [%].
Limit of Detection (LOD) and Limit of Quantification (LOQ) Based on the Specific Calibration Curve
Specific calibration curves were prepared using samples containing FA and MA in the low range of their concentrations. The limit of detection and limit of quantification were calculated from the equation of the graph of the area obtained by the spots of FA and MA versus the concentration. The LOD and LOQ were calculated using the equations:
where:
σ is the standard deviation (standard deviation of the intercept
σa and residual standard deviation
σxy of the calibration curve), and
S is the slope of the calibration curve.
Next, the mean values of LOD and LOQ were calculated for FA and MA.
The experiments were performed in six different analyses.
2.3. Use of RP-TLC for the Evaluation of Chromatographic Parameters of the Lipophilicity
The methanolic solutions of FA and MA were spotted on RP8F
254s, RP18WF
254, and CNF
254s chromatographic plates. The plates were developed by using two mobile phases, namely, using a mixture of methanol and water, as well as a mixture of dioxane and water, in different volume compositions. After drying the chromatographic plates, FA and MA were detected with methyl red (100 mg in 100 mL methanol) as a visualizing reagent. The analyses were repeated three times. The obtained R
F values were converted to R
M values. Next, from the R
M values (
Tables S2–S5) and the content of the organic component in the mobile phase, the R
MW chromatographic parameter of lipophilicity was calculated using the Soczewinski–Wachtmeister equation [
3]:
where:
RM is the value of the examined substance by content φ of the volume fraction of the methanol or dioxane in the mobile phase;
RMW is the theoretical value of the
RM of the analyte extrapolated to zero concentration of the methanol or dioxane in the mobile phase; S is the slope of the regression curve; φ is the volume fraction of the methanol or dioxane in the mobile phase.
2.4. New Ways of Calculation of the logP for the FA and the MA
New ways of calculating the logP value were suggested for fumaric acid and maleic acid using Equations (5)–(8):
where:
0B,
3Bq, χ
012,
A, and
W are the topological indices; and the
RM values are of MA and FA analyzed on RP8F
254s, RP18WF
254, and CNF
254s plates with the mixtures of methanol and water, as well as dioxane and water in a volume composition of 2:8 (
v/
v).
The topological index χ
012 is based on the adjacency matrix [
30] and selected topological indices: Pyka (
0B,
1B,
A) and Wiener (
W) were computed using the elements of the distance matrix defined by Pyka [
31,
32] and Barysz et al. [
33].
2.5. Statistical Analysis
The computer software Statistica 13.0 (Statsoft, Kraków, Poland) was used for all calculations.
4. Conclusions
So far, the RP-TLC technique has not been used to study the lipophilicity of fumaric acid and maleic acid. Another novelty is the use of the NP-TLC technique for the quantitative determination of fumaric and maleic acids in the aqueous phase after their extraction in the n-octanol–water system. All the methods used in this work, i.e., the classic shake-flask method, the RP-TLC method in combination with the Soczewiński equation, and the new methods using topological indices, determining the lipophilicity of fumaric acid and maleic acid are useful and yield consistent results. The logPexp values obtained in this work in the n-octanol–water system and by the Soczewiński method, and by the new methods using topological indices, have higher values for FA than MA. Under most methodological conditions, maleic acid showed a higher affinity to the water phase, while fumaric acid showed a higher affinity to the lipid phase. The methods employed to determine lipophilicity are of particular importance in the aspect of studying cis- and trans-configuration compounds, because generally available computer programs based on various algorithms (Virtual Computational Chemistry Laboratory and Molinspiration Cheminformatics) indicate that fumaric acid and maleic acid have identical logP values. The new theoretical calculations of the logP values, based on topological indices, and also the chromatographic determination of the lipophilicity of the examined compounds, complement well-established methods and applications, such as the traditional shake-flask method. Because of the experimental difficulties, including the solubility limits, chemical instability, formation of emulsions, or impurities of the organic compounds, the evaluation of the logP values by the proposed RP-TLC method is well-founded in this paper. The methodology for the lipophilicity assessment for maleic and fumaric acids described in our paper can be used for the further study and estimation of the lipophilic properties of other organic compounds, indicating a different biological significance. The comparison of these results indicates that our new method of the calculation of the partition coefficients using topological indices, and also the chromatographic lipophilicities, are powerful tools in the determination of the logP values of maleic and fumaric acids.